Abstract
Cyclosporine-A (CsA) is an immunosuppressive drug which has been used to prevent rejection after organ transplantation and to treat certain autoimmune diseases. However, its therapeutic use is limited by nephrotoxicity. In this study, the modulator effect of allicin on the oxidative nephrotoxicity of CsA in rats was investigated. Furthermore, the effect of allicin on CsA-induced hypersensitivity of urinary bladder rings to acetylcholine (ACh) was estimated. Rats were divided into three groups, control, CsA (15 mg/kg, subcutaneously), and CsA/allicin (50 mg/kg, orally). At the end of the study, all rats were killed and then blood, urine samples, and kidneys were taken. CsA administration caused a severe nephrotoxicity which was evidenced by elevated kidney/body weight ratio, serum creatinine (Cr), blood urea nitrogen, lactate dehydrogenase, and urinary protein with a concomitant reduction in serum albumin and Cr clearance as compared with control. A significant increase in renal contents of malondialdehyde, myeloperoxidase, and tumor necrosis factor-alpha with a significant decrease in renal reduced glutathione, superoxide dismutase activities, and nitric oxide (NOx) content was detected upon CsA administration. Exposure to CsA increased the sensitivity of isolated urinary bladder rings to ACh. Histological analysis revealed that CsA caused tubular necrosis and moderate diffuse tubular atrophy. Allicin protected kidney tissue against the oxidative damage and the nephrotoxic effect of CsA and significantly reduced the responses of isolated bladder rings to ACh. Our study indicates that allicin administration has the potential to protect against CsA-induced renal injury by reducing oxidative stress and inflammation and restoring NOx level.
Introduction
Despite the widespread use of cyclosporine-A (CsA) as an immunosuppressant in organ transplantation and in control of several autoimmune diseases, 1 its nephrotoxic impact continues to present serious challenges in medicine. One key element in the initiation and progression of CsA nephrotoxicity is the imbalance between renal vasodilator and vasoconstrictor capacities leading to renal ischemia and deterioration of renal function. 2 Apart from primates, the rat appears to be one of the few laboratory species susceptible to CsA-induced nephropathy and has been proposed as a model of the morphologic and functional changes that occur in the human. 3
The exact mechanisms of CsA-induced hypertension and nephrotoxicity remain obscure. Experimental studies revealed that several mechanisms may be involved. These include activation of the renin–angiotensin system 4 and increased synthesis of endothelins. 5 Several studies suggest that a defect in intracellular calcium handling, 6 magnesium deficiency, 7 oxidative stress, 8 and nitric oxide (NOx) system 9 are involved. During the last decade, considerable attention has been focused on the involvement of reactive oxygen species (ROS) in various organs including the kidney. ROS have been proposed as mediators of different kidney diseases especially in toxic, ischemic, or immunological conditions. 10 Several studies suggest that many antioxidants and free radical scavengers provide marked functional and histopathological protection against CsA nephrotoxicity. 11 Nephrotoxic effects of CsA involve acute vasoconstriction related to afferent glomerular arterioles as well as a pro-fibrotic effect in the chronic phase.
In the past few years, much interest has been paid to the role of naturally occurring dietary substances for the control and management of various chronic diseases. 12 Allicin (diallyl thiosulfinate), the major pharmacological component of garlic, 13 has attracted attention of the international medical field gradually due to its potential for disease prevention and treatment. It is formed by the action of the enzyme alliinase on alliin in crushed fresh garlic cloves. It possesses antioxidant activity and is shown to cause a variety of actions potentially useful for human health. Allicin exhibits hypolipidemic, antiplatelet, antibacterial, and antifungal effects. It has been reported that allicin inhibits various cancer cells demonstrating anticancer and chemopreventive activities. 14,15
The present study aimed to evaluate the potential protective effect of allicin in an experimental model of cyclosporine-induced nephrotoxicity.
Materials and methods
Chemicals
Allicin was obtained as pharmaceutical drug (Allimax capsule containing 100% allicin powder; Allisure® AC-23) obtained from (Allimax Nutraceuticals, Chicago, Illinois, USA). Cyclosporine was purchased as a pharmaceutical drug (cyclosporine oral capsules 50 mg; Sandimmune neoral, Novartis, Switzerland). All other chemicals and reagents used were of the highest analytical grade commercially available.
