Abstract
Introduction
Cyclosporine is a calcineurin inhibitor that is widely used to decrease the incidence of organ transplant rejection and in the management of immunological diseases. However, these effects may be associated with increased incidence of nephrotoxicity. This work aimed to the exploration of the impact of omarigliptin with or without shikonin on a rodent model of cyclosporine nephrotoxicity and the precise determination of the mechanisms that may represent the basis of these effects.
Methods
In a Wistar rat model of cyclosporine-elicited nephrotoxicity, the effect of omarigliptin and shikonin, each alone and in combination, was determined at the level of the biochemical parameters and the histomorphological changes.
Results
Omarigliptin and/or shikonin administered to cyclosporine-injected animals induced a significant restoration of renal functions and glucagon-like peptide-1 (GLP-1) and augmentation of the antioxidant defenses, associated with increased sirtuin 1 expression and its related signaling changes in comparison to animals that received cyclosporine alone. Additionally, omarigliptin and/or shikonin elicited a significant amelioration of the inflammatory response and cellular differentiation and a significant improvement of the renal tissue disruptive changes elicited by cyclosporine. These effects were evident with omarigliptin/shikonin combination when compared to the groups treated with each agent alone.
Conclusion
Omarigliptin/shikonin combination suggests potential therapeutic benefit for the mitigation of cyclosporine nephrotoxicity, possibly via their effects on dipeptidyl peptidase 4 activity and GLP-1 levels with subsequent modulation of the redox status, cellular proliferation, and the inflammatory pathways.
Introduction
Cyclosporine is an immunosuppressive agent that has proven efficacy in decreasing the incidence of organ transplant rejection.1,2 However, cyclosporine has several adverse effects, with nephrotoxicity being the most serious one. 3 The renal-damaging effects of cyclosporine may be attributed to interference with renal blood flow, which may induce oxidative and nitrosative stress, with activation of the inflammatory processes and renal fibrosis. 4 The ongoing research is directed towards the exploration of the molecular mechanisms that underlie these nephrotoxic effects. 5
Sirtuin-1 (SIRT1) is a nicotinamide adenine dinucleotide-dependent deacetylase that is involved in a wide range of cellular processes, including oxidative stress, inflammation, and cellular metabolism. 6 Recent reports highlighted a crucial role for sirtuins in the amelioration of the renal pathogenic effects induced by cyclosporine. 7 SIRT1 enhances the activity of the antioxidant enzymes and combats the harmful effects of reactive oxygen species (ROS). 8 In addition, SIRT1 deacetylates and inhibits the transcriptional factors that drive inflammation in the renal tissues, including high mobility group box 1 (HMGB1), which mediates the renal damage induced by cyclosporine.9–11 Moreover, an important antifibrotic effect for SIRT1 was detected in the renal tissues. 12
Glucagon-like peptide-1 (GLP-1) is a hormone that represents the keystone for glucose homeostasis. 13 Recent studies have emphasized the pleotropic effects of GLP-1 that have shown an outstanding efficacy for the amelioration of chemotherapy-induced renal toxicity.14,15 These effects include abrogation of oxidative stress, inhibition of the release and activity of the pro-inflammatory cytokines, and downregulation of the activity and proliferation of the renal fibroblasts. 16 These effects, in addition to improvement of the renal hemodynamic circulation, may drive attention towards exploration of its effects on cyclosporine-induced nephrotoxicity. 17
Omarigliptin is a dipeptidyl peptidase-4 (DPP-4) inhibitor that has proven efficacy in the management of type 2 diabetes mellitus. 18 Recent research has proven multiple favorable effects of DPP-4 inhibitors that may be of benefit for the mitigation of cyclosporine-elicited nephrotoxicity. These include reduction of renal inflammation, increased activity of the antioxidant enzymes, and diminution of ROS. 19 Moreover, treatment with DPP-4 inhibitors was proven to combat the pathways that facilitate activation of the fibroblasts and extracellular matrix deposition and improve the renal blood flow. 20 Therefore, assessment of the effects of omarigliptin on the renal pathological changes induced by cyclosporine may represent a hot research point.
Shikonin is a natural agent derived from the root of Lithospermum erythrorhizon. 21 Recently, shikonin was proven to possess a wide variety of pharmacological properties that may be beneficial for the different body systems. 22 These include its ability to counteract the different inflammatory mediators, augment the antioxidant defenses, and combat the fibrotic changes. 23 This, in addition to its effect on the renal vasculature, may impart a role for shikonin in the amelioration of cyclosporine-induced nephrotoxicity. 24
To our knowledge, the present study is the first one that examined the effect of omarigliptin with or without shikonin on cyclosporine-elicited nephrotoxicity in rats, via determination of the role of the redox mechanisms, inflammation, GLP-1, SIRT1, and HMGB1 signaling in the pathogenesis of this condition. The use of omarigliptin/shikonin combination in the present study may be attributed to the recent reports that shikonin improves the endothelial functions and promotes angiogenesis in diabetic wounds via modulation of the PI3K/AKT signaling pathway. 25 In addition, the ability of shikonin to reduce inflammation may ameliorate the chronic inflammatory responses associated with hyperglycemia and improve insulin sensitivity. 26 This may originate from its inhibitory effect on protein-tyrosine phosphatase 1B (PTP1B), which is a negative regulator of insulin signaling and is linked to chronic inflammation in diabetic patients. 27 Moreover, shikonin was proven to be an effective inhibitor of P-glycoprotein, 28 an effect that improves absorption and augments the bioavailability of DPP-4 inhibitors, including omarigliptin. 29 Furthermore, shikonin acts on multiple cellular signaling pathways, which may provide broader therapeutic coverage when combined with omarigliptin’s targeted action. This preclinical trial aimed at exploring the impact of omarigliptin with or without shikonin on a rodent model of cyclosporine nephrotoxicity and determination of the molecular mechanisms that may represent the basis of these effects.
Materials and methods
Ethical disclosure
The experimental procedures performed in the present work and the reporting of the animal testing experiments were in accordance with the ARRIVE guidelines for animal experiments. The protocol of the present study was ethically approved by the Research Ethics Committee of the Faculty of Medicine, Tanta University, Tanta, Egypt (Approval code 36264PR1078/2/25). To minimize selection bias and ensure equal distribution of potential confounding variables, a total of 50 male Wistar rats were randomly assigned into five equal experimental groups (n = 10 per group) using a computer-generated randomization sequence. The randomization was carried out using the RAND function in Microsoft Excel, which assigned a random number to each animal. Rats were then sorted based on these random numbers and sequentially allocated to one of the five groups. All individuals involved in animal handling, conducting biomarker analyses, and evaluating the histopathologist results were blinded to group allocation throughout the study to maintain objectivity and reduce observer bias. Animals were monitored daily for signs of distress or discomfort, and humane endpoints (weight loss >20%, lethargy, severe dehydration, respiratory distress, severe renal impairment) were established to minimize suffering. Animals were euthanized early if they exhibited any of these signs to uphold ethical standards and minimize animal suffering.
Chemicals, reagents, and drugs
Thermo Fisher Scientific, Waltham, Massachusetts, United States, was the source of cyclosporine A (99+%, CAS No. 59865-13-3). Omarigliptin powder was purchased from AbMole BioScience, Houston, Texas, United States (Purity >99%, Catalog No. M4936). Tokyo Chemical Industry Co., Tokyo, Japan, was the supplier of shikonin powder (CAS No. 54952-43-1, Product Number: H1780, Purity> 98.0% (HPLC), Molecular Formula/Molecular Weight: C16H16O5 = 288.30, Melting point: 149°C). Both olive oil and methyl cellulose were obtained from Hebei Weibang Biotechnology Co., Shijiazhuang, Hebei, China (Purity >99%, CAS No. 8001-25-0 and 9004-67-5, respectively). Cyclosporine was dissolved in olive oil to reach a final concentration of 20 mg/ml. Each of omarigliptin and shikonin was prepared as a suspension in a 0.5% methyl cellulose solution. El Goumhoria Trade Pharmaceuticals and Chemicals and Medical Supplies Co., Tanta, Egypt, supplied the other reagents and chemicals used in the present study.
