Abstract
The emissions from internal combustion engines (ICEs) contribute to climate change, air pollution, and pose risk to human health. This review is the first part of a research study that aims to employ computational fluid dynamics (CFD) to develop a clean and efficient combustion strategy focussing on ammonia as an alternative fuel. This article discusses background related to ammonia economy, safety, and its impact on the environment and human health. Combustion characteristics of ammonia are analysed in this article. The review explores various techniques such as blended and dual fuels that are employed to optimize the ignition, including developments in recent ammonia commercial engines. It discusses challenges for minimizing nitrogen-based emissions and also delves into investigations of hydrogen generation through ammonia decomposition, with a focus on microwave heating as an efficient method. Literatures on CFD models tailored to ICEs are also examined. The methodologies of CFD model and techno-economic approach of ammonia decomposition by microwave heating are described. Key findings referred that nearly 4% of ammonia production currently utilizes sustainable sources. Hydrogen with selective catalytic reduction reduces the temperature of reaction to below 200 °C and produces water vapour as a byproduct. Ammonia decomposition by microwave heating occurs at temperatures 100–200 °C lower than conventional methods. Moreover, the localized ammonia decomposition by microwave heating for hydrogen production proves to be 4.5% more efficient than hydrogen storage for transportation. The preliminary CFD investigation from this study suggests the feasibility of using ammonia as a carbon-free fuel for spark ignition ICEs. Compared to methane (CH4) combustion, ammonia shows a 2.5% increase in thermal efficiency and approximately 40% reduction in NOx emissions. This study lays the groundwork for future exploration of ammonia and hydrogen as potential cornerstone for cleaner and more sustainable ICEs.
Introduction
There is a crucial international demand for reducing carbon emissions, as well as replacing fossil fuel to meet Net Zero carbon emissions target by 2050. 1 The emissions are gases discharged from fossil fuel combustion. The sources of emissions are classified in USA into about 28% transportation, 25% electric power generation, 23% industrial plants, 13% commerce and residence, and 10% agriculture. 2 These emissions are called greenhouse gases (GHGs) and nitrogen oxides. GHGs impact on the environment, which contributed to rise the global average temperature by 1.2 °C between approximately 1900 and 2020 as documented in the sex assessment report by Intergovernmental Panel on Climate Change (IPCC). 3 The effects of emissions are global warming, ice melting, desertification, acid rains, and ozone layer depletion. 4 The prolonged exposure to polluted air causes heart attack, respiratory illnesses, asthma, and lung cancer. 5 The implications of these conditions can be increase in number of hospital admissions, emergency rooms, and death. Additionally, the demand for energy is expected to be two to three times higher by 2050 due to the growing world population and economic development, while fossil fuels are finite resources which will be depleted in next generations. 6 Therefore, it is vital to achieve decarbonization or Net Zero carbon emissions by switching from fossil fuel to green-renewable energy, 3 and cut off carbon emissions from transportation, 7 power, 7 industry 8 and commercial sectors. 9 A critical step in this process is identifying a replacement to fossil fuels used for internal combustion engines (ICEs). The substitute would be an energy having no carbon, sustainable, efficient, and cost-effective. Hence, research efforts in the last years have been concentrated to reduce emission of exhaust gases, 10 and particulate matter 11 using various techniques centred in traditional fuel replacement, energy cell employment, hybrid system use, combustion process development, and in-situ emissions capture. The fossil fuels can be altered or mixed with biofuels. There are several research studies that used this technique such as biodiesel, 12 biodiesel-diesel-ethanol, 13 biodiesel-alcohol, 14 and biodiesel-hydrogen 15 in diesel engine, and biogas as a clean energy for power generation and heating. 16 Another technique related to fuels is using water injection in gasoline injection engine, 17 and water emulsion fuel. 18 Furthermore, techniques of hybrid systems can be seen in research, such as power plant combined with solid oxide fuel cells, 19 and power generator with waste of thermal energy and R245fa Rankine cycle, 20 solar organic Rankine systems, 21 and hybrid batteries – diesel generator. 22 Recently, a technique that has been reported to reach a high-level of efficiency with low-level of emissions, which is a homogeneous charge compression ignition (HCCI) engine fuelled with hydrogen and nitric oxide. 23 On the other hand, air pollution control devices, 24 and metal-aided microwave irradiation for in-situ fast soot oxidation, which regenerates the diesel particulate filter while minimizing energy cost 25 are techniques used to develop emission trappers in ICEs. Overall, many techniques are used to reduce the emissions, however, there is another promising method for fuelling vehicles by clean energy that requires further study, which is ammonia.
The objective of this article is to carry out a literature review as a foundation for a broader study aims to developing a CFD model for ammonia combustion in ICEs to investigate critical aspects, such as effect of concentrations of fuel components (ammonia and hydrogen), engine design parameters, and operating conditions on ICE performance and emissions. This review focuses on the state-of-the-art ammonia combustion technology, combustion characterization of ammonia, economy, safety, environmental and health impacts, techniques for reduction of NOx emissions from ICEs, and computational modelling of ICEs, supported with research methodology and preliminary results of CFD investigation.
Economy and safety
Ammonia is produced in the form of fertilizers for agricultural purposes. It is produced by using a reaction between nitrogen and hydrogen under a catalytic system. Nitrogen is secured from air, which contains 78% nitrogen. Hydrogen can be obtained from water breaking. The annual manufacture of ammonia is about 180 million tons, which contributes to about 2% of global energy production. 26 The Haber–Bosch method is primarily employed to manufacture ammonia. Productions can be categorized into two according to their sources. First production is called brown ammonia, which is synthesized by extracting N2 via liquefaction from air and reforming of natural gas or coal producing H2. 27 This process produces a byproduct of about 290 Mt of CO2 as an annual global total. The production of ammonia, mainly, via fossil fuel consumes about 1.8%–3.0% of all global energy. Also, it is possible to produce ammonia from electrolysed hydrogen, where the estimated production is around 0.5% of global ammonia. 28 The second type is named green ammonia as it is environmentally friendly. 27 In this type, ammonia is synthesized by utilizing a sustainable source, such as wind, solar, ocean tidal, thermal energy, nuclear, and biomass fuel as a power for water electrolysis producing H2. 29 The amount of ammonia produced utilizing renewable sources is roughly 4%. 30 Power consumption to produce 1 kg of green NH3 is about 14.248 kW/kg, 31 while for brown NH3 it is estimated by 17.113 kW/kg. 32 Currently, Envision has officially commissioned the largest and advanced green hydrogen and ammonia production plant in the world located in Chifeng, China. The plant is completely supplied by power produced from wind and solar-based systems and delivers 320,000 ton of green ammonia yearly. 33 Ammonia can compete hydrogen economically as shown in Table 1.
Comparison between ammonia and hydrogen related to economy.

Notes: The efficiency by energy means the division of produced or stored or carried content energy on consumed chemical and electrical energies. Transport efficiency calculated over a distance of 1000 mile.
Significantly, the use of H2 vehicles needs a great and costly infrastructure. Construction of stations for hydrogen fuelling is beneficial when there is an increase in the amount of H2 fuel vehicles production, nevertheless, vehicle production of H2 fuel may be halted if the infrastructure of hydrogen is improperly grown. 37 However, another way to provide hydrogen is ammonia decomposition. The speculated cost of unit H2 produced from ammonia decomposition including capital and operating costs was about 6.27 $/kg for 30 m3/h while the cost of hydrogen production from gasoline was 10.65 $/kg. The capital cost of ammonia decomposition was calculated as 10.8% and the operating cost was 89.2% of the total cost considering the use of Ru-based catalysts (Ru/La(x)-Al2O3). 38 In addition to that ammonia is possible to be stored as a gas or liquid in tanks. For example, insulated tanks can be used to store about 50,000 ton of ammonia at temperature of −33 °C and pressure of 1 bar. Liquid NH3 may be stored in 60 km3 tank, which contains energy near to 211 GWh, equals to an annual generation of around 30 wind turbines. 39 The profit is that NH3 production can be sustainable from variable sources, cost effective, world-wide storage, carried by vehicles with large amount and low cost, and safe. 36
Environmental and health impacts
Ammonia is considered by National Fire Protection Association, USA as a toxic substance without colour, having a strong-penetrating odour. 26 The effect of ammonia on human health depends on concentration and time of exposure. Table 2 shows different legislations about limitations of ammonia exposure and their effects on human health. NH3 is an alkaline, which has the ability to react with other materials such as acid, halogen, and oxygenizing agent. 26 Ammonia mixing with water (pH 11.6) is a corrosive liquid for metals, such as copper, brass, and zinc creating a greenish/blue colour. 26 Interestingly, hydrogen easily diffuses through metals making metal parts more brittle and capable of breakage. 40 Anhydrous ammonia is non-flammable while vapour of ammonia in air is flammable, and it is possible to explode if it is exposed to ignition, however, ammonia is less explosive than other fuels.
A view on legislations/guidelines relating to ammonia exposure limitations.
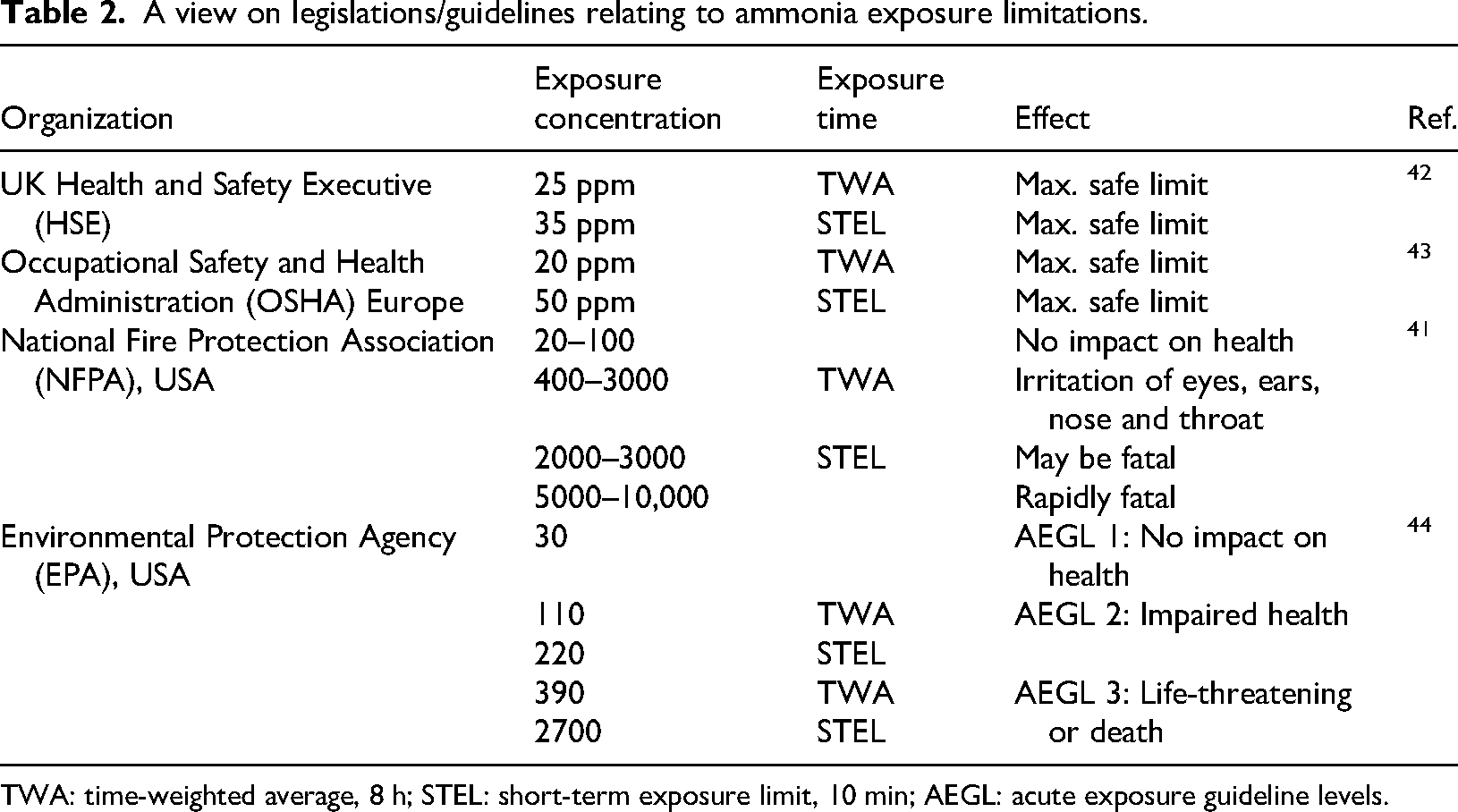
TWA: time-weighted average, 8 h; STEL: short-term exposure limit, 10 min; AEGL: acute exposure guideline levels.
