Abstract
Dried leaves of oleander were orally given at a single dose of 500 mg/kg body weight to 20 clinically healthy male chickens. Clinical signs of toxicosis began to appear about 1 h after receiving the oleander and included hypersalivation, vomiting, diarrhea, deep depression, and sudden death. Also, hyperemia in the combs and wattles was obviously seen. Electrocardiograms (ECG) were repeatedly recorded at 30 min intervals. ECGs findings included increasing the QRS duration in some birds and various kinds of arrhythmias. Bradycardia was the most frequently detected finding (30.43%). During necropsy, there were congestion and hemorrhages in the visceral organs particularly in heart, liver, kidney, and lung. Histopathology revealed myocardial cell necrosis with hyperemia and hemorrhage, severe diffuse pulmonary congestion and edema, severe renal congestion and hemorrhage with tubular necrosis, and coagulative necrosis of hepatocytes with hyperemia and hemorrhage. There were also congestion, mild epithelial necrosis and desquamation with infiltration of mononuclear inflammatory cells in the proventriculus of all birds. There was also mild to moderate congestion in the intestines with scattered necrosis of surface enterocytes. The lack of information about the toxicity of oleanders in poultry was the main cause for this study. The results suggest that chickens appear to respond to oleander poisoning in a manner similar to other species.
Introduction
Nerium oleander (common oleander) and Thevetia peruviana (yellow oleander) are potentially lethal plants after ingestion for human and animals. All parts of these plants are toxic. Oleander (Nerium oleander) is an evergreen perennial shrub originating from Mediterranean and is widely cultivated in tropical and subtropical regions as an ornamental plant. The plant grows rapidly and often used for highway barriers, road sides, and other areas that require screening from noise and pollution. Oleander is cultivated throughout Iran and particularly in eastern and southern provinces. All parts of oleander either fresh or dried are toxic and contain a mixture of cardiac glycosides of cardenolides. Oleandrin and neriine are the most potent of those glycosides.1,2 Oleander is one of the most toxic and lethal plants in birds. Accidental and/or experimental cases of oleander poisoning have been described in several species, including cattle,3,4 sheep,1,5,6 goats,7,8 camels, 9 horses, 10 donkeys, 11 cats,12,13 dogs, 14 monkeys, 15 turkeys, 7 geese, 16 canaries, 17 ducks, 18 and budgerigars. 19 The constituents of oleander responsible for exerting cardiotonic effects are the cardiac glycosides, which are of particular importance in the toxicity of the plant. The mechanism of action involves the inhibition of the plasma membrane Na+/K+-ATPase, which leads to alteration in the intracellular potassium, sodium, and calcium levels. 2 Fast-growing broiler chickens are very sensitive and mortalities due to heart-related conditions are observed predominantly in that age group. There has been little discussion about N. oleander intoxication in broiler chickens. The objective of this study was to determine the clinical and pathological features of oleander intoxication in broiler chickens.
Materials and methods
Initially, in a pilot project, 10 birds were used. Dried oleander powder (oleander leaves were dried at room temperature and ground to powder) and was orally administered to the chickens as an aqueous suspension using a syringe (gavage) in doses of 100, 300, 700, and 1000 mg/kg body weight. The amount of 500 mg/kg of oleander was determined as appropriate dose for experiment based on pilot study and experiments in other birds. Twenty clinically healthy male chickens (42-days old; Ross 307 strain) with an average weight of 2162 ± 36.9 g were used for study. In those birds, poisoning was induced by dried oleander powder (500 mg/kg). Electrocardiography was done (KENZ ECG 106.Model: SZK/K106-003 21.6V. 600 mAh), using a limb leads, at −15 as the baseline, and at 15, 30, 60, 90, 120, 150, 180, 210, and 240 min after oleander administration. For this purpose, alligator clips were attached to the skin at the base of the right and left wings and gastronemius muscle of the right and left limbs as described elsewhere20,21 (Figure 1 ). All ECGs were standardized at 1 mV = 10 mm, with speed of 50 mm/s. The ECGs were evaluated for heart rate, rhythm, and also abnormal patterns. Clinical findings were recorded and necropsy of chickens was also performed immediately after death and gross pathology of organs was recorded. For histological examination of lesions, tissue samples were collected and fixed in 10% neutral buffered formalin, and processed for routine histology. Tissue sections were stained by hematoxylin and eosin. 22 Some relevant data were checked for normality before performing analysis. All analyses utilized parametric statistical methods. All data were analysed using SPSS for Windows, version 16.0. In order to compare the mean control values of QRS interval (before oleander administration) with the mean values of the same parameter measured in intoxicated chickens on 15, 30, 60, 90, 120, 150, 180, 210, and 240 min after oleander administration, repeated ANOVA measure and LSD post hoc test were used. Necessary descriptive statistics and paired t test were used to express arrhythmia. The level of significance for each test was at p < 0.05. The experiment was approved by the animal welfare committee of the school of Veterinary Medicine of Mashhad.

