Abstract
Organophosphorus exposure affects different organs such as the lung, gastrointestinal tract, liver, and brain. The present experiment aimed to evaluate the effect of ginsenoside Rg3 on lung injury induced by acute omethoate poisoning. Rats were administered with omethoate subcutaneously at a single dose of 60 mg/kg, followed by ginsenoside Rg3 (5, 10, or 20 mg/kg) treatment. Histopathological examination of the lung was performed at 24 h after the omethoate exposure. The antioxidative parameters in the lung were also assayed. Moreover, the activities of acetylcholinesterase, myeloperoxidase, and the content of tumor necrosis factor α (TNF-α) in the lung were determined. The results showed that ginsenoside Rg3 attenuated omethoate-induced lung injury. Ginsenoside Rg3 increased the level of glutathione in the lung (p < 0.05 or p < 0.01). The altered activities of superoxide dismutase and catalase in the lung were also ameliorated by ginsenoside Rg3 treatment (p < 0.05 or p < 0.01). Ginsenoside Rg3 caused significant reductions in the contents of malondialdehyde, TNF-α, and the activity of myeloperoxidase (p < 0.05 or p < 0.01). The present study demonstrated that ginsenoside Rg3 had a protective effect against omethoate-induced lung injury in rats, and the mechanisms were related to its antioxidant potential and anti-inflammatory effect.
Introduction
The widespread use of pesticides in agricultural programs has caused severe environmental pollution and potential health hazards, including acute and chronic cases of human poisoning. Organophosphorus compounds are used widely as pesticides. Organophosphorus pesticide poisoning is a major health issue worldwide and is particularly severe in developing countries. 1 Organophosphorus pesticide poisoning causes death mainly by inducing acute respiratory failure, which occurs and develops rapidly, resulting in respiratory arrest. The lipophilic nature of organophosphorus facilitates its interaction with the cell membrane and leads to perturbations in the phospholipids bilayer structure, enhancing the production of reactive oxygen species (ROS), which in turn generate oxidative stress in different tissues. 2 Previous study indicated that the respiratory tract and in particular the lungs are the main target of ROS generated by several toxicants. 3 Exposure of organophosphorus can cause pulmonary impairments in mice and rabbits, such as alveolar congestion, hemorrhage, neutrophil infiltration, emphysematous changes, and cellular aggregation in the vascular walls and air spaces. 4 It is also reported that the organophosphorus-treated rats showed lung injury characterized by oxidative stress and inflammation. 5
Panax ginseng Meyer is a well-known medicinal plant from eastern Asian countries, especially China, Korea, and Japan. Ginsenosides are the bioactive ingredients of ginseng that is used widely in traditional medicine. Extensive studies evaluated the different pharmacological activities of ginsenosides, such as their ability to suppress inflammation and oxidative stress as well as their vasorelaxation effect. 6 Ginsenoside Rg3 (Figure 1, formula: C42H72O13) is one of the most effective ginsenosides. 7 It is reported that ginsenoside Rg3 showed antioxidative and anti-inflammatory activities in cultured mammalian cell lines. 8 Ginsenoside Rg3 displays neuroprotection against the cerebral ischemia-induced injury in rat brains through reducing lipid peroxides, scavenging free radicals. 9 Ginsenoside Rg3 exhibits anti-inflammatory activity in the vasculature partly by downregulating the expression of cell adhesion molecules and proinflammatory cytokines in human endothelial cells. 10

The chemical structure of ginsenoside Rg3.
To date, most studies have focused on identifying protective antioxidant and anti-inflammatory agents against organ injury induced by organophosphorus compounds. 11,12 Omethoate (O,O-dimethyl-S-[2-(methylamino)-2-oxoethyl] phosphorothioate), an important organophosphorus pesticide, is frequently used against a wide range of insects and mites and for indoor control of houseflies. According to previous report, exposure to omethoate causes several abnormalities like benign and malignant neoplasm of the liver, atrophy of the testes, chronic renal disease, and lung tissue damage. 13 Recent results also demonstrated that the exposures of animal to omethoate increased malondialdehyde (MDA) level and fluctuated antioxidant enzyme activities and reduced glutathione (GSH) content. 14,15 To our knowledge, there is little information available on the potential ability of ginsenoside Rg3 to attenuate lung toxicity induced by omethoate. Therefore, the present study aims to determine the efficiency of ginsenoside Rg3 in antagonizing oxidative damage and inflammation in the lungs of rats exposed to omethoate.
