Abstract
This study aimed to investigate the possible relationship between ovarian functionality and the oxidative response during cystogenesis induced by hyperandrogenization with letrozole and examine protective effect of the peroxisome proliferator-activated receptor gamma (PPAR-γ) agonist, pioglitazone (PIO), in polycystic ovary (PCO). Ovarian cysts were induced by oral administration of letrozol (1 mg/kg/day) for 21 consecutive days in the female rats. Effective dose of PIO (20 mg/kg/day) was administrated orally for 21 days. Serum estradiol (E), progesterone (P), testosterone (T), and the ovarian immunomodulator prostaglandin E (PGE) were analyzed as biomarkers of ovarian function. To determine the role of oxidative stress in PCO, the level of cellular lipid peroxidation (LPO), superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), and peroxynitrite (ONOO), and tumor necrosis factor alpha (TNF-α) as a marker of inflammation and apoptosis were measured in serum and the ovaries. Letrozole-induced PCO in rats exhibited a significant increase in LPO and ONOO in serum and ovary while significantly decreased serum and ovarian SOD, CAT, and GPx. Serum T and TNF-α, and ovarian PGE were increased in animals with cysts compared with healthy controls, while E and P diminished. When compared to control group, letrozole-treated group showed irregular sexual cycles, polycystic ovaries characterized by high incidence of sub-capsular ovarian cyst with diminished or scant granulosa cell layer, increased number of atretic pre-antral and antral follicles and absence of corpus luteum. There were almost no primary, secondary, and tertiary follicles observed in PCO rats. All measured parameters were improved by PIO and reached close to normal levels. The present study further supports the role of oxidative/nitrosative stress and infiammatory responses in the pathogenesis of letrozole-induced hyperandrogenic PCO rats. Results indicate that PIO is able to exert direct antioxidative and anti-inflammatory effects on the endocrine, biochemical, and pathological alterations independent of its possible effects mediated via increased insulin sensitivity in hyperandrogenized PCO.
Introduction
Polycystic ovary syndrome (PCOS) affects 4–8% of women in their reproductive years, who are diagnosed with hyperandrogenism and ovulatory dysfunction with an increased ovarian stroma and sub-capsular follicular cysts. 1 An ovarian cyst is an atretic fluid-filled follicle with a very thin layer of granulosa cells resulting from hormonal imbalance and creation of large number of the cysts in the ovaries. Close to 40% of women suffering from PCOS have anovulatory infertility problem, 2 experiencing higher rate of spontaneous abortion and pregnancy complications. 3,4 PCOS patients have higher luetinizing hormone but lower follicle-stimulating hormone levels, chronic anovulation, hyperinsulinemia/insulin resistance, glucose intolerance, obesity, altered lipid profile, prevalence of type II diabetes, 5,6 and infertility. 7 Although the precise mechanisms underlying PCOS are still not completely understood, but information about oxidative stress (OS) in PCOS is beginning to emerge. Reactive oxygen species (ROS) act not only as important signal molecules in the regulation of physiological functions in female reproduction such as folliculogenesis, oocyte maturation, steroidogenesis, corpus luteal function, and luteolysis 8 but also have a key role in pathological processes in female reproduction. 9,10 OS is a condition in which the equilibrium between body antioxidant capacity and toxic oxygen/nitrogen-derived products are impaired and thus free radicals are insufficiently detoxified by cellular antioxidants. Therefore, what we know is that the role of OS in female reproduction cannot be neglected, 11–13 and OS is involved in the pathogenesis of PCOS. 14,3 In one report of 30 women with PCOS, a decrease in total antioxidant status was noted in comparison to controls. 15 In another study, an increase in free radical activity in the peritoneal fluid environment of infertile women, some of whom had PCOS, was noted. 16 In PCOS, stimulated generation of ROS from mononuclear cells by hyperglycemia may play a key role in inflammation through the release of tumor necrosis factor alpha (TNF-α). 17,18 In this regard, insulin sensitizers including metformin and thiazolidinedione derivatives (roziglitazone and pioglitazone [PIO]) 19 are now commonly used as successful therapeutic interventions in the management of infertility in PCOS women through a mechanism that involves activation of the gamma isoform of the peroxisome proliferator-activated receptor (PPAR), a nuclear receptor. 20 This might directly affect ovarian function to restore regular menstrual cyclicity and ovulation primarily by decreasing insulin resistance. 21 PIO was approved for management of type 2 diabetes, 22 improvement of insulin sensitivity, insulin secretion, hyperandrogenism, and ovulation rates. 23 PIO by the mentioned mechanisms seems effective in stimulation of ovary and IVF outcome 24 and as an alternative to conventional therapies like clomiphene, dexamethasone, or metformin in infertile PCOS patients. 25 However, this drug is being clinically used without a complete understanding of the mechanisms involved. More recently, we found that hyperandrogenization of female rats with letrozole, an aromatase inhibitor, induced ovarian cysts and altered endocrine parameters such as serum testosterone (T), estradiol (E), progesterone (P), and TNF-α levels and ovarian prostaglandin E (PGE) concentration that were all imbalanced in the ovarian oxidant–antioxidant status, evidenced by peroxynitrite (ONOO) overproduction, the increased lipid peroxidation (LPO) index, and the diminution of enzymatic activities of antioxidants (Unpublished data). In the present work, we aimed to investigate the mechanisms of possible benefit of PIO in murine polycystic ovary (PCO) after hyperandrogenization with letrozole using biochemical, inflammatory, and pathological parameters.
