Abstract
Helping other people can entail risks for the helper. For example, when treating infectious patients, medical volunteers risk their own health. In such situations, decisions to help should depend on the individual’s valuation of others’ well-being (social preferences) and the degree of personal risk the individual finds acceptable (risk preferences). We investigated how these distinct preferences are psychologically and neurobiologically integrated when helping is risky. We used incentivized decision-making tasks (Study 1; N = 292 adults) and manipulated dopamine and norepinephrine levels in the brain by administering methylphenidate, atomoxetine, or a placebo (Study 2; N = 154 adults). We found that social and risk preferences are independent drivers of risky helping. Methylphenidate increased risky helping by selectively altering risk preferences rather than social preferences. Atomoxetine influenced neither risk preferences nor social preferences and did not affect risky helping. This suggests that methylphenidate-altered dopamine concentrations affect helping decisions that entail a risk to the helper.
Keywords
Individuals differ in how much they help others. Such behavioral variation in helping is often considered a reflection of differences in social preferences—the value that individuals assign to the welfare of another person (Fehr & Schmidt, 1999). However, helping other people can put the helper at risk: When medical volunteers treat infectious patients, they risk getting the disease themselves. Or when people attempt to intervene in a fight, they risk getting injured themselves. As with social preferences, people also differ in the risks they are willing to take—the degree to which a decision-maker tolerates uncertain outcomes (Frey et al., 2017)—and this might influence people’s willingness to help (e.g., Exley, 2016). Indeed, public debate about whether or not to help often revolves around the risks involved. For example, attempts to bring Ebola patients to hospitals in the United States were met with strong public concerns over infection risks (Yang, 2015).
How do people decide whether or not to help in situations where helping is risky? Is this determined by their social preferences, their risk preferences, or an interplay of both? And in the latter case, how are social and risk preferences integrated into a decision to help? Because social and risk preferences have been mostly studied in isolation, the precise form and function of these two preferences in determining whether people help in risky situations remains poorly understood. Here, we examined how social and risk preferences are psychologically and neurobiologically integrated when helping comes at a risk to oneself.
Evidence suggests that the brain represents the possible consequences of different decision options on a common subjective-value scale by integrating and trading off different option features or conflicting internal motives, such as risk aversion or social preferences (Gross et al., 2014; Levy & Glimcher, 2012; McNamee et al., 2013). Specifically, corticostriatal circuits encompassing the dorsolateral prefrontal cortex and the striatum are associated with both risk taking (Knoch et al., 2006; Mohr et al., 2010) and social preferences (Baumgartner et al., 2011; Christov-Moore et al., 2016; Crockett et al., 2017; Gross et al., 2018). This corticostriatal circuitry is targeted by the neurotransmitter dopamine, which independently modulates both risk taking (Fiorillo et al., 2003; Schultz et al., 2015) and social preferences (Crockett et al., 2015; Sáez et al., 2015; Soutschek et al., 2017; for a review, see Crockett & Fehr, 2014). For example, studies in rats and monkeys have shown that subcortical dopamine release is associated with risk taking (Fiorillo et al., 2003; Simon et al., 2011) and that dopamine firing rates reflect the curvature of the utility function—the mathematical representation of subjective value that indicates risk attitudes (Schultz et al., 2015).
In humans, risk taking increases following the administration of the dopamine precursor levodopa (Kobayashi et al., 2019; Rigoli et al., 2016; Rutledge et al., 2015) and the D2/D3 agonist pramipexole (Campbell-Meiklejohn et al., 2011; Riba et al., 2008). Although there are fewer data on dopaminergic modulation of social preferences, levodopa has been shown to reduce generosity in dictator games (Pedroni et al., 2014) and hyper-altruism in a harm-aversion task (Crockett et al., 2015), whereas tolcapone—a drug that enhances dopamine transmission in the prefrontal cortex—increases inequity aversion (Sáez et al., 2015).
Combined, these studies implicate a common neural circuitry for risk and social preferences that is sensitive to dopamine manipulations and may be engaged in risky helping decisions. Here, we examined this possibility by manipulating dopaminergic neurotransmission using methylphenidate and atomoxetine. By blocking the dopamine and norepinephrine transporters, methylphenidate increases dopamine concentration in the striatum (Kim et al., 2009; Smith et al., 2006; Volkow et al., 2012) and both dopamine and norepinephrine concentration in the ventromedial prefrontal cortex and dorsolateral prefrontal cortex (Arnsten & Li, 2005). In line with findings on the dopaminergic modulation of risk preferences, studies have shown that methylphenidate can increase risk taking in rats (Yates et al., 2020) and humans (Campbell-Meiklejohn et al., 2012; Mandali et al., 2021). Similar to methylphenidate, atomoxetine increases synaptic levels of both norepinephrine and dopamine in the prefrontal cortex. In contrast to methylphenidate, however, atomoxetine does not affect striatal dopamine levels (Bymaster et al., 2002; Schulz et al., 2017), and there is little evidence for norepinephrine modulation of risk or social preferences in humans (Crockett et al., 2010; Crockett & Fehr, 2014). Studies in rats suggest that norepinephrine modulates risk taking only when combined with dopamine reuptake inhibition (Montes et al., 2015). It is therefore possible to probe the differential contributions of dopaminergic and noradrenergic systems on risky helping by comparing the effects of methylphenidate and atomoxetine on behavior.
Statement of Relevance
People help others at sometimes substantial costs to themselves. What has been largely overlooked is that helping can not only be costly but also be risky. When, for example, medical volunteers treat patients with infectious diseases, they may become infected themselves. Volunteers who try to rescue shipwrecked refugees risk injury or drowning. Here, we identified individual differences in social preferences (predicting willingness to help) and risk preferences (predicting willingness to take risks) and examined how these distinct preferences alone and in combination predict decision-making when helping creates a risk for the helper. We found that even people with high social preferences are more likely to help in risky situations when their risk tolerance is sufficiently high. Our findings both advance neurobiological theory and have practical implications by further showing that the drug methylphenidate increases risky helping because it alters risk preferences rather than social preferences.
On the basis of previous findings, we hypothesized that methylphenidate will causally alter risk preferences. What remains unknown is whether methylphenidate also affects social preferences and whether and how methylphenidate impacts the integration of both risk and social preferences and, thereby, influences helping decisions that come with a risk to oneself. Meanwhile, because of scant evidence implicating norepinephrine in risk or social preferences in humans, we did not expect to observe effects of atomoxetine on risky helping behavior. We performed two studies to examine these possibilities. In both studies, we used incentivized behavioral tasks that confront the individual with a decision to help or refuse to help another person when helping is not only costly but also risky. By manipulating risk and social consequences independently within the same task, we measured social and risk preferences in isolation to study how these preferences are integrated when helping is risky. Study 1 tested the integration of social and risk preferences by observing participants’ behavioral decisions when choices are risky, have consequences for other individuals, or carry both risks to oneself and consequences for others. Study 2 complemented behavioral observations of Study 1 with a double-blind, placebo-controlled neuropharmacological intervention using methylphenidate and atomoxetine to manipulate dopaminergic and noradrenergic neurotransmission during decision-making.
