Abstract
Ceramic green bodies fabricated by stereolithography-based three-dimensional printing technology often have high loading of ultraviolet curable resins, which produce undesirable phenomena such as cracking, deformation, and blistering during the debinding process. Results showed that compared to argon and vacuum, air atmosphere provided higher flexural strength owing to the elevated density. The differences in microstructure between specimens prepared under these atmospheres were attributed to exothermic oxidation reactions occurred under air when compared to endothermic pyrolysis reactions under vacuum and argon. The debinding atmosphere showed little effect on crystallite size due to the elevated sintering temperature, which would determine the final crystallite size of Al2O3. Debinding under air atmosphere resulted in flexural strength of 176.69 MPa and open porosity of 23.4%. The flexural strength of the ceramics debinded in air was 21.6% higher than the ceramics debinded in argon atmosphere.
Introduction
Three-dimensional (3D) printing, also known as additive manufacturing, is a technique that builds objects through layer-by-layer mode based on digital model files, such as powder metal, plastic, or other sticky materials.1–3 3D printing technology is suitable to fabricate parts with complex structures that are difficult to be made by traditional methods.4–6 In addition, it possessed the feature of high efficiency and low cost.7–10 Stereolithography (SL) was first patented in 1986 by Chuck Hull, and relied on layer-by-layer curing of liquid photopolymer resins using ultraviolet (UV) laser. 11 The 3D printing technology based on SL is a high precision rapid prototyping technology. 12 Currently, 3D printing technology gained increasing interest among industry and academic fields and was called “the third industrial revolution.”13–15 Many recent research works have promoted the development of 3D printing technology.16–18
The rapid development of 3D printing technology for metal and resin materials also promoted the development of 3D printing technology for ceramics. 19 Alumina-based ceramics are very stable materials with excellent resistance to acid, alkali, and high temperature.20–22 They can be used in artificial bones, crucibles, cutting tools, abrasives, catalyst carriers, and sealing materials, and so on.23–25 The traditional production methods of alumina ceramics mainly include various moldings based on rotational, extrusion, spray drying, oil ammonia column, hot oil column, and water column. 26 However, these routes are often very difficult to produce ceramics with complex shapes and fine structures. 3D printing technology can overcome these problems, as well as facilitates ceramic part processing and shortens long preparation cycles. Consequently, tremendous efforts have been devoted to fabricating ceramics by SL technology. For instance, Hu et al. 27 designed a light-curing forming system for high solid content ceramic suspensions to overcome problems linked to high viscosity suspension and non-uniform green bodies. Zhang et al. 28 used short-chain dicarboxylic acid to modify alumina particles and optimize their rheological properties to meet the requirements of UV curing molding. Adake et al. 29 modified alumina with stearic acid and oleic acid to better disperse alumina in monomer with hydrophobic backbone. Johansson et al. 30 studied the effect of surfactant on dispersion of alumina in photopolymerizable monomers by SL.
However, many technical problems of SL technology in ceramics still exist and require solutions. 31 To this end, a large number of resins are added into ceramic slurries, which are subsequently removed from the green bodies during the debinding process. During debinding process, the volatilization of organic compounds can easily lead to the shrinkage, deformation, and surface cracking of the ceramics. Hence, the thermal treatment process of 3D printing ceramic materials has been extensively studied, in which debinding determines the quality of obtained ceramics. 32 Housaer et al. 33 studied the influence of debinding on the properties of carbon nanotubes reinforced aluminum matrix composites at 400 °C for 5 min which provide a guidance for SL debinding technology. Shang et al. 34 studied the gel-tape-casting of aluminum nitride ceramics and noticed that all organic additives were burned during debinding at temperatures as high as 500 °C. Wu et al. 35 concluded that both bimodal particle size distribution and vacuum debinding would provide an effective route for 3D printing of ceramics. In addition, specimens subjected to vacuum debinding showed higher density than those treated by traditional thermal debinding. He et al. 36 fabricated complex and dense zirconia ceramics, and the sintered ceramics displayed significant shrinkage of 35.26%. Wu et al. 37 prepared Al2O3–ZrO2 ceramics without defects by application of a two-step debinding, and concluded the relationship between sintering temperature and relative density. The sintered ceramics possessed a maximum density of 4.28 g/cm3 when sintered at 1600 °C. Doreau et al. 38 noticed similar flexural strength of alumina ceramics prepared by SL as that of alumina ceramics prepared by traditional pressing techniques. Corbel et al. 39 fabricated alumina ceramic parts by SL technology and then debinded the samples at 500 °C. They found that the optimal debinding temperature for polymers can be determined by thermogravimetric (TG) analysis. Liu et al. 40 employed SL technology to fabricate Si-O-C ceramics followed by thermal treatment at 1000 °C to obtain compact non-porous materials. Eckel et al. 41 reported polymer-derived ceramics with uniform shrinkage and no porosity through SL-based 3D printing technology. Zhao et al. 42 used SL technology to produce full-ceramic crowns after debinding at 600 °C and sintering at 300 °C to yield crack-free and uniform shrinkage denture model. However, SL-based 3D printing technology for alumina ceramics still faces some problems, such as significant shrinkage and low flexural strength. These issues are mainly determined by the debinding process, and would affect the mechanical properties of the ceramic parts. However, whether the debinding atmosphere improves the mechanical properties needs further investigation.
