Abstract
The aim of this study is to investigate the protective effects of thymoquinone (TQ) and ebselen (Eb) on arsenic (As)-induced renal toxicity in female rats. Sodium arsenite was orally administrated at a dose of 20 mg/kg body weight daily for 28 days, either alone or 1 h before TQ (10 mg/kg) or Eb (5 mg/kg) administration. Renal tissue As concentration and oxidative stress markers, including lipid peroxidation (LPO), nitrite/nitrate, and glutathione (GSH) levels, were determined. In addition to the oxidative stress response, antioxidant enzyme activities including that of superoxide dismutase, catalase, glutathione peroxidase, and glutathione reductase were measured. Exposure to As elicited a significant increase in As concentration and significant modifications to the redox state of the kidney, as was evidenced by a significant elevation in LPO and nitrite/nitrate concentration, with a concomitant reduction in GSH content and antioxidant enzyme activity. The oxidant/antioxidant imbalance observed in As toxicity was associated with a significant elevation in renal tumor necrosis factor α, interleukin 6, B-cell lymphoma 2 (Bcl-2)-associated X protein, and caspase 3 levels, in addition to a significant decrease in Bcl-2 levels. Post-administration of TQ and Eb markedly prevented As-induced oxidative stress, inflammation, apoptosis, and As accumulation in the renal tissue and reduced histological renal damage. These findings demonstrate that TQ, the main bioactive phytochemical constituent of Nigella sativa seed oil, and Eb, an organoselenium compound, could significantly inhibit As-induced oxidative damage, apoptosis, and inflammation, and significantly attenuate the accumulation of As in renal tissues by facilitating As biomethylation and excretion.
Introduction
Among many environmental toxicants, arsenic (As) is considered to be a ubiquitous pollutant diffused everywhere in the world. Currently, As poisoning affects tens of millions of people worldwide. 1 Although both metalloid forms are toxic, the inorganic forms of As (arsenite and arsenate) are more toxic than the methylated forms. 2 Exposure to inorganic forms for both long and short periods may lead to cancer of the skin, lungs, bladder, kidneys, and liver, 3,4 and non-cancer diseases such as diabetes and cardiovascular diseases. 5 However, the biological effects of As and associated disease risks remain unclear. 6
The toxicity resulting from As exposure is mediated by increased generation of reactive oxygen species (ROS), which induce oxidative injury in the kidney. Because the biochemical properties of phosphate and As are similar, As can replace phosphate in energy transfer phosphorylation reactions, resulting in unsuccessful adenosine triphosphate (ATP) synthesis. 7 Another possible mechanism of As-induced toxicity results from the ability of As to react with sulfhydryl groups that can induce structural modifications in proteins, 8 thereby leading to the inactivation of many enzymes. 9 In this regard, As inhibits pyruvate dehydrogenase and impairs ATP production by blocking the tricarboxylic acid cycle, which is critical for providing reducing equivalents that are important for electron transport to the mitochondria. 2
Many promising studies have been conducted to prevent As toxicity using antioxidants. 10 –12 Antioxidants have been widely investigated as free radicals scavengers and lipid peroxidation (LPO) suppressors in many diseases and metal-induced toxicity. 13,14 Ebselen (Eb, 2-phenyl-1,2-benzisoselenazol-3(2H)-one, PZ51) is a promising antioxidant that shows glutathione peroxidase (GPx)-like activity. This lipid-soluble selenium-containing compound displays potent cytoprotective, anti-atherosclerotic, and anti-inflammatory activities, 15 including therapeutic activity in neurological disorders, acute pancreatitis, noise-induced hearing loss, and cardiotoxicity. 16 In addition to synthetic compounds, several antioxidants from plant origin show potent activity against many aliments. Thymoquinone (TQ, 2-isopropyl-5-methyl-benzoquinone) is the bioactive phytochemical constituent of Nigella sativa seed oil and has shown promising pharmacological and therapeutic activities in in vitro and in vivo disease models with anticancer, hepatoprotective, anti-inflammatory, and antioxidant effects. 17
The nephroprotective effects of TQ and Eb on As-induced nephrotoxicity have not received any attention in the literature. This study was therefore designed to evaluate the potential roles of TQ and Eb on sodium arsenite (NaAsO2)–induced kidney injury and oxidative stress in rats. This study provides an experimental animal model for the application of TQ and Eb in the treatment of kidney damage resulting from occupational As poisoning.
