Abstract
Increasingly, nephrologists struggle with providing care to patients with complex diseases who are heading towards the end of life. In this vignette, we illustrate how to recognize and acknowledge disease progression, tailor treatments to frailty status, and extend high-quality kidney care through to the time of death. Questions answered include how to discuss prognosis while retaining feelings of hope, and tips on how to recognize that the end-of-life may be approaching are included as part of the case discussion. We advocate for modest changes to nephrology care guidelines that promote integration of both high clinical standards and holistic and practical kidney care.
Case vignette
John S is a 78-year-old retired high school teacher, who has a 20-year history of smoking-associated health issues. He struggled with smoking cessation, but with the help of a community group program, he has not smoked for the past 5 months. His past medical history includes myocardial infarction 14 years ago and further revascularization 6 years later; two transient ischemic attacks, both more than 2 years ago; fully optimized chronic obstructive airways disease; and significant heart failure with reduced ejection fraction (HFrEF). Over the last 3 years, he has been admitted 12 times for symptoms associated with HFrEF. His course has been complicated by progressively worsening cardiorenal disease over the past 11 months.
A left ventricular assist device (LVAD) was placed 8 months ago with good effect. However, John has recently been advised that he is no longer eligible for heart transplantation, and the LVAD is a long-term destination therapy. He has progressive kidney disease with a rising serum creatinine, despite an 8 kg muscle weight loss and increasing diuretic resistance. He is compliant with medications and treatments. Prior to the current admission, he was still participating in light household chores, but in the last couple of weeks, he has been experiencing severe dyspnea even at rest and swelling of his legs and scrotum, and he started sleeping in a reclining chair. On bedside examination now, he has improved respiration but still has pitting edema extending to his lower thighs. Although he has responded to intravenous diuretics, he continues to have edema and is increasingly fatigued, now being unable to sit unaided for more than 15 min a day. He is currently undergoing assessment for peritoneal dialysis.
Key points
A therapy that is primarily used to alleviate symptoms, but not modify the disease progression, can be considered a palliative intervention (palliare = to cloak).
Stratifying patients by clinical frailty status can allow treatment targets to be adapted to the patient's needs and likely outcomes. Frail patients may benefit from a frailty-informed clinical approach to care.
Prognosis is best conveyed using a continuum of conversations across time and personnel. Preference to terms such as months to years, weeks to months, and hours to days is given rather than timelines.
Patients, and families, often experience rhythmic variations in disease and well-being (“good days and bad days”). Helping patients, and families, recognize this rhythmic cadence can lead to more open discussions about the health trajectory.
Conversations about the expected changes around the time of death, such as difficulty breathing or somnolence, aid families in recognizing and accepting death as a terminal event.
Questions
What is the role of peritoneal dialysis in palliating symptoms of cardiorenal syndrome?
John has been increasingly experiencing symptoms of dyspnea at home despite high doses of diuretics. Hospitalization has been triggered when he becomes increasingly short of breath, when seated upright, at rest. His wife notes that at these times, he sleeps poorly and presents with irritability and drowsiness, as well as overt respiratory distress. When not experiencing dyspnea, he is able to participate in all self-care activities at home, and greatly values time spent supervising his 5-year-old granddaughter's home activities for 1–2 h each weekday. It is anticipated that ultrafiltration from peritoneal dialysis may improve symptoms, allowing him, at least for some time, to continue to color and read with his granddaughter.
