Abstract
In the present study, the antioxidative and anticlastogenic effects of curcumin and piperine separately and in combination have been investigated against benzo(a)pyrene (BaP)-mediated toxicity in mice. Male Swiss albino mice were pretreated with curcumin (100 mg kg−1 body weight) and piperine (20 mg kg−1 body weight) separately as well as in combination orally in corn oil for 7 days; and subsequently, after 2 h of pretreatment, BaP was administered orally in corn oil (125 mg kg−1 body weight). A single dose of BaP in normal mice increased the levels of lipid peroxidation (LPO), protein carbonyl content (PCC), and frequency of bone marrow micronucleated polychromatic erythrocytes (MNPCEs) but decreased significantly the levels of endogenous antioxidants such as superoxide dismutases (SODs), glutathione peroxidase (GPx), glutathione reductase (GR), catalase (CAT) and reduced glutathione (GSH) in the liver. Pretreatments with curcumin and curcumin plus piperine before administration of single dose of BaP significantly decreased the levels of LPO, PCC, and incidence of MNPCEs but elevated the level of GSH and enzyme activities of GPx, GR, SOD, CAT, and glutathione-S-transferase (GST) when compared to the BaP-treated group. The effect of curcumin plus piperine is more pronounced as compared to curcumin in attenuating BaP-induced oxidative insult and clastogenicity.
Introduction
Human beings are exposed to a multitude of xenobiotics in their environment, and majority of the cancers are considered induced by chemical carcinogens. 1 Polycyclic aromatic hydrocarbons (PAHs) are environmental carcinogens present in the atmosphere from the combustion sources such as diesel exhaust, cigarette smoke, residential burning of organic material, refuse burning, industrial coke production, volcanic eruption, and oil contamination by effluents and oil spills. 2 Benzo(a)pyrene (BaP) a potent pro-carcinogen is the prototypical and best characterized member of PAHs family of chemical carcinogens. 3 The food chain is considered as a dominant pathway for routine human exposure and accounts for 97% of the total daily uptake of BaP. 4
BaP is extensively metabolized by cytochrome P450 1A1 (CYP1A1); the microsomal epoxide hydrolase and glutathione S-transferase α (α-GST) detoxify a variety of metabolites that are responsible for initiating tumorigenesis. 5 A large proportion of the metabolic profile of BaP includes the BaP quinones (BaPQs) that are produced biologically by cytochrome P450 isozymes and peroxidases, as well as environmentally through UV light. 6 BaPQs are highly chemically active compounds that have been demonstrated in chemical systems to undergo one electron redox cycling with their semiquinone radicals resulting in the formation of superoxide anion (O2 –), hydrogen peroxide (H2O2), and hydroxyl radical (OH–), collectively called reactive oxygen species (ROS). 7
BaP is a known genotoxic carcinogen and causes DNA damage by both direct and indirect mechanisms. 8 The indirect mechanism involves induction of pro-oxidant status causing lipid peroxidation (LPO) through generation of peroxides/reactive oxygen species (ROS), which act as clastogenic factors (low-molecular-weight components that break chromosomes at the same or remote tissues). These lipid peroxides and active oxygen species mostly act as secondary agents that produce secondary DNA damage in biochemical reactions. 9 An increase in ROS is associated with abnormal cancer cell growth and reflects a disruption of redox homeostasis due to either an elevation of ROS production or a decline in the ROS-scavenging capacity, a condition known as oxidative stress. 10 Therefore, maintaining ROS homeostasis is crucial for normal growth and survival. The cells control the ROS levels by balancing ROS generation with their elimination by ROS-scavenging systems such as antioxidant enzymes (superoxide dismutases [SODs], glutathione peroxidase [GPx], glutathione reductase [GR], catalase [CAT]) and reduced glutathione (GSH). 11
BaP is an omnipresent environmental pollutant and is believed to be a risk factor for human chemical carcinogenesis. So, it is important to identify the naturally occurring/synthetic agents that could modulate the molecular events leading to BaP-induced tumorigenesis. Recent progress in cancer biology is directed toward developing strategies in chemoprevention. 12 Curcumin derived from the rhizome of the herb Curcuma longa has been shown to exhibit antioxidant and anticarcinogenic properties. But the cancer chemopreventive property of curcumin cannot be exploited to full potential because of its poor bioavailability and rapid metabolism in the liver and intestinal wall. So, to increase the bioavailability of curcumin different adjuvants are being tried, which can block metabolic pathways of curcumin. 13 It has been reported that coadministration of piperine and curcumin to humans and rats enhanced the bioavailability of curcumin by 2000% and 154%, respectively. 14 Keeping the above information in mind, the present study was undertaken to appraise antioxidative and anticlastogenic effect of curcumin alone and with piperine (adjuvant) against BaP-mediated toxicity in Swiss albino mice.