Materials and methods
Chemicals
NaAsO2 (CAS number 7784-46-5), Eb (CAS number 60940-34-3), TQ (CAS number 490-91-5), and Tris-hydrochloride (HCl) were obtained from Sigma-Aldrich (St Louis, Missouri, USA). Tumor necrosis factor α (TNF-α, catalog number: RTA00) and interleukin 6 (IL-6, catalogue number: R600B) enzyme-linked immunosorbent assay (ELISA) kits were obtained from R&D Systems (Minneapolis, Minnesota, USA). Urea and creatinine kits were provided by Biodiagnostic Co. (Giza, Egypt). The chemicals and reagents used were all of analytical grade.
Animals and experimental design
Forty-two female Sprague-Dawley rats (150–180 g; 8–10 weeks old) were used. The rats were obtained from the animal facility of the VACSERA
After acclimation, rats were randomly allocated to six groups of seven animals each as follows: control group animals receiving distilled water (control group), arsenite-intoxicated animals receiving NaAsO2 (As group), animals receiving arsenite and TQ posttreatment (As.TQ group), animals receiving arsenite and Eb posttreatment (As.Eb group), animals receiving only TQ (TQ group), and animals receiving only Eb (Eb group).
Animals were inoculated with their respective doses daily for 28 days. In the As.TQ and As.Eb groups, TQ and Eb were administered approximately 1 h after As. TQ and Eb were orally administered at a dose of 10 mg/kg body weight (bw) and 5 mg/kg bw, according to Kassab and El-Hennamy 18 and Basarslan et al., 19 respectively. As was orally administered at a dose of 20 mg/kg bw, according to Yadav et al. 20
Twenty-four hours after the last administered dose, rats were killed by means of mild ether anesthesia, and blood samples were collected. Serum was isolated via centrifugation at 1300 × g for 10 min and used to determine the kidney function parameters. The right kidneys of rats were quickly removed, weighed, and washed twice in ice-cold 0.1-M Tris-HCl, pH 7.4. The kidneys were homogenized in 10 volumes of ice-cold 0.1-M Tris-HCl (pH 7.4), and kidney homogenates were centrifuged at 1300 × g for 10 min at 4°C. The supernatants were used to perform various biochemical assays. The left kidneys were kept for histological examination.
Changes in the kidney index of rats
The relative kidney weight was calculated using only the weight of the right kidney according to the following formula: right kidney weight/bw × 100.
Kidney oxidative damage
Kidney homogenates were subjected to a thiobarbituric acid reactive substance formation assay to determine the LPO levels, expressed in terms of the level of malondialdehyde (MDA) formed. 21 Nitrite/nitrate (nitric oxide (NO)) and glutathione (GSH) levels were determined after protein removal according to the methods described by Green et al. 22 and Ellman, 23 respectively.
Antioxidant status
Kidney homogenate supernatants were used to determine the activity of superoxide dismutase (SOD, according to the method described by Fisher et al.), 24 catalase (CAT, according to the method described by Aebi 25 ), GPx, and glutathione reductase (GR, according to the method described by De Vega et al. 26 ).
Quantification of renal TNF-α and IL-6
The quantification of renal TNF-α and IL-6 levels was performed using ELISA kits in kidney supernatants according to the manufacturer’s instructions. Briefly, kidney supernatants were prepared using a protease inhibitor cocktail (catalogue number: P8340; Sigma-Aldrich) from kidney samples that were collected and frozen at −20°C until analysis. Pro-inflammatory cytokine levels were measured in duplicate and expressed as picogram per gram tissue.
