Abstract
In this pilot study, we examined the validity and usefulness of hair nicotine–cotinine evaluation as a biomarker of monitoring exposure to tobacco. Head hair samples were collected from 22 infants (<2 years of age) and 44 adults with different exposures to tobacco (through either active or passive smoking) and analyzed by liquid chromatography–mass spectrometry (LC-MS) for nicotine and cotinine. Hair samples were divided into three groups, infants, passive smoker adults and active smoker adults, and into eight subgroups according to the degree of exposure. The limit of quantification (LOQ) was 0.1 ng/mg for nicotine and 0.05 ng/mg for cotinine. Mean recovery was 69.15% for nicotine and 72.08% for cotinine. The within- and between-day precision for cotinine and nicotine was calculated at different concentrations. Moreover, hair nicotine and cotinine concentrations were highly correlated among adult active smokers (R 2 = 0.710, p < 0.001), among adult nonsmokers exposed to secondhand smoke (SHS; R 2 = 0.729, p < 0.001) and among infants (R 2 = 0.538, p = 0.01). Among the infants exposed to SHS from both parents the noted correlations were even stronger (R 2 = 0.835, p = 0.02). The above results identify the use of hair samples as an effective method for assessing exposure to tobacco, with a high association between nicotine and cotinine especially among infants heavily exposed to SHS.
Introduction
Exposure to secondhand smoke (SHS) is a significant threat to global health. 1 A mass of evidence from research studies, meta-analyses and health reports have directly indicated the associations of causality between exposure to SHS and cardiovascular disease, cancer, respiratory illness and a plethora of other causes of death and disability. 1–3 Cigarette smoking emits a number of toxins, including carbon monoxide, ammonia, formaldehyde, nicotine, hydrogen cyanide and also a plethora of potent carcinogens. When tobacco smoke constituents reach the small airways and alveoli of the lung, it is rapidly absorbed and transferred through the blood stream to the rest of the body where it is extensively distributed to body tissues. 4 In regard to nicotine, in most mammalian species, it is metabolized by the liver into 6 primary metabolites, of which in humans 70–80% is converted into cotinine. 4,5 This transformation is performed within two steps. The first is mediated by a cytochrome P450 system (mainly CYP2A6 and CYP2B6) and leads to the production of the nicotine iminium ion, while the second step is catalyzed by aldehyde oxidase (AOX). Another primary metabolite of nicotine is nicotine N′-oxide (NNO), although only about 4–7% of nicotine absorbed is metabolized via this route. Nicotine and its main metabolite, cotinine, can be detected in a substantial number of biological matrixes such as in human blood, hair, urine, saliva, nasal fluid, amniotic fluid and in breast milk. 4,6 –12
Although questionnaire data are routinely used in providing an indicator of the extent of SHS exposure, within the context of dose–response studies, accurately quantified exposure is an essential component of being able to attribute cause and effect.
The aim of this pilot study was not only to resolve issues that have arisen from previous research but more to assess the exposure to SHS among infants and adults using hair analysis. Taking the above into account, within this pilot study, we present the validity of the toxicological assessment of nicotine and cotinine in human hair as a marker of exposure to SHS in both infants and adults within a population-based study design in Greece.
Methods
Head hair sample collection
Sixty-six hair samples were collected from participants in Crete and in Athens, Greece, following written informed consent and institutional review board approval. From each subject, 10–100 mg of hair was collected from the occipital region of the scalp. The hair was cut at the scalp, and the full length was collected. Samples were divided into three groups. Twenty-two were from infants (below 2 years of age) with either no, one or two smoking parents (three subgroups). Parents were asked to answer a questionnaire regarding the number of parents that smoke in the household, as a proxy of home SHS exposure and were categorized as such. Nineteen hair samples were from nonsmoking adults not routinely exposed to SHS (no home/work exposure to SHS) according to their own self-report and divided in two subgroups (rare and frequent SHS exposure). Furthermore, 25 samples were collected from active smokers and divided into 3 subgroups according to the number of cigarettes smoked per day (<10 cigarettes/day = light smokers, 10–20 cigarettes/day = medium smokers and >20 cigarettes/day = heavy smokers).