Arrangement of the experimental animal groups and the study design
By using an OpenEpi test for the calculation of the appropriate sample size, 50 adult male Wistar rats (10 weeks old, Body weight 160–220 g, purchased from the animal house of the faculty of science, Tanta University, Tanta, Egypt) were used in the present study. These animals were kept in special metabolic wire cages with unrestricted access to food and water ad libitum for an acclimatization period of 2 weeks. They were kept under constant experimental circumstances throughout the entire study period, including a 12 L/12D lightening system, humidity of 50–70%, and a temperature of 23–26°C. Then, the experimental animals were randomly allocated into five equal groups (10 rats/group) as illustrated in Figure 1 as follows: the control group that received daily subcutaneous injection of olive oil starting from the 8th day of the experiments for 21 days concomitantly with daily oral 0.5% methyl cellulose solution by gastric tube from the start of the experiments for 28 days; cyclosporine group in which rats were injected subcutaneously with cyclosporine (20 mg/kg) daily starting from the 8th day of the experiments for 21 days
30
; cyclosporine + omarigliptin group that received omarigliptin (5 mg/kg) daily by gastric tube
31
concomitantly with daily subcutaneous injection of cyclosporine as previously described; cyclosporine + shikonin group that received shikonin (25 mg/kg) daily by gastric tube
32
concomitantly with daily subcutaneous injection of cyclosporine as mentioned above; cyclosporine + omarigliptin/shikonin combination group that received omarigliptin/shikonin combination daily by gastric tube in the above mentioned doses concomitantly with daily subcutaneous injection of cyclosporine as previously described. Both omarigliptin and shikonin were administered daily by gastric tube from the start of the experiments for 4 weeks. A diagrammatic scheme representing the experimental design of the study. Bax: BCL-2-associated X protein; BCL-2: beta cell lymphoma protein 2; CAT: catalase; CsA: cyclosporine; DPP-4: dipeptidyl peptidase-4; GLP-1: glucagon-like peptide-4; HMGB1: high mobility group box 1; HO-1: heme oxygenase-1; IL-1β: interleukin-1 beta; IL-2: interleukin-2; KIM-1: kidney injury molecule-1; MCP-1: monocyte chemoattractant protein-1; MDA: malondialdehyde; MPO: myeloperoxidase; NAG: N-acetyl beta-D-glucosaminidase; NF-κB: nuclear factor kappa B; Nrf2: nuclear factor erythroid 2-related factor 2; P-p38 MAPK: phospho-p38 mitogen-activated protein kinase; P-ERK: phospho-extracellular signal-regulated kinase; P-JNK: phospho-Jun N-terminal Kinase; RAGE: receptors for advanced glycation end products; S.C.: subcutaneous; SIRT1: sirtuin-1; SOD: super-oxide dismutase; TLR4: toll-like receptor 4.
In the current study, the doses of cyclosporine, shikonin, and omarigliptin were determined in view of the Reagan-Shaw equation, which provides a safe, effective, and tolerable doses.33,34 This equation is described as follows: Human equivalent dose (mg/kg) = Animal dose (mg/kg) multiplied by Animal Km/Human Km (Km of rats was six and Km of humans was 37). Also, the doses of cyclosporine, omarigliptin, and shikonin used in the current work were employed in previous animal studies performed by Ateyya et al., 30 Ayoub et al., 31 and Balaha et al., 32 respectively, who reported that these doses were in accordance with the human equivalent dose formula. 33
Harvesting the urine, blood, and renal tissue samples
At the 29th day of the experiments, rats were kept in individual metabolic cages for 24 h of urine collection. The protein content of the collected urine samples was quantified according to the Lowry et al. procedure. 35 The activity of N-acetyl beta-D-glucosaminidase (NAG) was determined in urine samples according to Price. 36 The urinary creatinine concentration was assayed using the Uchida et al. method. 37 Then, the animals were fasted overnight, and the next morning, blood samples were collected from the retro-orbital plexus under thiopental sodium anaesthesia (50 mg/kg, intraperitoneal injection). 38 Sera were separated from the collected blood samples by centrifugation at 1200× g in a cooling centrifuge (R-81 M, REMI Sales & Engineering Ltd, Maharashtra, India) for 15 min. Then, rats were euthanized by cervical dislocation, and the kidneys were harvested via a longitudinal midline abdominal incision and washed with ice-cold saline. Parts of the harvested kidneys were homogenized with 10 mL of radioimmunoprecipitation assay (RIPA) buffer mixed with a cocktail of protease inhibitors in Q800R3 Sonicator (Qsonica L.L.C., Newtown, CT 06470, United States) to yield a 10% w/v homogenate which was then centrifuged in a cooling centrifuge (R-81 M, REMI Sales & Engineering Ltd, Maharashtra, India) at 15,000× g for 10 min. Then, the resulting supernatant represented the working solution in which the tissue biochemical parameters were assayed. The remaining parts of the harvested kidneys were processed for further histological examination and quantification.
Assessment of the renal function tests in the blood and renal tissue samples harvested from the studied groups
The levels of blood urea and serum creatinine were assayed using commercially available kits purchased from Biodiagnostic Co., Giza, Egypt (Catalog No. UR2110 and CR12 50, respectively). Creatinine clearance was measured by applying the Cockroft and Gault formula, in which urinary creatinine is multiplied by the 24-h urinary volume, then divided by serum creatinine multiplied by 1440. 39 Elabscience, Houston, Texas 77079, United States, was the source of kits used for the assessment of kidney injury molecule-1 (KIM-1) in the renal tissues (Catalog No. E-EL-R3019) according to the enclosed supplier’s instructions.
Evaluation of the redox state in the harvested renal tissues
The redox state of the renal tissues was evaluated by measuring the levels of malondialdehyde (MDA), myeloperoxidase (MPO), catalase (CAT), and superoxide dismutase (SOD) in the supernatant of the centrifuged renal tissue homogenate. MDA content of the renal tissues was assessed following Casini et al. 40 Bradley et al. 41 was the guide upon which the renal tissue levels of MPO were assayed. The Aebi method was the tool used for quantification of CAT levels in the renal tissues. 42 ELISA kits provided by FineTest, Wuhan, 430074, Hubei, China, represented the tools used for assessment of SOD in the renal tissues (Catalog No. ER0332).
Quantification of the levels of SIRT1, nuclear factor erythroid 2-related factor 2 (Nrf2), and heme oxygenase-1 (HO-1) in the harvested renal tissues
LSBio, Newark, CA 94560, United States was the supplier of sandwich ELISA kits used for determination of the renal tissue levels of SIRT1 (Catalog No. LS-F35967). The renal tissue content of Nrf2 was assayed using kits supplied by Shanghai Korain Biotech Co., Shanghai, China (Code No. E1083Ra), while renal HO-1 levels were quantified using the instructions enclosed within the kits purchased from Assay Genie Ltd, Dublin 2, Ireland (Catalog No. AEES00428).
Determination of the tissue levels of the renal cytokines
The renal tissue levels of interleukin-1 beta (IL-1β) and IL-6 were determined by sandwich ELISA kits supplied by Shanghai Korain Biotech Co., Shanghai, China (Code No. E0119Ra and E0135Ra respectively). RayBiotech Life, Inc., Peachtree Corners, GA 30092, United States was the source of ELISA kits which represented the analytical tools used for assessment of monocyte chemo-attractant protein-1 (MCP-1) levels in the renal tissues (Accession No. P14844). Nuclear factor kappa B (NF-κB) p65 levels in the supernatant obtained from the centrifuged homogenate of the renal tissues were assessed using kits supplied by FineTest, Wuhan, 430074, Hubei, China (Catalog No. ER1187).
Quantification of the renal tissue levels of phospho-p38 mitogen-activated protein kinase (MAPK), phospho-extracellular signal-regulated kinase 1/2 (ERK1/2), and phospho-Jun N-terminal Kinase 1/2 (JNK1/2)
The renal tissue levels of phospho-p38 MAPK and phospho-ERK1/2 were estimated using kits purchased from RayBiotech Life, Inc., Peachtree Corners, GA 30092, United States (Accession No. Q16539 and P27361|P28482, respectively) according to the vendor’s guide. Thermo Fisher Scientific Inc., Waltham, Massachusetts, United States, was the source of ELISA kits used for the quantification of phospho-JNK1/2 (Thr183/Tyr185) levels in the renal tissues (Catalog No. 85-86197).
Assessment of the renal tissue content of glucagon-like peptide-1(GLP-1) and DPP-4 activity
The renal tissue content of GLP-1 was assayed using kits provided by FineTest, Wuhan, 430074, Hubei, China (Catalogue No. ER0996). Elabscience, Houston, Texas 77079, United States was the supplier of the fluorometric kits used for determination of DPP-4 activity in the renal tissues (Catalogue No. E-BC-F053).
Evaluation of HMGB1/receptors of advanced glycation end products (RAGE)/toll-like receptor 4 (TLR4) axis in the harvested renal tissues
HMGB1/RAGE/TLR4 axis was evaluated in the renal tissues by following the vendor’s instructions enclosed within the ELISA kits supplied by LSBio, Newark, CA 94560, United States (Catalog No. LS-F4039) for HMGB1, Proteintech Group, Inc., Rosemont, IL 60018, United States (Catalog No. KE20030) for RAGE, and Bio-Techne, Minneapolis, MN 55413, United States (Catalog No. NBP2-76571) for TLR4.
