Abstract
Cyanate and its active form isocyanate are formed mainly in the process of nonenzymatic urea biodegradation. Cyanate is capable of protein S- and N-carbamoylation, which can affect their activity. The present studies aimed to demonstrate the effect of cyanate on activity of the enzymes implicated in anaerobic cysteine metabolism and cyanide detoxification and on glutathione (GSH) level and peroxidative processes in the kidney. In addition, we examined whether a concomitant treatment with lipoate, a dithiol that may act as a target of S-carbamoylation, can prevent these changes. The studies were conducted in Wistar rats. The animals were assigned to four groups, which received injections of physiological saline, cyanate (200 mg/kg), cyanate (200 mg/kg) + lipoate (100 mg/kg) and lipoate alone (100 mg/kg). The animals were killed 2 h after the first injection, the kidneys were isolated and kept at −80°C until biochemical assays were performed. Cyanate inhibited rhodanese (TST) and mercaptopyruvate sulfotransferase (MPST) activity, decreased GSH level and enhanced peroxidative processes in the kidney. All these changes were abolished by cyanate treatment in combination with lipoate.
Introduction
End-stage renal failure (ESRF) patients suffer from defective metabolism and decreased glomerular filtration, which leads to accumulation of different uremic toxins in the body. The concentration of urea, the main product of protein metabolism, is used as an indicator of uremic toxemia.1,2 Uremic toxins include also the products of nonenzymatic and enzymatic urea biodegradation, i.e. the isomers isocyanate/cyanate (NCO−/OCN−)3–5 (Figure 1 ).
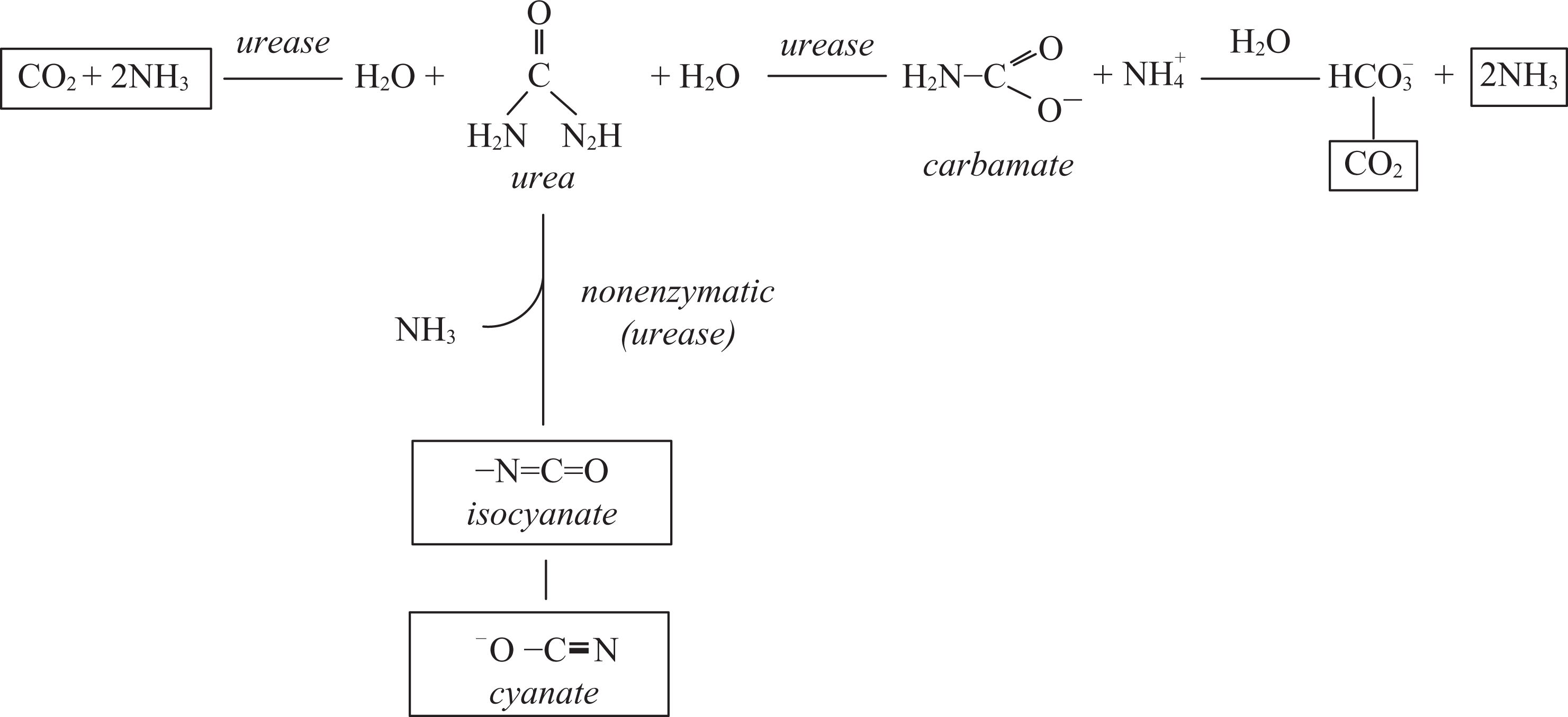
Urea breakdown. Hydrolytic breakdown to ammonia and carbon dioxide catalyzed by urease. Nonenzymatic (in aqueous environment) and enzymatic elimination reaction yielding isocyanate (NCO-) and cyanate (OCN−).
