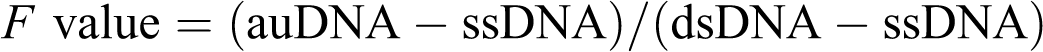Abstract
Cisplatin (CP) is a well-known chemotherapeutic drug that displays dose-limiting nephrotoxicity. In this study, tannic acid (TA), a naturally occurring plant polyphenol, was evaluated for its antioxidant and antigenotoxicity potential against the CP-induced renal oxidative stress and genotoxicity in Swiss albino mice. The mice were given a prophylactic treatment of TA orally at a dose of 40 and 80 mg/kg body weight (b wt) for 7 consecutive days before the administration of a single intraperitoneal (i.p.) injection of CP at 7 mg/kg b wt. The modulatory effects of TA on CP-induced nephrotoxicity and genotoxicity were investigated by assaying oxidative stress biomarkers, serum kidney toxicity markers, DNA fragmentation, alkaline unwinding, micronuclei assay, and by histopathological examination of kidney architecture. CP administration altered the antioxidant levels, enhanced lipid peroxidation, induced DNA strand breaks, and altered the levels of micronuclei among polychromatic erythrocytes (PCEs) significantly (p < 0.001). Pretreatment of TA in mice showed significant (p < 0.001) recovery in antioxidant status, viz., reduced glutathione content and its dependent enzymes, quinone reductase and γ-glutamyl transpeptidase. TA significantly (p < 0.001) reinstated the normal serum levels of blood urea nitrogen (BUN) and creatinine. TA showed strongly inhibited (p < 0.001) micronuclei induction, DNA strand breaks, and DNA fragmentation. Thus, TA as a phytochemical protects kidneys through its antigenotoxic activity and antioxidant potential.
Introduction
The use of chemotherapy in the treatment of cancer has opened new possibilities for improvement in the quality of life of cancer patients. Despite its success, treatment with some of the most effective anticancer drugs shows a number of symptoms of direct toxicity. Additionally, many anticancer drugs have been shown to be mutagenic, teratogenic, and carcinogenic in experimental model systems. Secondary malignant tumors are also known to be associated with several therapeutic treatments. 1 Cisplatin (CP) is a potent chemotherapeutic agent that has gained widespread use against various malignant tumors in different experimental animals 2 and in a variety of human malignancies. 3 Many of the biological properties and effects of CP have been well documented,4,5 with numerous reports indicating that the cellular DNA could be the primary target in its anticancer activity. 6 High doses of CP are more effective for the suppression of cancer. The high-dose CP chemotherapy, however, produces untoward side effects of nephrotoxicity, bone marrow toxicity, gastrointestinal toxicity, ototoxicity, and peripheral neuropathy. 4 The preferential accumulation of CP in kidney tissue contributes to CP-induced nephrotoxicity. 7 CP chemotherapy induces a decrease in plasma antioxidant levels, leading to a failure in the antioxidant defense armory against free radical damage generated by antitumor drugs. 8
During the recent past, considerable attention has been paid into the investigations of naturally occurring agents that are able to stimulate defense mechanisms. 9 Plants have limitless ability to synthesize aromatic substances such as polyphenols, mainly flavonoids, and phenolic acids, which exhibit antioxidant properties due their hydrogen-donating and metal-chelating capacities. 10 Polyphenols are secondary metabolites of plants and are widely distributed in plant-derived foods, such as cereals, legumes, nuts, vegetables, fruits, and in beverages such as green or black tea, fruit juice, and so on. 11 They are second only to flavonoids in terms of their dominance, suggesting that naturally occurring polyphenols have been recently recognized as functionally active molecules, possessing antioxidant, anticancer, antimutagenic properties, as well as exerting protective effects against several other diseases. 12
Tannins are such a class of polyphenols which are reported to possess multiple biological activities including anticancer, 13 antioxidant, 14 and antimicrobial activities. 15 Based on their chemical structures, tannins are further divided into hydrolysable and condensed tannins. 13 The main components of hydrolysable tannins is tannic acid (TA), also referred to as gallotannins. 9 In vitro, TA inhibited the proliferation of various cancer cell lines and induced cancer cell apoptosis. Also, when given in drinking water, TA enhanced the survival rate of mice bearing syngenic tumors. 16 Earlier studies provide evidence that co-treatment of TA with CP increases the nephrotoxicity while the post-treatment prevents it to a certain extent in rats. 17 None of the studies on the effect of pretreatment with TA before CP intoxication in mice has been carried out. Therefore, we found it reasonable to investigate whether pretreatment of TA, being the main hydrolysable tannin component of a variety of dietary elements, has the capacity to ameliorate the nephrotoxicity as well as the genotoxicity of CP in Swiss albino mice.
