Abstract
We investigated the toxic effect of nevirapine (NVP; Viramune®), an antiretroviral drug, on the liver, kidney and testis of Wistar rats. Twenty-one rats were assigned into 3 groups of 7 animals each. The first group served as control, and the second and third groups received NVP at 18 and 36 mg/kg body weight, respectively. Clinical signs of toxicity were not observed in the animals. NVP at both doses did not significantly (p > 0.05) alter the body weight gain, relative weights of kidney and testis, serum protein, urea, creatinine and alkaline phosphatase levels of the animals. However, NVP2 significantly (p < 0.05) increased the relative weight of liver, level of serum total bilirubin and activities of γ-glutamyl transferase, alanine and aspartate aminotransferases. NVP administration caused a dose-dependent, significant (p < 0.05) elevation of lipid peroxidation measured as malondialdehyde (MDA) content in the liver, kidney and testis of the rats. Hepatic, renal and testicular MDA were increased by 107%, 80% and 163%, respectively, in NVP2-treated rats. Elevation in MDA was accompanied by a significant (p < 0.05) decrease in the activities of hepatic, renal and testicular superoxide dismutase and catalase. NVP2 caused 43% and 32% decrease in spermatozoa motility and live/dead sperm count, respectively, and 94% increase in total sperm abnormalities. Histopathological findings showed that NVP2 caused degeneration of seminiferous tubules in testis, and severe necrosis in liver slides. NVP induced oxidative stress with corresponding decrease in antioxidant status of the rats. The changes in sperm parameters and, elevation of liver marker enzymes suggest an interference of NVP2 with these organs.
Introduction
Infection with human immunodeficiency virus 1 (HIV-1) results in a progressive loss of the number and function of CD4+ lymphocytes, which is associated with the risk of developing opportunistic infections and neoplasms. The use of highly active antiretroviral therapy (HAART) as the standard treatment of HIV infection has led to a dramatic decline in HIV-associated morbidity and mortality. 1 The success of current regimens has transformed HIV infection into a chronic condition requiring management over the course of years and decades. Nonnucleoside reverse transcriptase inhibitors (NNRTIs) have been shown to be superior alternatives to protease inhibitors (PIs), owing to serious metabolic complications caused by PI in HIV-infected patients and have become popular as the first-line therapies. 2,3 However, the drugs in this class have been associated with early adverse effects. 4 Nevirapine ([NVP] Viramune; Boehringer Ingelheim; Figure 1) is an NNRTI used in first-line treatment regimens, and it has also shown efficacy in decreasing the vertical transmission of HIV-1. 5 The acute hepatic toxicity of NVP has led to a change in the product label in both the United States and the European Union. 6 Hepatotoxicity, liver enzymes elevation, and drug interactions are a significant problem in HIV patients on NVP. In patients commencing antiretroviral therapy (ART) 14–20% may experience elevations of liver enzymes. 7 Also, because of severe hepatic injury and marked elevation of liver enzymes, 2–10% of patients are often advised to discontinue ART. 8 The diagnosis of liver disease by hepatologists in HIV patients are mainly based on abnormal liver enzymes. However, abnormalities in liver function may be caused by HIV itself, hepatitis viruses, systemic opportunistic infections, malignancies, and drug-induced hepatotoxicity. 9 Because of shared routes of transmission, coinfection with hepatitis B and/C viruses is common in HIV patients, and liver disease is becoming a later-onset toxicity that may reflect metabolic idiosyncrasy. 10 Thus, coinfection with hepatitis viruses may increase the risk of hepatotoxicity induced by NVP. From the aforementioned, liver toxicity observed during NVP therapy may or may not be linked directly to the drug since other predisposing factors may be available in the patients. Therefore, this study was designed to evaluate the possible toxic effects of NVP in the tissues (liver, kidney and testis) of male Wistar rats, given the human therapeutic and double therapeutic doses of the drug.

Structure of nevirapine (Viramune®; chemical IUPAC name is 1-cyclopropyl-5,11-dihydro-4-methyl-6H-dipyrido [3,2-b:2′,3′-e][1,4] diazepin-6-one).
Materials and methods
Chemicals
NVP (Viramune) produced by Boehringer Ingelheim (Germany) was used for this study and was obtained from PEPFAR (President’s Emergency Plan for AIDS Relief) Clinic, University College Hospital, Ibadan, Nigeria. Glutathione, hydrogen peroxide, 5,5′-dithios-bis-2-nitrobenzoic acid (DTNB) and epinephrine were purchased from Sigma Chemical Co., Saint Louis, MO, USA. All other chemicals used were supplied by Zayo Company, Jos, Nigeria, which is an accredited supplier for Sigma and BDH.