Under physiological conditions, isocyanate shows a capacity to react reversibly with –SH groups of proteins and non-protein thiols yielding carbamoylthiols (reaction 1). It can also react irreversibly with N-terminal –NH2 groups of proteins and α and ϵ amino groups of amino acids, mostly lysine yielding ϵ-carbamoyllysine (homocitrulline; reaction 2).6,7

Studies in biological systems indicated that isocyanates were characterized by a higher activity for –SH groups. 8 It suggests that S-carbamoylation reactions can be yet another type of reversible covalent modification of –SH groups, and ESRF patients are at particular risk of this transformation. The processes of S- and N-carbamoylation of proteins introduce changes in their structure and charge, which can lead to suppression of activity of many proteins.9–11
Our previous studies have documented a drop in plasma sulfane sulfur level in ESRF patients, accompanied by a decrease in total concentration of thiols and activity of 3-mercaptopyruvate sulfotransferase (MPST), an enzyme engaged in sulfane sulfur formation and transfer.12,13 Compounds containing sulfane sulfur (a reactive sulfur atom occurring in 0 or −1 oxidation state) play an important role in cyanide detoxification to thiocyanate (SCN−) catalyzed by sulfotransferases (Figure 2 ).14–16

Metabolism of cysteine to sulfane sulfur compounds catalyzed by: cysteine aminotransferase (CT) to 3-mercaptopyruvate; cystathionase (CST). MPST: 3-mercaptopyruvate sulfotransferase, TST: rhodanese.
Based on the above-described observations, we expected that the decrease in total thiol concentration in erythrocytes and the decrease in sulfane sulfur level in plasma of ESRF patients can be related to an increased concentration of cyanate (OCN−), which is an uremic toxin (Figure 2). In addition to cyanate, the levels of cyanide and its detoxification product thiocyanate (SCN−) are elevated in ESRF patients and in smokers.17,18 Cyanate can also be formed via thiocyanate (SCN−) oxidation catalyzed by eosinophil peroxidase or myeloperoxidase.19,20
It indicates that thiocyanate (SCN−) on the one hand is a product of CN− detoxification, catalyzed by sulfotransferases and with sulfane sulfur (S*) participation, and on the other can be also a precursor of toxic cyanate (Figure 2). Hence, there are close links between cyanide, thiocyanate and cyanate in ESRF patients and in smokers.
Since oxidative stress in plasma is aggravated in ESRF patients, we also investigated whether cyanate contributes to it, as one of the uremic toxins. Our earlier studies demonstrated that in addition to its strong antioxidant potential, lipoate could also stimulate the formation of sulfane sulfur-containing compounds in vivo in the liver and kidney. 21 Since ESRF patients suffer from chronic inflammation due to enhanced oxidative processes, efficient antioxidant defense is crucial in these patients. Therefore, lipoate has been suggested to play a role of an antioxidant and a protectant from cyanate action. In order to authenticate this suggestion, we performed in vivo studies on the effect of cyanate alone and in combination with lipoate on the activity of sulfotransfereases, concentration of sulfane sulfur and glutathione (GSH) and intensity of prooxidative processes in Wistar rats. We focused on the kidney for the studies because it is characterized by the highest level of sulfane sulfur and cysteine, and because ESRF is a cause of increased plasma cyanate levels. 22
Materials and methods
Animals

The first group received 3 injections of 0.9% NaCl at 30 min intervals.