Materials and methods
Chemicals
Tetraethoxypropan or malonedialdehyde, 3-mercapto-1,2-propanediol (thioglycerol), bovine serum albumin (BSA), buffer, Comassie blue, dithiothreitol, Eosin-Y, hematoxylin, phosphate-buffered saline, ethanol, paraffin, ethylenediamine tetra acetic acid (EDTA), glutathione (GSH), glutathione reductase (GR), hydrogen peroxide (H2O2), nicotinamide adenine dinucleotide phosphate, n-butanol, nitroblue tetrazolium (NBT), potassium cyanide, sodium phosphate buffer, sucrose, sulfuric acid, trichloroacetic acid, Tris-HCl, xanthine oxidase, xanthine, tetramethylbenzidine, citric acid, superoxide dismutase (SOD, 4400 U/mg protein, from Escherichia coli), rabbit antiserum, BSA, Kreb’s Ringer bicarbonate, ethyl acetate, sulfanilamide, N-1-napthylenediamine dihydrochloride, and 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) buffer from Sigma-Aldrich company (Germany), Hbt nitrotyrosine immunoassay kit, rat TNF-α and PGE enzyme-linked immunosorbent assay (ELISA) kits from Bender MedSystem (Austria), sex hormones radioimmunoassay kits from Neogen (Tehran), Letrozole from SOHA Pharmaceutical Co. (Tehran), and PIO from Osvah Pharmaceutical Co. (Tehran, Iran) were used in this study.
Animals
Sixty adult female albino rats of the Wistar strain weighing 200–210 g with normal estrous cycle were randomly divided into 4 groups of 15 animals each and accommodated in standard humidity and temperature with free access to water and food. Throughout the whole treatment, all animals were weighed daily and checked daily for 4 days of sexual cycle using vaginal smears up to the day of autopsy. Only rats showing at least three consecutive 4–5 days of regular estrous cycles were used in the experiment.
Treatments
One group of animals received only vehicle (water) orally once a day and assigned as control. PCO group were gavaged with letrozole once a day at a concentration of 1 mg/kg orally dissolved in water. PIO group received PIO 20 mg/kg/day. In the (PCO+PIO) group, PIO was administered 0.5 h before letrozole treatment. The treatment period in all groups was 21 days.
Sampling
Twenty-four hours after the last treatment, all the rats were weighted, anesthetized by ether, and blood samples were taken from the heart directly and serum was separated by centrifuge and stored at −70°C for further analysis of E, P, T, TNF-α, and OS markers. A part of dissected ovaries was immediately fixed in 4% (weight/volume [w/v]) paraformaldehyde for histomorphological studies while the other part was immediately frozen at −70°C for further determination of PGE and oxidant–antioxidant markers. The experiments were conducted according to the ethical rules approved by TUMS Review Board.
Morphological studies
After processing of formalin-fixed ovaries through paraffin embedding, they were longitudinally and serially sectioned at 4 µm with rotary microtome, placed on a glass slide, stained with H&E, and analyzed under microscope by two pathologist blinded to the origin of the sections. The measured outcomes were the number of follicles containing an oocyte with a nucleus as classified into primordial follicles (an oocyte surrounded by a single layer of granulosa cells, and a diameter <100 µm), growing follicles (an oocyte surrounded by several layers of granulosa cells, without an antrum, and a diameter of 100–300 µm) or grafian follicles (a peripheral oocyte surrounded by cumulus cells and several layers of granulosa cells, an antrum, and a diameter > 300 µm). Follicular atresia were examined for scattered pyknotic nuclei in the granulosa cell layer, detachment of the granulosa cell layers, loss of oocyte–granulosa cell communication, fragmentation and malformation of the oocyte, disruption of zona pellucida (ZP), or presence of cell debris in the antrum of the follicle.