Histological examination
The left kidney was fixed in 10% neutral-buffered formaldehyde for a day, dehydrated in ethyl alcohol, cleared in xylene, and mounted in molten paraffin wax. The paraffin-embedded kidneys were sectioned to a thickness of 4–5 µm and stained with hematoxylin and eosin. Sections were examined using a light microscope (Nikon; Eclipse E200-LED, Tokyo, Japan).
Statistical analysis
All results are expressed as means ± standard error of the mean. Multiple variable comparisons were performed via Student’s t-test and one-way analysis of variance. For comparison of significance between groups, Duncan’s test was used post hoc. The acceptable level of significance was established as p < 0.05. Correlations were evaluated using Pearson’s test for normal distribution.
Results
At the end of the treatment course, female rats injected with As alone showed a significant increase in kidney weight (p < 0.05; F = 36.09) compared to that in control rats. Post-administration of TQ and Eb, however, significantly reduced kidney weight compared to that in As-exposed animals (Figure 1).
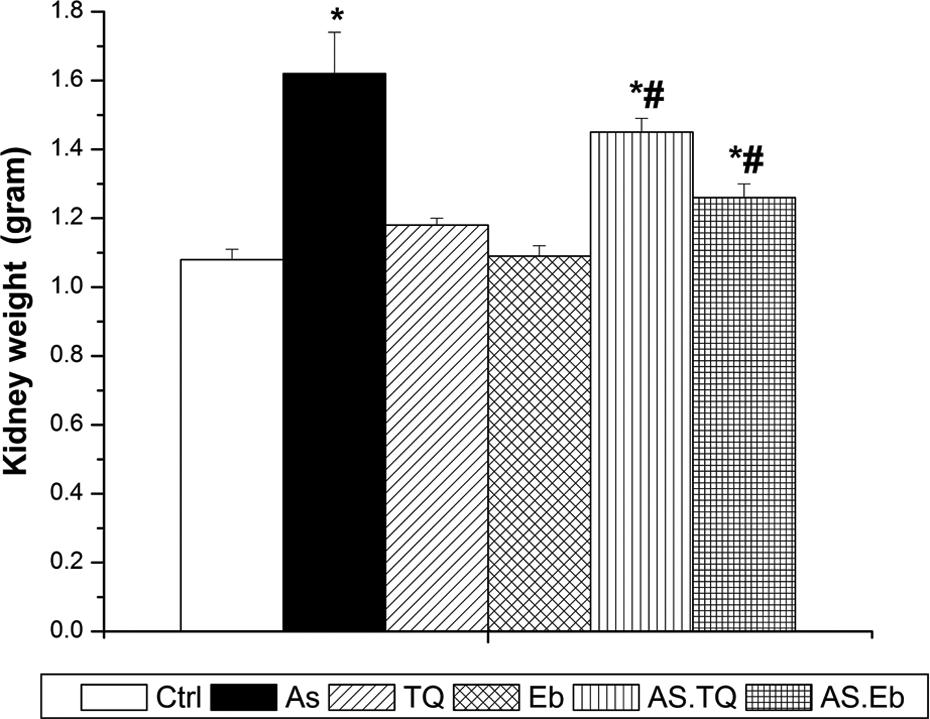
TQ and Eb prevent kidney weight change in female Sprague-Dawley rats exposed to arsenite for 28 days. Values are means ± SEM (n = 7). *p < 0.05: significant change with respect to control; #p < 0.05: significant change with respect to As for Duncan’s post hoc test. TQ: thymoquinone; Eb: ebselen; As: arsenic; As.TQ: As and TQ posttreatment; As.Eb: As and Eb posttreatment; SEM: standard error of the mean.
As concentration in kidney tissue was significantly elevated in As-treated rats compared to that in control rats (p < 0.05; F = 255.12; Figure 2). However, the post-administration of TQ or Eb significantly prevented the accumulation of As in the kidneys of rats in the As.TQ and As.Eb groups compared to that in As-treated rats.