The risk assessment of ammonia leakage on health and environment comparable with other fuels is illustrated in Table 3. 41
Ammonia impact on health and environment when compared to other fuels.

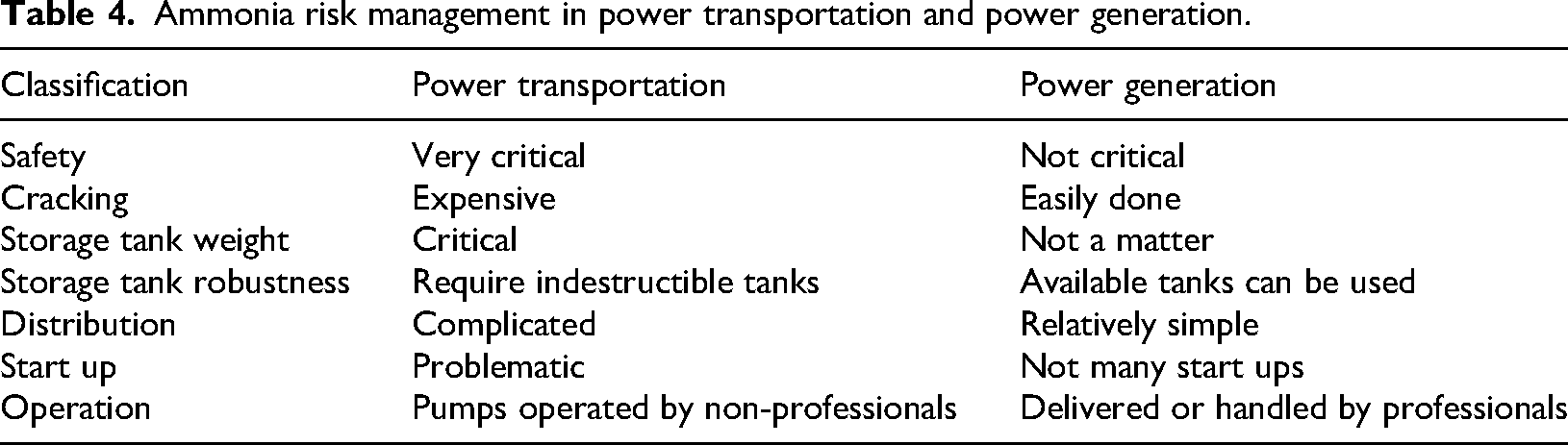
The risk management of ammonia use is dependent on the industrial environments, whether static plants such as electricity production and power generation or transportation, especially vehicles, which is classified in Table 4. 41
Ammonia risk management in power transportation and power generation.
On the other hand, ammonia depletes ozone indirectly via the production of NOx in the atmosphere.
45
Nitrous compounds are catalytically destroying ozone according to the following equations:



An investigation on the negative health impact of environment pollution on Pearl River Delta, which is a region of a significant economy in China, concluded that the mortality through a short term in 2013 reached between 13,217 and 22,800, and the loss in total economy was 1.4%–2.3% of the local gross domestic product due to NO2 and PM10 emissions, and O3 depletion. 46
Ammonia characterizations for combustion
This section discusses five fields of research concerning NH3 as a fuel, which are (1) ammonia as an alternative clean fuel; (2) combustion features; (3) characterization of blended fuel; (4) characterization of dual fuel; (5) commercial ammonia ICEs.
Ammonia as a clean alternative fuel
Ammonia can be a clean fuel and alternative to fossil fuels because it contributes significantly to reduction of CO2 emissions compared with carbon-based fuel. 47 Also, a review 48 showed that ammonia is the best replacement for hydrogen to be used as a fuel for engines for multiple justifications. Firstly, ammonia is possibly supplied as a liquid in tanks at atmospheric temperature and low pressure. Secondly, guides of safety and management for distribution and processing of NH3 are internationally published. Thirdly, the products of combustion are water and nitrogen. Moreover, it is possible to add ammonia to fossil fuels to reduce carbon emissions, where many vehicles could be fueled by 90% gasoline mixed with 10% liquid ammonia or perhaps it will be more in the future. 49 Hydrogen-assisted NH3 is also an alternative clean fuel enhancing NH3 combustion characteristics. 40
Combustion features
The concentrations of ammonia combustion emissions were experimentally measured by using premixed laminar burner fuelled by ammonia mixed with methane at various equivalence ratios (0.8, 0.9 and 1). 50 The measurements included discharges of NOx, CO and unburned NH3 in different conditions. 300 W was the input thermal energy. The molar fraction of ammonia with respect to a mixture of NH3 and CH4 was variable from 0 to 0.7. Results showed that the ammonia fuel had a low flammability carbon-free energy. Amount of NOx emissions grew with an increase in ammonia fraction until it reaches 0.5 of the mixture. Emissions of NOx dropped with equivalence ratio reduction towards lean conditions. Emissions of CO and unburned NH3 were properly low articulating that the combustions of CH4 and NH3 were complete processes. In the same trend, other experiments used alumina–zirconia burner of porous media that was supplied with two types of premixed fuels, which were NH3/CH4/air and NH3/H2/air to measure emission concentrations of NOx, CO, and unburned NH3. 51 The experiment consequences of burning NH3/CH4 and NH3/H2 stated that the ammonia autoignition was conducted at high temperature, the flame speed was low, and vaporization heat was high. The maximum emissions of NOx from NH3/CH4 mixture combustion were at approximately 0.5 of NH3 molar fraction whereas a great emission from NH3/H2 burning was at around 0.5 and 0.8 molar fraction of NH3. The amount of CO emissions referred that the CH4 combustion was complete. However, NH3 existence in exhaust gases meant its combustion was incomplete.
A study that was conducted to predict the ability of fuels containing nitrogen to be employed for each of compression ignition (CI) and spark ignition (SI) engines showed ammonia having the ability to be used for two types of ICEs as a carbon-free fuel. 52 However, there were disadvantages, such as temperature of autoignition and heat of vaporization were very high, speed of flame was low, and flammability was limited by 16–25% (volume in air). It was mentioned that a pure ammonia fuel for CI engines demanded a big compression ratio like (35:1), and fuels of high cetane number can be used with NH3 to enhance the features of ignition. Pure ammonia fuel for SI engines required multi spark plugs of high-power igniter and long duration spark for the purpose of igniting the NH3 at multiple locations to break the resistance of ammonia to combustion. Therefore, the suggestion was using natural gas and synthetic gasoline directly in the vehicle instead of producing ammonia from natural gas and using it as a fuel due to the energy content (LHV) of NH3 is 18.8 MJ/kg while it is 50 MJ/kg for natural gas. Nevertheless, using biomass could be the best sustainable source to produce ammonia.
On the other hand, it was documented that NH3 is a hydrogen carrier, which includes 17.8% hydrogen by mass, and it is available and capable of being stored and transported. It is possible for ammonia to be used for fueling different power usages despite the process of ammonia combustion being of high ignition energy and NOx emissions with low burning velocities. Low flammable speed and NOx emission can be controlled by developing ammonia synthesis catalysts or blended fuels. 53 Therefore, the proposed fuels were hydrogen, dimethyl ether (DME), gasoline, diesel, and biodiesel to be mixed with NH3 to overcome the downsides of ammonia fuel. 54
Results of an experimental work about blending gases of NH3 and H2 with air in SI engine 55 showed that a mixture of up to 20% hydrogen (by volume) frustrated misfires, and gave a better work and efficiency close to stoichiometric conditions. H2 of higher fractions caused reduction in efficiency due to the increase in thermal losses through walls and increased NOx and NH3 slip. The duration of combustion was low due to the low velocity of burning mixtures. A less than 10% volume of H2 to NH3 enhanced the combustion and engine stability.56,57 Best value of hydrogen to ammonia ratio for stability and approximately zero NH3 emissions during the period of cold engine start until warmed up was 2:1 by mole under lean conditions. 58 Hydrogen accelerated the initial combustion and the overall combustion.59,60
The findings of mixing liquid ammonia with dimethyl ether for fuelling high-pressure CI engine 61 revealed that the use of NH3 with dimethyl ether led to delay in ignition and limitation in conditions of engine load because the temperature of ignition was elevated, and combustion rate or flame speed was weak. Pressure and temperature of combustion decreased due to existing of NH3, producing a high emission of CO and HC. It also raised emissions of NOx and NH4.
Ammonia can be mixed with gasoline efficiently through a feature named solubility, which means it can be kept in a liquid phase by using emulsifier, such as ethanol or methanol. Pure gasoline can emulsify NH3 of 23 g/L at temperature and pressure of 286.65 K and 345 kPa, respectively, while gasoline mixed with each of 10% ethanol and 10% methanol keeps about 51.4 g/L and 61.8 g/L, respectively of NH3 in a liquid phase at similar conditions of temperature and pressure. 62 Gasoline content of 30% as an igniter necessitated a 10:1 compression ratio for ideal operation.
A comparison between two dual fuels of NH3/diesel and H2/diesel for a high-pressure heavy-duty engine was run out experimentally and numerically. 63 The major fuels were H2 and NH3 that were directly injected at maximum pressure of 500 bar. H2 and NH3 were ignited by a slight amount of diesel as a pilot fuel. Diesel was directly injected at a pressure up to 1800 bar. The comparison showed that the ignition of ammonia was delayed without any further actions because ammonia has lower flammability, higher evaporation energy and higher ignition temperature than hydrogen. This meant NH3 requiring a high value of activation energy for combustion, which delayed the starting of heat release rate (HRR). However, utilizing high ammonia injection pressure and temperature improved the vaporization speed and shifted the heat release rate early. Additionally, the preheating of NH3 enhanced the combustion. Furthermore, double pilot fuels led to ignite NH3 early. The heating of NH3 to 450 °C with a very high injection pressure showed that the trend of the HRR of ammonia approximately close to the HRR trend of hydrogen. The hydrogen burned almost near to the injector, and a long-term operation might be damaging the injection due to the elevated flame temperature close to the injector.