Materials and methods
Chemicals
BaP, curcumin, piperine, 1-chloro-2,4-dinitrobenzene (CDNB), 5,5-dithiobis-2-nitrobenzoic acid (DTNB), reduced glutathione (GSH), oxidized glutathione (GSSG), nitro blue tertazolium (NBT), bovine serum albumin (BSA), thiobarbituric acid (TBA), reduced nicotinamide adenine dinucleotide (NADH) and reduced nicotinamide adenine dinucleotide phosphate (NADPH) were obtained from Sigma Aldrich company (USA). All other reagents used were of analytical grade and were obtained either from Merck (India), SRL (India) or from Hi Media (India) India Ltd.
Animals
Male Swiss albino mice, 6–8 weeks old, weighing 26 ± 2 g were procured from the National Institute of Pharmaceutical Education and Research, Mohali, Punjab, India, and maintained in departmental animal house. They were randomly distributed into eight groups, housed in cages (six per cage) and maintained under standard conditions of 22 ± 2°C, 45 ± 10% relative humidity, and 12-h light/12-h dark cycles each day. The animals were housed in polypropylene cages bedded with rice husk and provided ad libitum access to clean drinking water and standard animal pellet diet (Ashirwad Industries, Ropar, Punjab, India) throughout the experiment. Experiments were carried out according to the guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), India, and approved by the institutional animal ethical committee (Approval No. CAH/420 dated April 22, 2009).
Experimental design
Mice were pretreated with curcumin (100 mg kg−1 body weight) or with piperine (20 mg kg−1 body weight) or with curcumin plus piperine dissolved in corn oil for 7 days through oral intubation gavage. Normal mice and mice pretreated with curcumin and piperine separately and in combination received BaP (125 mg kg−1 body weight) dissolved in corn oil (10 ml kg−1 body weight) orally after 2 h of pretreatments. 14-16
Eight treatment groups of six animals each were maintained as described below:
Group I: normal control administered with corn oil.
Group II: curcumin treated.
Group III: piperine treated.
Group IV: curcumin plus piperine treated.
Group V: BaP treated.
Group VI: curcumin plus BaP treated.
Group VII: piperine plus BaP treated.
Group VIII: curcumin plus piperine plus BaP treated.
All the animals were anesthetized and killed by cervical dislocation on the 8th day, 24 h after the last dose of BaP Their livers were removed and perfused immediately with ice-cold saline (0.9% sodium chloride [NaCl] solution). The tissues were then homogenized in 100 mM potassium phosphate buffer (pH 7.4) containing 150 mM potassium chloride (KCl) to obtain 10% homogenate (weight/volume [w/v]). The homogenates were subjected to cold centrifugation at 10,000g for 30 min. The pellets were discarded and the supernatants obtained were used for various biochemical estimations.
Biochemical analysis
LPO level in the liver was measured as thiobarbituric acid reactive substances (TBARS) by using the method of Beuge and Aust. 17 The protein carbonyl content (PCC) was determined spectrophotometrically using dinitrophenylhydrazine (DNPH)-labeling procedure as described by Levine et al. 18 The GSH was estimated by the method of Moron et al. 19 GR activity was determined by the method of Carlberg and Mannervik. 20 GPx activity was assayed by the method of Mohandas et al. 21 GST assay was done according to the method described by Habig et al. using CDNB as a substrate. 22 The method of Luck was used for the estimation of CAT. 23 The activity of SOD was estimated by following the method of Kono. 24 The protein content of the tissue homogenates mentioned earlier was determined by following the method described by Lowry et al. using BSA as a standard. 25 To perform the micronucleus test, bone marrows were flushed out of the femur bone with fetal calf serum into the centrifuge tubes. The cells were dispersed by gentle pipetting and collected by centrifugation at 150g for 5 min at 4°C. Acridine orange staining method of Hayashi et al. was used for the analysis. 26 A volume of 10 μl of 1 mg/ml acridine orange aqueous solution was spread homogeneously on a warmed glass slide. A volume of 5 µl of bone marrow cell suspension was placed on the center of an acridine orange-coated slide and covered immediately with a cover slip. Supravitally stained cells were examined by fluorescence microscope and the frequency of micronucleated polychromatic erythrocytes (MNPCEs) was obtained by observation of 2000 polychromatic erythrocytes (PCEs) per slide in randomly chosen fields of vision (magnification ×400). Immature erythrocytes, that is, PCEs were identified by their orange-red color, mature erythrocytes by their green color, and the micronuclei by their yellowish color. To evaluate cytotoxicity, 200 cells (PCE and normochromatic erythrocyte [NCE]) were scored.
Statistical analysis
The data were expressed as mean ± SD for six animals in each group. The statistical comparisons were performed by one-way analysis of variance (ANOVA) followed by Tukey’s honesty significant difference test. The results were considered statistically significant if the p values were equal to 0.05 or less.