The ECG recording in chicken.
Results
Clinical and ECG findings
Clinical findings were vomiting, diarrhea, restlessness, and incoordination, limb paresis, hypersalivation, recumbency and death that began about 1 h after the administration of oleander (Figure 2 ). Sudden death was observed, without any clinical signs in some birds that were very active and excited. Hyperemia in the sclera, combs, and wattles was observed. Some neurological symptoms in poisoned birds include turning the neck-to-around were observed. Seven of 20 birds died within 3 h after administration of oleander. One bird died during lavage of oleander. All intoxicated chickens died within 3 to 24 h after receiving oleander (the mean time for death was 370 ± 59 min).

Hypersalivation in intoxicated chicken.
The mean and standard deviation of heart rate in healthy chickens before oleander administration were 380 ± 40.82 and the mean and standard deviation of heart rate before death was 303.16 ± 37.72. In the healthy chickens (before taking the toxic dose), due to high heart rate, in most cases the P wave was mixed with previous T wave (as PT form) and the Q wave was not found in the ECGs (lead II). The S wave was higher than R wave in the majority of leads and the T wave was observed positive in some leads (I, II, III, and AVF) and negative in leads of aVR and aVL. Decreasing of the amplitude of S and T waves, and changing the direction of RS complex and T waves were observed in some traces after experimental poisoning with oleander. In some oleander-fed birds, significant increase of QRS duration was observed. The mean and standard deviation of QRS interval in these cases was significant in 90, 120, 150, 180, 210, and 240 min after oleander administration (0.032 ± 0.004 and 0.042 ± 0.007 s, respectively, before and after intoxication). Also ECG tracing of intoxicated chickens revealed sinus bradycardia, second degree AV blocks, ventricular premature beats, sinoatrial blocks, sinus arrhythmia, ventricular tachycardia, and ventricular fibrillation (Figure 3 ).

(a) Bradycardia. (b) Ventricular tachycardia. (c) Sinoatrial block. (d) Ventricular fibrilation. (e) Ventricular premature beat. (f) (Ectopic) ventricular premature beat. (Unipolar limb leads, 1 mV = 10 mm, 50 mm/s).
Pathology
During necropsy, congestion and hemorrhages in the visceral organs such as heart, liver, and lungs in all chickens were seen. Histopathological examination revealed myocardial cell necrosis with hyperemia and hemorrhage (Figure 4 ), severe diffuse pulmonary congestion and edema (Figure 5 ), severe renal congestion and hemorrhage with tubular necrosis (Figure 6 ), and coagulative necrosis of hepatocytes with hyperemia and hemorrhage (Figure 7 ). There were also congestion, mild epithelial necrosis, and desquamation with infiltration of mononuclear inflammatory cells in the proventriculus of all birds. There was also mild to moderate congestion in the intestines with scattered necrosis of surface enterocytes.

Myocardial cell necrosis with hyperemia and hemorrhage. Hematoxylin and eosin, ×320.
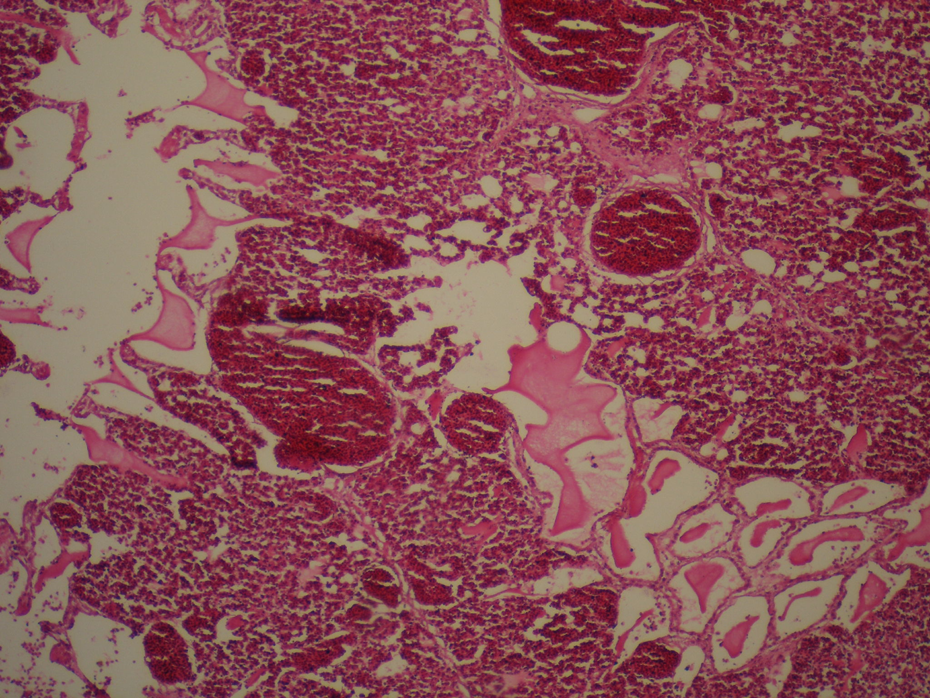
Severe diffuse pulmonary congestion and edema. Hematoxylin and eosin, ×160.

