Abstract
Background:
In 2016, Peritoneal Dialysis Assist (PDA) was implemented in British Columbia, Canada, as a pilot program to allow patients with physical, cognitive and social impairments to access an independent dialysis modality. This is a presentation of the usage and 5-year clinical outcomes of our provincial assisted peritoneal dialysis (PD) program.
Methods:
Patients who utilised long-term or respite PDA services in British Columbia, Canada, from 2016 to 2021 were included in this program evaluation. Incident and prevalent patient numbers were characterised annually as well as indications for PDA and patient demographics both annually and over time. Outcomes of interest included death, transfer to haemodialysis, transplantation and cessation of the PDA program but retention on PD.
Results:
Three hundred twenty-two total patients received services through the PDA program. The percentage of PD patients supported by long-term PDA service has grown to 11.2% in the most recent year. Patients spend a median of 13.6 (95% CI: 11.0, 16.1) months on long-term PDA, prolonging overall patient duration on PD by a little over a year. Of the patients who exited the long-term PDA program, 73 (37.4%) were able to utilise the service until they died.
Conclusion:
PDA is an accessible, patient-centric service with clear and standardised referral criteria. Through the implementation of a local PDA program, patients have accessed PD and may have extended their PD life span, through avoidance of in-centre haemodialysis, by over 13 months during this 5-year study period. A significant proportion of patients on long-term PDA were able to use their preferred kidney replacement modality at home until they reached end of life.
This is a visual representation of the abstract.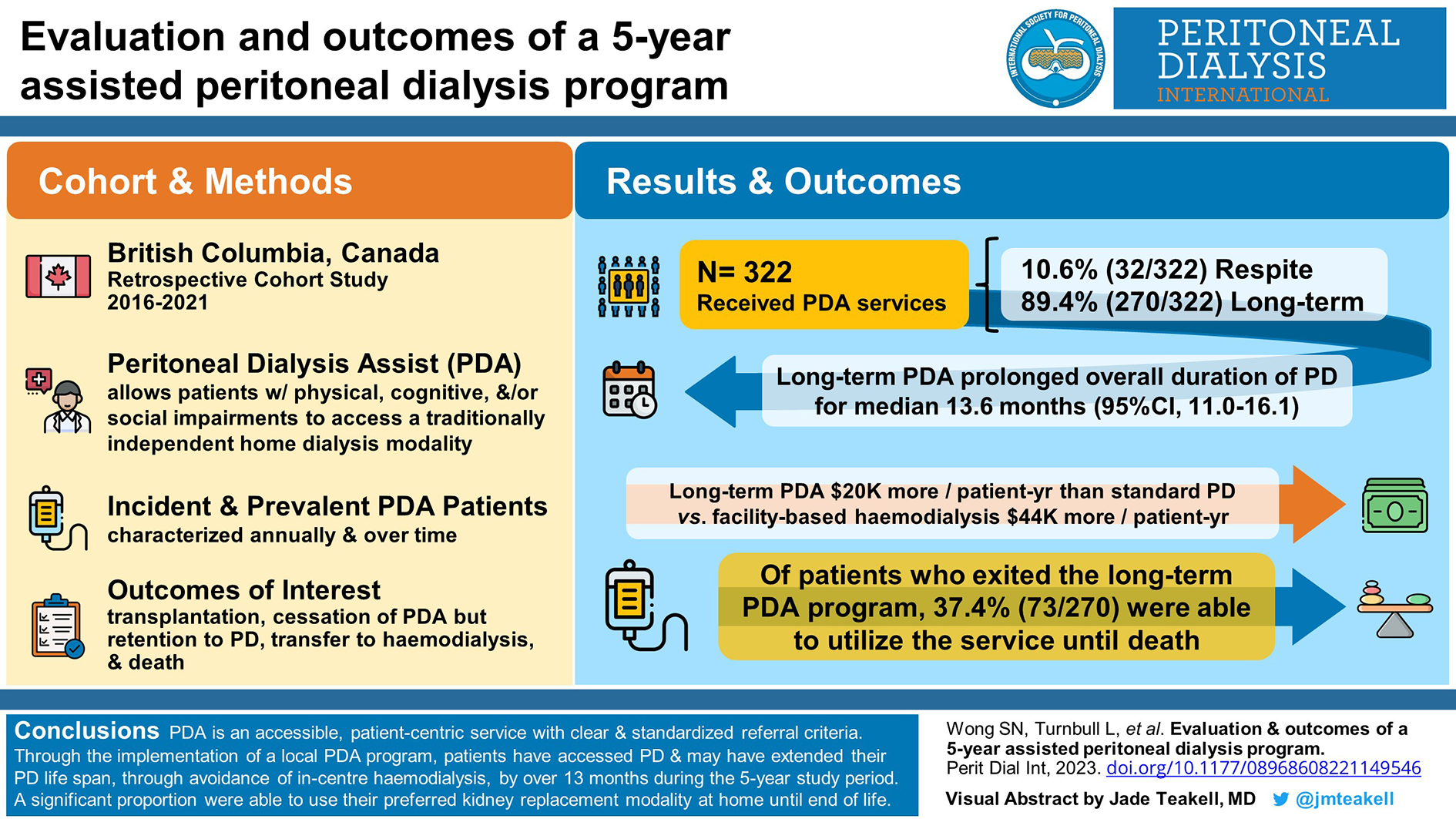
Background
Peritoneal dialysis (PD) is an underutilised kidney replacement modality, with only 24.1% of incident kidney failure patients on PD in British Columbia. 1 Historically, elderly patients with greater levels of physical and functional dependence have been excluded from PD; however, as the age demographic for dialysis shifts upwards, this patient population arguably has the most to benefit from this therapy. For many patients, PD offers greater independence and quality of life by avoiding the burden of frequent transportation and time-consuming in-centre haemodialysis treatments. 2 –4 As well, PD minimises the haemodynamic instability and increased risk of bacteremia that accompanies haemodialysis. 5 –7
Assisted PD has become a popular option to overcome patient-related barriers by utilising trained nurses and care aides to help manage various aspects of PD within the patient’s home. The first program was initiated in France in 1977, and both long-term and short-term respite models have since developed worldwide to increase the eligibility and accessibility of PD. 8,9 Various approaches to provide Peritoneal Dialysis Assist (PDA) exist across Canada. Ontario and Nova Scotia have developed provincial strategies for PDA. Winnipeg has partnered with Home and Community Care to provide this service while the Southern Alberta Renal Program provides PDA as part of care provision for PD patients who reside within the city limits of Calgary. A recent review of the Ontario program found that assisted PD provides at least similar health and quality-of-life outcomes with in-centre haemodialysis and reduces associated costs. 4
In 2015, British Columbia initiated a pilot project to explore the feasibility of a provincial PD Assist program. Our PDA program outsourced trained caregivers to perform specific tasks associated with cycler PD. Eligibility for this service was limited to patients who were able to perform their own cycler connections independently. Positive outcomes were observed from a clinical, financial and qualitative perspective. 3 In 2016, a formal home-based provincial PD Assist program was implemented to support patients who were identified as high risk for technique failure, with the goal of retaining them on PD. Here, we evaluate the usage and 5-year clinical outcomes of our provincial assisted PD program.
Methods
Description of service