Materials and methods
Chemicals
Oxidized and reduced glutathione, nicotinamide adenine dinucleotide phosphate reduced (NADPH), hydrogen peroxide (H2O2), dithionitrobenzene (DTNB), bisbenzamide, 1-chloro-2,4-dinitrobenzene (CDNB), glutathione reductase, TA, and proteinase K were purchased from Sigma Chemical Co. (St. Louis, MO, USA). Pharmaceutical grade CP under the brand name ‘Cytoplatin50’; was purchased from Cipla Ltd., India. May-Grünwald, Giemsa, and hematoxylin and eosin (H&E) stains were purchased from Hi-Media Labs, Mumbai. All other chemicals and reagents used were of highest purity and commercially available.
Animals
Eight-week-old adult male Swiss albino mice (20–25 g) were obtained from the Central Animal House Facility of Hamdard University, New Delhi, and were housed in plastic cages in a ventilated room at 25 ± 2°C, 60 ± 10% humidity under a 12-hr light/dark cycle. The animals were acclimatized for 1 week before the study and had free access to standard laboratory feed (Hindustan Lever Ltd., Bombay, India) and water ad libitum. The study was approved by the Committee for the Purpose of Control and Supervision of Experimental Animals (CPCSEA).
Study design and treatment
For the study of biochemical parameters and micronuclei assay, 30 male Swiss albino mice were divided into 5 groups. Group I served as a control and received saline (0.85%) orally for 7 consecutive days and on 7th day, 1 hr after receiving the oral saline dose, the mice received a single i.p. injection of saline (0.85%). Group II served as toxicant group and received saline (0.85%) orally for 7 consecutive days. TA was orally administered at two doses, 40 and 80 mg/kg body weight (b wt), to groups III and IV, respectively, for 7 consecutive days. On the 7th day of pretreatment, a single i.p. dose of CP (7 mg/kg b wt) after oral treatment of TA was given to the animals in groups II, III, and IV. Group V received only higher dose of TA orally for 7 consecutive days and on 7th day 1 hr TA treatment received a single i.p. injection of saline (0.85%) to ensure that Group V received only the higher dose of TA, this was done in order to test that the higher dose did not produce any kind of toxic effects. All the animals were killed after 24 hr of intoxication with CP. The time of killing was based on the preliminary studies. 10 Kidneys and femur bones were removed and processed for performing different assays. A preliminary dose-dependent pilot study was carried out to select an appropriate dose of CP, which produced acute renal failure and genotoxicity but did not suppress cell proliferation, in combination with the highest TA dose. The above-mentioned doses of TA were selected on the basis of preliminary dose-escalation studies to determine the threshold dose producing a measurable response between toxicant- and TA-treated mice (data not shown).
Micronucleus test
For this test, mice were killed 24 hr after treatment with a single i.p. dose of CP (7 mg/kg b wt). For micronucleus assay, bone marrow were collected from femur bones . The mouse bone marrow micronucleus test was carried out according to the method of Schimd. 18 The air-dried slides were stained with May–Grunwald and Giemsa as described by Schimd, made permanent, and coded. A total of 1500–2000 polychromatic erythrocytes (PCEs) and Normochromatic erythrocytes (NCEs) were scored per animal by the same observer for determining the frequencies of micronucleatedPCEs (MnPCEs). To detect possible cytotoxic effects, the PCE:NCE ratio in 200 erythrocytes per animal was calculated. 19 Coded slides were scored with a light microscope at a ×400 magnification.
DNA isolation
The DNA was isolated from the kidney tissue of mice by standard phenol chloroform isoamyl method with some modifications. 20 The amount of DNA was quantitated and its purity was checked by 260/280 nm ratio by adding 2 µl of stored DNA on a Nanodrop.
Gel electrophoresis and DNA fragmentation
The sample was mixed with 10 ml of loading dye (10 mM EDTA [pH 8.0], 1% (weight/volume [w/v]) bromophenol blue, and 40% [w/v] sucrose) preheated to 70°C.The DNA samples were loaded onto a 1.5% (w/v) low melting point agarose gel containing ethidium bromide (EtBr; 0.5 µg/ml). The DNA fragments were separated by electrophoresis at 25 V for 12 hr at 4°C in Tris/borate/EDTA buffer. The DNA was visualized under Gel documentation System (Alpha Innotech, USA).