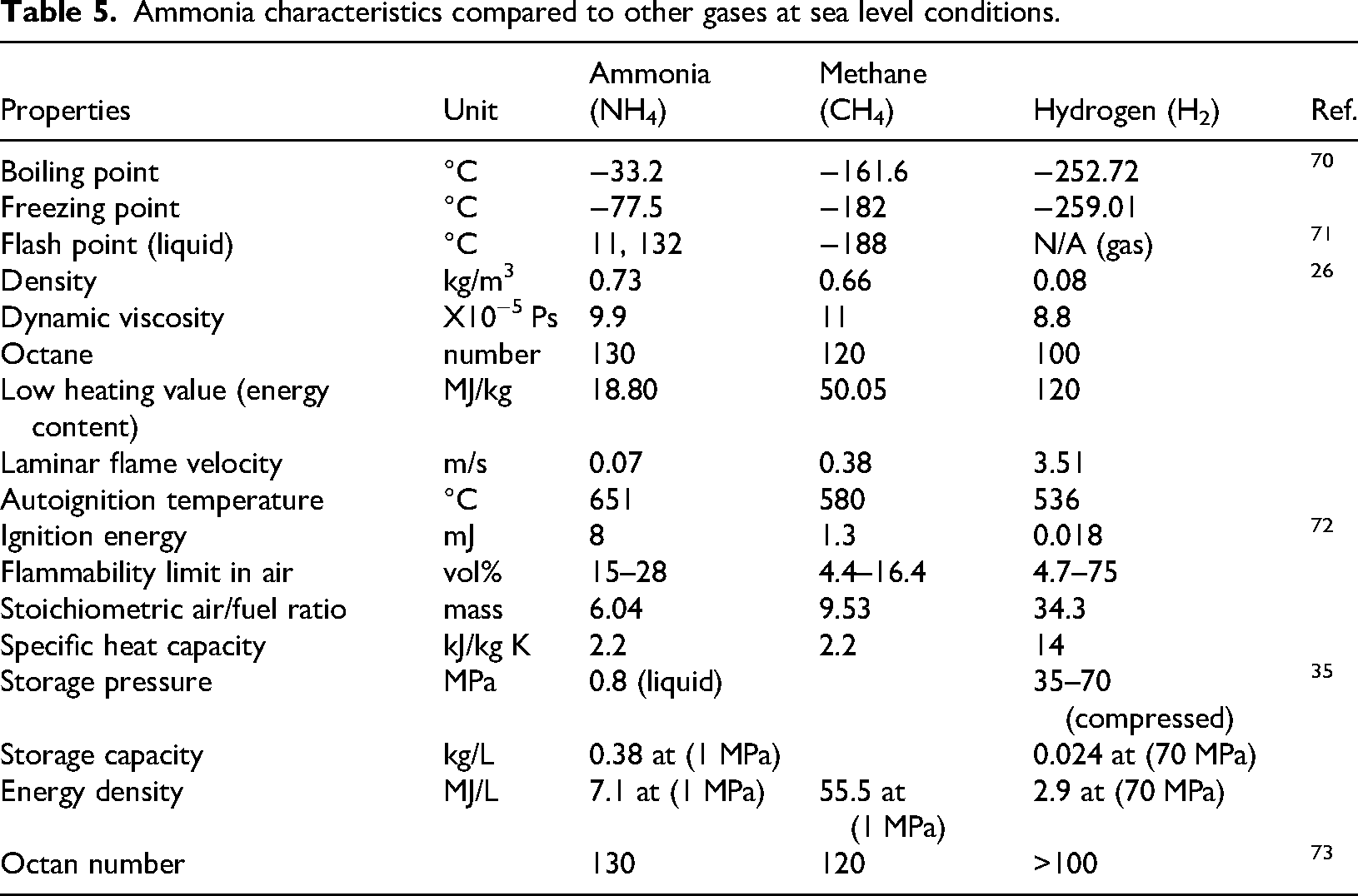
The characteristics of NH3 compared to CH4 and H2 are recorded in Table 5. One of the significant features is that the high-octane number of ammonia (130) resists and reduces knocking in reciprocating SI engines.35,55 An interesting observation from this research is that two different values have been reported for the flash point of liquid ammonia 132 °C64,65 and 11 °C66–69 as shown in Table 5. It can be deduced that more accurate research is required to remove this uncertainty.
Ammonia characteristics compared to other gases at sea level conditions.
Characterizations of blended fuel
Ammonia can be blended with other types of fuels to enhance the features of combustion for the blended fuel. The mixing of ammonia with gasoline prevents knock operation in the SI engine. 74 The amount of fuel mixing was limited to 30% gasoline and 70% ammonia on an energy basis for normal operation in spark ignition engines. Higher ratios of ammonia can be used for engines with superchargers. Subsequently, an experiment compared 100% gasoline with 80% ammonia to 20% gasoline. 75 The results stated that the exhaust temperature increased from 369.9 °C to 480.1 °C because ammonia was ignited under a higher temperature. The power output decreased from 3689.2 W to 3572.8 W due to the energy content of gasoline was higher than that of NH3. Efficiencies of energy and exergy decreased by about 7% and 8.5%, respectively, due to the reduction in output power. The reduction in emissions of CO2 was 0.0434 g/s. Next, it was explored 76 that the combustion of premixed NH3 with H2 in air inside spark ignition engines produced low NO comparable with hydrocarbon fuels. Best conditions for mixture of NH3 with H2 were equivalence ratios of range 1.0–1.05, and hydrogen fractions of 40–60%. Compression ratios of high values were appropriate for the mixture due to the reduction in knock. A recent experimental study on the combustion process of ammonia/hydrogen in air showed that there was an ability to improve the combustion characteristics of ammonia/hydrogen under high fraction of ammonia by employing a turbulent jet ignition. 77 However, in order to tackle the challenges of engine operating at cold start and unstable combustion in a SI engine fuelled with pure ammonia, a numerical investigation was done to crack ammonia by heating it with recycled exhaust gases using reformer producing hydrogen under a temperature of approximately 773 K. 78 The results of mixing ammonia with hydrogen demonstrated that the increase in H2 ratio from 2.5% to 12.5% shortened the duration of combustion by 34.9 CA degrees. Moreover, the pressure inside the cylinder was increased to a maximum value of 7.12 MPa at λ = 0.9 due to the excess in exothermic heat release while the minimum value of the pressure and the heat release rate inside the cylinder were at λ = 1.2 due to the decrease in velocity of burned mixture. Mixing 10% H2 of the reformate at stoichiometric conditions achieved 96.3% combustion efficiency and 43.6% thermal efficiency. NOx emissions were under 1.05 g/kWh at λ less than 1.0 and all values of hydrogen fractions, while the highest value of NOx was 24.4 g/kWh at λ = 1.2 and hydrogen fraction 12.5%. Unburned NH3 increased with a decrease in λ and hydrogen fraction due to the drop in temperatures in crevices preventing the complete combustion of ammonia. N2O emissions reduced with an increase in hydrogen from 2.5% to 12.5% but they increased with λ increase. 78 Table 6 shows a summary about blended ammonia.
Summary about spark ignition engines fuelled by bended ammonia.

Crucially, the research findings in literatures manifest no standard for fuel fraction can combine performance and emissions related to combustion of pure ammonia and ammonia with hydrogen blends. This highlights there is a need for establishing more comprehensive research to innovate the optimum value and condition for ammonia use in ICEs that can be harnessed by Net Zero industry.
Characterizations of dual fuel
The combustion features of ICEs are possibly developed by using ammonia as an additional or secondary fuel. Experimentally, the combustion and emission of a dual fuel of (diesel and ammonia) in CI engine were investigated. 79 The investigation referred that replacing 40% (by energy) or less of diesel with ammonia fuel lowered emissions of NOx compared to 100% diesel fuel. However, emissions of CO and HC were greater due to the reduction in temperature of combustion. Emissions of soot were not significantly affected by low fraction of ammonia. While, replacing 40–60% (by energy) diesel with ammonia caused ignition delay, raising the quantity of unburned NH3 due to ammonia is a big reluctant for autoignition. The peak in-cylinder pressure and combustion efficiency dropped due to the reduction in temperature of combustion. The engine brake specific fuel consumption was high. Moreover, the increase in ammonia fuel above 60% (by energy) led to higher NO emissions and specific fuel consumption. Emissions of soot decreased as a result of the reduction in the amount of soot producer, which was diesel. Emission of NH3 were more than the safe limit. Furthermore, a review about ammonia as a fuel for a CI engine presented that ammonia was a carbon-free fuel that could be employed for diesel engines. 80 However, ammonia fuel as a low autoignition temperature required diesel for combustion. Dual fuel combustion containing ammonia produced huge emissions of unburned NH3 and NOx because ammonia was nitrogen-based fuel. The advanced injection strategies could take part to improve performance of engines and reduce pollutant emissions. Hence, an experiment was conducted using two direct injections in a spark-ignition engine to enhance the engine performance. 81 First injection was gasoline, and the second injection was gaseous ammonia. In the experiment the time taken for ammonia injection was figured out. The results showed the increasing ammonia reduced the engine power. The optimal injection for ammonia was 320 BTDC and 370 BTDC at low load and high load, respectively. The peak in-cylinder pressures of the dual fuel of gasoline and ammonia was a little bit less than gasoline alone due to the depressed value of flame speed and temperature of ammonia fuel lowered the temperature of overall combustion. The efficiency by using ammonia-gasoline was close to efficiency of gasoline alone. The growth in the amount of injected ammonia fuel caused a growth in the emissions of unburned NH3 and NOx, and decreased CO emission. The combustion and emission characterizations of dual fuel (diesel and ammonia) spray engine were studied numerically at low- and high- pressure injections. 82 Diesel was used as a pilot igniter, then diesel and liquid ammonia were injected as a main fuel. The consequences showed that the best ratio was about 80% by energy for ammonia at low pressure dual fuel (LPDF) injection. The raise in ratio of ammonia increased the possibility of misfire happening. The optimum ratio of ammonia at high pressure dual fuel (HPDF) injection was achieved at 97%. The comparison between HPDF injection and pure diesel injection stated that the indicated thermal efficiencies were similar, and HPDF injection decreased the greenhouse gas emissions and NOx with a slight increase in emissions of N2O and NH3. The comparison between LPDF injection and pure diesel injection indicated that LPDF injection has the possibility to rise the indicated thermal efficiency, however, the contribution of LPDF to reduce emission of NOx, N2O and NH3 was limited due to the difficulty in burning ammonia. Recently, it was demonstrated numerically that the utilization of ammonia energy fraction between 30% and 70% in dual fuel (diesel and ammonia) reactivity-controlled compression ignition (RCCI) engine with early injection of diesel indicated mean effective pressure and lower the emissions of HC, CO, N2O, and unburned NH3 compared to diesel RCCI engine. 83 Also, there is another recent research study that employed dual fuel of diesel and ammonia to operate CI engine experimentally using ammonia share from zero to 55% by energy. 84 Research results concluded that the increasing replacement of diesel by ammonia pumped in-cylinder pressure slightly, climbed peak of heat release rate by just above 20 J/deg, and enhanced each of the combustion efficiency and engine efficiency by a range 3%–4% while increased specific fuel consumption by about 100 g/kWh. The increase in ammonia energy fraction from 23% to 55% increased the delay of ignition time, and the duration of combustion. The increase in ammonia shares up to 55% slumped soot emissions from 0.47 g/kWh to 0.012 g/kWh and plunged CO by roughly 25% whereas the trend of CO2 increased by 30 g/kWh at 11% ammonia and then collapsed by 200 g/kWh. The increase in ammonia fraction until 44% by energy increased emissions of NOx by 30% in comparison with using diesel alone. Recently, an experimental work was carried out 85 to predict impacts of ammonia and biodiesel mixture on the efficiency and emission of IC engine. The findings showed that the rise in ammonia fraction to 70% by energy caused a decrease in brake thermal efficiency from 31.8% (pure biodiesel) to 29.4% (highest ammonia fraction) because the combustion of the mixture is unsuccessful at full load. Ammonia boosts the rate of pressure rise. There was a delay in the start of combustion when the ammonia contribution was increased. The emissions of CO2 and CO were reduced by 510 g/kWh and 30.1 g/kWh, respectively when ammonia contribution was maximized to 69.4%. Another work that was carried out to investigate experimentally and numerically the leverage of ammonia/diesel ratio on performance and emissions of engines concluded that the indicated thermal efficiency elevated by growing ammonia to 84.2% as input energy lowered the GHGs compared to diesel, increased NOx and NH3 emissions, produced N2O offset the reduction of CO2. 86 The detailed results showed that in-cylinder peak pressure rose with ammonia increasing to 61.62% by energy. Combustion starting and duration dipped by 32 CA and 6.8 CA, respectively, due to increasing the ammonia energy share. Ammonia at 84.2% by energy dropped drastically the emissions of CO, CO2, and particulate matter, while NO1, NO2, N2O, and unburned ammonia emissions were increased. Ammonia combustion produces N2O of 90 ppm. Additionally, diesel should be replaced with ammonia at 35.9% as a minimum to lower the GHGs emission.
