Abstract
Paullinia cupana Mart. var. Sorbilis, commonly known as Guaraná, is a Brazilian plant frequently cited for its antioxidant properties and different pharmacological activities on the central nervous system. The potential beneficial uses of Guaraná in neurodegenerative disorders, such as in Parkinson's disease (PD), the pathogenesis of which is associated with mitochondrial dysfunction and oxidative stress, has not yet been assessed. Therefore, the main aim of the present study was to evaluate if an extract of commercial powdered seeds of Guaraná could protect human dopaminergic neuroblastoma SH-SY5Y cell line against rotenone-induced cytotoxicity. Two concentration of Guaraná dimethylsulfoxide extract (0.312 and 0.625 mg/mL) were added to SH-SY5Y cells treated with 300 nM rotenone for 48 h, and the cytoprotective effects were assessed by means of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay, measuring lactate dehydrogenase (LDH) levels, and analyzing nuclear integrity with Hoechst33258 stain. Results showed that the addition of Guaraná extract significantly increased the cell viability of SH-SY5Y cells treated with rotenone, in a dose-dependent manner. On the other hand, LDH levels were significantly reduced by addition of 0.312 mg/mL of Guaraná, but unexpectedly, no changes were observed with the higher concentration. Moreover, chromatin condensation and nuclear fragmentation were significantly reduced by addition of any of both concentrations of the extract. The results obtained in this work could provide relevant information about the mechanisms underlying the degeneration of dopaminergic neurons in PD and precede in vivo experiments. Further studies are needed to investigate which active constituent is responsible for the cytoprotective effect produced by Paullinia cupana.
Keywords
Introduction
Parkinson's disease (PD) is a complex, chronic neurodegenerative disorder with motor abnormalities, autonomic disturbances, psychiatric sequelae, and cognitive impairments as major clinical manifestations. 1 It is one of the most common neurodegenerative disorders, only second to Alzheimer’s disease. 2
The pathophysiology of PD is mainly characterized by a selective and significant loss of dopaminergic neurons in the substantia nigra pars compacta, which disrupts nigro-striatal connections, and leads to depletion of striatal dopamine levels below 60%−70% of its normal levels. These events trigger tremor, rigidity, and akinesia or bradykinesia, which are the core clinical features of PD. 3,4 Regarding its etiology, for most of the cases, it remains unknown, although it is thought that both genetic and environmental factors play an interrelated role in the development of the disease. 5 In spite of its probably multiple etiology, most of the identified factors appear to converge on common pathogenic mechanisms, like protein mishandling and oxidative stress. 1 The latter has received particular attention due to the fact that metabolism of endogenous dopamine results in the formation of reactive oxygen species (ROS) in the substantia nigra. 6 In fact, the oxidative phenomenon is the main contributing factor to nigral neuronal death, 7 and the analyses of postmortem brains had established mitochondrial dysfunction and oxidative stress as major participants in the pathogenesis of the disorder. 8
A classic model developed for the study of possible mechanisms underlying neurodegeneration in PD consists in the administration of the plant-derived pesticide rotenone (a specific inhibitor of mitochondrial complex I) to the human dopaminergic neuroblastoma cell line, SH-SY5Y. Rotenone induces apoptosis in SH-SY5Y and in mesencephalic dopaminergic cells by, at least in part, the generation of mitochondrial ROS. 9 –11 Rotenone can be used not only in in vivo models of PD 12,13 but also in an in vitro system in order to search new compounds with neuroprotective properties that could work as interesting prototypes for new drugs. Plant-derived substances have been thought to be important sources of these prototypes. 14
