Abstract
Rapid intravenous administration of
Introduction
α-
The development of an effective and clinically safe therapeutic regimen using the intravenous route in critically ill patients especially with cardiac diseases requires a comprehensive assessment of potential toxic effects of the drug in laboratory animals and in human beings. The study reported here was designed to evaluate the potential clinical, behavioral, hematological, biochemical, gross pathological and histological toxic effects associated with intravenous administration of
Materials and methods
Test substance
Natural
Preparation of test substance for intravenous injection
Preparation of the test substance for the injection was carried out in sterile conditions. The test substance was dissolved in sterile water for injection (USP; 4.2%, pH 6.9, 270 mOsm/L), approximately 30 min prior to injection. The solution was then filtered using syringe-driven membrane filters (0.22 μm pore size; polyvinylidene fluoride (PVDF); Jet Biofil, Shanghai, China) to ensure sterility. Samples from dosage formulations were analyzed using high-performance liquid chromatography (HPLC) to verify the concentration; all samples were within 10% of the targeted concentrations. The ready-for-use solution was tested to confirm its sterility, stability, homogeneity and absence of endotoxins (limulus amebocyte lysate (LAL), Clongen Laboratories, CA, USA). Freshly prepared solution was used for injection each time.
Sterile 0.9% saline was also prepared and used for injection in control groups. The volume of the test substance administered to each animal was calculated based on the individual body weight (10 mL/kg). The test substance was administered intravenously, once daily, 7 days a week for 28 days, at approximately the same time each day.
Animals and study design
All experimental protocols were approved by the Animal Care and Use Committee (ACUC) of Jordan University of Science and Technology. Healthy, adult rabbits (Valencia V-line), weighing 2.0–2.5 kg were obtained from Alexandria University, Egypt. 18 Immediately after arrival, the rabbits were assigned individual IDs by permanent ear tattoos and placed individually in cages for observation during an acclimatization period of 1 week. Rabbits were housed in rooms with controlled temperature (21–23°C) and humidity (35%–70%). Lighting was set at 12-h light and 12-h dark cycles. Animals were fed with commercially available rabbit ration (local supplier, Jordan) and were offered fresh water as described in the following sections.
A total of 18 rabbits were selected for the study. Rabbits were distributed into three groups using a random distribution method based on body weight. Six animals (three females and three males) were used in each group. Rabbits in group 1 (
Rabbits were fasted for approximately 12 h prior to injection. Drinking water was freely provided during this period. Slow intravenous injection (5–10 min) was achieved via the marginal ear vein using 25–27 gauge sterile butterfly needles (Sunphoria Co., Ltd, New Taipei, Taiwan). To reduce damage to the sensitive ear veins due to repeated daily injections, the smallest needle possible was used, and the right and left ear veins were used alternatively. 19 Rabbits were restrained in a wooden rabbit restrainer (locally produced, Jordan) for injection. Lamp heat was used to help dilate the vein before injection.
Clinical and behavioral observation
Cage-side observation was performed every 30 min in the 4-h post administration period and twice daily after that for 28 days. Animals in the satellite group were observed for further 14 days after the last injection. Rabbits were monitored for any abnormal clinical signs or changes in behavior. The body weight, food and water intake were recorded twice per week.
The body weight was measured using a digital scale with an accuracy of up to 0.1 kg. Food was offered once daily, that is, 3 h after the completion of administration. To measure feed consumption, feed left-over was collected and its weight was obtained before a fresh amount was offered. Water was offered in clean bottles and was changed every 12 h and the amount consumed was calculated. Both feed and water consumed was reported as amount consumed per gram of body weight per day.
Laboratory evaluation
Hematology and plasma biochemistry evaluations were performed on the day of killing. Approximately 2 mL of whole blood was collected from the central ear artery/vein from all rabbits. Blood was placed in tubes containing 3.8% sodium citrate for hematology, plasma biochemistry and prothrombin time (PT) measurements. To prevent dilution effects, the amount of anticoagulants in the tubes was reduced according to the amount of blood obtained. Blood samples were transported immediately after collection to the laboratory on ice for analysis. The following hematology parameters were determined using an electronic cell counter (ABC Vet hematology analyzer, ABX Diagnostics, France): total white blood cell (WBC) and differential cell counts including percentages of segmented neutrophils, lymphocytes, monocytes, eosinophils and basophils, red blood cell count, hemoglobin concentration, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin concentration and platelets count.