Assessment of the apoptotic markers in the harvested renal tissue specimens
The effect of the different treatments on the apoptotic status of the renal tissues was assessed by measurement of the renal tissue content of caspase three and Bax (BCL-2-associated X protein) (by kits purchased from Novus Biologicals, Centennial, CO 80112, United States, catalog No. NBP2-75024 and NBP2-69938 respectively), and beta cell lymphoma protein 2 (BCL-2) (by kits provided by LSBio, Newark, CA 94560, United States, catalog No. LS-F4135). The methods of determination of these parameters followed the vendors’ instructions.
Evaluation of the histomorphic changes in hematoxylin and eosin (H & E)-stained sections of the harvested renal tissue specimens
Slices of the harvested kidneys were rinsed with ice-cold saline and fixed by incubation for 24 hrs at 25°C in 10% neutral-buffered formaldehyde solution. These slices were then dehydrated in ethyl alcohol and embedded in paraffin wax. Olympus CUT 4055 motorized microTec 1996 microtome (Olympus, Germany) was used to cut the resulting blocks into tissue sections of 5 µm thickness. These sections were then mounted on glass slides and stained with H & E. The AmScope B100 Series 40X-2000X Binocular Biological LED Microscope (AmScope Microscope and Accessories, United States) was utilized for direct visualization of the histopathological changes, which were then quantified with the aid of ImageJ software (Version 1.52f, National Institutes of Health, Bethesda, MD, United States). Quantification of the degree of renal tissue damage was carried out by using the endothelial/glomerular/tubular/interstitial (EGTI) damage score as described by Chavez et al. 43 This score ranges from 0 which denotes absence of renal histomorphic damage, to 14 which means severe renal damage. This score has four components, including endothelial (0 to three scores), glomerular (0 to three scores), tubular (0 to 4 scores), and interstitial (0 to 4 scores) components. The total EGTI damage score is the sum of the scores of the four components.
Evaluation of the extent of expression of Ki-67 in the renal tissues by the immunohistochemical techniques
The paraffin-embedded kidney specimens were cut with an Olympus microtome (Olympus CUT 4055 motorized microTec 1996, Germany) into slices of 5 μm thickness. Then, these slices underwent deparaffinization using a sequence of xylene and ethyl alcohol. After that, the deparaffinized sections were rehydrated in graded solutions of ethyl alcohol (concentrations of 100%, 95%, 80%, and 70%). Ki-67 antigen retrieval was carried out by the enzyme-induced epitope retrieval method, which includes application of an enzyme solution such as trypsin, pepsin, or proteinase K followed by incubation at 37ᵒC for 30 min. Then, the non-specific binding sites were blocked by UltraVision protein block (Thermo Fisher Scientific, Waltham, Massachusetts, United States). After that, Ki-67-specific rabbit monoclonal primary antibody (Roche Diagnostics International AG, Rotkreuz, Switzerland, catalog No. 790-4286) was applied to these sections and incubated together at room temperature for 1 h. Then, Ki-67-specific secondary antibody (Thermo Fisher Scientific, Waltham, Massachusetts, United States, catalog No. PA5-16785) conjugated with HRP was applied to these sections and incubated together at room temperature for 10 minutes. After that, these sections were incubated for 5 minutes with DAB Quanto Chromagen and DAB Quanto Substrate mixture (DAB, Quanto), which acts as a chromogen substrate to visualize the Ki-67 immunostaining. Finally, these sections were counterstained with Gill’s Hematoxylin (Bio-Optica Milano S.p.A. Via San Faustino 58 - 20134 Milano, Italy, catalog No. 05-06014/L) and examined at 40× magnification. Quantification of the percentage of Ki-67 immunoexpression in the renal tissue specimens was performed using Image-J software (Version 1.52f, National Institutes of Health, Bethesda, MD, United States).
Exploration of the effect of the different treatments on the electron microscopic picture of the harvested renal tissue specimens
The harvested specimens that contain the renal cortex were immediately fixed in glutaraldehyde solution and divided into small portions. In order to enhance the contrast, these portions were later on fixed in osmium tetroxide solution followed by dehydration in a series of different concentrations of ethyl alcohol. Then, these portions were hardened by embedding into resin to be ready for cutting into ultra-thin sections of 50 nm thickness by Epredia microtome (Thermo Fisher Scientific, Waltham, Massachusetts, United States). The yielded sections were then stained with uranyl acetate and visualized using JEM-1200EX electron microscope (Jeol, Japan).
Analytical statistics for the obtained data
GraphPad Prism, version 8 (GraphPad Software, LLC, San Diego, CA, United States) was the tool utilized for statistical evaluation of the findings of the current study, and the data were represented as mean ± standard deviation (SD). The normal distribution of the obtained results was verified by the means of the Shapiro–Wilk normality test followed by Bartlett’s test to assess the degree of the variances’ homogeneity. The groups with equal variances were compared to one another using the one-way analysis of variance (One-way ANOVA) followed by the post hoc Tukey test. The animal groups that had unequal variances were compared to one another using Welch ANOVA followed by Games Howell test. The level of statistical significance was settled to be at a p-value equal to or less than 0.05.
Results
Omarigliptin with or without shikonin combatted the perturbations of the renal function tests induced by cyclosporine
Cyclosporine administration elicited a significant perturbation of the renal functions manifested by a significant elevation of the levels of blood urea (p < 0.001), serum creatinine (p < 0.001), renal KIM-1 (p < 0.001), and urinary NAG (p < 0.001) associated with a significant decrement in creatinine clearance (p < 0.001) when compared versus the control group. Interestingly, treatment with either omarigliptin or shikonin elicited a significant improvement of the renal functions evidenced by a significant mitigation of the aforementioned changes in the renal function tests (p < 0.001) with the most favorable results being encountered in rats treated with omarigliptin/shikonin combination when compared versus cyclosporine-injected rats treated with either omarigliptin or shikonin alone (p < 0.001) (Figure 2). Effect of omarigliptin and/or shikonin on the renal function tests in rats injected with cyclosporine. Data are presented as mean ± standard deviation (SD) (n = 10) and statistically analyzed using one-way ANOVA, confirmed by Tukey’s multiple comparison test. Omarigliptin with or without shikonin restored the normal renal functions in cyclosporine-injected rats. p-value <0.05 was considered significant. In (a) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0003 when comparing the CsA + OMG group with the CsA + shikonin group. p = 0.001 when comparing the CsA + OMG group with the CsA + OMG/shikonin combination group. In (b) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0024 when comparing the CsA + OMG group with the CsA + shikonin group. p = 0.0457 when comparing the CsA + OMG group with the CsA + OMG/shikonin combination group. In (c) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG or CsA + OMG/shikonin combination, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0006 when comparing the CsA group with the CsA + shikonin group. p = 0.0034 when comparing the CsA + OMG group with the CsA + shikonin group. p = 0.0005 when comparing the CsA + OMG group with the CsA + OMG/shikonin combination group. In (d) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, the CsA + OMG group with the CsA + OMG/shikonin combination group, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0065 when comparing the CsA + OMG group with the CsA + shikonin group. In (e) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.1125 when comparing the CsA + OMG group with the CsA + shikonin group. p = 0.0003 when comparing the CsA + OMG group with the CsA + OMG/shikonin combination group. CsA: cyclosporine; OMG: omarigliptin; KIM-1: kidney injury molecule-1; NAG: N-acetyl beta-D-glucosaminidase.
Omarigliptin with or without shikonin curbed the oxidative stress-triggered responses in the renal tissues induced by cyclosporine
Figure 3 illustrated that cyclosporine-injected rats exhibited a significant increase in the tissue oxidative stress parameters including MDA (p < 0.001) and MPO (p < 0.001) concomitantly with a significant abrogation of the antioxidant enzymes activity (CAT and SOD) (p < 0.001) relative to the control values. These changes were reversed with administration of either omarigliptin or shikonin with the most admirable responses being evidenced in animals treated with omarigliptin/shikonin combination. Effect of omarigliptin and/or shikonin on the redox status in the renal tissues of rats injected with cyclosporine. Data are presented as mean ± standard deviation (SD) (n = 10) and statistically analyzed using one-way ANOVA, confirmed by Tukey’s multiple comparison test. Omarigliptin with or without shikonin restored the redox balance in the renal tissues of cyclosporine-injected rats. p-value <0.05 was considered significant. In (a) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, the CsA + OMG group with the CsA + shikonin group, the CsA + OMG group with the CsA + OMG/shikonin combination group, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. In (b) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, the CsA + OMG group with the CsA + shikonin group, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0008 when comparing the CsA + OMG group with the CsA + OMG/shikonin combination group. In (c) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, the CsA + OMG group with the CsA + OMG/shikonin combination group, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0005 when comparing the CsA + OMG group with the CsA + shikonin group. In (d) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, the CsA + OMG group with the CsA + OMG/shikonin combination group, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0009 when comparing the CsA + OMG group with the CsA + shikonin group. CsA: cyclosporine; OMG: omarigliptin; MDA: malondialdehyde; MPO: myeloperoxidase; SOD: superoxide dismutase.