Reagents
Hexane (97%), (–)-nicotine (99%) and (–)-cotinine (98%) were obtained from Sigma (Sigma-Aldrich Co., St Louis, MO, USA). Dichloromethane (99.5%), sodium hydroxide (99%) and hydrochloric acid (37%) were obtained from Merck (Merck, D-6100 Darmstadt, FR Germany) and methanol (analytical grade) from Scharlau (Scharlau hemie SA La Jota, Barcelona, Spain).
Nicotine and cotinine extraction from hair samples
Hair treatment was based on a previous published method with some minor modifications. 7,8 Hair samples were washed twice with water, hexane and dichloromethane to remove any external contamination and dried in an oven at a temperature of 50°C. Subsequently, 10 μl of an internal standard (10 ppm, ketamine) and 2 ml NaOH (1 M) were added to the hair and digested for 90 min at 60°C. After cooling at room temperature, hair samples were extracted mechanically twice with 3 ml of dichloromethane for 15 min. The organic phases were separated, combined and 50 μl of HCl (1 M) were added to adjust the pH (pH = 2) and to prevent nicotine loss, evaporated to dryness by N2 at 30°C, dissolved in 100 μl methanol and analyzed by liquid chromatography–mass spectrometry (LC-MS).
LC-MS analysis
All analyses were performed using a LC-MS (Shimadzu LCMS—2010 EV) system equipped with an atmospheric pressure chemical ionization (APCI) interface, an autosampler, solvent degasser, binary pump and a heated/cooled column compartment. The column was a Discovery C18 HPLC Column (25 cm × 4.6 mm, 5 μm, Supelco [Bellefonte, PA, USA]). Both MS and high-performance liquid chromatography (HPLC) inlet were controlled by Shimadzu LCMS solution software (LCMS Solution version 3) which also was used for data acquisition and processing. The instrument was tuned and calibrated using autotune procedures recommended by the manufacturer. Interface, curved desolvation line (CDL) and heat block temperatures were 400, 250 and 200°C, respectively. The detector voltage was 1.5 kV and the nebulizing gas flow was 2.5 L/min. The scan range was routinely from 50 to 500 m/z.
Thirty microliters from each extracted sample were added to the chromatographic column at a temperature of 45°C. A gradient of 50 mM ammonium acetate, pH = 5.25 (solvent A) and an acetonitrile gradient (solvent B) were selected for routine use: starting at 5% of solvent B, 95% B (20 min linear ramp), 95% B (3 min) and 5% B (7 min). The total flow rate of mobile phase was 0.6 ml/min. The detection was done by selected ion monitoring (SIM) mode using ion fragments with m/z 163, 204 for nicotine, m/z 177, 218 for cotinine and m/z 238, 279 for ketamine. The fragments which were used for quantification were m/z 163, 218 and 238 for nicotine, cotinine and ketamine, respectively.
Statistical analysis
The descriptive data are presented as mean ± standard deviation for continuous variables while analysis of variance (ANOVA) tests were used for the comparisons. Correlations between hair nicotine and cotinine values were performed and the R 2 values were noted. All reported analyses are based on two-sided tests, while statistical significance was noted as p < 0.05. The statistical package SPSS 18.0 was used to perform the analysis.
Results
Method validation
Linearity
The linear calibration model was checked using blank hair samples spiked with standard solutions at seven concentration levels for each analyte. Hair samples from five infants that had no reported exposure to SHS were combined and used as a blank. Prior to merging, each of these samples were individually analyzed for the presence of nicotine and cotinine and then combined. The final combined head hair sample was analyzed and provided a nondetectable level of nicotine and cotinine (below the limit of quantification [LOQ]). Our results indicated a good linearity in the concentration range between 0 and 5 ng/mg of hair. The linearity of the spiked curve is 0.9974 for nicotine and 0.9975 for cotinine (Table 1).
