Abstract
Acute pancreatitis is a potentially fatal disease with no known cure. The initial events in acute pancreatitis may occur within the acinar cells. We examined the effect of sesamol on (i) a cerulein-induced pancreatic acinar cancer cell line, AR42J, and (ii) cerulein-induced experimental acute pancreatitis in rats. Sesamol inhibited amylase activity and increased cell survival. It also inhibited medium lipid peroxidation and 8-hydroxydeoxyguanosine in AR42J cells compared with the cerulein-alone groups. In addition, in cerulein-treated rats, sesamol inhibited serum amylase and lipase levels, pancreatic edema, and lipid peroxidation, but it increased pancreatic glutathione and nitric oxide levels. Thus, we hypothesize that sesamol attenuates cerulein-induced experimental acute pancreatitis by inhibiting the pancreatic acinar cell death associated with oxidative stress in rats.
Introduction
Acute pancreatitis, a common disease, is potentially fatal and has no known cure. 1 The incidence of acute pancreatitis has been increasing in recent years. 2,3 In the United States, where more than 300,000 persons per year are hospitalized because of this disease, 4 the average mortality rate of acute pancreatitis is 9–15%. 5,6 In Taiwan, the incidence rate of acute pancreatitis in 2005 was 42.8 per 100,000 persons. The patients with severe acute pancreatitis (20.4%) accounted for 22.1% of total hospital days, for 40.8% of total hospital charges, and for 18.9% of hospital mortality of all patients with acute pancreatitis. 7 Although approximately 80% of all cases can be attributed to either gallstones or alcohol, 8 acute pancreatitis has no distinct etiologies. The disease is still treated with traditional methods, which are generally palliative and nonspecific. 9
The initial events in acute pancreatitis may occur within the acinar cells. 10 The pathological conversion of zymogens to active enzymes, particularly proteases, in the pancreatic acinar cell is central for initiating acute pancreatitis. 10 Products released by the zymogen and lysosomal granules lead to the intracellular activation of digestive enzymes, such as trypsin, and to acinar cell injury. 10–12 In addition, DNA damage and cell death initiated by oxidative stress are important in the pathogenesis and development of acute pancreatitis. 13–15
Sesame oil is effective against various diseases, including atherosclerosis, hypertension, and the effects of aging. 16–18 Sesamol (3,4-methylenedioxyphenol) is one important water-soluble antioxidant in sesame oil. 19 Sesamol decreases oxidative stress and protects against organ injury in animal models of sepsis. 19,20 However, whether sesamol attenuates acute pancreatitis in rats has never been investigated. We first examined the effect of sesamol on the cerulein-induced pancreatic acinar cell death and oxidative stress in a murine pancreatic acinar cancer cell line (AR42J). We then examined the effects of sesamol on experimental cerulein-induced acute pancreatitis in rats.
Materials and methods
Chemicals
Cerulein and sesamol were obtained from Sigma-Aldrich (St. Louis, MO, USA).
AR42J cells
A murine pancreatic acinar cancer cell line (AR42J) was purchased from the American Type Culture Collection (Rockville, MD, USA), and the cells were cultured in Dulbecco's modified Eagle's medium (GIBCO BRL, Life Technologies, Inc., Grand Island, NY, USA) supplemented with 10% fetal bovine serum, glutamine (2 mM), penicillin (100 U/mL), and streptomycin (100 U/mL) in a humidified atmosphere of 5% CO2 at 37°C.
Animals
Male Wistar rats (weight range: 200–300 g) were obtained from our institution’s laboratory animal center. They were housed individually in a room with a 12-h light/dark cycle and central air conditioning (25°C, 70% humidity). The animal care and experimental protocols were in accord with nationally approved guidelines. The Institutional Animal Care and Use Committee approval number is 99054.
Experimental design
Experiment 1. Effects of sesamol on amylase release and cell survival in cerulein-treated AR42J cells
AR42J cells (5 × 105 cells/mL) were divided into five groups. C-group cells were treated with phosphate-buffered saline; Cr-only-group cells were treated with cerulein (10 μM) only; CrS10-, CrS100-, and CrS1000-group cells were treated with cerulein plus sesamol (10, 100, and 1000 μM, respectively). After 24 h, medium amylase, lipid peroxidation (a marker for oxidative stress), 8-hydroxydeoxyguanosine ([8-OHdG] a marker for oxidative DNA damage 21), and cell survival were determined. All tests were done six times.