Animals
Inbred 8 weeks old male Wistar albino rats weighing between 220 and 225 g were purchased from the animal house of the Department of Veterinary Physiology, Biochemistry and Pharmacology, University of Ibadan, Nigeria. The animals were kept in well-ventilated cages at room temperature (28–30°C) and under controlled light cycles (12-h light/12-h dark). They were maintained on normal laboratory chow (Ladokun Feeds, Ibadan, Nigeria) and water ad libitum. Rat handling and treatments conform to the guidelines of the National Institute of Health (NIH publication 85-23, 1985) for laboratory animal care and use.
Study design
Twenty-one male albino rats (Wistar strain) were randomly distributed into three groups of seven animals each. Animals were given a period of 2 weeks for acclimatization before the experiment. The first group served as the control and was given corn oil (vehicle for drugs). The second group received NVP at a dose of 18 mg/kg body weight (NVP1; equivalent of human therapeutic dose) and the third group received NVP at a dose of 36 mg/kg body weight (NVP2; Twice therapeutic dose). Previous studies revealed the human therapeutic dose of NVP and thus informed the choice of dosage for this study. 11 NVP was prepared with corn oil and given daily to the animals by oral gavage for 4 consecutive weeks.
Preparation of tissues
Rats were killed 24 h after the last dose of drugs and an overnight fast by cervical decapitation. Liver, kidney and testis were quickly removed and washed in ice-cold 1.15% KCl solution, dried and weighed. A section of the liver and kidney samples were fixed in 10% formalin, while the right testis was fixed in Bouin’s solution. Other parts were homogenized in 4 volumes of 50 mM phosphate buffer, pH 7.4 and centrifuged at 10,000g for 15 min to obtain post-mitochondrial supernatant fraction Post Mitochondrial Fraction (PMF). The samples were stored at −80°C until used. All procedures were carried out at temperature of 0–4°C.
Preparation of serum
Blood was collected from the heart of the animals into plain centrifuge tubes and was allowed to stand for 1 hour. Serum was prepared by centrifugation at 3000g for 15 minutes in a Beckman bench centrifuge. The clear supernatant was used for the estimation of serum urea, creatinine and enzymes.
Biochemical assays
Protein levels in the samples were assayed by the method of Lowry et al. 12 using bovine serum albumin as standard. Serum urea and creatinine levels were determined by the method of Talke and Schubert, 13 and Jaffe, 14 respectively. The activities of alanine and aspartate aminotransferases (ALT and AST) were assayed by the combined methods of Mohun and Cook 15 and Reitman and Frankel. 16 PMF γ-glutamyl transferase (GGT) activity was assayed using Bayer diagnostic kit according to the method of Fossati et al. 17 The estimation of serum alkaline phosphatase (ALP) activity was based on the method of King and Armstrong. 18 ALP activity was measured spectrophotometrically by monitoring the concentration of phenol formed when ALP reacts with disodium phenyl phosphate at 680 nm. Serum total bilirubin level was assayed by the method of Rutkowski and Debaare, 19 the method involved the reaction between bilirubin and diazotized sulphanilic acid in alkaline medium to form a blue-coloured complex, which was read spectrophotometrically at 546 nm. Serum and PMF lipid peroxidation levels were assayed by the reaction between 2-thiobarbituric acid (TBA) and malondialdehyde (MDA), an end product of lipid peroxides as described by Walls et al. 20 PMF-reduced glutathione (GSH) level was assayed by measuring the rate of formation of chromophoric product in a reaction between DTNB and free sulphydryl groups at 412 nm as described by Moron et al. 21 PMF superoxide dismutase (SOD) activity was measured by the nitro blue tetrazolium (NBT) reduction method of McCord and Fridovich. 22 PMF catalase (CAT) activity was assayed spectrophotometrically by measuring the rate of decomposition of hydrogen peroxide at 240 nm as described by Aebi. 23
Sperm analysis
Sperm motility was assessed by methods described by Zemjanis 24 and Rezvanfar et al. 25 Epididymal sperm count was determined by the method described by Pant and Srivastava, 26 while total sperm abnormalities (TSA) were assayed by the method of Wells and Awa. 27
Histopathology of tissues
Tissues fixed in 10% formalin and Bouin’s solution were dehydrated in 95% ethanol and then cleared in xylene before embedded in paraffin. Microsections (about 4 μm) were prepared and stained with haematoxylin and eosin (H&E) dye and were examined under a light microscope by a histopathologist who was ignorant of the treatment groups.