Animals
Twenty-four adult male Sprague–Dawley rats weighing 150–200 g purchased from Vacsera center, Helwan, Egypt, were used for the experimental procedures. Rats were allowed 1 week to adapt to the surroundings before beginning any experimentation. Animals were housed in individual plastic cages with bedding. Standard rat food and tap water were available ad libitum for the duration of the experiments unless otherwise noted. Temperature was maintained at 25°C with 12/12-h light/dark cycle. All animal experiments described in this study comply with the ethical principles and guidelines for the care and use of laboratory animals adopted by “Research Ethics Committee, Faculty of Pharmacy, Mansoura University.”
Experimental design
The animals were randomly divided into three groups containing eight rats in each group. Group (1): Control group, rats did not receive any solvent or drug during the experiment and were on a usual diet. Group (2): CsA group, CsA was administrated to rats (15 mg/kg, subcutaneously) diluted in olive oil with a ratio 1:1 for 4 weeks.
16
Group (3): CsA/allicin (AL) group, rats were injected with CsA (15 mg/kg, subcutaneously) along with AL (50 mg/kg, orally)
17
from the first day till the end of experimental study.
After the last dose, all control and experimental animals were immediately kept in individual metabolic cages (without prior adaptation) for collection of 24-h urine samples. These samples were centrifuged for 15 min at 3000 r/min and kept frozen until analyzed. Blood samples were obtained from overnight fasted animals through retro-orbital sinus, under diethyl ether anesthesia, into non-heparinized tubes. The collected blood samples were allowed to clot for 30 min at 25°C. After clotting, they were centrifuged at 1000 × g, 4°C for 15 min using cooling centrifuge (Damon/IEC Division, Model: CRU-5000, Needham, Massachusetts, USA). Sera were collected and stored frozen for the determination of levels of creatinine (Cr), blood urea nitrogen (BUN), albumin, and lactate dehydrogenase (LDH). The animals were killed after anesthesia with diethyl ether by cervical dislocation, then the lower abdomen was opened and the contractile response of the isolated urinary bladder rings toward ACh was tested. Fatty adherents from the kidneys were removed and the kidneys were weighed using a digital balance to calculate the kidney body weight ratio. The left kidneys were excised immediately, rinsed in ice-cold normal saline (0.9% w/v), homogenized in 0.1 M phosphate buffer (pH 7.4) to yield 10% w/v tissue homogenates that were then used for biochemical assays. The right kidneys were harvested for histopathological examination.
Determination of serum Cr and BUN
Cr was measured in rat sera as described by Bartels et al. 18 Urea was measured enzymatically in rat sera according to Fawcett and Scott. 19
Determination of CCr
Glomerular filtration was assessed by creatinine clearance (CCr) based on serum and urine Cr levels, with values expressed in mL/min, computed with the formula:
Urine flow was calculated by dividing 24 h of urine volume by 1440, which corresponds to the number of minutes in 24 h (60 min × 24 h = 1440): urine flow (mL/min) = value of urine volume (24 h)/1440 and it was expressed as mL/min.
Determination of protein in urine
Protein in urine was determined by Folin–Lowry colorimetric method. 20
Determination of serum albumin and LDH
Albumin was measured in rat sera, as described by Doumas et al. 21 LDH activity was assessed according to the method described by Henry. 22
Determination of lipid peroxidation
Lipid peroxidation was determined by the method of Ohkawa et al. 23
Estimation of SOD activity
Superoxide dismutase (SOD) activity in kidney homogenates was measured spectrophotometrically by monitoring the SOD-inhibitable autooxidation of pyrogallol, as described by Marklund. 24
Estimation of GSH activity
Reduced glutathione (GSH) was determined according to the method described by Ellman. 25
Measurement of NO2 −/NO3 − concentration
Nitrite/nitrate (NO2 −/NO3 −) production, an indicator of NO synthesis, was measured in the supernatant of the kidney homogenate using a commercially available NO assay kit (R&D Systems Inc., Minneapolis, Minnesota, USA) following the manufacturer’s instruction.