Experiment 2. Dose-response of sesamol on cerulein-induced acute pancreatitis in rats
Rats were divided into four groups of four. C-group rats (healthy controls) were intraperitoneally (i.p.) injected with saline (1 mL/kg) only; Cr-only group rats were injected with cerulein (20 μg/kg, i.p.) at 0, 1, 2, and 3 h; CrS10- and CrS30-group rats were subcutaneously (s.c.) injected with a single dose of sesamol (10 and 30 mg/kg, respectively) at 0 h and then injected with cerulein (20 μg/kg, i.p.) at 0, 1, 2, and 3 h. Pancreatic tissue from these rats was histologically examined 5 h after the first saline or cerulein injection.
Experiment 3. Protective effect of sesamol against cerulein-induced pancreatic damage and oxidative stress in rats
Rats were divided into four groups of six. C-group rats (healthy controls) were injected with saline (1 mL/kg, i.p.) only; Cr-only-group rats were injected with cerulein (20 μg/kg) at 0, 1, 2, and 3 h; CrS10- and CrS30-group rats were injected with a single dose of sesamol (10 and 30 mg/kg, s.c., respectively) at 0 h, and cerulein (20 μg/kg, i.p.) at 0, 1, 2, and 3 h. Serum amylase and lipase levels as well as pancreas tissue edema, lipid peroxidation, glutathione, and nitrite levels were determined 5 h after the first saline or cerulein injection.
Blood biochemistry analysis
We used blood biochemical analysis (Fujifilm Dri-Chem 3500s; Fujifilm, Kanagawa, Japan) to measure the levels of two enzymes, amylase and lipase, which are the indicators of pancreatic dysfunction.
Assaying cell survival
AR42J cell viability was analyzed using a variation in a colorimetric 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Cells were seeded in 96-well plates overnight. After 24 h, the cells had been treated with cerulein plus various doses of sesamol, and the culture medium was renewed with a medium containing 0.5 mg/mL of MTT and cultured for 4 h. After an equal volume of dimethyl sulfoxide had been added to the cell culture, it was spectrophotometrically measured at a wavelength of 570 nm. 21
Measuring medium 8-OHdG
Medium 8-OHdG was measured using an 8-hydroxy-2-deoxy guanosine enzyme immunoassay kit (Item No. 589320; Cayman Chemical Company, Ann Arbor, MI, USA); the spectrophotometer (DU 640B; Beckman Coulter, Inc., Fullerton, CA, USA) was read at 405 nm. 22
Measuring lipid peroxidation
Pancreatic tissue was homogenized in Tris-HCl (20 mmol; pH 7.4). The tissue homogenate (500 μL) was then centrifuged at 2500g for 10 min at 4°C. Supernatant (200 µL) or medium (200 µL) was used to measure lipid peroxidation (Lipid Peroxidase Assay Kit; Merck Biosciences GmbH, Darmstadt, Germany); the spectrophotometer was read at 586 nm. 19,20
Measuring nitrite concentration
Briefly, the nitrite concentration in medium and tissue were measured after the Griess reaction by incubating 100 μL of medium or tissue homogenate with 100 μL of Griess solution at room temperature for 20 min. The spectrophotometer was used to measure the absorbance at 550 nm. Nitrite concentration was calculated by comparing it with a standard solution with a known sodium nitrite concentration. 20
Histological examination
A histological examination was used to further assess pancreas damage. Briefly, organ tissues were fixed, at room temperature, in 4% formaldehyde that was buffered with a phosphate solution (0.1 M; pH 7.4). Organ fragments were washed in phosphate buffer, dehydrated in graded concentrations of ethanol, and then embedded in paraffin. From each tissue, 4-μm-thick sections were created and stained with hematoxylin and eosin. 23 In addition, in the histological examination, we used the Schoenberg grading system 24 to evaluate pancreatic injury. Edema grades ranged from 0 to 3: grade 0 = no tissue edema; grade 1 = interlobular edema; grade 2 = moderate interlobular edema and intraacinar edema; and grade 3 = severe interlobular and intraacinar edema. Neutrophilic inflammatory response stages ranged from 0 to 3: stage 0 = no infiltration; stage 1 = intravascular margination of granulocytes; stage 2 = granulocytes present in the perivascular tissue; and stage 3 = diffuse infiltration of the entire pancreas. Finally, fatty necrosis grades ranged from 0 to 3: grade 0 = no necrosis; grade 1 = one to four necrotic cells; grade 2 = 5–10 necrotic cells; and grade 3 = 11–16 necrotic cells (per microscopic area).
