Abstract
The aim of this study was to investigate the protective effects of taurine (Tau) on experimental acute pancreatitis (AP) in a rat model by measuring cytokines and oxidant stress markers.
Forty rats were randomly divided into four groups: sham, AP, Tau and AP + Tau. AP was induced with sodium taurocholate. No treatment was given to the AP. All rats were killed 5 days later. Pancreatic tissues of rats and blood samples were obtained.
Tau treatment significantly decreased serum amylase activity (p < 0.001), total injury score (p < 0.001), malondialdehyde levels (p < 0.001) and myeloperoxidase (MPO) activity (p < 0.001). There was no significant difference between the Tau and AP + Tau groups in serum and pancreatic tumor necrosis factor-α, interleukin (IL)-1β and IL-6 levels (p = 1.000). Histopathologic scores in the AP + Tau and Tau groups were significantly lower compared with the AP group (both p < 0.001).
These results showed that Tau reduces lipid peroxidation, amylase and MPO activities and the concentrations of proinflammatory cytokines secondary to AP and also increases superoxide dismutase and glutathione peroxidase activities in rats with sodium taurocholate-induced AP. It also has a marked ameliorative effect at histopathologic lesions. With these effects, Tau protects the cells from oxidative damage, reduces inflammation and promotes regression of pancreatic damage.
Introduction
Acute pancreatitis (AP) is a common disease associated with severe inflammatory and life-threatening complications. AP is characterized by interstitial edema, acinar cell vacuolization, and infiltration of neutrophils. 1 There are four major events that initiate the disease in acinar cells: increased capillary permeability, decreased pancreatic secretion, the activation of proteases and massive inflammatory reaction with infiltration of neutrophils and macrophages. 2 Increasing inflammation extends from the pancreas to the other organs. 2 The extrapancreatic effects of AP include extensive inflammation, disseminated fat necrosis, lung injury and shock. 3
AP may present a clinical course from a mild form to a severe necrotizing disease characterized by multiple organ damage. Experimental and clinical studies have shown that the pathogenesis of AP is complex and involves multiple mechanisms, such as oxygen free radicals, increasing inflammation that releases tumor necrosis factor-alpha (TNF-α), interleukin (IL)-1, IL-6 and proteolytic enzymes. 4 –7 The severity of AP is also largely determined by the actions of these factors. 6 Some of these factors are released by pancreatic acinar cells. 8
Taurine (Tau; 2-aminoethanesulfonic acid) is a sulfur-containing β-amino acid. 9 It has been reported that Tau and its derivatives have a key role in the innate immune response and has potential use in the prevention and treatment of various infections and inflammatory diseases. 10 In an experimental pulmonary fibrosis model, it has been shown that extension of pulmonary fibrosis and degree of inflammation are reduced by oral Tau treatment. 11 Furthermore, it also has cytoprotective effects by antioxidant mechanisms. 12 These effects of Tau rely upon its intracellular concentration. 13 Particularly, Tau levels in tissues exposed to oxidants are extremely high. 14 It was reported that Tau inhibits the production of nitric oxide (NO) and cytokines in a dose-dependent manner. 13 Some studies have reported that it may be an antioxidant and a protective agent inhibiting inducible nitric oxide synthase (iNOS). 15 Tau also has anti-inflammatory properties. 14
An effective therapeutic treatment for AP and its complications has still not been developed. The current management of AP is still symptomatic treatment. Several clinical and experimental studies for exploring anti-inflammatory and antioxidant actions of Tau in various diseases have been prophylactically treated with Tau. 9 The underlying mechanisms of AP are still not fully understood. However, the inflammatory response system and oxidant stress are most often implicated. Therefore, Tau may represent a new therapeutic option in the treatment of AP. In the present study, we aimed to investigate the protective effects of Tau on experimental AP in a rat model.
Methods
Animals
Forty adult male Sprague-Dawley rats (Health Sciences Institute, Gulhane Military Medical Academy, Ankara, Turkey), each weighing 250–350 g were used in our study. They were randomly assigned into four groups (n = 10) as follows: sham, AP, Tau and AP + Tau groups. They had free access to food and water, were kept in an air-conditioned room at 21 C with a 12 h:12 h light–dark cycle and were handled humanely, in accordance with the European Union Directive 609/86 for care and use of laboratory animals. This study was performed in the Research Center of the Gulhane Military Medical Academy with the approval of the Gulhane Military Medical Academy Animal Ethics Committee, Turkey.
