Abstract
Propolis has a broad spectrum of biological activities; however, whether its essential oils have neuroprotective effects is unknown. In this study, we found that propolis essential oil (PEO) could significantly reverse the anxiety-like behavior of restraint-stressed mice, and has no effect on locomotor activity. Furthermore, PEO significantly decreased the plasma levels of cortisol (CORT), adrenocorticotropic hormone (ACTH) and malondialdehyde (MDA), whereas it increased the activity of superoxide dismutase (SOD) in restraint-stressed mice. These results strongly suggest that PEO has therapeutic effects on anxiety through antagonizing the hyperfunction of hypothalamic–pituitary–adrenal (HPA) axis and improving the ability of antioxidation in brain tissue.
Keywords
Introduction
Anxiety is a substantive public health problem all over the world. Generalized anxiety disorder (GAD) and panic disorder are the two primary classifications of pathological anxiety in humans. The prevalence of GAD in the general population ranges from approximately 2% to 5%, with particular prevalence in certain special populations, such as older adults, patients and adolescence who are associated with potentially stressful challenges. 1 The associated individual and societal burden is substantial, but many of those who could benefit from treatment are not recognized or treated. 2 Due to the side effects of traditional anxiolytic treatment, there has been increasing interest in the use of alternative therapies, including aromatherapy. 3 Several studies have shown that essential oils used in aromatherapy decrease anxiety-related behavior in humans and animals.4,5
Propolis is a resinous material collected by honeybees (Apis mellifera L.) from leaf buds and cracks in the bark of the plants and mixed with wax and bee enzymes. Raw propolis is composed of 50% resin, 30% wax, 10% essential oils, 5% pollen and 5% various organic compounds. It has been reported to have various biological activities, such as antioxidant, antimicrobial, anti-inflammatory and anticancer.6–8 For these reasons, propolis is extensively used in folk medicine, cosmetology and food industry for health foods, beverages and nutritional supplements.9,10 The essential oil from propolis mainly contains terpenoids, aromatic and aliphatic compounds. Previous studies have shown that propolis essential oil (PEO) has antibacterial activities11–13 and inhibits lipid peroxidation.14,15 Interestingly, recent studies have found that propolis has neuroprotective effects on neuronal death, such as retinal ganglion cells, neuronal-like pheochromocytoma cells (PC12), human neuroblastoma cells (SH-SY5Y)16–18 and function on mitigating scopolamine-induced learning and memory impairment in mice. 19 However, whether PEO has therapeutic effects on anxiety is still unknown. In present study, we demonstrated the roles of PEO on anxiety-like behavior in restraint-stressed mice.
Materials and methods
Animals
Male ICR mice weighing 18–22 g were provided by the Shanghai Laboratory Animal Center of the Chinese Academy of Sciences (Certificate of animal quality: Zhong Ke Dong Guan No.003). All animals were housed in a mouse breeding room with air conditioning and an air defecation filtering device. The mice were allowed a standard pellet diet, free access to tip filter water and maintained at 23 ± 2°C, 50–70% humidity, a noise level <50 dB, and with 12-h dark/12-h light cycles. All the conditions were keeping with the Helsinki accord, and the experimental protocol was approved by the Animal Ethics Committee of Zhejiang University.
Drugs and reagents
Brazilian green propolis was obtained from Zhejiang Beewords Apiculture Group Co, Ltd. Mouse cortisol (CORT) and adrenocorticotropic hormone (ACTH) test kit were obtained from KapidBio Lab Calabasas (CA, USA); superoxide dismutase (SOD), glutathione (GSH) and malondialdehyde (MDA) test kits were obtained from Nanjing Jiancheng Bioengineering Institute; and diazepam tablets (DZP) were obtained from Hangzhou Huadong Medicine Group Co, Ltd; all other chemicals and solvents used were of analytical grade.