Preventive effects of TQ and Eb on As accumulation in the renal tissue of female Sprague-Dawley rats treated with arsenite for 28 days. Values are means ± SEM (n = 7). *p < 0.05: significant change with respect to control; #p < 0.05: significant change with respect to As for Duncan’s post hoc test. TQ: thymoquinone; Eb: ebselen; As: arsenic; As.TQ: As and TQ posttreatment; As.Eb: As and Eb posttreatment; SEM: standard error of the mean.
To evaluate the potential protective role of TQ and Eb treatment following As exposure, kidney function tests were conducted in renal tissue. As caused a marked elevation in urea and creatinine levels (55.10% and 51.56%, respectively), while animals in the groups treated with both As and TQ or Eb showed a decrease in these physiological markers compared to As-treated animals (Table 1).
Protective effects of TQ and Eb on urea and creatinine in rats exposed to arsenite for 28 days.a

As: arsenic; TQ: thymoquinone; Eb: ebselen; As.TQ: arsenic and thymoquinone posttreatment; As.Eb: arsenite and ebselen posttreatment; SEM: standard error of the mean.
a Values are means ± SEM (n = 7). Percentage of change was calculated using this formula: ((y 2 − y 1)/y 1) × 100, where y 2 is the treated value and y 1 is the control value.
b p < 0.05: significant change with respect to control.
c p < 0.05: significant change with respect to As for Duncan’s post hoc test.
Kidney sections of control, TQ, and Eb group rats showed normal kidney architecture with normal glomeruli as well as proximal and distal tubular cells (Figure 3(a), (c), and (d), respectively). However, histopathological examination of the kidney revealed severe damage characterized by congestion of the blood vessels and glomeruli associated with focal hemorrhage between the degenerated tubules (Figure 3(b)). Moreover, cellular debris in the lumen of the renal tubules, severe inflammatory leukocyte infiltration, cytoplasmic vacuolation, and desquamation were observed in As-treated rats. In contrast, the kidneys of rats post-treated with TQ or Eb showed remarkable improvements in kidney architecture (Figure 3(e) and (f), respectively). However, mild congestion in the stromal blood vessels of the kidneys of TQ post-treated rats was observed.

Photomicrographs of female Sprague-Dawley rats’ kidneys treated with As and/or TQ and Eb. (a) Kidney tissues from control group animals show normal kidney structures with the presence of intact basement membranes of glomeruli and renal tubules. (b) In the arsenite-intoxicated rats, congestion in the blood vessels and glomeruli associated with focal hemorrhage between the degenerated tubules, as well as desquamation and cytoplasmic vacuolation were observed. Kidney tissues from animals in the (c) TQ and (d) Eb groups show normal kidney structures with the presence of intact basement membranes of glomeruli and renal tubules. (e) Posttreatment with TQ markedly attenuated renal damage caused by As. However, mild congestion in the stromal blood vessels was observed. (f) Posttreatment with Eb markedly attenuated renal damage caused by arsenite. Samples were stained with H&E, 400× magnification. As: arsenic; TQ: thymoquinone; Eb: ebselen; H&E: hematoxylin and eosin.
To investigate the oxidative status in the renal tissue following As administration, LPO and nitrite/nitrate levels were assessed. As treatment at a dose of 20 mg/kg bw for 28 consecutive days induced oxidative reactions in the renal tissue. The data illustrated in Figure 4 revealed that MDA and nitrite/nitrate levels were significantly elevated (p < 0.05; F = 55.81 and 42.83, respectively) in the renal tissues of As-treated rats (38.77% and 28.02%, respectively) compared to those of rats in the control group. Rats post-treated with TQ and Eb showed a significant decrease in the levels of these oxidants compared to As-treated rats.