Method
The risky helping task
To investigate risky helping as a function of social and risk preferences, we designed a two-player incentivized “risky helping task” (Fig. 1a). It involves a decider who repeatedly decides whether or not to help another participant, the receiver. The decision to refuse help leads to a sure outcome of 15 monetary units (MU) for the decider and 0 MU for the receiver. The decision to help is risky: With a probability p, helping is unsuccessful, in which case both decider and receiver earn 0 MU. When helping is successful (with probability 1 – p), both earn 13 MU. Note that helping always reduces inequality between decider and receiver to zero. Further, helping is costly to the decider: Even when successful, the decider incurs a cost of 2 MU. Hence, the risky helping task confronts the decision-maker with a dilemma between avoiding risk to oneself and helping another person.
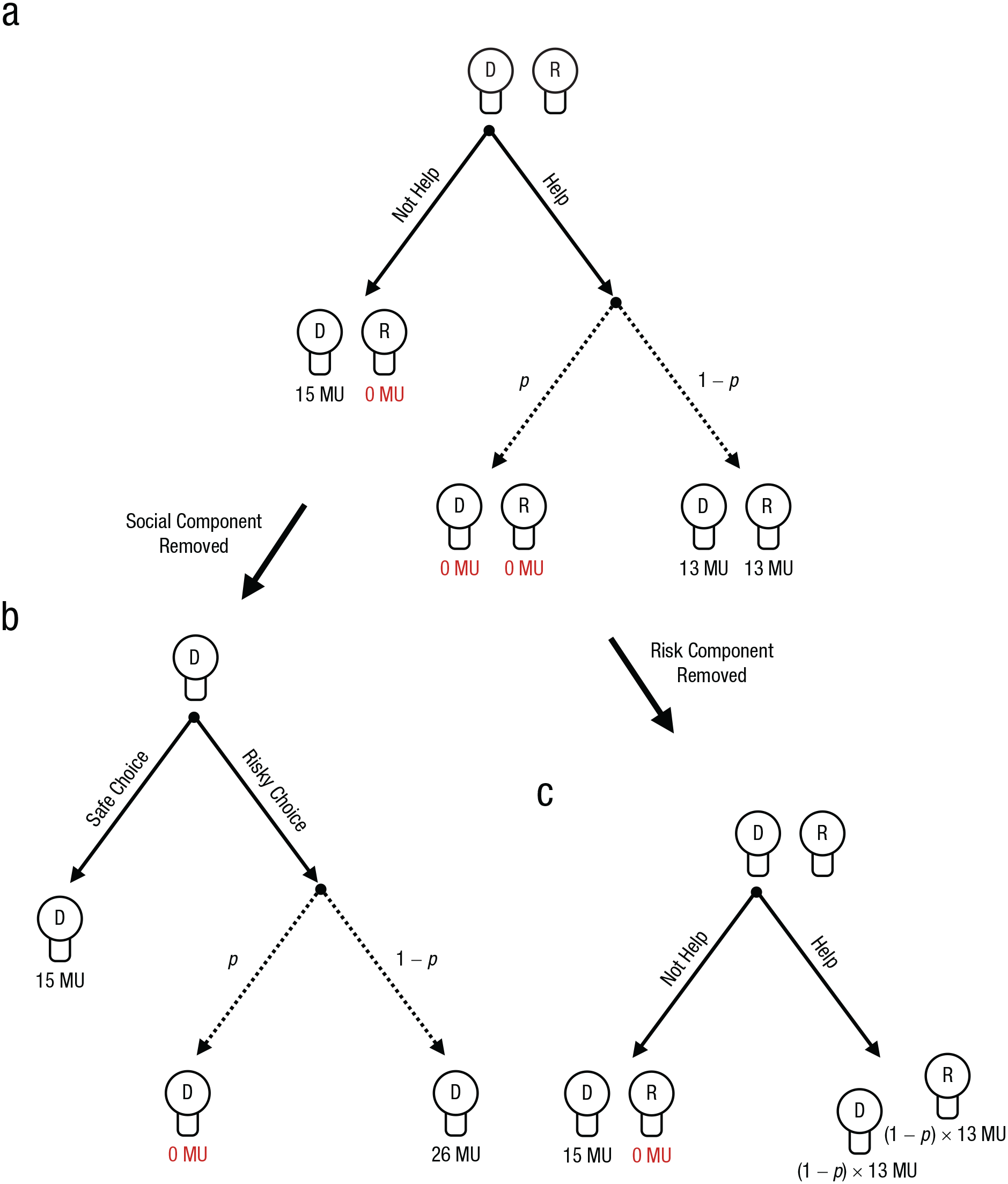
Schematics of the risky helping, risk, and helping tasks. In the risky helping task (a), the decider (“D”) decides whether to help or not help the receiver (“R”). If the decider decides to help, helping is unsuccessful with probability p, and both decider and receiver earn 0 monetary units (MU). With probability 1 – p, helping is successful, leading to an equal outcome of 13 MU for both. If the decider decides not to help, they earn 15 MU, but the receiver earns nothing. In the risk task (b), the decider decides between a safe option, leading to a payoff of 15 MU, and a risky option, leading to either a payoff of 0 MU with probability p or a payoff of 26 MU with probability 1 – p. In this task, the receiver is not affected by the decider’s decision. Thus, the social component of the risky helping task is removed. In the helping task (c), the decider decides whether to help or not help the receiver. Across trials, helping leads to a sure outcome equal to the expected value of possible outcomes of the risky helping task. Thus, the risk component of the risky helping task is removed. Not helping means that the decider earns 15 MU, and the receiver earns nothing. In the actual task, the labels “help,” “not help,” “risky choice,” and “safe choice” were replaced with neutral labels to avoid framing effects.