Extraction of the essential oil
Essential oil was extracted from propolis by microwave-assistant extraction (MAE). The extraction was performed at atmospheric pressure using a multimode microwave reactor 2.45 GHz with a maximum delivered power of 1000 W variable in 10 W increments. Temperature was monitored by an external infrared sensor. In MAE procedure, 20 g propolis was inserted into an extraction vessel and 200 ml of ligarine was added it. The extraction time was 15 min and the extraction temperature 35°C, and the microwave extractor was set at a maximum power. After cooling, the vessel was opened and the supernatant was filtered; then the filtrate was stored in the freezer for 2 h in order to precipitate fixed waxes and oils. After the second filtration, the extract was reduced by rotary evaporation and crude extract of PEO was obtained. The crude PEO was re-extracted by 15 times volume of absolute alcohol in order to eliminate the micro-amount beewax and alcohol-insoluble residue. Finally, the filtrate was reduced by rotary evaporation. The extract was collected, dried under anhydrous sodium sulphate and stored at 4°C until used.
Chemical investigation
Essential oil constituents were analysed by gas chromatography–mass spectrometry (GC-MS) using the Agilent 6890N Network GC system combined with Agilent 5975 B Inert MSD detector (quadrupole) with electron impact ionization (70 eV). A HP-5-MS (5% phenyl methyl siloxane) column (30-m long, 0.25 mm id and 0.25 μm film thickness) was used. Temperature programming of the oven included an initial hold at 80°C for 5 min and a rise to 185°C at 5°C/min held for 5 min and post 270°C for 2 min.
The injector was set at 280°C. The samples were diluted with n-hexane (1/10, v/v) and a volume of 1.0 μl was injected to the GC with the injector in the split mode (split ratio: 1/20). Carrier gas, helium (He), was adjusted to a linear velocity of 1 ml/min. The components were identified based on the comparison of their relative retention time and mass spectra with those of NBS75K library data of the GC-MS system, literature data and standards of the main components.20,21
Stress mouse model
The restraint stress is used in this study as a model of stress which consists of immobilizing the animal in a reduced space. The restraint stress mouse model was described previously. 22 Briefly, after 2-h acclimatization to the testing room, a separate cohort of mice were exposed to 18-h restraint stress using a modified transparent 50 ml plastic conical tube with air holes to increase ventilation.
Grouping and drug administration
Mice were randomly divided into six groups via fasting body weight (n = 10): normal control, model control, low-dose PEO (50 mg/kg), middle-dose PEO (100 mg/kg), high-dose PEO (200 mg/kg) and positive control (DZP 2 mg/kg) group. Normal and model control groups were administrated with vehicle (Tween-80 3% v/v in saline) at the rate of 0.1 ml/10 g body weight. PEO and DZP were dissolved in vehicle before use, and the solution was orally administered to animals at 0.1 ml/10 g body weight for 14 days before exposure to restraint stress.
Behavioral measures
Locomotor activity assay
Locomotor activity was measured in a box by optical means (Opto-Varimex apparatus, Yiyan Science and Technology Development Co., Ltd. Shandong, China). An increase in the number of transitions through the light beams reflects augmented locomotor activity.
Elevated plus maze (EPM) test
As described previously, 23 spatiotemporal measures in the EPM included frequency of entries and duration (measured in seconds) of time spent in the open and closed arms and centre square. Percentage measures of open and closed arms were derived from these. Behaviors scored were frequency of head dipping and rearing. Head-dipping and rearing behaviors were further differentiated by their occurrence on the maze. Any head dips (or rears) that occurred from the closed arms or centre square were classed as protected head dips (or rears), and from this percentage of protected head dips (or protected rears) were derived. The test was performed in the same session immediately after the locomotor activity assay.