In this article, the effects of debinding atmosphere on the mechanical properties of alumina ceramics were investigated. The thermal stability of the obtained green bodies and different chemical reactions occurred under each atmosphere were analyzed by thermogravimetry. The microstructures and phase compositions were analyzed by scanning electron microscopy (SEM) and X-ray diffraction (XRD). The influences of debinding atmosphere on the mechanical properties of alumina ceramics were investigated by analyzing the crystallite size, relative density, porosity, shrinkage, and flexural strength of the sintered ceramics.
Materials and experiments
Preparation of Al2O3 ceramic slurries
The alumina ceramic slurries were consisted of alumina powder and photosensitive resin. 27 The alumina powder (Henan Hecheng Inorganic New Material Co., Ltd., China) consists of two kinds of particles of different sizes, which were mainly 0.1 and 0.8 μm. 43 Al2O3 powder was first dried at 200 °C for 5 h, and then used to prepare the ceramic slurries. The photosensitive resin (Beijing Ten Dimensions Technology Co., Ltd., China) consisted of acrylic monomer, diluent, photoinitiator, and absorbent. Afterward, Al2O3 powder (495 g) was slowly added to 100 g photosensitive resin. After stirring, the slurry was milled for 2 h with planetary mill at 400 r/min and vacuum defoamed for 10 min to yield ceramic slurries.
Fabrication of Al2O3 ceramic green bodies
The model (50 mm × 4 mm × 3 mm) was first drawn by the Unigraphics NX software and then imported into STL format. Next, the file was transferred to the 3D printer and alumina ceramic slurry was introduced into the resin tank of the 3D printer (AutoceraM, Beijing Ten Dimensions Technology Co., Ltd., China). The irradiation source and the wavelength were LED light source and 405 nm, respectively. The irradiation dose was 10 mW/cm2, the exposure time of single layer was 10 s, and the thickness of printed green bodies was 100 μm. After the 3D printing process, the green bodies were washed with ethanol.
Debinding process
The green bodies were debinded in a tubular furnace (Hefei Ke Jing Materials Technology Co., Ltd., China). They were heated to 80 °C with a heating rate of 5 °C/min, and sustained for 2 h. Then heated to 180 °C with a heating rate of 1 °C/min, and sustained for 2 h. Finally, heated to 450 °C with a heating rate of 0.25 °C/min, and sustained for 2 h. The debinding profile is shown in Figure 1.