To measure the relative influence of the decider’s social and risk preferences, we derived two variants of the risky helping task: the risk task and the helping task. In the risk task, we removed the social component of the task to measure risk preferences in isolation. In this task, the receiver is not affected by the decisions of the decider. Instead, the decider chooses between a sure outcome and a lottery for each possible p that affects only their payoff (Fig. 1b). In other words, the risk task confronts the decision-maker with a dilemma between choosing a safe option and choosing a gamble that can lead to a higher payoff (with probability 1 – p) but also entails the risk of earning nothing for that trial (with probability p). The outcome for each decision option is the sum of money at stake in the risky helping task (i.e., 15 MU for the safe choice and 26 MU for the risky choice) to make sure that the safe choice would not simply dominate the risky choice in both risk and payoff for all ps. In essence, the risk task measures risk preferences by reducing the risky helping task to a set of paired lottery choices that have consequences only for the decider but not for the other person.
In the helping task, the decider chooses between helping and not helping. We removed the risk component from the risky helping task to measure social preferences in isolation (Fig. 1c). To achieve this, we replaced the helping outcome of the risky helping task with the expected value for each p—that is, p × 0 + (1 – p) × 13—as illustrated in Table 1. For example, in one trial of the risky helping task, the decision-maker faces the following choice between option A and option B: (a) 15 MU for you, 0 MU for the other person (b) with p = .5: 0 MU for you, 0 MU for the other person; with 1 – p = .5: 13 MU for you, 13 MU for the other person
Decision Consequences in the Risk, Helping, and Risky Helping Tasks Depending on Risk Probability (p)
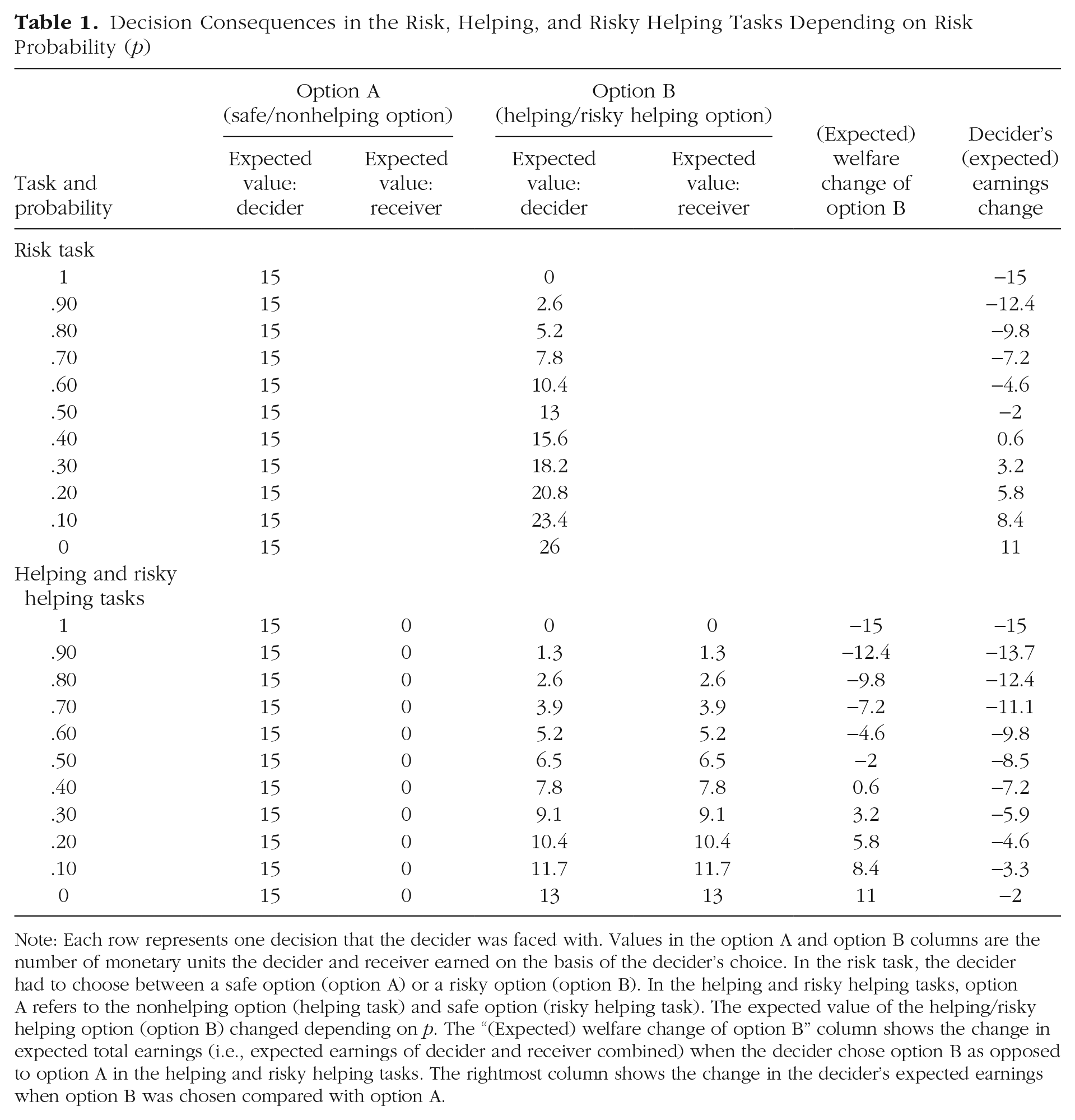
Note: Each row represents one decision that the decider was faced with. Values in the option A and option B columns are the number of monetary units the decider and receiver earned on the basis of the decider’s choice. In the risk task, the decider had to choose between a safe option (option A) or a risky option (option B). In the helping and risky helping tasks, option A refers to the nonhelping option (helping task) and safe option (risky helping task). The expected value of the helping/risky helping option (option B) changed depending on p. The “(Expected) welfare change of option B” column shows the change in expected total earnings (i.e., expected earnings of decider and receiver combined) when the decider chose option B as opposed to option A in the helping and risky helping tasks. The rightmost column shows the change in the decider’s expected earnings when option B was chosen compared with option A.
The equivalent trial in the helping task is as follows: (a) 15 MU for you, 0 MU for the other person (b) 6.5 MU for you, 6.5 MU for the other person
Thus, the decider chooses between two nonrisky outcomes in the helping task: One option maximizes one’s own payoff but also leads to inequality, whereas the other option requires sacrificing one’s own monetary units to benefit the receiver and eliminate inequality (similar to a mini dictator game with different efficiency gains). In other words, the dilemma for the decision-maker reduces to making a choice between a selfish option that leads to 0 MU for the receiver and a prosocial option that benefits the receiver at a cost to the decider.
Experimental implementation
In the experiments, neutral labels for helping and risk were used to avoid demand or framing effects, and decisions were completely anonymous to avoid reputation effects. In both studies, each participant performed the tasks in front of a personal computer terminal. Comprehension checks were used to make sure that participants understood the instructions (for task instructions and the decision interface, see the Supplemental Material available online).