On the other hand, the dual fuel of H2 and NH3 for fuelling an engine of heavy-duty truck was simulated using a special design. 87 The design replaced the spark plug and diesel injector with a fuel injector and added a small volume pre-chamber connected with the main chamber via orifice. The pre-chamber supplied with another spark plug and fuel injector to control the ignition starting. The simulation ran the engine in different modes, and the results revealed that the fuel efficiency was improved due to a reduction in the amount of fuel heat that was used to rise the temperature of the cold NH3 to ignition temperature.
Commercial ammonia internal combustion engines
The major worldwide commercial ammonia-powered ICEs that have undergone full-scale bed tested produced near zero carbon emissions are listed in Table 7, according to the Ammonia Energy Association on March 11, 2025, 88 and NYKline on 14 May 2025. 89
Full-scale tested commercial ammonia fuelled ICEs.
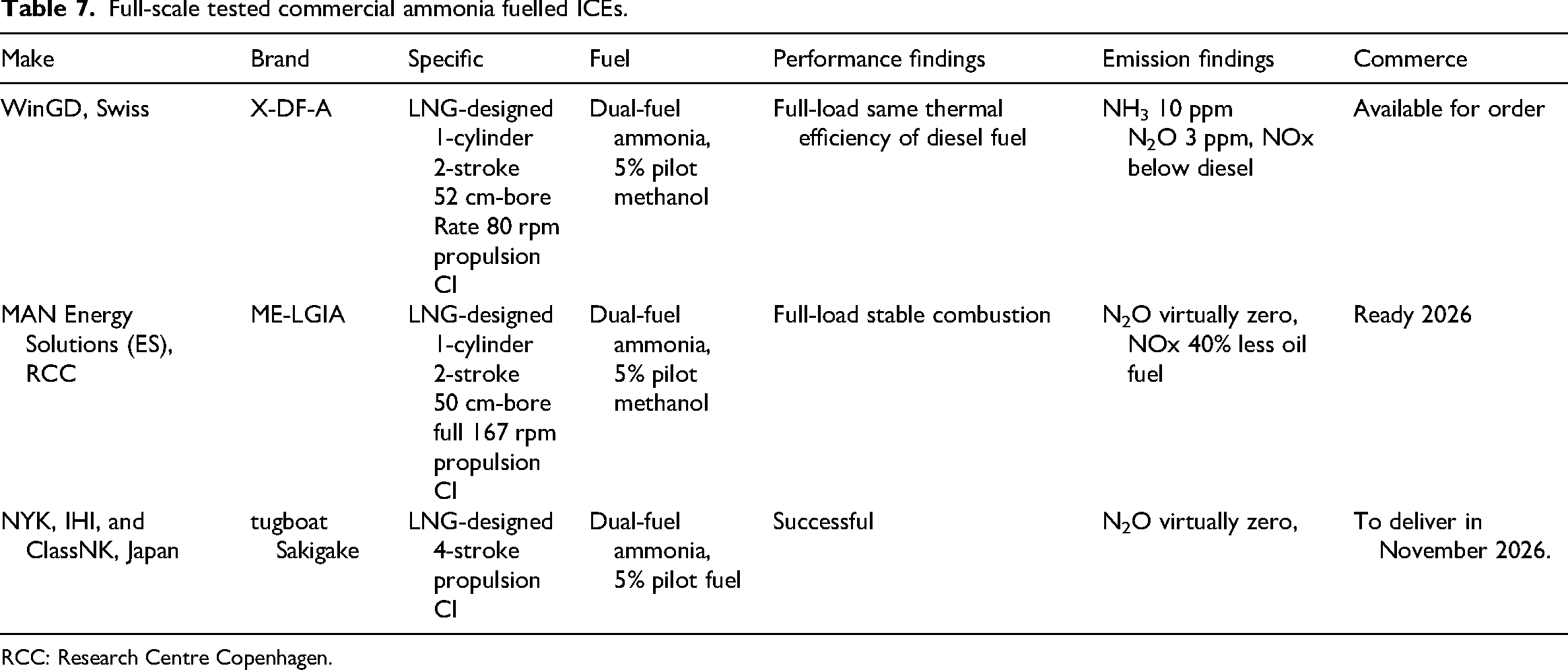
RCC: Research Centre Copenhagen.
However, several companies and institutions are playing a role in development of ammonia powered engines either blended fuel spark ignition or dual fuel compression ignition as listed in Table 8. 70
Key players involved in development of ammonia fuelled engines.

Reduction of NOx emissions from ICEs
The techniques for reduction of NOx emissions from ICEs are classified into (1) general techniques to reduce NOx; (2) techniques for reduction of NOx emissions by ammonia decomposition; and (3) reduction of NOx emissions by ammonia decomposition via microwave.
General techniques to reduce NOx emissions from ICEs
The selective catalytic reduction (SCR) and selective non-catalytic reduction (SNCR) are the most effective techniques used in exhaust gas aftertreatment of cars and ships for NOx reduction. The SCR is activated by injecting the exhaust gas flow with chemicals, such as ammonia, urea, hydrogen, hydrocarbons, and carbon monoxide that react with NOx and unburnt NH3 converting them into N2 and H2O with catalyst assistance. SNCR is an injection of nitrogen content selective reductant, such as NH3 and urea into a post-combustion flue gas to react with NOx, producing N2. Ammonia is an efficient factor for reduction of NOx emissions. Urea is a safe and low-toxicity alternative to ammonia. Hydrocarbon-based SCR systems are lowering operating temperatures and improving efficiency and it is environmentally friendly, particularly in low-temperature operations. 91 Other common techniques for NOx emission reduction are EGR, TWC, LNT, HAM, and fuel emulsions. EGR is exhaust gas recirculation, which recirculates exhaust gas into intake air to lower combustion temperature and NOx. TWC is a three-way catalytic converter, works under stoichiometric conditions, to convert NOx, CO, and HC to N₂, CO₂, and H₂O. LNT is lean NOx trap that stores NOx during lean operation and reduces it during rich operation. Humid air motor (HAM) adds water vapor to intake air to lower NOx. Fuel emulsions (water in fuel) reduce NOx through the decrease in combustion temperature. Table 9 shows the difference between cars and ships related to NOx techniques and regulations.92,93 Also, there are works that were conducted trying to mitigate NOx emissions from ICEs, especially the ammonia combustion engines are demonstrated in Table 10.
Nox emission reductions and regulations used in cars and ships.

Works aimed to reduce NOx emissions from ICEs.

Techniques for reduction of NOx emissions by ammonia decomposition
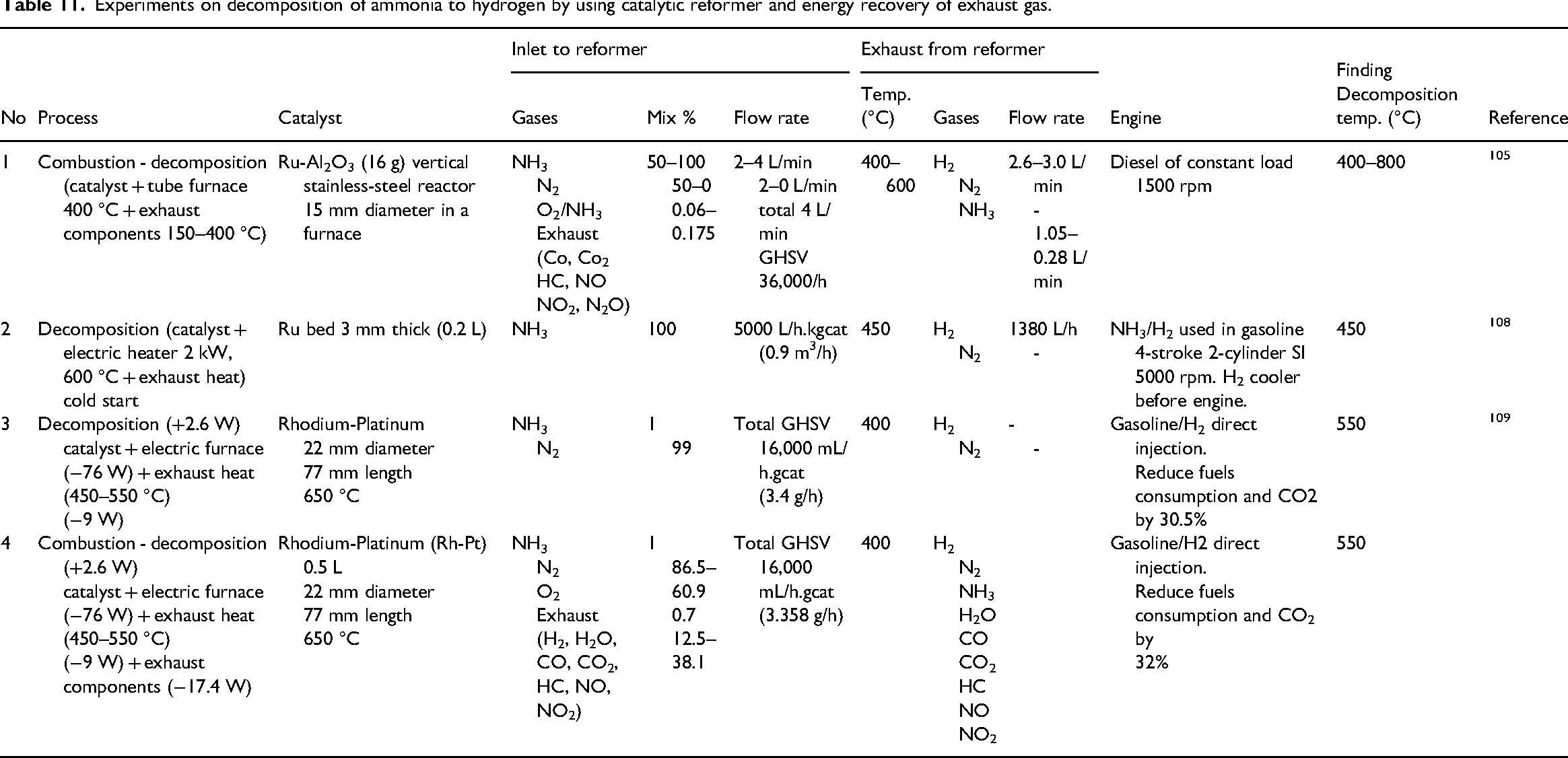
To overcome the challenges associated with ammonia combustion and its emissions, several studies suggested alternative approach such as decomposing NH3 to H2 and N2 using hydrogen as a fuel 103 or ignition promoter. 104 Hydrogen reduces nitrogen-based emissions. However, ammonia decomposition inside an engine's cylinder is difficult to be controlled and the endothermal reaction reduces the temperature and pressure inside the cylinder. Hence, ammonia decomposition should be before the engine intake. The common method that is employed for rising ammonia temperature uses heat recovery of exhaust gases incorporating a reformer.72,78 The required temperature for catalytic decomposition of ammonia is approximately 500 °C, however, the temperature of exhaust gases is less than 400 °C, 105 especially at cold start. Hence, other methods were considered to raise the temperature of ammonia such as, chemical interaction 106 and plasma. 107 Also, catalytic furnaces or electric heaters were used together with exhaust gases to increase NH3 temperature as demonstrated in Table 11.