Alkaline unwinding assay
The procedure used for alkaline unwinding was essentially the same as that outlined by Shugart,
21
with slight modifications. In the alkaline unwinding assay, the rate of transition of double-stranded DNA (dsDNA) to single-stranded DNA (ssDNA) under pre-defined alkaline denaturing condition was proportional to the number of breaks in the phosphodiester backbone and thus was used as a measure of DNA integrity. Bisbenzamide was used as a DNA-binding dye from the fluorescence of which various types of DNA were quantitated. For the fluorescence determination of dsDNA, ssDNA, and partially unwound DNA (au-DNA), three equal portions of diluted DNA sample was prepared. The amount of dsDNA was obtained from the fluorescence of the sample without any treatment; while ssDNA was determined from the sample that had been boiled for 30 min. Fluorescence of the DNA sample which had been subjected to alkaline treatment (pH 12.2) on ice for 30 min provided an estimate of the amount of au-DNA. The fluorescence of initial or dsDNA was determined by placing 100 mM DNA sample, 100 ml sodium chloride (NaCl; 25 mM), and 2 ml SDS (0.5%) in a prechilled test tube, followed by the addition of 3 ml of 0.2 M potassium phosphate (pH 9) and 3 ml bisbenzamide (1 mg/ml). The contents were mixed and allowed to react in the dark for 15 min to allow the fluorescence to stabilize. The fluorescence of the sample was measured using a spectrofluorimeter (Ex: 360 nm, Em: 450 nm). The fluorescence of ssDNA was determined as above but using the DNA sample that had already been boiled for 30 min to completely unwind the DNA. 50 ml sodium hydroxide (NaOH; 0.05 N) was rapidly mixed with 100 ml of DNA sample in a prechilled test tube. The mixture was incubated on ice in the dark for 30 min
22
followed by rapid addition and mixing of 50 ml hydrochloric acid (HCl; 0.05 N). This was followed immediately by an addition of 2 ml SDS (0.5%) and the mixture was forcefully passed through a 21-G needle 6 times. Fluorescence of alkaline unwound DNA sample was measured as described above. Measurement of the alkaline unwound sample was performed in triplicate and the average was reported. The ratio between dsDNA and total DNA (F value) was determined as follows:
Post-mitochondrial supernatant preparation
Kidneys were removed quickly, cleaned free of extraneous material, and immediately perfused with ice-cold saline (0.85% sodium chloride). The kidneys were homogenized in chilled phosphate buffer (0.1 M, pH 7.4) containing potassium chloride (KCl; 1.17%) using a Potter Elvehjen homogenizer. The homogenate was filtered through muslin cloth and was centrifuged at 800 g for 5 min at 4°C by REMI Cooling Centrifuge to separate the nuclear debris. The aliquot so obtained was centrifuged at 12,000 rpm for 20 min at 4°C to obtain post mitochondrial supernatant which was used as a source of enzymes. All the biochemical estimations were completed within 24 hr of killing the animal. 23
Reduced glutathione estimation
One milliliter sample of PMS was precipitated with 1.0 ml of sulfosalicylic acid (4%). The samples were kept at 4°C for 1 hr and then centrifuged at 1200 g for 20 min at 4°C. The assay mixture contained 0.1 ml filtered aliquot, 2.7 ml phosphate buffer (0.1 M, pH 7.4), and 0.2 ml 1,2-dithiobisnitrobenzoic acid (DTNB; 100 mM) in a total volume of 3.0 ml. The yellow color developed was read at 412 nm on a spectrophotometer. 24
Glutathione reductase activity
The reaction mixture consisted of 1.65 ml phosphate buffer (0.1 M, pH 7.6), 0.1 ml EDTA (0.5 mM), 0.05 ml oxidized GSH (1 mM), 0.1 ml NADPH (0.1 mM) and 0.1 ml 10% PMS in a total volume of 2 ml. Enzyme activity was quantitated at 25°C by measuring disappearance of NADPH at 340 nm and was calculated as nmol NADPH oxidized min–1 mg protein–1 using molar extinction coefficient of 6.22 × 103 M–1 cm–1. 25
Glutathione-S-transferase activity
The reaction mixture consisted of 2.5 ml phosphate buffer (0.1 M, pH 6.5), 0.2 ml GSH (1 mM), 0.2 ml CDNB (1 mM), and 0.1 ml of the cytosolic fraction (10%) in a total volume of 3.0 ml. The changes in absorbance were recorded at 340 nm, and enzymatic activity was calculated as nmol CDNB conjugate formed min–1mg protein–1 using a molar extinction coefficient of 9.6 × 103 M–1 cm–1. 26
Glucose-6-phosphate dehydrogenase (G6PD) activity
The reaction mixture consisted of 0.3 ml Tris-HCl buffer (0.05 M, pH 7.6), 0.1 ml nicotinamide adenine dinucleotide phosphate (NADP; 0.1 mM), 0.1 ml glucose-6-phosphate (0.8 mM), 0.1 ml magnesium chloride (MgCl2; 8 mM), 0.3 ml PMS (10%), and 2.1 ml distilled water in a total volume of 3 ml. The changes 27 in absorbance were recorded at 340 nm and enzyme activity was calculated as nmol NADP reduced min–1 mg–1 protein using a molar extinction coefficient of 9.6 × 103 M–1 cm–1.