Omarigliptin with or without shikonin reversed the changes in the renal tissue levels of SIRT1, Nrf2, and HO-1 induced by cyclosporine
Figure 4 showed a significant decrement in the renal tissue levels of SIRT1 (p < 0.001), Nrf2 (p < 0.001), and HO-1 (p < 0.001) as a consequence of cyclosporine injection relative to the control group. Animals treated with either omarigliptin or shikonin experienced a significant elevation of the renal tissue levels of the aforementioned parameters when compared versus animals injected with cyclosporine alone (p < 0.001). Amazingly, the group treated with omarigliptin/shikonin combination exhibited a significant elevation of these parameters to approximate the control values when compared to the groups treated with either omarigliptin or shikonin (p < 0.001). Effect of omarigliptin and/or shikonin on the renal tissue levels of sirtuin-1 (SIRT1), nuclear factor erythroid 2-related factor 2 (Nrf2), and heme oxygenase-1 (HO-1) in rats injected with cyclosporine. Data are presented as mean ± standard deviation (SD) (n = 10) and statistically analyzed using one-way ANOVA, confirmed by Tukey’s multiple comparison test. Omarigliptin with or without shikonin mitigated the changes in renal SIRT1, Nrf2, and HO-1 levels induced by cyclosporine. p-value <0.05 was considered significant. In (a) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, the CsA + OMG group with the CsA + shikonin group, the CsA + OMG group with the CsA + OMG/shikonin combination group, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. In (b) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG or OMG/shikonin combination, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0085 when comparing the CsA group with the CsA + shikonin group. p = 0.0303 when comparing the CsA + OMG group with the CsA + shikonin group. p = 0.0032 when comparing the CsA + OMG group with the CsA + OMG/shikonin combination group. In (c) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG or OMG/shikonin combination, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0001 when comparing the CsA group with the CsA + shikonin group. p = 0.0372 when comparing the CsA + OMG group with the CsA + shikonin group. p = 0.0176 when comparing the CsA + OMG group with the CsA + OMG/shikonin combination group. CsA: cyclosporine; OMG: omarigliptin; SIRT1: sirtuin-1; Nrf2: nuclear factor erythroid 2-related factor 2; HO-1: heme oxygenase-1.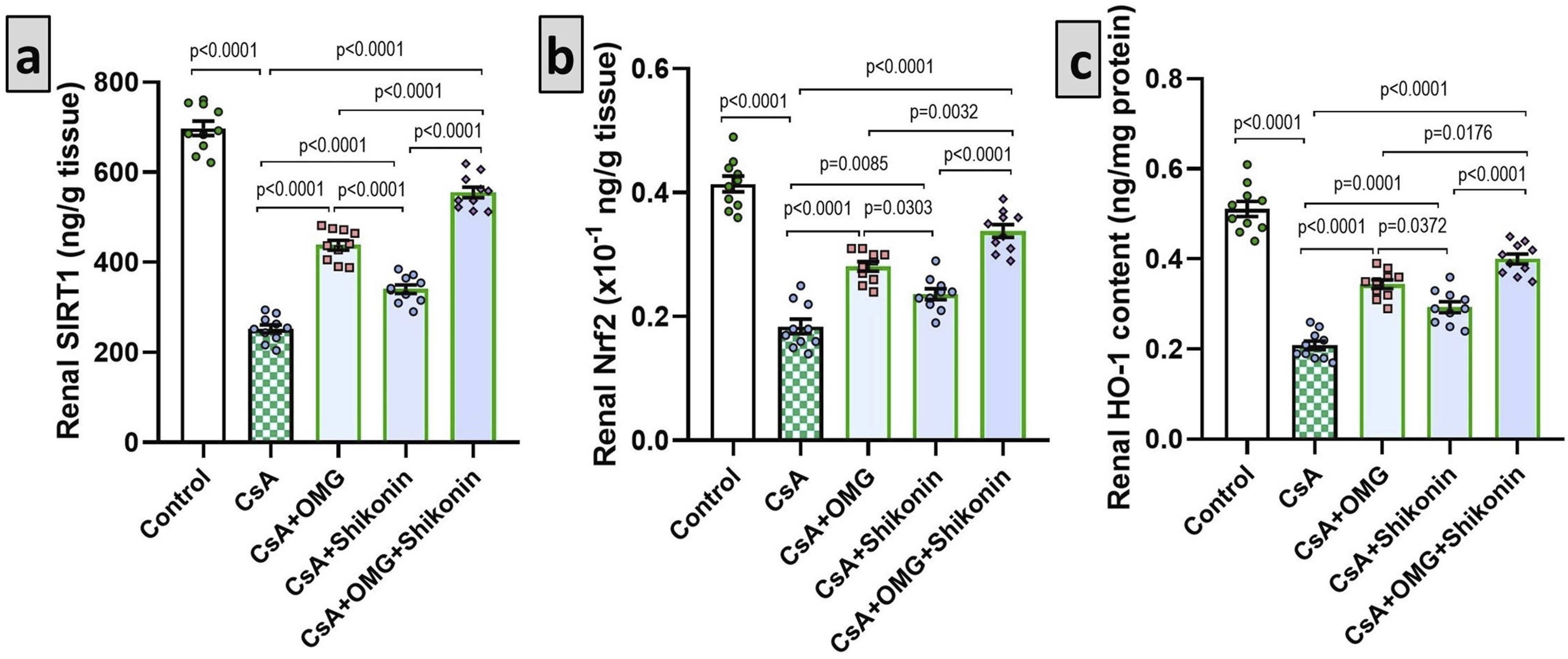
Omarigliptin with or without shikonin combatted the proinflammatory responses in the renal tissues triggered by cyclosporine
When compared with the control animals, the group injected with cyclosporine alone experienced a significant increase in the proinflammatory responses in the renal tissues manifested by a significant augmentation of the renal tissue levels of IL-1β (p < 0.001), IL-6 (p < 0.001), MCP-1 (p < 0.001), and NF-κB p65 (p < 0.001). Interestingly, these deleterious effects were significantly ameliorated in the groups treated with omarigliptin or shikonin (p < 0.001) with the mostly evidenced suppression of the proinflammatory responses being detected in animals treated with omarigliptin/shikonin combination (Figure 5). Effect of omarigliptin and/or shikonin on the renal tissue levels of interleukin-1 beta (IL-1β), IL-6, monocyte chemoattractant protein-1 (MCP-1), and nuclear factor kappa B (NF-κB) p65 in the renal tissues harvested from rats injected with cyclosporine. Data are presented as mean ± standard deviation (SD) (n = 10) and statistically analyzed using one-way ANOVA, confirmed by Tukey’s multiple comparison test. Omarigliptin with or without shikonin abrogated the changes in renal IL-1β, IL-6, MCP-1, and NF-κB p65 levels induced by cyclosporine. p-value <0.05 was considered significant. In (a) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, the CsA + OMG group with the CsA + shikonin group, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0033 when comparing the CsA + OMG group with the CsA + OMG/shikonin combination group. In (b) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, the CsA + OMG group with the CsA + shikonin group, the CsA + OMG group with the CsA + OMG/shikonin combination group, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. In (c) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, the CsA + OMG group with the CsA + OMG/shikonin combination group, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0006 when comparing the CsA + OMG group with the CsA + shikonin group. In (d) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, the CsA + OMG group with the CsA + shikonin group, the CsA + OMG group with the CsA + OMG/shikonin combination group, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. CsA: cyclosporine; OMG: omarigliptin; IL-1β: interleukin-1 beta; MCP-1: monocyte chemoattractant protein-1; NF-κB: nuclear factor kappa B.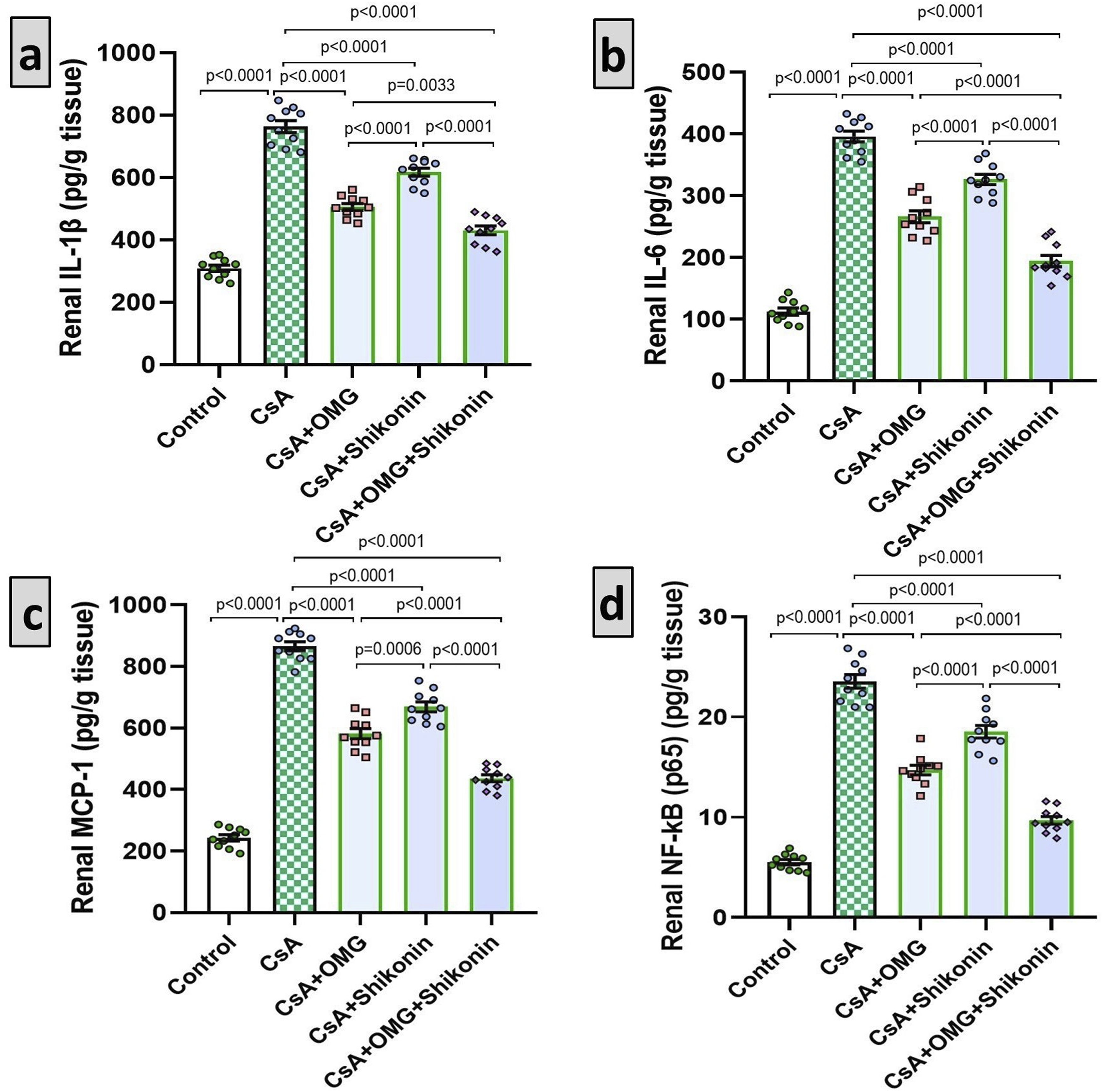
Omarigliptin with or without shikonin interfered with the MAPK transduction in the renal tissues of animals injected with cyclosporine