PD program clinicians assessed patients for long-term or respite PDA to identify those who were eligible. Patients were required to be on continuous cycling peritoneal dialysis (CCPD) and excluded patients using continuous ambulatory peritoneal dialysis (CAPD). The patient or an identified support person was required to perform connections to and from the cycler machine and manage non-cycler aspects of PD care including fluid management, weight and blood pressure checks, effluent assessment and supply ordering. The patient or support person was required to troubleshoot cycler complications that may occur during therapy and communicate any identified concerns to their PD team.
Referrals to the long-term PDA service occurred if there were irreversible physical, cognitive or social barriers to performing independent PD. Patients with dexterity deficits affecting their ability to lift dialysate solution bags, difficulty troubleshooting cycler machine alarms during setup or dismantle or limited available support were candidates for this service. Referral to respite PDA occurred if short-term, temporary assistance was required (2 weeks to 3 months) from a caregiver until a return to independent PD.
After referral, trained caregivers provided once-daily home visits of 1 h, up to 7 days a week. The caregivers were hired, trained and supervised by a contracted service provider, Nurse Next Door. Caregivers must be a minimum level 3 caregiver as identified by the British Columbia Registered Care Aide Program or have equivalent experience. Each caregiver completed a 2-day training program to develop competencies in the activities, roles and responsibilities of PDA. Respite and long-term PDA activities were identical. BC Renal developed all training materials. The caregiver responsibilities included dismantling and setup of the cycler machine, measuring blood pressures and weights, adding heparin to dialysate, moving PD supplies within the home, assisting in disposal of PD garbage, documenting findings and notifying the PD unit of any issues. PDA patients or their families were responsible for CCPD connections, treatment decisions, troubleshooting and management of medical comorbidities. If machine troubleshooting was too onerous, the patient or family was required to perform an emergency disconnect from the machine at minimum.
Data source
The Patient Records and Outcome Management Information System (PROMIS) is a British Columbia province-wide registry of all patients attending multidisciplinary kidney care clinics and receiving kidney replacement therapy. This information system, under the governance of BC Renal, supports a wide range of work ranging from individual patient care management to program planning and policy development. PDA data registry was available and accessible through PROMIS.
Design, setting and participants
The primary cohort of interest was patients who participated in long-term or respite PDA service between 1 January 2016 and 31 August 2021.
Two comparator cohorts, matched CCPD and general CCPD, were included to assess the impact of the PDA service on the duration of PD treatment. The matched CCPD cohort was comprised of patients on CCPD during the same enrolment period as the PDA group and was propensity matched using a 1:1 greedy nearest neighbour matching algorithm with a caliper size of 0.2 times the pooled estimate of the common standard deviation of the logits of the propensity score. The variables included in the propensity scores calculation included age, sex, dialysis vintage and comorbidities. The general CCPD cohort consisted of all the remaining prevalent adult CCPD patients in British Columbia during the same enrolment period as the PDA group.
Outcomes of interest
The purpose of this PDA program was to reduce attrition from PD by supporting frail patients to remain at home on this kidney replacement therapy, instead of transferring to in-centre haemodialysis. To ascertain time on PD, patients were followed until termination of PD treatment, loss to follow-up, movement out of the province or August 31, 2021, whichever event occurred first. The reasons for PD termination were grouped into three categories: (a) dying on PD as defined by death occurring within 6 weeks of the last PD treatment, (b) transplantation and (c) transfer to HD for more than 90 days. We were also interested in the annual activity and growth of the PDA program by describing the number of entries and exits each year as well as the number of patients remaining on the program at the end of the year.