γ-Glutamyl transpeptidase activity
The reaction mixture in a total volume of 0.1 ml contained 0.2 ml PMS, which was incubated with 0.8 ml of the substrate mixture (containing 4 mM γ-glutamyl p-nitroanilide, 40 mM glycine, and 11 mM MgCl2 in 185 mM Tris HCl buffer, pH 8.25) at 37°C. At 10 min after initiation of the reaction 1.0 ml of trichloroacetic acid (TCA; 25%) was added and mixed to terminate the reaction. The solution was centrifuged and the supernatant fraction was read at 405 nm. The enzyme activity 28 was calculated as nmol p-nitroaniline formed min–1 mg protein–1 using a molar extinction coefficient of p-nitroaniline as 1.74 × 103 M–1 cm–1.
Malondialdehyde formation
The reaction mixture in a total volume of 1.0 ml contained 0.60 ml phosphate buffer (0.1 M, pH 7.4), 0.2 ml homogenate, and 0.2 ml ascorbic acid (100 mM). The reaction mixture was incubated at 37°C in a shaking water bath for 1 hr. The reaction was stopped by adding 1.0 ml 10% TCA. Following the addition of 1.0 ml 0.67% thiobarbituric acid (TBA), all tubes were placed in boiling water bath for 20 min and then shifted to crushed ice bath before centrifuging at 2500 g for 10 min. The amount of malondialdehyde (MDA) formed in each of the samples was assessed by measuring the optical density of the supernatant at 535 nm using spectrophotometer (Perkin Elmer Lamda EZ 201) against a reagent blank. The results 29 were expressed as nmol MDA formed hr–1 g tissue–1 at 37°C using molar extinction coefficient of 1.56 × 105 M–1 cm–1.
Xanthine oxidase activity
The reaction mixture consisted of 0.2 ml PMS that was incubated for 5 min at 37°C with 0.8 ml phosphate buffer (0.1 M, pH 7.4). The reaction was started by adding 0.1 ml xanthine (9 mM) and kept at 37°C for 20 min. The reaction was terminated by adding of 0.5 ml ice-cold perchloric acid (PCA; 10% v/v). After 10 min, 2.4 ml of distilled water was added 30 and centrifuged at 4000 rpm for 10 min and the microgram of uric acid formed minute–1 mg protein–1 was recorded at 290 nm.
Catalase activity
The reaction mixture consisted of 1.95 ml phosphate buffer (0.1 M, pH 7.4), 1.0 ml H2O2 (0.019 M), and 0.05 ml 10% PMS in a final volume of 3 ml. Changes in absorbance were recorded at 240 nm. Catalase activity 31 was calculated as nmol H2O2 consumed min–1 mg protein–1.
Quinone reductase activity
The 3 ml reaction mixture consisted of 2.13 ml Tris-HCl buffer (25 mM, pH 7.4), 0.7 ml BSA, 0.1 ml flavin adenine dinucleotide (FAD), 0.02 ml NADPH (0.1 mM), and 50 μl PMS (10%). The reduction of dichlorophenolindophenol (DCPIP) was recorded calorimetrically at 600 nm and the enzyme activity 32 was calculated as nmol of DCPIP reduced min–1mg protein–1 using molar extinction coefficient of 2.1 × 104 M–1cm–1.
Estimation of blood urea nitrogen
Protein-free filtrate was prepared by adding serum and equal amount of 10% TCA, then the mixture was centrifuged at 2000 rpm and the supernatant was taken. To 0.5 ml of protein-free filtrate, 3.5 ml of distilled water, 0.8 ml diacetylmonoxime (2%), and 3.2 ml sulfuric acid–phosphoric acid reagent (reagent was prepared by mixing 150 ml 85% phosphoric acid with 140 ml water and 50 ml of concentrated sulfuric acid) were added. The reaction mixture was placed in a boiling water bath for 30 min and then cooled to room temperature. The absorbance 33 was read at 480 nm.
Estimation of creatinine
Protein-free filtrate was prepared. To 1.0 ml serum 1.0 ml sodium tungstate (5%), 1.0 ml sulfuric acid (0.6 N), and 1.0 ml distilled water were added. After mixing thoroughly, the mixture was centrifuged at 800 g for 5 min. The supernatant was added to a mixture containing 1.0 ml picric acid (1.05%) and 1.0 ml NaOH (0.75 N). The absorbance 34 was read at 520 nm exactly after 20 min.