Plasma was obtained by centrifugation of blood samples at 500 g for 10 min. The following plasma chemistry parameters were determined using a Sp-2100 spectrophotometer (Hinotek Technology Co., Ltd, Ningbo, China): glucose, cholesterol, blood urea nitrogen (BUN), creatinine, total protein, albumin, alanine aminotransferase (ALT) and aspartate aminotransferase (AST). BUN/creatinine ratio was calculated manually. Plasma concentrations of sodium, potassium and chlorides were determined using Easyvet (Medica Co., MA, USA). PT was determined using thrombotest reagent (Axis-Shield, Shanghai, China).
Pathological examination
Before euthanasia, the terminal body weight was recorded. Animals in groups 1 and 2 were humanely euthanized on day 29 of the experiment and those in group 3 on day 43. Animals were euthanized by exsanguination under general anesthesia using 2.5% thiopental sodium (Abbott Laboratories, North Point, Hong Kong) at 20 mg/kg intravenously. A sharp surgical blade (No. 20) was used to transect both carotid arteries.
A complete necropsy examination was performed on all animals by a certified pathologist under the supervision of the primary investigator. After careful gross pathological examination of body organs, all periorgan fat and adventitia were trimmed and organ weights were determined. The following organs were collected and weighed immediately: adrenals, brain, heart, spleen, kidneys, liver, lungs, thyroids, prostate, seminal vesicles, testes, epididymis, ovaries and uterus.