Results
The pretreatment effect of curcumin, piperine, and curcumin plus piperine on BaP-induced hepatic TBARS, protein carbonyl, GSH levels and bone marrow micronuclei are given in Figures 1, 2, 3 and 4, respectively. TBARS and protein carbonyl levels were significantly elevated and increased frequency of micronuclei was observed in BaP-treated animals as compared to normal animals. Administration of curcumin and curcumin plus piperine with BaP challenge significantly lowered the elevated levels of TBARS, protein carbonyl and reduced the frequency of micronuclei formation in the bone marrow. The concentration of GSH was significantly decreased in the BaP-treated group when compared with normal group, which was significantly elevated following pretreatments with curcumin and curcumin plus piperine. Piperine pretreatment followed by BaP treatment did not alter the levels of TBARS, protein carbonyl, and GSH in liver tissue as compared to the BaP-treated group. There was no statistically significant difference in the ratio of PCEs to the total erythrocytes among the different groups (Table 2).
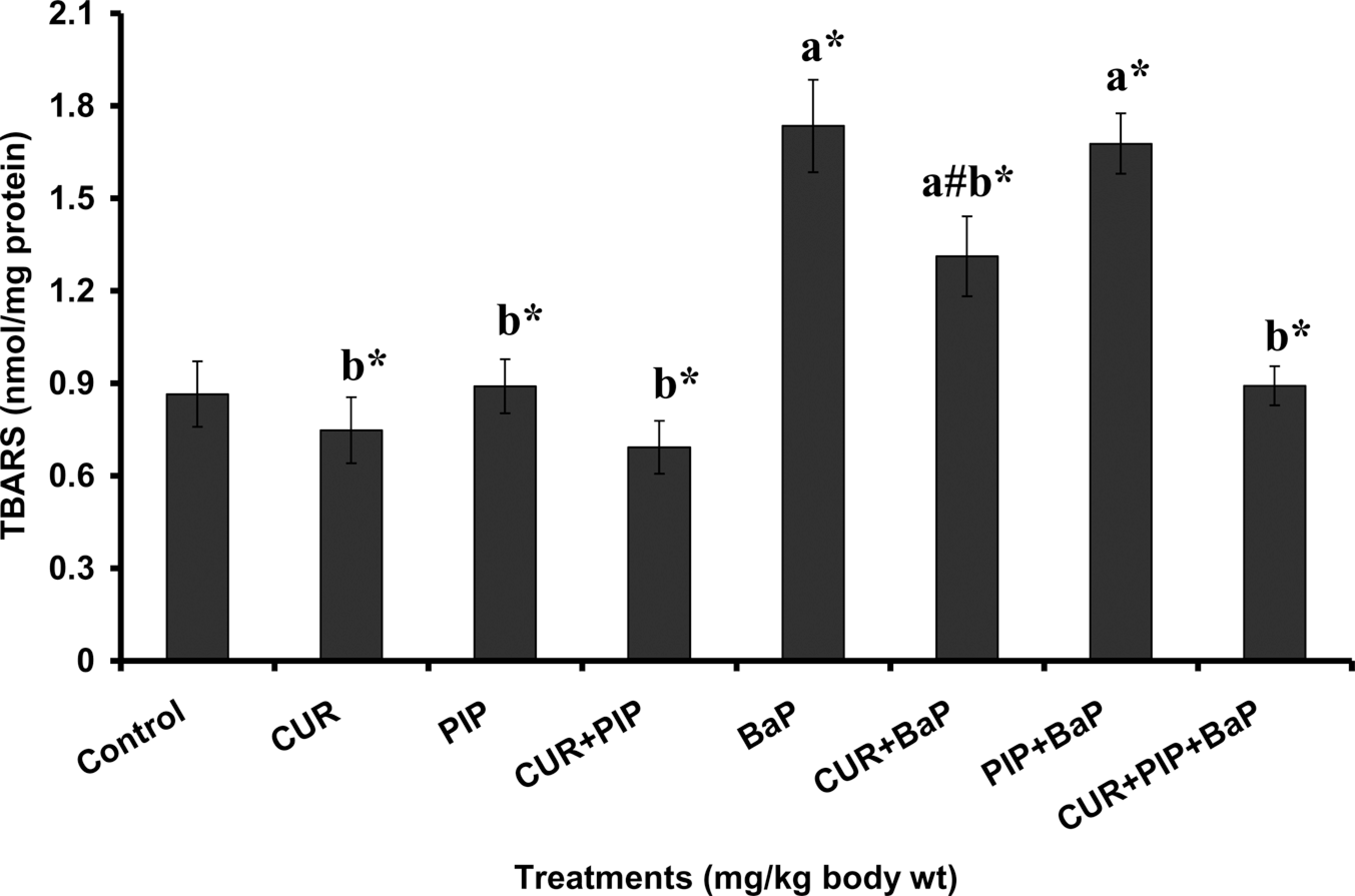
Effect of pretreatment with curcumin and piperine separately and in combination on liver thiobarbituric acid reactive substance (TBARS) level in mice treated with BaP. Values are expressed as mean ± SD; n = 6. CUR: curcumin (100 mg kg−1), PIP: piperine (20 mg kg−1), and BaP: benzo(a)pyrene (125 mg kg−1). (a) As compared with normal control group and (b) as compared with BaP group. Statistical significance: # p < 0.05, *p < 0.01.