Biological assays
Under ether anesthesia, blood was collected in test tubes with heparin sodium (50 U/ml blood) following cardiac puncture, and the brain was quickly removed. Blood samples were centrifuged at 3000 rpm/min for 15 min at 4°C in a refrigerated centrifuge (Sigma Co, Germany) to obtain the plasma, and the levels of CORT and ACTH were determined by ELISA kits. Brain samples were homogenized in chilled 0.01 M phosphate buffered saline (PBS; pH 7.4) using an ULTRA-TURRAX T8 homogenizer (GmbH Co, Germany) and centrifuged at 1000 rpm/min for 10 min at 4°C. A 1% brain homogenate was used to determine the protein concentration using a Coomassie brilliant blue kit with bovine serum albumin as the standard. Content of GSH and MDA and the activity of SOD in brain tissue were also investigated.
Data analysis
Data were presented as mean ± standard deviation (SD). One-way analysis of variance (ANOVA) followed by Dunnett’s t test were used for statistical analyses of the results. The level of statistical significance adopted was p < 0.05.
Results
The major constituents of PEO
To identify the major constituents of PEO, GC-MS analysis was performed. Table 1 showed the 21 constituents with a relative amount more than 1% in a total of 51 identified constituents. Longipinene (24.89%), α-eudesmol (6.94%), β-eudesmol (6.08%), β-caryophyllene (5.27%) and guaiol (5.06%) were the major components of the essential oil.
Chemical composition of PEO analysed by GC-MS

GC-MS: gas chromatography–mass spectroscopy, PEO: propolis essential oil, Rt: retention time.
a Compounds listed in order of elution from a HP-5 MS column.
Effects of PEO on spontaneous locomotor activity test in restraint-stressed mice
To test whether PEO can affect the locomotor activity in restraint-stressed mice, we performed the spontaneous locomotor activity test, a measure for evaluating emotional state of central nervous system in tested mice. As shown in Figure 1, there were no significant difference between the model control group and PEO-treated groups (p > 0.05, Figure 1). However, the spontaneous locomotor activities of positive control group were significantly suppressed by DZP 2 mg/kg (p < 0.05, Figure 1). These results suggested that PEO neither improved nor impaired the spontaneous locomotor activity of restraint-stressed mice.

Effects of propolis essential oil (PEO) on spontaneous locomotor activity test in restraint-stressed mice (n = 10). Results are expressed as mean ± SD. Mice were given a 5-min test 10 min after relief from restraint. Significant differences calculated from two-tailed Dunnett’st test are expressed as *p < 0.05, **p < 0.01, versus corresponding vehicle-treated model control mice after one-way analysis of variance (ANOVA).
Effects of PEO on EPM behavior in restraint-stressed mice
To further test whether PEO has therapeutic effects on anxiety in restraint-stressed mice, EPM behavior assay was performed. As shown inTable 2 andFigure 2 , compared with normal control group, restraint stress produced anxiety-like effects as determined by the decrease in time spent in the open arms (p < 0.01, Table 2), percentage of open arm entries (p < 0.05, Figure 2A) and percentage of time spent in the open arms (p < 0.01, Figure 2B); in addition, percentage of protected head dipping (p < 0.01, Figure 2C) and rearing (p < 0.05, Figure 2D) were significantly higher in restraint-stressed mice than normal mice. However, no significant trends in total arm entries and close arm entries were identified across the model control and normal control groups (p > 0.05, Table 2). 14-day administration with PEO resulted in anxiolytic-like effect on restraint stress mice. Compared with the model control group, low- and middle-dose PEOs could significantly increase the open arm entries (p < 0.01, Table 2) and time spent in the open arms (p < 0.05, p < 0.01, Table 2); increase the percentage of open arm entries (p < 0.05, p < 0.01, Figure 2A) and percentage of time spent in the open arms (p < 0.05, p < 0.01, Figure 2B); all PEO-treated groups significantly decreased the percentage of protected head dipping (p < 0.05, p < 0.01, Figure 2C); middle- and high-dose PEOs significantly decreased the percentage of protected rearing (p < 0.05 and p < 0.01, Figure 2D). The positive drug (DZP 2 mg/kg) was effective in increasing the percentage of open arm entries, percentage of time spent in the open arms, decreasing the percentage of protected head dipping and rearing in restraint-stressed mice (p < 0.01, Figure 2). No significant difference was identified in close arm entries across the control and treatment groups (p > 0.05, Table 2). Taken together, these results suggested that PEO had therapeutic effects on anxiety-like behavior in restraint-stressed mice.