Materials and methods
Materials
Ginsenoside Rg3, a white amorphous powder, was provided by the Department of Medicinal Chemistry, Jilin University, China. Its purity (>99%) was determined by high-performance liquid chromatography. Omethoate (78%, w/w) was obtained from Changshuang Co., Ltd (Changchun, China, lot number: 130619). Acetylcholinesterase (AChE), myeloperoxidase (MPO), MDA, superoxide dismutase (SOD), catalase (CAT), GSH, and protein level test kits were purchased from the Institute of Jiancheng Bioengineering (Nanjing, China). Tumor necrosis factor α (TNF-α) enzyme-linked immunosorbent assay (ELISA) kits were purchased from R&D Systems, Inc. (Minneapolis, Minnesota, USA). Other chemicals and reagents used in this study were of analytical grade.
Animals and treatment
The experiments were performed according to the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals (publication 86-23, revised in 1986) and were approved by the local Ethics Committee of Jilin University (J130926). Male Sprague Dawley rats weighing 250 ± 20 g were obtained from the Experiment Animal Center of Norman Bethune University of Medical Science (Jilin, China). All the rats were housed in diurnal lighting conditions (12 h/12 h) and allowed free access to food and water. The animals were acclimatized to laboratory conditions for 7 days. Based on the doses of ginsenoside Rg3 in previous experiment, 16 the animals were randomly divided into five groups with 10 animals in each group: control group, omethoate group, ginsenoside Rg3 (5 mg/kg) group, ginsenoside Rg3 (10 mg/kg) group, and ginsenoside Rg3 (20 mg/kg) group. The rats in the omethoate and ginsenoside Rg3 groups were administered subcutaneously with omethoate at a single dose of 60 mg/kg. Ten minutes after omethoate administration, the rats in the ginsenoside Rg3 groups were treated with ginsenoside Rg3 at a dose of 5, 10, or 20 mg/kg via tail vein, whereas the animals of the other groups were injected normal saline in equivalent volumes. There were no dead rats after omethoate exposure.
Histopathological examination
Twenty-four hours after omethoate administration, the animals were killed through an overdose of chloral hydrate (600 mg/kg, intraperitoneally). The lungs of two rats in each group were removed and then fixed in phosphate-buffered 10% formalin solution. Paraffin-embedded specimens were cut into 5-μm thick sections and stained with hematoxylin-eosin. The sections were examined under a light microscope by an experienced observer, who was blind to the treatment, and then photomicrographs were taken.
Preparation of the homogenate of lungs
To prepare the homogenate, eight lungs of each group were harvested and weighed. They were then homogenized in 4 volumes of 0.1 mol/L ice phosphate buffer (pH 7.4) and centrifuged at 5000 g for 10 min at 4°C. The supernatants were immediately stored at −80°C and used for the determination of the biochemical analysis and ELISA assay. The total protein was estimated by the previous method. 17
Assays of AChE in lung tissues
AChE activity was determined according to the previous methods. 18
Assays of MDA, SOD, CAT, and GSH in lung tissues
The determination of MDA content was performed according to previous method. 19 SOD activity was determined by the nitro blue tetrazolium reduction method. 20 CAT activity in lung tissues was measured at 37°C by following the rate of disappearance of hydrogen peroxide (H2O2) at 240 nm. 21 GSH was measured in the cytosolic fraction fluorimetrically following the method of Cohnand and Lyle. 22
Assays of MPO and TNF-α in lung tissues
The MPO activity in lung tissues was determined according to previous method. 23 The MPO activity was calculated from the absorbance of the mixture at 450 nm, using horseradish peroxidase as the standard. The levels of TNF-α in lung tissues were estimated with an ELISA plate reader, using commercially available ELISA kits and following the manufacturer’s protocols. Duplicate samples were analyzed for each sample.
Statistical analysis
Data were expressed as means ± standard deviation. Statistical analysis was carried out using one-way analysis of variance, followed by Student–Newman–Keuls’s test. The value of p < 0.05 was considered as statistically significant.