Experimental design
Anesthesia was induced with inhaled sevoflurane (sevorane liquid, 250 mL; Abbott Laboratories, Istanbul, Turkey). Laparotomy was performed through a midline incision. With the aid of an operating microscope, a microaneurysm clip was placed around the common biliopancreatic duct at its entry into the duodenum to avoid reflux of enteric contents into the duct. A 28-gauge, 1/2-inch microfine intravenous needle attached to a 1-mL U-40 insulin syringe was introduced into the common biliopancreatic duct. Induction of pancreatitis was performed in the AP and AP + Tau groups; 1 mL/kg of 5% sodium taurocholate (Sigma–Aldrich, St. Louis, Missouri, USA) was injected into the common biliopancreatic duct with steady manual pressure, as described by Liu et al, 16 1 mL/kg saline was injected into the common biliopancreatic duct of all rats in the sham group. Once the rats were injected, the microclips were removed and the abdomen was closed at two layers. All procedures were performed using sterile techniques.
Tau administration
Tau (Sigma–Aldrich, St. Louis, Missouri, USA) was administered at a dosage of 300 mg/kg intraperitoneally once daily, immediately after the induction of pancreatitis in the AP + Tau group and was administered at the same dosage intraperitoneally in the Tau group.
On the fifth day, surviving animals were anesthetized with ketamine (60 mg/kg) and xylazine (10 mg/kg) intramuscularly and killed. The pancreatic tissues were removed and immediately frozen in liquid nitrogen and then stored at −80°C until assay.
Whole blood samples were drawn into tubes without anticoagulant (for preparation of serum). The serum fraction was obtained by centrifugation (2000g, 10 min, 4°C) after storing the whole blood at room temperature for approximately 1 h. Serum aliquots were stored frozen at −80°C until analysis which was performed within 30 days after sampling.
Tissue preparation
The frozen pancreatic tissues were homogenized in 50 mM phosphate buffer (pH 7.4) by means of a homogenizator (Heidolph Diax 900; Heidolph Elektro GmbH, Kelheim, Germany) on an ice cube. Homogenates were centrifuged at 7530g in 4°C for 10 min. The protein content of the pancreatic homogenates was measured according to the method described by Lowry et al. 17
Biochemical assays
Serum amylase levels were evaluated with a spectrophotometric technique by the Olympus AU-2700 autoanalyzer using commercial kits (Olympus, Hamburg, Germany). The serum and pancreatic tissue TNF-α, IL-1β and IL-6 levels were measured by enzyme-linked immunosorbent assay (ELISA) kits (Bender MedSystems GmbH, Vienna, Austria), and myeloperoxidase (MPO) levels were measured by ELISA kit (Cusabio Biotech Co. Ltd, Wuhan, Hubei Province, China).
Superoxide dismutase (SOD) activity was measured as previously described by Aydin et al. 18 Briefly, 50 µL supernatant was mixed with 850 µL of substrate solution containing 0.05 mmol/L xanthine sodium and 0.025 mmol/L 2-(4-iodophenyl)-3-(4-nitrophenol)-5-phenyltetrazolium chloride in a buffer solution containing 50 mmol/L 3-(cyclohexylaminol)-1-propanesulfonic acid and 0.094 mmol/L ethylenediaminetetraacetic acid (pH 10.2). Xanthine oxidase (80 U/L), 100 µL, was added to the mixture and the increase in absorbance was followed at 505 nm for 3 min. SOD activity was expressed as unit per milligram protein.
Glutathione peroxidase (GPx) activity was measured as previously described by Aydin et al.18 Briefly, a reaction mixture containing 1 mmol/L disodium ethylenediamine tetraacetate, 2 mmol/L reduced glutathione, 0.2 mmol/L nicotinamide adenine dinucleotide phosphate, 4 mmol/L sodium azide and 1000 U glutathione reductase in 50 mmol/L tris(hydroxymethyl)aminomethane buffer (pH 7.6) was prepared. The supernatant, 50 µl, and the reaction mixture, 950 µl, were mixed and incubated for 5 min at 37°C. The reaction was initiated by adding 8.8 mmol/L hydrogen peroxide and the decrease in absorbance recorded at 340 nm for 3 min. GPx activity was expressed as unit per milligram protein.
Lipid peroxidation was estimated by the measurement of thiobarbituric acid reactive substances in the supernatant by the method as described by Al-Fawaeir et al.19 After the reaction of malondialdehyde (MDA) with thiobarbituric acid, the reaction product was followed spectrophotometrically at 532 nm, using tetrametoxypropane as a standard. The results were expressed as nanomole per milligram protein.