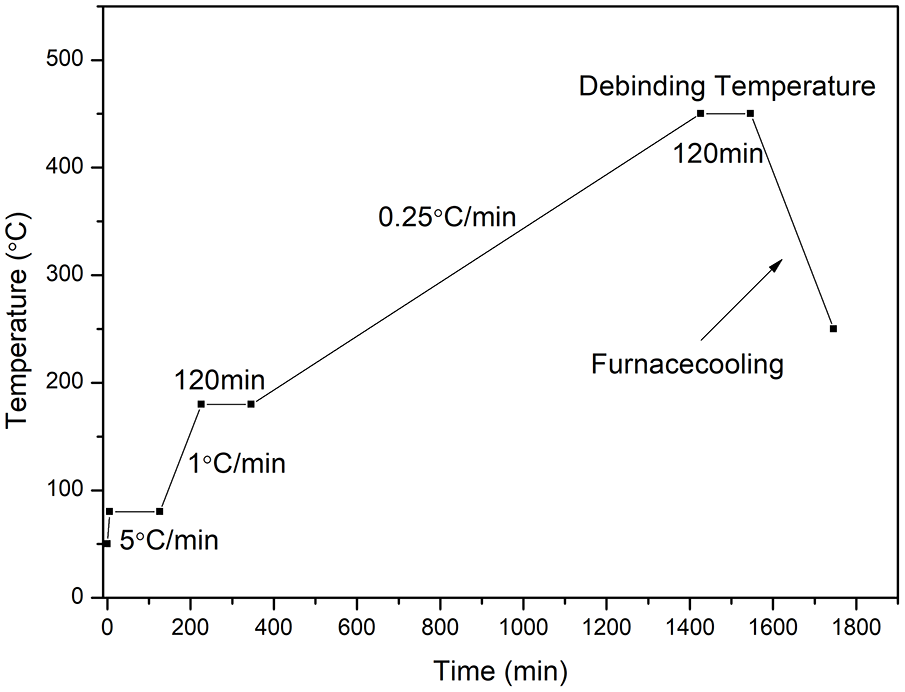
Debinding profile of the green bodies.
The debinding process was carried out in argon, air, and vacuum. The green bodies were denoted as Q(GB). The specimens debinded under argon, air and vacuum were denoted as “Q(Ar),”“Q(Air),” and “Q(Vacuum),” respectively.
Sintering process
After debinding, the specimens were sintered in a KSL-1700X high-temperature muffle furnace (Hefei Ke Jing Materials Technology Co., Ltd.) at 450 °C with a heating rate of 5 °C/min, and sustained for 20 min. Then heated to 650 °C with a heating rate of 5 °C/min, and sustained for 2 h. Then heated to 850 °C with a heating rate of 5 °C/min, and sustained for 2 h. Finally, heated to 1500 °C with a heating rate of 5 °C/min, and sustained for 2 h. The sintering profile are depicted in Figure 2.
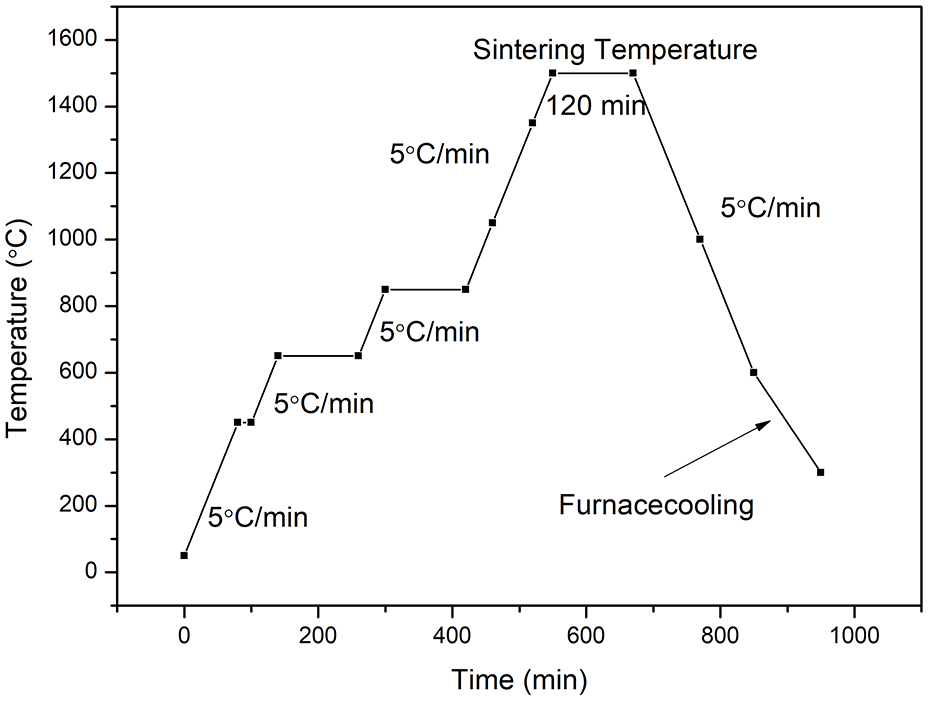
Sintering profile of the obtained specimens.
Characterization
The TG analyses (449F5 simultaneous TG–differential scanning calorimetry (DSC) apparatus, Netzsch, Germany) for the green bodies were carried out under flowing air or argon with a heating rate of 10 °C/min from 50 °C to 600 °C. The 449F5 simultaneous TG–DSC apparatus possessed a high vacuum closed system and the test temperature can reach to 1500 °C.