Results
Effect of ginsenoside Rg3 on pathological changes in lungs
The histology of lungs in the control group showed a normal single layer of squamous epithelium, a thin layer of connective tissue, and an absence of infiltration of inflammatory cells in lung tissues. However, omethoate administration caused significant numbers of inflammatory cells to migrate into the alveolar and interstitial spaces, with destruction of epithelial and endothelial cell structure, alveolar congestion, and hemorrhage. In contrast, treatment with ginsenoside Rg3 significantly attenuated the pathological changes induced by omethoate administration, manifested by less alveolar septal thickening, less inflammatory cell infiltration, and less alveolar congestion (Figure 2).
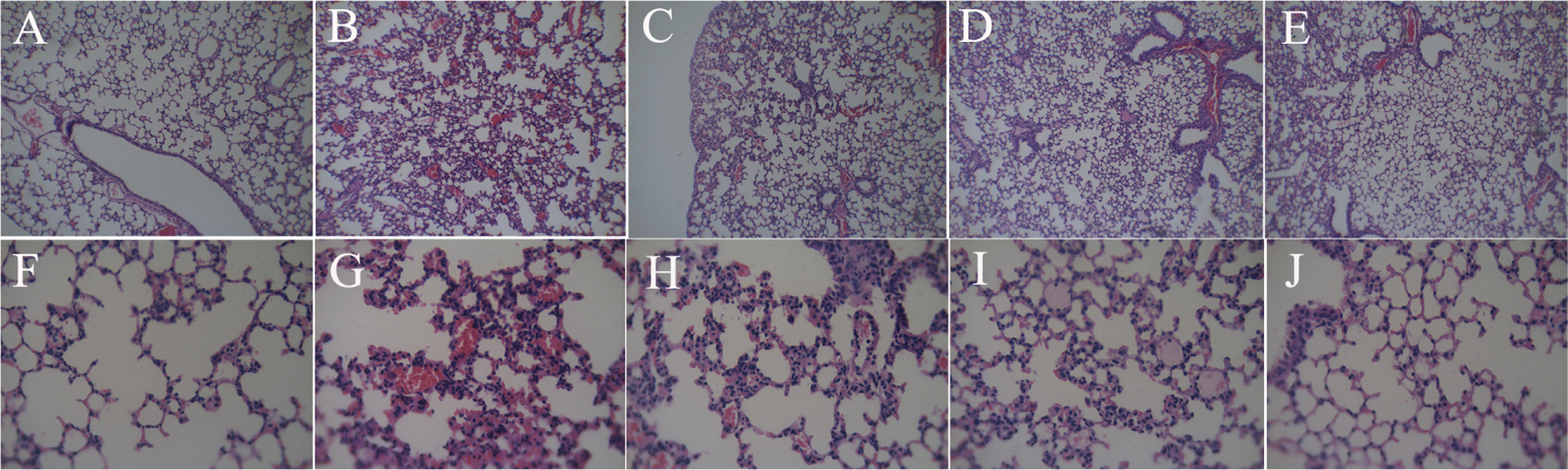
Effect of ginsenoside Rg3 on the pathological changes in lungs of rats (H&E). (a) Control group (×200), (b) omethoate group (×200), (c) Ginsenoside Rg3 (5 mg/kg) group (×200), (d) ginsenoside Rg3 (10 mg/kg) group (×200), (e) ginsenoside Rg3 (20 mg/kg) group (×200), (f) control group (×400), (g) omethoate group (×400), (h) ginsenoside Rg3 (5 mg/kg) group (×400), (i) ginsenoside Rg3 (10 mg/kg) group (×400), and (j) ginsenoside Rg3 (20 mg/kg) group (×400). H&E: hematoxylin-eosin.
Effect of ginsenoside Rg3 on AChE activity in lungs
Omethoate administration inhibited significantly the AChE activity in the lungs. Ginsenoside Rg3 had no effect on the reduction of AChE activity induced by omethoate exposure (Figure 3).

Effect of ginsenoside Rg3 on AChE activity in lungs. Data are expressed as means ± SD (n = 8). Statistical analysis was carried out using one-way ANOVA followed by Student–Newman–Keuls test. ## p < 0.01: compared with control group; AChE: acetylcholinesterase; ANOVA: analysis of variance; SD: standard deviation.
Effect of ginsenoside Rg3 on the level of MDA in lungs
The lipid peroxidation end product MDA was quantitated in the lungs of rats. Compared with the control group, the levels of MDA were significantly increased following omethoate exposure. Treatment with ginsenoside Rg3 at a dose of 5, 10, or 20 mg/kg attenuated the increase of MDA content (Figure 4).

