Abstract
Methyl parathion (MP) is a high venenosus insecticide. It has been used in pest control of agriculture for several years. The present study is performed to investigate the protective effect of sodium aescinate (SA) on lung injury induced by MP. Forty male Sprague-Dawley rats are randomly divided into five groups, with 8 animals in each group: control group, MP administration group, MP plus SA at doses of 0.45 mg/kg, 0.9 mg/kg and 1.8 mg/kg groups. Acetylcholinesterase (AChE) activity and nitric oxide (NO) level in plasma, myeloperoxidase (MPO) activity, NO level, and antioxidative parameters in lung tissue are assayed. Histopathological examination of lung is also performed. The results show that SA has no effect on AChE. Treatment with SA decreases the activity of MPO in lung and the level of NO in plasma and lung. The level of malondialdehyde in lung is decreased after SA treatments. SA increases the activities of superoxide dismutase, glutathione peroxidase and the content of glutathione in lung. SA administration also ameliorates lung injury induced by MP. The findings indicate that SA could protect lung injury induced by MP and the mechanism of action is related to the anti-inflammatory and anti-oxidative effect of SA.
Introduction
Organophosphate (OP) compounds are the most extensively used insecticides worldwide. The poisoning with these compounds is an important environmental problem for developing countries.1,2 Methyl parathion (O-O-dimethyl-O-p-nitrophenyl phosphorothioate; MP), an organophosphorus compound that may only be used lawfully as an insecticide on agricultural crops, has received attention recently as a consequence of its illegal use as a pesticide in private homes and other buildings in several states of the United States of America. 3 Acute organic phosphorus poisoning (AOPP) can manifest three different phases of toxic effects, namely, acute cholinergic crisis, intermediate syndrome and delayed neuropathy. The well-known effect of the OP is inhibition of acetylcholinesterase enzyme (AChE), which causes the accumulation of acetylcholine (ACh) in the body. However, AChE inhibition does not explain all the symptoms of OP intoxication.4–6 It is reported that lung injury, including thickening of the alveolar-capillary membrane, degeneration in alveoli, alveolar congestion, hamorrhage, neutrophil infiltration or aggregations and so on, does occur in AOPP. 7 Recent studies indicated that mechanisms other than AChE inhibition may be involved in the progression of AOPP, including oxidative stress, mitochondrial energy metabolism impairment,8,9 microcirculation disturbance,10–12 and acute inflammation.13–15
Escin is one of the main bioactive constituents of Aesculus hippocastanum, which is a plant distributed all over the world because of its excellent resistance to environmental conditions. Escin is widely used in clinical therapy because of its anti-oedematous, anti-inflammatory, and anti-oxidative effects. It can not only suppress traumatic brain injury in rats 16 but also inhibit brain ischemia injury-induced apoptosis in rats. 17 However, there are few reports about the effect of escin on lung injury induced by phosphate pesticide. Sodium aescinate is the sodium salt of escin. The present study focuses on evaluating the effect of sodium aescinate on lung injury induced by AOPP.
Materials and methods
Chemicals and instruments
MP (80%, w/w) was obtained from Shandong Dacheng Co, Ltd (Zibo, China) and the lot number is 20080814; SA was from Shandong Luye Pharmaceutical Co., Ltd (Yantai, China) and the lot number is 200809164; acetylcholinesterase (AChE), nitric oxide (NO), myeloperoxidase (MPO), malondialdehyde (MDA), superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), glutathione (GSH), and protein level test kits were purchased from the Institute of Jiancheng Bioengineering (Nanjing, China) and the lot number is 20100403. All other chemicals and reagents used in this study were of analytical grade.
The TDL-40B refrigerated centrifuge was obtained from Shanghai Anting instrument factory (Shanghai, China). Synergy HT Microplate Reader was from Bio-TEK Corporation (Winooski, Vermont, USA). The FJ-200S high-speed tissue homogenate machine was from Shanghai specimen and model factory (Shanghai, China).
