Abstract
Among the various metallic implant materials, titanium (Ti) alloy is the best choice for the long-term hard body tissue replacements such as hip and knee joints. It has excellent mechanical, superior biological and strong anticorrosion properties. Besides the appropriate implant material, its surface topology also plays a very important role in the success of any surgery. The surface topology is supposed to be the key factor for the promotion of osseointegration. This research article focuses its attention mainly on surface modification of Ti-based alloys by electric discharge machining process. Electric discharge machining is one of the most popular nonconventional manufacturing methods. Besides machining and surface modification of implants, it finds applications in almost all areas of manufacturing industries. This article reviews the various research work done on surface modification of Ti alloy by electric discharge machining and attempts to bring out the current scenario along with future directions. The area under focus for this research review is orthopedics applications. Furthermore, a new promising advancement in the area of enhancement of electric discharge machining performance for surface modification, particularly with regard to biocompatibility and surface quality of biomaterials, has also been introduced.
Introduction
Need for joint replacement
Over the last several decades, demand for joint replacement has increased rapidly. 1 It is estimated that 90% of the people above the age of 40 years suffer from degenerative and inflammatory diseases. Problems such as weakening of the bones (osteoporosis), inflammation in the bone joints (osteoarthritis), inflammation of synovial membrane (rheumatoid arthritis) and weakening of cartilage (chondromalacia) cause degradation of bone in continuous cyclic loading conditions and, thus, may cause degradation of mechanical properties which results in annoyance and loss in function. 2 This contributes to a rapid growth in the number of surgical procedures involving prosthesis implantation. To solve these problems, total joint replacement (TJR) is the solution where load-bearing joints or dysfunctional joint is replaced with artificial joint or implant. 3 Bio-implant is an artificial organ used for restoring the functionality of a dysfunctional natural organ or tissue of the body without adverse effect to other body parts. These can be implanted in the human body and let them become fixed and stable by natural biological self-bone formation process in order to improve the quality of life and longevity of human being. 4 Among all these implants, the replacements of knee, hip and spinal cord are extremely high. Figure 1 shows a typical hip and knee joint replacement.
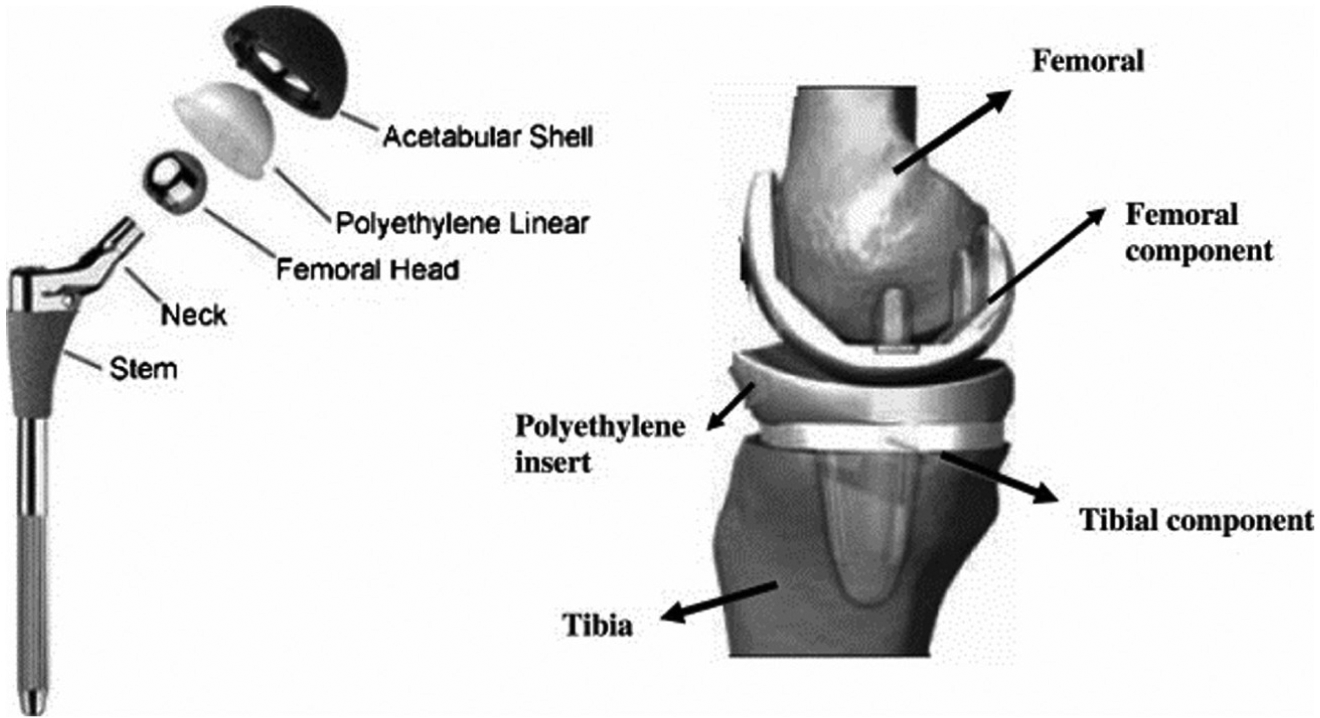
Total hip and knee joint implant replacements.
Properties required for bio-implants
The selection of the right biomaterials for a specific biomedical application depends upon their comprehensive properties such as biodegradability, biocompatibility, mechanical properties, nontoxicity, corrosion or wear resistance and mainly on surface chemistry and morphology. 5 A biomaterial must satisfy the following requirements.
Biocompatibility
Biocompatibility is determined by the surface characteristics of the implant material. The surface of biomaterials should be nontoxic and remain stable for a long time without causing any inflammatory or allergic responses. The ability of an implant material to withstand an appropriate host response in a specific situation measures the biocompatibility of a material. 6
Mechanical properties
Bio-implants are taken to defy in the host body under repeated cyclic loads or stresses. It is important that an implant material mimics as closely as possible the mechanical properties of the natural bone, especially elastic modulus equivalent to that of bone (5–30 GPa). 2 Other properties such as hardness, tensile strength and elongation should also be kept in mind while selecting the material for implant.
Surface topography
The nano-porous structure of interconnected pores with controllable size is required, which increases the surface area and provides a vehicle to interface implant with large-scale cells and tissues. The size of porosity has a favorable impact on cell adhesion, proliferation and new bone formation. 7 Controlled surface roughness and surface chemistry play a critical role in the formation of new bone ingrowths around implant.
Corrosion and wear resistance
The material of implant should have corrosion and wear resistance; otherwise, implant in body fluid may release ions causing allergic and toxic reactions. 2 The life of the implant is evaluated by its wear and abrasion performance. The low wear resistance causes loosening of implants and results in excessive pain and loss in functions.
Materials for bio-implant
To meet the joint replacement challenges, the development of various implant material has been witnessed, and the need of biomaterials is increasing rapidly. 1 The biomaterials are categorized as metallic, ceramics and polymers. Common metallic biomaterials are stainless steels (SUS-316L), cobalt-chromium (Co-Cr), titanium (Ti) and its alloys. 8 The nonmetallic biomaterials are polymeric based such as ultrahigh molecular weight polyethylene (UHMWPE), polyethylene terephthalate, lactide-co-glycolide and polytetrafluoroethylene. Silicon carbide (SiC), silicon nitride (Si3N4/TiN), zirconium oxide (ZrO2), titania (TiO2), titanium carbide (TiC), titanium nitride (TiN), bioglass, hydroxyapatite (HA) and aluminum oxide (Al2O3) are examples of ceramic biomaterials. 9 The mechanical properties of commonly used metallic implant materials compared with bone are summarized in Table 1.
Mechanical properties of metallic implant materials. 4
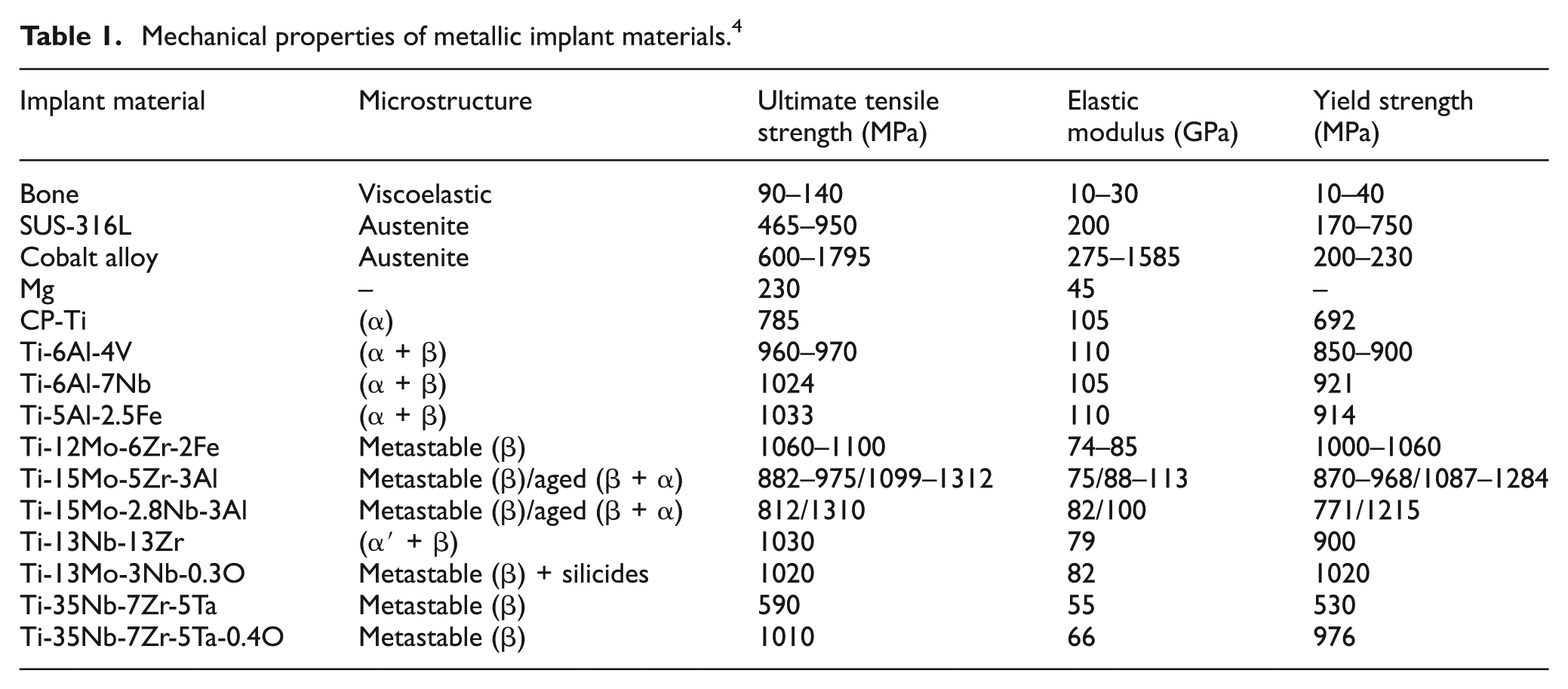
Due to wear and corrosion in the human body environment, the released elements such as cobalt (Co), nickel (Ni) and chromium (Cr) of SUS-316 and Co-Cr are found to cause toxicity within the individual. 2 In addition to this, ceramics and SUS-316 exhibit higher Young’s modulus and lower fracture strength than bone. 10 Ti and its alloys gained more attention in recent scenario, due to their superior biocompatibility and excellent mechanical properties especially low Young’s modulus, high fatigue performance, high corrosion resistance and low density as compared with other metallic biomaterials. 2 Earlier, CP-Ti and Ti-6Al-4V were proposed as biometals by the medical industries. 10 But the presence of Al and V restricts the applicability of Ti-6Al-4V because during abrasion, it releases ions and creates cytotoxicity which results in allergic reactions. 11 Despite this fact, the results of CP-Ti and Ti-6Al-4V cannot satisfy the requirements of biomaterials. So, the researcher developed biocompatible V andAl-free β-phase Ti alloy such as Ti-Nb, 12 Ti-Zr, 12 Ti-Nb-Zr, 13 Ti-Nb-Ta-Mo, 13 Ti-Nb-Ta-Sn 13 and Ti-Nb-Ta-Zr (TNTZ) alloys to overcome the toxicity of titanium–vanadium-based alloy. 14 Very recently developed β-type Ti alloys have lower elastic modulus of 55 GPa (near to bone 20 GPa), high strength, low density, excellent biocompatibility, good corrosion and wear resistance than CP-Ti and Ti-6Al-4V. 15
Corrosion fatigue behavior of metallic biomaterial
Titanium and its alloys are used for the preparation of implant and instruments used for clinical uses, such as plates, screws and nails, which always serve under cyclic load condition and sometimes fail due to fatigue and corrosion. Fatigue and corrosion fatigue performance are very important mechanical properties of the Ti alloys for biomedical application. Biomedical implants are subjected to pitting and crevice corrosion when intact with the aggressive environment of the body. From the beginning, stainless steel has been used in orthopedic applications. The failure of stainless steel by corrosion fatigue has been studied by Amel-Farzad et al. The dynamic condylar screw (DCS) barrel plate failed due to crevice corrosion. 16 Tavares et al. investigated the root causes of premature failure of stainless steel as orthopedic implant was removed from the patient’s body. The premature failure had occurred due to corrosion fatigue initiated by the high surface roughness. Pitting intergranular corrosion occurred in stainless steel–based implant under corrosion fatigue environment. Stainless steel releases ions and gets oxidized causing toxic and allergic reactions, leading to cause failure of implant. 17 Pitting corrosion occurred in cobalt-based alloy.18,19 Sudhakar et al. investigated the failure of cobalt alloy–based bone plate by corrosion fatigue. Crevice and pitting corrosion was found at the fractured surface after removal of the implant from the host body. 20 There are well-documented reports on the failure of cobalt-based implants by corrosion fatigue. 21 The crack growth was predicted under air or corrosive environment accelerated by fatigue performance. Moreover, recently, a case report pointed out the failure of cobalt-based femoral neck component of total hip arthoplasty. This was the first reported instance of Co-Cr modular neck failure. Co-Cr-based modular neck was subjected to fretting corrosion. 22 Figure 2 shows the failed modular neck of total hip arthoplasty. Figure 2(a) represents the enlarged view of the fracture site in the modular neck (the distal part is still in the female part of the Morse taper of the stem), and Figure 2(b) represents the proximal piece that has been turned over in the horizontal plane, and the two white arrows indicate the anterior and medial parts of the removable cone.