Preventive effects of TQ and Eb post-administration on (a) lipid peroxidation, (b) nitrite/nitrate, and (c) glutathione alterations in female Sprague-Dawley rats exposed to arsenite for 28 days. Lipid peroxidation is expressed as the amount of MDA formed. Values are means ± SEM (n = 7). *p < 0.05: significant change with respect to control. #p < 0.05: significant change with respect to As for Duncan’s post hoc test. TQ: thymoquinone; Eb: ebselen; As: arsenic; MDA: malondialdehyde; SEM: standard error of the mean.
With regard to the effect of treatment on the nonenzymatic antioxidant system in the renal tissue, data shown in Figure 4 revealed that the administration of As induced a significant decrease (p < 0.05; F = 61.24) in GSH content (38.36%) compared that in corresponding control rats. However, rats post-treated with TQ and Eb showed markedly increased GSH levels when compared to As-treated rats (28.16 and 23.94%, respectively). Enzymatic antioxidant system activities are shown in Figure 5. The daily oral administration of As produced a significant reduction in the activities of SOD (−54.27%; F = 78.68), CAT (−37.20%; F = 72.61), GPx (−44.87%; F = 57.18), and GR (−43.57%; F = 30.57) when compared to those in control rats. However, the activities of these enzymes were ameliorated in the As.TQ and As.Eb groups, reflecting the potent antioxidant capacities of TQ and Eb in renal tissue following As-induced oxidative damage.
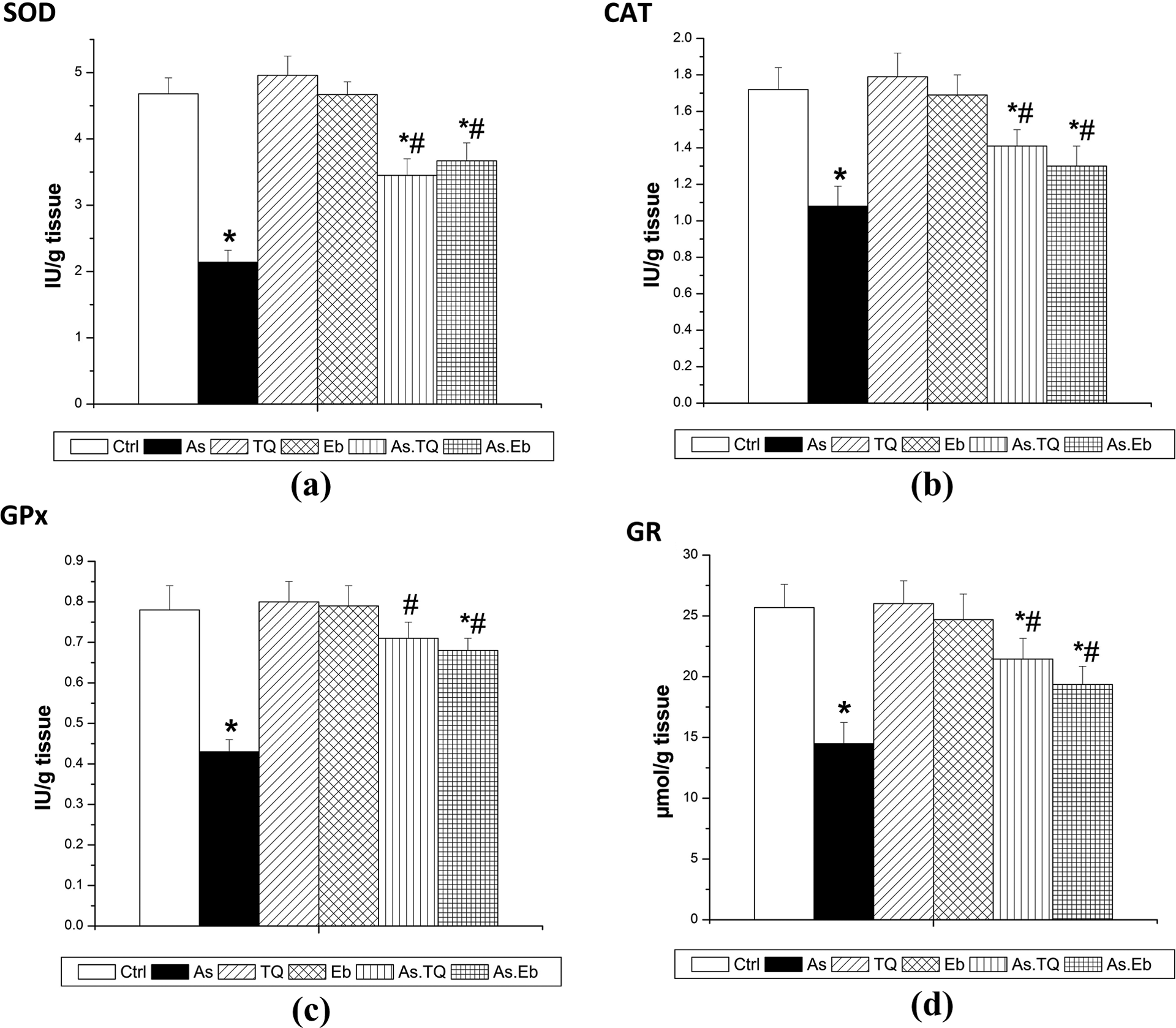
Preventive effects of TQ and Eb post-administration on antioxidant enzyme inhibition in female Sprague-Dawley rats exposed to arsenite for 28 days. (a) Superoxide dismutase, (b) catalase, (c) glutathione peroxidase, and (d) glutathione reductase. Values are means ± SEM (n = 7). *p < 0.05: significant change with respect to control; #p < 0.05: significant change with respect to As for Duncan’s post hoc test. TQ: thymoquinone; Eb: ebselen; As: arsenic; SEM: standard error of the mean.