Experiments on decomposition of ammonia to hydrogen by using catalytic reformer and energy recovery of exhaust gas.
Reduction of NOx emissions by ammonia decomposition via microwave
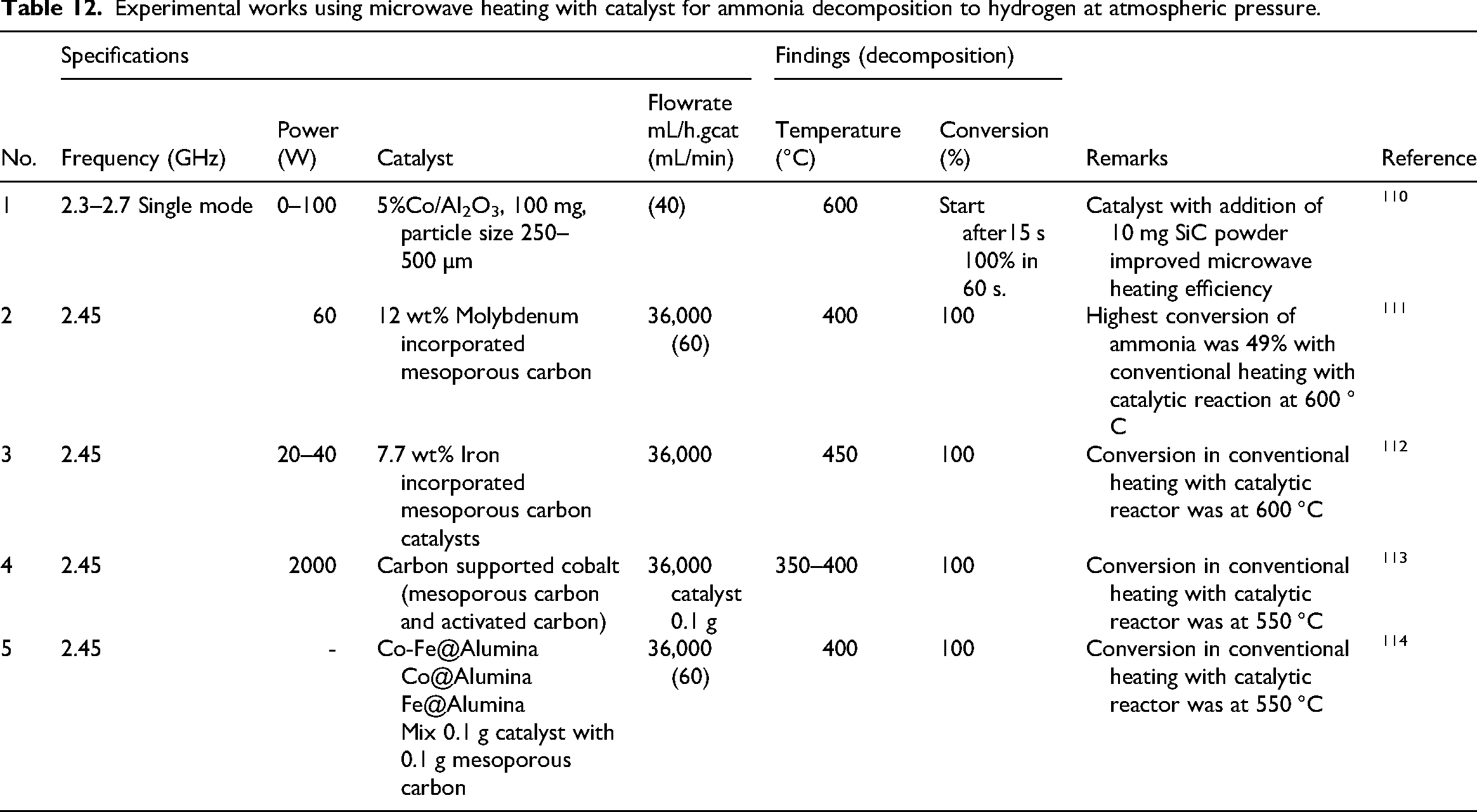
Several experiments used microwave heating with catalyst to assist the exhaust gases or replace them at cold engine start in order to facilitate ammonia heating and decomposition producing hydrogen fuel as shown in Table 12.
Experimental works using microwave heating with catalyst for ammonia decomposition to hydrogen at atmospheric pressure.
CFD studies related to ICEs
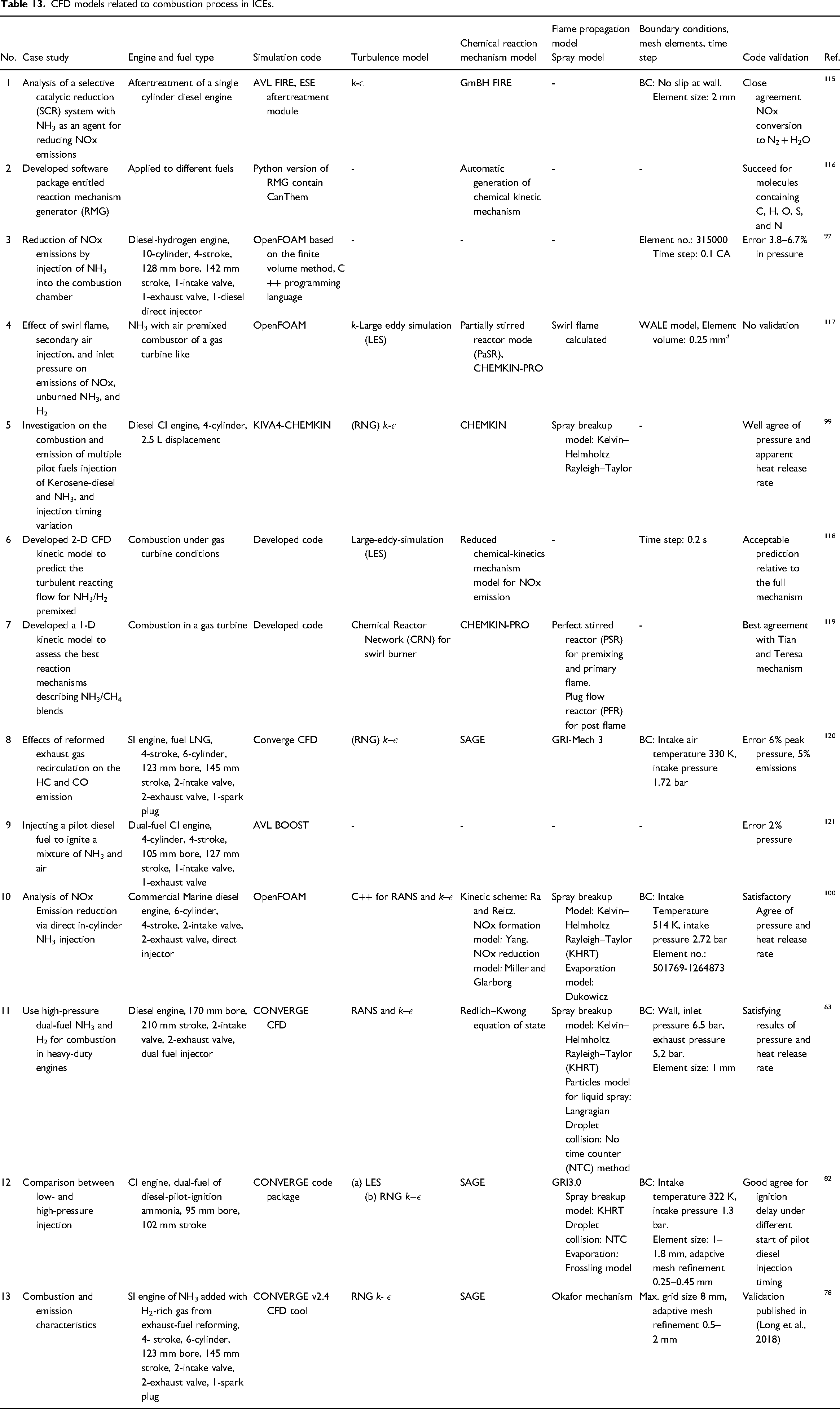
The review of literatures that studied CFD models for analysing ammonia combustion, or performance and emissions of ICEs is shown in Table 13.
CFD models related to combustion process in ICEs.
Methodology
CFD model for internal combustion engines
The methodology for assessing the thermal engine efficiency, work, and power of ICEs involves a comprehensive analysis of various parameters over time and space. These parameters include pressure, temperature, density, chemical heat release, and thermal losses within the engine cylinder. To achieve this, finite volume method with SIMPLE algorithm is employed for solving sets of unsteady three-dimensional governing equations.122–124 These equations encompass mass, momentum, energy, chemical reaction mechanisms, and state equations. Additionally, the methodology incorporates the re-normalized group k–ε model, which is pivotal in accurately modelling turbulent flow. This model is essential for understanding and predicting the complex behaviour of fluids within the engine. Furthermore, flame propagation models are integrated into the analysis providing a more detailed and precise representation for the process of combustion. The flowchart in Figure 1 shows the methodology of CFD model for spark-ignition engines that is designed by authors of this article. This holistic approach ensures a thorough understanding of the internal dynamics of ICEs, leading to more efficient and environmentally friendly engine designs. The CFD investigations were applied on two different premixed spark ignition ICEs as shown in Table 14, by employing ANSYS Forte. The computational geometry was built with moving piston and valves. The computational mesh of the engine geometry was generated automatically on-the-fly during the simulation by Ansys Forte using 2- and 2.5-mm global mesh size and refined by utilizing additional meshes as shown in Table 15. The CFD model setup are stated in Table 16.