Failure of Co-Cr based modular neck (a) Represents the enlarged view of the fracture site in the modular neck (the distal part is still in the female part of the Morse taper of the stem). (b) Proximal piece has been turned over in the horizontal plane, and the two white arrows indicate the anterior and medial parts of the removable cone.
Fleck and Eifler studied the fatigue behavior of Ti alloy in air and Ringer’s solution. The fatigue performance in Ringer’s solution is less compared to air. The fatigue performance of (α + β)-type Ti-6Al-7Nb and Ti-6Al-4V was found higher than that of CP-Ti. 23 The fatigue performance of Ti alloys depends upon the microstructure,24–26 processing technique 27 and heat treatment methods. 28 So, the fatigue and the corrosion fatigue performance of Ti alloy vary accordingly. The fatigue crack growth behavior of Ti-6Al-4V in the air and NaCl solution was studied in detail. 29 The authors studied the fatigue behavior of Ti alloy with distinct microstructure obtained with different heat treatment processes. The results obtained from the study showed that the crack growth increases in saline solution as compared to air. Gregory 30 reported that the crack growth accelerated the corrosion fatigue failure in the aggressive body environment. Lee et al. 31 investigated the corrosion fatigue performance of Ti-6Al-4V in vacuum, air and 3.5% NaCl solution. They found that the crack growth was much higher in air and saline solution than in vacuum. In addition to these, they also pointed out the effect of microstructure on the fatigue performance of the Ti alloy. Niinomi et al. studied the fatigue behavior of Ti-5Al-2.5 Fe alloy in air and Ringer’s solution and compared it with Ti-6Al-4V extra low interstitial (ELI) alloy. The crack growth was accelerated slowly in air for both materials, and from the plots of da/dN versus ΔK, the pure corrosion fatigue behavior was observed in both the materials. 32 Williams and Chawla 33 recently reported the failure of Ti-6Al-4V-based hip implant neck by fretting and crevice corrosion. The fretting and crevice corrosion reduces the fatigue strength of Ti-based neck of hip implant. The fatigue crack growth was created by the aggressive body fluid which accelerates the crevice corrosion resulting in failure of the joint. Figure 3 shows the image of fractured neck of the hip joint and presents the evidence of superficial fretting corrosion. From the best knowledge of the authors, most of the published literature available on the failure of Ti-6Al-4V-based implants occurred at the neck.34–42 Paliwal et al. reported the fatigue failure of three cementless Ti-6Al-4V-based hip stems. The main cause of the failure was surface cracks during fretting fatigue. 43 In another study, the hydrogen embrittlement was found to be the main cause for the fretting and corrosive corrosion. 44 Yokoyama et al. 45 also reported that Ti-based dental implant becomes brittle and fracture following the exposure to hydrogen. In the case of Ti-6Al-4V alloys, the vanadium ions dissolve and release by crevice corrosion in the aggressive environment of the body, causing an allergic reaction leading to adverse effect to human tissues. In the replacement of harmful vanadium (V) and aluminum (Al) elements, the establishing alloying elements niobium (Nb), tantalum (Ta) and zirconium (Zr) have been offered as an alternative with excellent biocompatibility.28,46–48 Rubitschek et al. 49 studied the corrosion fatigue behavior of ultrafine-grained NbZr alloy. The NbZr alloy offers no crack imitation at high cycle corrosion fatigue tests in simulated body fluid (SBF).
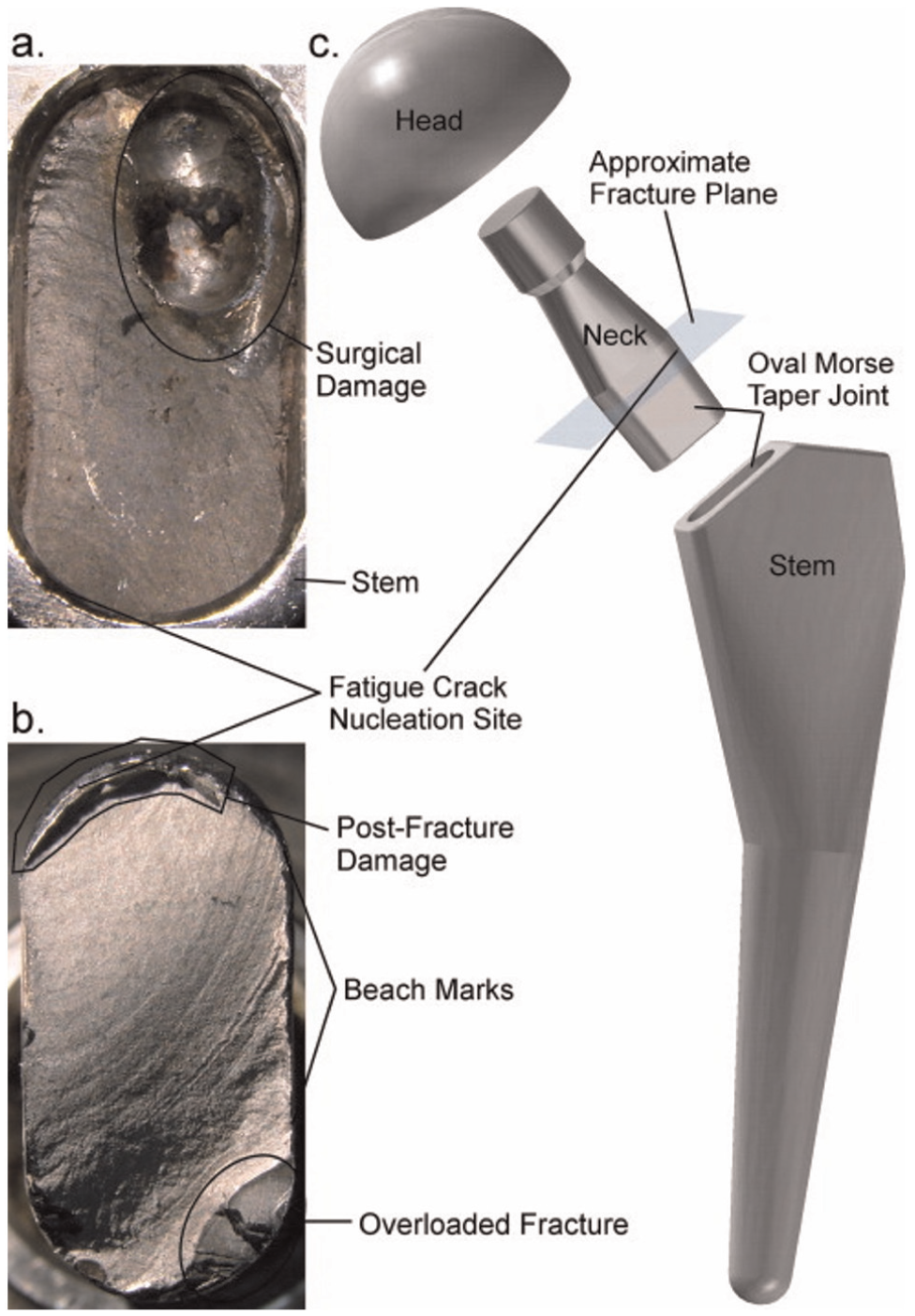
(a) Optical photograph of the fracture surface of the neck that remained in the stem. The surgical damage was created by a tool used to remove the implant from the body. (b) Optical photograph of the fracture surface of the neck that remained attached to the head. Post-fracture damage occurred when the fractured neck slid across the inside of the stem’s taper surface. (c) Exploded model of the modular hip implant, showing where neck fracture occurred. Both the neck and stem were made of Ti-6Al-4V.
Akahori et al. studied the fatigue behavior of TNTZ alloy and compared its performance with Ti-6Al-4V ELI and Ti-6Al-7Nb alloys in air and Ringer’s solution. The specimens were prepared with distinct conditions. The plates are designated as TNTZCR when these solutionized bars were cold rolled to the plates with a thickness of 2.5 mm and by a reduction ratio of 87.5% at ambient temperature. These plates are designated as TNTZST when solutionized at 1063 K for 3.6 ks in Ar gas atmosphere followed by water quenching (WQ). The specimens did not fail up to 107 cycles in air at room temperature. Figure 4 shows the comparison of Ti alloy with distinct treatment conditions. The results show that the fatigue strength of a TNTZ alloy increases as the aging temperature increased. The fatigue performance of Ti-6Al-4V ELI equiaxed α is higher than other Ti alloy which is around 880 MPa. The fretting fatigue of TNTZST and TNTZST aged at 673 K was also studied in Ringer’s solution. It was found that TNTZ alloy is not degraded in Ringer’s solution. 50 Baptista et al. studied the corrosion fatigue behavior of another type of β-phase Ti alloy (Ti-13Zr-13Nb) in air and 0.9 wt% NaCl solution. It was found that the alloy was insensitive to both the environments. 51 Boehlert et al. 52 have also reported the insensitivity of β-phase-type Ti-15Al-33Nb and Ti-21Al-29Bn alloys against corrosion fatigue.