Analytical parameters for the performed nicotine and cotinine analysis using head hair samples
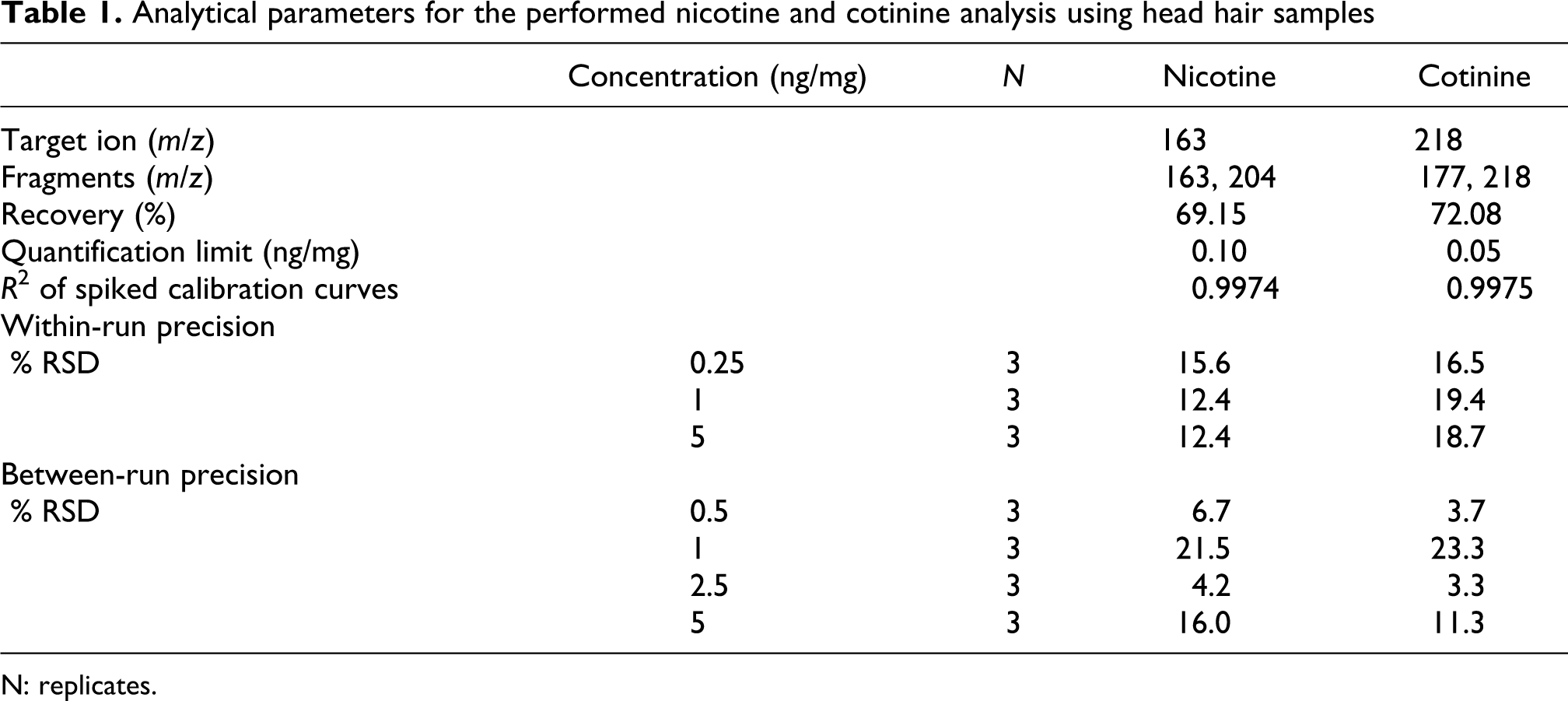
N: replicates.