Statistical analysis
All values were expressed as the mean ± SD of seven animals per group. Data were analyzed using one-way analysis of variance (ANOVA) followed by the post hoc Duncan multiple range test for analysis of biochemical data using SPSS (10.0). Values were considered statistically significant at p < 0.05.
Results
Effect of nevirapine on the body weight gain and relative weights of liver, kidney and testis in rats
Administration of NVP at 18 and 36 mg/kg body weight/day (NVP1 and NVP2, respectively) did not significantly (p > 0.05) alter the body weight gain and relative weights of kidney and testis of the animals when compared to controls. However, there was a significant increase (p < 0.05) in the relative weight of liver in rats treated with NVP2 when compared to controls (Table 1).
Changes in the body weight and relative weight of organs of rats treated with nevirapine for 4 weeks a
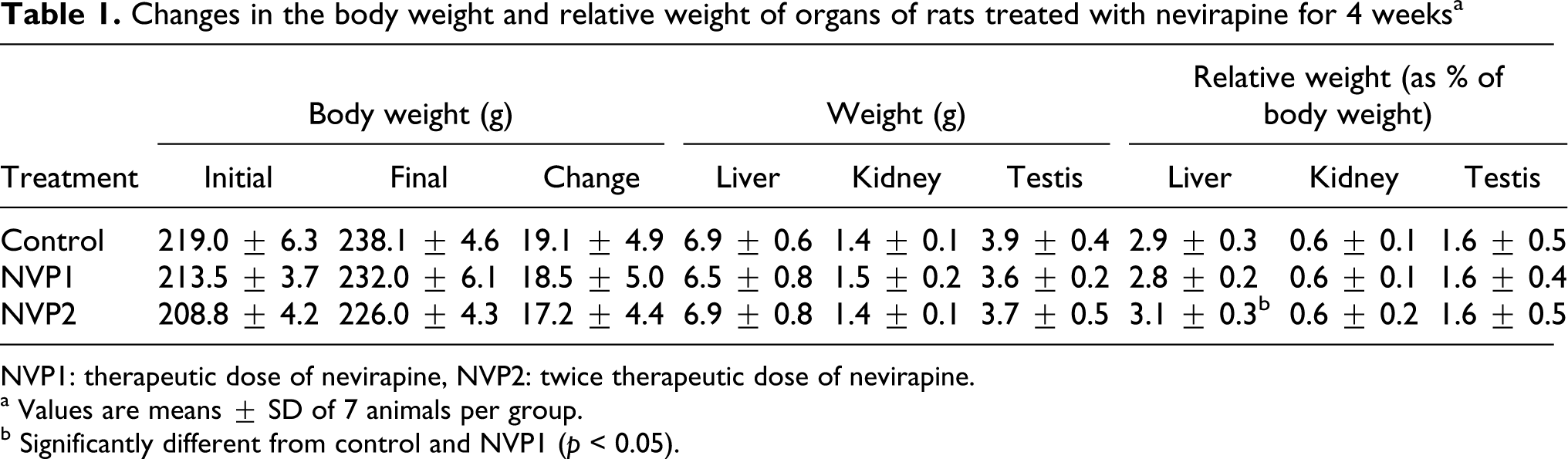
NVP1: therapeutic dose of nevirapine, NVP2: twice therapeutic dose of nevirapine.
a Values are means ± SD of 7 animals per group.
b Significantly different from control and NVP1 (p < 0.05).
Effect of nevirapine on the activities of serum enzymes and levels of urea and bilirubin in rats
There were no significant (p > 0.05) differences in the activities of serum ALT and AST, GGT and ALP of rats treated with NVP1 relative to controls (Table 2 and Figure 2). Similarly, treatment with NVP1 produced no significant (p > 0.05) effect on the levels of serum urea, creatinine and total bilirubin in the animals (Figures 2 and 3). In contrast, NVP2 intoxication significantly (p < 0.05) increased the activities of serum ALT, AST, GGT and the level of total bilirubin in the animals (Table 2; Figures 2 and 3). Precisely, ALT, AST, GGT and total bilirubin were increased by 61%, 43%, 86% and 76%, respectively.