Effect of ginsenoside Rg3 on the level of MDA in lungs. Data are expressed as means ± SD (n = 8). Statistical analysis was carried out using one-way ANOVA followed by Student–Newman–Keuls test. ## p < 0.01: compared with control group; *p < 0.05; **p < 0.01: compared with omethoate group; MDA: malondialdehyde; ANOVA: analysis of variance; SD: standard deviation.
Effect of ginsenoside Rg3 on the activities of SOD and CAT in lungs
A significant reduction in the activities of SOD and CAT in omethoate-challenged rats was observed as compared to the control group. In contrast, administration of ginsenoside Rg3 at a dose of 5, 10, or 20 mg/kg to omethoate-challenged animals partially prevented the reduction in the activities of SOD and CAT in the lungs (Figures 5 and 6).

Effect of ginsenoside Rg3 on the activity of SOD in lungs. Data are expressed as means ± SD, (n = 8). Statistical analysis was carried out using one-way ANOVA followed by Student–Newman–Keuls test. ## p < 0.01: compared with control group; *p < 0.05: compared with omethoate group; SOD: superoxide dismutase; SD: standard deviation; ANOVA: analysis of variance.

Effect of ginsenoside Rg3 on the activity of CAT in lungs. Data are expressed as means ± SD (n = 8). Statistical analysis was carried out using one-way ANOVA, followed by Student–Newman–Keuls test. ## p < 0.01: compared with control group; *p < 0.05; **p < 0.01: compared with omethoate group; CAT: catalase; SD: standard deviation; ANOVA: analysis of variance.
Effect of ginsenoside Rg3 on the level of GSH in lungs
Omethoate caused a significant decrease of GSH in lung tissues. Treatment with ginsenoside Rg3 at a dose of 5, 10, or 20 mg/kg significantly inhibited the decrease of GSH levels (Figure 7).

Effect of ginsenoside Rg3 on the level of GSH in lungs. Data are expressed as means ± SD (n = 8). Statistical analysis was carried out using one-way ANOVA, followed by Student–Newman–Keuls test. ## p < 0.01: compared with control group; *p < 0.05, **p < 0.01: compared with omethoate group; GSH: reduced glutathione; SD: standard deviation; ANOVA: analysis of variance.
Effect of ginsenoside Rg3 on MPO activity in lungs
As shown in Figure 8, the MPO activity in the omethoate group showed a significant increase compared with the control group. Also, ginsenoside Rg3 significantly reduced the MPO activity in lung tissues.

Effect of ginsenoside Rg3 on MPO activity in lungs. Data are expressed as means ± SD (n = 8). Statistical analysis was carried out using one-way ANOVA followed by Student–Newman–Keuls test. ## p < 0.01: compared with control group; *p < 0.05, **p < 0.01: compared with omethoate group; MPO: myeloperoxidase; SD: standard deviation; ANOVA: analysis of variance.
Effects of ginsenoside Rg3 on the level of TNF-α in lungs
In this study, omethoate increased the levels of TNF-α in lung tissues significantly. However, treatment with ginsenoside Rg3 inhibited significantly the increase of the TNF-α level in the lungs (Figure 9).

Effect of ginsenoside Rg3 on the level of TNF-α in lungs. Data are expressed as means ± SD (n = 8). Statistical analysis was carried out using one-way ANOVA followed by Student–Newman–Keuls test. ## p < 0.01: compared with control group; *p < 0.05, **p < 0.01: compared with omethoate group; TNF-α: tumour necrosis factor α; SD: standard deviation; ANOVA: analysis of variance.
Discussion
Omethoate, an organophosphate insecticide, is known to produce oxidative stress, which causes respiratory muscle injury when induced in rats. 24 It is also reported that inflammation is associated with the blood–brain barrier dysfunction induced by omethoate poisoning. 25 The present study showed that omethoate caused remarkable lung damage in rats compared with untreated animals, as reflected by significant numbers of inflammatory cells migrating into the alveolar and interstitial spaces, with destruction of epithelial and endothelial cell structure, alveolar congestion, and hemorrhage. However, treatment with ginsenoside Rg3 attenuated omethoate-induced lung injury in rats. These results demonstrated that ginsenoside Rg3 has a potential to prevent lung injury resulting from omethoate administration.