Histopathological analysis
Pancreatic tissues from each animal in all groups were fixed in formalin, embedded in paraffin, and cut with a microtome set at a thickness of 5 μ. The pancreatic tissue sections were stained with hematoxylin and eosin and then examined with a light microscope. Each slide was evaluated by an expert investigator, and histological changes were graded in a blind manner as previously described. 20 Briefly, for the evaluation of the pancreatic tissue injury, from each slides 10 randomly chosen microscopic fields were examined. Histological changes were scored on a scale of 0–3 (normal to severe) in five categories (edema, hemorrhage, infiltrations, necrosis and fibrosis) for a total score of 0–15.
Statistical analysis
All the statistical analyses were performed using SPSS 15.0 (SPSS Inc., Chicago, Illinois, USA) software. Distributions were evaluated using the one-sample Kolmogorov–Smirnov test, testing whether the continuous variables are normally distributed or not. Then, we used the Kruskal–Wallis test for the ones that are not normally distributed. For pairwise comparisons, we used the Bonferroni-corrected Mann–Whitney U test with respect to distribution. For comparing the pancreatic injury, χ 2 test was used. The results were expressed as median (minimum–maximum). A probability level of p < 0.05 was considered statistically significant.
Results
In our study, no death was observed in any of the groups of rats until the end of the experiment. Serum amylase, serum and pancreatic tissue TNF-α, IL-1β and IL-6 levels are shown in Table 1. Serum and pancreatic MPO, SOD and GPx activities and pancreatic MDA levels are shown in Table 2.
Biochemical parameters in serum and pancreatic tissue in all the experimental groups.

AP: acute pancreatitis; TNF-α: tumor necrosis factor-alpha; IL: interleukin; Tau: taurine.
All data were expressed as median (min−max).
aKruskal–Wallis test.
bBonferoni-corrected Mann–Whitney U test.
The parameters of oxidative status in pancreatic tissue and serum in all the experimental groups.

AP: acute pancreatitis; MDA: malondialdehyde; SOD: superoxide dismutase; GPx: glutathione peroxidase; MPO: myeloperoxidase; Tau: taurine.
All data were expressed as median (min−max).
aKruskal–Wallis test.
bBonferoni-corrected Mann–Whitney U test.
Serum amylase levels in the AP group were significantly higher when compared to the other groups (p < 0.001). Serum amylase levels in Tau group were significantly lower than the other groups (p < 0.001). Treatment with Tau caused a significant decrease in serum amylase levels when compared with the AP group (p < 0.001).
There was no significant difference between the Tau and AP + Tau groups in terms of serum TNF-α, IL-1β and IL-6 levels (p = 1.000). However, there was a significant difference in pancreatic TNF-α levels (p < 0.001). Similar results were also shown for serum and pancreatic tissue MPO activities. Tau treatment significantly decreased serum and tissue MPO activities in the AP + Tau and Tau groups compared with the AP group (p = 0.030, p < 0.001) and (p < 0.001, p < 0.001), respectively. And there was significant difference between the Tau and AP + Tau groups for serum and pancreatic MPO activities (both, p < 0.001).
The tissue MDA levels in the AP group were significantly higher when compared to other groups (p < 0.001). In the sham group, tissue MDA levels were lower than the AP group (p < 0.001). In the AP group, the tissue SOD activities were significantly lower than the sham group (p < 0.001). There is no significant difference in terms of the tissue SOD activities between the Tau and AP + Tau groups (p = 1.000). The tissue GPx activities in the AP group were significantly lower than the sham group (p < 0.001). The GPx activities were higher in the AP + Tau group when compared with AP group rats (p < 0.001). On the other hand, there was no difference between the AP + Tau and the Tau group (p = 0.210). The difference among all groups in terms of the tissue GPx activities was statistically significant (p < 0.001).
Histopathological scores are shown in Table 3. Histological lesions were more severe in the AP group than that of the other groups (p < 0.001). Marked edema, hemorrhage, inflammatory cell infiltration, severe acinar necrosis and fibrosis were detected in the rats with AP. The mean histopathological score of sham and Tau groups were significantly lower than that of the other groups (p < 0.001). Histological examination of pancreatic tissues confirmed amelioration by treatment of Tau on AP (Figure 1). Microscopically, edema, hemorrhage, acinar necrosis and inflammatory cell infiltration were improved more in the AP + Tau group than in the AP group (p < 0.001). Tau treatment caused a clear reduction in the severity of pancreatitis.