Powder XRD with Cu Kα radiation was performed on a Bruker D8 Focus (Bruker Corporation, Germany) X-ray diffractometer at diffraction angles ranging from 10° to 90°, voltage of 40 kV, current of 30 mA, and scanning speed of 0.02°s−1 per step. 44 The X-ray diffractometer possessed 1 kW X-ray source and one-dimensional (1D) detector. The crystallite size of Al2O3 was calculated from the peak at 2θ = 35.3° following the Scherrer equation
where d is the average crystallite size of Al2O3 (nm), K is the Scherrer constant (0.89 in the present case), λ is the wave length of X-ray (0.154056 nm), and B is the peak width at half height of Al2O3.
SEM images were obtained using the Helios G4 CX (America) at working voltage of 5.0 kV. The Helios G4 CX possessed accelerating voltage of 200 V–30 kV for electronic gun and beam intensity of 0.8 pA–100 nA for electron beam.
The densities of sintered specimens were measured by the Archimedes method. 45 The accuracy of the balance was set to 0.0001 g (Mettler Toledo, Switzerland). The shrinkage of the sintered samples was measured with the following formula
where
The flexural strength 46 of the sintered specimens was measured by three-point flexural method on an electronic universal testing machine (CMT4304, SUNS, China) at loading speed of 0.5 mm/min and span of 30 mm following equation (3)
where
Results and discussion
TG analysis and microstructure
Figure 3 shows the TG profiles of the green bodies. An endothermic reaction was observed for the green body heated under argon atmosphere (Figure 3(a)). By comparison, an exothermic process occurred for the green body heated under air (Figure 3(b)). In Figure 3(a), the mass loss was only estimated to 1.5% in the temperature range of 50 °C–350 °C (Figure 3(a)). When debinded in the temperature range of 350 °C–550 °C, the change in mass loss was estimated to 15.1%, which was almost 10 times higher than the mass loss in temperature range of 50 °C–350 °C. Also, decomposition of the green body occurred mainly in the range of 350 °C–550 °C, where endothermic reaction took place with DSC peak at 399 °C. Therefore, the heating rate should be slowed down to prevent the formation of cracks in the green body during debinding. At temperatures above 550 °C, no mass loss was noticed and only alumina remained. The DSC curve also showed a weak peak at 98 °C, corresponding to slight mass loss due to evaporation of residual water in the green body. The TG–DSC profile obtained here looked similar to that reported by Xue et al. 47 for Al2O3 ceramics. However, the decomposition temperature was about 100 °C lower than their results. Hence, resin composition was the main factor determining the decomposition temperatures of green bodies. The TG analysis of the green bodies indicating that the atmosphere during debinding would affect the chemical reaction, subsequently have a significant effect on the physical and mechanical properties of the ceramics. The heating rate was rather slow during the debinding process to avoid violent chemical reactions and guarantee the quality of the samples after the debinding process.