Estimation of protein concentration
The protein concentration in all samples was determined by the method of Lowry. 35
Histological examination
The kidneys were quickly removed after killing the mice and were fixed with 10% neutral buffered formalin solution. Histolologic sections were prepared, stained with H&E and then examined under microscope. The examiner of the histological slides was blinded to the protocol regime.
Statistical analysis
Differences between groups were analyzed using analysis of variance (ANOVA) followed by Dunnett’;s multiple comparisons test. All data points are presented as the treatment groups mean ± standard error of the mean (SEM).
Results
Administration of CP to Swiss albino mice caused induction of micronuclei in PCEs with higher frequency, loss of DNA integrity, and modulation of antioxidant defense system significantly. As observed during the study, CP alone (7 mg/kg b wt) led to the modulation of several parameters of oxidative stress, relative to control animals receiving saline only, with concomitant enhancement of its metabolizing enzymes.
As evident from Table 1, CP treatment decreased the kidney GSH content and the activities dependent enzymes glutathione-S-transferase and glutathione reductase significantly (p < 0.001). The TA-pretreated groups, that is, Group III and Group IV showed improvement in GSH content, GST and GR activities as compared to toxicant-only group. Table 2 shows concomitant and significant enhancement in xanthine oxidase levels and renal microsomal membrane lipid peroxidation (p < 0.001) in the CP-only treated group as compared to saline-treated control. A marked reduction was noted in XO levels (p < 0.01) and renal microsomal membrane LPO (p < 0.001) at higher dose of TA as compared to the CP-only group. Whereas, G6PD levels in mice decreased due to CP injection (p < 0.001). TA pretreatment replenished the low expression of this enzyme at both the doses, though more significantly at a higher dose (p < 0.001). Table 3 reveals that CP induced the release of kidney toxicity markers, that is, blood urea nitrogen (BUN) and creatinine into the serum significantly (p < 0.001). However, higher dose of TA significantly (p < 0.001) attenuated the release of both the serum toxicity markers. Gammaglutamyl transpeptidase exhibited marked increase in its activity after CP administration. But higher dose of TA-treated group displayed GGT at levels (p < 0.001) approximate to control. The level of quinone reductase (QR) and catalase enzymes in the toxicant-only group depleted significantly (p < 0.001) as shown in the Table 4, respectively, but was restored in the TA-pretreated groups.
Results of pretreatment of TA on antioxidant enzymes like GSH, GST and GR on cisplatin administration in kidney of Swiss albino mice a

CP: cisplatin, GSH: reduced glutathione, GST: glutathione-s-transferase, GR: glutathione reductase TA: tannic acid, D1: 40 mg/kg b wt, D2: 80 mg/kg b wt, CDNB: 1-chloro-2,4-dinitrobenzene, NADPH: nicotinamide adenine dinucleotide phosphate reduced.
aResults represent mean ± SE of six animals per group. Results obtained are significantly different from control group
(b p < 0.001). Results obtained are significantly different from CP-treated group
(c p < 0.01) and
(d p < 0.001).
Results of pretreatment of TA on antioxidant enzymes like G6PD, XO and LPO on cisplatin administration in kidney of Swiss albino mice a

CP: cisplatin, G6PD: glucose-6-phosphate dehydrogenase, XO: xanthine oxidase, LPO: lipid peroxidation TA: tannic acid, D1: 40 mg/kg b wt, D2: 80 mg/kg b wt, MDA: malondialdehyde.
aResults represent mean ± SE of six animals per group. Results obtained are significantly different from control group
(b p < 0.001). Results obtained are significantly different from CP-treated group
(c p < 0.01) and
(d p < 0.001).
Results of pretreatment of TA on serum kidney toxicity markers BUN, creatinine, and GGT on cisplatin administration in kidney of Swiss albino mice a

BUN: blood urea nitrogen, CP: cisplatin, GGT: gammaglutamyl transpeptidase, NS: results obtained are not significantly different from CP-treated group, TA: tannic acid, D1: 40 mg/kg b wt, D2: 80 mg/kg b wt.
aResults represent mean ± SE of six animals per group. Results obtained are significantly different from control group
(b p < 0.001). Results obtained are significantly different from CP-treated group
(c p < 0.001).