Assessing pancreatic edema
The edema index was calculated by measuring the pancreatic wet weight per 100 g of body weight. 25
Assessing glutathione level
Collected tissue samples were homogenized in ice-cold 10% (w/v) trichloracetic acid ([TCA] 1 g of tissue in 10 mL of TCA) using a tissue homogenizer. Briefly, after the tissue had been centrifuged at 3000g for 10 min, 0.5 mL of supernatant was added to 2 mL of 0.3 M of Na2HPO4·2H2O solution. A 0.2-mL solution of dithiobisnitrobenzoate (0.4 mg in 1 mL of 1% sodium citrate) was added and then the absorbance was measured at 412 nm. 26
Statistical analysis
Data are represented as means ± standard deviation (SD). Group comparisons were done using one-way analysis of variance (ANOVA) and then Tukey’s honestly significant difference (HSD) post hoc test. Statistical significance was set at p < 0.05.
Results
The effect of sesamol on amylase release and cell survival in cerulein-treated AR42J cells
To examine the protective effect of sesamol against cerulein in acinar cells, amylase activity in culture medium and cell survival were assessed. Amylase activity was significantly higher and cell survival was significantly lower in the Cr-only group than in the C group (Figure 1). Amylase activity was dose dependently lower in the three CrS groups than in the Cr-only group (Figure 1A). Cell survival was significantly higher in the CrS1000 group than in the Cr-only group (Figure 1B).

Effects of sesamol on amylase release and cell survival in cerulein-treated AR42J cells. AR42J cells were divided into five groups: C-group cells were treated with phosphate-buffered saline; Cr-only-group cells were treated with cerulein (10 μM) only; CrS10-, CrS100-, and CrS1000-group cells were treated with cerulein plus sesamol (10, 100, and 1000 μM, respectively). Culture medium amylase levels (A) and cell survival (B) were determined at 24 h. Data are means ± SD. Significant differences between groups were analyzed using one-way ANOVA. Different symbols (* and #) above the bars indicate significant (p < 0.05) differences.
The effect of sesamol on medium lipid peroxidation and 8-OHdG levels in cerulein-treated AR42J cells
To examine the effect of sesamol on cerulein-induced oxidative stress, medium lipid peroxidation (LPO) and 8-OHdG were measured. Medium lipid peroxidation and 8-OHdG levels were higher in the Cr-only group than in the C and CrS groups, and significantly so in the CrS1000 group compared with the Cr-only group (Figure 2A and B).

Effects of sesamol on culture medium lipid peroxidation (LPO) and 8-hydroxydeoxyguanosine (8-OHdG) levels in cerulein-treated AR42J cells. AR42J cells were divided into five groups: C-group cells were treated with phosphate-buffered saline; Cr-only-group cells were treated with cerulein (10 μM) only; CrS10-, CrS100-, and CrS1000-group cells were treated with cerulein plus sesamol (10, 100, and 1000 μM, respectively). Levels of malondialdehyde (MDA) (A) and 8-OHdG (B) were determined at 24 h. Data are means ± SD. Significant differences between groups were analyzed using one-way ANOVA. Different symbols (* and #) above the bars indicate significant (p < 0.05) differences.
The effect of sesamol on histological changes in the rat pancreas in cerulein-induced acute pancreatitis
To evaluate the protective effect of sesamol on cerulein-induced acute pancreatitis, the dose response of sesamol on cerulein-treated rats was studied. Significant histopathological changes—edema, inflammatory infiltration, and fatty necrosis—occurred in the pancreases of rats in the Cr-only group compared with the C group, but fewer histopathological changes in the CrS30 group than in the Cr-only group (Figure 3).