Pancreatic tissue sections demonstrate histological changes in the study groups. (a) Sham-operated group, pancreatic tissue section shows normal acinar (Ac) structure and the islets of Langerhans (L). (b) AP group, massive destruction is seen in acinar glandular structure and islet cells of pancreatic tissue. (c) Tau group, normal structure of pancreatic tissue is seen in the section. (d) AP + Tau treatment group, relative preservation in pancreatic structure is seen compared with the AP group. (H&E, Scale bars 200 µm). AP: acute pancreatitis; H&E: hematoxylin and eosin; Tau: taurine.
Histological injury criteria in pancreatic tissues in all groups.
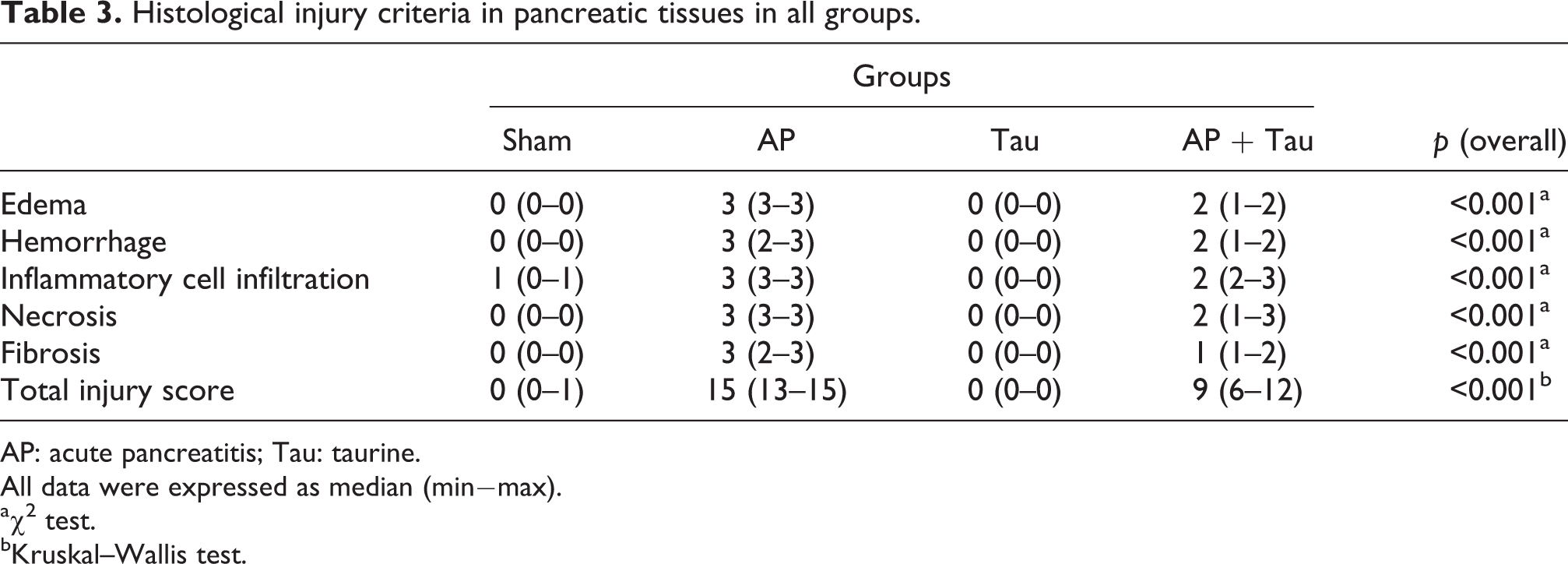
AP: acute pancreatitis; Tau: taurine.
All data were expressed as median (min−max).
a χ 2 test.
bKruskal–Wallis test.