Measurement of TNF-α level
Tumor necrosis factor-alpha (TNF-α) concentrations were measured in kidney tissue homogenates using an enzyme-linked immunosorbent assay kit (R&D Systems Inc.). Kidney tissue homogenate was added to a microtiter plate precoated with a monoclonal antibody specific for rat TNF-α. Incubation, plate washing, and quenching of reactions were carried out according to the kit manufacturer’s instruction and TNF-α levels were expressed as picograms per milligram protein (Bender Med Systems GmbH, Vienna, Austria).
Determination of MPO activity
The neutrophils accumulation in the kidney was measured by assaying myeloperoxidase (MPO) activity, as described by Schierwagen et al. 26
Isolation and preparation of urinary bladder rings
Following the collection of blood samples, the rats were killed by after anesthesia with diethyl ether by cervical dislocation; the lower abdomen was opened and the urinary bladder was exposed, the connective tissue and accompanying blood vessels were cut away, and the bladder was cut into rings and placed in a warm physiological salt solution (PSS). The composition of the PSS 27 in grams per liter was as follows: sodium chloride, 6.9; sodium bicarbonate, 2.1; potassium chloride, 0.35; magnesium sulfate, 0.15; monopotassium phosphate, 0.16; calcium chloride, 0.28; and glucose, 2.0. The rings were mounted horizontally between a clamp and a force transducer for the measurement of the isometric tension in an organ bath that was filled with 10 mL of the PSS at a temperature of 37°C and gassed with 95% oxygen to 5% carbon dioxide. The rings were allowed to equilibrate for 30 min prior to the experiment under a resting load of 1 g. 28 During this time, the bath solution was replaced every 5 min. Isometric tension generated by the smooth muscle was measured by means of an isometric transducer (serial no. 88576, Biegestab K30, Hugo Sachs Elektronik, Federal Republic of Germany) recorded with a Powerlab unit/400 linked to a PC running Chart version 4.2 software (AD Instruments Pty Ltd, Australia). Concentration–response curves to acetylcholine (ACh) were constructed. The rings were exposed to different concentrations of ACh in a cumulative manner. Exposure to each concentration of ACh was maintained until the maximal response to that concentration was reached. The responses of the bladder rings were calculated as grams tension per gram tissue.
Histological examinations
The kidney tissues of rats were fixed in 10% neutral buffered formalin solution for 24 h and embedded in a paraffin wax. Tissues were then sectioned at 5 μm, stained with hematoxylin eosin. A minimum of 20 fields were assessed in each biopsy and graded semiquantitatively by a pathologist blinded to treatment groups. For tubular injury: 0 = no tubular injury; 0.5 = <5% of tubules injured; 1 = 5–20%; 1.5 = 21–35%; 2 = 36–50%; 2.5 = 51–65%; and 3 = >65% of tubules injured. 29
Statistical analysis
The values are expressed as mean ± standard error of mean (SEM), for eight rats in each group. Bladder contraction was calculated as grams tension per gram tissue. The highest response obtained was considered as the maximum response (E max). pEC50 (negative log of the concentration producing 50% of maximal response) was determined from nonlinear regression analysis (four-parameter curve fit). Statistical analyses were performed using one-way analysis of variance followed by a Tukey–Kramer post hoc test.
Kruskal–Wallis test was used followed by Dunn test as a nonparametric measure.
Differences were considered significant at p < 0.05. Statistical analyses were carried out using Graph pad Prism software (GraphPad Software Inc. V4.03, San Diego, California, USA).
Results
Kidney/body weight ratio
The results in Figure 1 show that CsA produced a significant increase in the kidney body weight ratio compared to control rats. In the CsA/AL-treated group, the kidney body weight ratio was significantly lower than that of the CsA-treated group.

Effect of AL (50 mg/kg) on kidney/body weight in CsA-treated rats. Values are expressed as means ± standard error of mean (n = 8). Comparisons performed using one-way ANOVA followed by Tukey–Kramer multiple comparisons post hoc test. *p < 0.05 versus control; #p < 0.05 versus CsA-treated group. CsA: cyclosporine-A; AL: allicin; ANOVA: analysis of variance.