Animals and treatment
Forty male Sprague-Dawley rats weighing 220 ± 20 g were provided by the Experimental Animal Center of Shandong Engineering Research Center for Natural Drugs (Yantai, China), and the certificate number was 20030020. All experimental procedures carried out in this study were performed in accordance with the Guidelines for the Care and Use of Laboratory Animals of Yantai University. The rats were kept with free access to food and water on a 12 h light/dark cycle. They were housed in plastic cages and randomly divided into five groups with eight animals in each group: control group, MP administration group, MP plus SA at a dose of 0.45 mg/kg group, MP plus SA at a dose of 0.9 mg/kg group, and MP plus SA at a dose of 1.8 mg/kg group. 18 Rats of the MP and MP + SA groups received MP at a single dose of 15 mg/kg intragastrically to establish the acute MP poisoning rat model. 19 The animals of MP + SA groups were treated with SA via the tail vein at 2.5 h after MP administration while the animals of the other groups were treated with normal saline in equivalent volumes. All the animals were anesthetized with chloral hydrate (300 mg/kg, i.p.) at 24 h after MP administration. Five milliliters heparinized blood (1% heparin, 100 μL) was collected from abdominal aorta, and then the animals died of blood loss under anesthesia. The lungs were excised and cut into two for each immediately and washed with chilled normal saline. One was fixed in paraformaldehyde (4%, diluted in 0.1 mol/L phosphate buffer solution, pH 7.4). The other was weighed and homogenated.
Histopathologic observation
The histopathologic observation was performed as the method described previously. 19 In short, lung was fixed by paraformaldehyde (4%), paraffin-embedded lung samples were cut into 4 μm thick sections, deparaffinized in xylene, and rehydrated through a series of decreasing concentrations of ethanol. Sections were stained with hematoxylin-eosin (H&E). Pathological observation of tissue was performed under light microscopy.
Biochemical analysis
Blood samples were drawn into heparinized tubes for biochemical analysis. After immediate centrifugation (2500 g for 10 min at 4°C), the plasma was stored at −80°C until biochemical analysis.
The lungs were weighed and homogenized using a homogenizer in ice-cold normal saline (1/9, w/v) at a speed of 5000 rpm/min (15 s × 5). The homogenate was then centrifuged at a speed of 2500 g for 10 min at 4°C, and the supernatants obtained were stored at −80°C until further use. 19
According to the instruction of test kit, the standard curve was made first. Then the AChE activity 19 in plasma, NO level in plasma and NO, MDA and GSH levels 20 in lung homogenate supernatants, SOD and GSH-Px activities 19 in lung were determined, respectively.
Statistical analysis
One-way ANOVA was used to analyze the significant differences within the different groups. The comparison between two groups was determined by Student’s unpaired t-test, using SPSS 11.5 statistical software, p < 0.05 was accepted as indicating a statistically significant difference between groups. All data in this study were expressed as mean values ± SD.
Results
Effect of SA on the pathological changes in lung of rats
The effect of SA on the lungs of rats was determined at 24 h after MP challenge by histochemical staining with H&E. Normal single layer of squamous epithelium, thin layer of connective tissue, and no inflammation change in the lung tissues were observed in the control group (Figure. 1A). A number of inflammatory cells had migrated into the alveolar spaces and interstitial spaces in MP group. MP administration also caused the destruction of epithelial and endothelial cell structure, alveolar congestion, and hemorrhage (Figure. 1B). In contrast, SA significantly ameliorated the pathological changes induced by MP administration, manifested by less alveolar septal thickening, less inflammatory cell infiltration, and less alveolar congestion (Figure 1C, D, and E).

Effect of sodium aescinate (SA) on the pathological changes in lung of rats (haematoxylin and eosin staining, magnification ×400). (A) Microscopic appearance of lung tissue in the control group. (B) Microscopic appearance of lung tissue in the methyl parathion (MP) group. (C) Microscopic appearance of lung tissue in the SA 0.45 mg/kg group. (D) Microscopic appearance of lung tissue in the SA 0.9 mg/kg group. (E) Microscopic appearance of lung tissue in the SA 1.8 mg/kg group. The arrows indicate neutrophils infiltration and alveolar hemorrhage.
Effect of SA on AChE activity and NO level in plasma after MP administration
The AChE activity was inhibited significantly after MP administration. SA has no effect on the reduction of AChE activity; compared with control group, NO level in MP group significantly increased. SA suppressed the elevation of NO level in plasma (Table 1 ).