Paullinia cupana Mart. var. Sorbilis, commonly called Guaraná, is a Brazilian plant that belongs to the Sapindaceae family. The chemical composition of the seed of Guaraná is quite complex, being catechin, epicatechin, ent-epicatechin, procyanidins B1, B2, B3, B4, A2, C1, and phenolic compounds some of its constituents. 15 Some of these components had nigrostriatal dopaminergic neurons protective and antioxidant activities demonstrated. 16,17 The presence of xanthine bases such as caffeine, theophylline and theobromine, and purine alkaloids, which have been often described as having antioxidant properties, is also well known. 18 –22 Some of the frequently cited pharmacologic activities of Guaraná are consequences of its stimulant actions on the central nervous system (CNS). 23 For instance, the effectiveness of Guaraná has been demonstrated for postradiation fatigue and depression. 24 Guaraná is still used as a painkiller, febrifuge, to treat hypertension, migraine, neuralgia, and dysentery. 23 It was also reported that its seeds posses anti-platelet aggregatory properties. 25 Moreover, the antioxidant activity of ethanol extract from Paullinia cupana has also been reported. 23 Despite this growing body of data on Guaraná properties, its effects on CNS are much less frequently assessed, thus the potential beneficial uses of this plant, or its components, in neurodegenerative diseases, such as PD, are little explored. Therefore, the main aim of the present study was to evaluate if commercial powdered seeds of Guaraná could have cytoprotective effects, by using rotenone-induced apoptosis in SH-SY5Y cells as an in vitro model of degeneration of dopaminergic neurons. The results of this work might help to get new insights for the development of new therapeutic tools for neurodegenerative disease, such as PD.
Materials and methods
Materials
Neurobasal medium DMEM HAM F-12, fetal bovine serum, and penicillin/streptomycin antibiotics were obtained from Cultilab (Campinas, São Paulo, Brazil). Bis-benzimide (Hoechst 33258), MTT-tetrazole (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide), sodium dodecylsulfate (SDS), dimethylsulfoxide (DMSO), Triton X-100 (T-Octyiphenoxypolyethoxyethanol), p-formaldehyde, and bovine serum albumin were purchased from Sigma-Aldrich (St. Louis, Missouri, USA). Human neuroblastoma SH-SY5Y cells were kindly provided by Dr. Pablo Mendez (Instituto Cajal, Madrid, Spain).
Preparation of extract
Paullinia cupana (Guaraná) was acquired as a powder from pulverized dry seed (Vitalab Ltda., São Paulo, Brazil). Extract was prepared with 125 mg of this powder diluted in 1 mL DMSO during 24 h and centrifuged at 900 × g for 15 min (concentrations above 125 mg/mL exceed the solubility limit of the drug in this solvent). Supernatant was filtered using membrane FG hydrophobic with pores of 0.2 µm (MILLIPORE, Billerica, Massachusetts, USA) and used in the experiments.
Cell culture and rotenone-induced oxidative damage in SH-SY5Y cells
Human neuroblastoma SH-SY5Y cells were cultured at 37°C in DMEM HAM F-12 medium supplemented with 10% fetal bovine serum, 100 IU/mL penicillin, and 100 µg/mL streptomycin in a humidified atmosphere of 5% CO2 and 95% air. The culture medium was changed every 2 days. The cells were seeded in 96-well plates at a density of 62.5 × 103 cells/cm2 and were cultured for 72 h prior to treatments.
To apply the best experimental conditions, cells were treated with rotenone dissolved in DMSO and mixed with the culture medium (final DMSO concentration of 0.5% v/v) at concentrations ranging from 10 nM to 1000 nM for various intervals (24, 48 and 72 h) to examine cell viability. Nonlinear regression was used to determine the effective concentration of rotenone that was able to kill 50% of cells (EC50) using GraphPad Prism (version 4.03, GraphPad Software Inc., San Diego, California, USA). Eight replicates were used for each concentration, and all the experiments were performed at least in triplicate. Concentration of 300 nM rotenone during 48 h was used in all subsequent experiments.
Treatment with Paullinia cupana
The extract of Paullinia cupana was subjected to a serial dilution test. To observe possible toxic effects, SH-SY5Y cells were treated with the extract in concentrations ranging from 0.0097 mg/mL to 0.6250 mg/mL (maximum concentration possible for 0.5% v/v DMSO from the 125 mg/mL initial extract). Control cells were treated with DMSO alone. After that, two concentrations (that were not toxic) were chosen for a better evaluation of the cytoprotective effects using analysis of cell viability (detected by MTT assay and by measuring lactate dehydrogenase levels) and nuclear morphological changes (by staining with Hoeschst 33258).