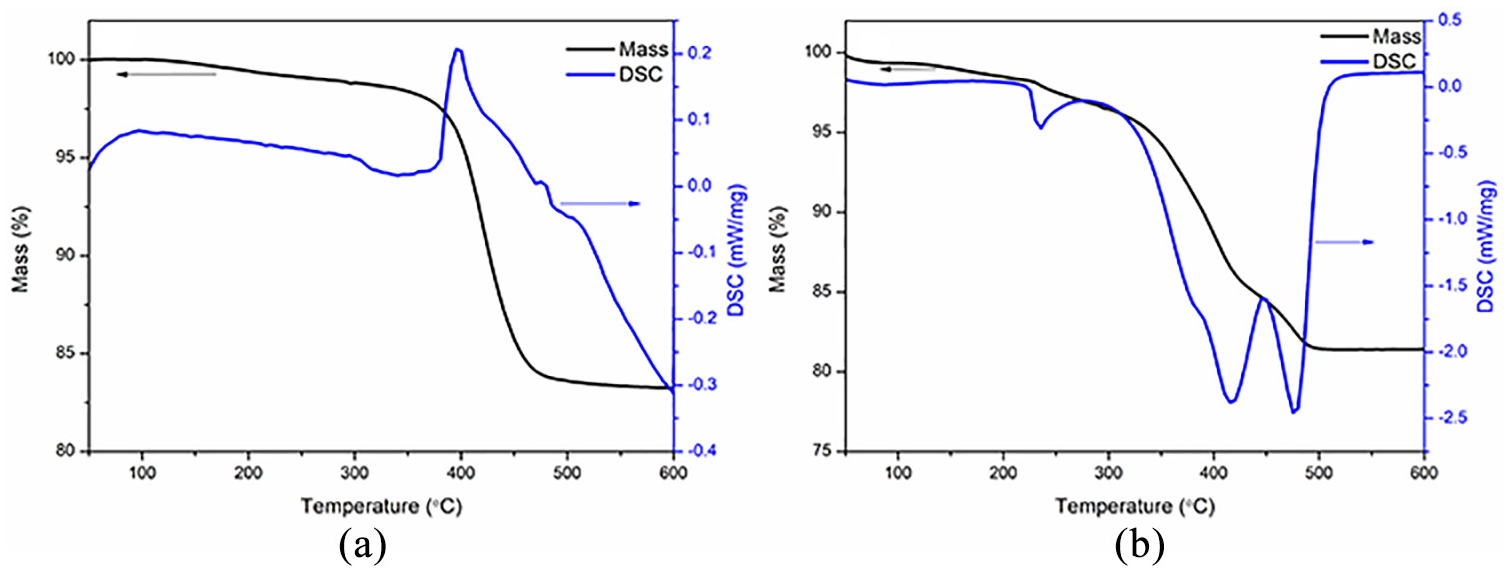
TG–DSC curves of the green body: (a) under argon and (b) under air.
Figure 4 shows the SEM morphologies of the obtained green bodies, and obvious differences between specimens were observed. The green bodies were composed of cured photosensitive resin and alumina powder. The samples debinded under argon atmosphere exhibited the loose and porous structure (Figure 4(b)), leading to irregular granular structures due to pyrolysis of the resins after thermal treatment. Some pores were still present on the surface because of the relatively low debinding temperature. The microstructure of the samples debinded under air changed the porous structure into larger block morphology (Figure 4(c)). In addition, numerous small particles were uniformly distributed over the large blocks. The morphology of debinding in air was obviously different from that of other specimens. The morphological characteristics under vacuum debinding appeared similar to those under argon debinding but not identical. Furthermore, irregularly shaped particles were randomly distributed (Figure 4(d)). However, the stacking of these particles was denser and particle size was smaller when compared to those obtained after debinding under argon. The samples debinded in argon and vacuum atmosphere exhibited a similar microstructure, which is obviously different with the samples debinded in air atmosphere. This phenomenon is mainly due to that the chemical reactions were different in various atmospheres. When the samples were debinded in argon and vacuum atmospheres, they belong to endothermic reaction. But when the samples debinded in air atmosphere, they belong to exothermic reaction.

Microstructures of the green body and specimens debinded in different atmospheres: (a) green body, (b) argon, (c) air, and (d) vacuum.
Phase composition and physical properties
Figure 5 shows the XRD results of the prepared samples. The chemical composition of sintered specimens looked the same, consisting of α-alumina. The 25.6°, 35.1°, 37.8°, 41.7°, 43.4°, 46.2°, 52.6°, 57.5°, 59.8°, 61.2°, 66.5°, 68.2°, 70.4°, 74.3°, 77.2°, 80.7°, 84.4°, and 86.4° were attributed to peaks of α-alumina of (012), (104), (110), (006), (113), (202), (024), (116), (211), (122), (214), (300), (125), (208), (119), (220), (223), and (312) crystallographic planes, respectively, (PDF#65-3103).48–50 After the debinding and sintering processes, the phase composition was almost the same since the peak position of different specimens looked identical. Hence, specimens treated under different atmospheres would have the same crystal faces. Although the debinding atmosphere was different, the phase compositions all contained α-alumina as the most alumina stable phase. No phase changed could occurred when debinded in different atmospheres, since α-Al2O3 is the most stable phase among various crystal forms in alumina. Therefore, Figure 5 exhibited the identical phase composition for different samples.