Cost analysis
The total annual cost of PDA care delivery included direct patient care, travel costs and administrative costs, including training of the external health service provider (Nurse Next Door) and BC Provincial Renal Agency administrative costs. To determine the economic implications of the program, the annual cost of PDA was compared with baseline PD costs as well as other options for patients failing self-care PD, including transfer to HD or transition to long-term care.
Statistical analysis
Demographics and baseline characteristics of the three cohorts were reported as median and interquartile range (IQR) for continuous variables, and frequency and percentages for categorical variables.
Kaplan–Meier curves were used to describe the probability of remaining on PD treatment, and the probability of remaining on PD treatment prior to transferring to haemodialysis while censoring for death and transplantation. The difference among cohorts were assessed based on a Log-rank test and Cox Proportional Hazard model adjusting for age, sex, dialysis vintage, diabetes and cardiovascular disease comorbidities.
All analyses were performed in SAS software, version 9.4 (SAS Institute Inc., Cary, North Carolina) and R software version 4.1.2.
Results
Time on PD
The average patient accessing PDA services was over 70 years old (median age of 72 and 74 years old for respite and long-term PDA, respectively), with a higher proportion of male patients with diabetes and cardiovascular disease represented in the long-term PDA group as compared to respite PDA. There was a longer dialysis vintage in the respite PDA group of 17 [IQR: 4.5, 28.2] months, compared to 10 [IQR: 2.8, 28.7] months in the long-term PDA group. Approximately 18% of the long-term group leveraged the PDA service to initiate their PD (Table 1).
PD Assist program patient characteristics.
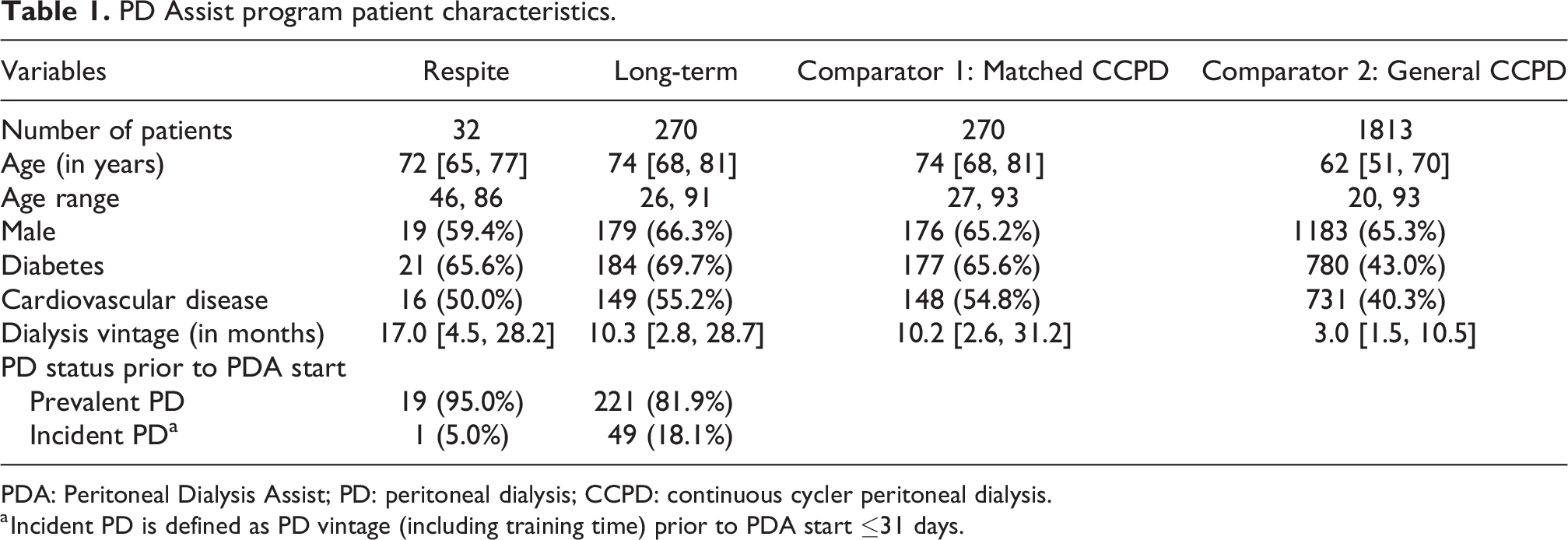
PDA: Peritoneal Dialysis Assist; PD: peritoneal dialysis; CCPD: continuous cycler peritoneal dialysis.
a Incident PD is defined as PD vintage (including training time) prior to PDA start ≤31 days.
By comparison, prevalent CCPD patients were younger (median age of 63 years old) and had a lower proportion of diabetes and cardiovascular disease than patients using PDA services. Prevalent CCPD patients also had a shorter dialysis vintage of 3 [IQR: 1.6, 13.0] months as compared to patients in the PDA program.