Sensitivity limits
The LOQ and limit of detection (LOD) were determined by injecting decreasing concentrations of spiked solutions of nicotine and cotinine. We defined as a quantification and detection limit the peaks that gave a signal to noise ratio of 10 and 3, respectively. The LOQ and LOD were calculated as 0.1 and 0.02 ng/mg for nicotine and 0.05 and 0.015 ng/mg for cotinine, respectively (Table 1).
Recovery and precision
The recovery value was calculated using 6 hair samples (blanks), 100 mg each spiked with nicotine and cotinine in concentrations between 0.1 and 5 ng/mg. After elaboration, samples were measured twice. The recovery was defined by comparing the ratio between the peak of the substance and the peak of ketamine (IS) of spiked samples to the ratio of standard solutions. The recoveries were estimated at 69.15% for nicotine and at 72.08% for cotinine (Table 1).
The within- and between-day precision were evaluated by extracting and analyzing three replicates of spiked hair samples at three and four different concentrations, respectively. Within precision was determined on the mean of three replicates for each concentration. The percentage relative standard deviation (RSD) was calculated for within-day precision at 15.6%, 12.4% and 12.4% at concentrations of 0.25, 1 and 5 ng/mg, respectively, for nicotine and 16.5%, 19.4% and 18.7% for cotinine at the same concentrations. Regarding the between-day precision, the percentage RSD were 6.7%, 21.5%, 4.2% and 16.0% for nicotine and 3.7%, 23.3%, 3.3% and 11.3% for cotinine at concentrations of 0.5, 1.0, 2.5 and 5 ng/mg, respectively (Table 1).
Biomonitoring
Infants
As depicted in Table 2, among infants, the mean concentration of nicotine for head hair samples was 6.72 ± 6.72 ng/mg with a range from 0.18 to 28.71 ng/mg. The corresponding values of cotinine were 0.74 ± 0.42 ng/mg with a range between 0.13 and 1.57 ng/mg. Among these infant hair samples, cotinine and nicotine concentrations were found to be higher among those with both parents smokers (11.98 ± 8.71 ng/mg) in comparison to both infants with either one parent a smoker (5.52 ± 3.72 ng/mg) and infants nonexposed to SHS (0.78 ± 0.72 ng/mg), with this noted difference statistically significant, p = 0.012. Hair cotinine concentrations were also found to differ between the three groups assessed albeit not to a statistically significant level (p = 0.221). Moreover, the distribution of nicotine and cotinine levels among infants by their extent of exposure to SHS is depicted in Figure 1.

Hair nicotine and cotinine concentrations (ng/mg) in infants, by parental smoking status.
Mean concentrations, standard deviations and value ranges of hair samples of the pilot study participants

a ANOVA, based on two-sided tests, p < 0.05 flagged as statistically significant.
Adults
Among the adult participants, active smoking was a strong determinant of hair nicotine and cotinine levels. Among these participants, the mean concentration of nicotine was 27.97 ± 22.32 ng/mg (with a range of 2.01–79.30 ng/mg) and of cotinine was 1.16 ± 0.75 ng/mg (with a range between 0.08 and 2.49 ng/mg). Furthermore, a dose–response relationship was revealed between the number of cigarettes smoked per day and hair nicotine (p < 0.001) and hair cotinine levels (p = 0.002), with the biomarker indexes found to increase with the number of cigarettes smoked per day as depicted also in Table 1. Specifically, hair nicotine concentrations were measured at 3.70 ± 1.68 ng/mg, 11.50 ± 3.79 ng/mg and 42.58 ± 19.46 ng/mg for light, moderate and heavy smokers, respectively (p < 0.001), with hair cotinine concentrations found to follow a similar pattern.