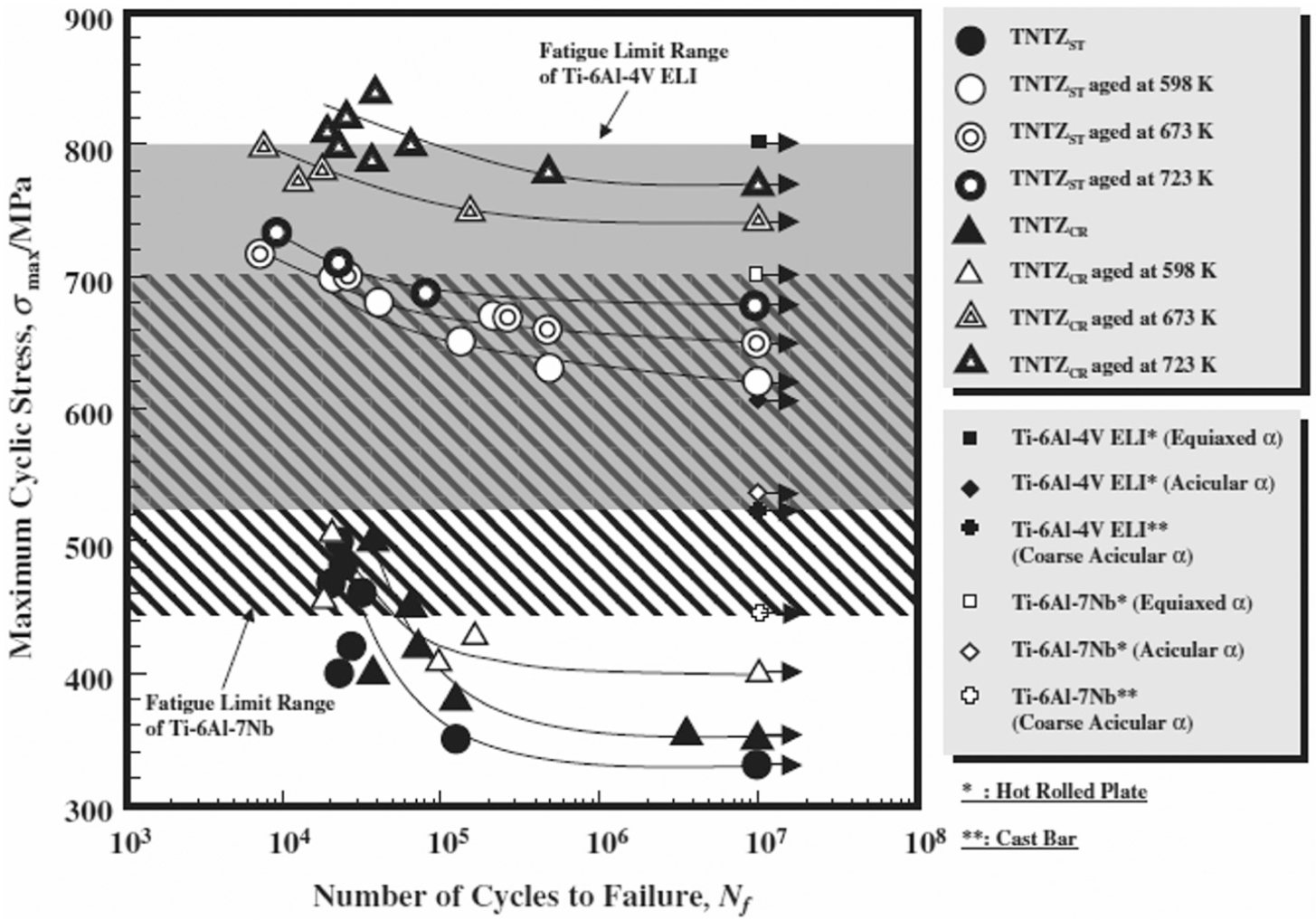
S-N curves of TNTZST, TNTZCR and TNTZST, and TNTZCR conducted with aging at 598, 673 and 723 K for 259.2 ks with those of Ti-6Al-4V ELI and Ti-6Al-7Nb in air.
Need for surface modification
Despite their excellent mechanical properties, Ti-based implants have the shortcoming of inability to meet the requirements of osseointegration, that is, the incomplete bonding between the implant and the surrounding tissues. The surface of implant needs to be subjected to some mechanical treatments to maximize its bioactivity when interfacing with surrounding natural tissue. Surface modification is required for better stability and biological fixation of the implant, so that a homogeneous stress transfer takes place between bone and implants. Surface modification is a process of alteration of the key bulk properties of the material by removing or addition of material on the surface of implant. 7 In other words, surface modification is basically required for successful bone regeneration and healing. The main events after implantation include adaptation of surrounding tissues and start bone formation by the biological self-healing process. The interaction between the bone and the implant surface at different topographical scales is shown in Figure 5.
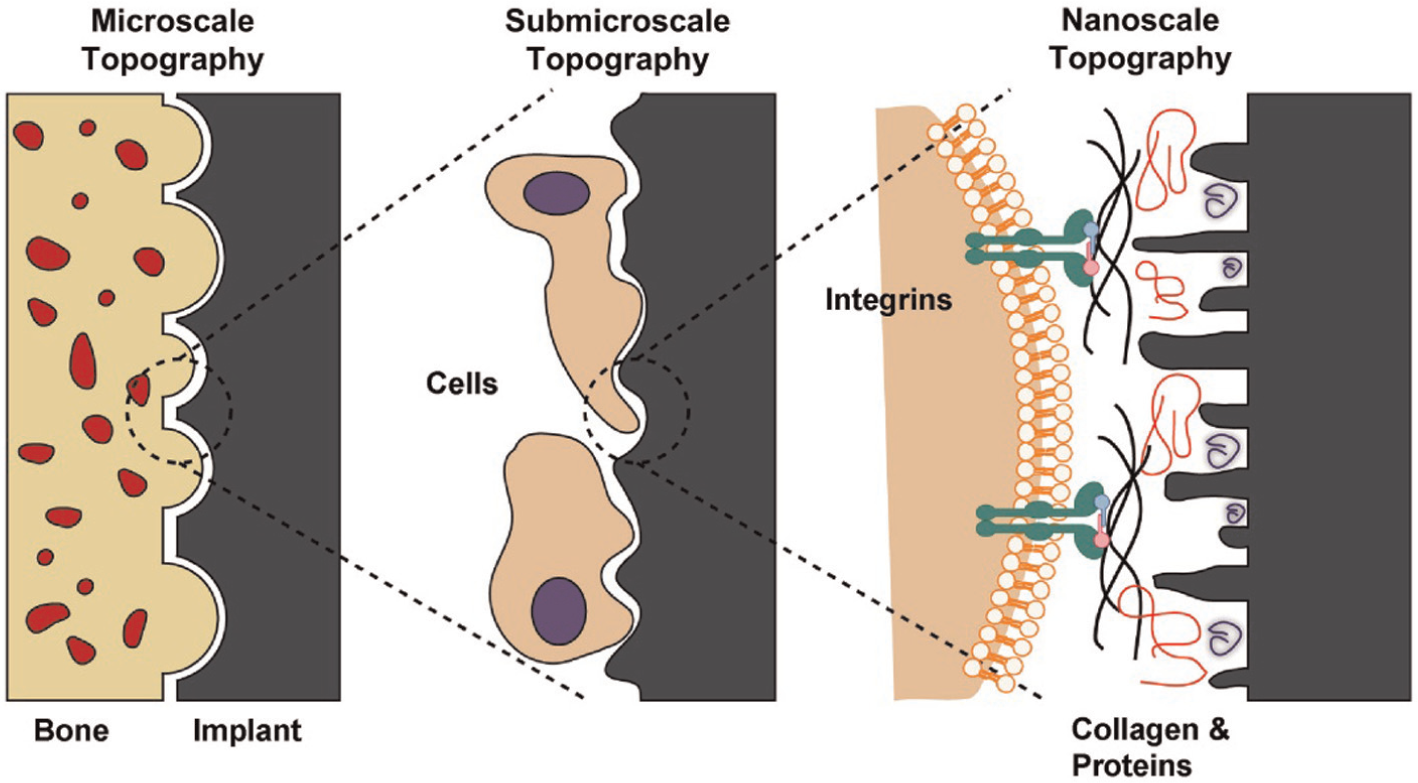
Schematic representation of the interactions between bone and implant surface at different topographical scales.
The past research work shows that surface properties play a critical and a very important role in new bone formation 53 and further growth of tissues. 54 Surface properties such as surface roughness, 55 surface topography, 56 surface porosity, 57 surface chemistry58,59 and surface energy60,61 have found significant impact on new tissue generation and collagen or protein absorption during in vivo or in vitro study.62,63 The surface roughness has been categorized into three different scales, namely, macro (100 µm < Ra > millimeters), micro (1 µm < Ra > 100 µm) and nano (100 nm < Ra > 1 µm).64,65 These different scales of surface roughness have their own impact on bone ingrowths. 56 Generally, micro-scale rough surface is preferred because large surface indentations provide a better interaction of implant to the surrounding bone tissues. 66 This large to surface volume provides a vehicle to interface implant with large-scale cells and tissues; 67 thus, macro-roughness usually promotes cell adhesion and their spreading. 68 However, it was found that the surface of micro-scale roughness indentations is an arguable characteristic promoting cells and tissues around implant surface. 69 Over the last several decades, few researchers have drawn their attention toward the surface with nano-scale roughness irregularity. 7 It was found that nano-scale surface roughness of the material surface has been considered as the most advantageous and advisable factor for the formation, adhesion, growth and differentiation of new bone cells. 70 The cell reacts with nano-scale surface and their surrounding. 71 The growth behavior of rat calvaria bone cell on the surface with nanometric features was found. 72 The nano-structured surface increased the function of osteoblast or cell growth. 73
The proper and appropriate surface treatment or modification not only adds or removes material on the implant surface but also improves surface morphology and chemistry as per required by different biomedical applications. Table 2 presents the various surface modification techniques used to enhance the performance of implant. 8 The design and development of surface modification techniques such as plasma spraying, 74 physical vapor deposition (PVD), 75 chemical vapor deposition (CVD), 76 nitriding,77,78 anodization 79 and laser cladding 80 are essential for improving their surface quality. However, the practice of using these techniques as surface modification for Ti alloys frequently becomes ineffective over a long duration due to poor adhesion and low physical bond strength and enables to serve under cyclic loading condition in a corrosive environment. 2 These techniques required surface preparation before coating such as shot peening (SP) and grit blasting, which increases the processing cost. More recently, the importance of electric discharge machining (EDM) has been widely acknowledged in fabricating nano-structured biocompatible surface and has found favorable impact on the biological attachment of human osteoblastic cell. 81 EDM is a potential candidate to produce biocompatible surfaces. There is no requirement of surface preparation before EDM and also converts the surface into an oxide layer of controlled thickness, which in turn increases the biocompatibility of the substrate surface and provides a better vehicle for cell attachment and growth. In comparisons to the other surface modification techniques, EDM offers the formation of carbides on the top machined layer which increased the hardness (Hv), 82 wear 83 and corrosion resistance 84 of the material surface. Apart from this, complex geometry and three-dimensional shapes can be easily fabricated by EDM process. EDM is a nonconventional machining process and has been widely used to machine hard and tough materials which are difficult to machine by conventional machining method. It uses repeated series of electrical discharges to machine the conductive materials. In EDM process, there is no direct contact between the tool and the workpiece. So, vibration and chatters are almost 0, and thus, any conductive and complex shapes within very close tolerances can be machined or manufactured or modified easily. 85 Because of all these advantages, it finds applications in mold, die, automotive, aerospace and medical industries. 86
Surface modification techniques for titanium alloy as biomedical implant. 8
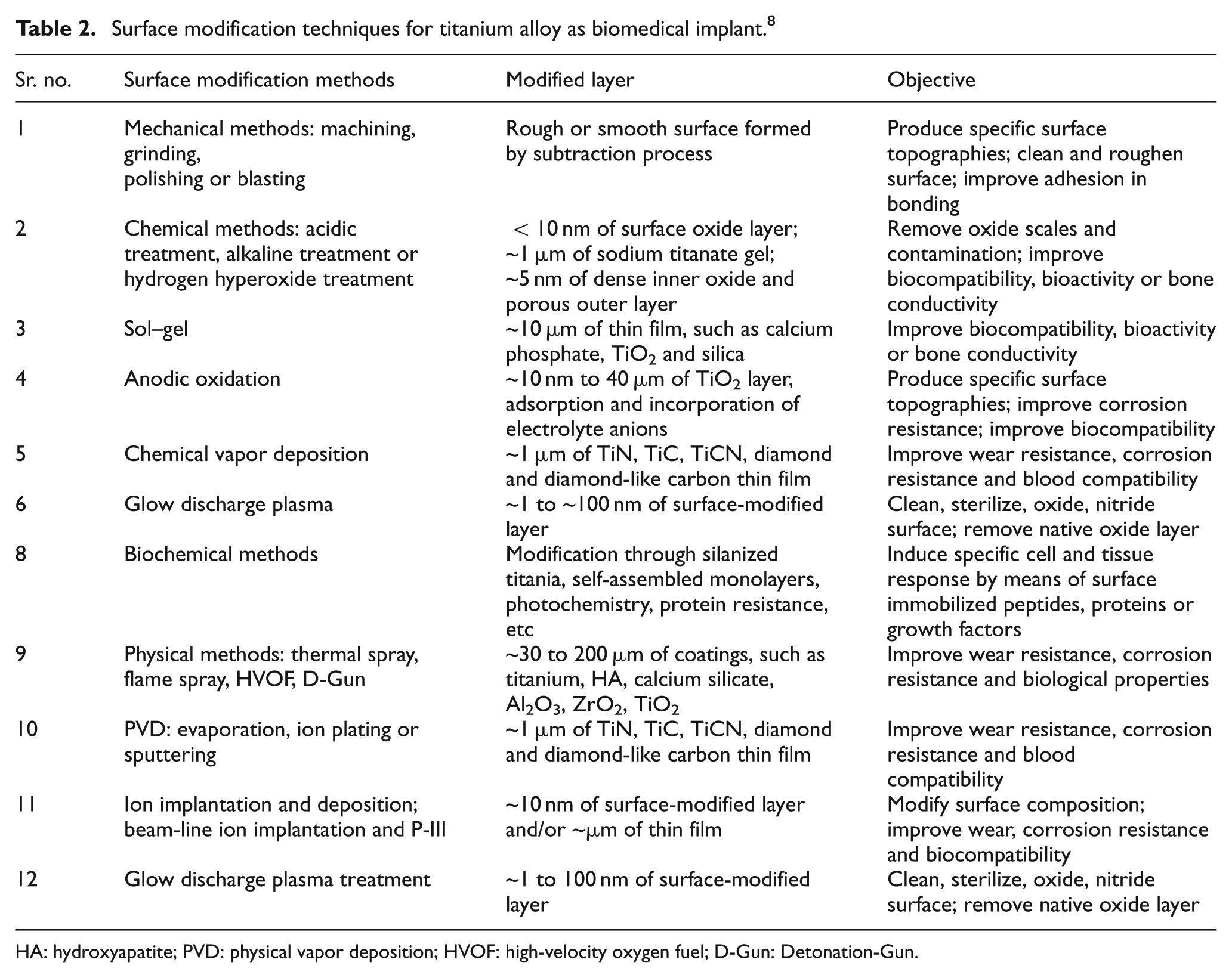
HA: hydroxyapatite; PVD: physical vapor deposition; HVOF: high-velocity oxygen fuel; D-Gun: Detonation-Gun.