Changes in the levels of serum alanine aminotransferase and creatinine in rats after 4 weeks of treatment with nevirapine (NVP). *Significantly different from control and NVP1 at p < 0.05. ALT: alanine aminotransferase, NVP1: rats given therapeutic dose of nevirapine, NVP2: rats given twice therapeutic dose of nevirapine.

Changes in the levels of serum urea and total bilirubin in rats after 4 weeks of treatment with nevirapine (NVP). *Significantly different from control at p < 0.05. NVP1: rats given therapeutic dose of nevirapine, NVP2: rats given twice therapeutic dose of nevirapine.
Changes in the levels of some biochemical indices in rats treated with nevirapine for 4 consecutive weeks a
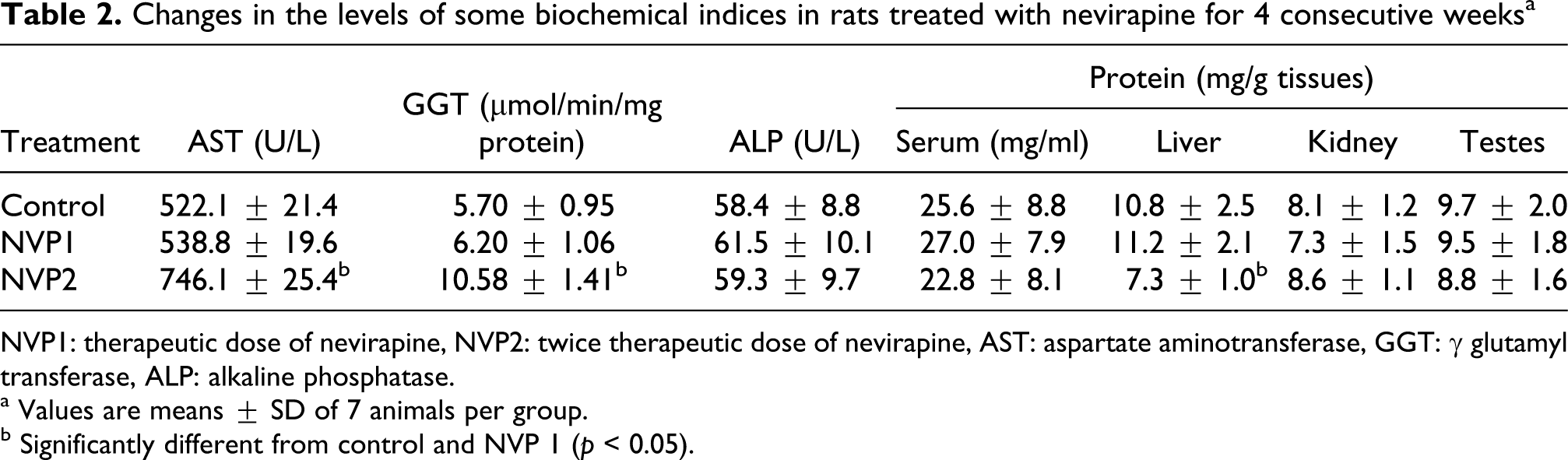
NVP1: therapeutic dose of nevirapine, NVP2: twice therapeutic dose of nevirapine, AST: aspartate aminotransferase, GGT: γ glutamyl transferase, ALP: alkaline phosphatase.
a Values are means ± SD of 7 animals per group.
b Significantly different from control and NVP 1 (p < 0.05).
Effect of nevirapine on the levels of antioxidant enzymes and lipid peroxidation in rats
NVP dose dependently caused significant increase (p < 0.05) in lipid peroxidation products as assessed by the accumulation of MDA in liver, kidney and testis of the animals. Specifically, NVP1 increased the MDA levels in the liver, kidney and testis by 102%, 75% and 92%, respectively, while NVP2 caused 107%, 80% and 163% increases in MDA levels of liver, kidney and testis, respectively (Figure 4). In NVP2-treated rats, the increase in MDA was accompanied by concomitant and marked (p < 0.05) decline in hepatic and testicular GSH levels. However, there were no significant (p > 0.05) differences in the levels of hepatic, renal and testicular GSH of rats treated with NVP1 when compared to controls (Figure 5). Administration of NVP at both doses significantly decreased (p < 0.05) the activities of hepatic SOD and CAT relative to controls (Figures 6 and 7). In addition, NVP2-treated rats had significantly decreased (p < 0.05) renal and testicular SOD and CAT activities when compared to controls (Table 3).

