Abstract
This is a visual representation of the abstract.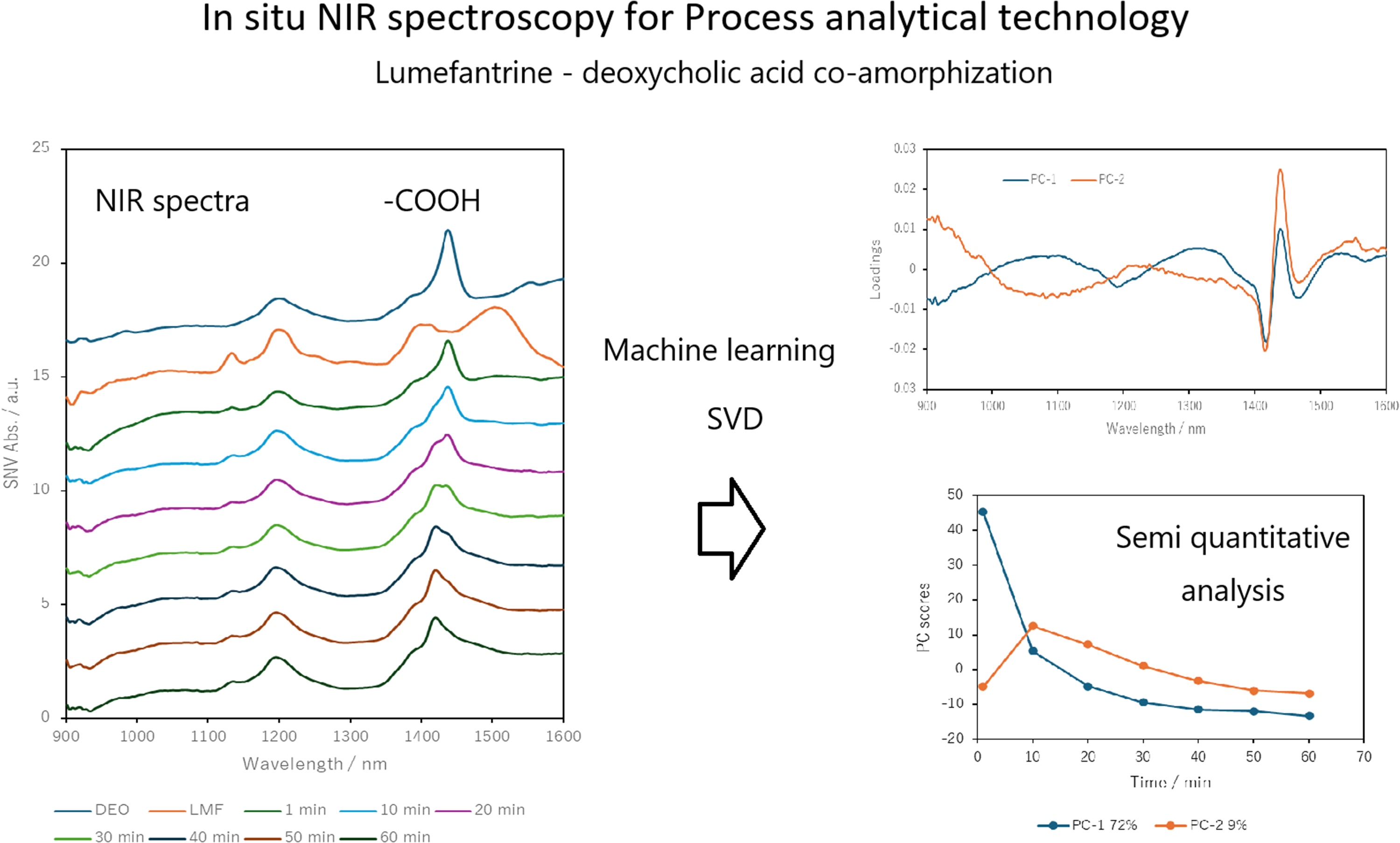
Background
Various methods have been reported for improving the water-insoluble drugs in oral administration formulations. Among them, amorphization has been attracting attention and developed as a method for solubilizing API (active pharmaceutical ingredient)s by changing their physicochemical properties. Molecular complexation is also known as a method for solubilizing APIs by synthesizing cocrystals, etc. Co-amorphization, which achieves both molecular complexation and amorphization, is effective and has attracted attention. Thus, co-amorphization has been proven to be an effective approach to solubilization.
Objective
This study aims to improve the solubility of lumefantrine, used here as a model compound, through co-amorphization with deoxycholic acid.
Significance
The physicochemical properties are an important factor in developing pharmaceutical ingredients. Hydrogen-bonded co-amorphization has gained attention as a method to enhance the physicochemical properties of hydrophobic drugs.
Methods
The co-amorphous Lumefantrine-deoxycholic acid system was prepared using a mechanochemical synthesis method based on ball milling. The synthesis process was monitored by powder X-ray diffraction and near-infrared spectroscopy. The products and materials were analyzed by thermal analysis.
Results
Spectroscopic analysis revealed that the two molecules were complexed through intermolecular hydrogen bonding interactions. The produced co-amorphous has no melting point was found by thermal analysis.
Conclusions
Process monitoring also indicated the presence of a metastable crystalline Lumefantrine (LMF) intermediate.
Get full access to this article
View all access options for this article.