Effect of sodium aescinate (SA) on plasma acetylcholinesterase (AChE) activity and nitric oxide (NO) level after methyl parathion (MP) administration a
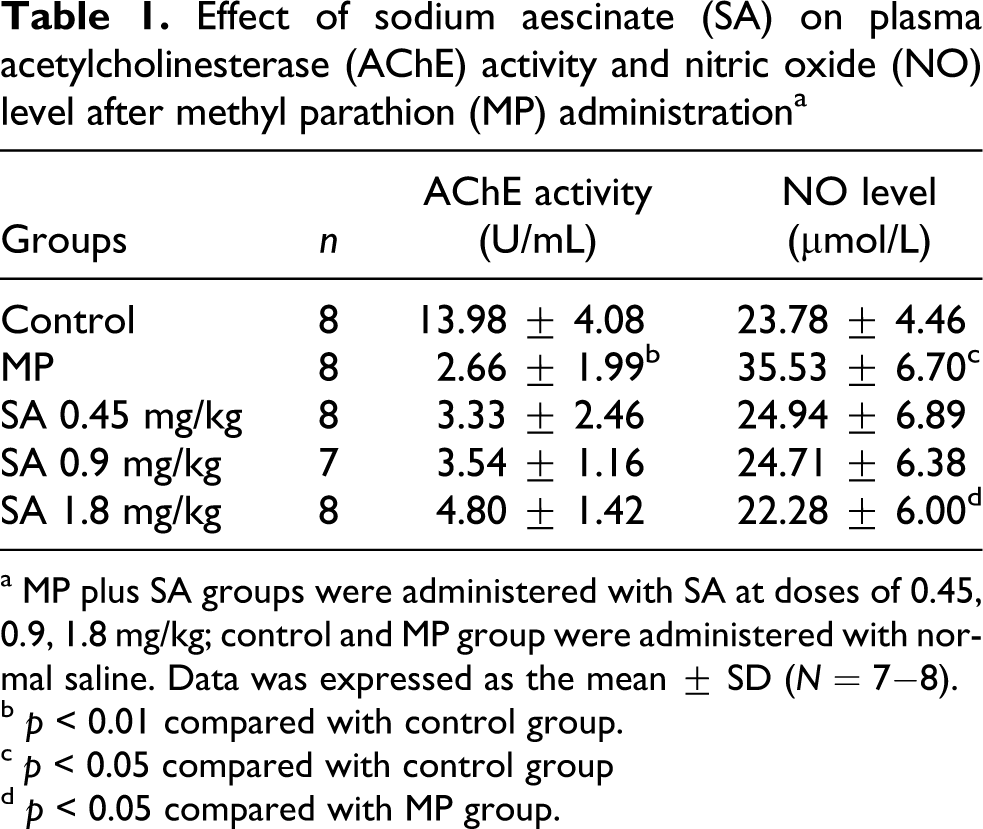
a MP plus SA groups were administered with SA at doses of 0.45, 0.9, 1.8 mg/kg; control and MP group were administered with normal saline. Data was expressed as the mean ± SD (N = 7−8).
b p < 0.01 compared with control group.
c p < 0.05 compared with control group
d p < 0.05 compared with MP group.
Effect of SA on MPO activity and NO level in lung after MP administration
The activity of MPO increased markedly after MP administration. SA (1.8 mg/kg) treatment obviously decreased MPO activity (p < 0.05). The NO content in MP group significantly increased. Compared with control group, SA suppressed the elevation of NO content in lung tissue (Table 2 ).
Effect of sodium aescinate (SA) on myeloperoxidase (MPO) activity and nitric oxide (NO) level in lung after methyl parathion (MP) administration a

a MP plus SA groups were administered with SA at doses of 0.45, 0.9, 1.8 mg/kg; control and MP group were administered with normal saline. Data was expressed as the mean ± SD (N = 7−8).
b p < 0.01 compared with control group.
c p < 0.05 compared with control group.
d p < 0.05 compared with MP group.
Effect of SA on MDA level and SOD, GSH-Px activities, GSH level in lung after MP administration
MP administration led to an increase in the level of MDA. Furthermore, antioxidative parameters analysis showed that administration of MP resulted in a significant decrease in the activities of SOD, GSH-Px, and the level of GSH. However, treatment with SA attenuated the levels of MDA, GSH, and the changed activities of SOD, GSH-Px (Figures 2–5).

Effect of sodium aescinate (SA) on malondialdehyde (MDA) level in lung. SA treatment group was administered with SA at doses of 0.45, 0.9, 1.8 mg/kg; Control and methyl parathion (MP) group was administered with normal saline. Data was expressed as the mean ± SD (N = 7−8). ## p < 0.01 compared with control group, * p < 0.05 compared with MP group.

Effect of sodium aescinate (SA) on superoxide dismutase (SOD) activity in lung. SA treatment group was administered with SA at doses of 0.45, 0.9, 1.8 mg/kg; Control and methyl parathion (MP) group was administered with normal saline. Data was expressed as the mean ± SD (N = 7−8). # p < 0.05 compared with Control group, *p < 0.05 compared with MP group.