As depicted in Figure 6, administration of cyclosporine instigated a significant activation of the MAPK pathway as evidenced by a significant elevation of the levels of the phosphorylated forms of p38 MAPK (p < 0.001), ERK1/2 (p < 0.001), and JNK1/2 (p < 0.001) when compared versus the control animals. A unique ability to counteract these changes were detected in animals treated with either omarigliptin or shikonin with the most significant decline in these parameters being evidenced in the group treated with omarigliptin/shikonin combination relative to the groups that were only treated with either of these agents alone (p < 0.001). Effect of omarigliptin and/or shikonin on the mitogen-activated protein kinase (MAPK) transduction in the renal tissues of rats injected with cyclosporine. Data are presented as mean ± standard deviation (SD) (n = 10) and statistically analyzed using one-way ANOVA, confirmed by Tukey’s multiple comparison test. Omarigliptin with or without shikonin ameliorated the changes in MAPK transduction induced by cyclosporine. p-value <0.05 was considered significant. In (a) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, the CsA + OMG group with the CsA + shikonin group, the CsA + OMG group with the CsA + OMG/shikonin combination group, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. In (b) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, the CsA + OMG group with the CsA + shikonin group, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0026 when comparing the CsA + OMG group with the CsA + OMG/shikonin combination group. In (c) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, the CsA + OMG group with the CsA + shikonin group, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.01 when comparing the CsA + OMG group with the CsA + OMG/shikonin combination group. CsA: cyclosporine; OMG: omarigliptin; P-p38 MAPK: phospho-p38 mitogen-activated protein kinase; P-ERK1/2: phospho-extracellular signal-regulated kinase 1/2; P-JNK1/2: phospho-Jun N-terminal Kinase 1/2.
Omarigliptin with or without shikonin restored GLP-1 levels and inhibited DPP-4 activity in the renal tissues of animals injected with cyclosporine
As shown in Figure 7, cyclosporine injection elicited a significant decline in GLP-1 levels (p < 0.001) associated with a significant increase in DPP-4 activity (p < 0.001) in the renal tissues relative to the control animals. Treatment with either omarigliptin or shikonin was associated with a significant mitigation of these changes (p < 0.001) with the most significant increase in tissue GLP-1 levels and the most significant suppressing effect on tissue DPP-4 activity being detected in animals treated with omarigliptin/shikonin combination relative to the animals that received only one of these agents (p < 0.001). Effect of omarigliptin and/or shikonin on glucagon-like peptide-1 (GLP-1) content and dipeptidyl peptidase-4 (DPP-4) activity in the renal tissues harvested from rats injected with cyclosporine. Data are presented as mean ± standard deviation (SD) (n = 10) and statistically analyzed using one-way ANOVA, confirmed by Tukey’s multiple comparison test. Omarigliptin with or without shikonin restored GLP-1 levels and inhibited DPP-4 activity in the renal tissues of cyclosporine-injected rats. p-value <0.05 was considered significant. In (a) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.001 when comparing the CsA + OMG group with the CsA + shikonin group. p = 0.0003 when comparing the CsA + OMG group with the CsA + OMG/shikonin combination group. In (b) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG or CsA + OMG/shikonin combination, the CsA + OMG group with the CsA + shikonin group, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0118 when comparing the CsA group with the CsA + shikonin group. p = 0.0003 when comparing the CsA + OMG group with the CsA + OMG/shikonin combination group. CsA: cyclosporine; OMG: omarigliptin; GLP-1: glucagon-like peptide-1; DPP-4: dipeptidyl peptidase-4.
Effect of the different treatments on HMGB1/RAGE/TLR4 axis in the renal tissues
Administration of cyclosporine significantly enhanced the production of HMGB1 (p < 0.001), RAGE (p < 0.001), and TLR4 (p < 0.001) by the renal tissues when put in comparison to the renal tissues of the control animals. Interestingly, treatment with either omarigliptin or shikonin significantly mitigated cyclosporine-mediated changes in the HMGB1/RAGE/TLR4 axis (p < 0.001) with the most admirable results being detected in animals treated with omarigliptin/shikonin combination when compared to administration of only one of these agents (p < 0.001) (Figure 8). Effect of omarigliptin and/or shikonin on high mobility group box 1 (HMGB1)/receptors for advanced glycation end products (RAGE)/toll-like receptor 4 (TLR4) axis in the renal tissues harvested from rats injected with cyclosporine. Data are presented as mean ± standard deviation (SD) (n = 10) and statistically analyzed using one-way ANOVA, confirmed by Tukey’s multiple comparison test. Omarigliptin with or without shikonin abrogated HMGB1/RAGE/TLR4 axis in the renal tissues of cyclosporine-injected rats. p-value <0.05 was considered significant. In (a) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0041 when comparing the CsA + OMG group with the CsA + shikonin group. p = 0.0007 when comparing the CsA + OMG group with the CsA + OMG/shikonin combination group. In (b) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG or CsA + OMG/shikonin combination, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0002 when comparing the CsA group with the CsA + shikonin group. p = 0.0046 when comparing the CsA + OMG group with the CsA + shikonin group. p = 0.0035 when comparing the CsA + OMG group with the CsA + OMG/shikonin combination group. In (c) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG or CsA + OMG/shikonin combination, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0071 when comparing the CsA group with the CsA + shikonin group. p = 0.0005 when comparing the CsA + OMG group with the CsA + shikonin group. p = 0.0005 when comparing the CsA + OMG group with the CsA + OMG/shikonin combination group. CsA: cyclosporine; OMG: omarigliptin; HMGB1: high mobility group box 1; RAGE: receptors for advanced glycation end products; TLR4: toll-like receptor 4.