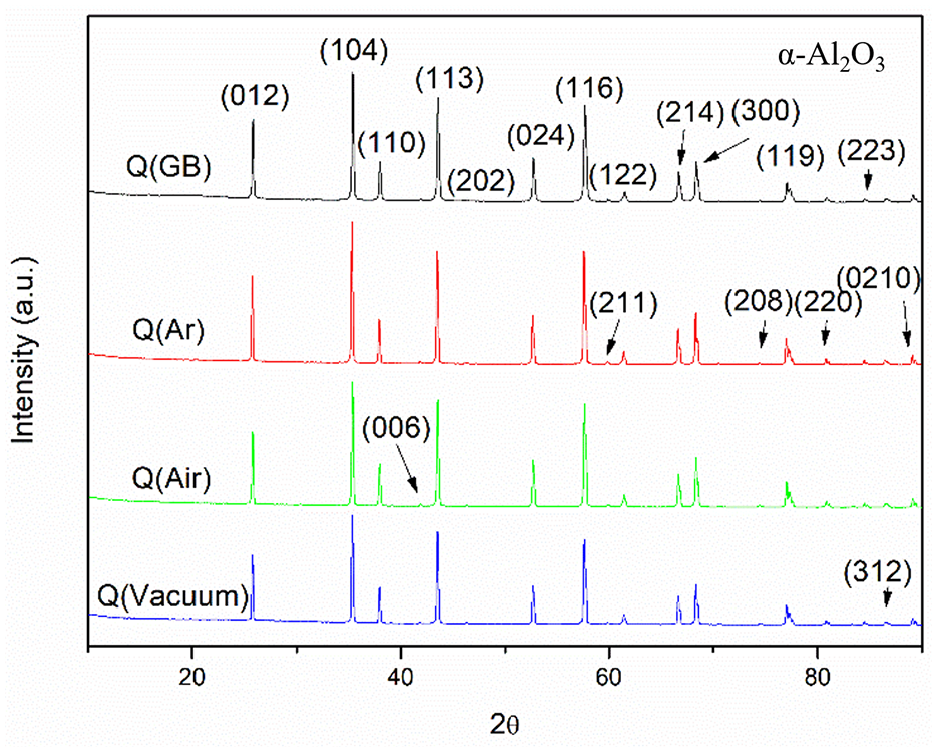
XRD patterns of the green body and specimens debinded under different atmospheres.
The crystallite size of α-alumina was calculated by the Scherrer equation and the results are shown in Table 1. The smallest crystallite size was determined as 37.46 nm. After sintering, the crystallite size of all specimens slightly changed to 43.37 nm, meaning preservation of crystallite size after sintering. This may be due to the fact that crystals were grown at high temperatures, and then affected the final product. Krell et al. 51 noticed the primary crystallite size of α-Al2O3 as small as 45 nm, which was very close to our results. Morin et al. 52 measured the crystallite size of α-Al2O3 as 75 nm, which was much larger than ours. These differences may be due to variation in thermal treatment procedures.
Crystallite size of the samples.
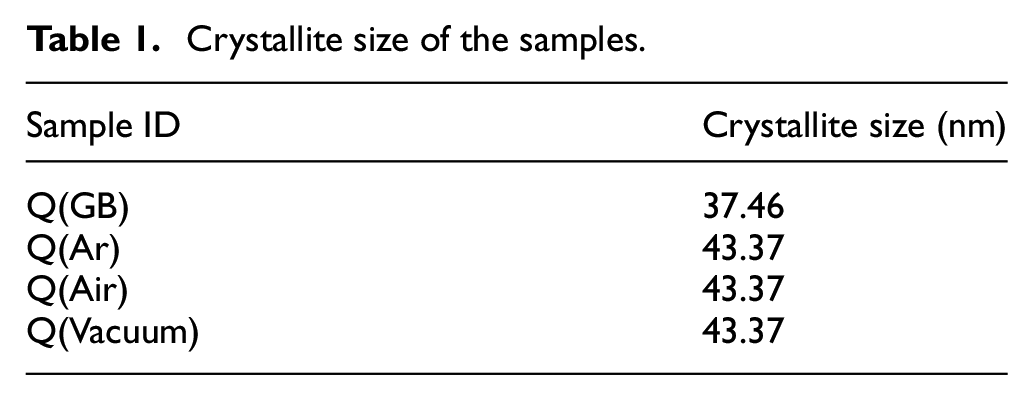
Table 2 shows the density, relative density, and open porosity of each specimen debinded in different atmospheres. The density of the original green body was determined as 2.83 g/cm3 and its relative density was 71.66%. After the debinding and sintering processes, the density and relative density of the ceramics increased due to high-temperature treatment. Debinding under air raised the density to maximum of 3.01 g/cm3 and relative density to 76.15%. Under argon atmosphere, the density and the relative density were slightly lower than in air atmosphere, determined as 3.00 g/cm3 and 76.03%, respectively. However, the density substantially declined under vacuum to yield only 2.78 g/cm3, which was slightly lower than that of the original green body. The relative density was 70.37%, and specimens with more pores led to lower densities, consistent with SEM data (Figure 4). Debinding under air will quickly evaporate the organic matter present in the green body, beneficial to orderly growth of crystallite size during debinding and sintering. In vacuum debinding, the internal structure rich in pores and relatively smaller crystallite size lead to low densities. The volatilization of the resin from the green bodies formed more pores in the sintered ceramics, with resulted in the relative densities lower than those of alumina obtained by traditional preparation method. 53 According to Maleksaeedi et al., 54 the porosity of the 3D printed alumina ceramics was between 20% and 50%, which was consistent with the results shown in Table 2.