To measure risk and social preferences and the extent of helping in a risky environment on the individual level, we had each decider engage in all three separate tasks (risky helping task, risk task, helping task) in a random order (within-subjects design). Probability p (i.e., risk) was systematically varied between 0 and 1 across 21 randomly presented trials in the risk task and the risky helping task.
The decisions of the decider had real financial consequences for decider and receiver that were paid out in cash at the end of the experimental session (one round of each task was selected at random for payment; 1 MU was worth €0.20 in Study 1 and 36 British pennies in Study 2). In Study 1, receivers were also confronted with every decision that the decider had to face in the risk, helping, and risky helping tasks but, instead, had to guess how the decider would decide. Guessing correctly was financially incentivized (for the results, see the Supplemental Material).
Participants
In Study 1, 292 participants (142 women, age: M = 22.3 years, SD = 3.7) were recruited and split into 146 deciders and 146 receivers. Sample size (aimed at ~300) for this within-subjects design was based on earlier work assessing individual differences in social-value orientation (van Lange, 1999), which typically found that roughly 45% (40%) of the sample held prosocial (pro-self) preferences. On the basis of this heuristic, we expected a good spread of social preferences in our sample, with approximately 50 participants having strong and approximately 50 participants having weak social preferences. Data were not analyzed before the full data collection was finished.
Participants were free to withdraw from the experiment at any time. Only individuals who voluntarily entered the recruiting database were invited, and informed consent was obtained from all participants by electronic acceptance of an invitation to attend an experimental session. The experiment was conducted following the peer-approved procedures established by the Center for Research in Experimental Economics and Political Decision Making (CREED) at the University of Amsterdam and was fully incentivized. The experiment was performed in a large lab with 30 separated cubicles. This allowed participants to see that other people took part in the experiment while not being able to identify with whom they were paired or how other participants decided during the experiment. Following standard laboratory protocols, we did not use deception and invited participants from a pool of individuals who were aware of this no-deception policy.
In Study 2, a sample of 154 participants (77 women, age: M = 23.7 years, SD = 3.9) was recruited in the role of deciders, and only one participant took part per experimental session (we aimed for 150 participants). This sample size equaled the one used in Study 1 and followed our earlier work on drug administration and behavioral decision-making, in which a sample of approximately 50 participants per treatment condition enabled us to detect small to medium effect sizes (Baas et al., 2020; Crockett et al., 2015). As in Study 1, data were not analyzed before the full data collection was finished.
Participants completed a prescreening questionnaire so that we could select only those who had no history of drug consumption, had a limited alcohol and caffeine intake, had no clinically relevant depression scores, and were generally in a healthy condition. Participants underwent another screening via telephone the day before the study to make sure they had, for example, not consumed any alcohol or other medication. The experiment received ethical approval from the University of Oxford’s Medical Sciences Interdivisional Research Ethics Committee. Because dopamine function can vary throughout the day (Hood et al., 2010), we conducted each experimental session in the same place at roughly the same time of the day (starting around 9 a.m.). Before substance administration and after preadministration checks, female participants took a test to confirm they were not pregnant, and we measured height, weight, and blood pressure of all participants. Supervised by a medical doctor throughout the study, participants received either 30 mg of methylphenidate, 60 mg of atomoxetine, or a placebo; each was contained in an identical blue capsule. Following ethical requirements, we informed participants about the three substances they might be taking, but neither they, the experimenters, nor the medical doctors knew which substance was administered.
Procedure
To make sure that testing coincided with peak absorption rate, we started the experiment 90 min after drug administration. Participants then took part in the risk task, the helping task, and the risky helping task as part of a larger test battery. Participants also completed a 15-item visual analogue mood scale at two time points of the experiment—namely, before drug administration and after the experiment was finished. For each item on the scale, participants had to indicate their mood by placing a mark along a straight line with two opposing adjectives on either side (e.g., muzzy vs. clearheaded, happy vs. sad, tense vs. relaxed), which resulted in a continuous measure between 0 and 1 for each item. Hence, we measured subjective mood states before and after the drug administration (which also allowed us to compute mood changes).
Analyses
In each task, deciders made the binary decision to help or not (helping task and risky helping task) or to take the risky or the safe choice (risk task). Trials differed in risk (risk and risky helping task) or helping consequences (helping task). Hence, in each trial, we observed a binary decision. The binary decisions within each task revealed when a participant would take the risky or social option and when they would switch to the certain or selfish option. We used these switching points as a measure of risky helping, social preferences, and risk preferences.
For example, if a participant decided to choose the lottery for a risk, p = [0, .05, .10, .15, .20, .25], and switched to the sure outcome for p = [.30, . . . , 1] in the risk task, the participant was assigned a value of .30 as their measure of risk preference. Note that this implies that a switching point of .45 or lower indicates risk aversion (or risk neutrality) because the decision-maker switched to the safe option before the expected value of the lottery decreased below the fixed payoff of the safe option. A switching point of .50 and higher indicates risk seeking because the decision-maker accepted lotteries for which the expected value of the lottery was lower than the fixed payoff of the safe option.
Between 14% and 18% of our participants had multiple switching points, thereby violating monotonicity. This is not unexpected because trials were randomly and sequentially presented within each task. Accordingly, we interpreted violations of monotonicity as noise and averaged the switching points in these cases. Thus, a decision pattern such as [B, B, B, A, B, A, A, . . . , A] for risk levels of [0, .05, .10, .15, .20, .25, .30, . . . , 1] led to a switching point of .20. Excluding participants who violated monotonicity from the analysis did not alter the reported conclusions below (see the Supplemental Material).
To analyze switching points, we used Tobit regressions. Tobit regressions deviate from a linear regression model in that they account for the (left) censoring in the data as they assume a latent variable y* that linearly depends on the predictors (y = y* if y > 0 but deviates in the lower bound). For y = y*, we used the likelihood function of a t distribution, t(y|bX, σ2, df). For y ≠ y*, the likelihood function was based on the normal cumulative distribution and, hence, modeled the probability that y* would take a value less than or equal to y, given the observed predictors. This approach allowed us to derive unbiased estimates for predictors and error variance in the presence of heteroscedasticity.
Results
Here, we summarize the main findings from our two studies. The regression models and results, alongside additional models that include control variables and robustness checks, are reported in the Supplemental Material.