Analysis of cell viability by metabolic parameters
Cell viability was measured by the MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) method. 26 The MTT is a yellow substratum for mitochondrial dehydrogenases that generates a blue product (formazan) measurable by photometric techniques. 27
Human neuroblastoma SH-SY5Y cells in 96-well plate were submitted to the treatments of interest (rotenone, extract, and both) and after this, the MTT reagent dissolved in phosphate buffered saline (PBS) was added to each well (0.04 mg/mL per well). Following additional 2-h incubation, 100 µL of SDS (50% DMF and 20% SDS in water) was added to breach the cells and to dissolve the formazan crystals. Then the absorbance was measured at 595 nm of wavelength using a microplate reader (THERMO PLATE, model TP-reader - type B). Wells without cells were used as blanks and were subtracted as background from each sample. Results were expressed as a percentage of control and represented in a graph.
Analysis of cell viability by membrane integrity evaluation
Analysis of membrane integrity was performed in order to demonstrate the deleterious effects of rotenone and protective effects of the extract on this structure, and also to confirm results from MTT assay, since membrane integrity is a classic parameter for analysis of cell viability. 28 Membrane integrity was determined by measuring lactate dehydrogenase (EC 1.1.1.28; LDH) levels in the culture medium using a commercial colorimetric system (kit from Doles - Brazil). After 48-h incubation with the respective treatments, the culture medium of each 35 mm-plate was collected, centrifuged, and the supernatant was tested following fabricant's instructions. The absorbance was then measured at 510 nm of wavelength using the same microplate reader used for MTT assay. Data were expressed as international unit per liter (IU/L).
Analysis of nuclei morphology by staining with Hoeschst 33258
To visualize nuclear morphology, cells were washed twice with 1 mM PBS after the treatments and then fixed in 4% paraformaldehyde for 10 min. Cells were permeabilized with 1% Triton X-100, diluted in PBS with 2.5% serum, for 20 min at room temperature (RT), and stored at −20°C. The cells were then stained with 2.5 µg/mL DNA dye Hoechst 33258 in PBS for 10 min at RT and analyzed by fluorescence microscopy (Olympus AX70 microscope - ex/em 340/510 nm), using a 20× objective. Viable healthy human neuroblastoma SH-SY5Y cells nuclei were uniformly stained. Cells with condensed or fragmented nuclei were counted on adjacent fields for a total of 140−160 cells. The percentage of condensed/fragmented nuclei was determined on six replicates for each condition and normalized to controls.
Statistical analysis
Statistical analysis data was performed by one-way ANOVA, followed by Tukey's HSD pair-wise comparison tests. When the assumption of normal distribution of the data was violated (checked by Shapiro-Wilk test) and/or the data did not show homocedasticity (checked by Levene's test), the statistical evaluation was carried out by the non-parametric Kruskal-Wallis test followed by post-hoc pair comparisons using Mann-Whitney test. In the latter case, p values were adjusted by Bonferroni method in order to control familywise error rate. Differences with a probability of 5% or less were considered to be significant (p ≤ 0.05). Data were expressed as a percentage of the control group mean ± SEM, unless otherwise noted. Graphical representation of the results was done using the GraphPad Prism (version 4.03, GraphPad Software Inc), while the statistical analysis was performed using the SPSS 13.0 for Windows (SPSS Inc, Chicago, Illinois, USA).