The second group received first lipoate (50 mg/kg), then 0.9% NaCl after 30 min and again lipoate (50 mg/kg) 30 min later. The third group first received 0.9% NaCl, then potassium cyanate (200 mg/kg) after 30 min and again 0.9% NaCl 30 min later. The forth group (the combined treatment of lipoate and KCNO) was administered lipoate (50 mg/kg), then KCNO (200 mg/kg) after 30 min and again lipoate (50 mg/kg) after 30 min.
Animals were sacrificed 2 h after the second injection, the kidneys were isolated and stored at −80°C until experiments were performed.
The doses of cyanate and the time of cyanate action were determined according to the procedure described by Tor-Agbidye et al. 23 Doses of lipoate were determined in accordance with the article of Smith et al. 24
Biological material
The tissues were weighed and homogenized in 4 mL of the phosphate buffer, pH 7.4 per 1 g of the tissue.
Chemicals
Potassium cyanide (KCN), dithiothreitol, p-phenylenodiamine, N-ethylmaleimide (NEM), β-nicotinamide adenine dinucleotide reduced form (NADH), 5,5’-dithio-bis-2-nitrobenzoic acid (DTNB), NADPH, mercaptopyruvic acid sodium salt and lactic dehydrogenase (LDH), potassium cyanate (KCNO) and α-lipoic acid sodium salt were provided by Sigma Chemical Co. (St. Louis, Missouri, USA). Formaldehyde, ferric chloride (FeCl3), thiosulfate and all the other reagents were obtained from the Polish Chemical Reagent Company (P.O.Ch, Gliwice, Poland).
Methods
Activity of rhodanese (TST; thiosulfate: cyanide sulfotransferase, EC 2.8.1.1)
25
was determined using the method of Sörbo, based on sulfane sulfur transfer from thiosulfate to cyanide with thiocyanate (rhodanate – SCN−) formation. Thiocyanate forms a colored complex with Fe3+ ions showing maximum absorbance at 460 nm. Determination of 3-mercaptopyruvate sulfotransferase (MPST)
26
involves two steps: MPST catalyzes sulfur transfer from 3-mercaptopyruvate to sulfate (IV) yielding pyruvate and thiosulfate; Pyruvate (PV) is reduced to lactate by lactic dehydrogenase in the presence of NADH, which is oxidized to NAD+. This method utilizes a difference in absorbance of NADH and NAD+ at 340 nm, which is a measure of the amount of pyruvate formed in MPST-catalyzed reaction. Cystathionase activity was determined by the method of Matsuo and Greenberg
27
L-Homoserine is a substrate of this reaction and pyridoxal phosphate (PLP) is a coenzyme. α-Ketobutyric acid (KB) formed from homoserine is determined with the use of 3-methyl-2-benzothiazolinone hydrazone (MBTH). Sulfane sulfur (S*) level was determined by a cold cyanolysis method
28
It consists in nucleophilic attack of cyanide on sulfane sulfur-containing compounds: thiosulfate, polysulfides, persulfides etc. These compounds react quantitatively with cyanide in an alkaline solution at a temperature of 10−25°C. Thiocyanide formed in this reaction reacts with Fe3+ ions yielding ferric thiocyanate. Formaldehyde stabilizes the complex by reacting with cyanide excess. Determination of glutatione
29
To determine GSH level, the whole GSH is transformed into the reduced form with the use of NADPH and glutathione reductase. GSH determination is based on the reduction of 5,5’-dithiobis-(2-nitrobenzoic) acid by –SH groups of the tripeptide to 2-nitrobenzoic acid having a characteristic yellow color. Absorbance of the colored product is measured at 412 nm. Determination of protein content
30
The determination is based on the reaction of protein peptide bonds and aromatic amino acid residues with Folin-Ciocalteau reagent containing a mixture of phosphotungstic and phosphomolybdic acids. Reaction occurs in an alkaline environment in the presence of copper ions. Protein bound with copper ions reduces the above acids to respective oxides. Determination of lipid peroxidation (MDA)
31
The determination is based on the reaction of malondialdehyde (end-product of lipid peroxidation) with thiobarbituric acid (TBA) in an acidic environment at a temperature of 100°C. The colored product of the reaction shows maximum absorbance at 535 nm. Determination of reactive oxygen species (ROS) level
32
The method based is on fluorescence measurement (Ex 488, Em 525 nm) of 2,7-dichlorofluorescein (DCF) formed via 2,7-dichlorohydrofluorescein (DCFH) oxidation by ROS. DCFH is generated by enzymatic deestrification of the substrate 2,7-dichlorohydrofluorescein diacetate. The concentration of 2,7-dichlorofluorescein formed during a 30-minute incubation of the homogenate with the substrate at 37°C was read from a standard curve. The influence of cyanate on rhodanese activity in vitro.