Among the nonsmoking adults, the mean concentration of hair nicotine was 1.49 ± 0.68 ng/mg with a range between 0.12 and 2.58 ng/mg and of hair cotinine 0.13 ± 0.09 ng/mg with a range between 0.05 and 0.32 ng/mg. Significant differences in hair cotinine and nicotine levels according to the self-reported exposure to SHS were also noted (p < 0.001) and is depicted in Figure 2.

Hair nicotine and cotinine concentrations (ng/mg) among adults according to smoking status and level of exposure to SHS.
Correlation between nicotine and cotinine
Among all three study groups, hair nicotine and cotinine concentrations were highly correlated indicating the usefulness of either one or the other in assessing exposure to SHS or tobacco use. Specifically a Pearson’s correlation of R 2 = 0.710, p < 0.001 was noted among adult active smokers, an R 2 = 0.729, p < 0.001 among adult nonsmokers and an R 2 = 0.538, p = 0.01 among infants. The highest correlation between hair nicotine/cotinine concentrations was noted among infants exposed to SHS from both their parents (R 2 = 0.835, p = 0.02).
Discussion
In this study, head hair samples were collected for the determination and quantification of nicotine and cotinine, markers of exposure to tobacco and a standardized methodological approach was performed. Among both active and passive smokers, hair nicotine and cotinine levels increased with each participant’s extent of exposure (low vs. moderate vs. high active tobacco consumption and low vs. high passive exposure to SHS, respectively), while hair nicotine and cotinine concentrations in all cases correlated well. It is of interest to note that the strongest differences and correlations were noted among infants exposed to household SHS from both parents.
The usefulness of the methodological approach as a means of noninvasively assessing exposure to SHS among infants and nonsmoking adults as also assessing active smoking was identified through this research, with clear intervalidity indicated between tests. As hair is a part of the organism that is not metabolized and cannot be easily affected by external conditions, these parameters make hair compatible for a safe and accurate study on determining and measuring nicotine and cotinine levels in humans and in infants.
As a toxicological approach, a hair analysis has many advantages as a preselection method for the assessment of toxic substances that are harmful to health due to exposure to air, dust, sediment, soil, food and environmental toxins. 13 Hair specimens in general can be collected quicker and easier than blood, urine or other biological samples, as its methodological approach is noninvasive and does not need complicated storage procedures. Moreover, concentrations of most substances in the hair are significantly found in higher concentrations than in the blood or in other tissues. 11,14 In addition, the analysis of urinary nicotine and cotinine in bodily fluids such as serum, urine and saliva (which is commonly assessed in cohort studies) cannot distinguish between recent or more chronic exposure to nicotine as only recent exposure is assessed, conversely through the segmental analysis of hair this distinction can be made. 10,15–17 It has been estimated that each 1 cm of hair represents approximately 1 month’s exposure to tobacco smoke, thus allowing for a month-to-month exposure analysis. 18 On the other hand, while collecting hair may be relatively easy from a technical perspective, it must be noted that sometimes mothers are not willing to consent for testing the hair of their infants for aesthetic or religious reasons and this factor should be taken into account during protocol design. 19 Within our study, the different concentrations of nicotine and its main metabolite, cotinine, among smokers and nonsmokers were clear. A large variation in nicotine and cotinine concentrations was observed in all groups (smoker adults, nonsmoker adults and infants), suggesting that there is a large variation in individual uptake of nicotine to hair. 7 The large variation may be due to many possible causes, including differences in exposure sources, exposure intensity, duration, ventilation, proximity to a smoker, as also smoking habits/intensity among the smoking population. 4 ,20 ,21 It is of interest to also note that among our study participants, light and moderate adult smokers had comparable hair nicotine levels to infants heavily exposed to SHS, an issue which we partially attribute to the diminished nicotine metabolism in very young children, as the half-life of nicotine in neonates is three to four times longer in newborns exposed to SHS than in adults. 22 The fact that infants have higher hair nicotine concentrations than adults exposed to the same number of cigarettes has also been previously documented. 23
