Abstract
Cis-platin and other platinum complexes are important chemotherapeutic agents useful in the treatment of several cancers. However, therapeutic usage of cis-platin and other platinum complex are limited by their undesirable side effects including cardiotoxicity. In this context, we aimed to compare the damage caused in heart by cis-platin and novel platinum-N-heterocyclic carbene (Pt-NHC) complex. For this purpose, 35 Sprague-Dawley rats were divided randomly into five equal groups (n = 7 for each group). Cis-platin and novel Pt-NHC complex were intraperitoneally administered at a single dose of 5 mg/kg and 10 mg/kg and then sacrificed 10 days after this treatment. The heart tissues were taken from all rats for determination of oxidative and myocardial damage. Cis-platin and novel Pt-NHC complex caused oxidative and histological damage in the heart tissue in a dose-dependent manner (p < 0.05). On the other hand, at the same dose levels, cis-platin caused lower oxidative and histological damage in heart tissue compared to novel Pt-NHC complex. These results suggest that novel Pt-NHC complex is more cardiotoxic than cis-platin.
Introduction
Cis-platin or cis-diamminedichloroplatinium (CDDP, cis-[PtCl2(NH3)2]) is one of the most active antitumor agents in current clinical use. Apart from being curative against testicular tumors, it is effective against several other cancers such as ovarian, cervical and bladder. 1-3 Although its success, the treatment with cis-platin, is limited by undesirable side effects such as nephrotoxicity, ototoxicity, hepatotoxicity, gastrointestinal dysfunction and cardiotoxicity. 4-6 Many investigators 7-9 have indicated that cis-platin caused acute or chronic cardiovascular complications, including atrial fibrilation and supraventricular tachycardia, and could impair their quality of life in human beings. Besides, cis-platin cardiotoxicity leads to delay of treatment and decrease of dose in chemotherapy. Toxicological researches 10,11 showed that cardiotoxicity like hepatotoxicity and nephrotoxicitiy induced by cis-platin could be correlated with oxidative stress, characterized by an increase in lipid peroxidation and decrease in antioxidant system. Thus, the determination of thiobarbituric acid reactive substances (TBARS), reduced glutathione (GSH) levels and CuZn-superoxide dismutase (SOD) and catalase (CAT) activities might demonstrate toxicity of cis-platin and novel platinum-N-heterocyclic carbene (Pt-NHC) complex.
The side effect of the current anticancer drugs has stimulated the research on the novel platinum-based antitumor complexes. In this context, it becomes desirable to develop new series of platinum complexes displaying highly modular scaffolds, easily adaptable to drug design. With this aim, we synthesized N-heterocyclic carbene as ligand for cytotoxic platinum complexes. Pt-NHCs and other NHCs are readily accessible in few steps. Their substituent can be widely varied and their anticancer potential is greater than cis-platin in cell culture system. 12 Despite the existence of extensive studies on NHC complexes in catalysis, their biomedical applications are limited. 13,14 Therefore, the present study described the synthesis and characterization of a novel Pt-NHC complex. Also, the investigations were made to evaluate the effects of cis-platin and novel Pt-NHC complex on oxidative and histological damage in cardiac tissue of rats.
Material and methods
Chemicals
The novel Pt-NHC complex was synthesized in Inonu University, Organometallic research laboratory. Cis-platin (10 mg/ 10 mL, Code 1876A) was obtained from Faulding Pharmaceuticals Plc (Warwickshire, UK). All other chemicals were purchased from Sigma Chemical Co. (St. Louis, Missouri, USA) and were of analytical grade or of the highest grade available.
Animals and treatment
A total of 35 healthy young adult male Spraque-Dawley rats (between 2 and 3 months old and 200−250 g in weight) were obtained from Experimental Animal Institute, Malatya, Turkey, for this experiment. Animals were housed in sterilized polypropylene rat cages, in 12-h light-dark cycle, at an ambient temperature of 21°C. Diet and water from them were given ad libitum. Experiments were performed based on animal ethics guidelines of Institutional Animals Ethics Committee.
Rats were randomly divided into five equal groups (n = 7 in each group). Previous studies 7,15,16 demonstrated that the cis-platin caused cardiotoxic and other toxic effects at the 7 mg/kg single dose. Thus, we selected dose of cis-platin upper (10 mg/kg) and lower (5 mg/kg) range of toxic dose (7 mg/kg) for this experiment. Similarly, because the therapeutic dosage of novel Pt-NHC complex is not reported, we used the same dose of cis-platin for novel Pt-NHC complex. All drug applications were given intraperitonally (ip) by a single injection after all chemicals were dissolved in dimethylsulfoxide (DMSO). In the first (control) group, DMSO was administered to rats by a single injection. In the second and third (low dose) groups, cis-platin and Pt-NHC complex were administered to rats at the dose of 5 mg/kg. In the fourth and fifth (high dose) groups, cis-platin and Pt-NHC complex were administered to rats at the dose of 10 mg/kg. The animals were sacrificed under ether anesthesia after 10 days of drugs administration. Hearts were immediately removed and dissected over ice-cold glass. Tissues were stored at −45°C until analysis.
The homogenization of tissues was carried out in teflonglass homogenizer with 150 mM KCl (pH 7.4) to obtain 1:10 (w/v) dilution of the whole homogenates. The homogenates were centrifuged at 18.000 × g (4°C) for 30 min to determine thiobarbituric acid reactive substances (TBARS), total GSH levels and CAT, SOD activities.
Synthesis of Pt-N-heterocyclic carbene complex
All reactions for the preparation of benzimidazolium salt and Pt-NHC complex were carried out under argon in flame-dried glassware using standard Schlenk techniques. The used solvents were purified by distillation over the drying agents indicated and were transferred under Ar. Melting point was determined in glass capillaries under air with an Electrothermal-9200 melting point apparatus. FT-IR spectra were recorded as KBr pellets in the range 400–4000 cm–1 with an ATI UNICAM 1000 spectrometer. 1 H NMR and 13 C NMR spectra were recorded with a Varian AS 400 Merkur spectrometer operating at 400 MHz ( 1 H), 100 MHz ( 13 C) in CDCl3 with tetramethylsilane as an internal reference. Elemental analyses were performed by Turkish Research Council (Ankara, Turkey) Microlab.
Dichloro-1-ethyl-3-methylbenzimidazolidin-2-ylideneplatinium(II), 1
A solution of entetraamine (0.25 g; 0.72 mmol) and [PtCl2(COD)] (0.26 g; 0.70 mmol) in tetrahydrofurane (thf) (15 mL) was heated at 50°C for 4 h. Upon cooling, a white solid precipitated. It was filtered, washed with hexane (15 mL) and dried under vacuum. The crude product was recrystallized from CH2Cl2:Et2O. Yield: 0.35 g; 83%, m.p.: 340-341°C; υ(CN) = 1394 cm-1. 1 H NMR (CDCl3) δ: 1.43 (t, J = 15 Hz, 3H, NCH2CH 3), 4.42 (s, 3H, NCH 3), 4.68 4.68 (q, J = 15 Hz, 2H, NCH 2CH3), 7.26-7.45 (m, 4H, C6 H 4). 13 C(H)NMR (CDCl3) δ: 16.4 (NCH2 CH3), 36.9 (NCH3), 45.5 (NCH2CH3), 112.6, 125.7, 125.8, 134.9 and 136.9 (C 6H4), 160.8 (Pt-Ccarb ). Anal. Calcd for C20H24PtCl2N4: C, 40.96; H, 4.13; N, 9.55%; found: C, 40.83; H, 4.09; N, 9.58%.
Biochemical assay
The levels of TBARS, as an index of lipid peroxidation, were determined by thiobarbituric acid reaction using the method of Yagi.
17
The product was determined spectrophotometrically at 532 nm and results were expressed as nmol/g tissue. The GSH content of the heart homogenate was measured at 412 nm using the method of Sedlak and Lindsay
18
and was expressed as nmol/g tissue. SOD activity was measured by the inhibition of nitroblue tetrazolium (NBT) reduction due to O2
− generation by the xanthine/xanthine oxidase system.
19
One unit of SOD activity was defined as an amount of protein causing 50% inhibition of the NBT reduction rate. The product was analyzed spectrophotometrically at 560 nm and results were expressed as IU/mg protein. CAT activity of tissues was determined according to the method of
Determination of protein content
Tissue protein content was determined according to the method developed by Lowry et al. 21 using bovine serum albumin as standard.
Histological examination
For histological evaluation, heart samples were fixed in 10% formalin. Paraffin-embedded specimens were cut into 5 μm thick sections and were stained with hematoxylin-eosin (H&E) and Masson’s trichrome. The sections were examined by a Leica DFC 280 light microscope. An overall score of cardiac damage severity was semiquantitively assessed as follows: Cell with eosinophilic cytoplasm and pyknotic nuclei, cytoplasmic vacuolization, congestion, hemorrhage and interstitial edema. The microscopic score of each tissue was calculated as the sum of the scores given to each criteria. Scores were given as 0, none; 1, mild; 2, moderate and 3, severe for each criteria. Thus, the maximum score reached was 15.
Statistical analysis
All values were presented as mean ± SEM. Differences were considered to be significant at p < 0.05. A computer program SPSS 11.0 (SPSS Inc, Chicago, Illinois, USA) was used for statistical analysis. For biochemical values, statistical analyses were performed using one-way ANOVA and post hoc Tukey's honestly significant difference test. Histological results were compared with Kruskal-Wallis variance analysis. Where differences among the groups were detected, group means were compared using the Mann-Whitney U test.
Results
Preparation of Pt-N-heterocyclic carbene complex
The carbene ligand, arising from the deprotonation by tBuOK of the benzimidazolium bromide according to Lappert’s procedure,
22
was treated with [PtCl2COD] in thf at 50°C. After 4 h, the reaction was completed and platin-carbene complex,