Patients who used PDA were significantly less likely to terminate PD compared to both matched CCPD (adjusted HR 0.72; 95% CI: 0.60, 0.88; p value = 0.001) and general CCPD (adjusted HR 0.82; 95% CI: 0.70, 0.95; p value = 0.011) cohorts (Figure 1(a)). Similarly, patients accessing the PDA service were less likely to stop PD due to transfer to HD, as compared to the matched CCPD cohort (Figure 1(b)).
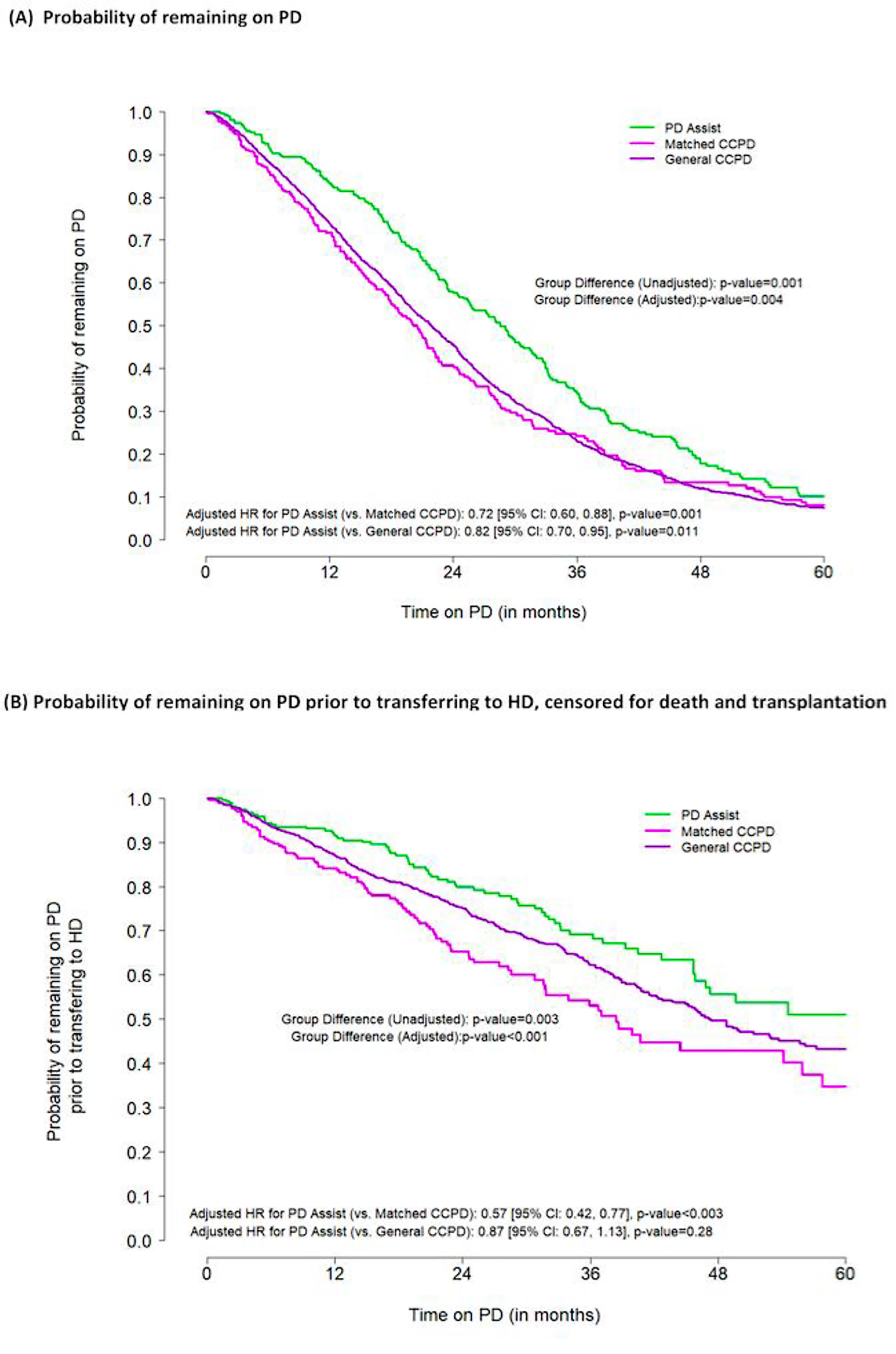
(a) Kaplan–Meier curves for probability of remaining on PD. (b) Kaplan–Meier curves for probability of remaining on PD prior to transferring to HD, censored for death and transplantation. PD: peritoneal dialysis; HD: haemodialysis.
Time on PDA
There were 270 patients who used the long-term PDA service between 1 January 2016 and 31 August 2021, which translates to 292.3 patient years on PDA. Survival on the PDA long-term service dropped by over 50% in the first year of its use (Figure 2). Patients spent a median of 13.6 (95% CI: 11.0, 16.1) months on long-term PDA, prolonging overall patient duration on PD by a little over a year.