Changes in hepatic, renal and testicular lipid peroxidation (LPO) levels in rats after 4 weeks of treatment with nevirapine (NVP). *Significantly different from control at p < 0.05. NVP1: rats given therapeutic dose of nevirapine, NVP2: rats given twice therapeutic dose of nevirapine, MDA: malondialdehyde.

Changes in the levels of hepatic, renal and testicular reduced glutathione (GSH) in rats after 4 weeks of treatment with nevirapine (NVP). *Significantly different from control and NVP1 at p < 0.05. NVP1: rats given therapeutic dose of nevirapine, NVP2: rats given twice therapeutic dose of nevirapine.

Changes in the activities of hepatic, renal and testicular superoxide dismutase (SOD) in rats after 4 weeks of treatment with nevirapine. *Significantly different from control at p < 0.05, **significantly different from control and NVP1 at p < 0.05. NVP1: rats given therapeutic dose of nevirapine, NVP2: rats given twice therapeutic dose of nevirapine.

Changes in the activities of hepatic, renal and testicular catalase (CAT) in rats after 4 weeks of treatment with nevirapine (NVP). *Significantly different from control at p < 0.05, **significantly different from control and NVP1 at p < 0.05. NVP1: rats given therapeutic dose of nevirapine, NVP2: rats given twice therapeutic dose of nevirapine.
Effect of nevirapine on sperm parameters and total percentage abnormalities in spermatozoa of Wistar albino rats a

NVP1: therapeutic dose of nevirapine, NVP2: twice therapeutic dose of nevirapine, TSA: total sperm abnormalities, TPC: testicular protein contents.
a Values are means ± SD of 7 animals per group.
b Significantly different from control and NVP1 (p < 0.05).
Effects of nevirapine on the histology of liver, kidney and testis of rats
Figure 8a–c show representative photomicrographs of liver, kidney and testes of rats treated with NVP. In NVP1-treated rats, these tissues showed normal architectural layout with little or no visible lesions and were comparable to controls. However, in NVP2-treated rats, the liver slides showed major distortion of architecture with marked necrosis and, severe portal and central venous congestion. Likewise, slides from the testis confirmed extensive degeneration of seminiferous tubules (ST), necrosis of spermatocytes and defoliation of many spematocytes into lumen of the ST. In contrast, the kidney slides from NVP2-treated rats were comparable to the control with no visible lesion. The results of histopathology correlated with the biochemical changes in the serum and urinary indices of the animals.
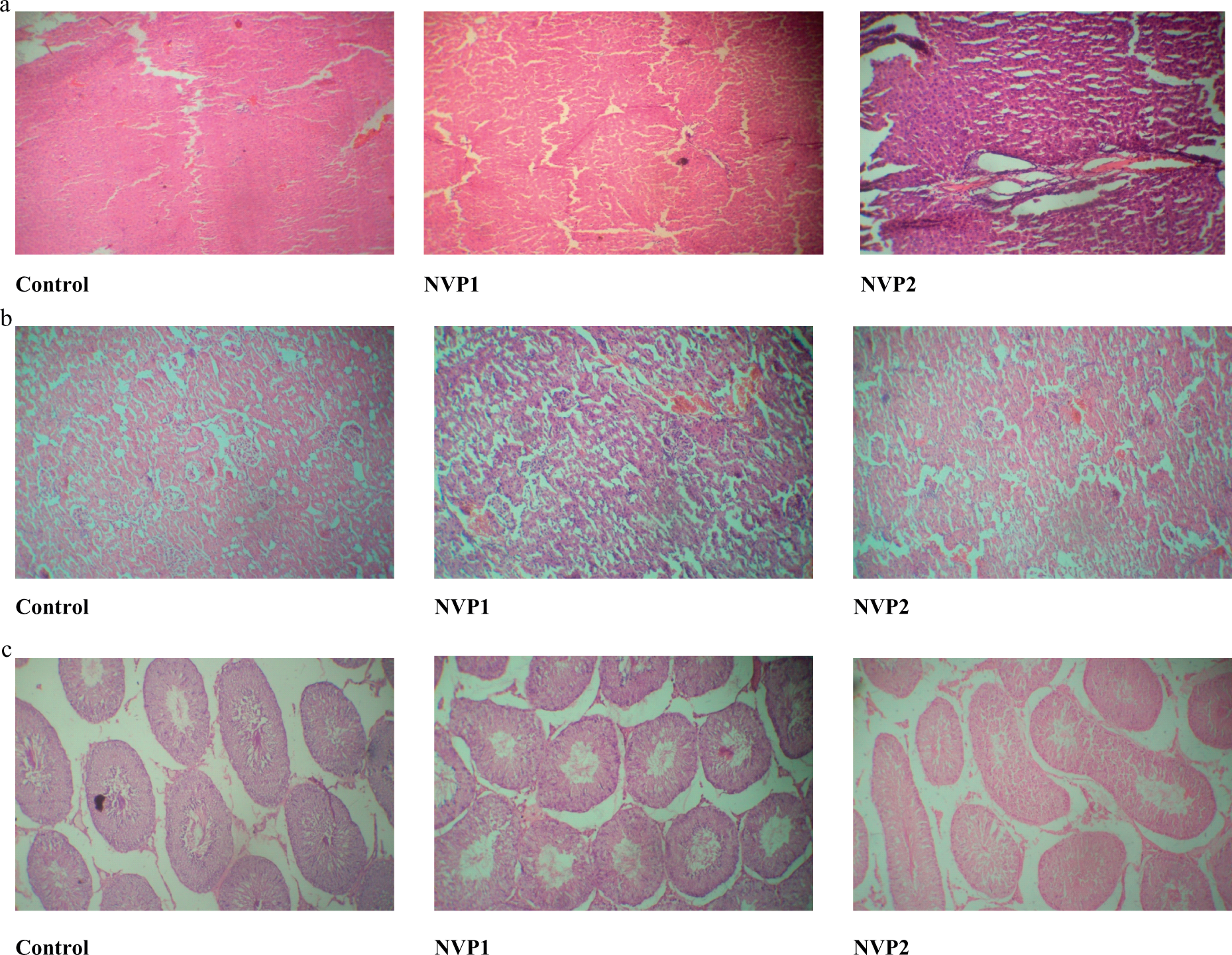