Effect of sesamol on cerulein-induced pancreatic histological changes in rats. Rats were divided into four groups of six. C-group rats (healthy controls) were i.p. injected with saline (1 mL/kg, i.p.) only; Cr-only-group rats were injected with cerulein (20 μg/kg) at 0, 1, 2, and 3 h; CrS10- and CrS30-group rats were s.c. injected with a single dose of sesamol (10 and 30 mg/kg, respectively) at 0 h, and cerulein (20 μg/kg, i.p.) at 0, 1, 2, and 3 h. Histological examination (A) and scoring (B) were done 5 h after the first saline or cerulein injection. Data are means ± SD. Significant differences between groups were analyzed using one-way ANOVA. Different symbols (*, #, and ‡) above the bars indicate significant (p < 0.05) differences. Abbreviations: A, acinus; IL, islet of Langerhans; ILD, intralobular duct; ICTS, interlobular connective tissue septa; SCI, severe cellular infiltration; SFN, severe fatty necrosis; MCI, mild cellular infiltration; FN, fatty necrosis (hematoxylin and eosin stain, original magnification ×100).
The effect of sesamol on serum amylase and lipase activities as well as pancreatic edema in cerulein-induced acute pancreatitis in rats
To confirm that sesamol protected rats against cerulein-induced experimental acute pancreatitis, serum amylase and lipase activities as well as pancreatic edema were measured. Serum amylase (Figure 4A), serum lipase (Figure 4B), and the pancreatic edema index (Figure 4C) were higher in the Cr-only group than in the C and S groups, but significantly lower in the CrS group than in the Cr-only group.

Effects of sesamol on serum amylase and lipase activities as well as pancreatic edema in cerulein-induced acute pancreatitis in rats. Rats were divided into four groups of six. C-group rats (healthy controls) were given saline (1 mL/kg, i.p.) only; S-group rats were given a single dose of sesamol (30 mg/kg, s.c.) at 0 h; Cr-only-group rats were given cerulein (20 μg/kg, i.p.) at 0, 1, 2, and 3 h; CrS-group rats were given a single dose of sesamol (30 mg/kg, s.c.) at 0 h and cerulein (20 μg/kg, i.p.) at 0, 1, 2, and 3 h. Serum amylase (A) and lipase (B) levels and pancreatic edema (C) were determined at 5 h. Data are means ± SD. Significant differences between groups were analyzed using one-way ANOVA. Different symbols (*, #, and ‡) above the bars indicate significant (p < 0.05) differences.
The effect of sesamol on pancreatic lipid peroxidation, glutathione, and nitric oxide levels in cerulein-induced acute pancreatitis in rats
To confirm the involvement of oxidative stress in sesamol-associated protection in cerulein-induced acute pancreatitis, pancreatic lipid peroxidation, glutathione (GSH), and nitrite levels were assessed. Pancreatic lipid peroxidation was significantly higher in the Cr-only group than in the C and S groups, but significantly lower in CrS group than in the Cr-only group (Figure 5A). In addition, pancreatic glutathione and nitric oxide levels were lower in the Cr-only group than in the C and S groups, but significantly higher in the CrS group than in the Cr-only group (Figure 5B and C).
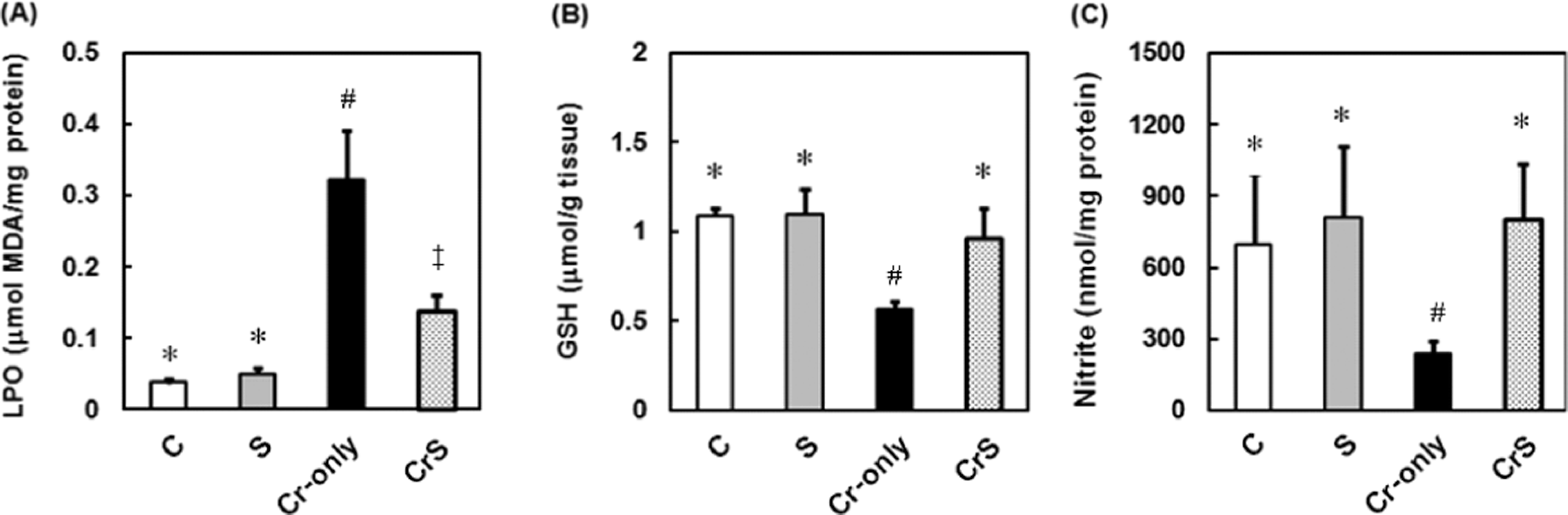