Serum biochemical parameters
Kidney functions
Cyclosporine caused a significant elevation in serum Cr and BUN and urinary protein when compared to the control group, with a significant decrease in serum albumin and CCr. Allicin administration significantly attenuated CsA-induced changes in serum Cr, BUN, CCr, serum albumin, and proteinuria compared to CsA-treated rats (Table 1).
Effect of AL (50 mg/kg) on kidney function parameters in CsA-treated rats.a

ANOVA: analysis of variance; CsA: cyclosporine-A; AL: allicin; SEM: standard error of mean; BUN: blood urea nitrogen; CCr: creatinine clearance.
aData expressed as means ± SEM (n = 8). Analyses performed using one-way ANOVA followed by Tukey–Kramer multiple comparisons post hoc test.
b p < 0.05 versus control.
c p < 0.05 versus CsA.
Serum LDH activity
Figure 2 shows that CsA produced a significant increase in LDH activity compared to control rats. In the CsA/AL-treated group, LDH activity significantly decreased compared to CsA-treated rats.

Effect of AL (50 mg/kg) on serum LDH activity in CsA-treated rats. Values are expressed as means ± standard error of mean (n = 8). Comparisons performed using one-way ANOVA followed by Tukey–Kramer multiple comparisons post hoc test. *p < 0.05 versus control; #p < 0.05 versus CsA-treated group. CsA: cyclosporine-A; AL: allicin; LDH: lactate dehydrogenase; ANOVA: analysis of variance.
Antioxidant status
Table 2 shows that after 4 weeks of treatment, CsA significantly increased the malondialdehyde (MDA) levels in rat kidney tissue homogenate but decreased both GSH and SOD activities. Allicin significantly decreased the CsA-induced changes in MDA levels and significantly increased GSH and SOD activities.
Effect of AL (50 mg/kg) on kidney anti-oxidant status in CsA-treated rats.a

ANOVA: analysis of variance; CsA: cyclosporine; AL: allicin; SEM: standard error of mean; MDA: malondialdehyde; GSH: reduced glutathione; SOD: superoxide dismutase.
aData expressed as means ± SEM (n = 8). Analyses performed using one-way ANOVA followed by Tukey–Kramer multiple comparisons post hoc test.
b p < 0.05 versus control
c p < 0.05 versus CsA.
Renal NOx content
The results in Figure 3 show that CsA produced a significant decrease in the renal NOx content compared to control rats. In CsA/AL-treated group, the renal NOx content significantly increased when compared to CsA-treated group.
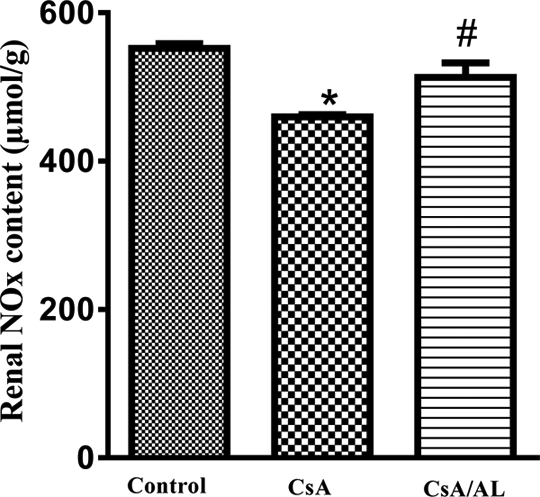
Effect of AL (50 mg/kg) on renal NOx content in CsA-treated rats. Values are expressed as means ± standard error of mean (n = 8). Comparisons performed using one-way ANOVA followed by Tukey–Kramer multiple comparisons post hoc test. *p < 0.05 versus control; #p < 0.05 versus CsA-treated group. CsA: cyclosporine-A; AL: allicin; ANOVA: analysis of variance; NOx: nitric oxide.
Renal TNF-α content
Figure 4 shows that CsA produced a significant increase in the renal TNF-α content compared to control rats. In the CsA/AL-treated group, the renal TNF-α content decreased significantly from the CsA-treated group.