OS biomarkers
ONOO– as a measure of nitrosative stress was assayed by Hbt nitrotyrosine immunoassay kit using nitrotyrosine detector as described previously. 26 Glutathione peroxidase (GPx) was determined by use of spectrophotometer measuring rate of oxidized form of GSH (GSSG) that is reduced by GR. 26 The basis of SOD assay was the reaction of SOD and NBT as the indicator molecule that was measured by the rate of increase at 560 nm over a 5-minute time period. 26 Catalase (CAT) activity was also determined by measuring the rate of lysis of H2O2. 26 The LPO product in serum and ovary was determined by thiobarbituric acid reactive substances expressed as the extent of lipid peroxides produced during an acid heating reaction as described previously. 27
Inflammatory biomarkers
Ovarian PGE was quantified using a rabbit antiserum and results were expressed as pg PGE/mg protein. 28 TNF-α was assessed using a rat sandwich ELISA kit and expressed as pg/mg protein. Details of the procedure have been described previously. 27 To report the concentrations of TNF-α and PGE per protein, Bradford method was used to measure protein content using concentrated Comassie blue as reagent and BSA as the standard.
Sex hormones
T, P, and E concentrations were determined by radioimmunoassay kits according to the manufacture’s brochure.
Statistical analysis
Mean and standard error values were determined for all the parameters and the results were expressed as mean ± SEM. All data were analyzed using analysis of variance and Tukey multiple comparison post hoc tests. Differences between groups was considered significant when p < 0.05.
Results
Letrozole-treated rats gained more weight than the controls (Table 1); but in the group that PIO was coadministered, the weight gain was not different from controls. When the ovary weight was compared between different groups, a significant increase in ovarian weight was observed (Table 1) whereas PIO-treated PCO animals exhibited ovarian weights similar to controls.
Comparative assessment of weight and histopathological changes of ovary in experimental and control groupsa

–: No change, +: slight change, ++: marked change, +++: sever change, PCO: polycystic ovary, PIO: pioglitazone.
a The symbols represent statistical significance: *: p < 0.05, **: p < 0.01, and ***: p < 0.001.
b Represents a significant difference between control and other treated groups.
c Represents a significant difference between PCO and PCO + PIO treated groups.
All control rats showed regular estrous cycles. Almost all letrozole-treated rats were completely acyclic and exhibited constant estrus. Treatment of letrozole-induced PCO rats by PIO showed similar estrus cyclicity as controls, suggesting the maintenance of normal sexual cycle.
Macroscopically, follicular cysts in the ovarian surface were evident. Histological parameters of ovaries taken from experimental and control groups are shown in Tables 1 and 2. Ovaries from control group exhibited follicles in various stages of development including pre-antral and antral growing follicles, and fresh corpora lutea (Figure 1: A and C). Histological examination of ovaries from letrozole-treated rats revealed an increase in the number of atretic and large cystic follicles (Table 2; Figure 1B). The morphology of cysts is characterized by a thin layer of granulosa cells undergoing compacted epithelial-like phenotype (Figures 1: D and E). There was a clear absence of growing and tertiary follicles as well as CL (Tables 1 and 2; Figure 1B). In most of atretic follicles, the oocyte was malformed and had shrinkage feature (Figure 1E). Follicles with 2–3 granulosa cell layers were exhibiting dispersed antral spaces between them (precautions antral formation) as a sign of follicular atresia (Figure 1: B and E). In some primary and secondary follicles, signs of luteinization were seen in granulosa cells confirming follicular atresia. In atretic follicles, pyknotic index in granulose cells were high, further revealing follicular atresia. There were numerous macrophages around and inside of the nonhealthy (atretic and cystic) follicles. The signs of breakdown and fragmentation were seen in ZP structure of oocyte in atretic follicles. PIO treatment of PCO rats exhibited a significant decrease in total population of ovarian atretic and cystic follicles and increased the number of corpora lutea (Tables 1 and 2; Figure 1F) in comparison to PCO rats.