Synthesis of platinum-N-heterocyclic carbene (Pt-NHC) complex.
Complex, which is very stable in the solid state, has been characterized by analytical and spectroscopic techniques. It exhibits a characteristic ν(NCN) infrared absorption typically at 1394 cm–1. The 1 H NMR spectra of complex is characterized by the absence of the NCHN protons which are fingerprints of the NHC precursor, thus suggestive of successful formation of the metal-NHC moieties. The 13 C chemical shifts, which provide a useful diagnostic tool for metal carbene complexes, show that C carb is substantially deshielded. δ ( 13 Ccarb) is 160.8 ppm and is typical value for benzimidazolin-2-ylidene complexes of platinum. 23
Biochemical evaluation
Changes in biochemical parameters, such as, CAT, SOD activities and TBARS, GSH levels in heart tissue of rats exposed to 5 mg/ kg (low dose) and 10 mg/ kg (high dose) doses of cis-platin and Pt-NHC complex are given in Table 1 . A significant decrease (p < 0.05) in SOD, CAT activities and GSH level was observed in the heart tissue of rat exposed to 5 mg/kg (low dose) and 10 mg/ kg (high dose) doses of cis-platin and novel Pt-NHC complex compared to the control group. However, the TBARS levels in heart tissue of rats were significantly (p < 0.05) increased at the low and high doses of cis-platin and novel Pt-NHC complex compared to the control group (p < 0.05). The present study revealed that administration of cis-platin and novel Pt-NHC complex changed SOD, CAT activities and TBARS, GSH levels in a dose-dependent manner.
Changes in SOD, CAT activities and GSH, TBARS levels in heart tissue of rats administered cis-platin and novel Pt-NHC complex (value ± SEM, n = 7)