Effects of sesamol on lipid peroxidation (LPO), glutathione (GSH), and nitrite levels in cerulein-induced acute pancreatitis in rats. Rats were divided into four groups of six. C-group rats (healthy controls) were given saline (1 mL/kg, i.p.) only; S-group rats were given a single dose of sesamol (30 mg/kg, s.c.) at 0 h; Cr-only-group rats were given cerulein (20 μg/kg, i.p.) at 0, 1, 2, and 3 h; CrS-group rats were given a single dose of sesamol (30 mg/kg, s.c.) at 0 h and cerulein (20 μg/kg, i.p.) at 0, 1, 2, and 3 h. Pancreatic tissue LPO (A), GSH (B), and nitrite (C) levels were determined at 5 h. Data are means ± SD. Significant differences between groups were analyzed using one-way ANOVA. Different symbols (*, #, and ‡) above the bars indicate significant (p < 0.05) differences.
Discussion
In the present study, we found that sesamol significantly decreased oxidative stress markers (lipid peroxidation) and oxidative DNA damage markers (8-OHdG) in cerulein-treated AR42J cells and increased acinar cell survival. Furthermore, sesamol significantly protected rats against cerulein-induced experimental acute pancreatitis. Sesamol decreased pancreatic lipid peroxidation and increased pancreatic glutathione and nitric oxide levels in cerulein-treated rats. Although the exact mechanism involved in the pathogenesis of acute pancreatitis is not fully understood, impaired acinar cells may be important in the development of acute pancreatitis. 9 Oxidative stress is one of the major causes of DNA damage and the death of acinar cells. 14 Some other antioxidants—lycopene, ascorbic acid, and N-acetylcysteine—also protect acinar cells against cell death and oxidative stress-induced damage in experimental acute pancreatitis. 15,27 It is likely that sesamol protects against acute pancreatitis by inhibiting oxidative stress-associated damage to acinar cells.
Oxidative stress is imposed on cells as a result of an increase in free radicals and a decrease in circulating antioxidants. 28 In biological systems, nitric oxide is both a free radical and a scavenger of free radicals, 29–31 depending on its level. 32 Nitric oxide acts with superoxide anion to form peroxynitrite and hydroxyl radical, both of which are toxic free radicals. 19 On the other hand, nitric oxide has a lipid peroxidation-decreasing property. 29,33–35 Other studies 32,36 have reported that increasing nitric oxide protects against acute pancreatitis in rats. In the present study, sesamol maintained the nitric oxide levels in the pancreas after the cerulein challenge. Therefore, we hypothesize that increasing nitric oxide production is at least partially involved in sesamol’s antioxidative effect in cerulein-induced acute pancreatitis.
Our study shows that sesamol has potential for clinically managing acute pancreatitis. However, because oral sesamol would be degraded in the digestive system, it would be more appropriate to inject sesamol (s.c. or intravenously) when treating patients with pancreatitis. Additional studies, and clinical trials, are needed to confirm our findings and to establish a safe and effective range of therapeutic doses for humans. In summary, we hypothesize that sesamol attenuates cerulein-induced experimental acute pancreatitis by inhibiting oxidative stress-associated pancreatic acinar cell death.
Footnotes
Acknowlegments
We thank Dr Yu-Chung Chang for valuable discussion.
This study was supported by grant NSC-96-2628-B-006-038-MY3 from the National Science Council, Taiwan.