Effect of sodium aescinate (SA) on glutathione peroxidase (GSH-Px) activity in lung. SA treatment group was administered with SA at doses of 0.45, 0.9, 1.8 mg/kg; Control and methyl parathion (MP) group was administered with normal saline. Data was expressed as the mean ± SD (N = 7−8). # p < 0.05 compared with Control group, ** p < 0.01 compared with MP group.
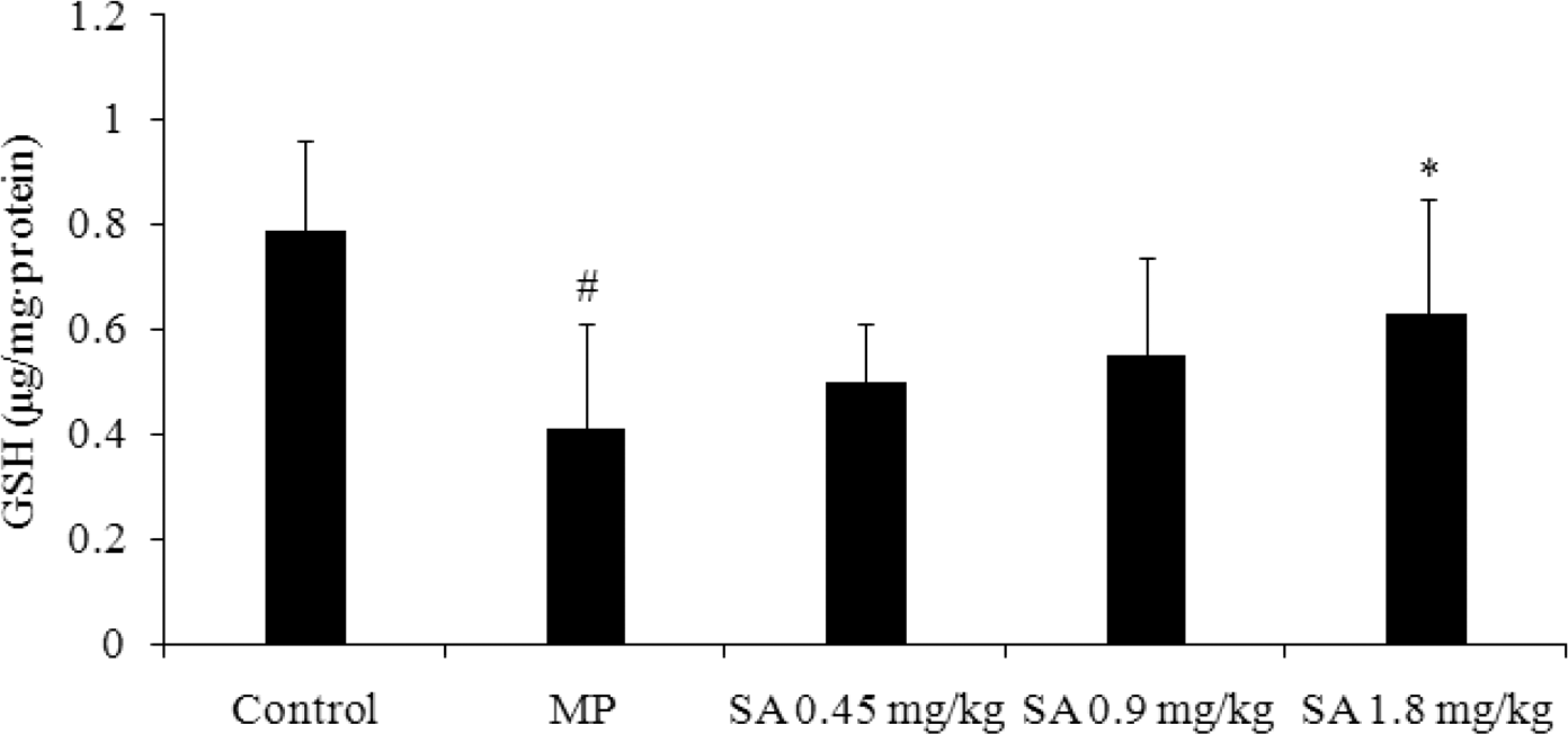
Effect of sodium aescinate (SA) on glutathione (GSH) level in lung. SA treatment group was administered with SA at doses of 0.45, 0.9, 1.8 mg/kg; Control and methyl parathion (MP) group was administered with normal saline. Data was expressed as the mean ± SD (N = 7−8). # p < 0.05 compared with Control group, *p < 0.05 compared with MP group.