Omarigliptin with or without shikonin aborted cyclosporine-mediated apoptosis in the renal tissues
Animals injected with cyclosporine alone exhibited a significant apoptosis enhancement manifested by a significant increase in the renal tissue levels of caspase three (p < 0.001) and Bax (p < 0.001) associated with a significant decline in the renal tissue levels of BCL-2 (p < 0.001) relative to the control animals. These changes were mitigated in animals treated with either omarigliptin or shikonin with the most significant abrogation of the apoptotic process being detected in animals treated with omarigliptin/shikonin combination (p < 0.001) (Figure 9). Effect of omarigliptin and/or shikonin on the apoptotic process in the renal tissues harvested from rats injected with cyclosporine. Data are presented as mean ± standard deviation (SD) (n = 10) and statistically analyzed using one-way ANOVA, confirmed by Tukey’s multiple comparison test. Omarigliptin with or without shikonin combated apoptosis in the renal tissues of cyclosporine-injected rats. p-value <0.05 was considered significant. In (a) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, the CsA + OMG group with the CsA + shikonin group, the CsA + OMG group with the CsA + OMG/shikonin combination group, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. In (b) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG or CsA + OMG/shikonin combination, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0002 when comparing the CsA group with the CsA + shikonin group. p = 0.0265 when comparing the CsA + OMG group with the CsA + shikonin group. p = 0.0123 when comparing the CsA + OMG group with the CsA + OMG/shikonin combination group. In (c) p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG or CsA + OMG/shikonin combination, the CsA + OMG group with the CsA + shikonin group, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0092 when comparing the CsA group with the CsA + shikonin group. p = 0.001 when comparing the CsA + OMG group with the CsA + OMG/shikonin combination group. CsA: cyclosporine; OMG: omarigliptin; Bax: BCL-2-associated X protein; BCL-2: beta cell lymphoma protein 2.
Effect of omarigliptin with or without shikonin on the histomorphic changes in the renal tissues induced by cyclosporine
In the current study, the control group showed the normal appearance of the renal architecture with typical glomeruli and intact renal tubules and interstitial tissues (Figure 10(a) and (g)). The group injected with cyclosporine alone exhibited a significant kidney damage manifested by diffuse glomerular atrophy, increased Bowman’s space, tubular degeneration and vacuolation, massive inflammatory cellular infiltration, and diffuse interstitial hemorrhage (Figure 10(b), (c), and (g)). These changes were significantly abrogated in the groups treated with either omarigliptin or shikonin alone (Figure 10(d) and (e) respectively, and Figure 10(g)) with the best results being encountered in the group treated with omarigliptin/shikonin combination (Figure 10(f) and (g)). Sections of the kidney (H and E ×100, scale bar 100 µm) from (a) The control group showing normal appearance of the renal architecture with typical glomeruli (Thin arrows) and intact renal tubules (Thick arrows) and interstitial tissues; (b) and (c) Cyclosporine group showing a significant kidney damage manifested by diffuse glomerular atrophy (Asterisk), increased Bowman’s space (Thin arrows), tubular degeneration (Thick arrows), extensive vacuolation (Arrow heads), and diffuse interstitial hemorrhage (V); (d) and (e) Cyclosporine-injected groups treated with omarigliptin and shikonin respectively exhibiting a significant increase in the number of the apparently intact glomeruli (Thin arrows) with scattered degenerated glomeruli (Asterisk) and scanty tubular degeneration (Thick arrows) with mild vascular congestion (V); (f) Cyclosporine-injected groups treated with omarigliptin/shikonin combination showing restoration of the normal morphological structure of the renal tissues with apparently normal glomeruli (Thin arrows) and renal tubules (Thick arrow) with minimal vascular congestion (V); (g) EGTI score in the studied groups. Data are presented as mean ± standard deviation (SD) (n = 10) and statistically analyzed using one-way ANOVA, confirmed by Tukey’s multiple comparison test. p-value <0.05 was considered significant. p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG or CsA + OMG/shikonin combination, the CsA + OMG group with the CsA + OMG/shikonin combination group, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0012 when comparing the CsA group with the CsA + shikonin group. p = 0.1553 when comparing the CsA + OMG group with the CsA + shikonin group. CsA: cyclosporine; OMG: omarigliptin.
Effect of omarigliptin with or without shikonin on the immuno-expression of Ki-67 in the renal tissues of rats treated with cyclosporine
The renal tissue sections harvested from animals treated with cyclosporine alone exhibited a significant augmentation of the immune-expression of Ki-67 relative to the renal tissue sections harvested from the control animals (Figure 11(a), (b), and (f)). The immune-expression of Ki-67 in the renal tissues of cyclosporine-injected animals was significantly mitigated with administration of either omarigliptin or shikonin alone with the maximal inhibition being detected in the animals treated with omarigliptin/shikonin combination (Figure 11(c)–(f)). The immunohistochemical expression of Ki-67 in the renal tissue sections (×200, scale bar 40 µm) harvested from (a) The control group; (b) The group injected with cyclosporine alone; (c) The cyclosporine-injected group treated with omarigliptin; (d) Cyclosporine-injected group treated with shikonin; (e) The cyclosporine-injected group treated with omarigliptin/shikonin combination; (f) Quantitative representation of Ki-67 immune expression in the renal tissues harvested from the different studied groups (% of the control). Data are presented as mean ± standard deviation (SD) (n = 10) and statistically analyzed using one-way ANOVA, confirmed by Tukey’s multiple comparison test. p-value <0.05 was considered significant. p < 0.0001 when comparing the control group with the CsA group, the CsA group with the groups treated with CsA + OMG and/or shikonin, and the CsA + shikonin group with the CsA + OMG/shikonin combination group. p = 0.0045 when comparing the CsA + OMG group with the CsA + shikonin group. p = 0.0396 when comparing the CsA + OMG group with the CsA + OMG/shikonin combination group. CsA: cyclosporine; OMG: omarigliptin.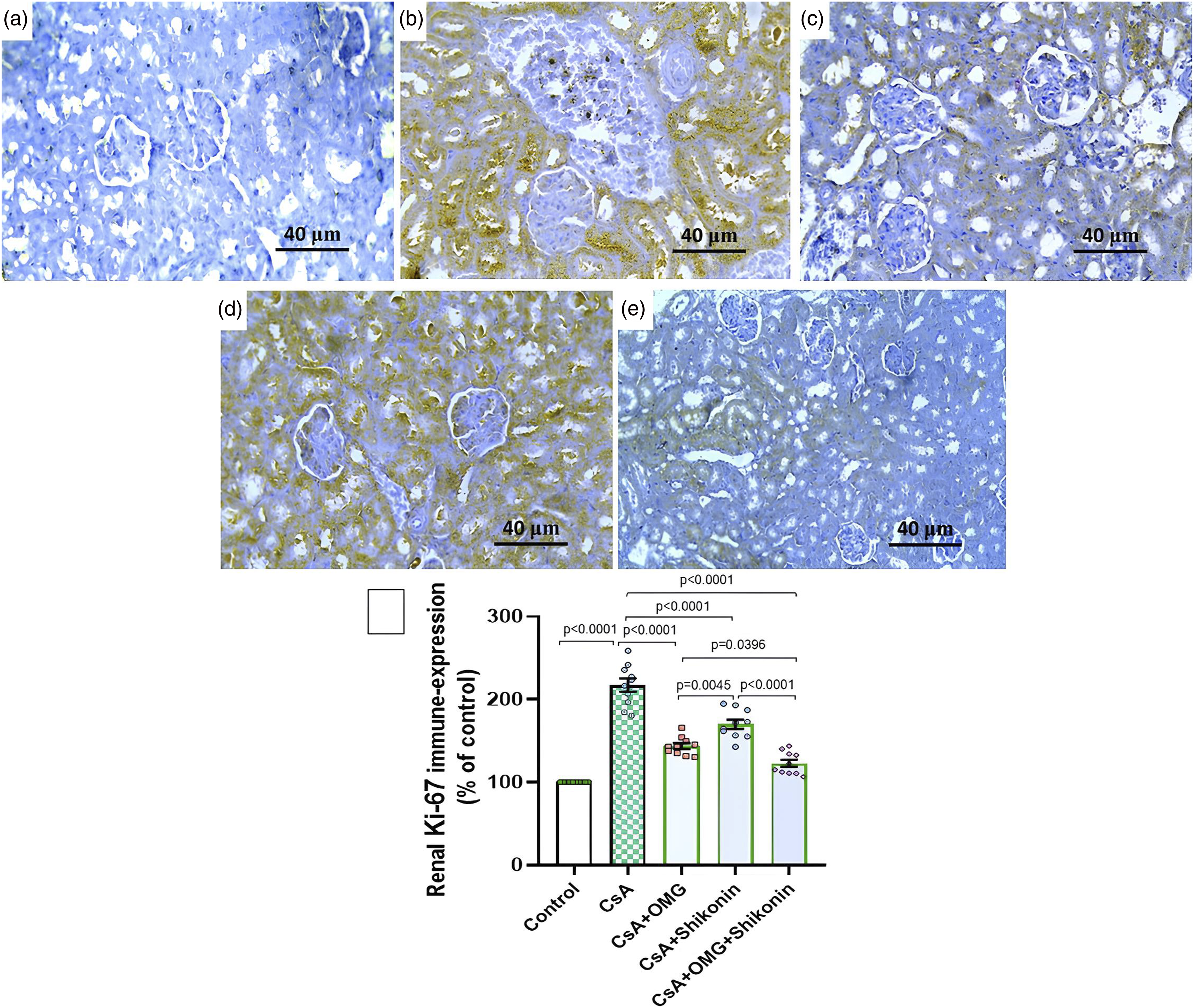