Tissue samples from the following organs were harvested and fixed in 10% neutral-buffered formalin and prepared for histopathology examination: adrenals, brain, heart, spleen, kidneys, liver, thymus, lungs, thyroids, pancreas, prostate, seminal vesicles, testes, epididymis, ovaries, uterus, aorta, sternum, cecum, colon, eyes, optic nerves, femur, injection site, lymph nodes, rectum, salivary glands, sciatic nerves, skeletal muscle, small intestine, spinal cord, stomach, tongue, trachea, urinary bladder and vagina. Tissue samples were further processed for histopathological (light microscopy) analysis after sectioning at 5 μm thickness and staining with hematoxylin and eosin stain. At least three stained slides from each organ from each animal were thoroughly examined by the pathologist.
Statistical analysis
It is important to note here that in this study, the smallest number of animals per group (three males and three females) recommended by our institutional ACUC was used to avoid excessive use of animals. This may explain minor variations that were noted in the results concerning some of the parameters included in the analysis.
Analysis was performed between all the groups using the parameters obtained within the first 4 weeks of the study only. Parameters entered in the analysis included initial body weight, weight gain, organ weights, food and water consumption and various parameters in the hematology and biochemistry analyses.
Statistical analysis was performed using the Student’s t test (two samples assuming unequal variances), from the Analysis Tool Pak of Microsoft-Excel 2003, using Windows XP Professional. Two-tail p < 0.05 was considered significant. Data were expressed in means ± SEM.
Results
All rabbits survived until the scheduled sacrifice day without any major clinical signs or behavioral alterations. In this study, male and female rabbits tolerated the intravenous administration of 4.2% solution of
There were no significant differences in body or organ weights between
Rabbits body weight, feed and water consumption over the study period (6 weeks) a
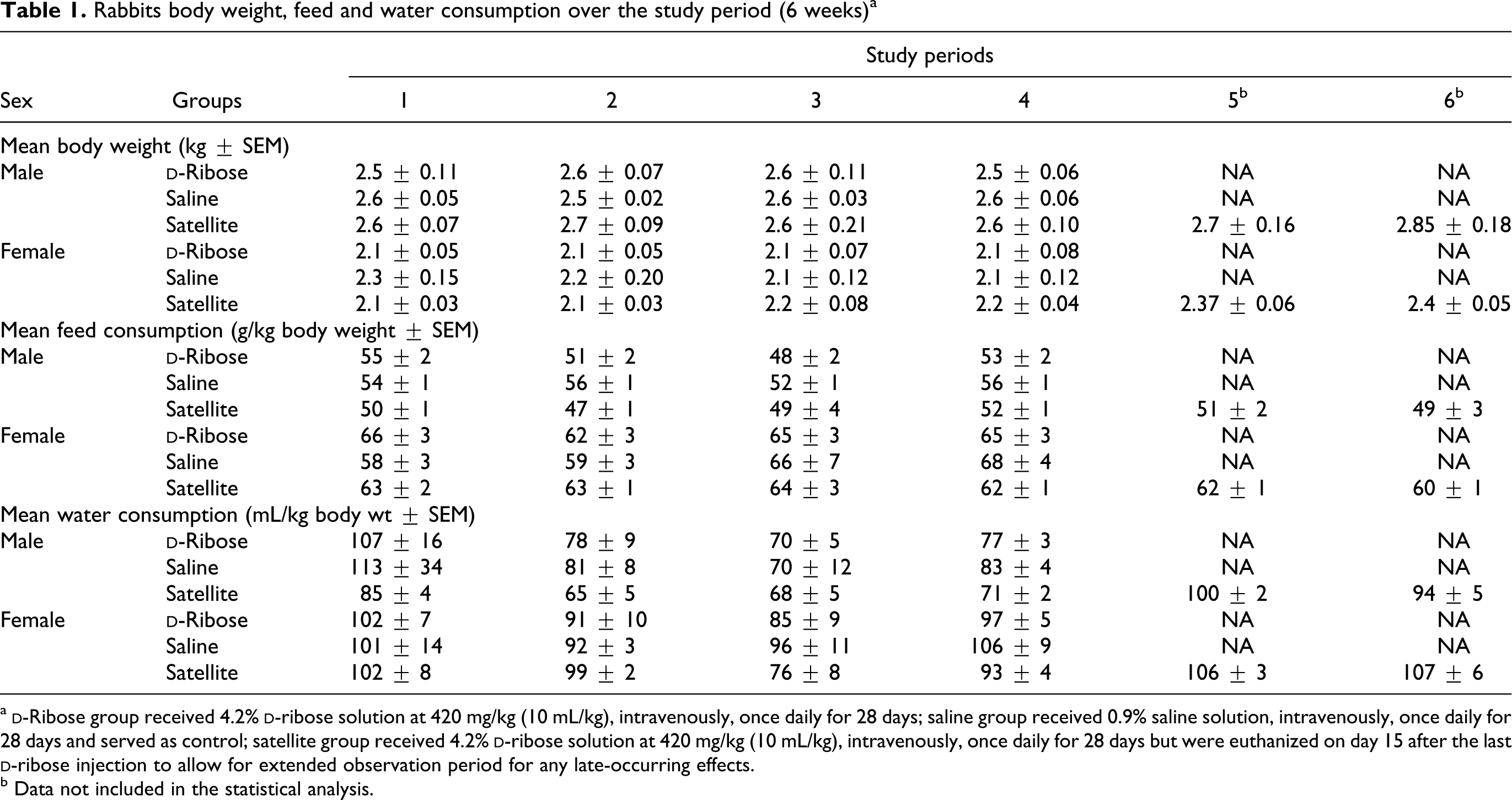
a
b Data not included in the statistical analysis.
Means ± SEM of organ weights (g) in rabbits administered 4.2%
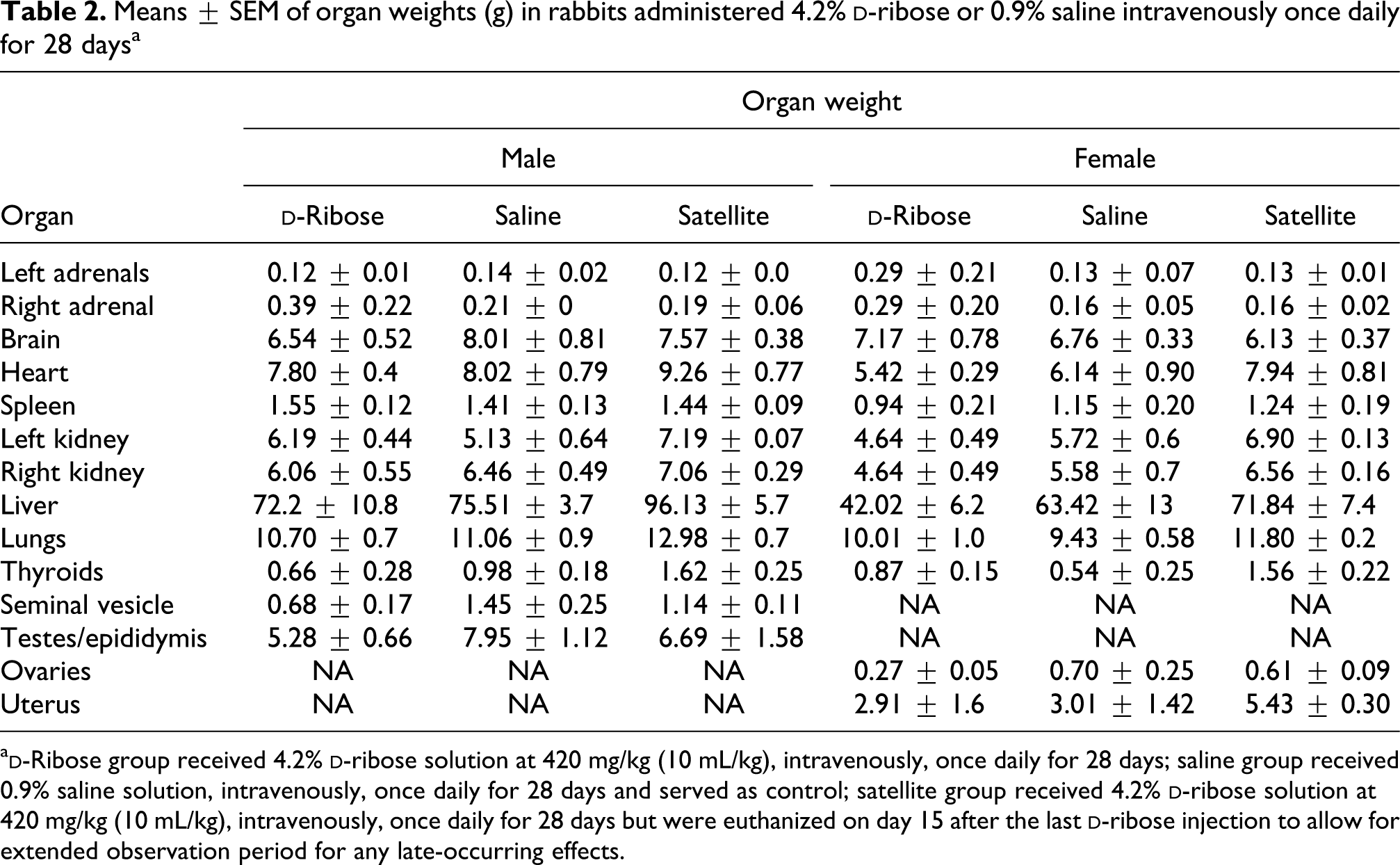
a
Results of hematology and differential leukocyte counts are presented in Tables 3 and 4. There were no significant differences in total WBC count between
Means ± SEM of red and white blood counts and percentages of lymphocytes, neutrophils, monocytes, eosinophils and basophils in rabbits administered 4.2%

RBC, red blood cell count; WBC, white blood cell.
a
b p < 0.05.
Means ± SEM of hematology parameters and PTs in rabbits administered 4.2%
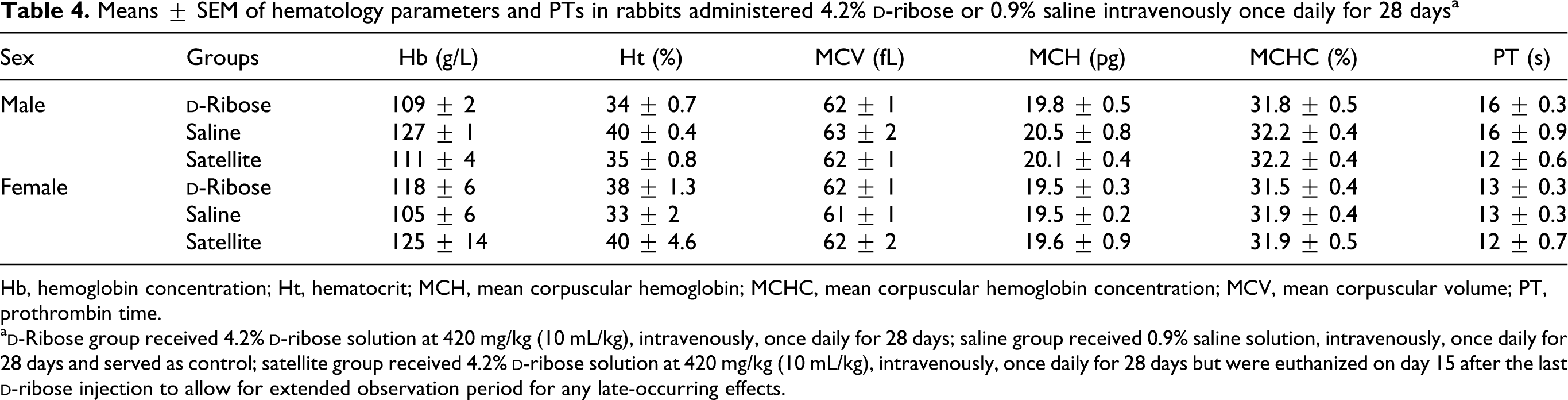
Hb, hemoglobin concentration; Ht, hematocrit; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; PT, prothrombin time.