Rhodanese was pre-incubated with various cyanate concentrations (final): 0, 0.5, 1.0, 2.0 mM at room temperature for 10 min. Residual enzyme activity was measured in the assay mixture containing 0.25 mg/mL of rhodanese, 50 mM thiosulphate, 47.5 mM sodium buffer pH 7.4 and 71.5 mM cyanide, incubated in a final volume of 525 µL for 5 min. The reaction was stopped by addition of formaldehyde. Reaction product, i.e. thiocyanate, formed coloured complex with ferric ions, which absorbance was measured spectrophotometrically at 460 nm.
Statistical analysis
The results are presented as the mean plus/minus standard deviation (SD) of the mean for each group. Statistically significant differences between groups were calculated using two-way ANOVA, followed (if significant) by an LSD-test.
Results
The effects of cyanate on anaerobic sulfur metabolism and prooxidative processes were evaluated following administration of potassium cyanate (200 mg/kg) in Wistar rats (see Materials and methods).
Cyanate administration was observed to significantly decrease renal rhodanese and 3-mercaptopyruvate sulfotransferase activities but did not influence γ-cystathionase activity (CST) or sulfane sulfur (S-S) level in the kidney (Table 1 ). Cyanate alone also significantly increased the concentration of reactive oxygen species (Figure 3 ) and malondialdehyde (MDA) in the kidney (Figure 4 ) and decreased glutathione level (Figure 5 ), thereby