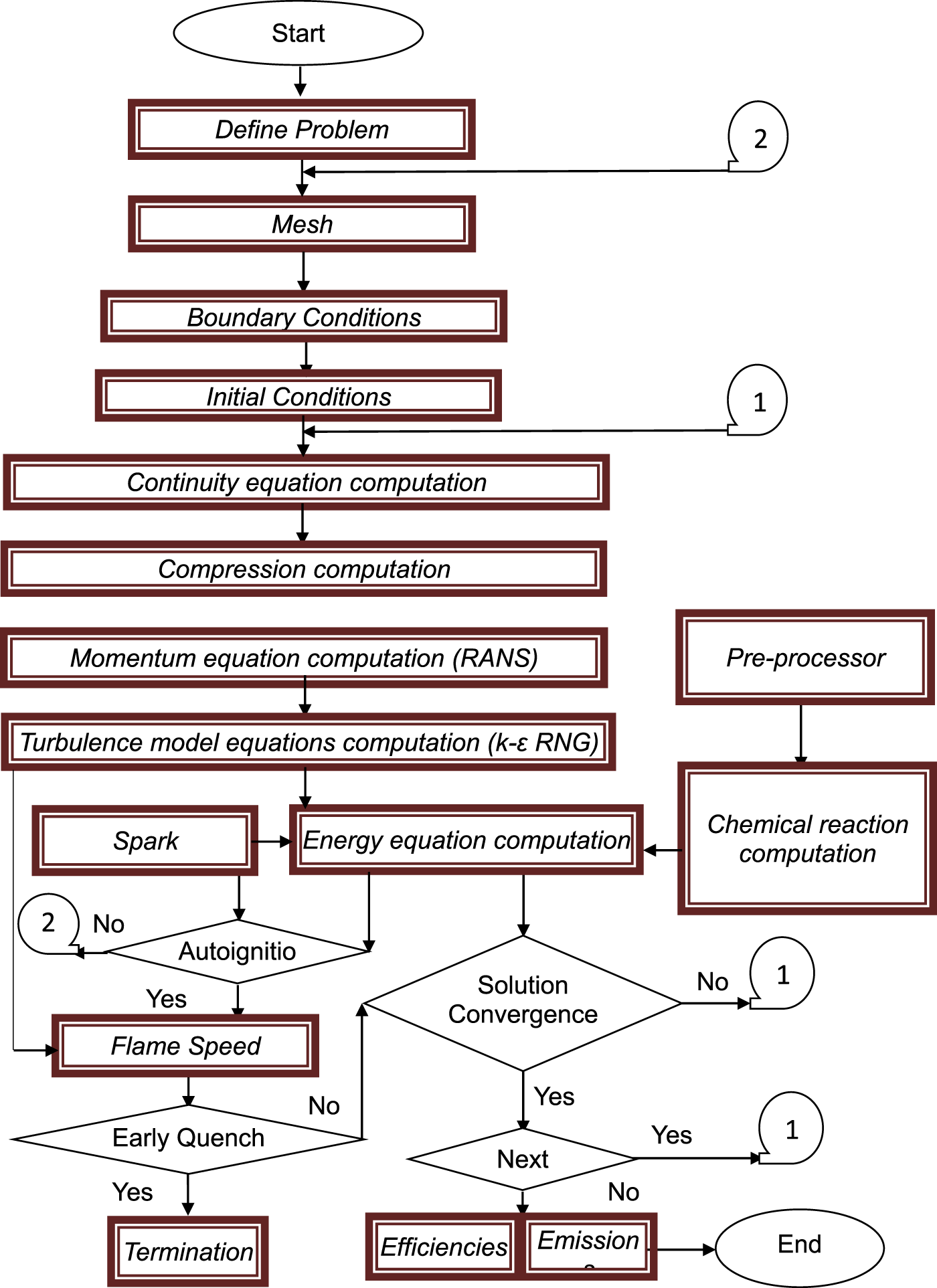
Flowchart of the methodology of CFD model for spark-ignition ICE.
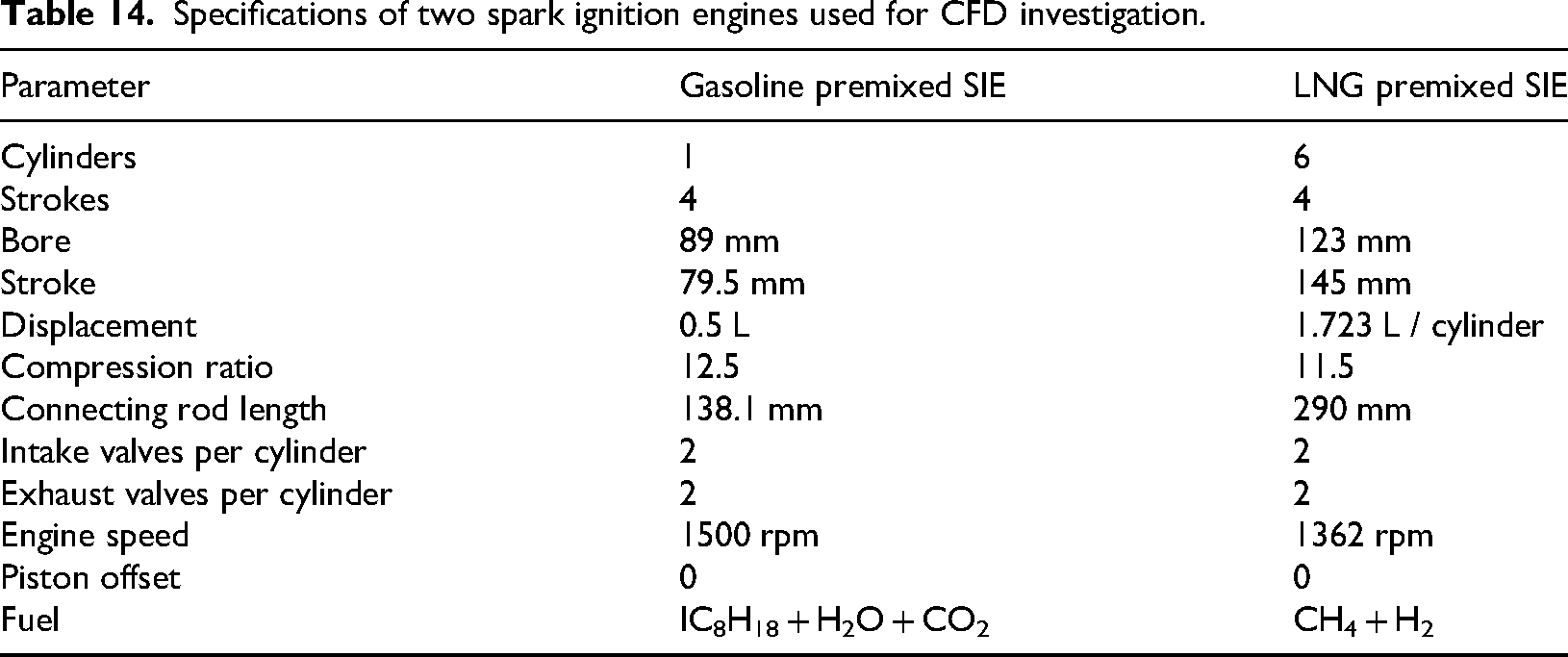
Specifications of two spark ignition engines used for CFD investigation.
Additional mesh types.

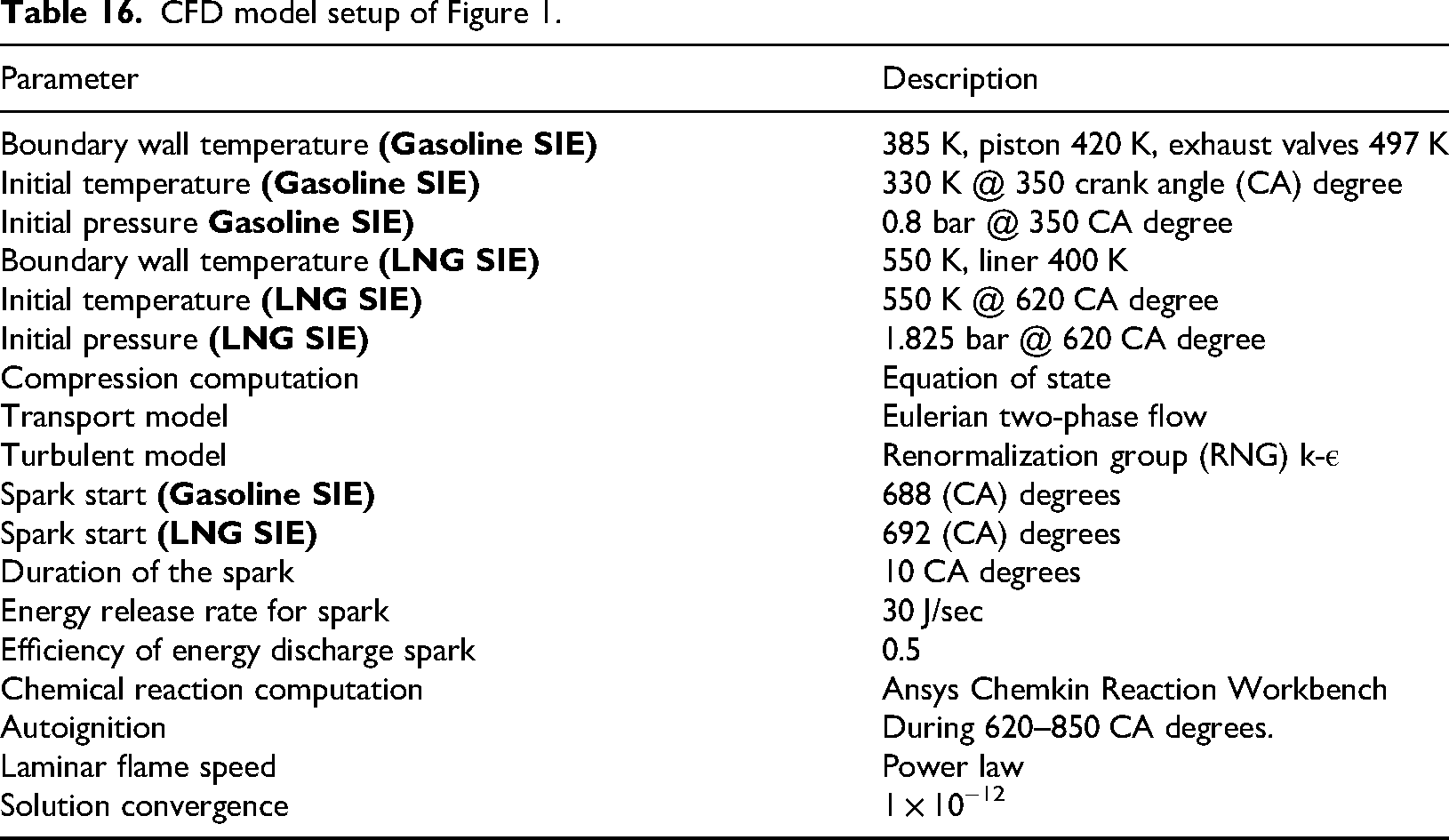
CFD model setup of Figure 1.
Techno-economic approach on hydrogen re-production by ammonia decomposition using microwave heating
The objective of this section is to pay attention on storage and heat energy investment by microwave heating to support the thought of ammonia hydrogen fuelling.