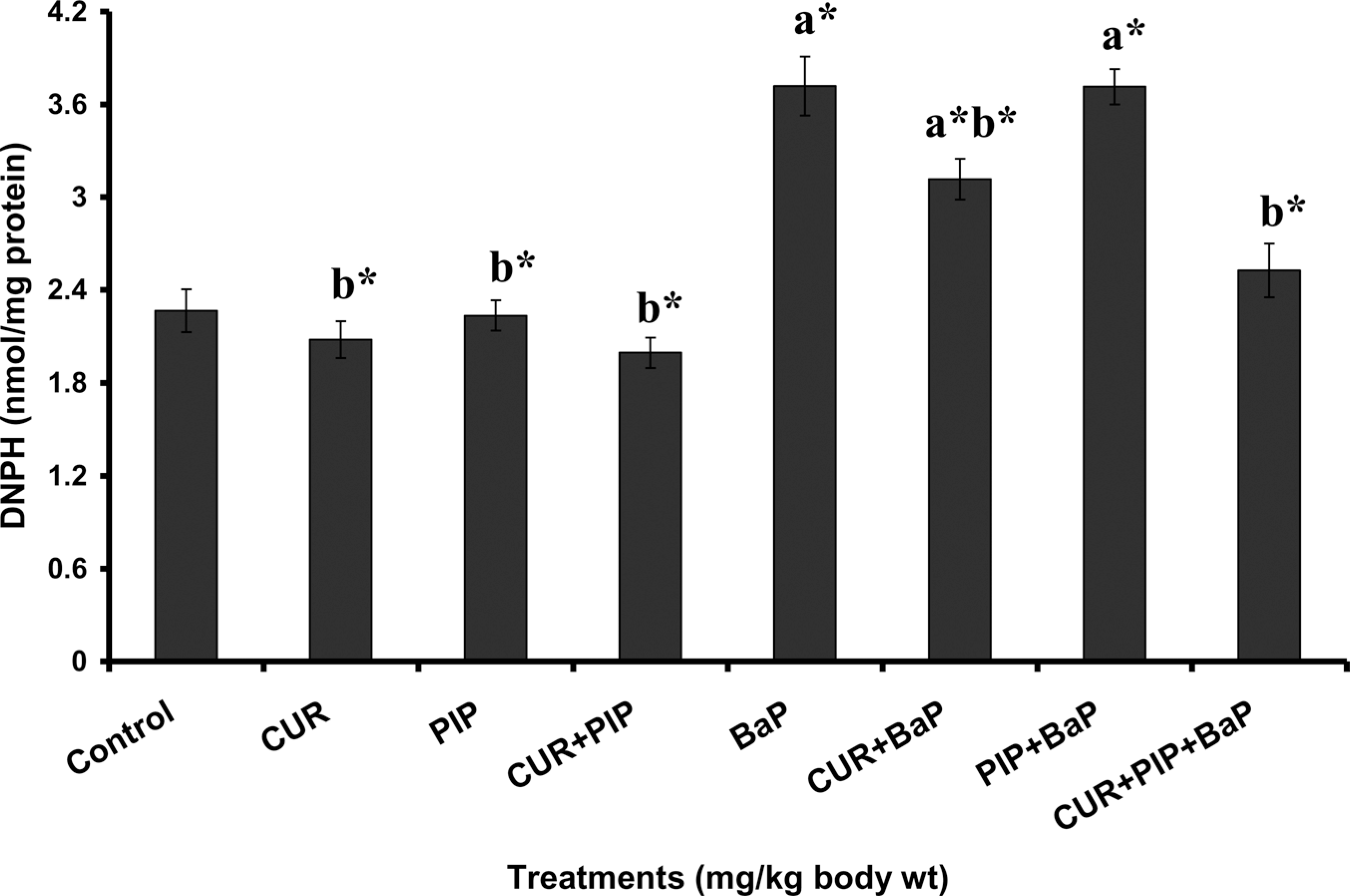
Effect of pretreatment with curcumin and piperine separately and in combination on liver protein carbonyl content in mice treated with BaP. Values are expressed as mean ± SD; n = 6. CUR: curcumin (100 mg kg−1), PIP: piperine (20 mg kg−1), BaP: benzo(a)pyrene (125 mg kg−1), and DNPH: dinitrophenylhydrazine. (a) As compared with normal control group and (b) as compared with BaP group. Statistical significance: *p < 0.01.