To assess the inflammatory status following As intoxication, the levels of pro-inflammatory cytokines were measured in the renal tissue. Post-administration of TQ and Eb significantly reduced (p < 0.05) the levels of the investigated cytokines (TNF-α, F = 130.19, and IL-6, F = 63.58) in the renal tissue when compared to those in As-treated rats (Figure 6), showing that these compounds possess anti-inflammatory activity.

Effects of TQ and Eb post-administration on (a) TNF-α and (b) IL-6 levels in female Sprague-Dawley rats exposed to arsenite for 28 days. Values are means ± SEM (n = 7). *p < 0.05: significant change with respect to control. #p < 0.05: significant change with respect to As for Duncan’s post hoc test. TNF-α: tumor necrosis factor-α; IL-6: interleukin-6; TQ: thymoquinone; Eb: ebselen; As: arsenic; SEM: standard error of the mean.
NaAsO2 administration resulted in significant increase in renal B-cell lymphoma 2 (Bcl-2) associated X protein (Bax) and caspase-3 levels and a significant decrease in Bcl-2 level compared to those in control rats (Figure 7). However, rats treated with TQ or Eb after As exposure showed significantly higher Bcl-2 levels (p < 0.05; F = 17.22), and lower Bax (F = 41.04) and caspase-3 (F = 64.51) levels when compared to those of As-treated group animals, indicating the antiapoptotic activity of these compounds.

Effects of TQ and Eb post-administration on (a) Bcl-2, (b) Bax, and (c) caspase 3 in female Sprague-Dawley rats exposed to arsenite for 28 days. Values are means ± SEM (n = 7). *p < 0.05: significant change with respect to control. #p < 0.05: significant change with respect to As for Duncan’s post hoc test. TQ: thymoquinone; Eb: ebselen; Bcl-2: B-cell lymphoma 2; Bax: Bcl-2-associated X protein; As: arsenic; SEM: standard error of the mean.