Peritoneal dialysis (PD) is increasingly used to aid symptom management in individuals with HFrEF when diuretics are no longer effectively controlling symptoms. The advantages of PD over hemodialysis (HD) are hemodynamic stability, gentle removal of fluids over a longer period of time, and no need for bloodstream access. Several observational studies confirm that PD use is associated with improved symptoms. A pooled meta-analysis showed improvement of New York Health Assessment (NYHA) class, with the mean difference (MD) in scores showing a reduction of 1.37 (95% CI −0.78 to −1.96, p < 0.0001) in those undergoing PD. There was considerable variability between studies, as indicated by I2 statistics >90%, making interpretation somewhat limited, but in most studies, patients also had a reduction in hospitalization duration (MD in hospitalization duration −34.8 days/patient/year, 95% CI −20.6 to 48.9, p < 0.0001). 1 Survival comparisons were confounded by indication bias, but available data suggest little improvement in the overall duration of life. 1 The use of dialysis in those with a destination LVAD is less widely studied, but it is being increasingly adopted across several centers, and initial concerns related to infection and other risks appear to be low. 2
Based on the medieval Latin verb “palliare,” meaning “to cloak,” peritoneal dialysis would be a component of holistic palliative care aimed at reducing the distress John is experiencing from his breathlessness and edema. 3 Ideally, when taking a palliative approach to care, both burden and benefits should be clearly estimated and communicated. In this case, the effort required for the introduction of the treatment, including the process of catheter placement and the activity of dialysate exchanges, would be balanced against the value John places on the potential improvement in his activity level when he is less breathless and sleeping better.
What is frailty-informed dialysis care?
John has voiced that he would prioritize reading to or drawing with his granddaughter over all other daily activities. This role defines his values, and requires a health state where he can sit, unaided, for 60–120 min without distress. This aligns with a frailty-informed dialysis care plan. For him, health benefits arising from better blood pressure control, or reduced lower limb edema, are less valuable than the reduction of fatigue and improved sitting balance.
The phrase “frailty-informed” is used in this context to describe an approach to medical care where treatment goals are adapted to the individual's experience of health or illness (Frailty Informed Pathway, Figure 1). 4 Similar to changes emerging from the common-sense oncology movement, where treatments are being modified to minimize symptoms rather than promote longevity, frailty-informed dialysis pathways may allow the patient to live their life as richly as possible until the moment of death. In oncology, frailty assessment is incorporated into the personalized treatment plan. 5 By doing so, there appears to be a significant reduction in the intensity of chemotherapy and therapy-related toxic side effects. Patients achieve higher rates of treatment completion and report improved patient-reported and caregiver-centered well-being.6,7