Discussion
AP is characterized by severe inflammation in the pancreatic tissue with infiltration of macrophages and neutrophils. These cells produce and release proinflammatory cytokines such as TNF-α, IL-1β and IL-6. 21 As frequently described in experimental studies, these cytokines have important roles in stimulating pancreatic damage. 22 For example, serum IL-6 levels have been suggested to predict the severity of disease. 23
The extent of the infiltration of neutrophils into the pancreatic tissue was shown by measuring the MPO activity in tissue samples. 24 It has been suggested that inhibition of both local and systemic inflammation might prevent organ damage in the AP. In inflammatory conditions, Tau acts as an important immunoregulatory factor that affects the synthesis of cytokines. 25
Several experimental and clinical studies have shown that reactive oxygen species (ROS) have a pivotal role in the pathogenesis of AP. In the early stage of AP, prominent ROS production leads to oxidative stress. 26 ROS and reactive nitrogen species also have an important role in the occurrence of local inflammation and systemic complications of the AP. 27 It was previously reported that ROS levels produced by neutrophils were enhanced patients with AP. It has been shown that ROS and cytokines released by neutrophils adversely influence the development of acinar cell necrosis. 28 In another study, Pereda et al. have reported that the interaction between proinflammatory cytokines and oxidative stress is defined as the major cause leading to amplification of uncontrolled inflammatory cascade and initiation of systemic inflammatory response in AP. 29
Tau has both antioxidant and anti-inflammatory properties. Tau has antioxidant effects through scavenger oxygen species and nitrogen free radicals. 15 Tau is known as a cytoprotectant and is the only known endogenous inhibitor of iNOS. 5 Tau regulates inflammatory process through the generation of Tau chloramine (TauCl). Particularly activated phagocytic cells, TauCl is produced by the reaction between Tau and hypochlorous acid. 9 TauCl inhibits the translation and transcription of iNOS gene, the production of superoxide anion, NO and proinflammatory cytokines. At the same time, it has been reported that exogenous Tau increases TauCl formation in the inflammatory cells. 30 Some effects of Tau depend on the amount of intracellular concentration.9 It has been suggested that the intracellular Tau levels are important for its antioxidant effects. 31
In a rat model of mastitis, Tau treatment significantly decreased the MPO activity in mammary tissue. 32 In another study, Akdemir et al. have shown that the Tau treatment significantly decreased the numbers of neutrophils and the level of necrosis in tissue. 33 These results have suggested that the infiltration of neutrophils is modulated by Tau. MPO activity has been used to determine quantitatively the extent of neutrophil infiltration. 24 In an experimental study, Yilmaz et al. have reported that pancreatic MPO levels were significantly elevated in rats with AP. 34 Similar findings were observed in our study. As shown in our results, MPO activities in both serum and pancreatic tissue decreased after Tau treatment. Therefore, Tau also demonstrates anti-inflammatory effect preventing the infiltration of neutrophils and consequently reducing the release of inflammatory cytokines in pancreatic tissue.
Some experimental studies have shown that TNF-α, IL-1β and IL-6 play important roles in promoting pancreatic damage and in propagating injury. 35 In our study, the concentrations of cytokines including TNF-α, IL-1β and IL-6 were increased in pancreatic tissue of the AP rats, not in serum. In this study, we also investigated the effects of Tau on AP. Our results showed that Tau not only decreased the severity of pancreatic damage but also inhibited the release of proinflammatory cytokines TNF-α, IL-1β and IL-6 in serum and pancreatic tissue. Our data demonstrated that Tau also represents a favorable therapeutic strategy for AP.
In our study, edema, hemorrhage, severe acinar necrosis and fibrosis occurred in pancreatic tissues of the rats with AP. We also showed inflammatory cell infiltration in the inflammatory pancreatic sections. In addition, it has been shown that Tau treatment reduced the infiltration of neutrophils to the inflammatory area of pancreas in the AP + Tau rats, which represents an important additional mechanism for its protective anti-inflammatory effects. After treatment with Tau, we also found significant improvements in the other histological findings in pancreas.
In conclusion, Tau reduces lipid peroxidation, amylase and MPO activities and the concentrations of proinflammatory cytokines secondary to AP and also increases SOD and GPx activities in rats with sodium taurocholate-induced AP. It also has a marked ameliorative effect on histopathological lesions. With these effects, Tau protects the cells from oxidative damage, reduces inflammation and promotes regression of pancreatic damage. These findings may be important for preventing the complications of AP. To evaluate the potential roles of Tau in AP, detailed future studies are necessary.
Footnotes
Acknowledgments
The authors would like to express their sincere appreciation to FAVOR (FMF Arthritis Vasculitis and Orphan Diseases Research/www.favor.org.tr) Web registries at Gulhane Military Medical Academy, Institute of Health Sciences for their supports in epidemiological and statistical advisory and invaluable guidance for the preparation of the manuscript.
Conflict of Interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