Effect of omarigliptin with or without shikonin on the electron microscopic picture of the renal tissues of rats injected with cyclosporine
The electron microscopic picture of the renal tissue specimens harvested from rats injected with cyclosporine exhibited significant disruption of the glomerular architectures relative to the control group, as evidenced by significant increase in the thickness of the glomerular basement membrane associated with widespread destruction of the foot processes with some of them fused together in some areas (Figure 12(a) and (b)). Treatment of cyclosporine-injected animals with either omarigliptin or shikonin elicited significant alleviation of cyclosporine-mediated electron microscopic changes as demonstrated by significant decrease in the thickness of the basement membrane associated with significant improvement of the electron microscopic picture of the foot processes of the glomeruli with scattered areas of fusion of the foot processes (Figure 12(c) and (d)). The most favorable results were detected in omarigliptin/shikonin combination group relative to the groups treated with either omarigliptin or shikonin (Figure 12(e)). Electron micrographs of the kidney tissue specimens that were excised from rats of (a) the control group showing the fenestrated capillary endothelium (f) on the luminal surface of the glomerular basement membrane (bm). On its outer surface, the secondary foot processes (2) of the podocytes are seen. The secondary foot processes are separated by filtration slits (Arrows); (b) cyclosporine-injected group showing thickened glomerular basement membrane (bm). The fenestrations (F) of the endothelium at the inner aspect of the basement membrane are seen. The secondary foot processes of the podocytes appear effaced and fused (Arrow) or thickened (Double arrows). Obliteration of the filtration slits is seen (Asterisk); (c) cyclosporine-injected group treated with omarigliptin and (d) cyclosporine-injected group treated with shikonin showing a significant decrease in the thickness of the basement membrane with patent fenestrations on the inner aspect of the basement membrane. Some filtration slits are preserved (Arrow) while others are obliterated (Asterisk). The secondary foot processes of the podocytes are either normal or thickened and truncated (2); and (e) cyclosporine-injected group treated with omarigliptin/shikonin combination showing restoration of the normal electron microscopic architecture of the glomeruli with apparently normal thickness of the basement membrane (bm), apparently normal fenestrations (f) at its inner aspect, and normal secondary foot processes (2) at its outer aspect with preserved filtration slits (Arrows) in-between (EM ×8000, scale bar 500 nm).
Discussion
Cyclosporine is an immunosuppressive agent that decreases organ transplant rejection and is an integral part of the management protocols of autoimmune disorders.
44
Despite its proven therapeutic effects, the increased incidence of renal damage represents a major obstacle that may hinder its clinical use.
45
The mechanisms of this nephrotoxicity are multifaceted and involve both direct and indirect effects (Figure 13).
46
This was in agreement with our study where cyclosporine-injected animals exhibited a significant interference with the renal functions relative to the control group. The possible mechanisms of cyclosporine-induced nephrotoxicity (This artwork was created using Reactome icon library and smart art servier items).
Induction of ROS generation and interference with their detoxification mechanisms are the main determinants of cyclosporine-induced nephrotoxicity. 47 Cyclosporine may impair mitochondrial functions leading to energy depletion and exacerbation of kidney damage. 48 This was evidenced in the present study where cyclosporine significantly interfered with the antioxidant enzyme systems which was negatively reflected on renal functions. Interestingly, the group treated with omarigliptin exhibited a significant increase in the antioxidant defenses when compared to the group that received only cyclosporine. This was explained by Kawanami et al. 49 who reported that DPP-4 inhibitors can suppress ROS production in the renal tissues. This together with the ability of omarigliptin to activate Nrf2/HO-1 signaling may explain its protective effects against oxidative stress detected in the current work. 50 In addition, the antioxidant properties of shikonin that were evidenced in the present study were in accordance with Peng et al. 51 who stated that shikonin donates electrons to the free radicals, thereby stabilizing them and preventing them from causing oxidative insult to the kidney. Moreover, shikonin enhances the activity of the antioxidant enzymes, providing significant protection to the renal tissues against oxidative stress. 52
Sirtuin-1 (SIRT-1) is a deacetylase enzyme that directly influences the various cellular processes involved in the pathogenesis of cyclosporine-induced nephrotoxicity. 53 Bazyluk et al. 54 reported that SIRT1 combats cyclosporine nephrotoxicity via abrogation of oxidative stress and increased activity of antioxidant enzymes in the renal tissues, an action that largely depends on activation of Nrf2/HO-1 signaling. In addition, SIRT1, via deacetylation and deactivation of the transcriptional factors, was proven to combat cyclosporine-elicited renal fibrosis. 55 Moreover, SIRT1 is a key regulator of the transcriptional factors that mediate the inflammatory processes in cyclosporine-induced nephrotoxicity.56,57 This coincided with our results where cyclosporine elicited a significant decline in tissue SIRT1 with subsequent inhibition of Nrf2/HO-1 signaling, affection of the tissue redox status, and increased expression of NF-κB p65 with consequent increase of the proinflammatory cytokines in the renal tissues.
In the present work, omarigliptin elicited a significant increase in the renal tissue SIRT1 with subsequent amelioration of the inflammatory responses and the oxidative renal tissue damage. This coincided with Zhang et al. 58 who detected an intimate relationship between DPP-4 inhibition and the increase in renal tissue SIRT1 with subsequent amelioration of the effects of the nephrotoxic agents. Similar to our results, Huang et al. 24 reported that treatment with shikonin was positively correlated with increased tissue expression of SIRT1, which was reflected on the antioxidant status and the inflammatory microenvironment of the renal tissues.