(A) Section of ovary from control rat containing large numbers of growing follicles and corpora lutea (×40). (B) Section of ovary from polycystic ovary (PCO) rats exhibiting many sub-capsular small and large cysts (×40). (C) A normal antral follicle with a clear oocyte surrounded by cumulus cells and several layers of granulosa cells (×400). (D) A fluid-filled large cystic follicle of PCO rats with a degenerated thin layer of granulosa cells (×400). (E) Another section of ovary from PCO rats showing large cystic dilated follicles (*). Also an antral atretic follicle is seen in the center of this section that is characterized by a malformed oocyte with shrinkage (↑) and disintegration of granulosa cells (×100). (F) Normal developing follicles (↑) and many corpus luteum (*) in PCO + PIO group (×40). PIO: pioglitazone.
Comparison of mean number of atretic and cystic primordial, growing and grafian follicles and CL in experimental and control groupsa

PCO: polycystic ovary, PIO: pioglitazone, CL: corpus luteum.
a The symbols represent statistical significance: *: p < 0.05, **: p < 0.01, and ***: p < 0.001.
b Represents a significant difference between control and other treated groups.
c Represents a significant difference between PCO and PCO+PIO treated groups.
In the ovarian tissues and serum of PCO rats, the LPO and ONOO were significantly higher than that of controls. The ovarian and serum SOD, CAT, and GPx activities decreased significantly in PCO rats when compared to controls. PIO ameliorated all of letrozole-induced changes (Table 3).
Comparative assessment of OS and inflammation parameters in blood and ovary of experimental and control groupsa

PCO: polycystic ovary, PIO: pioglitazone, LPO: lipid peroxidation, ONOO: peroxynitrite, SOD: superoxide dismutase, CAT: catalase, GPx: glutathione peroxidase, TNF-α: tumor necrosis factor alpha, PGE: prostaglandin E.
a The symbols represent statistical significance: *: p < 0.05, **: p < 0.01, and ***: p < 0.001.
b Represents a significant difference between control and other treated groups.
c Represents a significant difference between PCO and PCO+PIO-treated groups.
As shown in Table 3, PCO rats exhibited increased level of serum TNF-α when compared with controls, while PIO treatment lowered TNF-α in PCO rats even less than that of controls.
The ovarian PGE was increased after letrozole treatment in PCO rats as compared with controls (Table 3) but PIO induced PGE in PCO group to reach normal levels.
Serum hormone levels are shown in Table 4. Treatment by letrozole induced a significant increase in serum T as compared with controls. E and P were reduced in letrozole-treated rats in comparison to controls. Serum sexual hormone levels were normalized in PIO-treated PCO rats.
Sex hormone levels in experimental and control groupsa
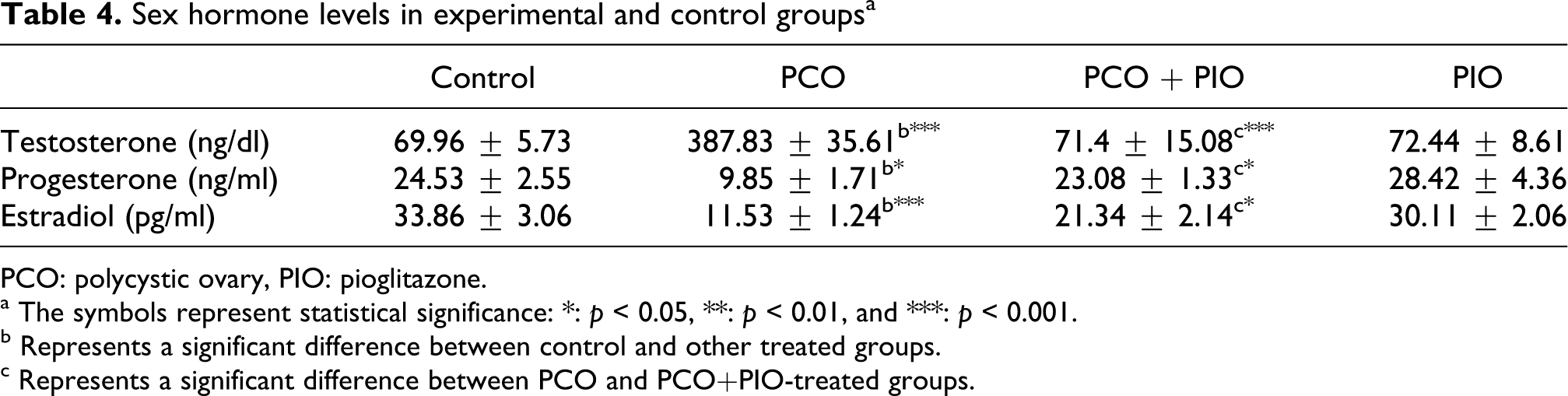
PCO: polycystic ovary, PIO: pioglitazone.
a The symbols represent statistical significance: *: p < 0.05, **: p < 0.01, and ***: p < 0.001.
b Represents a significant difference between control and other treated groups.
c Represents a significant difference between PCO and PCO+PIO-treated groups.