Density, relative density, and open porosity of the specimens.
Mechanical properties
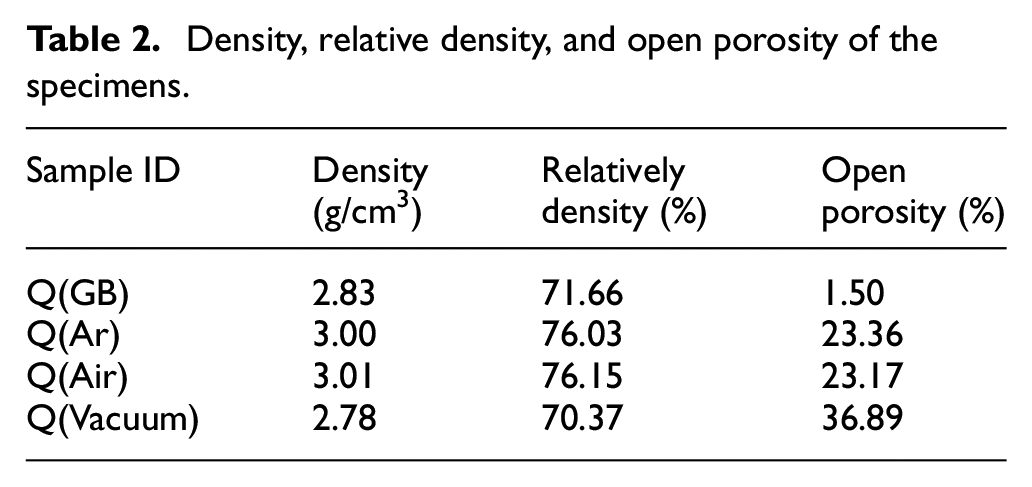
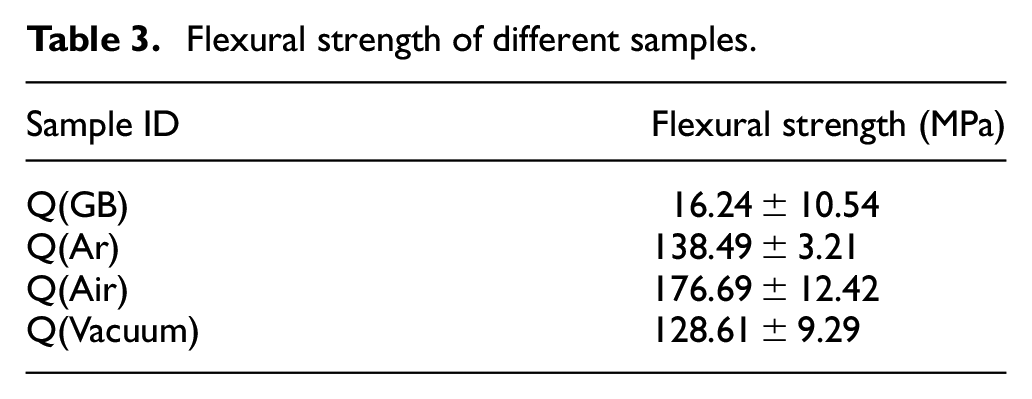
The flexural strength of the sintered ceramics is shown in Table 3. The flexural strength changed significantly with the debinding atmosphere. The flexural strength of the green body was determined as 16.24 MPa, representing the minimum value among these specimens. The undecomposed organic matter present in the green body weakened the binding strength between alumina powders and lowered the flexural strength. When debinding in air, the flexural strength of the sintered ceramics raised to a maximum value of 176.69 MPa, which was much higher than the values of other three specimens. Debinding in argon or vacuum resulted in flexural strengths of 138.49 and 128.61 MPa, respectively. After high-temperature sintering, the interaction forces between alumina powders became strong, and the flexural strength of sintered ceramics was higher than that of green body. Hence, there was a corresponding relationship between density and flexural strength. The flexural strengths of sintered ceramics were much lower than values obtained by Halloran 55 (85% for aluminum ceramics at 580 MPa). This is mainly due to the different sintering procedures. The different chemical reactions occurred during the debinding process lead to the various microstructures of the debinded samples, and then resulted in the discrepancy in flexural strength.