Discussion
The kidneys are highly affected by As poisoning because of their ability to reabsorb and accumulate As. High deposition of As in the kidney results from the high reabsorption rate of phosphate. Arsenite and phosphate have been shown to compete for the same binding sites, with a preference for As reuptake. Once taken up by the kidney, the inorganic forms of As are methylated to form the metabolites monomethylarsonic acid (MMAV) and dimethylarsinic acid (DMAV)and are then excreted in the urine. 1 However, reduction of the kidney clearance rate due to biomethylation process saturation leads to As deposition in the kidney. 27,28
Furthermore, this biomethylation process (oxidative methylation followed by a reduction to trivalent As) is considered to be a detoxification process, although it may be an activation process, considering the formation of reactive intermediates such as monomethylarsonous acid (MMAIII) and dimethylarsinous acid (DMAIII). 29 These metabolites were shown to be 20 times more toxic than trivalent As. 30 In addition to these metabolites, the sulfur-containing methylated As metabolites monomethylmonothioarsonic acid (MMMTAV) and dimethylmonothioarsinic acid (DMMTAV) were recently discovered in human urine. 31
Administration of As increased creatinine and urea levels, and this may result from the deleterious effect of As mediated by ROS-induced oxidative damage in the kidney. This may then lead to the loss of kidney membrane integrity and fluidity and impaired cellular structure, resulting in leakage of kidney biomarkers through damaged membranes. 32,33 High levels of kidney biomarkers are indicative of kidney damage. These results are in agreement with previous findings. 13,34 Histopathological investigations revealed alterations in Bowman’s capsule and the epithelial cells of proximal convoluted tubules. 34 This alteration may be due to excessive accumulation of As and its metabolites in the renal tissue. 13,35 Treatment with TQ and Eb prevented this tissue alteration, suggesting that both TQ and Eb may have the ability to protect kidney tissue from As-induced renal damage by enhancing the biomethylation process of As. Previous studies reported the nephroprotective effects of TQ and Eb induced by different agents, as manifested by improvements in the histological appearance of the kidney with a concomitant improvement in serum biomarker levels. 36,37 LPO is a prominent marker of oxidative stress as it is known to increase production of epoxides, hydroperoxides, and MDA, all of which may interact with cellular micromolecules such as proteins, deoxyribonucleic acid, and ribonucleic acid, thereby causing renal tissue damage. 38 Here, TQ and Eb were shown to improve As-induced nephrotoxicity via their considerable effects on the reduction of LPO. The effect of TQ was supported by results from a previous report by Sayed-Ahmed and Nagi 39 who demonstrated that TQ acted as an antioxidant by scavenging superoxide, hydroxyl radicals, and free radical oxygen. Furthermore, Ozgur et al. 37 reported that Eb showed antioxidant and free radical-scavenging properties.
In the current study, As exposure elevated the levels of NO, TNF-α, and IL-6 in the renal tissues of rats. Increased NO production is implicated in As-mediated nephrotoxicity and oxidative damage. TNF-α and IL-6 are key elements in the pro-inflammatory chemokine network that are activated in arsenite-intoxicated kidneys. The elevated NO production observed in our study may contribute to the ability of As to enhance pro-inflammatory processes through stimulation of nuclear factor kappa-B (NF-κB). 40 Activation of the NF-κB signaling pathway promotes the transcription of inducible NO synthase (iNOS), TNF-α, and IL-6 genes. The nephrotoxicity of NO increases greatly when it reacts with superoxide anions to form highly reactive peroxynitrite radicals (ONOO−) that cause further cell damage by oxidizing and nitrating cellular macromolecules. Furthermore, excess NO depletes intracellular GSH levels, thereby increasing the susceptibility of the renal tissue to As-mediated oxidative stress. 14
However, both TQ and Eb proved effective in reducing the generation of ROS and pro-inflammatory mediators. The antioxidant and anti-inflammatory properties of TQ and Eb are related to the ability of TQ to prevent the expression of iNOS, TNF-α, and IL-6 proteins, 41 whereas Eb prevents the activation of the NF-κB signaling pathway, which promotes the transcription of IL-6, TNF-α, and iNOS genes. 42 This is in accordance with our results, which revealed that TQ and Eb treatment significantly attenuated the overproduction of TNF-α, IL-6, and NO and maintained the antioxidant defense mechanisms in the kidneys of rats exposed to As intoxication.