A number of studies are available for the machining of aerospace superalloy 87 and titanium alloy 88 by EDM process. Ndaliman et al. investigated the influence of two dielectric fluids on the enhancement of surface properties of Ti-6Al-4V alloy by EDM process. Urea solution and distilled water were used as dielectric fluids, and Cu-TaC was used as electrode. It was found that the distilled water dielectric gave a better surface finish and the urea solution enhanced the surface hardness. 89 The performance of a special grade of graphite electrode was investigated in the machining of Ti-6Al-4v alloy by EDM. The best results were obtained at negative polarity of the electrode, and TiC was found in the recast layer regime. 90 Surface integrity, surface topography and electrochemical corrosion resistance of intermetallic titanium aluminide (γ-TiAl) were investigated after EDM process. Copper, graphite and aluminum were used as a tool electrode for surface treatment of γ-TiAl alloy. It was found that the graphite electrode produces carbide phases on the machined sample, and aluminum creates oxides. The samples machined by graphite offer more corrosion resistance than copper and aluminum. 91 The process capability of Ti and its alloy by wire-electric discharge machining (W-EDM) has been studied by various authors. Pure Ti 92 and Ti-6Al-4V 93 alloy have applications in aerospace, marine and biomedical industries and can be processed very precisely by W-EDM process. Three different grades of Ti and its alloys are machined using EDM after deep and shallow cryogenic treatment. 94 Due to deep and shallow cryogenic treatment, the thermal conductivity of the material was increased, as a result of which, increase in material removal was observed. Some elements were found in energy-dispersive x-ray spectroscopy (EDS), which shows the existence of material migration phenomena. The improvement in the fabrication accuracy of microhole in Ti-6Al-4V alloy by changing polarity in micro-EDM has been attempted recently, which has tremendous applications in biomedical microdevices and automotive, aerospace and microelectromechanical systems. 95
In the EDM process, a number of efforts have been made in the past to enhance its performance for surface modification. These are (a) rotary motion or orbital motion to electrode;96,97 (b) ultrasonic vibration assisted98,99 and (c) powder mixed additive in dielectric fluid of EDM.100–104 Among all these, powder mixed electric discharge machining (PMEDM) is extremely used, and a number of studies are available on the enhancement of EDM performance by adding conductive or semi-conductive micro-sized 105 and nano-sized powder. 106 Furthermore, Tan and Yeo explored that nano-sized powder mixed dielectric in micro-EDM plays a very vital role to enhance the surface quality and surface properties of workpiece specimen. The effect of nano-powders on surface roughness and recast layer thickness was studied. Low powder concentration, low sub-microsecond pulse duration and tool electrode rotation have shown a significant effect on the reduction of recast layer thickness. 107 Furthermore, they found the application of PMEDM for enhancing the surface hardness of stainless steel and titanium alloy. Tantalum carbide (TaC) powder at a concentration of 25 g/L was added to kerosene dielectric fluid to enhance the surface properties of stainless steel and the surface with improved hardness, and a high corrosion resistance was found. 108 The surface hardness of Ti alloy (Ti-6Al-4v) was enhanced by the use of 10 g/L urea concentration with Cu-TaC electrode. The highest micro-hardness of Ti alloy of 1795 Hv was achieved. 109
After the thorough scrutiny of the available literature on the use of Ti alloy after its machining and surface modification by EDM process, it has been concluded no research article is available which can summarize the latest developments along with the application and future trends in use of EDM for machining and surface modification of Ti alloy for biomedical applications. Thus, this research article summarizes the critically reviewed available literature on machining of Ti alloy by EDM for orthopedic applications. Current review initially describes that among Ti alloys, β-phases are the ultimate choice for biomedical applications. It also describes why surface modification of implant is required prior to implantation in the host body. In section “EDM,” the potential of EDM for surface modification of metallic implants has been explored. Section “Historical perspective for surface modification of metallic implants by EDM” traces the historical view of the diligence of Ti alloy machined or modified by EDM. A new innovative methodology which so far has not been used for surface modification of bio-implants is introduced in section “An innovative method for surface modification by material migration in EDM.” The application and future trends are followed by concluding remarks.
EDM
EDM involves the removal of material from workpiece by thermal energy created by a series of high-frequency electrical sparks between the tool and the workpiece. The conventional representation of the experimental setup is presented in Figure 6. The tool and workpiece maintained a very small gap in dielectric fluid. Figure 7 shows the concept of spark erosion in EDM process. 110 When a voltage is put up at a small gap, a high strength electric field is set up, which ionizes the dielectric fluid. Plasma channels are generated, and electrical discharges at very high frequency take place at least resistance point between the electrode and the workpiece. Due to this, a very high temperature around 20,000 °C is generated causing to melt the material from both the tool electrode and the workpiece. 111 As a result, small craters are formed on both the tool electrode and the workpiece surface. After the final stage of discharge duration, the temperature of plasma drops rapidly, 110 and the resulting products of spark erosion are cooled and flushed away in the form of debris, 112 and few remain solidified on the surface of the workpiece in the form of a recast layer which is composed of oxides and various carbides.113,114Figure 8 shows the schematic representation of the principle of spark erosion in EDM process by voltage and current waveform. The electrical sparks that follow are transferred to the next discharge to come about in the closest spot far from the previous release to maintain EDM process stability. Such a discharge location may be the spot where the gap is small or contaminated with eroded debris particles which may weaken the breakdown strength of dielectric liquid. 110 Accordingly, the pulse interval (pulse-off) time between pulse discharges must be sufficiently long, so that the plasma generated by the previous discharge can be deionized, and the dielectric breakdown strength around the previous discharge location can be recovered by the time the next pulse voltage is applied. 110 The material removal in EDM process is still arguable. 115
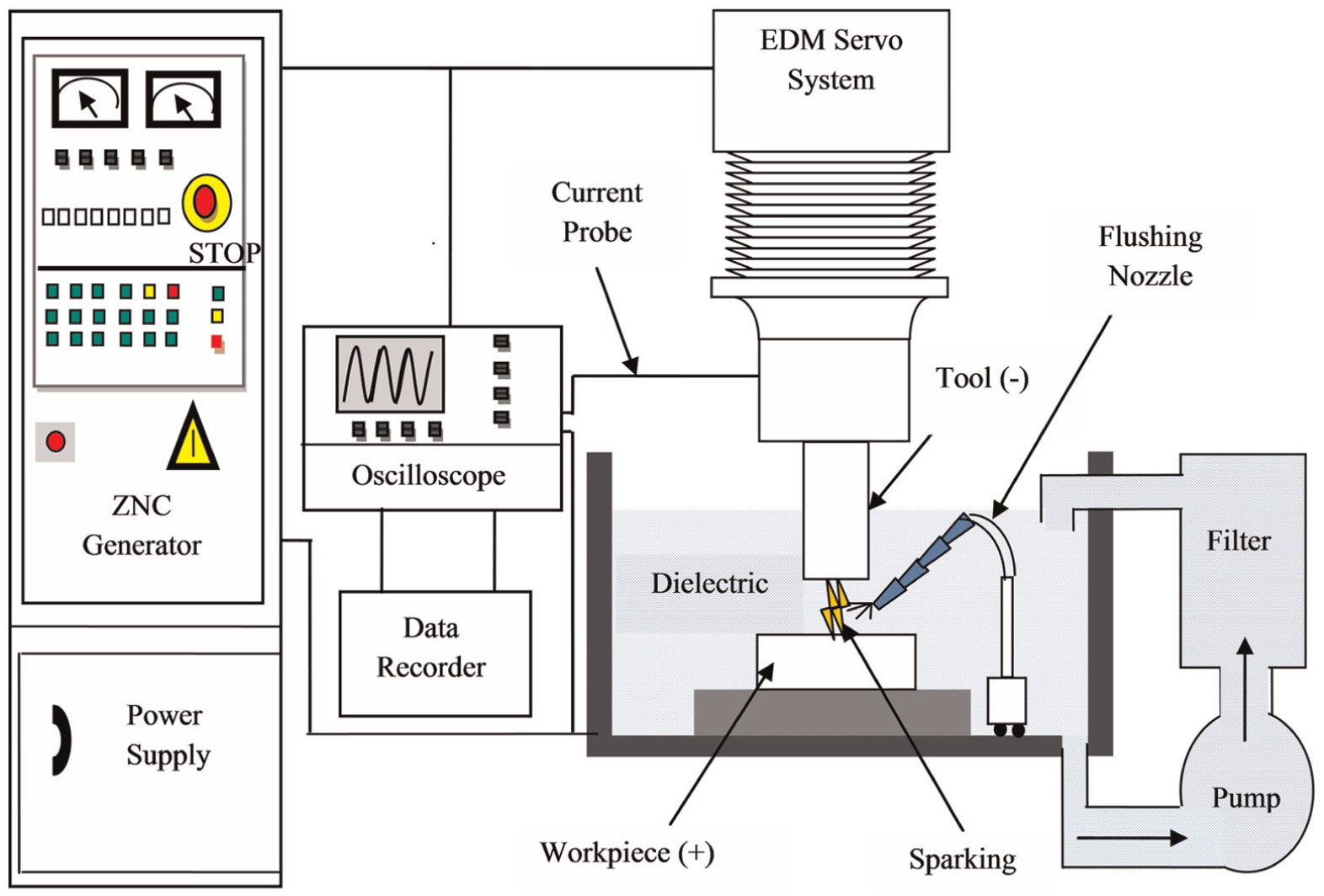
Typical experimental setup for EDM process.