Results of pretreatment of TA on the level of QR, catalase, and DNA strand breaks on cisplatin administration in kidney of Swiss albino mice a
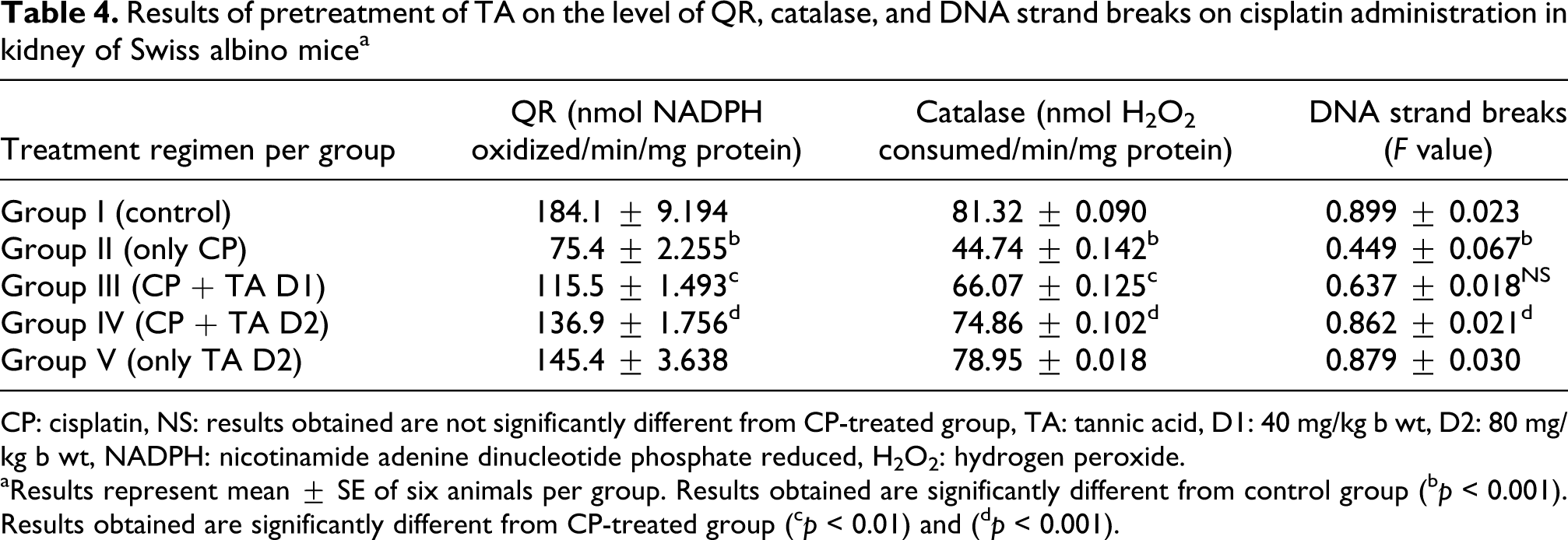
CP: cisplatin, NS: results obtained are not significantly different from CP-treated group, TA: tannic acid, D1: 40 mg/kg b wt, D2: 80 mg/kg b wt, NADPH: nicotinamide adenine dinucleotide phosphate reduced, H2O2: hydrogen peroxide.
aResults represent mean ± SE of six animals per group. Results obtained are significantly different from control group
(b p < 0.001). Results obtained are significantly different from CP-treated group
(c p < 0.01) and
(d p < 0.001).
Figure 1 shows that the CP induced the micronuclei in PCEs at higher frequency (p < 0.001), whereas the pretreatment at higher dose of TA significantly (p < 0.001) decreased the micronuclei induction in PCEs. The estimated PCE:NCE ratio in bone marrow preparations in the Figure 2 shows a statistical increase in hematopoiesis (p < 0.001) as a result of TA pretreatment at higher dose, indicating reversal of the cytotoxic effects caused by CP administration to mice. A simultaneous decrease in F value was noted in the DNA alkaline unwinding assay shown in Table 4, which is a marker for alteration in DNA integrity as evident from agarose gel electrophoresis in Figure 3. The results indicate that there was significant DNA fragmentation due to necrosis only in the toxicant group as compared to the control group, while there was less fragmentation in TA-pretreated groups. During DNA fragmentation assay, DNA damage due to necrosis is estimated in terms of smearing and lack of intact band control; only the TA-treated group showed less smearing and an intact band was also observed.

Results represent mean ± SE of six animals per group. Results obtained are significantly different from control group (***p < 0.001). Results obtained are significantly different from CP-treated group (## p < 0.01) and (### p < 0.001). NS: results obtained are not significantly different from CP-treated group, TA: tannic acid, D1: 40 mg/kg b wt, D2: 80 mg/kg b wt, CP: cisplatin, b wt: body weight.

Results represent mean ± SE of six animals per group. Results obtained are significantly different from control group (***p < 0.001). Results obtained are significantly different from CP-treated group (## p < 0.05) and (### p < 0.001). NS: results obtained are not significantly different from CP-treated group, TA: tannic acid, D1: 40 mg/kg b wt, D2: 80 mg/kg b wt, CP: cisplatin, b wt: body weight.