In the present work, As intoxication impaired the antioxidant system by depleting GSH content, the main endogenous antioxidant, and increasing LPO. GSH plays an important role in detoxifying As species as well as stimulating the excretion of methylated As compounds through its ability to scavenge free radicals. 35 The nephroprotective effects of TQ and Eb in maintaining GSH content at near-control levels should promote the detoxification of As and enhance the capacity of the endogenous antioxidant defense system. TQ and Eb may function by increasing steady-state GSH levels and/or the rate of synthesis, thereby conferring enhanced nephroprotection against As intoxication.
Reduced activities of antioxidant enzymes are frequently implicated in oxidative renal stress. Rats exposed to arsenite were reported to show significantly lower levels of antioxidant enzymes. As ions compete with metal ions (such as selenium, zinc, phosphate, and magnesium ions) that are essential for the activity of antioxidant enzyme, resulting in a loss of or decrease in antioxidant enzyme activity. 43 Interestingly, posttreatment with TQ or Eb prevented the activity of these enzymes from being suppressed, which suggests the regulatory role of TQ and Eb on antioxidant enzymes, and revealed their antioxidant and metal-chelating properties.
As is a well-known nephrotoxic agent, and several pathways, including apoptosis, have been documented as possible mechanisms of action. 44 Arsenite-induced nephrotoxicity may also enhance ROS generation and thus contribute to apoptosis. High ROS production in renal cells induces cytochrome c release from mitochondria and subsequently activates caspase 3, which stimulates renal apoptosis. 45 ROS may also downregulate Bcl-2 via oxidation of cysteine residues and thereby trigger apoptosis. Furthermore, Kim et al. 46 reported that arsenite induced apoptosis via both caspase-dependent and independent pathways. Our results are in agreement with the work of Uygur et al. 47 who reported that arsenite administration induced apoptosis in rat testes by activating caspase 3 and alerting Bax/Bcl-2 with a concomitant decrease in mitochondrial membrane potential, suggesting that As initiated mitochondria-mediated apoptosis. Interestingly, TQ and Eb posttreatment significantly decreased Bax and caspase 3 levels and increased Bcl-2 levels in renal tissue. Previous reports demonstrated the antiapoptotic effects of TQ and Eb. 47,48 The apoptotic prevention effect of TQ and Eb is similar. TQ mainly removes/eliminates ROS and prevents oxidation that could lead to apoptosis. 44 Furthermore, Eb activates nuclear factor erythroid 2-related factor 2, which exerts antioxidant and antiapoptotic activities and protects the cell from ROS-induced injury. 48
The nephroprotective activity of TQ and Eb was further confirmed by histological investigations and kidney function findings. Histopathological observations in As-treated rats showed the following tissue alterations: tubular necrosis, tubular degeneration, inflammatory cell infiltration, hemorrhage, swelling of tubules, and vacuolization. These tissue alterations were due to As ions, as As-increased formation of lipid peroxides and associated ROS production lead to damage in membrane integrity and other pathological changes in kidney tissue. 38,49 Administration of TQ and Eb, however, reduced the histological alterations provoked by As. These results may be attributed to the antiradical, antioxidant, anti-inflammatory, and metal-chelating activities of both TQ and Eb, which significantly reduced oxidative stress, leading to the reduction of histopathological alterations and restoration of the normal physiological state of the kidney. Further, the membrane-stabilizing properties of TQ and Eb may help to alleviate the physiological alterations caused by As in the kidney tissue of rats.
Conclusions
The present investigation demonstrated that TQ and Eb appear to preserve membrane integrity and functions in renal tissue. TQ and Eb enhance the endogenous antioxidant system, counteracting oxidative stress, inflammation, and the apoptotic cascade mediated by As toxicity in the kidney tissue.
Footnotes
Acknowledgement
The authors would like to thank Ton Duc Thang University, Ho Chi Minh City, Vietnam for supporting the current work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