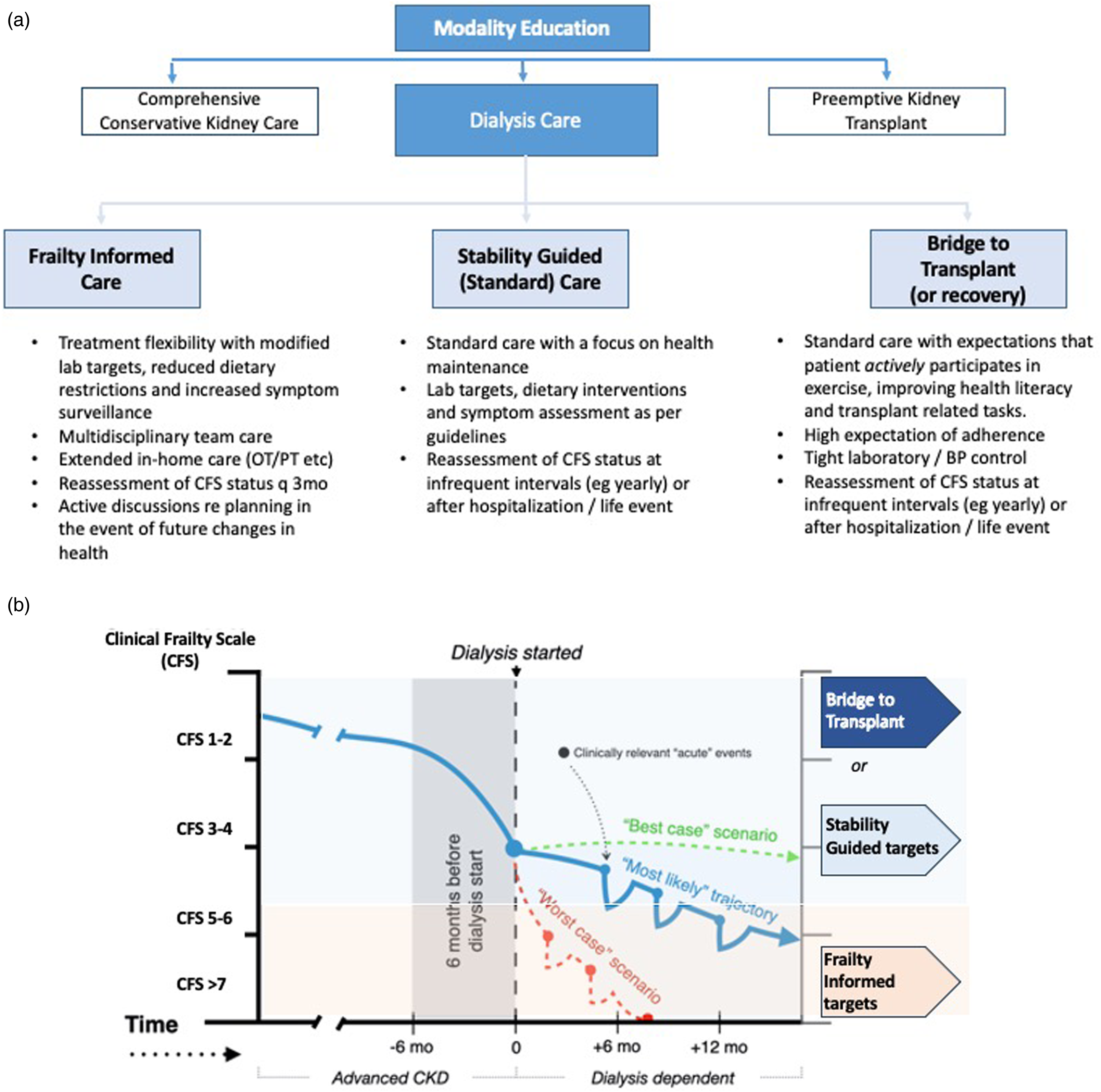
Frailty-informed approach to dialysis care. Suggested approach for how to incorporate frailty into clinical guidelines for the care of those undergoing maintenance dialysis. (a) Figure showing the different strata for patients who chose to pursue dialysis depending on their frailty status. (b) An illustration of how dialysis targets may be adapted to observed changes in health seen after starting dialysis. The lines reflect the typical trajectory over time for older individuals starting dialysis. We have used a Best Case (green dotted line), Worst Case (dashed red line), and Most Likely Case (solid blue line) approach. The individual, in the most likely case, initially manages well and is managed with a standard, stability-guided approach to targets; however, by 12 months has experienced recurrent ill-health (represented by the dips). As the individual becomes more frail, they may benefit from a change to frailty-informed health care and adapted targets. Please note the illustrated thresholds are suggestions based only on clinical experience and not supported by data. Adapted from Fonseca-Correa and Jassal. 8
Within a nephrology context, frailty-informed pathways would build upon the data from multiple studies showing that a high number of individuals starting on dialysis have high frailty levels.9–12 These individuals are those most at risk of ongoing symptom burden, and of treatment-associated morbidity and therefore, they stand to be those most likely to benefit from a tailored approach to care.12–14 In our suggested model of care, frail patients would have more access to multidisciplinary support (including physical and occupational therapy), less stringent blood pressure and laboratory targets, minimal dietary restrictions, and in some cases, a reduced frequency of regular lab draws. Furthermore, they could undergo medication assessment to reduce pill burden through, for example, discontinuation of statins, phosphate binders, and other medications used to limit disease but not symptoms. Multidisciplinary support could extend to caregivers, particularly as informal caregiver burden increases as the patient experiences higher symptom burden and lower mental quality of life. 15
There are numerous tools used to measure frailty. All have strengths and caveats. 16 The choice of frailty screening tool (and clinical thresholds) remains open to discussion. Ongoing work may lend credence to some tools more than others. We use the Clinical Frailty Scale (CFS) because it captures multidimensional frailty, has been shown to be meaningful in the dialysis environment, and is easy to apply. Its use by less experienced clinicians can be supported through the addition of a clinical classification tree.16,17
Within the frailty-informed approach, we suggest resilient patients (i.e., those doing well on PD or those bridging to transplant, Figure 1) be held to higher levels of health ownership. This may include an expectation that patients make positive lifestyle modifications such as increased exercise participation and dietary awareness, taking ownership of future health expectations, or tasks specific to the patient, for example, proactive measures as completing a priori paperwork towards future medication coverage for transplant medications.