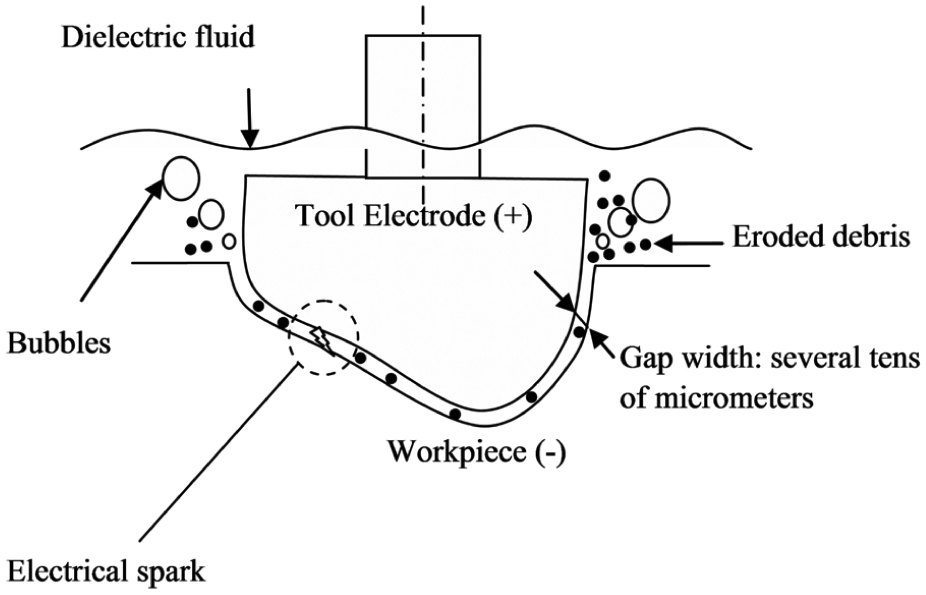
Concept of spark erosion in EDM.
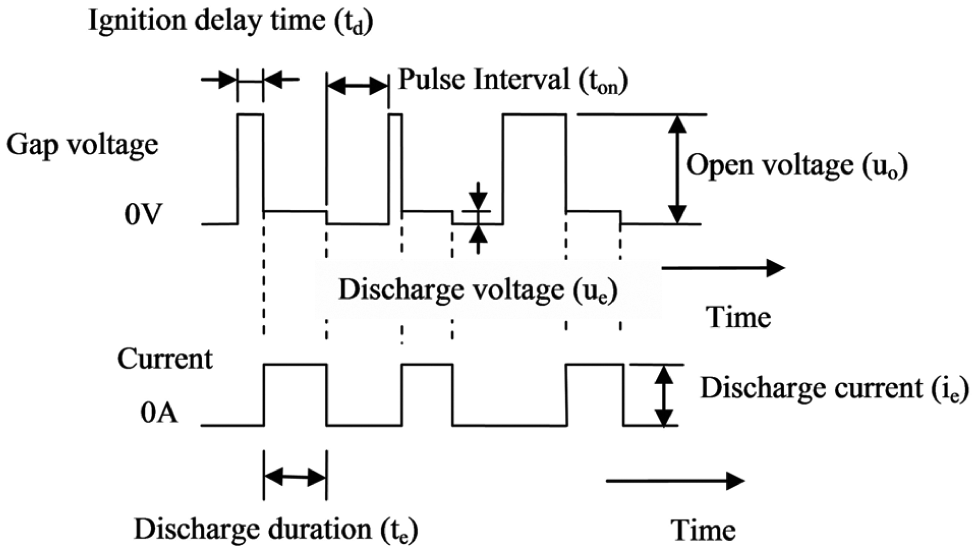
Principles of spark erosion in EDM process.
Historical perspective for surface modification of metallic implants by EDM
EDM process has been widely used to machine and modify the surface of new intelligent and hard materials. EDM is attracting broad interest in the current research scenario for surface modification of metallic implants for biomedical applications. 116 Ti and its alloys are one of them and extremely used in biomedical applications. The use of Ti and its alloy is increasing because of their excellent properties and biocompatibility, which has already been discussed in detail in the previous section. This section identifies the use of EDM in the past to improve the biocompatibility to enhance its biological applications.
Enhancing the osseointegration
EDM has been able to improve the surface properties of implants, in such a manner that the healing time is shortened and the bone formation increases. Following studies have been critically reviewed on the capability of EDM in in vitro and in vivo behavior.
In vitro cast study
Numerous studies have reported on EDM as surface modification method for improving the osteoconductivity of titanium and other alloys. The effect of cell attachment, proliferation and differentiation of human osteoblastic cell has been shown in in vitro analysis. Chen et al. 81 have examined the potential of EDM to modify the surface of Fe-Al-Mn alloy for orthopedics application and dental applications (Figure 9). It is clear from Figure 9 that the surfaces treated by EDM provide better and potential surface for osteoblastic cell attachment. It was also observed that cell highly adopted the specimen treated by EDM after 24 h of cell culture times. The nano-porous surface of Ti implant modified by EDM has found a favorable impact on the adhesion and proliferation of human osteoblast-like MG-63 cell responses to the specimen.117–119Figure 10 shows the scanning electron microscope (SEM) images of MG-63 cell attachment and proliferation on the unmachined and EDM-treated surface. The cells were widely spread and completely covered the unmachined surface and electric discharge machined (EDMed) surface. It was found that EDMed surface shows the higher growth factor for the attachment of human osteoblastic cells (MG-63).
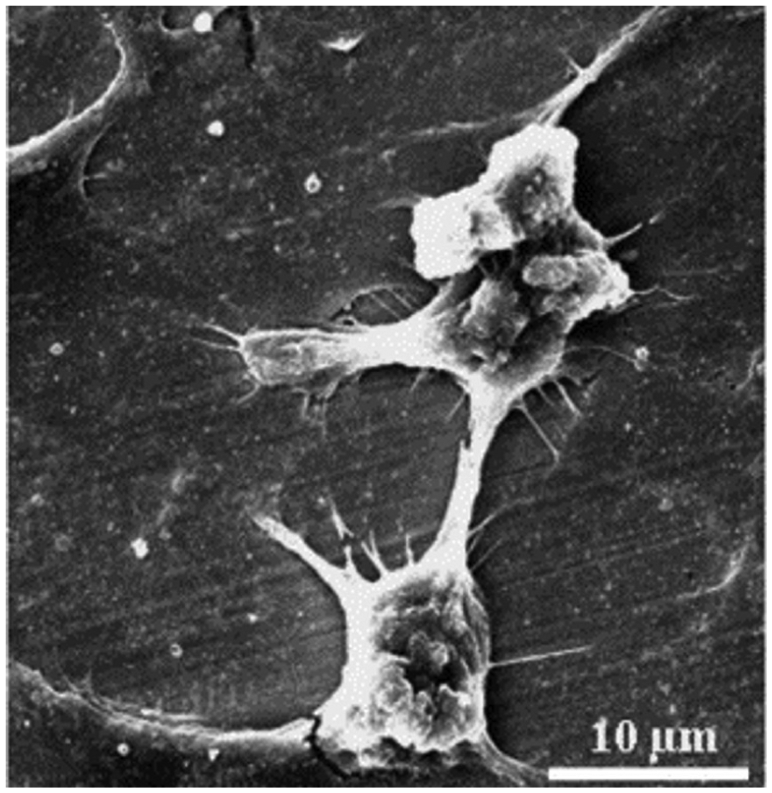
SEM image of Fe-Al-Mn alloy attachment after 24 h of culture times.

MG-63 cells attachment and proliferation on the (a) Untreated specimen. (b) EDMed specimen (at 10µs pulse duration). (c) EDM specimen (at 60µs pulse duration) after day 7 of cell culture.
The biological response in terms of osteogenic gene expression of mesenchymal stem cells on Ti surface processed by W-EDM was investigated by Tamaki et al. 120 Titanium surface processed by W-EDM promotes the cells attachment. Furthermore, Otsuka et al. 121 also explored the effect of surface topography and chemistry of Ti surfaces machined by W-EDM on the attachment and proliferation of MC3T3-E1 cells. Figure 11(a) and (b) shows an SEM image of the cell attachment morphology and spread of MC3T3-E1 cells cultured for 3 days on conventional machined surface and EDM surface, respectively. MC3T3-E1 cells attached with EDM-treated samples and proliferation reveals that cell numbers increased significantly on the EDM surfaces as compared to the machined surfaces. It was found that cells were elongated in all directions, and pseudopodium-like structures were observed on EDMed surface. The cells were polygonal in shape, with many thin filopodia attached to the Ti surface treated with EDM process.

(a) SEM image showing the morphology of spread MC3T3-E1 cells cultured for 3 days on a machined surface and (b) morphological assessment of cells cultured on an EDM surface.
A possible application of EDM to fabricate rough biocompatible surface for orthopedic applications was reported by Harcuba et al. 122 They reported that carbon-enriched highly macro-roughness has a favorable and better surface for adhesion and proliferation of human osteoblast-like MG-63 cells and compared with the sample prepared by plasma sprayed with TiO2 as represented in Figure 12. Strasky et al. 123 reported in their research that the Ti-6Al-4V alloy surface has been modified by combining EDM, acid etching and SP to fabricate biocompatible surface suitable for osteoblast proliferation. The treated surfaces were characterized by SEM and EDS. This work demonstrates that surface morphology has the micro-, macro- and nano-scale structures, which play an important role to promote osseointegration. MG-63 cells were seeded on machined surfaces, and the results show that the introduction of such structures enhances osteoblast differentiation and local factor production. Furthermore, a novel approach has been adapted by Havlikova et al. 124 to improve the biological performance of Ti-6Al-4V alloy for orthopedic applications by three-step mechanical treatments using the combination of conventional machining processes (SP and chemical milling (CM)) followed by EDM. The surface is modified to provide a more suitable and favorable surface for adhesion and proliferation of human osteoblast-like MG-63 cells.