a
Results of plasma biochemical analyses are presented in Table 5. There were no major alterations in tested parameters after
Means ± SEM of blood biochemistry parameters in rabbits administered 4.2%
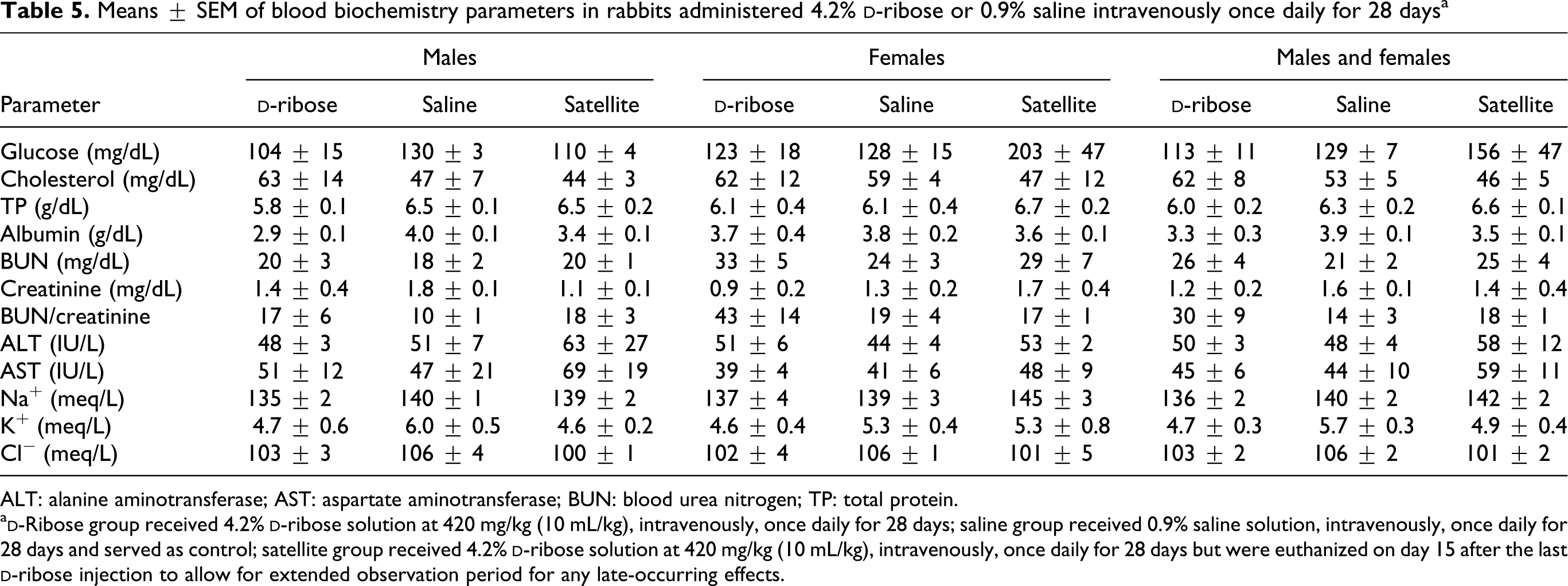
ALT: alanine aminotransferase; AST: aspartate aminotransferase; BUN: blood urea nitrogen; TP: total protein.
a
There were no significant gross pathological changes observed during postmortem examination in any of the rabbits. Extensive histopathological examination of all body organs revealed no significant abnormal changes in any of the studied organs.
Discussion
The potential therapeutic effects of
In order to eliminate possible toxic effects resulted from fluid overload, the volume of 10 mL/kg administered intravenously was not exceeded.
19,20
In this study, 420 mg/kg of
Previous clinical and preclinical toxicity studies have focused mainly on oral administration of
Griffiths and his colleagues
21
have reported a significant reduction in weight gain and no significant change in feed consumption in rats receiving 10% and 20% dietary supplement of
In the Griffiths study,
21
there was higher water consumption in rats receiving 10% and 20%
Hematology results indicate a significant increase in the percentage of neutrophils in male rabbits compared with controls. No changes were observed in female group. Similar to our results, an increased percentage of neutrophils with a reduction in lymphocyte percentages was observed in rats whose diet was supplemented with
In rats receiving 5%
BUN were increased in
Liver function parameters assessed in healthy human volunteers have shown slight but not significant changes after 7 and 14 days of oral
Griffiths and his colleagues
21
found no evidence of gross or histopathological changes in any organ after extended
In conclusion, isotonic solution (4.2%) of
Footnotes
Acknowledgements
The authors would like to thank the staff of Philadelphia Biological and Medical Product Development Centre, Jordan University of Science and Technology and The University of Jordan, Medical School for their continuous support and enthusiasm.
Funding
This project was gratefully sponsored by Heartland Biosciences International, Minneapolis, USA.