The key concept of energy fraction for ammonia decomposition by chemical equilibrium states that the energy content of H2 produced from NH3 decomposition is more than the reactant NH3 as shown in equations (5)–(7).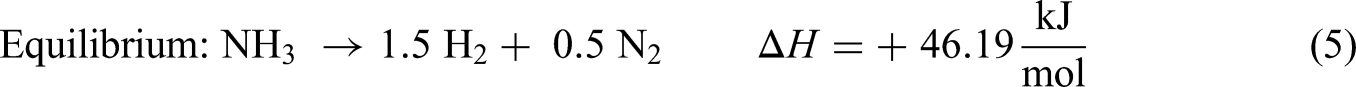
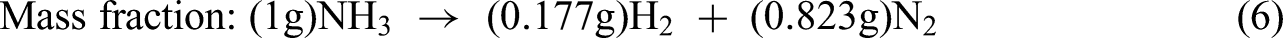
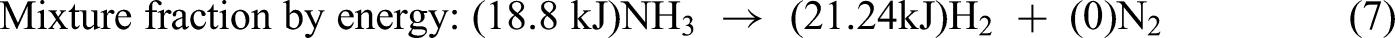
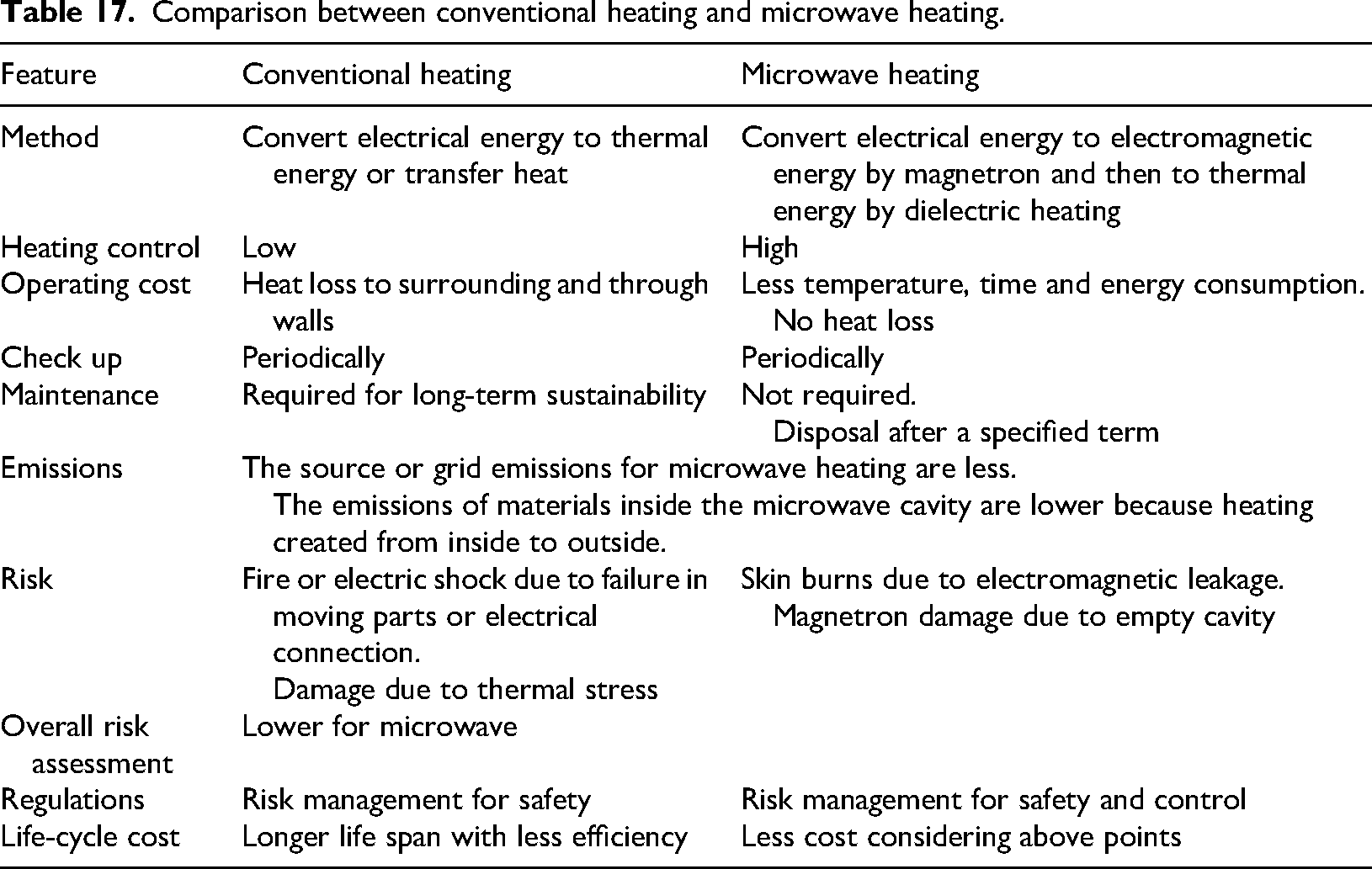
The microwave heating, aforementioned, outweighs conventional heating in aspects that are articulated in Table 17. Technically, microwave heating reduces the temperature of ammonia decomposition by about 100 °C–200 °C in comparison to conventional heating as revealed in Table 12. It should be noted that ammonia is a limited absorber for electromagnetic radiation, since the dielectric constant of ammonia is 16.61 at 20 °C. 125 Therefore, a good absorber of electromagnetic radiation, for example, carbon 126 is required to promote the absorption and dissipation of electromagnetic energy as a thermal energy, and then conduct heat to ammonia. The proposed technique to prevent the spread of electromagnetic waves is using meshes of spaces’ length shorter than electromagnetic wavelength.
Comparison between conventional heating and microwave heating.
The economic benefit for re-production of H2 from NH3 using microwave heating system can be analysed from a comparison between (a) re-production efficiency and (b) storage efficiency of H2.
NH3 production from H2 → Storage of NH3 → H2 production from NH3
Efficiency of NH3 production from H2 by Haber–Bosch process: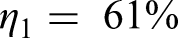
Efficiency of H2 production from NH3 by microwave heating:
Energy content for H2 and NH3 are available in Table 5.
The consumed microwave power is 100 W as shown in Table 12.
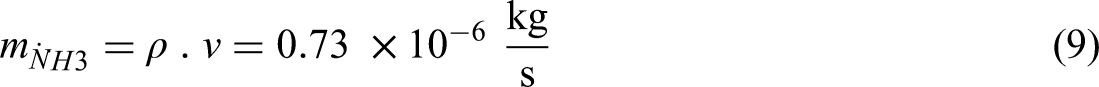
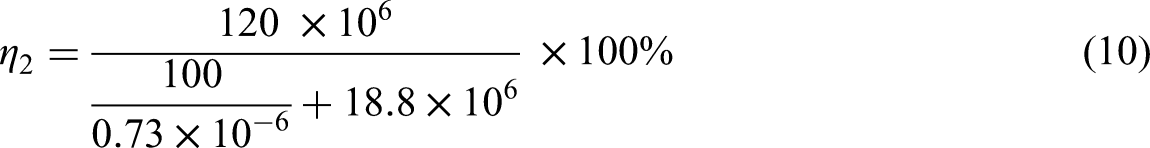
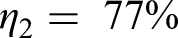
Efficiency of ammonia storage:
Energy density for NH3 and H2 are 7.1 MJ/L and 2.9 MJ/L, respectively from Table 5.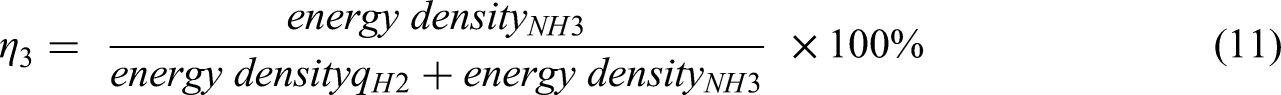
Efficiency of ammonia storage, (b) Storage of H2:
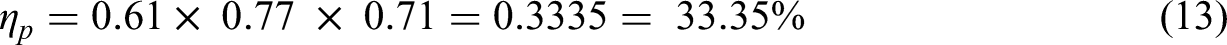
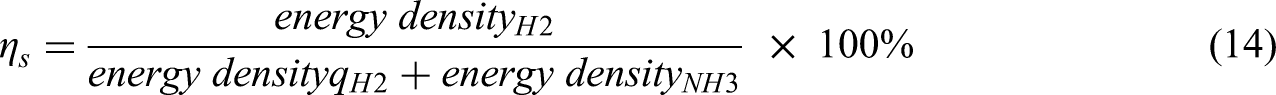
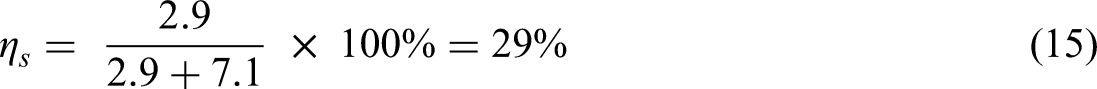
Therefore, the system efficiency of re-production of H2 (ηp) is around 4.5% more than the efficiency of H2 storage (ηs).
Preliminary results of current CFD research
The CFD software was verified by comparing the simulation results of gasoline engine with Ansys-Forte tutorials 124 and the results showed a good agreement as shown in Figure 2. The CFD model was validated by comparing the investigation results of LNG engine with experimental results. 120 The results stated that the operation time of 2 mm global mesh size lasted 4 times longer than that of 2.5 mm. Overall uncertainty of peak pressure, peak pressure location, and chemical heat release rate by using 2.5 mm global mesh size was 2.4%.
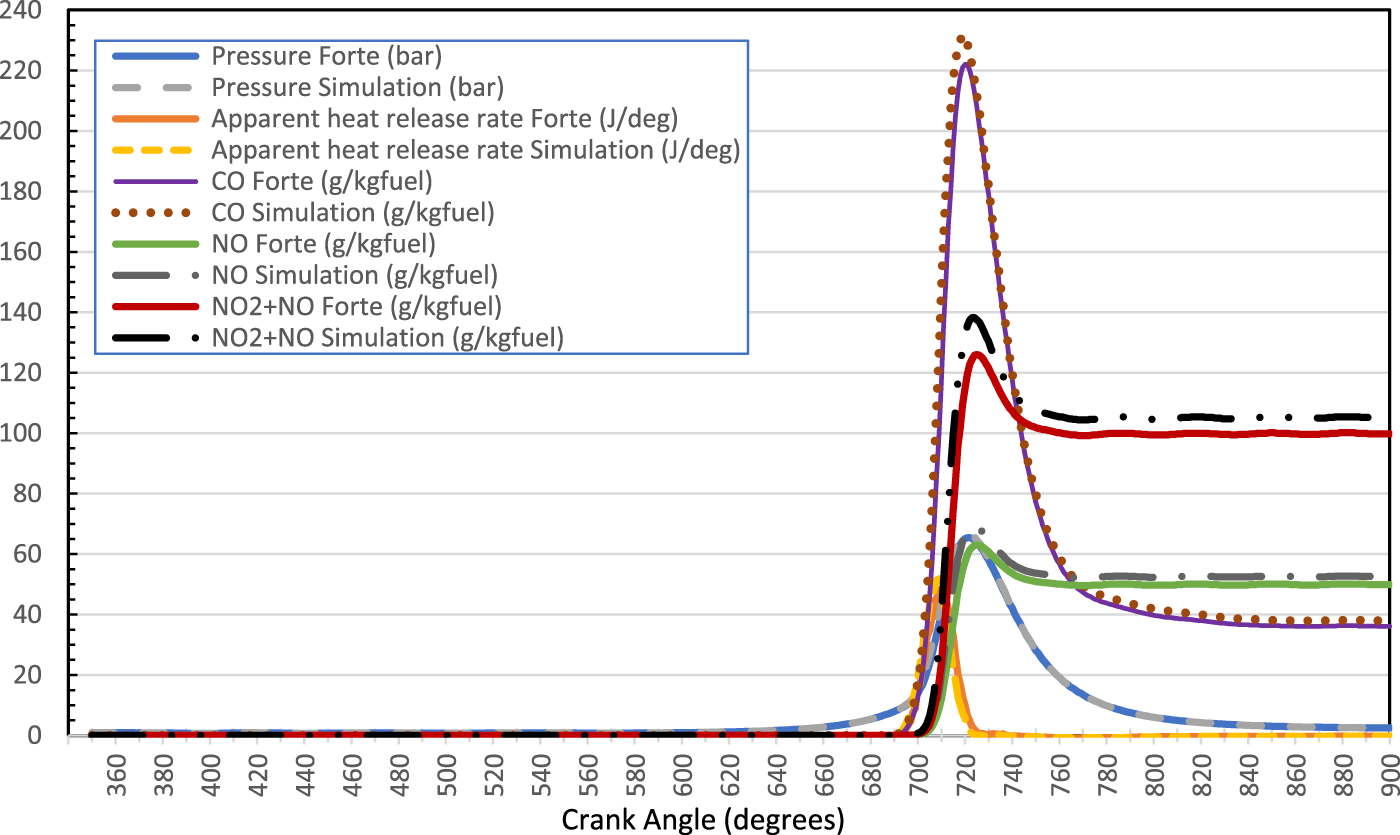
CFD investigation results show pressure, heat release rate, NO1, NOx and CO1 versus crank angles of gasoline spark ignition engine.