Previous research using hair nicotine concentrations has indicated that the association between children’s exposure to SHS at home and hair nicotine concentrations is stronger among younger children and children with longer exposure at home. 24 A plethora of researchers have previously evaluated the exposure of infants to SHS by assessing the burden of nicotine and cotinine in infants’ hair. 10,15,18,21,23,24 For instance, Pichini et al., among 3–36 months old infants (n = 24) with different degree of exposure, none, rare and frequent exposure, identified that the average concentrations of hair nicotine were 1.3 ± 1.7, 6.8 ± 2.1 and 15.4 ± 6.7 ng/mg, respectively, and in a similar dose–response manner with the findings of our study among infants 6–24 months of age. 10 Research using hair nicotine from participants in 31 countries also indicated that hair nicotine concentrations from children exposed to SHS varied in relation to the number of cigarettes smoked in the household. 23 Similarly our classification using the number of parents who smoke at home (none vs. one vs. two) could be regarded as a proxy for the number of cigarettes that the infants are exposed to. In line with this fact, we identified that hair nicotine and cotinine levels during infancy are higher correlated among children exposed through both their parents to SHS, in comparison to those either exposed through SHS from one parent, or those not exposed at all from their parents to SHS. The increased levels of SHS exposure among the infants exposed to SHS indicate the important role of adopting smoke-free households and promoting smoking cessation among parents with young children, for the sake of protecting their child’s health. Although children can be exposed to SHS in public places, we perceive the household to be the most significant source of exposure due to both the duration spent within the home (especially among infants) and the proximity to the smoker. 25 Moreover, the importance of adopting smoke-free legislations is corroborated by the fact that research has indicated that the implementation of a nationwide smoke-free ban may not only reduce childhood exposure to SHS in public places but may also have an effect on the extent a child is exposed to SHS in the home when one parent (namely the father in the above study) is a smoker. 26
While our results are indicative of the relationship between SHS exposure and hair nicotine/cotinine concentrations, we are unable to generalize these findings to other study populations within Greece. However, this was not the aim of this pilot study, which was to assess the feasibility of collecting, analyzing and measuring exposure to tobacco products or SHS using hair as the biological sample. While as noted above, hair nicotine/cotinine concentrations are useful for assessing more chronic exposure to SHS, hair samples have a number of drawbacks. Namely, there is a strong influence of hair pigmentation on nicotine and cotinine binding and uptake, as nicotine and its metabolites bind to melanin and thus darker colored hair could release more nicotine for the same exposure. 27 In the current study we were unable to assess hair color and this limitation should be met in future research.
In conclusion, our pilot findings indicate that assessing hair nicotine/cotinine concentrations as a proxy for SHS exposure is feasible within Greece and has a good capability in differentiating between levels of exposure. Moreover, hair nicotine and cotinine were strongly correlated and increase in a dose–response relationship according to either SHS or active exposure to tobacco smoking indicating hair cotinine/nicotine’s usefulness in monitoring exposure to SHS.
According to the best of our knowledge, this is probably the first study, using hair analysis, to assess the exposure of infants to SHS in Greece and provide data concerning the degree of exposure to SHS in Greek population (infants and passive smokers). This report is a part of large study in which the participation of infants, preschool children, teenagers and adults has been considered.
Our results are in agreement with those that were presented in previous published studies in the literature 5,9,14 and indicate a relationship of the measured nicotine and cotinine concentrations and the degree of secondhand exposure. The current study also provides information about the smoking habits of the Greek parents.
Footnotes
The authors declared no conflicts of interest.
The study was co-funded by grants 2600, 2590 and 2598 of the laboratory of Toxicology, University of Crete. Author CIV is currently supported by a Flight Attendant Medical Research Institute (FAMRI) grant (Clinical Investigator Award 072058).