Discussion
The outcome of this study provided the first evidence for a mechanistic link between letrozole-induced hyperandrogenism, oxidative/nitrosative stress, and proinflammatory condition, confirming the correlation of OS and cystogenesis/anovulation. Furthermore, we found that letrozole induces higher level of T, reflecting its blocking effects on conversion of androgens into estrogens. Also, PIO prevented letrozole-induced PCO by inhibitory effect on the synthesis or release of inflammatory mediators, reduction of OS and maintaining steroid status. These results indicate that administration of PIO reduces LPO (a marker of OS) and increases SOD, CAT and GPx (markers of antioxidant potential) of letrozole-induced PCO in rats. Also, the concentrations of ONOO (marker of nitrosative stress), TNF-α, and PGE were diminished by PIO. All the present protective effects of PIO were in consistent with histological evidences showing significantly improvement in microscopic characteristics of folliculogenesis when compared to the PCO group. In support of the present findings, it was reported that hyperandrogenism induces OS status 29 in the ovary. 30 Furthermore, the intraovarian hyperandrogenism is the main cause of follicular arrest. 31 Moreover, there is a good correlation between blood total antioxidant capacity (TAC) and follicular fluid TAC, suggesting that systemic OS could influence follicular fluid. 32 Therefore, our data support the hypothesis that there is an overproduction of toxic free radicals or failure to adequately recharge the blood and ovarian enzymatic antioxidants during ovarian cystogenesis. Lower E levels shown in the present study were one of our expectations, because letrozole as a nonsteroidal aromatase inhibitor blocks conversion of T to E. It has been found that excessive production of NO causes negative effects on steroidogenesis through inhibition of cytochrome P450 aromatase which is responsible for E production. 33 Another study has shown that E has antioxidant properties and thus lower E level correlates with follicular OS. 34 Moreover, P levels decreased in PCO rats indicating anovulation as already reported in PCOS patients. 35 The increase in the number of atretic and cystic follicles in PCO rats is due to the disturbance of folliculogenesis and oogenesis. Lack of CL along with persistent estrus indicates anovulation and increased ovarian weight resulting from endocrine system dysfunction due to letrozole-induced hyperandrogenism and OS.
In the present study, PIO decreased the formation of cystic follicles and follicular degeneration and atresia induced by letrozole. Thus, when PIO acts on the ovaries, ovulation occurs and follicular cysts do not largely develop. Explaining the mechanism of action of PIO, it has been well documented that PPAR-γ plays an important role in normal ovarian function. 36 Treatment with PIO is associated with significant higher ovulation rates and improvement in hyperandrogenism. 37 Explaining the mechanism of action of PIO, it has been determined that glitazones reduce androgen levels by reducing pituitary gonadotrophin secretion, ovarian androgen secretion, adrenal androgen secretion and by increasing the plasma levels of sex hormone binding globulin. 38 Also, the PIO-mediated increase in E led to diminished androgenicity (increased E/T) and enhanced estrogenicity and normalized folliclogenesis. 39 PPAR-γ is involved in steroidogenesis, angiogenesis, tissue remodeling, cell cycle, apoptosis, and lipid metabolism, which are critical for normal ovarian function. Using the mouse mutant model, granulosa cell-specific deletion of PPAR-γ resulted in a marked impairment of ovulation due to defective follicular rupture. 40 Activators of PPAR have been shown to influence steroid production and synthesis of steroid hormones in the theca 41 and granulosa cells. 42 Therefore, granulosa cells may be indicated as one of the sites of action for the positive effect of PIO that result in normalized hormonal levels. Also, PIO contributes to the production of P in CL. In previous studies, letrozole-treated rats exhibited lower serum P, 43,44 indicating possible luteal phase defects like those found in some PCOS women. 45 In the present study, PIO corrected serum P level most probably through increasing the number of CL and a direct stimulatory effect on CL function. 46 Therefore, maintenance of normal estrus cyclicity, following PIO treatment of PCO rats could be attributed to preserving of steroid status and enabling ovarian function to be normalized.