Effect of AL (50 mg/kg) on renal TNF-α level in CsA-treated rats. Values are expressed as means ± standard error of mean (n = 8) Comparisons performed using one-way ANOVA followed by Tukey–Kramer multiple comparisons post hoc test. * p < 0.05 versus control; #p < 0.05 versus CsA-treated group. CsA: cyclosporine-A; AL: allicin; ANOVA: analysis of variance; TNF-α: tumor necrosis factor-alpha.
Renal MPO activity
The results in Figure 5 show that CsA produced a significant increase in the renal MPO activity compared to control rats. In the CsA/AL-treated group, the renal MPO activity decreased significantly from the CsA-treated group.

Effect of AL (50 mg/kg) on renal MPO activity in CsA-treated rats. Values are expressed as means ± standard error of mean (n = 8). Comparisons performed using one-way ANOVA followed by Tukey–Kramer multiple comparisons post hoc test. *p < 0.05 versus control; #p < 0.05 versus CsA-treated group. CsA: cyclosporine-A; AL: allicin; MPO: myeloperoxidase; ANOVA: analysis of variance.
Bladder reactivity
The effects of CsA and AL on the responses of bladder rings to ACh are shown in Figure 6. The average increments in the control urinary bladder rings tension after ACh of 0.1, 0.3, 1, 3, and 10 µM were 2 ± 0.3, 5. 2 ± 0.5, 12.5 ± 0.8, 29.8 ± 0.7, and 63.8 ± 1.9 g tension/g tissue, respectively. Treatment with CsA significantly enhanced the responsiveness of the rings toward ACh, as compared with the control group. Thus, the average increments in tension in response to ACh of 0.1, 0.3, 1, 3, and 10 µM were 17.5 ± 0.7, 53.6 ± 2.5, 137.2 ± 4.1, 250.8 ± 5.2, and 380.9 ± 8.1 g tension/g tissue, respectively. Urinary bladder rings, isolated from CsA/AL-treated rats showed a significant reduction in their responsiveness to ACh when compared to the CsA-treated ones. The average increments in their tensions in response to ACh of 0.1, 0.3, 1, 3, and 10 µM were 6.2 ± 0.2, 18.8 ± 0.9, 43 ± 1.9, 94.5 ± 5.2, and 164.8 ± 7.6 g tension/g tissue, respectively.

Effect of CsA (15 mg/kg) and AL (50 mg/kg) on contractions to ACh in isolated rat urinary bladder rings. Values are expressed as means ± standard error of mean (n = 5). Cumulative dose–response curve to ACh (10−7 to 10−5 M) was measured. *p < 0.05 for E max value compared with control group; # p < 0.05 for E max value compared with CsA group using one-way ANOVA followed by Tukey–Kramer’s multiple comparison post hoc test. CsA: cyclosporine-A; AL: allicin; ACh: acetylcholine; ANOVA: analysis of variance.
Histopathological results
The kidney of control rats showed normal architecture of glomerulus and tubules (Figure 7(a)). Kidney of cyclosporine-induced rats showed acute drug toxicity with tubular dilatation, vacuolization, and cast formation (Figure 7(b) and (c)). Treatment of rats with 50 mg/kg/day allicin along with CsA (Figure 7(d)) ameliorated the degree of renal tissue damage. Semiquantitative analysis of tubular injury scores were demonstrated in Table 3.

Light micrographs of sections from rat kidneys stained with hematoxylin and eosin. (a) Control group: showed normal architecture of glomerulus and tubules; (b) CsA group: showed acute tubular necrosis with sloughed necrotic cells, moderate diffuse tubular atrophy with hyaline casts; (c) CsA group: showed marked hyaline cast formation; (d) CsA/AL group: The degree of renal tissue damage was significantly ameliorated in the rats pretreated with allicin. Original magnification (a) and (b) ×200; (c) and (d) ×400. CsA: cyclosporine-A; AL: allicin.
Mean scores of tubular injury.a

CsA: cyclosporine-A; AL: allicin; SEM: standard error of mean.
aValues represent the mean ± SEM of eight rats/group. Mean values were compared using Kruskal–Wallis followed by Dunn test (p < 0.05).
bSignificantly different from the mean value of the control group.
cSignificantly different from the mean value of CsA group.