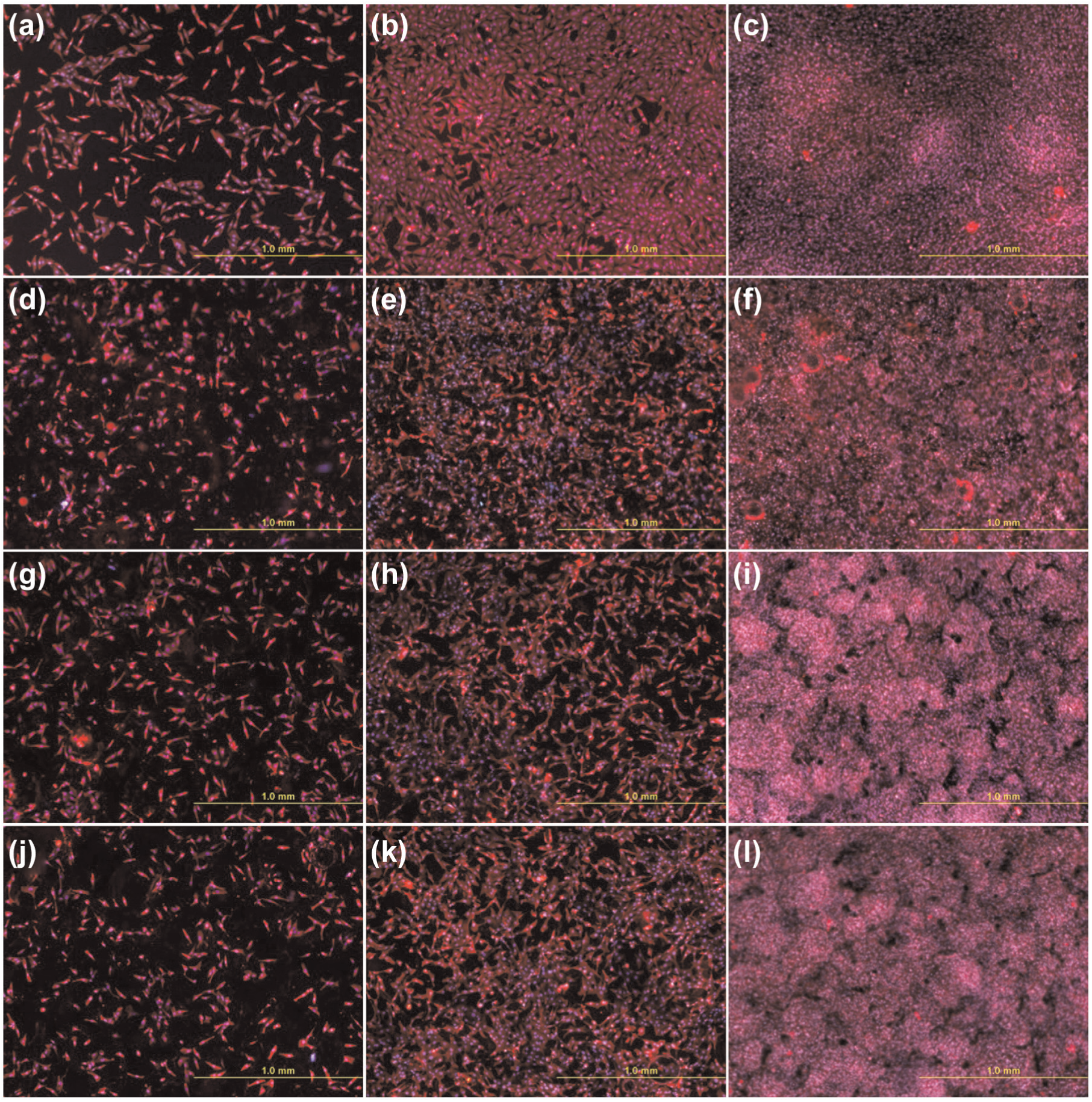
(a–c) MG-63 cells in cultures on polystyrene dishes, (d–f) Ti-6Al-4V treated with plasma spraying, (g–i) EDM and (j–l) EDM with subsequent annealing on (a, d, g, j) day 1, (b, e, h, k) day 3 and (c, f, i, l) day 7 after seeding.
Evaluation by in vivo
Carbon-enriched oxide surface has been fabricated on Ti-based implant. 125 White rabbits are sacrificed in this in vivo analysis. Bone formation response has been observed around EDMed titanium surfaces with different oxide layer thicknesses and sub-micro-meter topography. Furthermore, Yamaki et al. 126 also studied the bone biocompatibility in vivo. A biocompatible micro-fabricated surface with an irregular morphology was fabricated by W-EDM process as seen in Figure 13 (a) and (b), which show an SEM image of EDM surface and machined surface, respectively. The bone formation around screw shape implant processed by EDM and conventional machining process was investigated by in vivo study using four Japanese white rabbits. Figure 14 represents that higher bone formation takes place around implant processed by EDM than that at machined surface implant. Thus, EDM seems to be a possible technique for surface modification of the implants to support enhanced osseointegration.

SEM images of the surface morphology of (a) EDM surface and (b) machined surface.
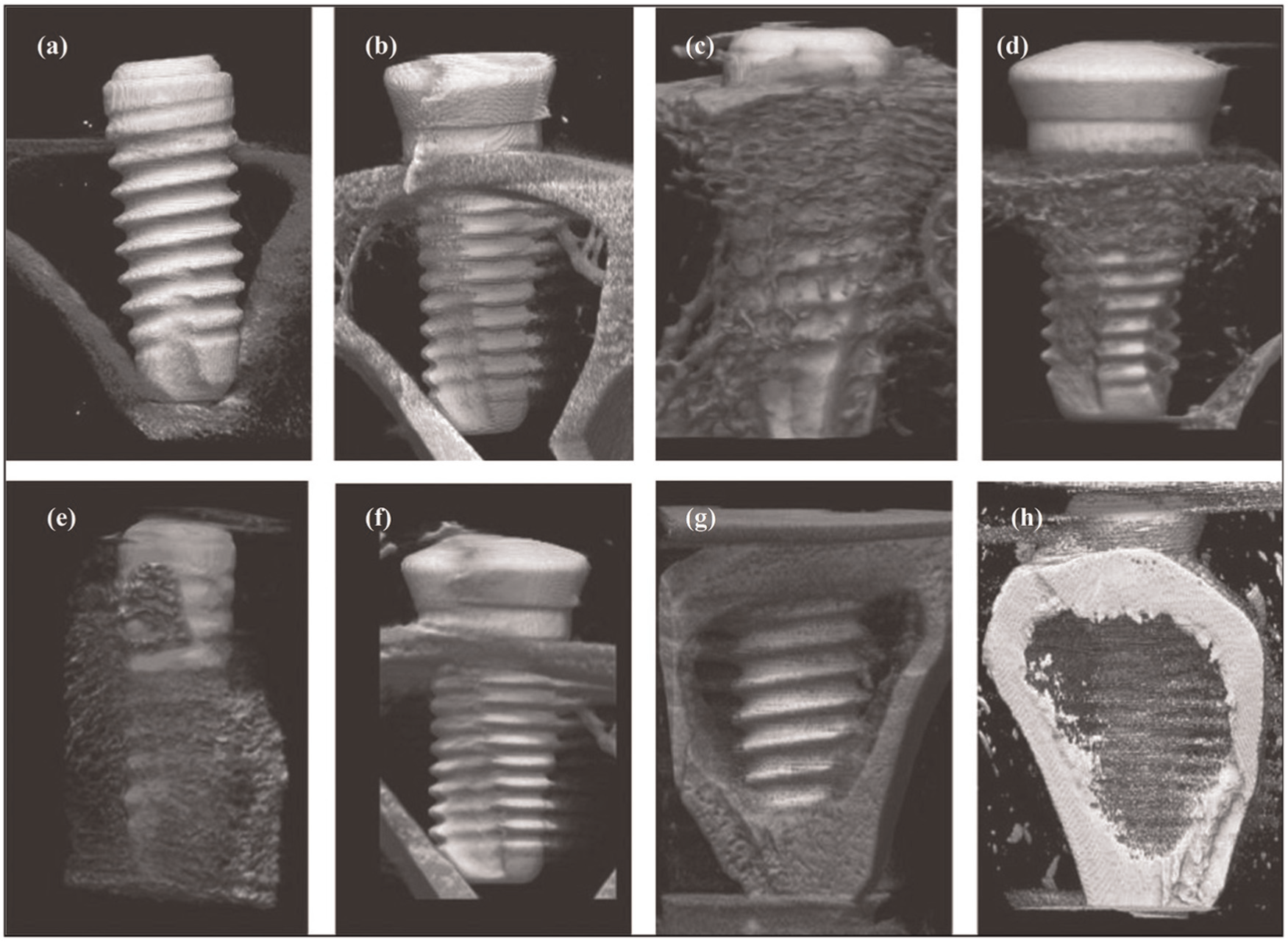
Micro-CT images of EDM-treated implant surrounded by bone tissue at (a) 0, (c) 1, (e) 2 and (g) 4 weeks of post-implantation and machined implant at (b) 0, (d) 1, (f) 2 and (h) 4 weeks of post-implantation.
Enhancing surface integrity
Controlled surface roughness
The spark-eroding process generates the rough oxide surface containing titanium-, oxygen- and carbon-enriched layers. They found that surface roughness allows bone formation and bone fixation due to surface modification of high roughness values (Ra≈ 7.8 µm). 122 Under the controlled machining condition, surface roughness has been reduced by micro-EDM. The surface roughness was achieved within the recommended values of Rz and Ra of 3.4 and 0.4 µm, respectively, for biological studies such as implant abutment. 127
Carbon-enriched highly rough surface at the macro-scale on the Ti-6Al-4V alloy can also be fabricated by the EDM process using high peak current. The graphite electrode with high peak current of 21–79 A was used for rough machining. This makes chemical changes to surface of titanium, and this form of imposing macro-roughness (15.6 µm) has a favorable and a better surface impact on the adhesion and proliferation of human osteoblast-like MG-63 cells. 122 An effort of the combined effect of CM and SP with EDM has been covered to melt off the surface roughness that is achieved by EDM process.123,124 Micro- and sub-micro-surface roughness and structure are favorable for orthopedic applications. Surface roughness of samples evaluated by combining (EDM + SP + etching) process was 2.7 µm. The nano-porous biocompatible surface with roughness value 184 nm has been fabricated on titanium alloy by EDM by Peng et al. The surface of nano-scale features has a favorable impact on osteoblastic responses. 118
Surface chemistry and topology
The formation of nano-porous biocompatible layer on titanium by EDM was studied by Peng et al. 117 It discussed that how EDM generates an α-phase and a γ-TiH-(γ-hydride)-phase on the recast layer by electrical discharging. Among the EDM parameters, the most important parameters are electric current density and pulse duration in the fabrication of biocompatible nano-porous surface. Nano-(δ + γ) hydrides play important role in the formation of nano-porous oxide layers. A γ-hydride microstructure was also formed on the recast layer following electrical discharging. Electric discharging for a short duration leads to the production of thick and nano-porous TiO2 bioactive layer on titanium. Figure 15 shows the SEM photographs of nano-porous TiO2 bioactive layer on titanium by EDM process at a scale of 200 nm. 117

SEM photograph of nano-porous TiO2 bioactive layer on titanium by EDM process.
Bin et al. explored the potential of EDM for surface modification of titanium alloy by electro-spark alloying (ESA). They investigated the ESA layers on Ti surface produced in air, nitrogen gas atmosphere and silicone oil as dielectric medium. 128 The thickness of ESA layer was found to be 10–20 µm. The layer obtained under silicone oil was a relatively smooth and dense surface with outstanding wear and corrosion resistance. Figure 16 shows the SEM photographs of ESA layer on Ti surface in air, N2 and silicone oil. The surface obtained in silicone oil during EDM machining was found to have good biocompatibility and was biologically active. The properties of Ti-6Al-4V alloy after EDM treatment have been investigated.

SEM of surface and cross section of ESA layer: (a and d) ESA in air, (b and e) ESA in N2 and (c and f) ESA in silicone oil.