Agarose (1.5%) gel electrophoresis of DNA fragments in mouse kidney. Lane 1: control; Lane 2: CP alone; Lane 3: CP + TA D1; Lane 4: CP + TA D2; Lane 5: TA D2 alone; and DNA ladder lane: mid-range ladder, TA: tannic acid, CP: cisplatin, D1: 40 mg/kg b wt, D2: 80 mg/kg b wt, b wt: body weight.
Histopathological examination of kidneys in mice showed normal glomerular and tubular histology in control mice both in cortical and medullary region. CP in the toxicant group was found to cause glomerular and peritubular damage which resulted in the invasion of inflammatory cells into the kidney cortical section. The morphological deterioration was characterized by widespread degeneration of tubular architecture, tubular congestion, swelling, and necrosis. In contrast, renal sections obtained from mice that were pretreated with TA at higher dose demonstrated marked reduction in the histological features of renal injury (Figure 4). Pretreatment of TA (40 mg/kg b wt) was also associated with a significant reduction in injury, but this reduction was less marked than in the higher dose TA (80 mg/kg b wt)-pretreatment group (data not shown).

Mice kidney histopathological examination (×400 magnifications). GL: glomeruli, BC: Bowman’;s capsule, NS: necrosis, ICI: inflammatory cell invasion. (A) Control; (B) Cisplatin = 7 mg/kg b wt; (C) CP + TA D1 (40 mg/kg b wt); (D) CP + TA D2 (80 mg/kg b wt); (E) TA D2 only (80 mg/kg b wt), b wt: body weight.
Discussion
CP is a potent anticancer therapeutic drug. The therapeutic effects of CP are significantly improved by dose escalation. However, high-dose therapy with CP is limited by its cumulative nephrotoxicity and neurotoxicity.
36
CP is toxic to the renal proximal tubules.
37
Recently, it has been postulated that oxidative stress and reactive oxygen species (ROS) are involved in the pathogenesis of CP-induced nephrotoxicity.
38
It has been shown that superoxide anion (O2•−), H2O2, and hydroxyl radical (•OH) are involved in CP-induced nephrotoxicity.
37
Thiols such as the sulfur of GSH will bind to the platinum molecule, replacing one of the chloride ions and preventing binding to other cellular nucleophiles.
39
Increased intracellular GSH concentrations correlate with decreased platinum-DNA binding in freshly isolated peripheral blood mononuclear cells.
40
The ROS generated during normal cellular processes are immediately detoxified by endogenous antioxidants like GSH, catalase, GR, G6PD, GST, and so on, but excessive ROS accumulation by CP causes an antioxidant status imbalance, leads to lipid peroxidation, and GSH depletion.
41
Our results in mice renal tissue demonstrated GSH depletion, enhanced GGT and XO activity, and increased lipid peroxidation after CP injection, which is in accordance with earlier findings.
42
The activity of catalase and GR also decreased after CP administration, which in turn decreased the ability of the kidney to scavenge toxic H2O2 and lipid peroxides. Therefore, lipid peroxidation due to CP administration is a consequence of GSH depletion and impaired antioxidant enzyme activities. These observations support the evidence that part of the mechanism of nephrotoxicity in CP-treated mice is related to depletion of GSH and imbalance in antioxidant system. The inhibition of antioxidant enzyme activity and GSH depletion in CP nephrotoxicity and the prevention of this inhibition in mice with prophylactic treatment of TA support the rationale for the use of GSH and other antioxidants to ameliorate CP nephrotoxicity. Significant attenuation of CP-induced apoptosis and renal failure in animal models have resulted from maneuvers such as treatment with the hydroxyl radical scavenger dimethylthiourea, targeted proximal tubular delivery of superoxide dismutase, and pretreatment with
During the recent past, a lot of focus has been given to dietary components. Majority of them being without side effects, thus taking the leading role in the fight against oxidative stress caused by different xenobiotics in humans. One such class of dietary components are the polyphenols found in various plant-derived foods. Polyphenols have been recently recognized as functionally active molecules, possessing antioxidant, anticancer, antimutagenic properties, as well as exerting protective effects against several other diseases. 12 Tannins are such a class of polyphenols which are reported to possess multiple biological activities including anticancer, 13 antioxidant 14 and antimicrobial activities. 15 Earlier studies have shown that TA has potent antioxidant and free radical scavenging activity, 43 suggesting that antigenotoxic and nephrotoxic activity of TA may be associated with its antioxidant activity. Consistent with the previous studies, TA was able to restore antioxidant balance by inducing GR, GST, and GSH possibly by quenching excessive ROS and free radicals generated by CP.