exhibiting a prooxidative action. Lipoate treatment alone significantly elevated sulfane sulfur level (Table 1) and GSH concentration vs saline group (Figure 5). In vitro studies confirmed that cyanate dose-dependently inhibited rhodanese activity (Figure 6 ).
The influence of cyanate, liponate and the combined treatment with cyanate and lipoate on the sulfotransferases activities and sulfane sulfur level in the rat kidney
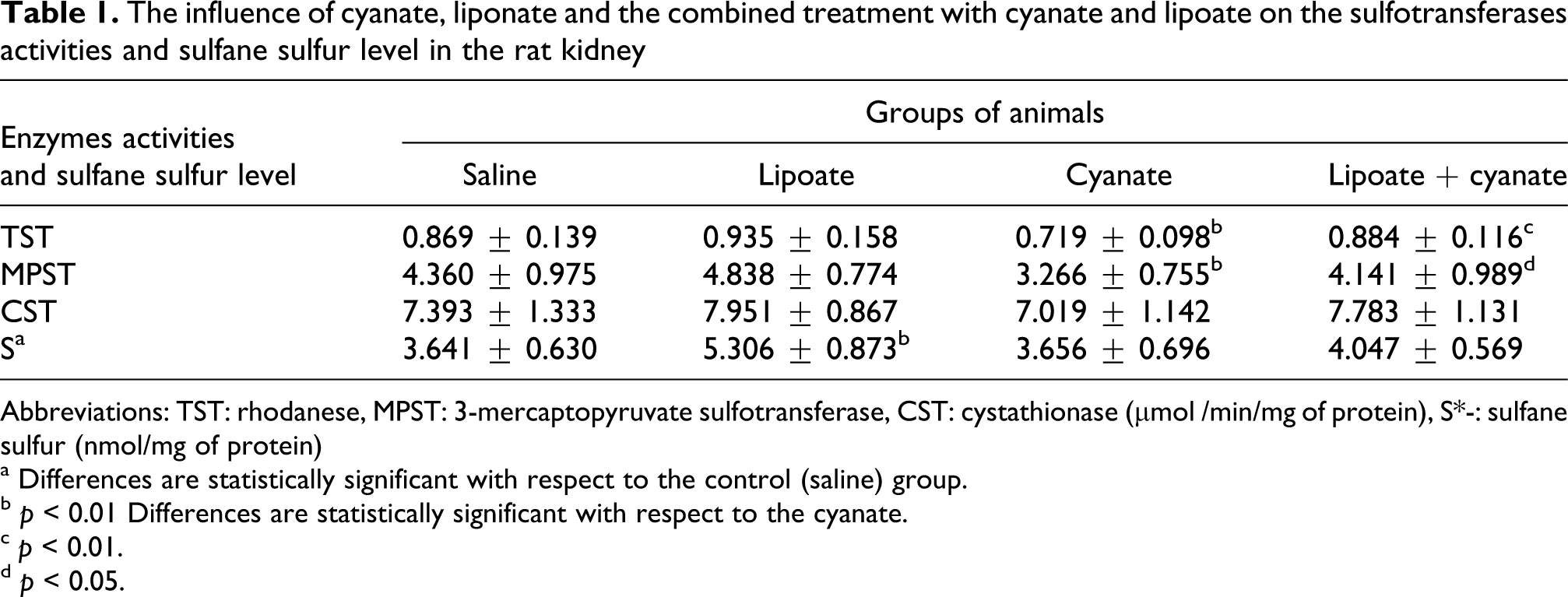
Abbreviations: TST: rhodanese, MPST: 3-mercaptopyruvate sulfotransferase, CST: cystathionase (µmol /min/mg of protein), S*-: sulfane sulfur (nmol/mg of protein)
a Differences are statistically significant with respect to the control (saline) group.
b p < 0.01 Differences are statistically significant with respect to the cyanate.
c p < 0.01.
d p < 0.05.

The influence of cyanate (C), liponate (L) administrated alone and in combination (LC) on the ROS level in the rat kidney. *Statistically significant difference between saline (S) group and experimental groups. *p < 0.05 *** p < 0.001, ▵▵▵ statistically significant difference between experimental cyanate (C) and LC group. Each group contained 7 rats. Assay was performed in triplicate.

The effect of cyanate (C), liponate (L) and the combined treatment with cyanate and lipoate (LC) on the malondialdehyde (MDA) level in the rat kidney. *Statistically significant difference between saline (S) group and experimental group (p < 0.05), ▵statistically difference between cyanate (C) and experimental group (p < 0.05). Each group consisted of seven rats. Assay was performed in triplicate.