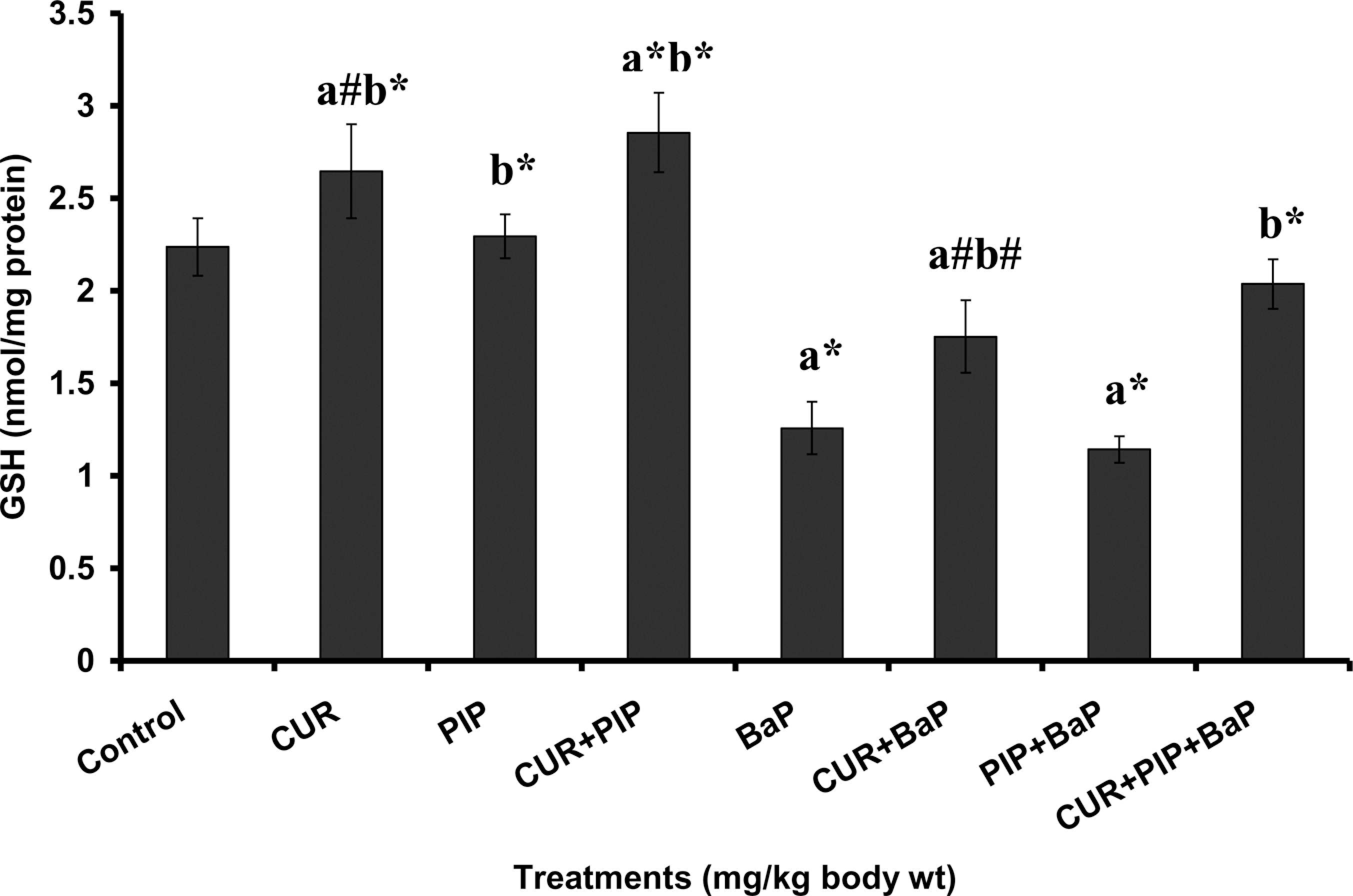
Effect of pretreatment with curcumin and piperine separately and in combination on reduced glutathione (GSH) level in the liver of mice treated with BaP. Values are expressed as mean ± SD; n = 6. CUR: curcumin (100 mg kg−1); PIP: piperine (20 mg kg−1); BaP: benzo(a)pyrene (125 mg kg−1). (a) As compared with normal control group and (b) as compared with BaP group. Statistical significance: # p < 0.05, *p < 0.01.