Abbreviations: CAT: catalase, GSH: reduced glutathione, TBARS: thiobarbituric acid reactive substances, SOD: superoxide dismutase, Pt-NHC: platinum-N-heterocyclic carbine.
Note: Means bearing different superscripts within same row were significantly different (P<0.05).
On the other hand, it was observed that, low dose novel Pt-NHC complex group SOD, CAT activities and GSH level were significantly (p < 0.05) decreased compared to low dose cis-platin group. Similarly, there were significant changes in SOD, CAT activity and GSH level of rat exposed to both high dose cis-platin and Pt-NHC complex. Additionally, TBARS levels of high dose novel Pt-NHC complex treatment group were significantly (p < 0.05) increased compared to same dose cis-platin treatment. In the same way, there were significant changes in SOD, CAT activities and GSH level of rat exposed to both low dose cis-platin and Pt-NHC complex.
Histological evaluation
The control group exhibit normal morphological findings with H&E staining methods (Figure 1). There were significant differences between control and other groups. Different grades of edema, congestion and hemorrhage among myocytes were detected in all study groups (Figures 2−4). Congestion and hemorrhage were more prominent in low and high dose cis-platin groups compared to low- and high-dose novel Pt-NHC complex groups. In addition, vacuolization, elevation in eosinophilia of degree in the cytoplasms of some cells and pyknotic changes in the nuclei were scattered randomly among the areas with normal morphology (Figures 5−7). Eosinophilic stained cells, shrinkage and darkening of nuclei were recognized as increased in high-dose novel Pt-NHC complex group (Figure 8). In areas with damage, there were spaces in intercellular field. These morphological changes seem to be related with edema formation. There was statistically significant difference between low- and high-dose Pt-NHC complex groups. The score of semiquantitative analysis in the heart tissue was reported in Table 2 .