Results
Rotenone induced a concentration- and time-dependent drop in cell viability
First of all, the best experimental conditions for the model were tested. The results of the Kruskal–Wallis test for each time tested were all significant (H = 23.324, df = 4, p < 0.01; H = 23.216, df = 4, p < 0.01; H = 29.393, df = 4, p < 0.01; for 24, 48 and 72 h, respectively). Post-hoc analysis by Mann-Whitney test revealed that the cell viability, 24 or 48 h after treatment with 10 nM rotenone, was not significantly different from the control (p = ns for both cases; see Figure 1). This was not the case after treatment with 10 nM for 72 h. In this condition, cell viability was significantly lower than in the control (p < 0.01; see Figure 1). For every time interval tested, concentrations of rotenone equal to or greater than 100 nM caused a significant drop in the cell viability (p < 0.05 for DMSO vs 100 nM rotenone for 24 or 48 h; p < 0.01 for DMSO vs 100 nM rotenone for 72 h; and for DMSO vs. 300 nM or 1000 nM rotenone for 24, 48, or 72 h; see Figure 1). These results indicate that rotenone induced cytotoxic effects on SH-SY5Y cells in a concentration- and time-dependent manner.

The toxic profile of rotenone on SH-SY5Y cells.
Rotenone-induced cytotoxicity after 48 h was fitted to Eq. 1:
Effects of Paullinia cupana extract on human neuroblastoma SH-SY5Y cells
To search for possible toxic effects, human neuroblastoma SH-SY5Y cells were treated with increasing concentrations of Paullinia cupana extract. As shown in Figure 2 , cell viability, assessed by MTT assay, was not affected by treatment with any concentration of Paullinia cupana extract after 48 h of exposition (F(7, 22) = 1.931, p = ns). Therefore, the highest concentrations tested (0.312 mg/mL and 0.625 mg/mL) were chosen for the cytoprotective assays.

The effect of Paullinia cupana extract on cell viability of human neuroblastoma cells (SH-SY5Y).
Cytoprotective effects of Paullinia cupana extract assessed by MTT assay
To study the cytoprotective effect of Paullinia cupana extract, the MTT assay was performed on cells previously treated with DMSO alone, with the addition of 300 nM rotenone, or treated with the addition of both 300 nM rotenone and one of two concentrations of the extract (0.312 mg/mL or 0.625 mg/mL). The ANOVA was significant (F(3, 10) = 81.989, p < 0.01). Post-hoc tests revealed that after the addition of 300 nM rotenone, the cell viability significantly dropped to 58.15% of the control condition (p < 0.01. See Figure 3), while the addition of 0.312 mg/mL of the extract increased cell viability to 83.04% of the control. This increment was significantly higher than the one observed in the treatment with 300 nM rotenone without the addition of the extract (p < 0.01; see Figure 3), but it was significantly lower than in the control (p < 0.01; see Figure 3). These data indicate a partial protection of the deleterious effects of rotenone on SH-SY5Y cells by the addition of 0.312 mg/mL of Paullinia cupana extract. Moreover, the addition of 0.625 mg/mL of the extract produced an even higher reduction of the cytotoxic effect of rotenone, since the cell viability increased to 95.45% of the control condition, and this percentage was significantly higher than in the treatment with 300 nM rotenone without the addition of the extract (p < 0.01; see Figure 3), and it was not significantly different from the control (p = ns; see Figure 3).

Cytoprotective effects of Paullinia cupana extract on rotenone-induced cell damage. The cell viability (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide [MTT] assay) has dropped significantly after treatment with 300 nM rotenone. **p < 0.01, in comparison to the control; †† p < 0.01, in comparison to treatment with 300 nM rotenone.
Effects of Paullinia cupana extract on rotenone-induced alteration of membrane integrity
Changes in membrane integrity were also assessed after 48-h treatment. Extracellular LDH levels have not shown significant changes neither by the presence of DMSO nor by the presence of DMSO with the addition of any of the chosen concentrations of Paullinia cupana extract, since the ANOVA was not significant (F(3, 8) = 1.346, p = ns; see Figure 4a). By contrast, the treatment with rotenone or with rotenone and the extract induced statistically significant changes of the extracellular levels of LDH, since the Kruskal-Wallis test was significant (H = 7.513, df = 3, p < 0.05). Post-hoc analysis by Mann-Whitney test revealed that the treatment with 300 nM rotenone, without the addition of the extract, induced a significant increase of the LDH levels in comparison to the LDH levels in the control (p < 0.05), indicating a rotenone-induced alteration of membrane integrity (see Figure 4b). When a concentration of 0.312 mg/mL of Paullinia cupana extract was added, LDH levels were significantly lower than the LDH levels induced by treatment with 300 nM rotenone alone (p < 0.05; see Figure 4b), and moreover, no significant changes were detected in comparison to the LDH levels in the control (p = ns; see Figure 4b), indicating a total reversion of the damage to membrane integrity induced by rotenone. Unexpectedly, when a concentration of 0.625 mg/mL of Paullinia cupana extract was added, LDH levels were not significantly different neither from those induced by treatment with 300 nM rotenone alone nor from those in the control (p = ns for both comparisons; see Figure 4b).