(a) Representative photomicrographs of liver slides from nevirapine (NVP)-treated rats. NVP1 received 18 mg/kg and NVP2 received 36 mg/kg (×100) haematoxylin and eosin (H&E). Normal architectural layout with no visible lesions (control); normal architecture with mild lesion (NVP1); major distortion with marked necrosis and severe portal and central venous congestion (NVP2). (b) Representative photomicrographs of kidney slides from NVP-treated rats. NVP1 received 18 mg/kg and NVP2 received 36 mg/kg (×100) H&E. Slides from control, NVP1 and NVP2 are identical. No visible lesion seen, no tubular necrosis and absence of protein casts in the tubular lumen and no interstitial hemorrhage. (c) Representative photomicrographs of slides from testis of NVP-treated rats. NVP1 received 18 mg/kg and NVP2 received 36 mg/kg (×100) H&E. Normal testicular architecture (control); normal architecture, mild necrosis (NVP1); major distortion, extensive degeneration of seminiferous tubules (ST) and severe necrosis of spermatocytes (NVP2).
Discussion
NVP is known to improve the metabolic complications in HIV-infected patients. 2,28 This study demonstrates that NVP produced a dose-dependent elevation of lipid peroxidation products in tissues of animals with concomitant reduction in antioxidant status. At double therapeutic dose (NVP2), the drug caused severe toxicities in liver and testis of the animals evidenced by abnormal values of both biochemical and histopathological parameters. It has been reported that NNRTIs induce more liver toxicity than other antiretrovirals, for example, up to 10–20% of HIV patients treated with NVP and efavirenz exhibit increases in liver enzymes that sometimes require discontinuation of therapy. 29,30 Furthermore, the risk of hepatotoxicity with NNRTIs is much greater when HIV coexists with hepatitis B and or C infection, 31,32 both of which are characterized by increases in mediators known to undermine metabolic dysfunctions. 33 In the present study, we can speculate that lower dose of NVP induces levels of dysfunction below those necessary to generate direct liver damage, whereas higher dose stimulates further metabolic stress to a point that it becomes evident as seen in the elevation of serum ALT, AST, GGT and total bilirubin of animals given NVP2. AST and ALT are the reliable makers for liver function. It is known that AST can be found in the liver, cardiac muscle, kidney, brain, pancreas, lungs, leukocytes and erythrocytes, whereas ALT is predominantly available in the liver. 34 The increased levels of these enzymes (AST and ALT) indicate an increased permeability and damage and/or necrosis of hepatocytes by NVP2. 35 The membrane-bound enzyme (GGT) is released into bloodstream during pathological phenomenon or chemical assault. 36 Therefore, the observed increase in these biochemical markers during NVP2 administration suggests that the drug caused severe damage to hepatocytes of treated animals, thereby causing these enzymes to leak into the serum.