Control group; normal histological appearance of cardiac muscle cells (hematoxylin-eosin [H&E], ×132).

Low-dose cis-platin group; severe hemorrhage (*) and congestion (arrows) are visible (hematoxylin-eosin [H&E], ×66).

High-dose cis-platin group; hemorrhage (*) and edema (E) are evident (hematoxylin-eosin [H&E], ×66).

Low-dose N-heterocyclic carbene (NHC)-platin group; moderate hemorrhage (*) and congestion (arrows) are seen (hematoxylin-eosin [H&E], ×66).

Low-dose cis-platin group; intracytoplasmic vacuolization (arrow heads), increase in eosinophilia cytoplasm and pyknotic changes in the nuclei (arrows) are observed (hematoxylin-eosin [H&E], ×132).

High-dose cis-platin group; cardiac muscles show eosinophilia cytoplasm and pyknotic changes in the nuclei (arrows) and intracytoplasmic vacuolization are visible (arrow heads; hematoxylin-eosin [H&E], ×132).
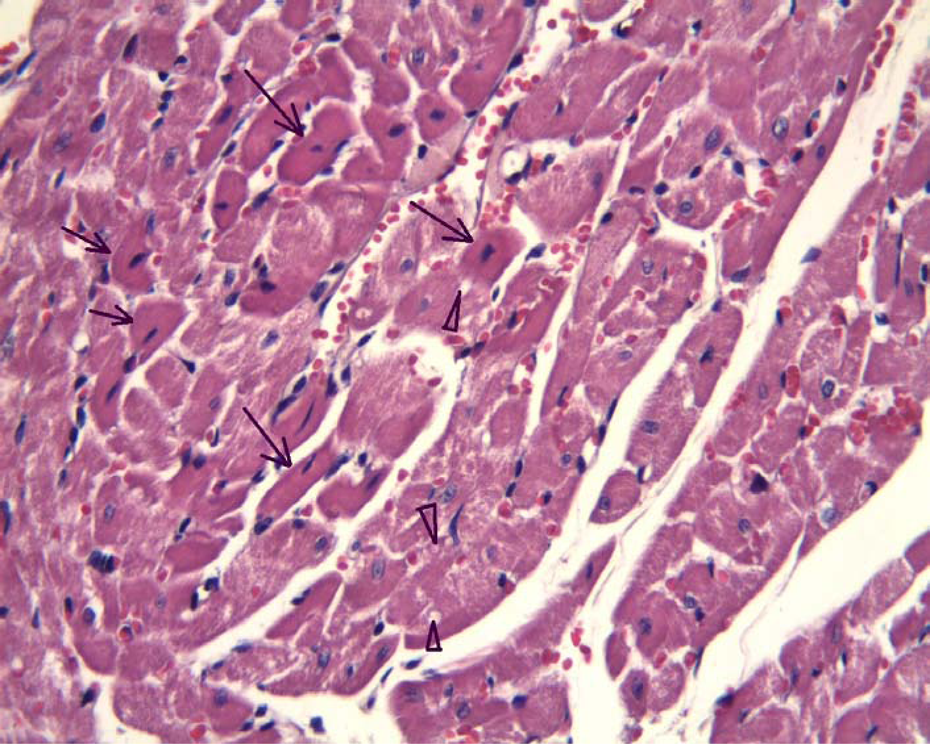
Low-dose N-heterocyclic carbene (NHC)-platin group; note the vacuoles in the cytoplasm (arrow heads), pyknosis of nuclei and eosinophilic cytoplasm (arrows; hematoxylin-eosin [H&E], ×132).