CP requires the GGT activity, a cell surface enzyme, for exerting its nephrotoxicity in kidney proximal tubules. GSH-conjugates of CP are metabolized extracellularly to toxic cysteinyl-glycine-conjugates by the cell surface enzyme GGT. Inhibition of GGT has been shown to block the nephrotoxicity of CP in both rats and mice.44,45 Another study has shown that CP is not nephrotoxic in GGT-knockout mice. 46 Therefore, pretreatment with TA probably downregulates GGT expression on the cell surface, thereby preventing the formation of toxic cysteinyl-glycine-conjugates which are one of the culprits of renal proximal tubular cellular damage. CP administration at a dose of 7 mg/kg b wt resulted in a significant decrease in G6PD activity in the mice kidney as in earlier studies, 47 which maybe due to depletion of GSH. But, TA was able to maintain the normal activity of G6PD in mice perhaps by restoring the normal GSH levels. While investigating the nephroprotective effects of TA in vivo, we found that the administration of TA attenuated CP-induced renal damage characterized by increases in serum BUN and creatinine. CP-induced elevations in the levels of BUN and creatinine as seen in this study have been previously reported in other murine models.48,49 Genomic instability is often considered the hallmark of cancer and it has been shown earlier that CP administration leads to genotoxicity and DNA strand breaks that is believed to result from its interaction with DNA by the mechanism of binding with N 7 of purine bases, forming CP-DNA adducts. 50 All these suggest that CP cytotoxicity and genotoxicity results from the generation of reactive oxygen and by forming DNA adducts. Prophylactic treatment of TA significantly prevented the DNA damage as is obvious from all the parameters of genotoxicity viz micronuclei induction, DNA fragmentation, and so on, possibly by quenching the free radicals generated by CP administration at the above-mentioned dose. It is evident from the present study that TA not only reduced the cellular damage but also suppressed the DNA fragmentation due to necrosis and MnPCEs formation in vivo, which are the hallmarks of genotoxicity in pretreated animals. The PCE:NCE ratio, a mark of cytotoxicity, was also restored to normal in the TA-treated groups.
CP nephrotoxicity primarily causes tubulointerstitial lesions. Mitochondrial swelling and nuclear pallor occur in the distal nephron. 37 Earlier studies in the rats have demonstrated that CP induces acute tubular necrosis of the straight portion of the proximal tubule located at the cortico-medullary junction. 51 In concordance with the results of earlier studies, our study on histological examination revealed major disruption in architecture of straight portion of proximal tubules due to CP toxicity. The toxicity was characterized by widespread degeneration of tubular architecture, tubular congestion, swelling and necrosis, luminal congestion with loss of brush border, and an infiltration of polymorphonuclear neutrophils. CP intoxication showed severe atrophy of glomerulus, which was apparent due to the reduction in its size. Marked dilation of proximal convoluted tubules with slogging of almost entire epithelium due to desquamation of tubular epithelium was evident. Cellular debris in the tubular lumen and increased tissue in the interstitium is also an indication of CP-induced renal necrosis. The changes obtained in the present study run parallel with the earlier report documented. 52 Prophylactic treatment with TA at both the doses protected the architecture of nephron from destruction in both corticular and medullary regions of kidney. Histopathological inspection robustly supported the results of biochemical assays, henceforth confirming the TA as ROS scavenger.
One of the recent study provides evidence that co-treatment of TA with CP increases the nephrotoxicity, while the post-treatment prevents it up to a certain extent. 17 Similar to the results of post-treatment in the former study, our study showed that prophylactic treatment mitigates the nephrotoxicity. The explanation most probably could be that during pretreatment the competitive binding between TA and CP for plasma albumins is less severe than during the co-treatment protocol. The decrease in the free plasma albumin due to the formation of TA-albumin complex may have been compensated by the rush of more albumins into the blood, induced by TA during 7 days of treatment period. CP administered on the 7th day would have got enough albumin molecules to bind with, hence CP-albumin complex was excreted through urine without getting much chance for DNA binding. To conclude, though the mechanism of TA preventing CP nephrotoxicity is complicated, our study hints that pretreatment of TA inhibits the formation of ROS and free radicals generated due to CP in kidneys. Moreover, TA pretreatment decreases the GGT expression on extracellular surface of proximal tubular cells, thereby decreasing the metabolism of CP into a nephrotoxic metabolite, which may further deteriorate the normal physiological processes of kidney. Further investigations at molecular level are warranted, which may support the extrapolation that TA can act as a potent antigenotoxicant and nephroprotective agent when taken as a prophylactic measure in anticipation of toxic insults like CP chemotherapy in humans.
Footnotes
Acknowledgements
The authors are thankful to the Indian Council of Medical Research (ICMR), New Delhi, India, for providing the funds to carry out this research work.
The authors of the present research work do not have any conflict of interest to present this research work.