Discussion
Cyclosporine continues to be the backbone of post-transplant immunosuppression. However, side effects associated with CsA treatment are numerous and kidney dysfunction is the main complication of CsA treatment. 30
This study demonstrated that allicin administration attenuated renal damage in rats treated with CsA. The protective effects of allicin may be multifactorial involving regulatory effects on inflammation and oxidative stress.
The present study demonstrated that CsA administration increased the kidney/body weight ratio of rats when compared to that of control group. This result agreed with Chandramohan and Parameswari 30 who showed that injection of CsA with a dose of 25 mg/kg for 21 days produce a significant increase in kidney/body weight ratio compared to the control group. This increase in kidney/body weight ratio may be due to inflammation and edema occurring in concomitant with weight loss in rats.
Allicin administration with CsA showed a significant decrease in kidney/body weight index compared to CsA-treated group; this can be explained by the study of Son et al. 31 that has proved that allicin downregulates gamma IR-induced intercellular adhesion molecule-1 (ICAM-1) expression. ICAM-1 plays a pivotal role in inflammatory responses, so this downregulation of this adhesion molecule leads to reduction of inflammation and edema, and also allicin exhibits anti-inflammatory effect by inhibiting cell-mediated T-helper 1 and inflammatory cytokines (TNF-α, IL-1α, IL-6, IL-8, T-cell, interferon-γ, and IL-2) while upregulating IL-10 production. 32
The results of this study show that CsA administration produced a typical pattern of nephrotoxicity which was manifested by marked increase in serum Cr and BUN levels. Elevated levels of these parameters are usually taken as early signs of renal insult. Allicin treatment decreased serum levels of Cr and BUN when compared to CsA-treated group. This renoprotective effect may be related to its antioxidant effect.
Urine analysis also provides important clues about the functional status of the kidney. In this regard, significantly higher quantity of urine protein was recorded with CsA administration to rats for 28 days. This could be attributed to proximal tubular structural damage observed in our study and consequently defects in reabsorption. The current study showed that CsA treatment produced reduction in GFR, manifested by the progressive reduction in CCr. Allicin administration caused a significant increase in CCr value which reflects the enhanced glomerular function. In addition, it significantly decrease the elevated levels of protein in urine.
In the present study, CsA resulted in a significant increase in serum level of LDH but a significant decrease in level of albumin. This result comes in line with Goksu Erol et al. 33 who detected that CsA caused lower serum albumin levels, probably as a result of its side effect of albuminuria. Allicin administration significantly reduced LDH activity when compared to CsA-treated group. This result agreed with Wang et al. 34 who reported the oral administration of allicin significantly decreased the damage indexes of AST, ALT, and LDH. Also, allicin significantly increased serum albumin level when compared to CsA group.
Oxidative stress stimulates the development of different vasoactive mediators that lead to renal vasoconstriction or reducing the glomerular capillary ultrafiltration coefficient and decreasing the glomerular filtration rate thus affecting renal function directly. In the present study, renal SOD and GSH were significantly decreased and conversely renal MDA was significantly increased in CsA-treated rats.
Allicin administration with CsA showed a significant decrease in MDA content compared to rats treated with CsA alone, a result that agreed with other reported data that demonstrated apparent. 35 This effect may be due to the ability of allicin to scavenge hydroxyl and peroxyl radicals and superoxide anion 36 and inhibit lipid peroxidation. 37 Also, treatment of rats with allicin showed a significant increase in the activity of GSH compared to the CsA-treated group, because it has antioxidant activity. 38 Also, it exerts its effect by increasing the activities of nonenzymatic antioxidant (GSH) and the detoxifying enzyme (glutathione S-transferase (GST)). 37 Allicin may provide the sulfur source required for the synthesis of GSH 39 and so it restores glutathione level and increases the activities of GSH and GST. Allicin administration with CsA resulted in significant increase in the activity of SOD compared to the CsA-treated group. This may be related to its effect as an antioxidant. 38
The current study showed that CsA administration significantly decreased the levels of NOx in kidney tissue homogenate. This result agreed with Chander et al. 40 who showed that CsA caused a significant decrease in tissue nitrite levels. Previous studies have shown that administration of CsA stimulates the production of vasoconstriction factors such as endothelin, thromboxane A2, and angiotensin II. 41 –43 Studies on endothelial cell cultures showed that exposure of cells to CsA results in structural damage, 44 and several in vitro studies reported that ACh-induced vasodilation is impaired in vascular beds of CsA-treated animals, suggesting a deficient endothelial NO synthesis, 45,46 although these findings can also be explained by enhanced generation of free radicals that inactivates NO. 47 Mansour et al. 48 have demonstrated that exogenous supplementation of l-arginine is effective in reducing renal damage induced by CsA, possibly through the NO pathway. In addition, De Nicola et al. 8 have shown that oral supplementation of l-arginine prevents nephrotoxicity induced by chronic supplementation of CsA due to formation of more NO which may enhance vasodilation and consequently reduce the kidney function impairment. This effect was reversed by allicin administration that resulted in significant increase in levels of NO when compared to CsA-treated group.