Study 1
In line with previous literature (Holt & Laury, 2002), results showed that participants were predominantly risk averse in the risk task: They switched to the risky option only for gambles for which the expected value of the risky option exceeded the value of the safe option (Fig. 2a). In the helping task, a majority of participants revealed social preferences. Most participants gave up some of their own resources to help the other person at least once (Fig. 2b). In the risky helping task, participants who engaged in risky helping did so up to a risk of p = .15, on average, and then switched to not helping (Fig. 2c). When participants engaged in risky helping, the gain to social welfare (i.e., the combined expected earnings of decider and receiver) significantly exceeded their own monetary sacrifice, showing that risky helping was not unconditional (reduction in earnings for deciders vs. expected value of decider and receiver; Wilcoxon signed-rank test, p < .001, d = 0.66). On average, deciders sacrificed 1 MU to achieve an expected welfare increase of 1.8 MU.
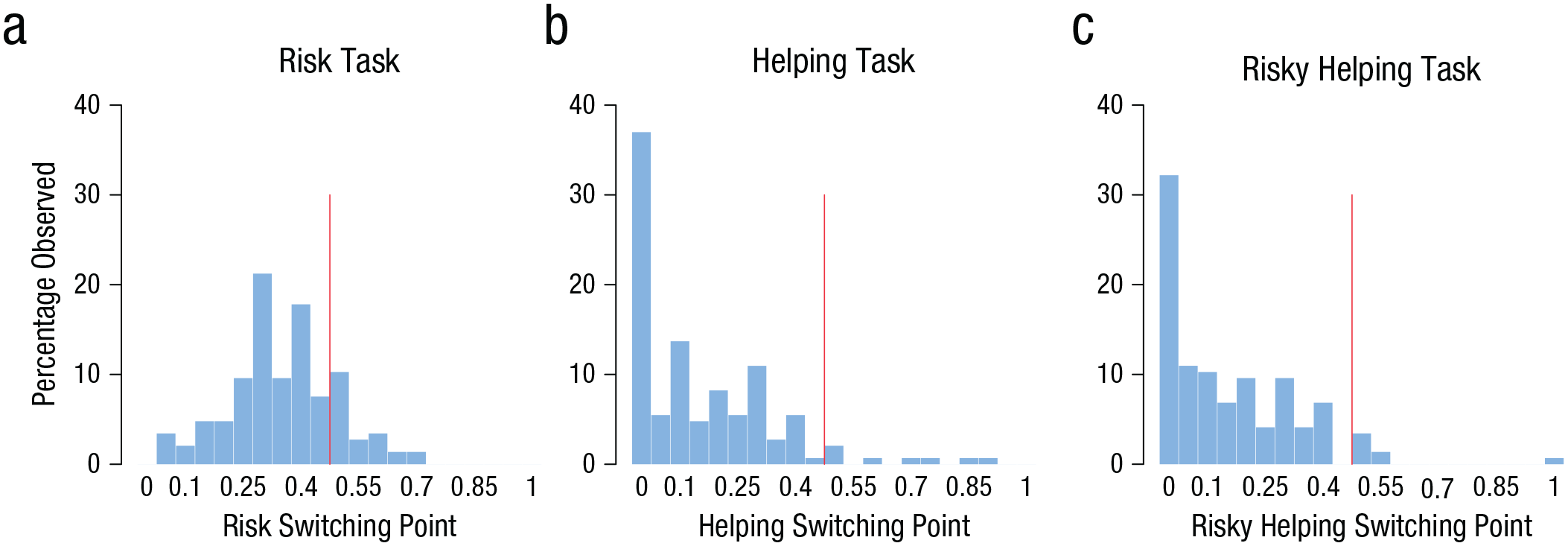
Distribution of switching points in the risk (a), helping (b), and risky helping (c) tasks in Study 1. The red line indicates the border between risk aversion and risk seeking (risk task) and between social efficiency and social inefficiency (helping and risky helping tasks). Social efficiency refers to making a helping or risky helping decision only when the combined (expected) payoff for decider and receiver exceeds the sure payoff of not helping. A switching point above the efficiency point means that participants chose to help even when the combined (expected) payoff for decider and receiver was lower than the sure payoff of 15 monetary units (MU) for not helping (i.e., 15 MU for the decider and 0 MU for the receiver).
To test whether risky helping decisions can be modeled as a function of risk and social preferences, we analyzed whether and how switching points in the risk and helping tasks predicted risky helping decisions. We found, first of all, that risk preferences derived from the risk task and social preferences derived from the helping task were linearly independent of each other (Spearman’s r = −.02, p = .787). Thus, an individual’s social preference could not be inferred from knowing their risk preference and vice versa, pointing to a behavioral dissociation of risk and social preferences.
Yet both risk and social preferences independently predicted risky helping decisions. We first calculated the change in switching points between helping and risky helping tasks on the individual level to see whether this change was accounted for by risk preferences. The change in switching points between helping and risky helping was significantly correlated with the switching point in the risk task (Spearman’s r = .25, p = .002). The more risk tolerant participants were (as measured in the risk task), the more they helped under risk. Conversely, a switching-point change between the risk task and the risky helping task was significantly correlated with the extent of helping in the helping task (Spearman’s r = .57, p < .001). In other words, stronger social preferences (as measured in the helping task) were associated with an increased willingness to help under risk (as measured in the risky helping task).
Another approach to test the extent to which risky helping can be predicted by risk preference, social preference, or their combination is to use regression models. As illustrated in Figure 3a, risky helping decisions were a function of both social preferences and risk preferences (risk preference: b = 0.26, SE = 0.09, p = .005; social preference: b = 0.83, SE = 0.07, p < .001). A post hoc test comparing the relative weight of social and risk preferences also revealed that although both variables were independently associated with risky helping, social preferences were a stronger predictor of risky helping than risk preferences (post hoc comparison: social-preference estimate – risk-preference estimate ≠ 0, b = 0.57, SE = 0.12, p < .001). In other words, helping under risk emerged only with moderate to high social preferences combined with low to moderate risk aversion. Taken together, the results of Study 1 provide evidence that risk and social preferences are behaviorally independent of each other yet systematically integrated in situations in which helping is risky.