The use of a frailty-informed approach aligns with, and enhances the current International Society of Peritoneal Dialysis (ISPD) guideline that places importance on prescribing high-quality goal-directed peritoneal dialysis. 18 It encourages the clinician to modify or adapt treatment goals in frail patients, based on personalized functional goals (physical, emotional, social, and spiritual) in preference to following conventional dialysis targets. By creating different strata of care based on frailty, we anticipate short- and intermediate-term improvements in care for both patients and the healthcare teams. The level of frailty may fluctuate over time, often in response to comorbid diseases, infections, or hospitalizations, leading to a trend over time represented by the slope of the lines in Figure 1(b).19,20 For those patients who show stability (green dotted line, Figure 1(b)) or improvement in their health trajectory over time, goals and targets can be revised to a higher stratum that applies more rigorous targets. For those who experience a decline in health (represented by the red dashed line, Figure 1(b)), goals may be revised to allow additional multidisciplinary care, focusing on the patient and caregiver experience with more relaxed clinical targets. This approach may lead to lower medicalization and patient distress, particularly in the final months to years of life. This approach can span across the full range of wellness and prognosis, so for example, those awaiting transplant may be expected to meet more stringent adherence criteria than those on a frailty-informed care path. The deliberate use of the word frailty, implicitly encourages family members to recognize their loved one's health state, but ideally without threat. By acknowledging the physical and/or psychosocial impairment(s) associated with their health state, patients will become more aware of how their health trajectory changes over time and adapt to more realistic expectations of their health. At an administrative level, adoption of formal terminology for each of the strata may be beneficial for setting clinical standards and quality measures such as those mandated by the Centers for Medicare and Medicaid Services in the United States.
What do I tell the patient about prognosis?
Prognostication in chronic kidney disease can be challenging, particularly at older ages. In a cohort of over 300,000 individuals, Bowling et al. showed substantial overlap in prognosis estimates across groups of healthy and unwell patients. 21 Their data showed wide confidence intervals around the estimates of life expectancy, such that point estimates of survival in those with normal kidney function fell within the confidence intervals applicable to those with advanced kidney disease. Effectively, this limits the ability of the clinician to give a numeric survival expectancy. As a result, we suggest clinicians communicate prognosis using the health trajectory.