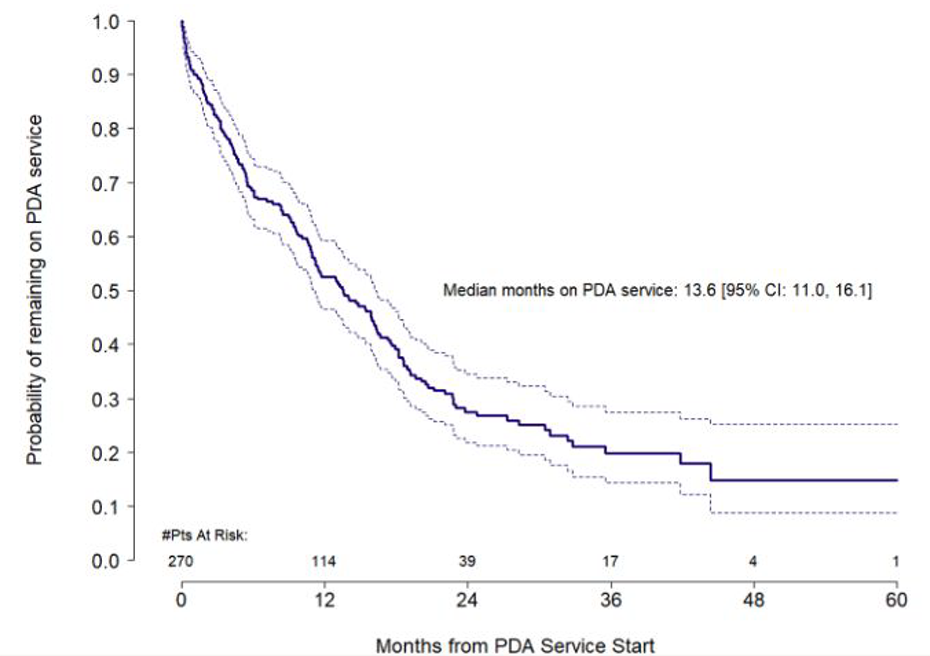
Kaplan–Meier curves for probability of remaining on PD Assist service over time. PDA: Peritoneal Dialysis Assist.
Causes for PDA termination
Among the 270 patients included in this analysis, 195 patients stopped using the long-term PDA service. Sixty (30.8%) patients transitioned to haemodialysis, predominantly for medical reasons. Thirty-one (15.9%) patients were able to continue PD at home or in long-term care. Twenty-two (11.3%) patients pursued comfort care measures shortly after discontinuation of PDA, 7 (3.6%) patients were transplanted and 2 (1.0%) patients moved out of province. Seventy-three (37.4%) patients used the long-term PDA service until they died. By comparison, 579 (29.5%) prevalent PD patients used this treatment modality until they died (Supplemental Figure 1).
Eighty-six (49.4%) patients died within 6 weeks of their last PDA treatment and 568 (28.9%) patients died within 6 weeks of their last PD treatment. These patients were examined retrospectively to see how many transitioned to haemodialysis before time of death. Within 6 weeks of exiting long-term PDA treatment, 6 (6.98%) patients transitioned to haemodialysis before death. Five (83%) of these patients were hospitalised and spent a median of 9 days [IQR: 2, 10] on haemodialysis before death. By comparison, within 6 weeks of exiting PD treatment, 61 (9.85%) patients transitioned to haemodialysis before death. Among these, 57 (97%) were hospitalised and spent a median of 12 days [IQR: 5, 20] on haemodialysis before death. The majority of time on haemodialysis was spent on an inpatient basis for both long-term PDA and PD patients who transitioned to this modality before time of death.
PDA growth
Between 1 January 2016 and 31 August 2021, a total of 32 patients received services through the respite PDA program and 270 patients received services through the long-term PDA program. An additional 20 patients used either services through multiple registrations, for reasons such as uncertainty around the COVID-19 pandemic.
The percentage of PD patients supported by the long-term PDA service has grown from 7% in 2018 (the first year the service was available across the province) to 11.2% in the most recent year (Figure 3). The overall use of PD has not significantly changed over time (ranging from 26.4% of prevalent dialysis patients on PD in 2016 to 25.1% in 2021). However, there has been a higher proportion of patients who withdrew from or died on PD and a lower proportion of PD patients who transitioned to haemodialysis since the PDA service was implemented. The average age of prevalent PD patients has increased over time from a median age of 65 to 69 years old, though the proportion of patients with medical comorbidities such as diabetes and cardiovascular disease has not significantly changed.