Surface roughness, distinctive irregular features and dense oxide layer were observed on the Ti surface by W-EDM process, which makes surface active for the interaction of osteoblastic cells. 121 Graphite electrode with high peak current of 21–79 A was used for rough machining. This causes chemical changes to surface of titanium. Figure 17 shows the carbon-enriched highly rough macro-level surface layer after EDM process. 122 Micro- and sub-micro-surface roughness and structure are favorable for orthopedic applications (Figure 18). A very high content of carbon and oxygen can be found on the machined surface during EDM treatment that was transferred from hydrocarbon oil. The titanium oxide (TiO2) fabricated due to the interaction of Ti and O present in the etchant and SP medium. This is because, Ti is an element with high chemical affinity, and during machining process, it is oxidized and forms titanium oxide, so different compounds of titanium oxides and titanium carbides are created on machined surfaces. Increased content of titanium oxide may have a positive effect on the bioactivity of the surface. EDM and subsequent CM lead to a significant improvement of osteoblast proliferation.123,124
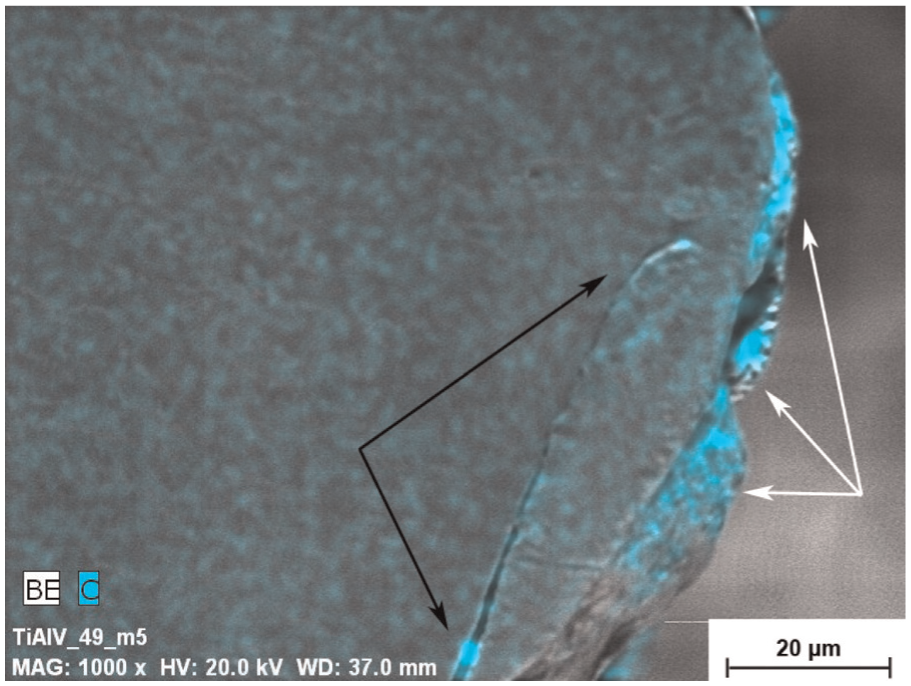
SEM image of carbon-enriched surface.

SEM images of the material surface after (a) EDM, (b) EDM + CM, (c) EDM + SP2 and (d) EDM + CM + SP2.
Enhancing the mechanical properties
Fatigue performance
The application of EDM for surface modification of biomaterials has been limited due to its poor fatigue performance. 129 It is a known fact that high surface roughness and surface micro-cracks after EDM processing decrease fatigue performance due to high notch sensitivity. This has been caused by tensile stresses in near heat-affected region, micro-cracks and high surface roughness. This performance can be further improved by suitable heat treatments. 130 An attempt has been made by Strasky and colleagues123,124 to improve the biological and fatigue performance. They have reported a combination of conventional machining techniques (SP and CM) followed by the EDM process to improve the poor fatigue strength of Ti-6Al-4V alloy. It was observed that EDM has least fatigue strength, and this performance would improve if the samples were prepared by a SP process followed by EDM process. Electro-polished standard samples have highest fatigue strength compared to EDM and EDM + SP. Furthermore, high fatigue strength was observed in combination of CM followed by SP and EDM. This proposed combination approach of EDM + SP + CM offers a potential surface for orthopedic applications. Klocke et al. reported that Ti-6Al-4V alloy treated by W-EDM offers higher fatigue performance than grounded. In 10%, 50% and 90% probability of failure, the W-EDM-treated samples offered higher fatigue limit stress than grounded samples. 131 The fatigue performance of Ti-6Al-4V alloy was studied for micro-specimens and compared with Inconel alloys. It was found that Ti alloys exhibit higher fatigue performance than Inconel alloys. 132 Mower 133 described in his study that EDM degrades the fatigue performance of the alloy by 15%–30% of milled samples. Intrinsic fatigue failure occurred due to crack initiation at interior location. The high surface roughness and thick recast layer were the major reason for the degradation of fatigue performance.
Enhancing micro-hardness, wear and corrosion resistance properties
Bin et al. investigated the deposition of TiC on Ti alloy by electrical discharge alloying graphite electrode. The surface micro-hardness was measured, and it was found that it improves by 3.5 times the initial. The high amount of induced hard TiC possessed excellent wear and corrosion resistance in SBF solution. 128
Summary of current research issues and future directions
The role of EDM process in recent investigations has demonstrated the potential to fabricate the biocompatible nano-porous surface on Ti and its alloy. The surface produced by EDM has superimposed topography that may offer a more suitable surface for osteoblast proliferation and improved implant fixation. EDM process modifies the Ti surface into various types of oxides and alloying elements. The primary aim is to produce biocompatible porous oxide structures for encouraging cell growth and adhesion. The corrosion and wear resistance are also improved using this technique. The present research trend shows that the oxide–carbide–porous layer formed on the titanium substrate by EDM process increases bioactivity. It is well defined from this research scenario that the trend of surface modification has been switched from conventional machining methods to advanced machining methods (laser, plasma, ion and EDM). The adaptation of bone tissues by Ti implants is a very critical and important issue. Surface modifications were often required for fixation and long-term stability of implant in particular biomedical application. EDM process can improve the surface properties and chemistry of biomaterial. EDM process has the potential to machine biocompatible materials, ranging from metals or alloys to ceramics, which have a wide range of application in dental, orthopedics, microdevices and so on. Table 3 describes the other studies carried out on the application of EDM process on Ti and its alloy in other areas.
Surface modification of Ti and its alloys by EDM for biomedical applications.
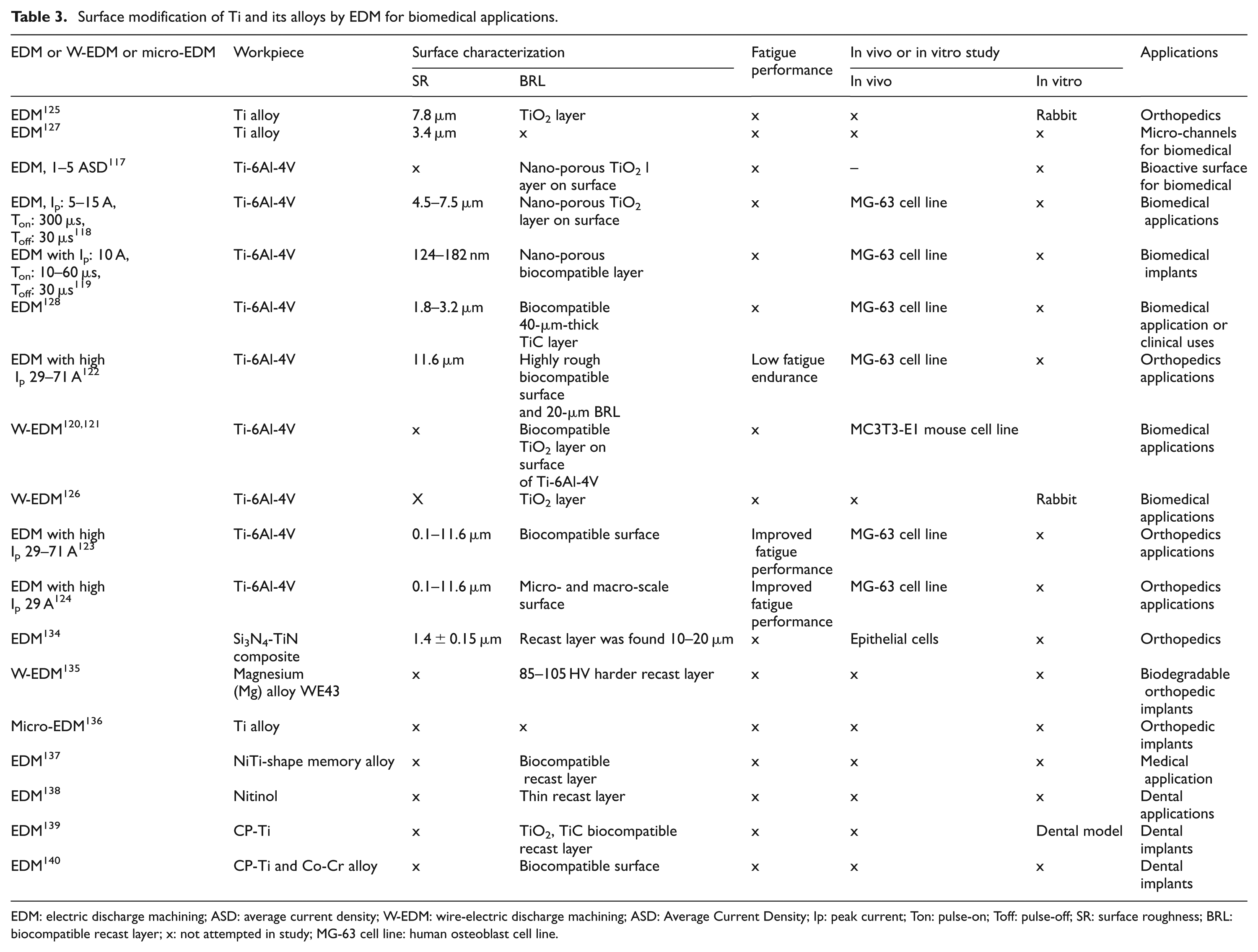
EDM: electric discharge machining; ASD: average current density; W-EDM: wire-electric discharge machining; ASD: Average Current Density; Ip: peak current; Ton: pulse-on; Toff: pulse-off; SR: surface roughness; BRL: biocompatible recast layer; x: not attempted in study; MG-63 cell line: human osteoblast cell line.
An innovative method for surface modification by material migration in EDM
To satisfy the need to generate bioactive implant surface, a new innovative technology is proposed here. This technology is well explored by Kansal and his team.100–102,141–144 This technology has not been employed before to generate the biocompatible surface for biomedical application. Before exploring its real potential, it has been presumed that PMEDM will also fulfill these objectives. A number of experiments have been carried on in parliamentary procedure to generate the biocompatible surface for biomedical application. The results of these explorations are coming up in the subsequent publications.
PMEDM
To enhance the performance of the EDM process, a relatively new advancement is the addition of conductive powder in the dielectric fluid of EDM oil. 143 This process is termed as PMEDM. PMEDM process as a surface modification technique has brought in a great deal of interest in recent years with regard to productivity and surface quality of workpiece. 141 Moreover, the surface developed by PMEDM process has high immunity to corrosion and abrasion. In PMEDM process, machining is performed in a separate tank of the main machining tank. This is to avoid the wastage of dielectric fluid mixed with different powders and at different concentrations. The new machining tank is made up of fiber glass and is transparent. The detailed experimental setup is adopted from Kansal et al. 143 The machining tank is filled up with dielectric fluid. For better circulation of powder mixed dielectric, a separate stirring system is integrated, which gently stirred dielectric and oil in the machining tank for assuring suitable uniformity of powder concentration in the dielectric fluid and decreasing the chance of particle settling at the tail end of the machining tank.