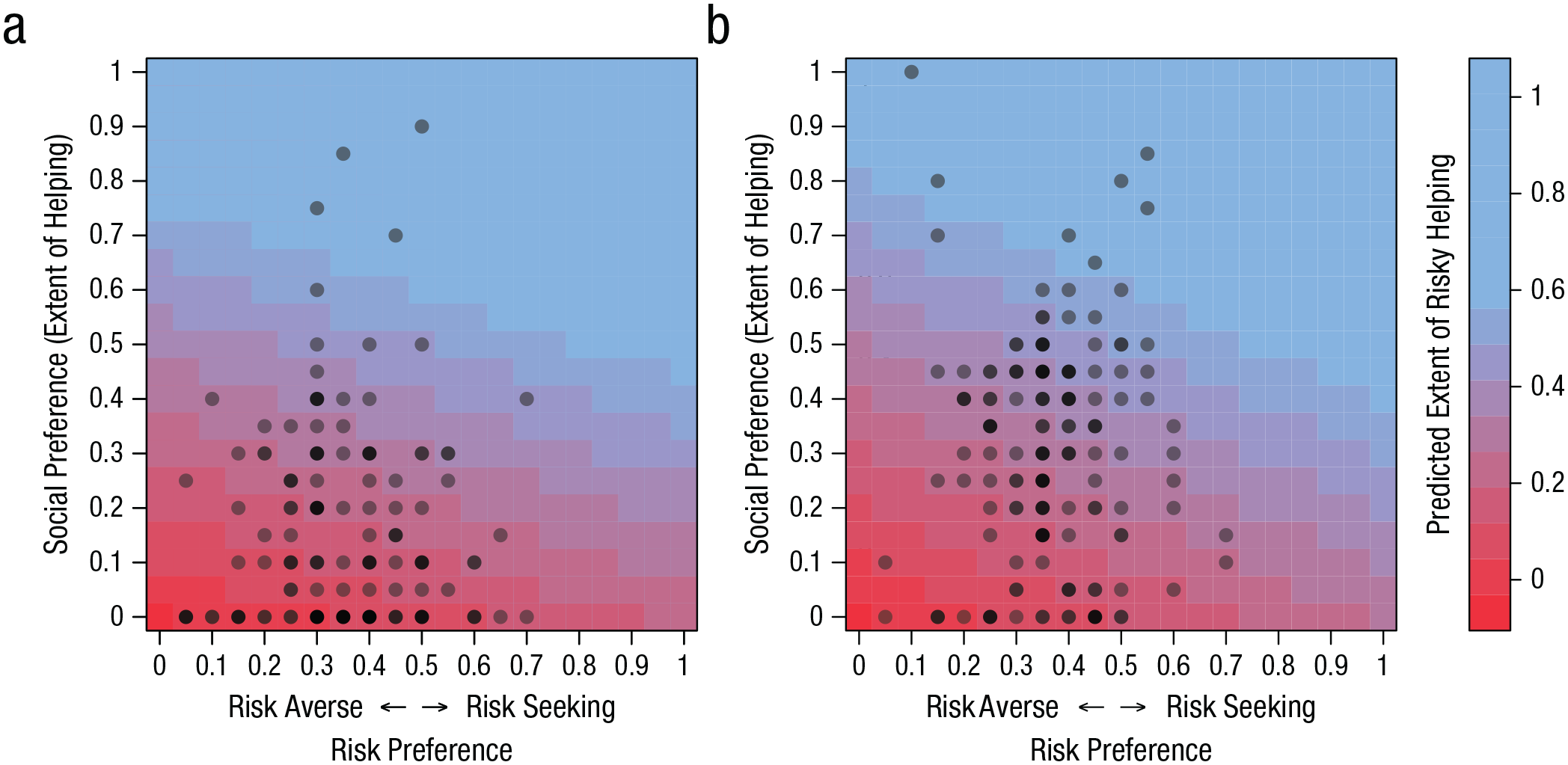
Predicted extent of risky helping (color surface) based on a linear combination of risk preference and social preference in Study 1 (a) and Study 2 (b). Dots show the observed risk preference and social preference of deciders. Darker dots indicate that multiple deciders had the same profile for risk and social preference.
Study 2
As in Study 1, individuals’ risk and social preferences were uncorrelated (Spearman’s r = .03, p = .800). Further replicating the results of Study 1, risk preferences in Study 2 were correlated with changes in helping between the helping and risky helping tasks (Spearman’s r = .24, p = .003). Participants who were more risk taking were also more likely to help when it was risky compared with when it was not. When participants were more risk averse, in contrast, they were less likely to help when it was risky. Within-individual changes in switching points between the risk and risky helping tasks were also positively correlated with social preferences (Spearman’s r = .61, p < .001). Hence, participants with stronger social preferences accepted more risk when it could benefit another person (and the other way around).
As shown in Figure 3b, risky helping decisions could again be modeled as a linear combination of social preferences and risk preferences (risk preference: b = 0.31, SE = 0.09, p = .001; social preference: b = 0.73, SE = 0.05, p < .001). This means that a person’s social preferences did not allow accurate predictions of risky helping without also factoring in this person’s risk preferences or the other way around: Knowing a person’s risk preferences did not allow for accurate prediction of risky helping without knowing their social preferences. And in line with Study 1, results of a post hoc test showed that social preferences were a stronger predictor of risky helping than risk preferences (post hoc comparison: social-preference estimate – risk-preference estimate ≠ 0; b = 0.42, SE = 0.11, p < .001).
Methylphenidate causally affected risk preferences (Fig. 4a). Participants who received methylphenidate accepted significantly more risk than participants who received a placebo (b = 0.05, SE = 0.02, p = .018). Whereas 90% of the participants given a placebo were classified as risk averse (similar to the percentage in Study 1), this dropped to 78% for those given methylphenidate. Risk taking did not differ between participants who received a placebo and those who received atomoxetine (b = 0.02, SE = 0.02, p = .295). We also found no statistically significant difference in risk taking between participants given atomoxetine and methylphenidate (b = 0.03, SE = 0.02, p = .185).