Discussion
The extracts from the seeds of Aesculus hippocastanum have been traditionally used in China as a carminative, stomachic, and analgesic agent and as an antipyretic and antihemorrhoidal agent. 21 The saponin mixture isolated from the seeds is a pentacyclic triterpene and is referred to as escin, which exists in α and β forms. It is the β form of escin that has been described to exhibit pharmacologic actions. 22 Escin was found to inhibit acute inflammation induced by acetic acid in mice and histamine in rats, 20 and the anti-inflammatory effect can last for 24 hours. 23
OP compounds affect the respiratory system. 24 Respiratory failure is a prominent feature of acute OP poisoning. Experimental studies report that OP-induced respiratory failure is implicated in pulmonary secretory, airway, and pulmonary dysfunction.7,25,26
The rats in MP-treated group showed lung injury, which was characterized by alveolar congestion, hemorrhage, inflammatory cell infiltration, and destruction of epithelial and endothelial cell structure. Administration with SA significantly alleviated the pathological changes induced by MP administration. Considering that SA has no effect on the reduction of AChE activity, it is reasonable to conclude that the pharmacological action of SA was not related to modulating AChE activity.
A number of reports revealed that MPO plays an important role in the anti-microbial activity of neutrophils. MPO was released during neutrophil activation and MPO activity reflects neutrophil and macrophage diapedesis. 27 The MPO activity also increases in the lung during sepsis, which confirms the infiltration of lung parenchyma by neutrophils and reflects the severity of neutrophil-mediated lung damage. Most frequently, the MPO content in lung is measured in patients with respiratory complications.28–30 The present study showed that MP caused a significant enhancement of MPO activity in rat lung parenchyma, which indicated there was a significant recruitment of neutrophils in lung parenchyma. However, SA inhibited the change of MPO activity in pulmonary parenchymal. The findings suggest SA possesses a protective effect on lung injury induced by AOPP.
NO, acting as a pro-inflammatory agent, plays an important role in lung injury. NO may be attributed to a stimulation of the lung inflammatory response and a persistent increase in neutrophil accumulation and MPO activity. 31 Otherwise, NO interacts with superoxide (O2 −) to produce peroxynitrite (ONOO−), which is a potent oxidant. 32 Increased level of NO is a natural sequence to the inhibition of AChE by OPs. 9 The results showed that there is a significant increase in nitrate (measured as nitrite) in plasma and lung in MP-treated group. Therefore, we make a conclusion that the change of NO level is harmful to lung tissue. Expectedly, SA treatment inhibited the increase of NO content induced by AOPP. The result indicates that the mechanism of effect of SA was related to attenuating the change of NO content induced by AOPP.
Pesticides have been reported to induce the generation of reactive oxygen species (ROS) in vitro and in vivo.33,34 ROS plays an important role in the toxicity of OP compounds. 35 Oxidative damage by free radicals or ROS could result in lipid peroxidation and protein modification, causing changes in membrane properties and cell dysfunction. 36 It has been reported that Aesculus hippocastanum could improve the antioxidative defense system. 37 So, we aimed to investigate the effects of SA on the antioxidant defense systems in lung. Our study found that the content of MDA enhanced while the SOD, GSH-Px activities and GSH level decreased after MP administration. These data imply that the oxidative stress plays a pivotal role in lung injury, which is corresponded to the previous literatures.38,39 SA treatment not only reduced the content of MDA but also increased the activities of SOD, GSH-Px and the level of GSH. So far, the pharmacological action of SA preventing MP-induced lung injury was implicated in its antioxidant effect.
Besides, the study about the adverse events of SA reported that only 0.6% of patients showed gastrointestinal disturbances. According to the risk of acute renal failure, three clinical trials assess the effects of escin on renal function. The result showed that there were no signs of renal impairment in the patients with normal renal function or of worsening of renal function. 40 These findings indicated that escin could not result in renal impairment.
In the present study, it is showed that SA possesses protective effect on lung injury induced by MP administration and the pharmacological action of SA is associated with its effect of anti-inflammation and anti-oxidation. According to the present experiment, SA is a promising agent against lung injury induced by AOPP.
Footnotes
Acknowledgement
The authors would like to thank Tong-Shen Liu for his technical assistance.
This study was supported by the 11th Five Years Key Programs for Science and Technology Development of China (Grant 2008ZX09202-008) and the National Natural Science Foundation of China (Grant 30772760).