Omethoate intoxication results primarily from the irreversible inhibition of AChE, which leads to acetylcholine accumulation and overstimulation of cholinergic receptors in the peripheral and central nervous systems. The results in this experiment showed that the activity of AChE was reduced markedly after omethoate poisoning. Ginsenoside Rg3 had no effect on the reduction of AChE activity caused by omethoate. These findings indicated that the protective effect of ginsenoside Rg3 against the omethoate-induced lung injury was not related to modulating AChE activity.
Free radicals and oxidative stress play a role in the pathogenesis of organ injury induced by organophosphate. 26 ROS induced by organophosphate in the lung tissues might damage cell membranes by increasing the lipid peroxidation. 27 Organisms have nonenzymatic and enzymatic defense mechanisms against the toxicity and tissue damage of the free radical. 28 The nonenzymatic antioxidant systems are mainly substances of low molecular weight, such as GSH, vitamins C and E, urate, retinyl esters, and β-carotene. The primary antioxidant protection against these species is provided by SOD and CAT, respectively. 29,30 SOD is one of the antioxidant enzymes and plays an important role in ameliorating cell damage by preventing ROS. SOD destroys the highly reactive radical by converting it into the less reactive H2O2 that can be scavenged by CAT reaction. GSH plays an important role in the defense against endogenous membrane peroxidation and subsequent changes by reducing H2O2 via GSH peroxidase. Hence, levels of GSH are believed to be rate limiting in the process of detoxification of H2O2 or other peroxides. Our results showed that there was a significant increase in the level of MDA in the lung tissues of rats administrated with omethoate. However, the increase of MDA levels in the lung tissues induced by omethoate was inhibited partially by ginsenoside Rg3. The activities of SOD, CAT and the level of GSH in the lung tissues were significantly reduced by omethoate exposure. On the other hand, ginsenoside Rg3 treatment resulted in a significant increase in the activities of SOD, CAT, and the level of GSH. These findings suggested that ginsenoside Rg3 possessed protective effects against the omethoate-caused oxidative damage in lung tissues.
The increase in the production of ROS does not fully account for the etiopathogenesis of omethoate-induced lung damage. It was demonstrated that the activity of MPO increased markedly after organophosphate administration. MPO is abundantly expressed in neutrophils and secreted from activated neutrophils. 31 Its overexpression is commonly associated with inflammatory diseases. 32 Therefore, MPO activity is considered an index for the evaluation of neutrophils infiltration and inflammation. 33 TNF-α also plays a significant role in inflammatory diseases of the lung. The most important lung diseases affected by TNF-α include acute lung injury, chronic bronchitis, chronic obstructive pulmonary disease, and asthma. 34 TNF-α also depletes cellular GSH levels. 35 Overexpression of TNF-α in transgenic mice induces differential changes in redox status and GSH-regulating enzymes 36 by depleting the total cellular GSH levels. Treatment of human pulmonary artery endothelial cells with TNF-α decreases the GSH levels of the cells, 37 and this depletion of GSH enhances the endothelial cell susceptibility to oxygen toxicity. 38 The current study showed that omethoate led to increases of MPO activity and TNF-α levels in the lung tissues of rats. These findings indicated that the lung injury induced by omethoate is related to the inflammatory process, at least in part. Ginsenoside Rg3 treatment reduced the activity of MPO and the content of TNF-α and then ameliorated the inflammation and neutrophil infiltration in lung tissues. Collectively, these results demonstrated that ginsenoside Rg3 can inhibit infiltration of neutrophils and subsequent inflammation to prevent omethoate-induced lung injury.
It is important to note that other biochemical factors such as hypoxia-inducible factor (HIF), chemokine in lung tissues, and systemic proinflammatory cytokine have not been evaluated in this study. HIF, chemokine, and proinflammatory cytokine play a pivotal role in lung injury. 39 Thus, additional studies addressing whether ginsenoside Rg3 has an effect on HIF, chemokine, and proinflammatory cytokine in omethoate-induced lung injury are clearly required.
In conclusion, this study demonstrated the protective effects of ginsenoside Rg3 against omethoate-induced lung injury in rats not only by virtue of its antioxidant potential but also due to its anti-inflammatory effect.
Footnotes
Acknowledgment
The authors thank Prof. Altaf S Darvesh for the English language revision.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (grant no. 81473378).