Flexural strength of different samples.
The cross-sectional morphologies of the sintered ceramics debinded under different atmospheres are shown in Figure 6. These images were obtained by testing the flexural strength of fractured sections. The microstructures and surface morphologies of specimens sintered under different atmospheres looked very different. After the sintering process, the debinded specimens under argon atmosphere were formed by compact accumulation of crystal particles with sharp edges and uneven sizes. Under the high-temperature sintering process, the particles of the irregular shape transformed into regular crystal structure. And some pores were formed between the particles due to the volatilization of cured photosensitive resin. At the same time, the size of the particle became uniform. When debinded in vacuum atmosphere, the morphologies of the sintered specimens contained many smaller crystal particles, which accumulated tightly around the larger crystal particles. Hence, the accumulation process would form many holes, which could lead to reduced flexural strength of the sintered ceramics. The microstructure of the sintered ceramics exhibited different appearance mainly due to the different chemical reactions occurred when debinded in various atmospheres.

Microstructures of the green body and ceramics debinded in different atmospheres: (a) green body, (b) argon, (c) air, and (d) vacuum.
The shrinking percentage data are shown in Supplemental Figure S1. After completion of debinding, the shrinking percentage became very small and ranged between 0.6% and 1.1% with insignificant differences. After sintering, the shrinking percentage increased to reach almost sixfold the values of debinded specimens (between 6.3% and 6.7%). Although the shrinking percentage after debinding was slightly different, the shrinkage after sintering remained almost the same. The debinding in different atmospheres induced different shrinkages due to changes in chemical reactions. After sintering, the alumina crystal particles were grown gradually and connect together through strong interaction forces during the sintering process, which lead to the similar shrinkage of the sintered ceramics.56–58
Conclusion
Alumina ceramic green bodies were fabricated by SL process then subjected to low-temperature debinding and high-temperature sintering. The influence of different atmospheres (argon, air, and vacuum) during the debinding process on microstructure and the mechanical properties were studied as follows:
The maximum density reached 3.01 g/cm3 when debinded in air and the minimum density of 2.78 g/cm3 when debinded in vacuum. The chemical reactions during the debinding process changed according to various atmospheres.
Air atmosphere leads to higher flexural strengths and relative densities while vacuum atmosphere leads to lower flexural strengths and relative densities due to the different chemical reactions occurred during the debinding process. The exothermic reactions took place in air and the endothermic reactions took place under argon.
The flexural strength was the highest when debinded in air. Using the same thermal treatment procedure, air atmosphere leads to elevated flexural strength and relative densities due to different chemical reactions. The flexural strength of the samples debinded in air was 21.6% higher than the samples debinded in argon atmosphere.
How to control the debinding conditions to obtain desired alumina ceramics needs further research. The temperature, heating rate, and holding time during debinding process need detailed investigation to promote the development of 3D printing technology for ceramics.
Supplemental Material
supporting_information_20200426 – Supplemental material for Investigating the relation between debinding atmosphere and mechanical properties of stereolithography-based three-dimensional printed Al2O3 ceramic
Supplemental material, supporting_information_20200426 for Investigating the relation between debinding atmosphere and mechanical properties of stereolithography-based three-dimensional printed Al2O3 ceramic by He Li, Yongsheng Liu, Yansong Liu, Kehui Hu, Zhigang Lu and Jingjing Liang in Proceedings of the Institution of Mechanical Engineers, Part B: Journal of Engineering Manufacture
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Research and Development Program of China (no. 2018YFB1106600), the Chinese National Foundation for Natural Sciences under Contracts (no. 51672217), Innovation Foundation for Doctor Dissertation of Northwestern Polytechnical University (CX202006).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