TNF-α has an early and crucial role in the cascade of pro-inflammatory cytokine production and subsequent inflammatory processes. In this study, CsA-treated rats showed a significant increase in TNF-α level in kidney tissue homogenate. This finding agreed with Abdel-latif et al. 49 who stated that CsA-treated rats exhibited a significant increase in the renal expression of TNF-α which may be due to renal ischemia and vasoconstriction that are commonly associated with CsA administration. Many researchers reported that renal ischemia with or without reperfusion leads to increase in renal TNF-α levels. 50,51 Furthermore, the oxidative stress and lipid peroxidation products possibly serve as activators of transcription factors, leading to induction of gene expression of pro-inflammatory cytokines and release of many inflammatory cytokines including TNF-α. 52 Allicin administration resulted in a significant decrease in levels of TNF-α when compared to CsA-treated group. This could be due to the ability of allicin to suppress the expression of mRNA of TNF-α and due to its antioxidant, anti-inflammatory activity. This result is in agreement with the investigation reported by Bruck et al., 53 who reported that allicin administration (300 mL/mouse/day) had the ability to decrease the elevation in serum level of TNF-α, and the hepatic necroinflammation was much improved.
MOP is a neutrophil and monocyte enzyme that amplifies the reactivity of hydrogen peroxide through the generation of hypochlorous acid, free radicals, and ROS. 54 MOP and its oxidative products play a key role for the enzyme in promotion of lipid peroxidation, protein nitration, and other oxidative modifications. 55 In this study, treatment with CsA increased the levels of renal MPO activity. This finding agrees with Gökçe et al. 56 who reported that the activity of MPO enzyme and MDA levels in the CsA group were significantly increased. Allicin administration significantly resulted in reversing the increase in MPO activity confirming its anti-inflammatory and antioxidant properties.
In the present study, CsA treatment produced a significant increase in responsiveness of urinary bladder rings toward ACh. Afferent arteriole vasoconstriction is considered the main mechanism responsible for the pathogenesis of CsA-induced acute renal injury. 57 This event is mediated by the imbalance of vasoactive factors, that is, an increase of vasoconstrictors (such as angiotensin II and endothelin) and a decrease in vasodilators (such as prostaglandins and NOx) causing vasoconstriction. 57 Generating ROS in the renal system could be suggested as the pathway mediating CsA-induced hyperresponsiveness of the urinary bladder rings toward ACh. Allicin administration significantly decreased the responsiveness of the isolated urinary bladder rings toward ACh. This effect may be due to its ability to reduce ROS through scavenging free radicals.
The histological studies of kidney from CsA-treated rats showed acute drug toxicity with tubular dilatation, vacuolization, and cast formation, however, allicin attenuated these changes.
In summary, we have confirmed that allicin has a protective role against nephrotoxicity induced by CsA exposure. According to our biochemical findings, which were supported by histopathological and sensitivity of urinary bladder rings to ACh, administration of allicin rescued the cells from the effects of CsA. These findings indicate that allicin administration may reduce CsA-induced renal injury. Therefore, we propose that allicin might be a potential candidate agent against CsA-induced nephrotoxicity via its antioxidant and anti-inflammatory properties.
Footnotes
Acknowledgment
The authors acknowledge Dr Mona Abdel Rahim, Urology and Nephrology center, Faculty of Medicine, Mansoura University, Mansoura, Egypt, for providing assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