Effects of PEO on elevated plus maze behavior in restraint-stressed mice a

PEO: propolis essential oil; p.o.: by mouth, SD: standard deviation.
a Results are expressed as mean ± SD.
b p < 0.01 versus model control.
c p < 0.05 versus model control.
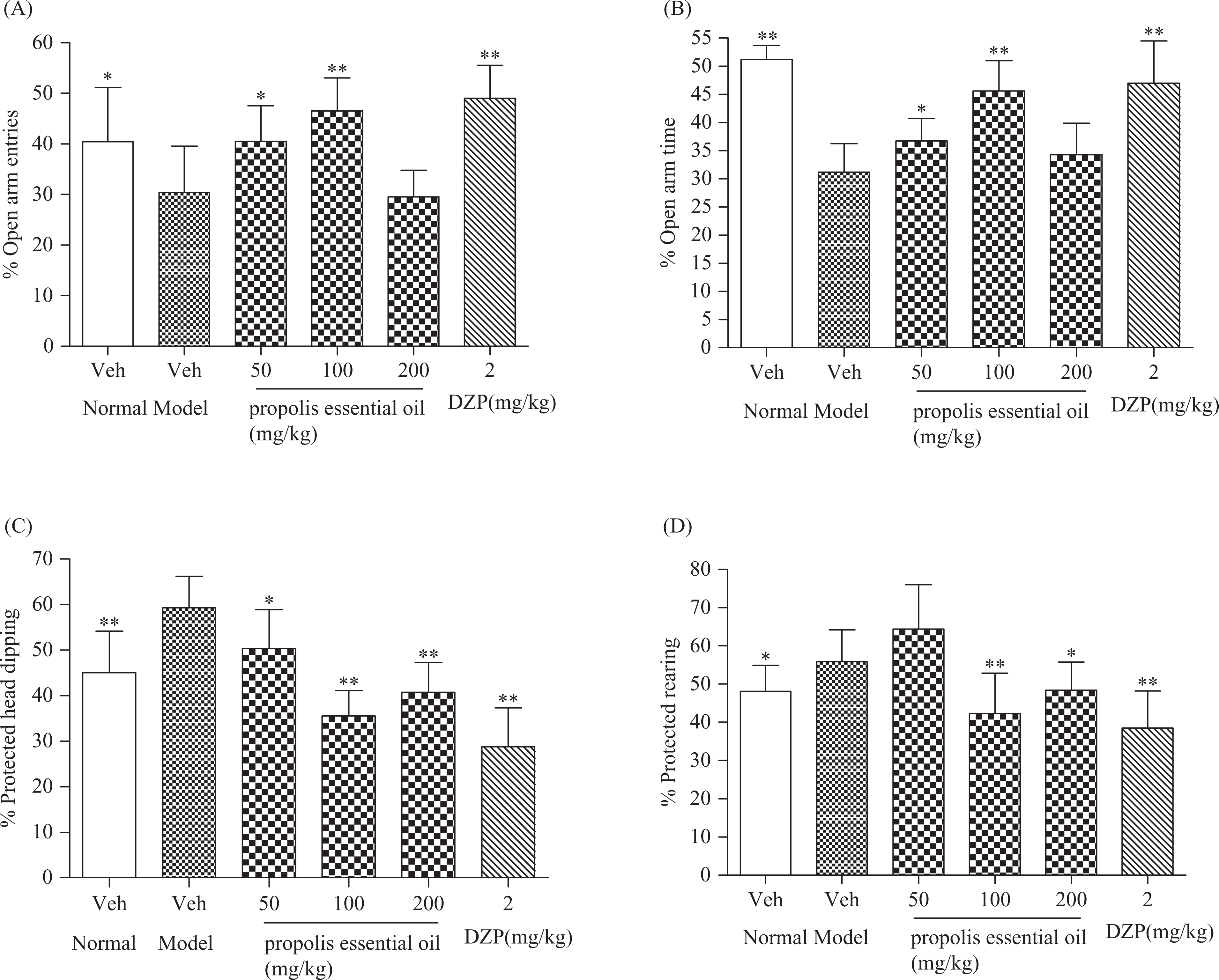
Effects of propolis essential oil (PEO) on elevated plus maze test in restraint-stressed mice (n = 10). Results are expressed as mean ± SD. The following parameters are shown % open arm entries (percentage of entries into open arms with respect to total entries into arms (A)); % open arm time (percentage of time spent in open arms with respect to total time spent in arms (B)); % protected head dipping (percentage of protected head dipping with respect to total head dipping (C)); % protected rearing (percentage of protected rearing with respect to total rearing (D)). Mice were given a 5-min test 30 min after relief from restraint. Significant differences calculated from two-tailed Dunnett’s t test are expressed as *p < 0.05, **p < 0.01, versus corresponding vehicle-treated model control mice after one-way analysis of variance (ANOVA).
Effects of PEO on plasma CORT, ACTH level in restraint-stressed mice
Since the activation of hypothalamic–pituitary–adrenal (HPA) axis has an important role in anxiety, we next tested whether PEO had therapeutic effects on anxiety through antagonizing the hyperfunction of HPA axis. As shown inTable 3 , plasma CORT and ACTH levels in restraint-stressed mice were significantly