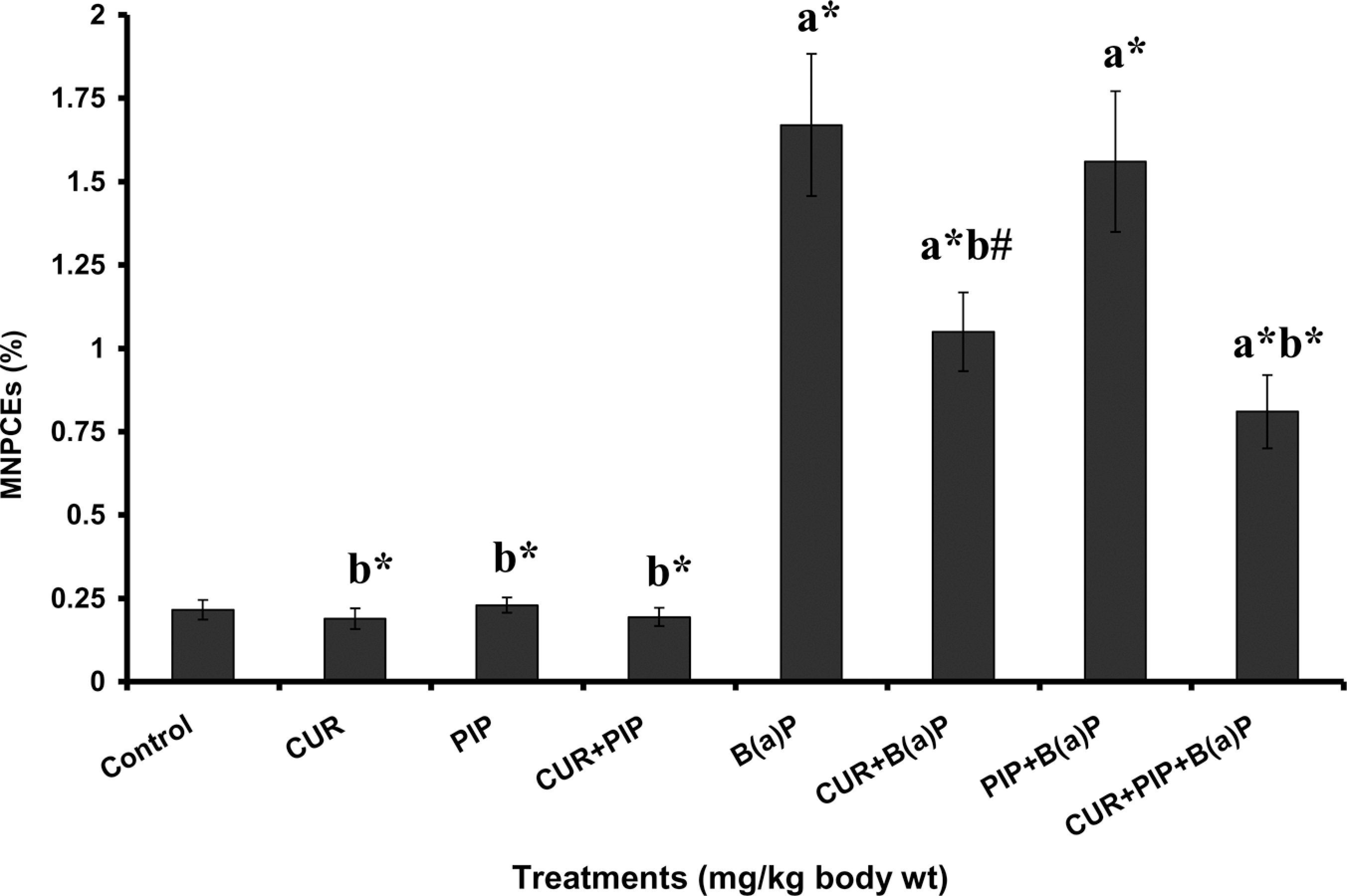
Effect of pretreatment with curcumin and piperine separately and in combination on benzo(a)pyrene (BaP)-induced bone marrow micronucleated polychromatic erythrocytes (MNPCEs). (a) As compared with normal control group and (b) as compared with BaP group. Statistical significance: *p < 0.01.
Effect of curcumin and piperine pretreatment separately and in combination on BaP-induced bone marrow micronucleated polychromatic erythrocytes (MNPCEs) a

CUR: curcumin (100 mg kg−1), PIP: piperine (20 mg kg−1), BaP: benzo(a)pyrene (125 mg kg−1).
a Values are expressed as mean ± SD; n = 6 for each treatment group. Percentage of polychromatic erythrocytes was calculated as follows: (PCEs/PCEs + NCEs) × 100.
b As compared with BaP group.
c As compared with normal control group.
d Statistical significance at p < 0.01.
e Statistical significance at p < 0.05.
Effect of pretreatment with curcumin and piperine separately and in combination on hepatic CAT, SOD, GR, GPx, and GST activities in mice treated with BaP a

CUR: curcumin (100 mg kg−1), PIP: piperine (20 mg kg−1), BaP: benzo(a)pyrene (125 mg kg−1).
a Values are expressed as mean ± SD; n = 6 for each treatment group. CAT is expressed in units μmol H2O2 consumed/min/mg protein, SOD in units/min/mg protein, GR in units nmol NADPH consumed/min/mg protein, GPx in units nmol NADPH consumed/min/mg protein, GST in units nmol CDNB conjugates/min/mg protein.
b As compared with BaP group.
c As compared with normal control group.
d Statistical significance at p < 0.01.
e Statistical significance at p < 0.05.