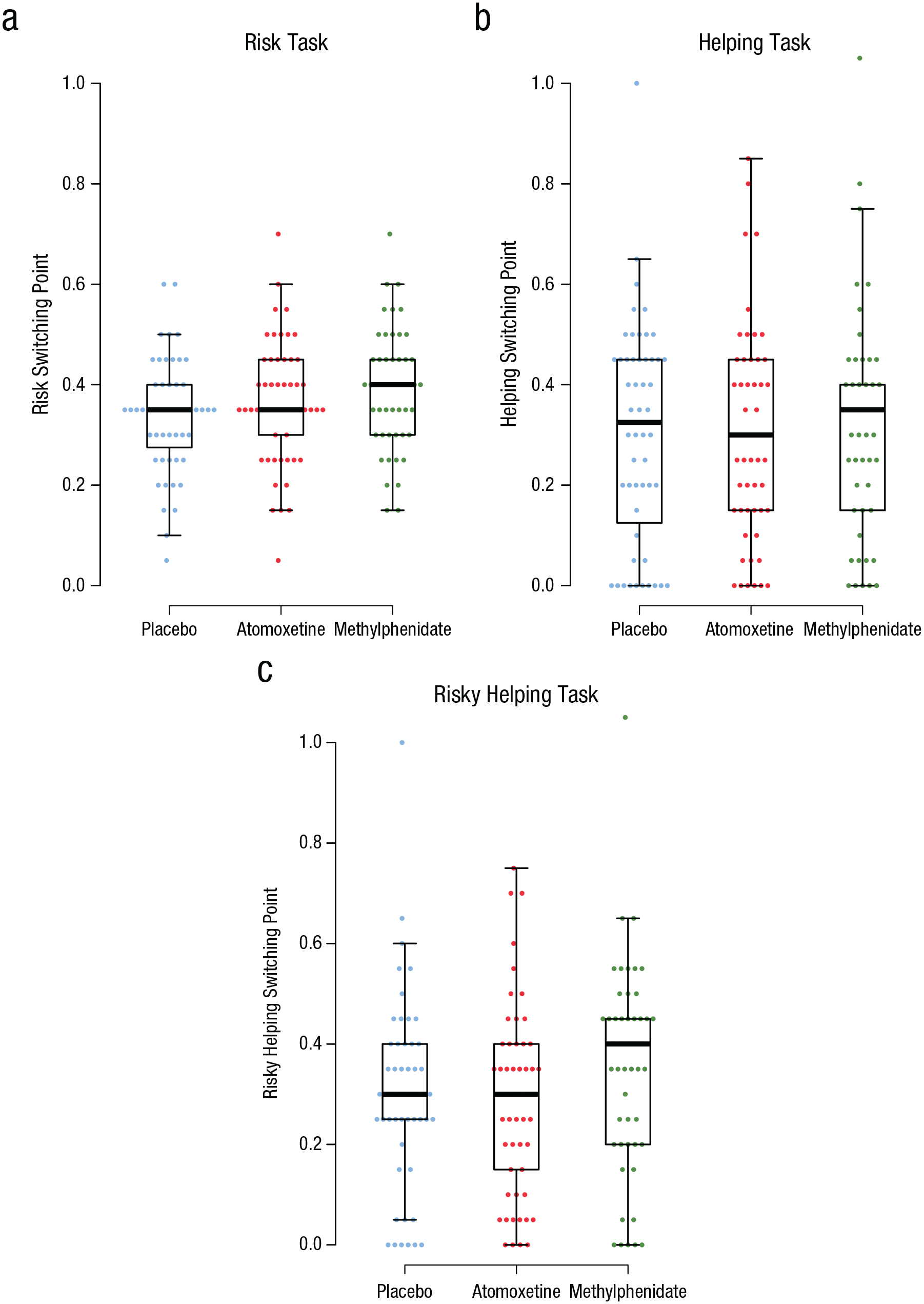
Distribution of switching points across drug conditions in the risk (a), helping (b), and risky helping (c) tasks in Study 2. In each box-and-whisker plot, the central line indicates the median, and the lower and upper hinges of the box indicate the 25th and 75th percentiles, respectively. Whiskers extend 1.5 times the interquartile range. Points show individual data.
There was no evidence that methylphenidate or atomoxetine affected social preferences (Fig. 4b; methylphenidate vs. placebo: b = 0.02, SE = 0.05, p = .728; atomoxetine vs. placebo: b = 0.00, SE = 0.05, p = .947; methylphenidate vs. atomoxetine: b = 0.02, SE = 0.05, p = .664). However, because of their higher willingness to take risks, participants who received methylphenidate helped more often in the risky helping task compared with participants who received atomoxetine or a placebo (Fig. 4c; atomoxetine vs. methylphenidate: b = 0.08, SE = 0.04, p = .030; methylphenidate vs. placebo: b = 0.07, SE = 0.04, p = .047). In contrast, atomoxetine did not increase risky helping compared with the placebo (b = −0.01, SE = 0.04, p = .787). Consequently, participants who received methylphenidate sacrificed more resources in the risky helping task, which resulted in higher overall welfare compared with participants who received a placebo or atomoxetine. Of participants who received methylphenidate, 22.4% helped even beyond the social-efficiency point under risk, compared with 11.5% and 13.2% who received a placebo and atomoxetine, respectively.
The effects of methylphenidate on risk attitudes and risky helping may have been driven by interindividual differences in drug-absorption rates, drug interactions with age or gender, or effects of drugs on mood. To examine these alternative explanations, we controlled for age and gender in the reported regressions and computed additional regressions in which we controlled for body mass index and regular medication (e.g., contraceptive pill), which both may influence absorption rates of the drug agent. We also controlled for mood and mood changes that have been shown to influence risk taking and social decision-making. Across these control analyses, drug treatment remained a robust predictor of risk and risky helping decisions (see Tables S6–S8 in the Supplemental Material). Further, when asked to guess what treatment they received, 38% of the participants guessed correctly (which is consistent with random guessing; i.e., chance level of 33%), χ2(1, N = 154) = 1.44, p = .230. Repeating the reported analyses only with participants who incorrectly guessed the treatment did not change the above conclusions (see Tables S6–S8).
Another possibility is that drug treatment makes decision-makers more erratic and thus increases choice inconsistency. This may have led to a spurious increase or decrease in switching points. Choice consistency in our tasks can be measured by looking at the number of switching points. A perfectly consistent decision-maker has one unique switching point in each task, whereas multiple switching points indicate intransitive choice. In the risk task, 76% of the participants had a unique switching point (compared with 86% in Study 1). In the helping task, 73% of the participants had a unique switching point (compared with 82% in Study 1). Lastly, in the risky helping task, 77% of the participants had a unique switching point (compared with 83% in Study 1). Although participants were slightly less consistent than in Study 1, we found no statistical evidence that the drug treatments made decisions noisier and more inconsistent in the risk task (methylphenidate vs. placebo: b = 0.50, SE = 0.98, p = .609; atomoxetine vs. placebo: b = 0.89, SE = 0.92, p = .337), helping task (methylphenidate vs. placebo: b = −1.55, SE = 1.06, p = .141; atomoxetine vs. placebo: b = 0.74, SE = 0.72, p = .306), or risky helping task (methylphenidate vs. placebo: b = −0.24, SE = 1.43, p = .862; atomoxetine vs. placebo: b = 1.64, SE = 1.28, p = .201). Reported results also remained robust when regression models controlled for choice consistency (see Tables S6–S8).