High-dose N-heterocyclic carbene (NHC)-platin group; notice increased number of cells with eosinophilic cytoplasm (arrows; hematoxylin-eosin [H&E], ×132).
Comparison of the histological grading (mean ± SEM) of control and study groups

Abbreviation: Pt-NHC: platinum-N-heterocyclic carbine.
Note: Means bearing different superscripts within same column were significantly different (p < 0.05).
Discussion
Cis-platin is one of the most active antitumor agents but its usage is limited by undesirable side effects such as cardiotoxicity. For this reason, Pt-NHC complex was synthesized to show whether it is less toxic to cardiac tissue or not. However, the results show that novel Pt-NHC complex is more cardiotoxic compound than cis-platin in terms of oxidative and histological damage in rats. This may be due to its superior anticancer properties as revealed by cell culture studies 12 ; this finding might also suggest that less therapeutic dose of novel platinum complex is needed, when it is used for chemotherapy.
Many studies showed that cis-platin generates ROS such as superoxide anions and hydroxyl radicals and stimulates cardiac lipid peroxidations and this leads to oxidative damage of cell components. 15,16,24 In this study, it is showed that cis-platin increased TBARS levels and myocardial damage. This result is consistent with the previous studies which showed that cis-platin leads to oxidative damage in rats. 15,16 Similarly, Wang et al. 7 suggested that cis-platin increased TBARS levels in heart tissue dose-dependently and caused myocardial damage in rats. There was no study about the effects of novel Pt-NHC complex on TBARS levels. In the present study, the novel Pt-NHC complex increased TBARS levels and caused oxidative damage and this effect were more pronounced compared to cis-platin.
Reduced glutathione (GSH) is a major non-enzymatic antioxidant molecule that is involved in the second line of defense against free-radical damage in the body. 25 In the current study, GSH levels decreased in rats treated with cis-platin. Similarly, it has been shown in various studies that 26,27 cis-platin administration decreases GSH level in some tissue of rats such as heart, liver and kidney. The present study showed for the fist time that novel Pt-NHC complex decreased GSH levels in heart tissue in a dose-dependent manner and that this decrease was more pronounced than that caused by cis-platin alone.
CAT, which act as a preventive antioxidant, and SOD, as a chain-breaking antioxidant, play an important role in protection against to the deleterious effect of lipid peroxidation. 28 In the present study, CAT and SOD activities decreased in rats treated with cis-platin and novel Pt-NHC complex. In the same way, Yuce et al. 24 showed that cis-platin significantly decreased CAT and SOD activities in the heart and liver tissues of rats. Additionally, several studies indicate that cis-platin decreased antioxidant enzyme activities in rats. 15,29 There is no study about the effects of Pt-NHC complexes on CAT and SOD activities in rat. Novel Pt-NHC complex decreased CAT and SOD activities dose-dependently .This decrease in CAT and SOD activities in novel Pt-NHC complex groups was more than cis-platin group. In this context, it was assumed that novel Pt-NHC complex caused damage of body’s antioxidant mechanism more than cis-platin.
Histopathological results showed that cis-platin and novel Pt-NHC complex caused myocardial damage such as congestion, hemorrhage, vacuolization, elevation of eosinophilia degree in the cytoplasm of some cells and pyknotic changes in the nuclei. The present study also showed that novel Pt-NHC complex caused more myocardial damage than cis-platin. Similarly, some investigators determined that myocardial changes were characterized with cis-platin therapy in rats. 7,24 Our results suggested that myocardial toxic effects of the novel NHC complex might be due to the oxidative damage caused by this agent.
Conclusions
In conclusion, both cis-platin and novel Pt-NHC complex induced oxidative and myocardial damage in rats. On the other hand, novel Pt-NHC complex caused more oxidative stress and myocardial damage in heart tissue than cis-platin. This may also suggest that therapeutic effects of novel Pt-NHC complex as an anticancer drug may be superior to cis-platin. Therefore, further studies are warranted to elucidate the efficacy of novel Pt-NHC complex in cancer treatment.
Footnotes
Acknowledgements
It is a great pleasure to thank TUBITAK for their financial support during our project.
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
We acknowledge the support of TUBITAK (Scientific and Technical Research Council of the Turkish Republic) under Grant 109T540.