Table 2 shows the effects of pretreatments with curcumin and piperine separately and in combination on hepatic CAT, SOD, GR, GPx, and GST activities in mice treated with BaP. CAT, SOD, GR, and GPx activities were significantly lowered in the BaP-treated group when compared with the normal control group, but no significant change was assessed in GST activity compared to normal mice. Administration of curcumin and curcumin plus piperine for 7 days before single dose of BaP significantly elevated the activities of GR, GST, SOD, CAT and GPx as compared to the BaP-treated group and restored the endogenous antioxidants near to normal levels following the combined treatment. Pretreatment with piperine followed by BaP exposure did not alter the activities of GR, GST, SOD, CAT, and GPx in liver as compared to BaP-treated group. Combined administration of curcumin plus piperine followed by BaP treatment significantly increased the activities of GR, GST, SOD, CAT, and GPx in liver with respect to BaP- and curcumin-treated mice, and the increase was more pronounced when seen in context with comparison of curcumin plus BaP- with BaP-treated group, thereby indicating that piperine does act as an adjuvant.
No significant difference in the levels of endogenous antioxidants was observed in curcumin, piperine, and curcumin plus piperine pretreated groups except an increase in the levels of GSH and GST in curcumin and curcumin plus piperine pretreated groups.
Discussion
The role of reactive oxygen radicals in the etiology of cancer is supported by epidemiologic studies. Specifically these epidemiologic studies illustrated the protective role of antioxidants against cancer development. 27,28 BaP is one of the PAHs classified as carcinogenic to humans by the International Agency for Research on Cancer. 29 One of its toxic effects is induction of oxidative stress which leads to various diseases such as cancer. BaP metabolism produces ROS in the form of O2 –, OH–, and H2O2. 7 These ROS can react with cellular macromolecules in a variety of ways and produce LPO, protein oxidation, and/or DNA oxidation. 30
Plant products are known to exert their protective effects by scavenging free radicals and modulating carcinogen detoxification and antioxidant defense system. Antioxidant status has been suggested as a useful tool in estimating the risk of oxidative damage induced by chemical carcinogens. In the present study, the antioxidant and anticlastogenic effect of curcumin alone and with adjuvant piperine has been investigated against BaP-mediated toxicity. A single dose of BaP increased the levels of LPO products significantly, which indicate induction of oxidative stress in hepatic tissue. BaP is reported to increase LPO products either by producing the ROS or by decreasing the activities of antioxidant enzymes that lead to cellular damage and cellular dysfunction. 31 LPO is used as a biomarker to show the index of oxidative stress and causes cell membrane damage resulting in gradual loss of cell membrane fluidity, decreased membrane potential, and increased permeability to ions. 32 The level of PCC which is measured as an index of protein oxidation was significantly increased in the liver tissue after treatment with BaP. Proteins are attacked by ROS directly or indirectly through LPO. The protein oxidative damage can result in the modification in structural, enzymatic activity, and signaling pathways. Previous studies have shown that BaP treatment increased LPO and PCC in the liver of mice. 31,33 Pretreatment with curcumin alone, which was followed by BaP treatment, caused a significant decrease in LPO and PCC levels which were further decreased when curcumin was administered along with piperine, thus signifying the adjuvantive effect of piperine. Several studies have shown that curcumin significantly decreased the levels of LPO products. 34,35
Cellular redox potential is largely determined by GSH content, which accounts for >90% of the cellular nonprotein thiols. 36 GSH cellular concentration is 500- to 1000-fold higher than the other redox regulatory proteins and is the most abundant low-molecular-weight thiol in mammalian cells. GSH antioxidant system plays a major role against ROS and other oxidant species. 37 BaP metabolism produces abundant ROS which oxidizes GSH sulfhydryl groups to disulfide compound GSSG (oxidized form) and leads to the depletion of GSH. Oral administration of curcumin alone and with piperine normalized the depleted level of GSH caused by BaP. Studies in cell culture suggest that curcumin can increase cellular GSH levels by enhancing the transcription of the two Gcl genes, that is, Gclc and Gclm for glutamate cysteine ligase, the rate-limiting enzyme in GSH synthesis. 38 The protection mechanism of curcumin could be through its activity as free radical scavenging, particularly against oxygen radical, which would inhibit SH-group oxidation. 35 GSH acts as a cofactor for antioxidant enzymes such as GPx and is involved in reduction of peroxides, including membrane lipid peroxides formed upon oxidative insults. In this study, the activity of GPx was decreased significantly in liver after BaP treatment and pretreatment with curcumin alone and with piperine restored the activity near to normal levels, thus making the cells more effective in detoxification of metabolites. GR is an NADPH-dependant enzyme, which converts GSSG (oxidized form) into GSH (reduced form). Its inhibition is likely to be deleterious to cells since it contributes efficiently in maintaining the basal levels of cellular GSH. GR activity was significantly reduced upon BaP treatment. The increased GR activity as observed in curcumin alone and in combination with piperine plays a significant role in the regulation of GSH-GSSG cycle.