The beneficial effects of PPAR-γ agonists in PCOS patients have been documented previously. 20,21,24 However, information about the effects of PPAR-γ agonists on OS and inflammatory response associated with PCOS are yet so limited. PIO is an antidiabetic agent with well-known antioxidant and anti-inflammatory effects against several oxidative/nitrosative stress conditions. 47,48 PIO is suggested to reduce overproduction of ROS/reactive nitrogen species directly and/or indirectly through inhibition of inflammation by reduction in LPO and decreasing nitrotyrosine and myeloperoxidase. 48 In addition, formation of LPO and nitrotyrosine are suppressed by such a treatment. 49,50 PIO imposes its neuroprotective and hepatoprotective effects via improving GSH, GPx, SOD, and CAT activity and reducing the LPO and TNF-α. 51–53 Although the molecular basis for the PPAR-γ ligand-induced increases in SOD activity, 54,55 has not been defined, it seems that Cu/Zn-SOD gene expression may be stimulated directly by PPAR-γ activation. 56 PPAR-γ is pharmacologically and pathologically associated with the expression of CAT and OS. 57 Therefore, the first thought that may come to mind is that the beneficial effects of PIO in PCO are mainly returned to its strong antioxidant potential.
Another mechanism for protective effects of PIO is downregulation of inflammatory responses. 58 A positive correlation has been demonstrated between hyperandrogenism and serum TNF-α in patients with PCOS. Mutation of the TNF receptor is associated with hyperandrogenism, 59 so that elevated serum TNF-α induces overproduction of androgen by theca cells in PCOS patients. 60 Also higher TNF-α production is involved in OS 5,61 and correlate with poor oocyte quality. 3 One of the outcomes of reduction in TNF-α would be a decline in many deleterious effects including inflammation, apoptosis, and cytotoxicity. On the other hand, it has been reported that TNF-α modulates steroidogenesis of both granulosa and theca–interstitial cells by a mechanism independent of those induced by insulin and insulin-like growth factor-I. 62 Further support of the present results come from studies indicating the relationship between PIO and TNF-α. 63–65 It is supposed that PPAR-γ may be involved in ovulation and luteinization by inhibiting the secretion of TNF-α. 66 In agreement with these findings, we found that letrozole-treated animals have higher serum TNF-α level than controls while administration of PIO restored TNF-α close to control levels. Therefore, the decrease in serum TNF-α seems an additional mechanism for positive effects of PIO.
As we expected, the ovarian PGE increased after letrozole treatment as previously reported in patients with PCOS. 67 In the present report, ovarian PGE was evaluated as a measure of inflammatory process. As previously reported, 28 hyperandrogenization causes a significant increase in ovarian PGE as a consequence of the enhanced ovarian cells LPO and cyclooxygenase-2 (COX-2) overexpression. Inhibitory effect of PIO on the production of NO by reducing the expression of COX-2 gene has been already reported. 68 Prostaglandins is known to be associated with the OS as overexpression of COX-2 induces generation of free radicals and lipid peroxides. 69 It has been clearly shown that ROS regulate the expression of many proinflammatory genes, including COX-2, 70 and administration of PIO is correlated with lower levels of COX-2. 68 Consequently, modulation of PGE by PPAR-γ agonist is another mechanism of the anti-inflammatory and antioxidative effects of PIO. Therefore, this mechanism of action of PIO would make it suitable for other free-radical-related diseases like diabetes 71 and colitis 72 .
To the best of our knowledge, this is the first study on potential relationships between oxidative/nitrosative stress and inflammatory response in letrozole-induced hyperandrogenic PCO rats. It is also the first study that differentiated direct antioxidative effect of PIO from secondary effects mediated via increased insulin sensitivity since letrozole-induced PCO is not associated with the induction of insulin resistance or reduction of insulin sensitivity. Thus, letrozole does not affect insulin-signaling pathways despite increasing testosterone levels. 73 A challenge for future studies will be to elucidate the mechanisms of actions related to potential protective effects of PIO on consequences of PCOS. The present results suggest that apart from the effects on insulin metabolism, other possible sites of action are mechanistically triggered by PIO.
Footnotes
Acknowledgements
The authors thank assistance of Osvah Pharmaceutical Co. for providing pioglitazone.
Funding
This study was partly supported by a grant (RC: 89-03-33-11332) from TUMS.