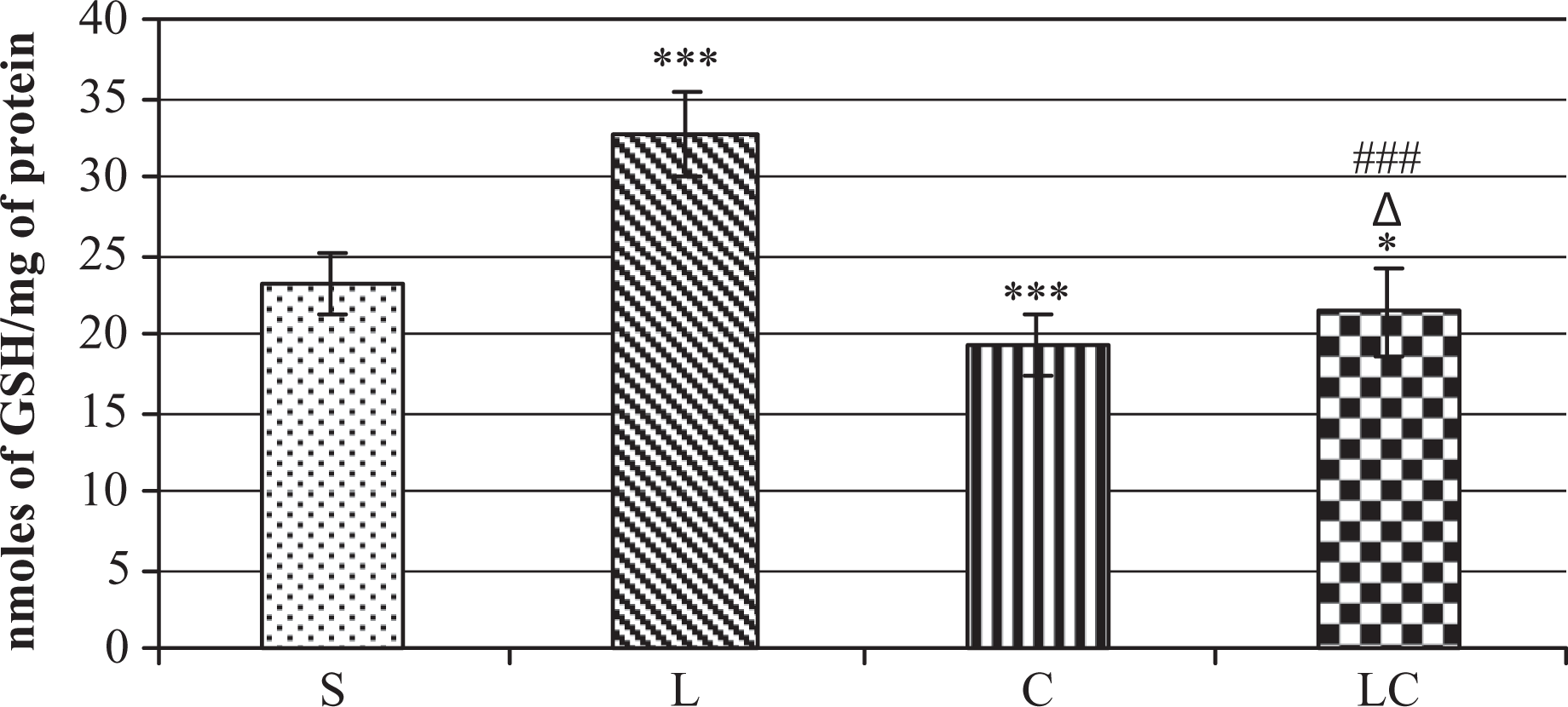
The influence of cyanate (C), liponate (L) and the combined treatment with cyanate and lipoate (CL) on the glutathione (GSH) level in the rat kidney. *Statistically significant difference between saline (S) group and experimental groups *p < 0.05), *** p < 0,001, Δstatistically significant difference between cyanate (C) and experimental group (Δ p < 0.05), ###statistically significant difference between cyanate (L) and experimental group (p < 0.001). Each group contained seven rats. Assay was performed in triplicate.

The effect of various cyanate concentrations: 0, 0.5, 1.0, 2.0 mM on the rhodanese (0.25 mg/ml) activity in vitro. SCN: thiocyanate.
The animals treated with a combination of cyanate (200 mg/kg b.w.) and lipoate L (50 mg/kg, given twice; LC group) showed a significant drop in ROS and MDA level vs cyanate alone group (Figures 3 and 4). The combined treatment with cyanate and lipoate (LC) increased also TST and MPST activity vs cyanate alone group (Table 1) and corrected cyanate-induced lowering of GSH (Figure 5). It indicates that lipoate administered jointly with cyanate can correct the prooxidative cyanate action and activity of sulfane sulfur-metabolizing and cyanide detoxifying enzymes in the kidney.