When a voltage of 80–320 V is applied between the electrode and the workpiece facing each other with a gap of 25–50 µm, an electric field in the range of 105–107 V/m is created. The powder particles become energized and behave in a zigzag fashion as depicted in Figure 19. Under the sparking area, the particles get close to each other and arrange themselves in the pattern of chain-like structures between the tool and the workpiece. The interlocking between the powders particles occurs in the direction of flow of current. The chain formation helps in bridging the discharge gap between the tool and the workpiece. Due to bridging effect, the insulating strength of the dielectric fluid decreases. The easy short circuit takes place, which causes early explosion in the gap. As a consequence, a series of discharges start under the electrode area. The faster sparking within a discharge takes place, which causes faster erosion of the workpiece surface and hence increases the erosion rate. At the same time, the added powder modifies the plasma channel. The plasma channel becomes enlarged and widened. The sparking is uniformly distributed among the powder particles, and hence, the electric density of the spark decreases. Consequently, uniform erosion (shallow craters) occurs on the workpiece surface. 141
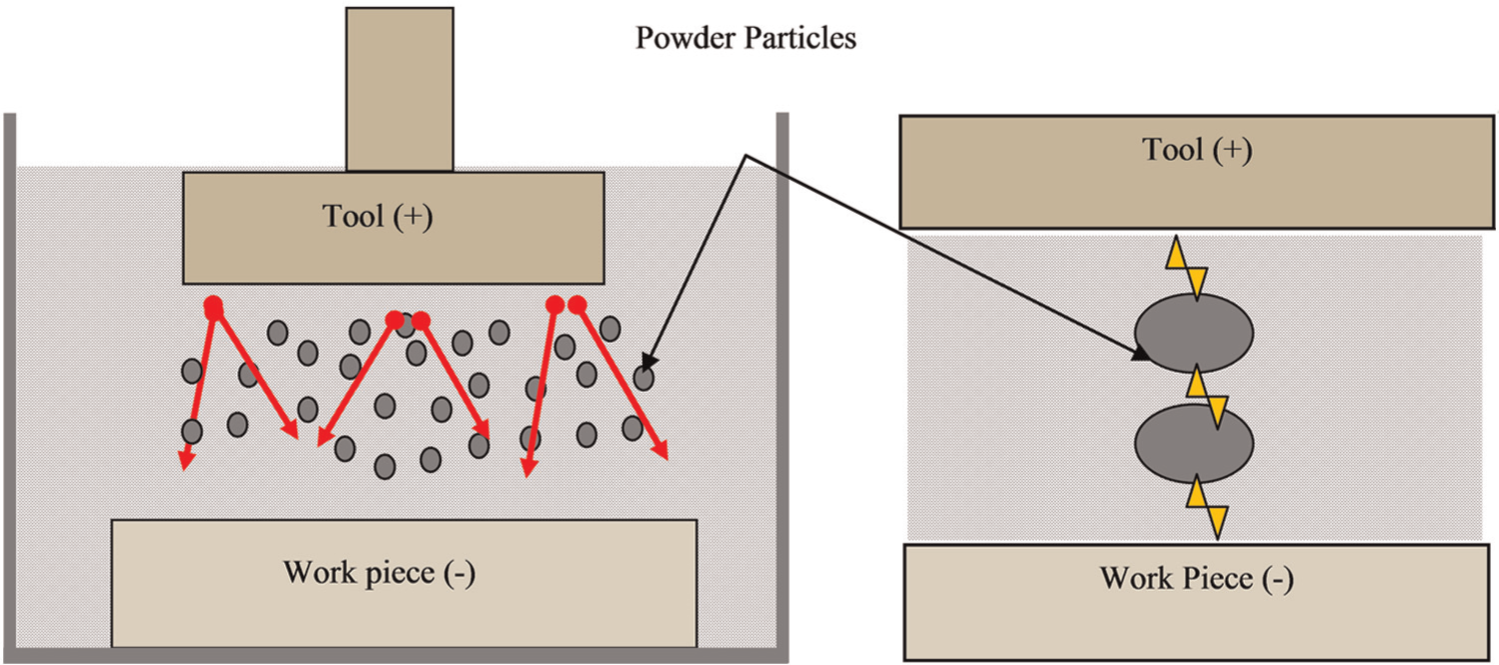
Schematic representation of mechanism of PMEDM process.
PMEDM process not only improves the surface finish but also allows the alloying of oxides and carbides in recast layer. During PMEDM process, at high concentration of the powder particles, the suspended powder particles get energized and form a chain-like structure between the tool and the workpiece. As a result, the sparking will take place intermediately between conductive powder particles as seen in Figure 19. Enormous amount of discharge energy is generated which melts the powder particles partially, and the material is removed from the workpiece surface in the form of debris. At the same time, the dielectric breaks down as a result of carbon and oxygen decomposed from it and react (alloyed) with the partially melted powder particles and the micro-droplets of molten metal to form oxides and carbides. Due to high concentration of powder, the eroded debris cannot be flushed out and is entrapped between the discharge gaps and gets migrated toward the workpiece surface and penetrate into the molten pool before solidification. 103 Followed by secondary discharges, these oxides and carbides get accelerated and induce a negative pressure by means of electrophoresis and are transferred toward the workpiece surface and penetrate the molten pool before solidification. The alloyed layer of oxides and carbides in the form of micro-droplets solidify at the top of the machined workpiece surface like a hemisphere. When these micro-droplets come under the contact of surface, its surface solidifies immediately, and its center part solidifies less rapidly. Advantageously, at the same time, enormous amount of absorbed gas is ejected, which leads to the creation of porosity and foamy-like shape onto the top surface as reported. 144 The migrated layer of alloy reduces the formation of micro-cracks on the workpiece surface and improves the mechanical properties. 145 The principle mechanism of material migration during PMEDM process is shown in Figure 20. The schematic representation of the material migration mechanism in PMEDM process takes place by the following steps: step (i) plasma channel of heat flux between powder particle and workpiece due to sparking; step (ii) after sparking, gathering of hydrocarbon (C) and oxygen molecule (O) between partially melted powder particles and micro-droplets of molten metal; step (iii) formation of carbides (XC, MC) and oxides (MO2, XO2) due to the presence of hydrocarbon (C), oxygen molecule (O) and powder particles (X) and step (iv) penetration of oxides and carbide in the molten pool of recast layer before solidification on the workpiece surface in PMEDM process. The previous results revealed that the high concentration of suspended powder, carbon (hydrocarbon) and oxygen (oxide) that decompose from dielectric and penetrate into the molten pool before solidification is substituted in the form of oxides and carbides on the workpiece surface. Furutani et al. 114 investigated the formation of titanium carbide (TiC) by PMEDM process and Janmanee and Muttamara 145 investigated the fabrication of very hard layer of tungsten carbide (WC) by electrical discharge coating (EDC) using titanium powder additives. The migrated layer of alloy reduces the formation of micro-cracks on the workpiece surface and improves the mechanical properties. This phenomenon is called as material migration from suspended powder particles in a dielectric fluid of EDM. Many researchers have investigated the modification of machined surfaces of material migration method. Wang et al. 146 reported the fabrication of 20-µm-thick compact layer of Ti ceramic using a Ti powder green compact electrode by EDM. TiN hard layer was obtained on the surface of titanium when urea solution in water is used as dielectric fluid. 147
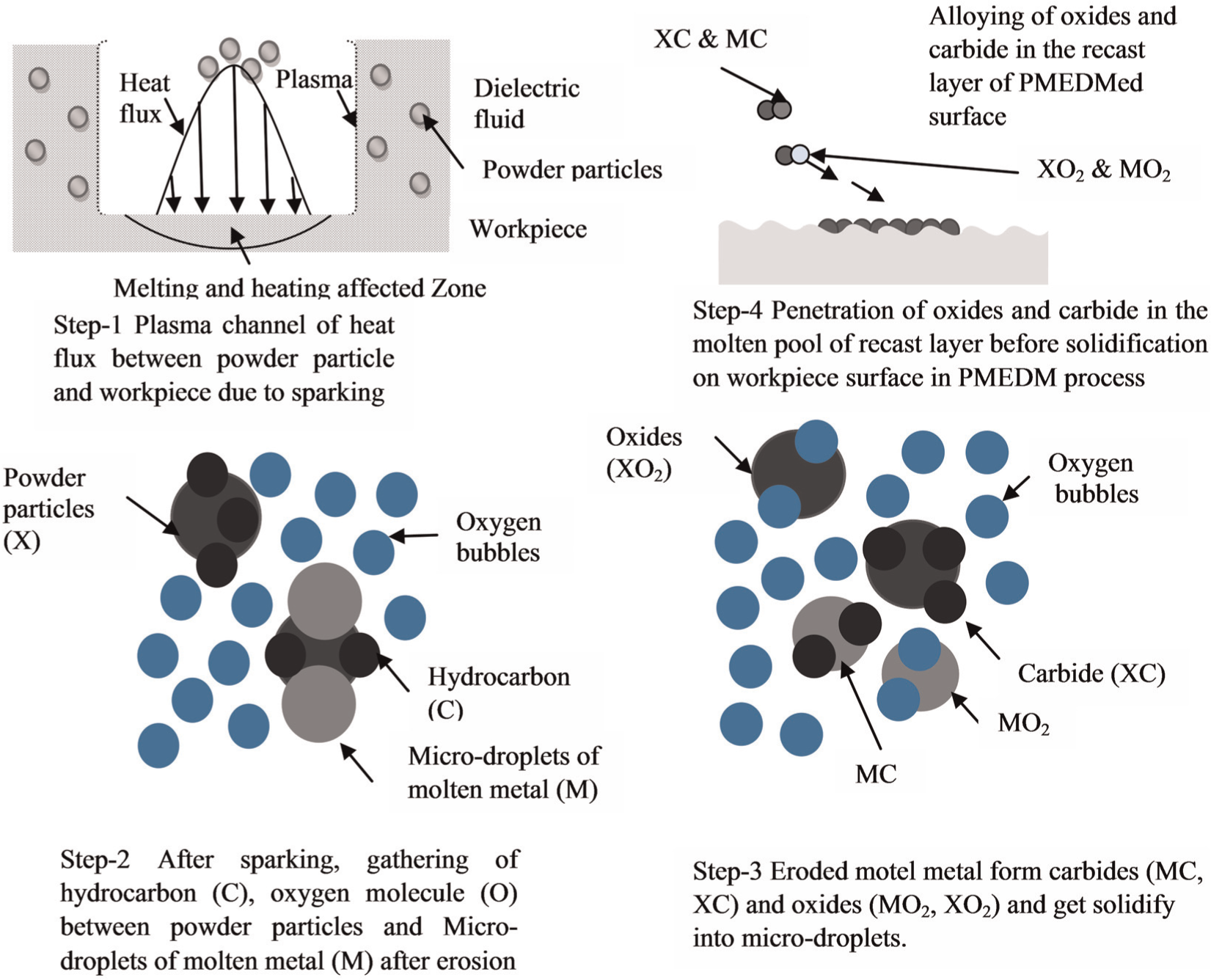
Schematic representation of mechanism of surface modification by material migration in PMEDM process.
From the literature survey of surface modification, it is obvious that material migration from suspended powder to workpiece surface is possible, and workpiece surface can be amended in various ways. The surface obtained from PMEDM process has carbide phases, and this increase in the percentage of carbon along the machined surface indicates that suspended powder particles react with products of hydrocarbon dielectric fluid (oxygen and carbon) after a dielectric breakdown at high temperatures of the plasma channel to form carbides. This surface alloying significantly enhances the surface micro-hardness. Moreover, surface hardness has a direct bearing on abrasion resistance, which might serve to improve the surface load-bearing capacity and results in outstanding wear resistance. As discussed in an earlier section, the requirements of biomedical implant surface are controlled surface roughness, surface topography, erosion and wear resistant. The studies indicate that PMEDM technique has potential to enhance the mechanical as well as biocompatibility of bio-implants. The experimental results of this process are clearly discussed in the upcoming publications.
Conclusions
The EDM has potential in treatment and modification of Ti alloy and other metallic materials used for biomedical applications. In comparison to the several existing techniques such as CVD, PVD and anodization, EDM shows better potential for surface treatment of metallic implants. The EDM has been more advantageously employed than other surface modification techniques. These are (a) no surface preparation before EDM; (b) converts the machined surface into oxide and carbide layer of controlled thickness, which increases the biocompatibility and generates a hydrophilic surface; (c) ability to produce nano-porous surface; (d) improves the surface hardness of material and (e) improves the wear and corrosion resistance of the machined surface. The only disadvantage is that the fatigue performance of the highly rough EDMed sample is low, which can be further improved by surface treatment method or by the reduction of recast layer by shot blasting, CM, SP and so on. However, the use of EDM process in the medical arena is still in its early life. The rapidly growing field of biomanufacturing faces significant challenges and opportunities. The role of EDM process for surface modification of biomaterials is still at the experimental stage. A few number of research studies have been reported by taking EDM as surface treatment or modification process for biomaterials; superior biocompatible surface characteristics have been reported, and capability of formation of biocompatible nano-porous surface has been reported. Despite this, there is a gap and the urgent need to study the effect of various input-independent parameters of EDM on the formation of biocompatible surface. Yet, many more issues need to be addressed before the method can be embraced by the biomedical industry.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This work was financially supported by the Department of Science and Technology (DST), Government of India under SERB project with reference number SR/S3/MERC/0028/2012.