Health trajectories are well described in the literature and have been used to help clinicians better understand disease. 22 In John's case, it is possible that peritoneal dialysis will reduce his dyspnea, and he may regain some function. It is unlikely he will return to his previous functional levels. His disease trajectory follows that of chronic organ failure and can be drawn using his personalized time scale over 14 years (Figure 2). Events, and recovery, illustrate his lived experience with health events, including how long it took to recover after hospitalizations, both 14 years ago and those more recently. When drawn freehand, during a conversation with the patient, it allows the patient to participate and often will illustrate the chronicity of the change and how they have adapted their lifestyle to their health status. Future expected declines in health may be implied but do not have to be explicitly stated. Often, patients will recognize their own prognosis through articulation of these health changes. Patients who ask about timelines for expected survival can be given information using terms such as days to weeks, weeks to months, or (e.g., for relatively stable patients) months to years. Furthermore, this anticipated trajectory can be incorporated into discussions using the Best Case, Worst Case, and Most Likely Case scenarios, facilitating goal setting to be adapted to focus on symptom-centered targets, rather than traditional targets.23,24
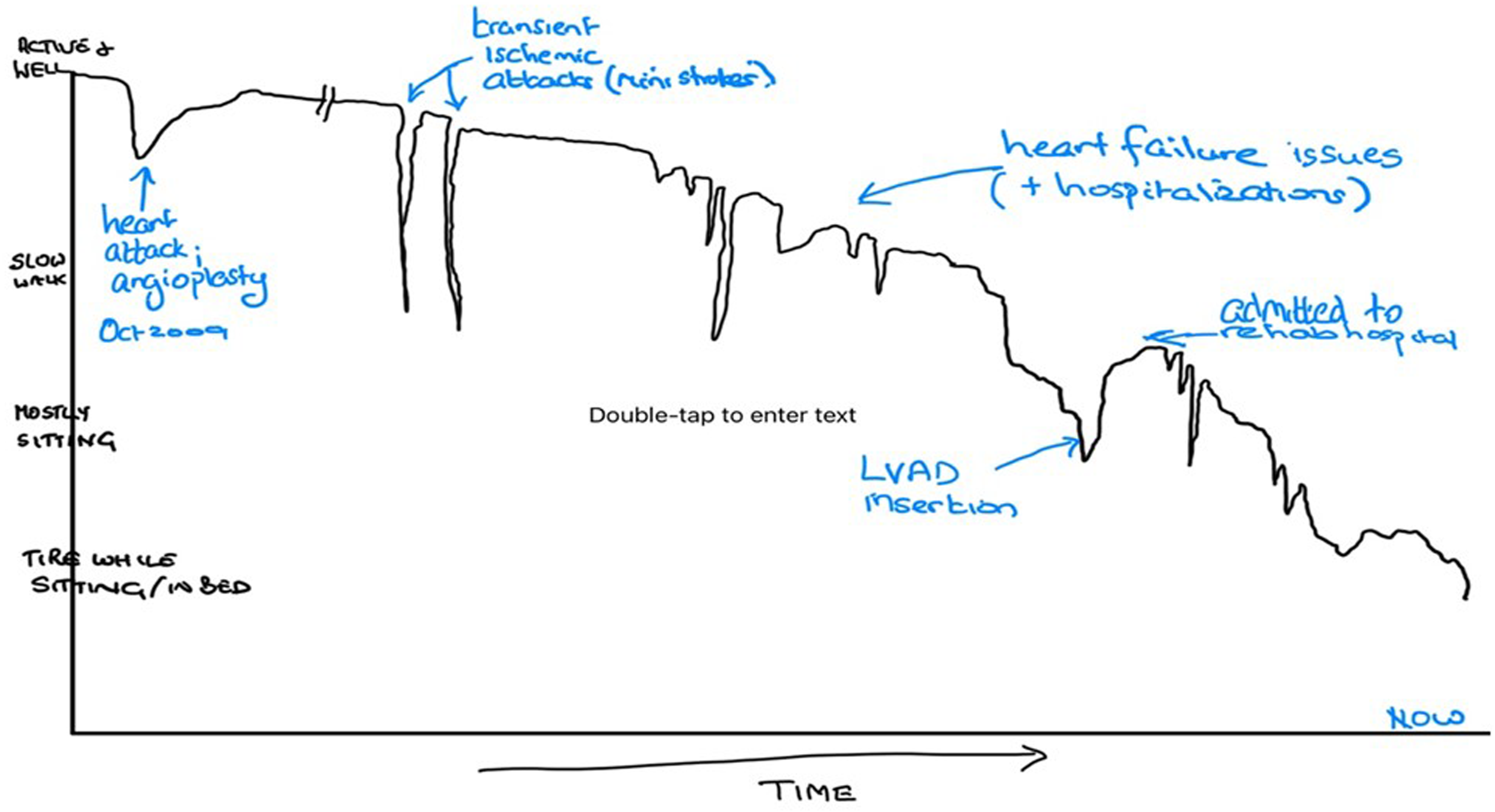
John's personalized health trajectory. This is an example of a hand-drawn health trajectory that represents John's well-being over the years. The black line has been drawn to illustrate the change in well-being and function reported by the patient, as his health varies with time. Some events have been labeled. The dips incorporate both the impact of the health event on his well-being and ability to participate in preferred activities, as well as the time taken to recover. For example, John reported that after his heart attack, he was able to manage most household tasks but worked shorter hours for some years (represented by a small vertical but wide horizontal line of recovery). In contrast, he found the transient ischemic attacks impacted his swallowing, speech, and ability to stand up or balance without assistance, but he had a rapid recovery. This saw-tooth appearance in a trajectory is often labeled as the chronic disease health trajectory.