increased than normal control mice (p < 0.01 and p < 0.05, Table 3). However, the treatment with PEO significantly decreased the level of CORT and ACTH in restraint-stressed mice (p < 0.01 and p < 0.05, Table 3) and the positive drug (DZP 2 mg/kg) also decreased the CORT level in restraint-stressed mice (p < 0.01, Table 3) but had no significant influence on ACTH level (p > 0.05, Table 3). These results suggested that PEO could decrease the plasma CORT, ACTH levels in restraint-stressed mice.
Effects of PEO on ACTH and CORT in restraint-stressed mice a
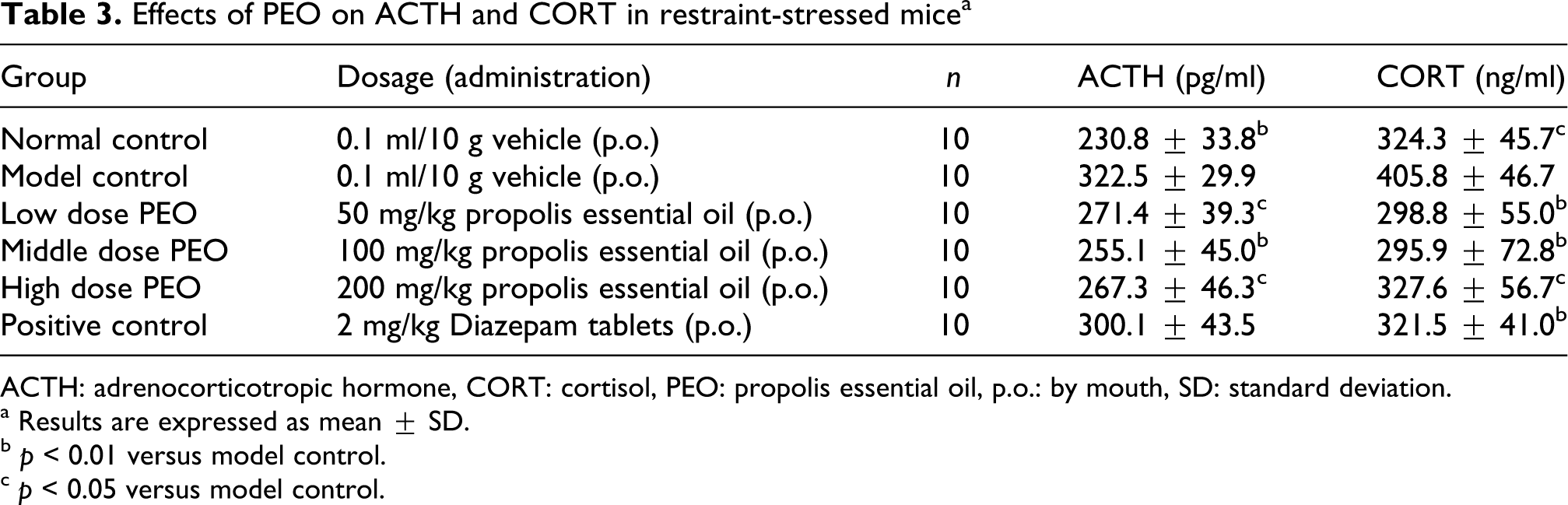
ACTH: adrenocorticotropic hormone, CORT: cortisol, PEO: propolis essential oil, p.o.: by mouth, SD: standard deviation.
a Results are expressed as mean ± SD.
b p < 0.01 versus model control.
c p < 0.05 versus model control.
Effects of PEO on MDA, GSH and SOD levels in brain tissue of restraint-stressed mice
As shown in Table 4, restraint stress produced an increase in MDA level and decreases in GSH and SOD levels in brain tissue compared with normal control mice (p < 0.01, Table 4). The treatment with all doses of PEO were effective in mitigating the level of MDA (p < 0.05, Table 4); high dose of PEO significantly improved the activity of SOD (p < 0.05, Table 4); and there was no significant effect of PEO on GSH in the brain tissue of restraint-stressed mice (p > 0.05, Table 4). Positive drug (DZP 2 mg/kg) significantly decreased the level of MDA (p < 0.05, Table 4) but showed no effect on SOD activity (p > 0.05, Table 4); and significantly decreased GSH level in the brain tissue of restraint-stressed mice (p < 0.05, Table 4).
Effects of PEO on MDA, SOD and GSH in brain tissue of restraint-stressed mice a

GSH: glutathione, MDA: malondialdehyde, PEO: propolis essential oil, SOD: superoxide dismutase, p.o.: by mouth, SD: standard deviation.
a Results are expressed as mean ± SD.
b p < 0.01 versus model control.
c p < 0.05 versus model control.
Discussion
There is evidence that exposure to an acute stressor can result in sustained increase in anxiety-like behavior. 24 Some researchers reported that a period of acute restraint stress could increase anxiety-like behavior in three well-established behavior tests, the EPM, the defensive withdrawal paradigm, and the light–dark box test.25,26 Consistent with these previous studies, in the present study, we found that 18-h restraint stress induced an anxiety-like state measured by EPM test. The activities on spontaneous locomotor activity test were not changed significantly, indicating that the locomotor activity was not affected by stress procedure and the anxiogenic effect after stress protocols was indeed related to anxiety and not to the locomotor activity of the mice. These specific effects were accompanied by hyperactivity of the HPA axis with an increase in plasma level of CORT and ACTH and modifications in oxidative status in the brain tissue. Thus, we use this animal model to evaluate the anxiolytic effect of PEO and underlying pathway.