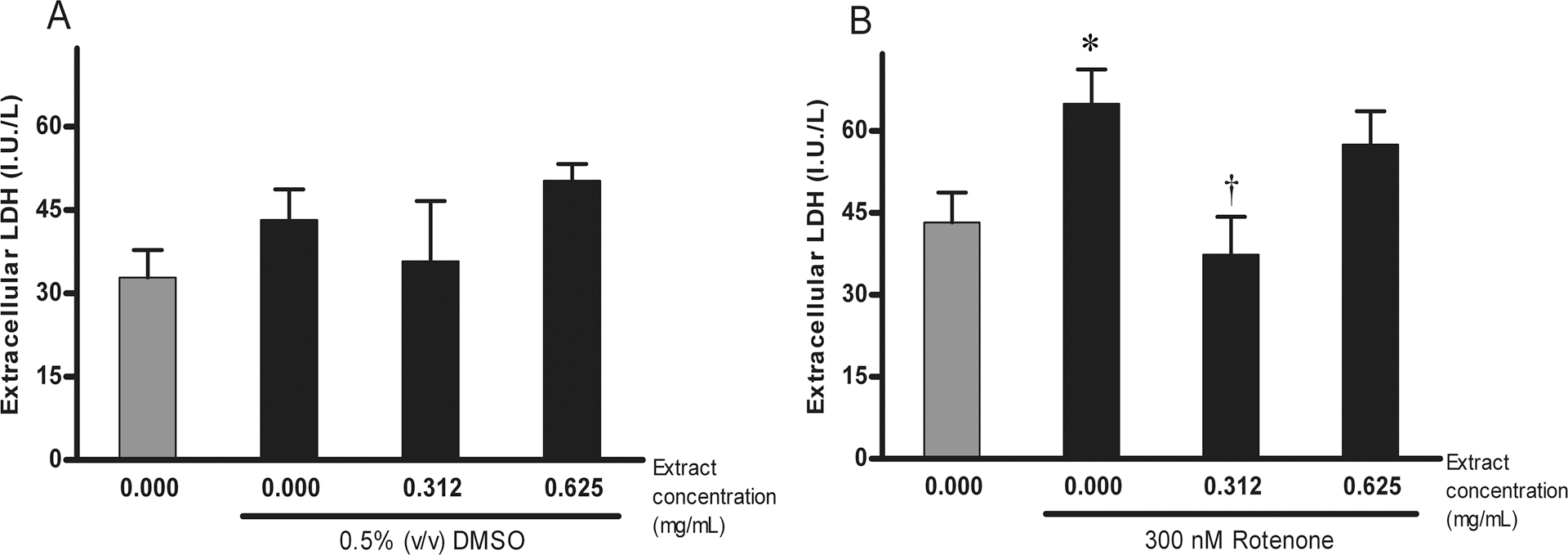
Lactate dehydrogenase (LDH) concentration in rotenone-induced alteration of membrane integrity and the effect of Paullinia cupana extract. *p < 0.05, in comparison to the control condition (DMSO); † p < 0.05, in comparison to treatment with 300 nM rotenone.