Severe renal congestion and hemorrhage with tubular necrosis. Hematoxylin and eosin, ×320.

Coagulative necrosis of hepatocytes with hyperemia and hemorrhage. Hematoxylin and eosin, ×320.
Discussion
Table 1 shows the frequency and type of dysrhythmias in poisoned chickens. Results showed that among all ECG lead systems, lead II, because of its high amplitude and more clarity of waves, is more appropriate in chickens. In contrast, in lead I, traces were either isoelectric or had very low amplitude.
Types of arrhythmia and their frequency in intoxicated chickens

Clinical, ECG, and pathological findings showed that the plant is highly toxic in broiler chickens and they are generally consistent with those reported in turkeys, 7 gees, 16 sheep, 1 goats, 7 cattle, 23 and human. 24 Different types of cardiac arrhythmias were observed in oleander intoxicated chickens. In some birds, the heart rate was decreased considerably upto 120–170 bpm.
Various cardiac arrhythmias such as AV blocks ventricular tachycardia and ventricular fibrillation were recorded in the ECGs of chickens. Developments of those arrhythmias are due to the direct and indirect effects of cardiac glycosides of oleander on the heart tissue particularly on the cardiac conduction system. 2 Cardiac glycosides may affect the atrioventricular node causing a decrease in electrical conduction resulting in AV blocks.23,24 In the present study, although 30.43% of cases showed obvious bradycardia, in 17 of 19 cases (89/4%), decrease in heart rate was observed. On the other hand, the canaries have been found very sensitive to oleander toxicity so that they died within minutes (about 5 min) after 120–700 mg of plant administration for each bird without any gross postmortem lesions. 17
The histopathological findings of the present study showed some degrees of cardiomyopathy in intoxicated chickens. Myocardial cell necrosis with hyperemia and hemorrhage may also have a rule in the development of cardiac arrhythmias. Mechanism of cardiomyopathy is not determined in cardiac glycoside toxicosis. 25 The pathological findings are consistent with earlier observations, which showed similar cardiomyopathy in cattle, 23 sheep, 1 and goats. 7 Some extracardiac changes were also observed in lung, kidney, liver, proventriculus, and intestines. The extracardiac lesions have also been found in previous reports.1,7,23 Pulmonary lesions may be produced by vascular endothelial damage and acute heart failure. The etiology of renal tubular necrosis in oleander toxicosis is unidentified. Tubular cells of kidneys are probably exposed to much amounts of oleander toxins due to their excretion from this organ. 7 The coagulative necrosis of hepatocytes and mild epithelial necrosis in the proventriculus and scattered necrosis of surface enterocytes are probably direct effects of the toxins on the vascular endothelial bed.
The median lethal dose (LD50) of oleander as well as biological half-life of its cardiac glycosides has not been determined in chickens. The lethal dose of the plant for chickens was greater than sheep and goats (4.54 times)1,7,25 and calves (10 times). 23 These findings suggest that in general, poultry are not very sensitive to oleander than other species. And so in comparison with other species, turkey poults are not very sensitive to oleander poisoning with 250 mg/kg. 7 Rodents are also insensitive to cardiac glycosides of N. oleander. 2 Low sensitivity of some animals may be related to the low affinity of their cardiac Na+/K+-ATPase towards cardiac glycosides. 2
Considerably, more work should be done in evaluation of Nerium intoxication such as determination of serum cardiac glycoside levels. This is important to notice that oleander poisoning is not restricted to N. oleander and acute yellow oleander (T. peruviana) poisoning is frequently reported in humans.2,24
Footnotes
Acknowledgements
The authors thank Dr Monjezi and Dr Hoseini for their assistance in this project.
Funding
The authors gratefully acknowledge the financial support for this work that was provided by the Vice Chancellor for Research at Birjand University.