Of the two concentrations administered, only 18 mg/kg body weight is within the usual therapeutic dosage, but given the high rate of interindividual variability in the pharmacokinetics of NVP and for better understanding of the toxicity of this drug, we believe that the higher dose employed in our experiments is also relevant. In this study, NVP at both doses induced oxidative stress in the tissues of the animals evidenced by the elevation of MDA levels. This observation is consistent with the findings of Hulgan et al., 37 which noticed an increase in the plasma F2-isoprostane concentrations in HIV patients receiving efavirenz. F2-isoprostanes are prostaglandin-like compounds produced by peroxidation of cell membrane lipids and are accurate and reliable markers of in vivo lipid peroxidation and systemic oxidative stress in humans. 38 Factors that could potentially contribute to oxidative stress during prolonged ART include ongoing HIV replication or persistent exposure to medications that increase stress. 39 Oxidative stress during HIV infection has been linked to changes in plasma and/or cellular markers such as glutathione, cystine, zinc and selenium. 40 The present study confirmed that higher dose of NVP caused severe depletion of tissues glutathione and loss of activities of antioxidant enzymes, especially SOD and CAT. GSH is a naturally occurring antioxidant that prevents free radical damage and helps detoxification by conjugating with endogenous molecules within the cells. Under oxidative stress, GSH is consumed by GSH-related enzymes to detoxify peroxides produced by increased lipid peroxidation, which is caused by chemical assaults. 41,42 In this study, the observed increase in lipid peroxidation and a concomitant depletion in the levels of GSH suggest that the metabolism of NVP at higher concentration induces oxidative stress with consequential depletion of GSH. Furthermore, the activities of tissue SOD and CAT were significantly decreased in NVP2-treated rats. The observed decrease in SOD activity suggests inactivation (or overutilization) of the enzyme probably caused by increased superoxide radical production, or an inhibition by the H2O2 as a result of corresponding decrease in the activity of CAT in NVP2 metabolism. The reduction in activity of CAT may reflect an inability of tissues to eliminate H2O2 produced by NVP2. This may be due to overutilization of the enzyme caused by excess reactive oxygen species. In addition, our current data showed that NVP2 produced no detrimental effect on total sperm quantity but caused significant reduction in sperm motility and viability with a discernable increase in sperm abnormalities in the rats. Testicular lipid peroxidation process induced by NVP may destroy the structure of lipid matrix in membranes of spermatozoa and may lead to rapid loss of intracellular ATP resulting to axonemal damage, decreased sperm viability and increased mid-piece morphological defects, and even inhibits spermatogenesis in extreme cases. 43 Histological examination revealed that the testicular architecture of rats treated with NVP2 was significantly disrupted as shown by marked degeneration of ST and necrosis of spermatocytes. This observation is the first report to show that NVP at higher concentration may disrupt sperm parameters in animals. This is open for further investigation in other animal models. Furthermore, NVP did not alter the levels of serum creatinine and urea of the animals. These indices are sensitive and reliable biochemical markers for evaluation of renal functions in animal model. 44 Increased serum urea and creatinine levels indicate impairment to kidney functions such as acute glomerulonephritis, nephrosclerosis and even tubular necrosis. 45 Therefore, NVP at the two doses administered did not interfere with the renal functions in the animals. This observation was supported by results from the histopathological slides from kidneys of the animals.
In conclusion, increased oxidative stress and liver and testis toxicities observed in animals treated with NVP2 may have important health implications for HIV-infected persons who are on NVP therapy. Future studies should define whether lipid peroxidation may be involved in the adverse effects of ART and its long-term clinical implications. The potential utility of antioxidant therapies during ART should also be considered.
Footnotes
Acknowledgements
The authors thankfully acknowledge the technical assistance of Messers Omoko Ejiro and Chuks Omoruyi of the Department of Veterinary Reproduction and Institute of Child Health, University of Ibadan, Nigeria, respectively.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of Conflict Interest
The authors declared no conflicts of interest.