How can I communicate that I anticipate John's health will decline without having him lose hope?
Patients may struggle with accepting their prognosis. Jackson et al., using a case vignette, describe how a continuum of conversations is an effective way of conveying prognosis (Figure 3). 25 In their article, we are reminded that each patient may be at a different stage of readiness to cope with accepting their prognosis. Many individuals with chronic health issues recognize that their disease is associated with increased mortality but do not internalize their own risk of death. Cultural beliefs may alter how discussions about death are held. In our case, John had initially hoped he would regain his health and become eligible for a heart transplant. While he accepted the LVAD, he imagined it only as a temporary measure. It is likely he did understand he may have ongoing health issues over time; however, he is still questioning why his symptoms continue to deteriorate, and expresses anger that is mired in unrealistic expectations. Peritoneal dialysis in this setting should be clearly described as only a symptom-modifying, and not disease-modifying, treatment. In some cases, it is advised to include education in advance about planned dialysis discontinuation, linking this option to circumstances when symptoms do not improve or patient goals are not met.
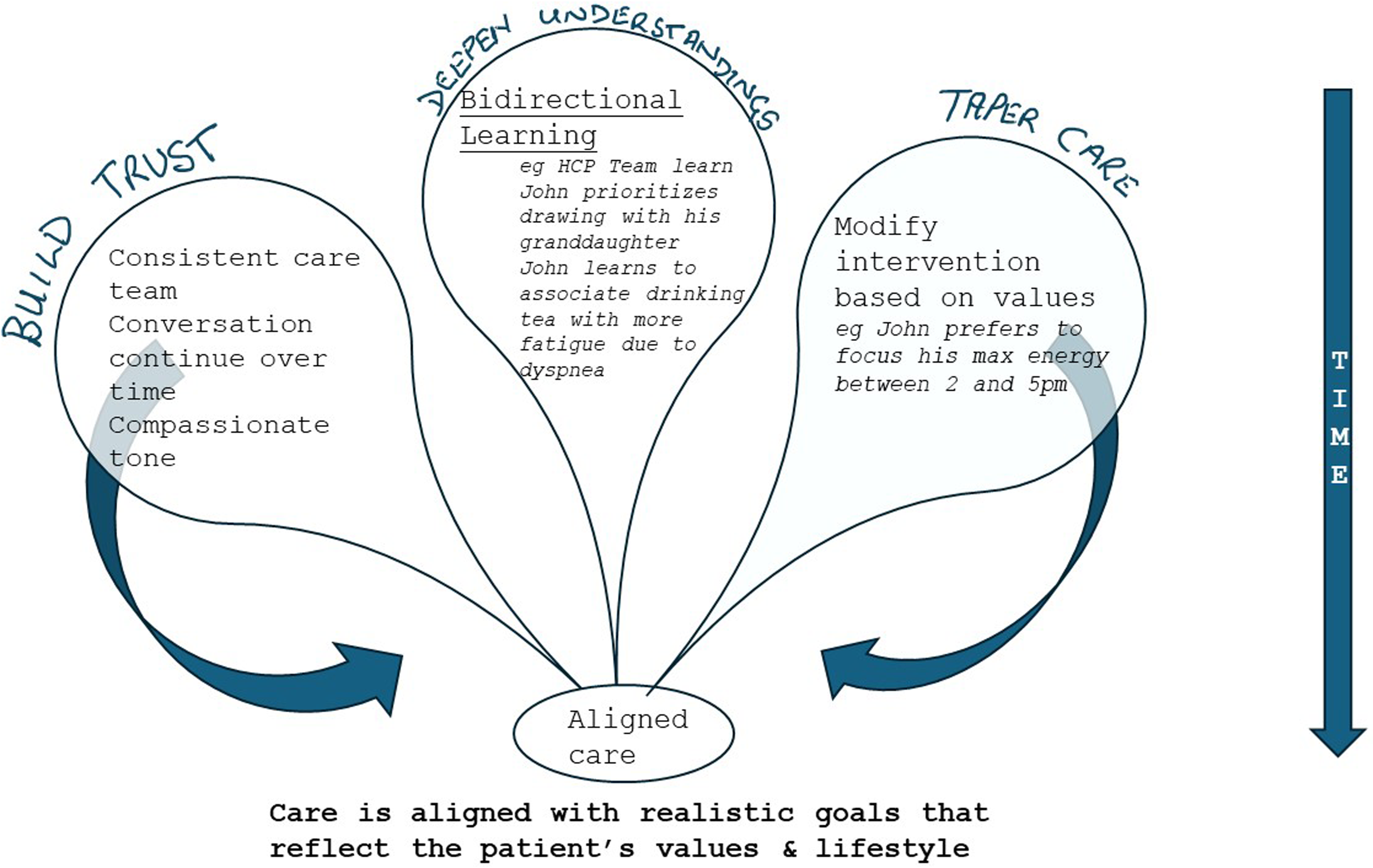
A conceptual model illustrating environmental methods that can improve prognostic awareness of likely outcomes. 25
Despite honest information about prognosis, he talks, during one visit of the importance of attending his granddaughter's university graduation. This apparent lack of acceptance of his prognosis is not uncommon and reflects a desire to balance reality with hope. In these circumstances, there is a need to recognize the patient's story or experience, and several tools can help clinicians respond with cognitive information.26,27 Often described as disease cadence, patients will recognize that they have good health days and bad health days, and they become accepting of the rhythmic nature of these. Engaging patients and families in recognizing these rhythms can be a helpful way to guide patients into better adaptive coping and more realistic health expectations. While it is tempting to correct him and remind him that it is unlikely he will be