Treatment with Paullinia cupana extract reduces rotenone-induced condensation of chromatin and nuclear fragmentation in human neuroblastoma SH-SY5Y cells
Human neuroblastoma SH-SY5Y cells that were treated with rotenone showed nuclear condensation and fragmentation (see Figure 5). We have previously seen this effect of rotenone on SH-SY5Y cells. 29 The quantification of condensed/fragmented nuclei stained with Hoeschst 33258 (see section 2.7 for a description of the procedure) revealed significant differences between the previously mentioned conditions (F(3, 12) = 139.767, p < 0.01). Post-hoc pairwise comparison by Tukey's HSD test showed that the percentage of the total number of nuclei, that were condensed or fragmented, after treatment with 300 nM rotenone alone (79.32%), were significantly higher than those seen in the control (p < 0.01; see Figure 6), which were only 1% of the total. The addition of a concentration of 0.312 mg/mL or 0.625 mg/mL of Paullinia cupana extract significantly decreased the percentage of condensed/fragmented nuclei to 30.88% and 36.56%, respectively, in comparison to those seen after treatment with 300 nM rotenone alone (p < 0.01, for both comparisons; see Figure 6). Nevertheless, the percentages of fragmented/condensed nuclei after treatment with any of the concentrations of the extract were significantly higher than the 1% of the control (p < 0.01, for both comparisons; see Figure 6). These data indicate that Paullinia cupana extract exerts a partial protective effect on rotenone-induced apoptosis in human neuroblastoma SH-SY5Y cells.

Nuclei stained with Hoeschst 33258 (a) in control conditions and (b) after 48 h of treatment with 300 nM rotenone. Note the nuclear fragmentation (arrows) and nuclear condensation (head of arrow) after treatment with rotenone.

Quantification of condensed/fragmented nuclei. The percentage of condensed or fragmented nuclei was significantly lower in extract/rotenone-treated cells than in rotenone-treated cells. DMSO (0.5% v/v) was present in every condition. Bars and error bars represent means ± SEM. **p < 0.01, in comparison to the control condition; †† p < 0.01, in comparison to treatment with 300 nM rotenone alone.
Discussion
Parkinson's disease is increasingly becoming a health priority since the worldwide population is getting older and age is the main known risk factor. 30 This disease remains without an effective treatment, although several therapeutic strategies to control its symptoms, with different degrees of success, are available. 31 The experimental model used in this study has been used previously in recent scientific works. 29,32,33 This strategy could procure relevant information related to some of the mechanisms that underlie PD.
In the last decades, a growing body of evidence has added support to the hypothesis that links mitochondrial dysfunction and oxidative stress with the pathogenesis of PD. 30,34,35 For instance, a history of exposure to the mitochondrial toxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) was associated to the development of motor symptoms resembling those seen in PD. 36 The active metabolite of MPTP, 1-methyl-4-phenylpyridine (MPP+), induces degeneration of the nigrostriatal dopaminergic system, mainly due to its inhibitory action on mitochondrial complex I activity. 37 –40 Moreover, it was found that complex I activity is decreased by 30% to 40% in the affected brain areas in idiopathic PD. 41 It has also been reported that coenzyme Q10 (CoQ10), a naturally occurring antioxidant, which accepts electrons from complexes I and II in the mitochondrial electron transport chain, was able to reduce the in vitro cytotoxicity of rotenone against SH-SY5Y cells 42 and showed neuroprotective effects in an in vivo model of PD. 43 Also, it is worth pointing out that it was found that CoQ10 has its levels reduced in mitochondria from individuals who suffer from PD. 44 These and other evidences motivated recent clinical trials that are aiming at whether CoQ10 might slow disease progression, 44 pointing once more oxidative stress targeting therapies as promissory approach for future treatment of PD. CoQ10 is also a good example of a compound with potential antiparkisonian action identified by in vitro tests using the same model of this work.
The long-term occupational exposure to pesticides, such as rotenone, which is a well-known inhibitor of mitochondrial complex I and can cross the brain-blood barrier due to its lipophilic properties, is also significantly associated with the development of PD. 45 –47 In the present study, we demonstrated that the treatment with extract of commercial Paullinia cupana seeds significantly reduced the rotenone-induced cytotoxicity in human dopaminergic neuroblastoma cell line (SH-SY5Y).