Coinciding with our results, HMGB1 levels were increased in the renal tissues of the cyclosporine group, which highlighted its role in the pathogenesis of cyclosporine-elicited nephrotoxicity. 59 Being released from the damaged tissues in response to cyclosporine, HMGB1 is a key player in cyclosporine nephrotoxicity. 60 It exacerbates kidney damage and aggravates the immune response via interacting with specific receptors on the surface of the immune cells, including RAGE and TLR4. 61 This increases the expression of the proinflammatory cytokines, resulting in sustained inflammation and renal tissue damage. 62 In addition, HMGB1 is a pro-fibrotic agent that stimulates the production of extracellular matrix proteins, resulting in progressive renal scarring. 63 Also, Zmijewska et al. 10 reported that HMGB1 can amplify the deleterious consequences of oxidative insult to the renal tissues, possibly via increased ROS production with subsequent interference with the antioxidant defenses, perpetuating the cycle of kidney injury induced by cyclosporine. These effects were ameliorated in the present study with administration of omarigliptin which significantly combatted the changes in HMGB1/RAGE/TLR4 signaling induced by cyclosporine. This agrees with Allam et al. 64 who stated that the anti-inflammatory effects of DPP-4 inhibitors are mediated via modulation of HMGB1 expression with subsequent affection of the levels of RAGE and TLR4. In addition, shikonin in the current work elicited a significant detrimental effect on the HMGB1/RAGE/TLR4 axis, which agreed with the previous reports that postulated that HMGB1 is one of the main targets of shikonin that explains its potent antioxidant and inflammation-mitigating properties.65,66
The expression of p38/ERK/JNK MAPKs plays a fundamental role in the pathogenesis of cyclosporine-elicited nephrotoxicity. 67 As evidenced in our study, activation of p38 MAPK by cyclosporine leads to increased production of the pro-inflammatory cytokines and augmentation of oxidative stress and apoptosis leading to significant kidney damage. 68 Also, cyclosporine-mediated activation of renal tissue ERK may aggravate the cellular stress, enhance apoptosis, and interfere with tissue survival. 67 Moreover, JNK MAPK was proven to have a direct connection to the stress responses and apoptosis of the renal tissues initiated by cyclosporine. 69 Interestingly, the present study’s findings demonstrated that treatment of cyclosporine-injected rats with omarigliptin significantly inactivated p38/ERK/JNK MAPKs and abrogated apoptosis relative to rats that received cyclosporine alone. This agrees with Shao et al. 70 who stated that DPP-4 inhibitors inhibit the genetic expression of the phosphorylated forms of MAPKs, which possibly offers therapeutic benefits. Additionally, the modulatory effect of shikonin on MAPKs expression and the apoptotic processes detected in the current work was explained by its ability to inhibit the expression of the phosphorylated ERK and c-Myc gene, thereby modulating the signaling pathways that control cell survival and proliferation.71,72
In agreement with our findings, recent reports stated that the renal tissues of animals treated with cyclosporine have significantly lower levels of GLP-1 and a significant increase in the activity of DPP-4, which is responsible for the degradation of GLP-1, relative to the control values. 73 Moreover, GLP-1 was proven to inhibit the release and abort the action of the pro-inflammatory cytokines, thereby mitigating the renal inflammatory responses. 19 Additionally, the anti-fibrotic effect of GLP-1 mediated via downregulation of the activity and proliferation of the fibroblasts may impart a role for GLP-1 to combat the effects of cyclosporine on the kidneys. 20 Furthermore, Haddock et al. 74 reported that GLP-1 improves renal perfusion via inducing vasodilation of the renal vasculature, thereby ensuring sufficient oxygen and nutrient supply to the renal tissues. These data may explain our findings where omarigliptin induced a significant suppression of DPP-4 activity associated with a significant increase in tissue GLP-1 levels with a significant amelioration of the redox imbalance, the inflammatory responses, and the renal histopathological changes elicited by cyclosporine.
Cyclosporine-injected rats treated with shikonin in the current work exhibited a significant decline in DPP-4 activity associated with a significant elevation in tissue GLP-1 levels relative to animals injected with cyclosporine alone. This was in agreement with Saeed et al. 27 who reported that shikonin has an antihyperglycemic effect via inhibition of DPP-4 activity with subsequent increase of GLP-1 levels.
In the current work, rats injected with cyclosporine exhibited a significant increase in Ki-67 immune expression relative to the control rats. Mizui et al. 75 reported that in cyclosporine-induced nephrotoxicity, Ki-67 can be used as a marker to assess the extent of cellular proliferation and damage. Studies on different animal models have reported that short-term cyclosporine treatment leads to increased renal tissue levels of Ki-67, alongside other markers such as proliferating cell nuclear antigen and transforming growth factor beta 1, denoting active cellular regeneration and remodeling of renal tissue. 76 Meanwhile, long-term therapy with cyclosporine leads to pathological fibrosis and profound chronic tissue damage, where the tissue levels of the proliferative markers, such as Ki-67, may decline as cells enter the senescence phase. 77 Therefore, Ki-67 can be considered not only as a biomarker for early renal injury but also provides insight into the dynamic balance between repair and degeneration in cyclosporine-induced renal damage. 76
In agreement with our findings, DPP-4 inhibitors may have anti-proliferative effects leading to decreased Ki-67 expression. 78 The exact mechanism isn’t yet fully understood and may involve complex pathways related to inflammation and the regulation of cellular differentiation. 79 Also, the inhibitory effect of shikonin on Ki-67 immune expression that was detected in our study may be attributed to its ability to target certain signaling pathways such as p53 and the phosphorylation of ERK and Akt, thereby downregulating the immune expression of Ki-67, inhibiting cellular proliferation, and inducing cell cycle arrest. 80
In the current study, cyclosporine-injected animals treated with the omarigliptin/shikonin combination exhibited the most favorable mitigating effects on cyclosporine-induced kidney injury relative to animals treated with each of these agents alone. This effect may rely on their ability to regain the renal oxidant/antioxidant balance and their capacity to modulate the inflammatory signals and regulate cellular differentiation in the kidney. Also, both agents can affect SIRT1/Nrf2/HO-1 axis and its downstream mediators in the kidney. Additionally, the inhibitory effect of both agents on DPP-4 enzyme activity, with subsequent restoration of the renal GLP-1 levels, leads to a significant amelioration of the HMGB1/RAGE/NF-κB axis and inactivation of MAPKs, thereby interfering with the inflammatory and fibrogenic processes in the kidney. Moreover, shikonin was proven to inhibit P-glycoprotein, 28 an effect that improves the absorption and enhances the bioavailability of DPP-4 inhibitors, including omarigliptin. 29
The current medical literature and the major drug interaction databases didn’t report any significant pharmacokinetic or pharmacodynamic drug interactions between cyclosporine and omarigliptin. 2 Cyclosporine is a potent immunosuppressive with a high potential for drug interactions, especially with agents that affect the cytochrome P-450 enzyme system, which is needed for its activity and excretion. 81 Being primarily excreted unchanged via the kidneys, omarigliptin has a low risk for drug interactions with cyclosporine. 82 However, the well-documented effect of shikonin on the cytochrome P-450 enzyme system may suggest a potential pharmacokinetic drug interaction with cyclosporine, which may influence its effectiveness and clearance. 83 Exploration of the effect of this potential drug interaction will be handled in our future research.
The results of the present work may offer preliminary insights into dosing, timing, and safety, which can be useful when considering the application of these results in humans. Also, assessment of the mechanisms of toxicity and the protective effects of omarigliptin with or without shikonin against cyclosporine-elicited renal damage in animal models can guide future human trials to develop safer therapeutic strategies for the mitigation of cyclosporine A nephrotoxicity. The limitations of the present study may include the relatively small number of animals used and the short duration of the study. Also, the biological differences between rats and humans in the pharmacokinetic and pharmacodynamic behavior of the drugs may be a challenge to our research. In addition, our study didn’t investigate whether omarigliptin with or without shikonin had an impact on the immunosuppressive properties of cyclosporine, which represents an important step for the possible application of the results of the present study in the clinical settings. Moreover, the present study didn’t investigate the potential adverse effects associated with either omarigliptin or shikonin. Also, the absence of a positive control group limits comparative assessment. Mechanistic interpretations are limited by reliance on ELISA/immunohistochemistry without Western blot or mRNA confirmation. In addition, the combination effects were assessed descriptively without formal synergy analysis.
Conclusion
Omarigliptin/shikonin combination may open new horizons towards amelioration of cyclosporine-elicited nephrotoxicity. This may be derived from their inhibitory effects on DPP-4 activity with a subsequent increase in the renal GLP-1 levels, which consequently affects the redox status, the cellular proliferative capacity, apoptosis, and the inflammatory microenvironment of the renal tissues. Further in-depth mechanistic research is vitally required to fully explore the molecular and cellular events that contribute to these effects, including Western blot analysis and mRNA measurement of the target proteins in addition to experiments on knock-out animals. Moreover, assessment of the effect of omarigliptin with or without shikonin on the immunomodulatory properties of cyclosporine should be carried out. In addition, further research efforts should be directed towards evaluation of the potential safety of this combination and the possibility of application of the findings of the present study in clinical settings.
Footnotes
Acknowledgement
The authors thank Scientific Research Deanship at the University of Ha’il, Saudi Arabia, for funding this research through project number RG-24 020.
Ethical consideration
The protocol of the present study was ethically approved by the Research Ethics Committee of the Faculty of Medicine, Tanta University, Tanta, Egypt (Approval code 36264PR1078/2/25).
Authors’ contribution
Conceptualization and Data curation: Ahmed M. Kabel; Methodology, Investigation, Formal analysis, Visualization, Validation: Marwa H. Abdallah, Hanan Abdelmawgoud Atia, Hemat A. Elariny, Amany M. Khalifa, Asmaa Saleh, Ahmed M. Kabel; Funding acquisition: Hanan Abdelmawgoud Atia; Writing- Original draft preparation: Ahmed M. Kabel; Reviewing and Editing: Ahmed M. Kabel. All authors contributed to and have approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Scientific Research Deanship at University of Ha'il, Saudi Arabia, through project number RG-24 020.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article and its supplementary information files.