around for his granddaughter's graduation, it is more important to help him link his optimism to the current pleasure he gets from interacting with her. “I can see she is the apple of your eye, so let's start by sitting you up and getting you to keep your balance so you can enjoy seeing her most recent colouring work.” As his health declines, he may slowly recognize that this time is shorter or less frequent and thus improve his prognostic awareness. Particularly when the whole team is involved, it is possible to accelerate this prognostic awareness (and acceptance) through small observations. The physiotherapist, for example, may say I know you need to read to your granddaughter when you are home, so lets start by getting you sitting up straight in bed for 2 hours each day. Next week we need to see if you feel strong enough to sit in a chair and maybe even help with her colouring homework.
The physician may integrate prognostication by including a preferred target and using the Wish, Worry Framework described by the Serious Conversation Guide.
28
Ah, your ultrafiltration is close to 200 mls today. I wish we had closer to 400 mls. Let's see how we can make this work. I wonder if you can cut back a bit more on your fluid intake so the breathing remains comfortable. I worry your breathing may become less comfortable over time if the UF does not increase.
Similarly, it is possible to convey negative messaging by placing emphasis on shorter term, less demanding goals “well, I’m not promising graduation, but lets start with…” Over time, it is likely that John and the family will see the underachievement and acknowledge his reality. Opportunity to address patient's wishes regarding future procedures, hospitalizations, and resuscitation may occur at any time, and clinicians should have a heightened awareness of such opportunities. I can see you are worried that the medications are not improving … (symptom) … and that you will end up in hospital again. Do you want to talk about what happens if you chose not to go back to hospital? Maybe, we can talk about what it looks like to spend your final days at home.
These conversations can explore his ideas about end-of-life care, that is, if he prefers to stay at home vs. hospital or hospice in his final days, as well as incorporate information and guidance on how to access local palliative care services.