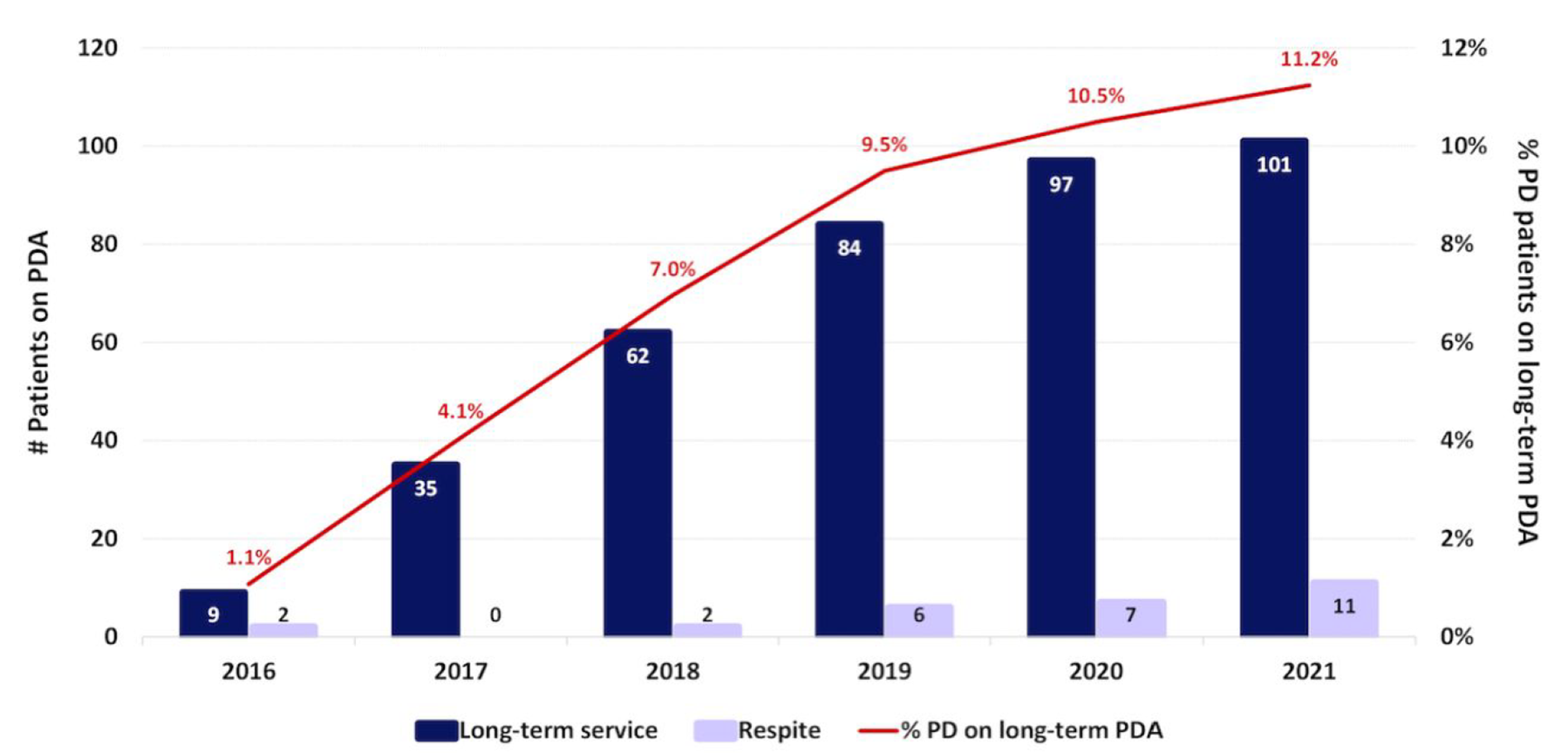
Growth of the PD Assist program over time as reflected by the number of active patients in the program on the last day of 2016–2020, and on 31 August 2021. PDA: Peritoneal Dialysis Assist.
Annual PDA initiation
When the PDA service became available province-wide, there were an average of 67 long-term and 14 respite referrals received. Sixty-seven long-term and 13 respite services started annually (Supplemental Figure 2). The wait time from referral to PDA start was similar between long-term (median days: 4.5 [IQR: 2, 10]) and respite (median days: 4 [IQR: 2, 7]) PDA. Providers were permitted to include multiple reasons for referral to the PDA program. The most common reasons for long-term PDA referral included physical deficits, dexterity deficits, social support deficits or cognitive deficits (Supplemental Figure 3). In contrast, the most common reasons for respite PDA referral were for physical deficits and post-hospitalisation assistance.
The PDA program was delivered in phases throughout the province: 3 regions in 2016, 4 regions in 2017 and all 5 regions in 2018. Fraser Health (FHA) and Vancouver Island Health Authorities (VIHA) were the most frequent users of the long-term PDA service. Respite PDA was used evenly by all regions in BC, particularly in the most recent 2 years (Supplemental Figure 4).
Over time, patients used more available PDA services, which included both cycler machine setup and dismantling as well as vital sign measurements. PDA was also used more frequently over time with 96% of enrolled patients currently receiving 7 visits per week.
Annual PDA attrition
Annually, on average 43 patients exited from long-term PDA and 9 patients from respite PDA (Supplemental Figure 2). Providers were permitted to document multiple reasons for exit from the PDA service. Long-term PDA exits primarily occurred due to death, followed by ‘other’ reasons, medical reasons, ineligibility for PDA or patient choice (Supplemental Figure 5). When explored further, ‘other’ options included transitioning to comfort care, transitioning to haemodialysis, or for peritonitis (3 episodes were cited as the reason for exit from the program). Patients who chose to leave the PDA program (i.e. patient choice) were more likely to continue PD independently as their modality of choice (59%) instead of switching to haemodialysis (24%). Common reasons for respite PDA exits included patient choice, improvement in clinical condition or death.
Costs of PDA
Per patient year costs for long-term PDA were approximately $20,000 more than standard PD. By comparison, transition to facility-based haemodialysis results in an annual cost of $44,000 more than standard PD. Similarly, long-term care, which may be a requirement for patients who are unable to independently perform PD, is associated with an annual cost of $38,500 more than standard PD 10 (Figure 4). Respite PDA had an estimated cost of $1250 for the median usage of 29 days, which is significantly less than the historical defaults of hospitalisation or transfer to haemodialysis.