The current CFD investigation is on replacing methane (LNG) with ammonia using same LNG engine and conditions to compare the effect of each of ammonia and methane combustion on spark ignition engine performance and emissions. The comparison results of 0.2668 mole fraction ammonia that equals (in energy) to 0.1 mole fraction methane are shown in Figure 3 for NOx vs equivalence ratio and Figure 4 for performance and emission parameters at equivalence ratio 1. The results state that NH3 in the conditions of current investigation can be combusted in a spark ignition ICEs producing in comparison to CH4 a mitigation in NOx emissions within the range of 0.8–1.2 equivalence ratio achieving around 40% reduction at equivalence ratio 1 due to the decrease in air-nitrogen. The thermal efficiency was increased by about 2.5% due to the increase in in-cylinder temperature and pressure, in addition to zero carbon emissions.
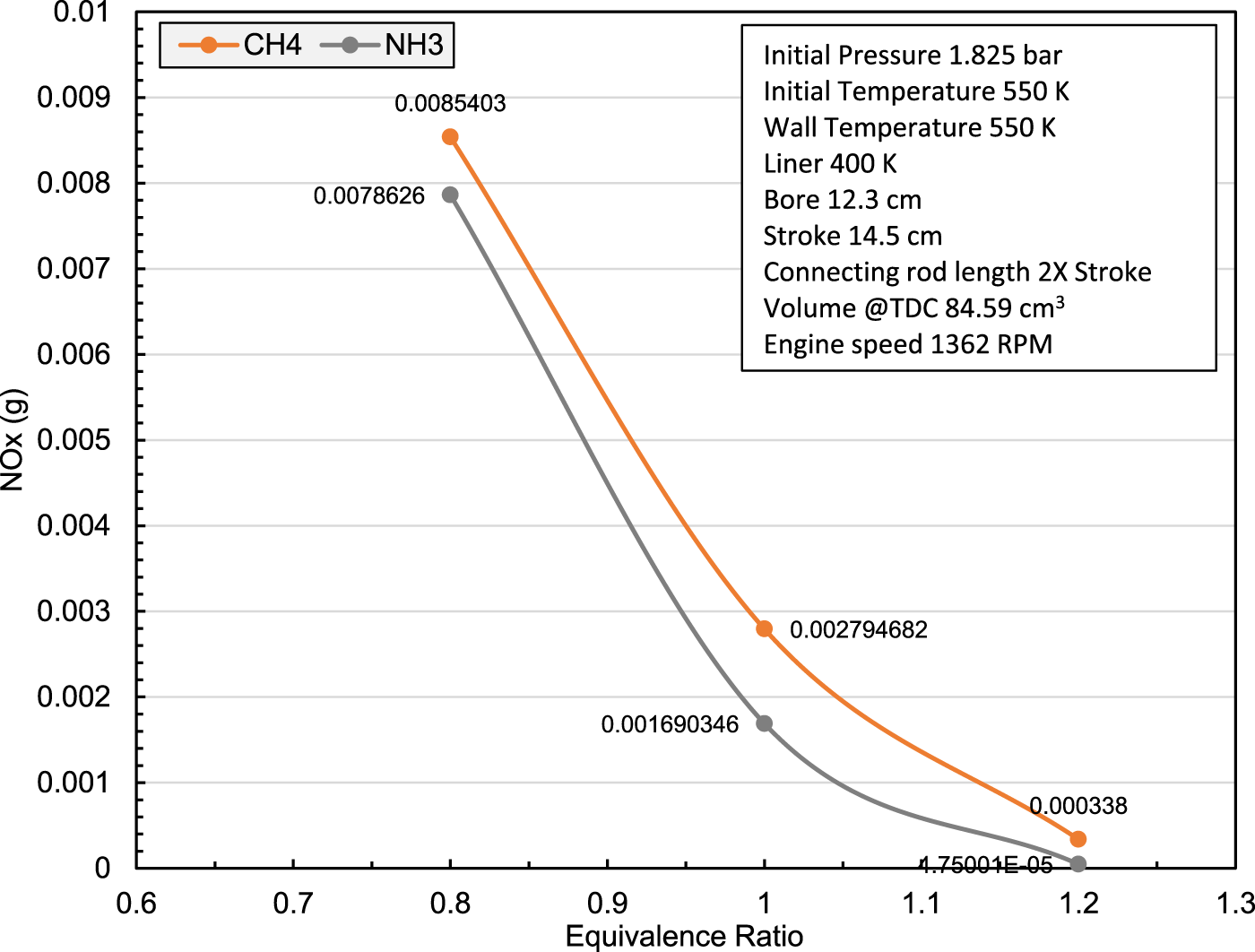
Preliminary results of the current CFD investigation show the effect of replacing CH4 by NH3 on NOx emissions of spark ignition ICE.
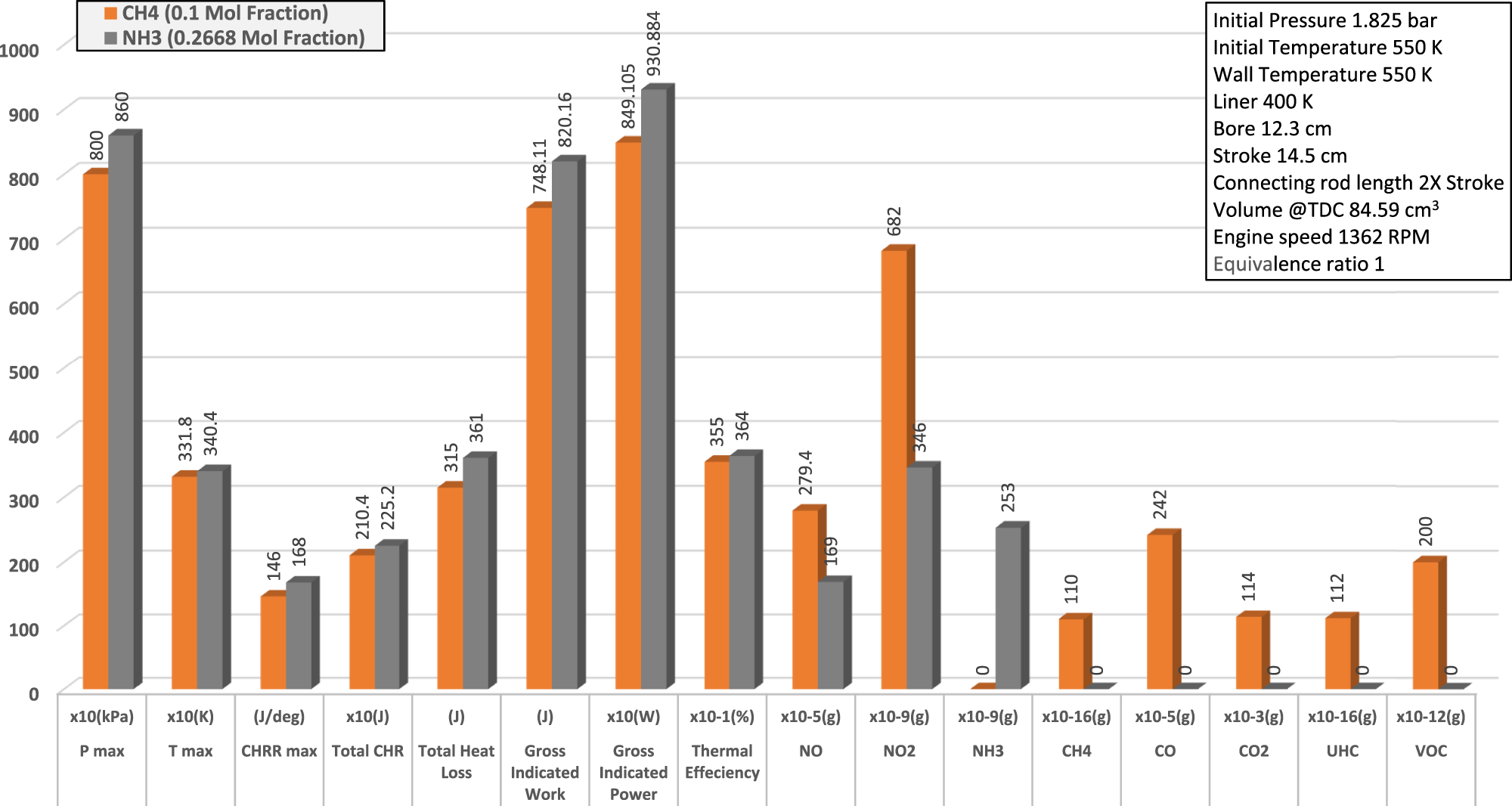
Preliminary results of the current CFD investigation show the effect of replacing CH4 by NH3 on performance and emissions of spark ignition ICE.
Conclusions
The conclusions that are analysed from the article are:
Ammonia as a sustainable fuel: Approximately 4% of ammonia production is derived from sustainable sources. Ammonia as a clean fuel: Ammonia is a free carbon energy. The undesired byproducts, such as unburned NH3 and NOx produce from the ammonia combustion can be treated by a selective catalyst reduction system. Economy of storage and transport: Ammonia is a hydrogen energy carrier. The energy of ammonia stored at 1 MPa is higher by about 2.5 times in comparison with that of hydrogen stored at 70 MPa. Safety of storage: Ammonia can be stored in insulated tanks at 0.8 MPa while hydrogen should be stored under high pressure of about 70 MPa. The boiling point of ammonia is −33 °C whereas it is −161 °C for methane and −252 °C for hydrogen. Environment and health risks: Ammonia is a highly toxic, corrosive, low flammable, non-active material without a reaction agent. It is preferred to be used for power generation than power transport for less risk. Productions of ammonia combustion are harmful gases for health and environment. Significant combustion characteristics of ammonia: The temperature and energy required for attaining ammonia combustion are high, which are 651 °C and 8 mJ, respectively causing great NOx emissions. Speed of flame is low. Flammability is limited by 16–25% (volume). Energy content (18.8 MJ/kg) is low comparable with other fuels. Impact of ammonia blends: Blending ammonia with 10% hydrogen by volume tackles ammonia autoignition problem and improves engine efficiency and work output in spark ignition engines. However, the concentrations of hydrogen and oxygen affect the amount of NOx production. Impact of ammonia dual fuel: Replacing up to 40% by energy of diesel with ammonia fuel lowers emissions of NOx compared to 100% diesel fuel. However, emissions of CO and HC are greater due to the reduction in temperature of combustion. Reduction of NOx emissions using innovative techniques: The in-cylinder ammonia injection at 58.4 CA ATDC achieves up to a 78.1% reduction in NOx emissions. The selective catalytic reduction (SCR) and selective non-catalytic reduction (SNCR) promise further NOx emission reduction. Premixed hydrogen injection enhances further the overall combustion and engine efficiency and catalyst-aftertreatment hydrogen injection reduces NOx emissions. Efficiency of microwave heating for ammonia decomposition: Ammonia decomposition using microwave heating occurs at temperatures 100–200 °C lower than conventional methods. This process results in an ammonia-to-hydrogen conversion efficiency of 100% at about 400 °C. This approach makes an approximately 4.5% increase in economic efficiency comparable to traditional hydrogen storage and transport methods. Additionally, the integration of microwave-heated ammonia decomposition into ICEs provides potentially a continuous supply of hydrogen with a risk less than the hydrogen storage. Advancements in computational fluid dynamics (CFD) modelling: The preliminary CFD results in comparison to CH4 state that it is potential to use ammonia as a clean fuel for spark ignition engines with 2.5% rise in thermal efficiency, zero carbon emissions and close to 40% lowering in NOx emissions.
Currently, a CFD model is under development to explore the optimum concentration of mixing H2 to NH3 and to optimize boundary conditions with operating conditions to get a best performance and less emissions for ICEs.
Footnotes
Acknowledgements
The authors would like to acknowledge Teesside University for the financial and technical support to conduct this research work. We would also like to thank Mr Oluwagbemiga A. Fabunmi from Teesside University for his valuable time for proofreading of this article and providing feedback.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