The tested extract demonstrated interesting activity in all cell viability tests. MTT assay clearly demonstrated this cytoprotective effect, which was partial at 0.312 mg/mL but total at 0.625 mg/mL of the extract. However, when cells exposed to rotenone were also treated with this concentration, the cell viability was not different from that of control. More interestingly, LDH levels in supernatants showed that 0.625 mg/mL of extract, which was not toxic in our experiments, was lesser effective than 0.312 mg/mL in membrane-integrity maintenance. The protective potential of Paullinia cupana indicated by cell viability tests was confirmed by nuclear morphology analysis, suggesting an anti-apoptotic action, as rotenone-mediated oxidative stress leads to apoptosis. 48
A possible underlying mechanism for this protection can be associated with the antioxidant potential of Guaraná that has compounds with radical-scavenging activity. 15 It is of significant importance that purine alkaloids have been identified between Paullinia cupana constituents, 18 since they are possible cytoprotective compounds due to their well-known antioxidant properties. 19 –21 Another antioxidant compound usually found in Paullinia cupana extracts are phenols, such as clorogenic acid and free phenolic acids, which are capable of scavenging free radicals, providing the antioxidant activity of these plant extracts, 49 as well as procyanidine B2, which has also strong antioxidant activity. 17 Other evidences supporting the antioxidant properties of Guaraná came from the ability of its ethanol extracts to reduce lipid peroxidation in 3T3-L1 cells 23 and in rat brain homogenates. 50 These antioxidant activities have also been observed in other plants from genus Paullinia 51 as well as in other species such as Valeriana officinalis, Anemopaegma mirandum, and Ginkgo biloba. 11,29,52 In fact, pretreatment with a standardized extract of Ginkgo biloba L. (EGb 761) reduced the motor deficits in the 6-OHDA rat model of Parkinson's disease. 53
In vitro studies demonstrated that the antioxidant effects of Paullinia cupana are also related to the inhibition of spontaneous oxidative processes, probably due to its high concentration of tannins, 50,54 which are known to present protective effects against DNA damage. Moreover, the genotoxic and cytotoxic damage produced by N-nitrosodiethylamine (DEN) on mouse liver cells was significantly reduced by treatment with a Guaraná extract. 55 In the present study, the treatment with a commercial extract of Paullinia cupana has significantly reduced the rotenone-induced condensation of chromatin and nuclear fragmentation in human neuroblastoma SH-SY5Y cells.
Rotenone administration leads to oxidative stress, and postmortem studies have consistently supported the theory that oxidative damage occurs in PD pathogenesis. 1 Therefore, compounds with potential antioxidant activity, like some of the constituents of Guaraná, are candidates for new therapeutic agents, since perspectives for treatment of PD in the future could include antioxidant therapy. 56 –58
The results of the present and other studies can be used as precedent in vivo experiments and clinical trials in the search of new adjuvant therapies for PD, although further studies will be needed to reveal which of the active constituents of Paullinia cupana are responsible for the cytoprotective effects against rotenone-induced cytotoxicity in SH-SY5Y cells. However, it is important taking into account that, perhaps, the phytocomplex is responsible for the observed effects. Therefore, the therapeutic use of Guaraná as phytotherapic agent may be considered of potential interest.
Footnotes
Pablo Galeano and Juan Ignacio Romero are fellowship holders from CONICET (Argentina).
This work was supported by Grant from Fundação de Amparo a Pesquisa do Estado da Bahía (PRODOC/ FAPESB 016/2004; Brazil), Conselho Nacional de Desenvolvimento Científico e Tecnológico (FAPESB/CNPq 159/203; Brazil), Agencia Nacional de Promoción Científica y Técnica (ANPCyT BID 1728/OC-AR PICT 15001; Argentina), and University of Buenos Aires Grants M047 and M020.
Acknowledgement
We thank Jorge Joaquín Llambías for his helpful comments on English language, and Deyse Valverde G de Andrade for technical assistance.