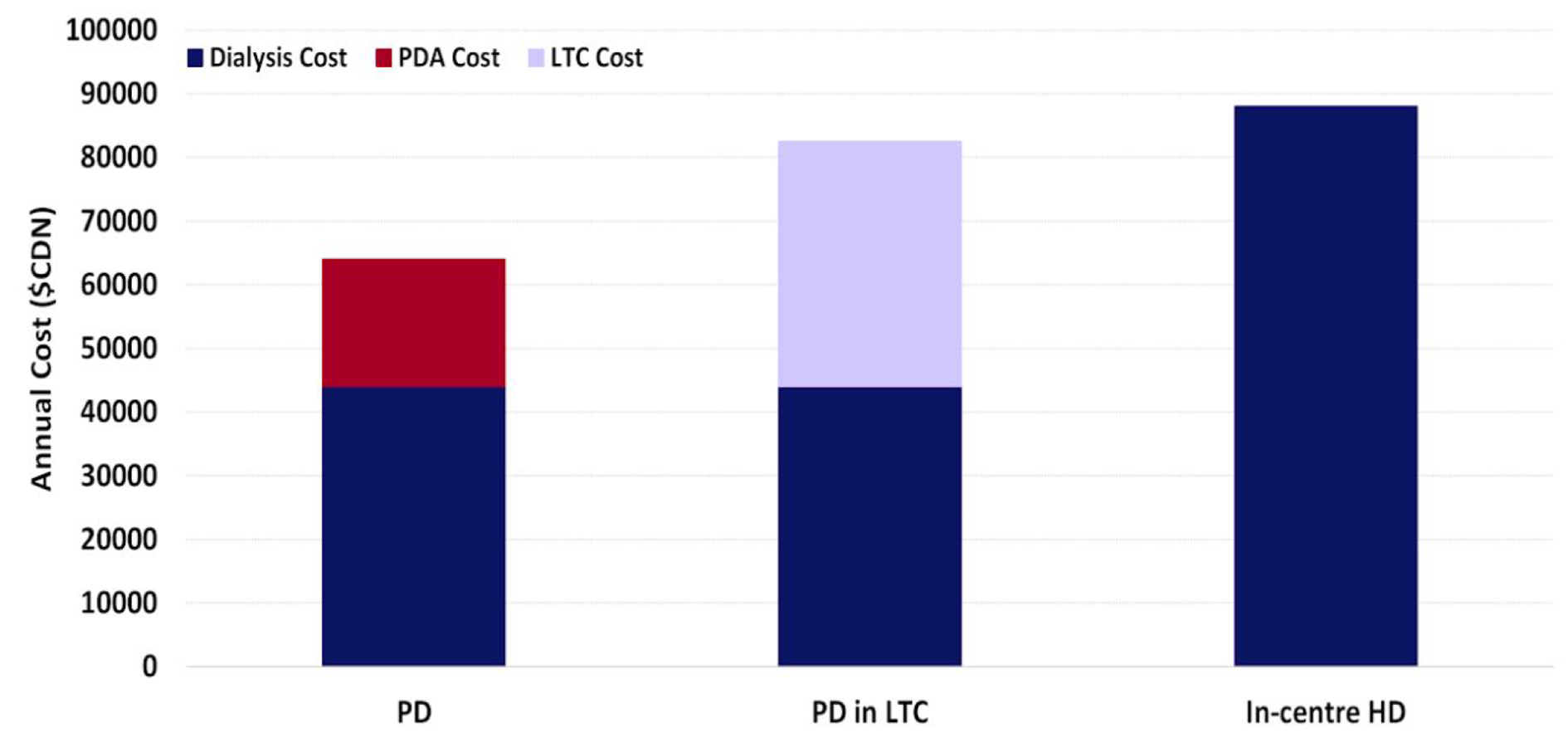
Annual cost of PDA and other options for patients failing self-care PD. PDA: Peritoneal Dialysis Assist; PD: peritoneal dialysis; LTC: long-term care; HD: haemodialysis.
Discussion
This 5-year program review demonstrates the success of an assisted PD model that offers accessible, patient-centric service with clear and standardised referral criteria to support PD patients at high risk of technique failure. Over the 5-year study period, our program allowed 270 long-term PDA patients to access and potentially extend their PD life span by over 13 months, through avoidance of in-centre haemodialysis. This program permitted many patients to stay home on their chosen treatment modality until end of life.
The average patient accessing PDA care was elderly and comorbid with a long dialysis vintage. These findings are consistent with patient demographics from other PDA programs. 2,4,11 Most patients used PDA on a long-term basis. Respite PDA provided the option for patients to continue their therapy of choice while receiving extra assistance for a potentially reversible medical condition or social situation. The program demonstrated steady growth over time with 11.2% of the PD population in British Columbia currently accessing PDA services. Local differences in PDA referrals across health authorities were noted, with Fraser (FHA) and Vancouver Island (VIHA) Health Authorities representing the majority of PDA referrals over the 5-year study period. This may reflect differences in local patterns of practice or different patient demographics in these regions.
Most exits from the respite PDA program occurred due to improvement in an underlying medical condition. These patients used the respite program appropriately for reversible situations with the eventual goal of returning to independent PD. By contrast, exits from the long-term PDA program occurred primarily due to patient death. A greater proportion of patients died on long-term PDA as compared to the baseline PD population. This may be explained by differences in patient characteristics, with more elderly and comorbid patients represented in the long-term PDA group. 3 PDA patients were also less likely to stop PD for reasons other than transplantation or death, as compared to the matched CCPD cohort. Long-term PDA provided the opportunity for patients to die or stay on an independent kidney replacement therapy without experiencing a disruptive and costly modality change to haemodialysis near end of life.