How do I recognize when a patient has reached the end of life?
Despite dialysis, John continues to get more and more fatigued and is increasingly somnolent. He rarely rises from bed. Having been discharged home with community palliative care services, he now has a hospital bed in the home that helps with caregiving. He has some lower limb edema bilaterally, but respiratory examination reveals a few crackles. His respiratory effort is shallow, and he is clearly fatigued. Dialysis exchanges are unremarkable, and ultrafiltration ranges between 400 and 600 mls per day using two icodextrin exchanges. He becomes fatigued when chewing and consequently has markedly reduced oral intake. It is felt that he is entering into the final days of life.
In several jurisdictions, end-of-life care may be supported in a hospice or by community palliative care services. In others, this may fall upon the nephrology team. Both families and patients value the long-term relationship they have built with their kidney care clinicians, and continuing to follow through end of life fulfills the responsibility of nonabandonment. Even if transferring care to a palliative care specialist, it can be valuable to continue to follow alongside the palliative care team for a time. 29
The term palliative care is sometimes confused with end-of-life care. The former, as noted above, is care directed at symptom mitigation and can occur over months to years of life. End-of-life care is better described as that care given as the patient enters into the final hours/final days of life. An honest conversation with family members at this stage is key, and despite the discomfort experienced by the clinician delivering the news, it can help alleviate anxiety, facilitate healthy grieving, and improve the collaborative relationship with family members. It is important to acknowledge the increased fatigue and somnolence that John is experiencing. If the family are receptive, engaging them in monitoring for symptoms (e.g., somnolence or distress) can help adjust their expectations as well and also allow medication titration. Simple statements such as “I see he is struggling to stay awake” can acknowledge the decline and open the discussion about the potential for dialysis discontinuation. Direct communication, such as “It may be a good time let family who live further away know things are changing” or “would it be helpful if we were to talk about other changes you may notice as the days go on?” may open a deeper conversation. Families can be taught about anticipated changes in breathing patterns or rhythm, involuntary movements (including twitching, jerks, or myoclonus), and delirium. Most cultures place a high value on food, so as the patient increasingly refuses food, it is important to communicate the relationship between the severity of illness and anorexia. Families taught to recognize reduced hunger as the discomfort of overeating (“as if they have just eaten a four-course meal”) have lower distress from the reduced intake. Finally, adequate opioid treatment of pain, distress, and dyspnea must be maintained while imparting an appreciation of how to manage opioid-related toxicities, such as myoclonus, delirium, and/or pain from constipation or hyperalgesia.
Conclusion
The principles and health trajectory outlined in this case are described for a patient with an unusually complex disease; however, they can be extended across the majority of cases presenting to nephrologists.
The proposed concept of frailty-informed dialysis pathways encourages the nephrologist to acknowledge the functional status of their patients, as this greatly impacts their health trajectory. It aids in adjusting treatment goals in patients with less resilience due to physical or psychosocial impairment in a deliberate way. This patient-centered approach prioritizes well-being and symptom management, rather than traditional targets, as a new “standard of care” in selected patients.
The incorporation of health trajectories into conversations offers a method through which the physician has an opportunity to communicate truthfully. This expands opportunities for advance care planning, including discussion about realistic options regarding future interventions, resuscitation, hospitalizations, and end-of-life wishes. In some settings, this may lead to closer collaboration with (nephro)geriatric or palliative services.
We acknowledge that the concepts proposed, where we create strata where different patients have different targets, require wider dialogue across both professional and patient stakeholders and funding agencies. With both aging, and an increase in complex comorbidity of the population we serve, discussion about new approaches becomes increasingly relevant. The use of frailty-informed dialysis pathways offers an alternative approach to health care beyond the simple provision of access to treatments, leading to a more holistic and more proactive strategy.
Footnotes
Author contributions
IVL conceptualization; writing – review and editing. SVJ conceptualization, writing – original draft, review and editing, visualization, supervision.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