Discussion
Helping can have unforeseen negative consequences for the helper that render prosocial behavior not only costly but also risky. Here, we identified individual differences in social preferences that predict willingness to help and risk preferences that predict willingness to take risks, and we showed that people systematically integrate their risk preferences with their social preferences when helping is risky. Hence, we demonstrated that one cannot reliably predict decision-making in risky helping situations on the basis of a person’s risk or social preferences alone. Instead, someone who refuses to help under risk could be driven by a lack of social preferences or by risk aversion (or a combination of both). This means that refusals to help other people can be rooted in risk aversion rather than selfishness. In contrast, our data on receivers also revealed that they often misattributed failures to help to a lack of social concern rather than a lack of risk tolerance (for further details on these results, see the Supplemental Material). Thus, interventions aimed at promoting helping under risk might need to be adjusted toward reducing risk aversion (e.g., by emphasizing when fears of risk are unfounded) rather than toward increasing people’s moral obligation to care for the welfare of others independently of risks.
Because participants’ risk and social preferences were uncorrelated, our results suggest that both preferences are, to some degree, independently processed and then systematically integrated when social concerns need to be traded off against risk concerns. In line with this, methylphenidate led to more helping under risk because the substance selectively altered risk preferences but not social preferences. Meanwhile, atomoxetine changed neither risk nor social preferences and, as a result, also did not affect risky helping. Atomoxetine increases both norepinephrine and dopamine levels in the prefrontal cortex. Methylphenidate also inhibits norepinephrine reuptake but, in contrast to atomoxetine, also enhances dopamine neurotransmission in subcortical brain structures such as the striatum (Bymaster et al., 2002; Schulz et al., 2017). Our results fit the possibility that risk preferences are modulated by striatal rather than prefrontal dopamine (Fiorillo et al., 2003; Onge et al., 2010). Furthermore, because both drugs increase norepinephrine levels but only methylphenidate modulated risk preferences, results also suggest that if norepinephrine modulates risk preferences, it may do so especially—or only—when combined with striatal dopamine reuptake inhibition (Montes et al., 2015). Finally, our results suggest that subcortical dopamine transmission appears not to modulate social preferences. Hence, the effects of dopaminergic manipulations on social preferences found in previous research might be partly attributable to altered risk preferences.
From a practical perspective, both methylphenidate (sold under the trade name Ritalin) and atomoxetine (sold under the trade name Strattera) are prescription drugs used to treat attention-deficit/hyperactivity disorder and are regularly used off-label by people who aim to enhance their cognitive performance (Maier et al., 2018). Thus, our results have implications for the ethics of and policy for the use of psychostimulants. Indeed, the Global Drug Survey taken in 2015 and 2017 revealed that 3.2% and 6.6% of respondents, respectively, reported using psychostimulants such as methylphenidate for cognitive enhancement (Maier et al., 2018). Both in the professional ethical debate as well as in the general public, concerns about the medical safety and the fairness of such cognitive enhancements are discussed (Faber et al., 2016). However, our finding that methylphenidate alters helping behavior through increased risk seeking demonstrates that substances aimed at changing cognitive functioning can also influence social behavior. Such “social” side effects of cognitive enhancement (whether deemed positive or negative) are currently unknown to both users and administrators and thus do not receive much attention in the societal debate about psychostimulant use (Faulmüller et al., 2013).
Conclusions are constrained by some limitations of our study. Because our sample consisted of healthy volunteers only and because drugs such as methylphenidate can have different effects in clinical and nonclinical populations (Moll et al., 2003), it is not possible to generalize our results to clinical populations who take these drugs for medical purposes. Second, our results suggest that behavioral differences in risky helping are driven by the dopamine-specific effects of methylphenidate, but our conclusions about the possible role of norepinephrine are largely based on null findings. Future research could be focused on these issues, using larger samples and neuroimaging techniques that provide more precision with respect to observing neurobiological mechanisms. Third, individuals in our experiments knew the exact probabilities associated with different decision outcomes. Oftentimes, however, probabilities are less well-defined, thus creating ambiguity rather than risk. The Ellsberg paradox demonstrates that people are ambiguity averse; they prefer bets with known rather than unknown probability distributions of the outcome space. Vives and FeldmanHall (2018) showed that interindividual differences in ambiguity tolerance, but not risk attitudes, predicted prosocial choice and trust (also see FeldmanHall et al., 2016). Future work is needed to identify how the biobehavioral dissociation of risk and social preferences shown here generalizes to ambiguous situations.
Conclusion
Decision-makers can rarely be sure about how their decisions affect other people or their own welfare. Accordingly, social decisions often require an integration of social preferences and risk concerns that, theoretically, can have divergent effects on decisions and create a dilemma between helping others and avoiding risk to oneself. Our results highlight that risk preferences and social concerns are orthogonal, behaviorally dissociable preferences. A failure to help can reflect a lack of concern for other people or a personal aversion to risk—or both.
Whereas previous research revealed that risk and social consequences are processed in overlapping neural circuitries, our drug manipulation suggests largely independent mechanisms. Methylphenidate selectively altered risk tolerance and left social preferences unchanged—it increased helping under risk but not helping without risk. Atomoxetine, on the other hand, had no effect on either risk or social preferences. These drug-induced changes in risk taking and risky helping thus reveal a neurobiological dissociation of the processing of risk and social consequences in humans.
Supplemental Material
sj-pdf-1-pss-10.1177_09567976211015942 – Supplemental material for When Helping Is Risky: The Behavioral and Neurobiological Trade-off of Social and Risk Preferences
Supplemental material, sj-pdf-1-pss-10.1177_09567976211015942 for When Helping Is Risky: The Behavioral and Neurobiological Trade-off of Social and Risk Preferences by Jörg Gross, Nadira S. Faber, Andreas Kappes, Anne-Marie Nussberger, Philip J. Cowen, Michael Browning, Guy Kahane, Julian Savulescu, Molly J. Crockett and Carsten K. W. De Dreu in Psychological Science
Footnotes
Transparency
Action Editor: Daniela Schiller
Editor: Patricia J. Bauer
Author Contributions
J. Gross and N. S. Faber are joint first authors of this article. J. Gross, N. S. Faber, and C. K. W. De Dreu conceived of the project, developed the behavioral tasks, and designed and implemented Study 1; M. J. Crockett, A. Kappes, N. S. Faber, J. Savulescu, and G. Kahane conceived of and designed Study 2 with input from P. J. Cowen and M. Browning. Data collection and preparation were coordinated by J. Gross (Study 1) and A. Kappes, A.-M. Nussberger, P. J. Cowen, and M. Browning (Study 2). J. Gross analyzed the data. J. Gross, N. S. Faber, M. J. Crockett, and C. K. W. De Dreu wrote the manuscript and incorporated the coauthors’ revisions. All the authors approved the final manuscript for submission.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