Many of the surviving patients that exited the long-term PDA program chose to continue PD independently or with family support. Those that transitioned to haemodialysis did so predominantly for medical reasons. This speaks to the willingness of patients to maintain their quality of life and independence by continuing with PD as their chosen treatment modality. The data are also consistent with the positive qualitative experiences voiced by participants of our 1-year pilot PDA program. 3
The program was also successful in allowing a significant proportion of patients on long-term PDA to use their preferred kidney replacement modality at home until they reached end of life. By comparison, those who were already near end of life and chose to switch treatment modalities spent most of this time hospitalised on inpatient haemodialysis before death. The number of hospitalisations or days on haemodialysis was not significantly different than patients in the baseline PD population, which suggests similar benefits of using long-term PDA or PD as compared to transitioning to haemodialysis near end of life. The long-term PDA service however, provided the additional benefit of allowing patients, who may otherwise not have been eligible, to access an independent kidney replacement modality for end of life care.
Our preliminary cost analysis demonstrates that there are clear financial benefits to long-term and respite PDA, as compared to transition to in-centre haemodialysis. As there is no zero-cost option for dialysis patients, so the goal is to provide care that produces the best clinical and quality-of-life outcomes at the lowest cost. Without the PDA program, this subset of frail patients would likely transition to facility-based haemodialysis or long-term care. The cost avoided for each patient who uses long-term PDA instead of HD is $29,000 per patient year, and the costs of the PDA program are recouped if every 3.9 patient years of PDA treatment results in the avoidance of 1 patient year of HD. In this way, PDA is a cost-minimisation initiative.
We acknowledge several limitations to our program review. The growth and success of each assisted PD program is influenced by the scope of practice that caregivers can provide. The PDA program in British Columbia was designed to reduce attrition from PD by addressing a specific need in the context of a universal healthcare system with resource limitations. It is by no means the only model of assisted PD. French registry data show that half of the incident PD patients use PDA services, 12 with many European programs able to offer multiple home visits per day. In comparison, Ontario delivers this service through integration with other home care services and dialysis care models and enrols 20–25% of PD patients in their PDA program. 8,13 We believe that our PDA program has steady growth and good uptake, with most referrals leading to patient enrolment. Our healthcare providers have a good understanding of the type of patient who would benefit from available services and are submitting appropriate referrals to the PDA program. Over the 5-year study period, PDA services were expanded to all Health Authorities in British Columbia, resulting in a higher frequency of home visits, particularly in urban areas. Unfortunately, the delivery of assisted PD was limited in rural communities, with fewer referrals received from these regions. Although the PDA program is supported by all PD programs, hiring of caregivers for some remote areas proved challenging. It was felt that this rural population likely received greater support from family and community members, which reduced the need for PDA. We recognise that this may become a more pressing issue in the future with rising labour shortages in all workplaces.
As the role of the trained caregiver changes over time, it will be interesting to evaluate whether an increase in scope of practice and responsibilities, such as antibiotic administration for peritonitis, exit site checks or cycler connections, will allow more kidney failure patients to participate in PD through the PDA program. We also recognise that many patients use PD until they reach the end of their lives. It will be worthwhile to explore how the PDA program can be tailored to facilitate this process with a focus on quality-of-life measures. Finally, more research and patient partnership is required to explore the role of PDA in rural and remote communities, and to explore barriers to its uptake and usage.
Conclusion
This 5-year program review describes the sustained growth and success of an assisted PD model in supporting PD patients at high risk of technique failure. Over a 5-year study period, our program has allowed 270 long-term PDA patients to access and potentially extend their PD life span through avoidance of in-centre haemodialysis by over one year. Because of this service, many patients were able to stay home on their chosen treatment modality until end of life. Our evaluation demonstrates responsible stewardship of an assisted PD program, and we hope to see continued benefits for patients and their families as the program moves forward.
Supplemental material
Supplemental Material, sj-docx-1-ptd-10.1177_08968608221149546 - Evaluation and outcomes of a 5-year assisted peritoneal dialysis program
Supplemental Material, sj-docx-1-ptd-10.1177_08968608221149546 for Evaluation and outcomes of a 5-year assisted peritoneal dialysis program by Shannon N Wong, Linda Turnbull, Sushila Saunders, Lee Er, Micheli U Bevilacqua, Adeera Levin and Rajinder S Singh in Peritoneal Dialysis International
Footnotes
Acknowledgements
We gratefully acknowledge all members of the PDA Committee for their contribution to this work.
Author contributions
Made a substantial contribution to the concept or design of the work: SW, LT, SS, LE, AL, RS, acquisition, analysis or interpretation of data: LT, LE, SW. Drafted the article: SW, LE, LT, revised it critically for important intellectual content: AL, MB, LT, SS, Approved the version to be published: SW, LT, SS, LE, MB, AL, RS.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Funding
The author(s) received financial support for the research, authorship, and/or publication of this article: S Wong is funded through the Ministry of Health, UBC Nephrology Core Fellowship program.
Informed consent
Not applicable.
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