Interestingly, we found that PEO at a dose of 100 mg/kg can readily mitigate anxiety-like behavior, inhibit HPA axis hyperactivity and the lipid peroxidation in brain tissue. DZP is a clinic medicine that has been shown to improve anxiety state in a number of animal model systems. Compared with the positive control group of DZP, the PEO-treated groups had the comparative effect on the mitigate anxiety in EPM test. However, in spontaneous locomotor activity test PEO showed no significant effect on locomotor activities, which indicated the ability of the oil to lessen anxiety without causing sedation. Furthermore, we also evaluated the levels of ACTH and CORT in plasma as well as the content of MDA, GSH and SOD in brain tissue, and the data support their effects on the mitigating anxiety.
As the composition of PEO may be different depending on geographical locations and plant sources, we listed the main chemical components present in this PEO sample (Table 1). Aromatic carboxylic acids and terpenoids were the main components identified in the essential oil. Terpenoids show various biological and pharmacological activities, including antioxidative, anti-inflammatory, antitumor and antibacterial effects. 27 Recently, increasing evidence have suggested that great amount of terpenoids in essential oil have neuroprotective effects in many models of neurodegenerative diseases in vitro and in vivo,28–31 the mechanisms of their neuroprotection are complicated and associated with multi-pharmacological effects, such as antioxidant activity, anti-inflammatory activity, inhibition of excitotoxicity, inhibition of cell cycle progression and protection of mitochondrial dysfunction occurring in ischemia.32,33
Several studies have provided evidence for an association between HPA axis functioning and psychiatric problems.34,35 In stressful situations, the hypothalamus secretes corticotrophin-releasing hormone, which stimulates the pituitary gland to secrete ACTH. Subsequently, ACTH is released, which causes the adrenal glands to produce CORT. Changes in CORT concentrations influence immunity, metabolism, growth, reproduction, depressive/anxiety and other important physiological processes. Consistent with previous studies, in present study, the results in hormone level suggested that PEO significantly inhibited HPA axis hyperactivity in restraint-stressed mice to mitigate increasing plasma ACTH and CORT levels. This effect may be attributed to its complex components. Some researchers reported that the essential oils from certain aromatic plants that had anxiolytic activities were related to the inhibition on HPA axis hyperactivity. Saiyudthong and Marsden had examined the acute effects of bergamot oil on anxiety-related behavior and CORT level in rats and revealed that bergamot oil exhibited anxiolytic-like effect via attenuated HPA axis activity. 36 Ceccarelli et al. reported that long-term exposure of lemon essential oil significantly inhibited anxiety and pain with decreasing CORT level. 37 In this study, we also found PEO had a selective effect of inhibition on HPA axis hyperactivity, which may result in reversing the anxiety induced by restraint stress. However, the actual mechanism has not been clearly known.
Neurodegenerative diseases such as anxiety are known to eventually be the result of neuronal cell death by many kinds of cause-induced excitotoxicity, oxidative stress, inflammation and apoptosis. 38 Oxidative stress has gained special attention recently in psychiatric medicine and has been reported to be associated with anxiety. One previous study suggested that neurotic and anxious female students tend to be exposed to oxidative stress 39 and the other clinical trial also indicated raised level of MDA in patients with increasing affective disorders. 40 In the present study, restraint stress caused significant oxidative damage as indicated by a raise in lipid peroxidation and depletes GSH and SOD activities. PEO treatment significantly attenuated lipid peroxidation and partially restored SOD activity in brain tissue of restraint-stressed mice. Thus, these results strongly suggested the activity of PEO on mitigation of anxiety in restraint-stressed mice may act partially through antioxidation properties.
In summary, PEO has therapeutic effects on anxiety, and this effect could be attributed to the inhibition of HPA axis hyperactivity in restraint-stressed mice and antioxidation. Further studies should elucidate the components of PEO and the exact site to achieve their potential anxiolytic effects.
Footnotes
Acknowledgments
We acknowledge the great support from the Center of Analysis and Measurement of Zhejiang University for the GC-MS analysis of essential oil.
This work is supported by the grant from Chinese Ministry of Agriculture (Project number: NYCYTX-43) and Zhejiang Provincial Natural Science Foundation of China (Project number: R3090332).