Discussion
The in vivo studies demonstrated that cyanate-induced inhibition of activities of rhodanese (TST) and 3-mercaptopyruvate sulfotransferase (MPST) was not accompanied by a drop in sulfane sulfur level or activity of γ-cystathionase, the main enzyme involved in its biosynthesis. Both TST and MPST are sulfotransferases responsible for the reduced sulfur transport to different acceptors, including CN− with SCN− formation. It indicates that the cyanate-induced TST and MPST inhibition in the kidney and other organs can lead to distorted cyanide detoxification and its excessive accumulation. It has been confirmed by reports indicating an increased cyanide concentration in erythrocytes of ESRF patients before hemodialysis (HD; i.e. when plasma cyanate concentration is the highest). 17 Theoretically, it might be expected that this should be accompanied by a drop in plasma SCN− level. However, the studies of Hasuike et al. showed that although plasma SCN− concentration dropped slightly after HD in comparison with the same patients before HD, the level of this anion still remained at the elevated level. It was probably connected with an inadequate efficiency of SCN− removal by hemodialysis, which is the only way of removal of this anion from plasma of ESRF patients. 18
It has been shown that thiocyanate (SCN−), a product of cyanide detoxification, is a strong atherogenic factor.3,17 In smokers, SCN− ions were demonstrated to dose-dependently enhance lipid peroxidation and to increase oxidized low-density lipoproteins (ox LDL) fraction.33,34 It has also been suggested that prooxidative action of SCN− can be consequent to its potential of being oxidized to cyanate and to the following enhancement of carbamoylation of proteins. It is corroborated by the observations that OCN− could promote formation of cholesterol deposits and could induce protein carbamoylation, thereby leading to development of inflammatory foci and atheromatous plaques. 3 LDL carbamoylated in vitro (cLDL) have been reported to cause multiple proatherosclerotic biological effects, including diminished LDL receptor recognition, smooth muscle cell proliferation and endothelial cell apoptosis.20,35 The studies of Asci did not confirm cLDL contribution to apoptosis induction but increased injury to endothelial cells in HD patients. 36 All in all, cyanate can promote protein carbamoylation and oxidative processes. Therefore, the following routes may be involved: cyanate can be formed either from urea or via SCN− oxidation, SCN− can be generated during CN− detoxification, catalyzed by sulfotransferases (TST and MPST), and these enzymes are inhibited by cyanate.
Our studies did not show a cyanate-induced decrease in cystathionase (CST) activity or any change in sulfane sulfur level (Table 1). It means that cyanate impaired neither renal routes of cysteine generation from methionine nor sulfane sulfur metabolism. The present studies documented prooxidative action of cyanate in the kidneys, which was reflected by the acceleration of peroxidative processes and S-carbamoylation of GSH or cysteine, a thiol amino acid, rate-limiting for GSH biosynthesis. 6 It is corroborated by the decrease in GSH concentration, caused by its carbamoylation, and the decrease in glutathione reductase activity in isolated rat hepatocytes. 37 Rhodanese is an enzyme participating in sulfane sulfur transfer to different acceptors, including CN−, thereby causing its detoxification to SCN−. The enzyme possesses four –SH groups in its structure, among which one is located in its active center. The cyanate-induced inhibition of rhodanese activity can occur due to S-carbamoylation and N-carbamoylation. Lipoate administered in combination with cyanate showed the ability to correct rhodanese and MPST activity in the kidney and to alleviate OCN−-induced oxidative stress (ROS and MDA). The protective effect of the dithiol lipoate can be attributed as well to its strong antioxidant properties as to protection of biomolecules against carbamoylation. Lipoate’s ability to cause recovery of rhodanese activity can also be attributable to the fact that lipoate (as demonstrated in vitro) can be an acceptor of sulfane sulfur from the rhodanese active center.38,39
In conclusion, the studies presented in this paper have demonstrated that cyanate-alone treatment induced the following effects in the kidney: (1) inhibition of TST and MPST activities, (2) decrease in GSH level (3) increase in MDA and ROS levels.
The increase in ROS concentration observed in rats after cyanate injections can be partially attributed to the inhibition of the respiratory chain by CN−, the level of which probably rises due to sulfotransferases blockade. Moreover, the decrease in the concentration of the main cellular antioxidant GSH cannot be compensated for by sulfane sulfur because of the fact that its concentration is not changed by cyanate injection. For these reasons, in our studies, cyanate showed mostly prooxidant action.
The studies have also shown restoration of sulfotransferases activities and peroxidative processes inhibition after treatment with cyanate in combination with lipoate. Among numerous antioxidants which can prevent this process, lipoate is the most beneficial not only because it is ‘an antioxidant of antioxidants' that modulates activity of other antioxidants but also as a factor restoring sulfotransferases activity, thereby lowering the level of toxic cyanide. It indicates that lipoate can efficiently prevent cyanate toxicity.
This observation is promising for ESRF patients since lipoate could play a double important role in these patients, namely it could be an efficient antioxidant defense and a protectant against cyanate toxicity.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not for-profit sectors.