SOD is essentially a protective enzyme which scavenges the superoxide ions produced as cellular by-products during oxidative stress. 39 It catalyzes the transformation of the superoxide radical into H2O2, which can then be transformed by the enzyme CAT into water and molecular oxygen. 40 Its decreased activity can lead to adverse effects because O2 – are extremely toxic and may accumulate in the cells. 41 In the present study, activities of SOD and CAT were decreased significantly in the liver of BaP-treated mice. It is reported that the activities of SOD and CAT in the tissues of rats (liver, kidney, and lung) were decreased after BaP treatment which suggests that BaP oxidatively alters antioxidant enzymes which might be associated with BaP carcinogenesis. 42 The levels of SOD and CAT were normalized by pretreatment with curcumin alone and in synergism with piperine in liver of BaP-treated groups. Further, it was documented that curcumin enhances the activities of antioxidant enzymes such as SOD and CAT in the liver homogenates of rats. 43
GST is a critical detoxification enzyme that functions primarily in conjugating ‘functionalized metabolites’ with endogenous ligands (GSH), favoring their elimination from the body of the organism. 44 As a result, carcinogenic metabolites of BaP derived from cytochrome P450 can be conjugated to GSH and be subsequently eliminated from the system. In our study, BaP treatment does not alter the activity of GST, but pretreatment with curcumin alone and in combination with piperine lead to induction of GST. There is persuasive evidence to support a correlation between the induction of GST and protection against wide spectrum of cytotoxic, mutagenic, and carcinogenic chemicals. 45,46 Enhancement of hepatic and pulmonary GST by dietary curcumin has been shown in previous studies. 47,48 Increasing the phase II biotransformation enzyme activity is generally thought to enhance the elimination of potential carcinogens. Several studies in animals have found that dietary curcumin increased the activity of phase II enzymes, such as GST.47,49
Oral pretreatment with curcumin and curcumin plus piperine significantly reduced the frequency of occurrence of BaP-induced bone marrow micronuclei. The induction of BaP-induced chromosomal aberrations, micronuclei formation, and sister chromatid exchanges (SCEs) were found to be inhibited by curcumin. 15 Moreover, our data demonstrate that the administration of curcumin plus piperine exhibited stronger effect in reducing BaP-induced genotoxicity. It is reported that curcumin and piperine in combination exhibited a potent antigenotoxic effect as compared to either agent alone in 7,12-dimethylbenz(a)anthracene (DMBA)-induced genotoxicity in golden Syrian hamsters. 50 Studies in rodents suggest that curcumin has poor systemic bioavailability when given orally (p.o.) and is cleared rapidly from the plasma when administered intravenously (i.v.). 51 Concomitant administration of curcumin with piperine to BaP-treated mice in the present study showed the overall pronounced antioxidative and anticlastogenic effects as compared to curcumin alone. Piperine a strong inhibitor of hepatic and intestinal aryl hydrocarbon hydroxylation and glucuronidation is known to enhance the bioavailability of a number of therapeutic drugs as well as phytochemicals. 52,53 An enhanced systemic availability of propranolol (40 mg) and theophylline (150 mg) was evidenced in healthy volunteers as a result of coadministration with piperine (20 mg/day). 54 Concomitant administration of curcumin (2 g/kg or 500 mg/kg) with piperine (20 mg/kg) increased the bioavailability of curcumin in rats. 14,55 Piperine enhanced the bioavailability of curcumin by not only reducing the rate of its metabolic breakdown but also enhancing the extent of intestinal absorption by increasing residence time, alteration in membrane lipid dynamics, and change in the conformation of enzymes in the intestine. 56 In another study, it has also been suggested that piperine could enhance curcumin’s effect by enhancing the bioavailability through inhibition of curcumin’s efflux via P glycoprotein (ABCB1 or MDR1) efflux pump. 57 Therefore, the present study concludes the effectiveness of curcumin in alleviating genotoxicity and oxidative stress increases when it is given alongwith piperine which acts as an adjuvant.
Further studies are in progress in our laboratory to explore the chemopreventive effects of curcumin and piperine against BaP-induced oxidative stress and DNA damage by quantitation of 8-oxo-7,8-dihydro-2-deoxyguanosine (8-oxo-dG) content, DNA adducts, and p53 gene expression in hepatic and pulmonary tissues of mice. The study of phytochemicals with chemopreventive effects and understanding their health-related interactions will lead to better use of dietary intervention in the prevention of cancer.
Footnotes
Acknowledgment and Funding
The authors are grateful to Indian Council of Medical Research, India, for providing financial assistance in the form of senior research fellow to A Sehgal.
