Abstract
Background:
People on peritoneal dialysis (PD) at risk of transfer to haemodialysis (HD) need support to remain on PD or ensure a safe transition to HD. Simple point-of-care risk stratification tools are needed to direct limited dialysis centre resources. In this study, we evaluated the utility of collecting clinicians’ identification of patients at high risk of transfer to HD using a single point of care question.
Methods:
In this prospective observational study, we included 1275 patients undergoing PD in 35 home dialysis programmes. We modified the palliative care ‘surprise question’ (SQ) by asking the registered nurse and treating nephrologist: ‘Would you be surprised if this patient transferred to HD in the next six months?’ A ‘yes’ or ‘no’ answer indicated low and high risk, respectively. We subsequently followed patient outcomes for 6 months. Cox regression model estimated the hazard ratio (HR) of transfer to HD.
Results:
Patients’ mean age was 59 ± 16 years, 41% were female and the median PD vintage was 20 months (interquartile range: 9–40). Responses were received from nurses for 1123 patients, indicating 169 (15%) as high risk and 954 (85%) as low risk. Over the next 6 months, transfer to HD occurred in 18 (11%) versus 29 (3%) of the high and low-risk groups, respectively (HR: 3.92, 95% confidence interval (CI): 2.17–7.05). Nephrologist responses were obtained for 692 patients, with 118 (17%) and 574 (83%) identified as high and low risk, respectively. Transfer to HD was observed in 14 (12%) of the high-risk group and 14 (2%) of the low-risk group (HR: 5.56, 95% CI: 2.65–11.67). Patients in the high-risk group experienced higher rates of death and hospitalisation than low-risk patients, with peritonitis events being similar between the two groups.
Conclusions:
The PDSQ is a simple point of care tool that can help identify patients at high risk of transfer to HD and other poor clinical outcomes.
Keywords
Introduction
Peritoneal dialysis (PD) is an attractive therapy for both patients and clinicians due to clinical and lifestyle advantages. Unfortunately, less than half the patients starting PD remain on the modality by the end of the second year of therapy. 1,2 Furthermore, transfer from PD to haemodialysis (HD) is common and associated with poor clinical outcomes. The transition from PD to home HD is infrequent, 3,4 and the majority of patients transitioning to in-centre HD start with a central venous catheter, indicating poor preparation for the transition, with associated higher costs and rates of infection and mortality. 2,5 –7
These challenges provide an opportunity to introduce more systematic approach to the evaluation and interventions of high-risk patients to improve care. However, given the impracticality of implementing a high-resource intervention across the entire population, a risk-stratification tool is necessary to focus additional resources on those at high risk. Several prediction models exist to identify patients at risk of transferring to HD, 1,2,8 but they only target specific groups, such as incident dialysis patients or those who experience peritonitis events. 9 Furthermore, existing prediction tools are relatively complex and not readily available at the point of care.
Clinicians naturally assess a patient’s risk for poor clinical outcomes as part of routine care and use this information to manage individual patients. In palliative care, the simple point of care surprise question (SQ) ‘Would you be surprised if this patient dies in the next 12 months?’ has been used to identify patients at high risk for mortality. 10 –12 It has also been shown to be predictive of mortality in patients undergoing HD. 13 In this prospective observational study, we modified the palliative care ‘surprise question’ and evaluated its performance in identifying a subgroup with higher risk of transfer from PD to HD. The proposed utility of this tool would be to supplement regular clinical practice for an identified subgroup of patients with systematic evaluation and interventions. These interventions would be tailored to support those who may benefit from remaining on PD and, where appropriate, to optimise transfer to HD.
Materials and methods
Study design
In this prospective observational study (Clinicaltrials.gov: NCT04666844), we used the electronic health record (EHR) system of a non-profit dialysis organisation to select all adult patients on PD as of December 2020. Patients engaged in PD training and those enrolled in hospice were excluded.
We asked the treating nephrologists and registered nurses, for patients under their care, the modified surprise question (PDSQ): ‘Would you be surprised if this patient transferred to HD in the next 6 months?’. We conducted brief didactic sessions to familiarise nurses with the PDSQ. We provided each nurse with a list of patients under their care with fields for their responses. Responses were collected via encrypted e-mail. Reponses from the nephrologists were collected for each of their PD patients over fax, phone or encrypted email after they reviewed a written explanation of the PDSQ. To minimise bias, nurses and nephrologists were blinded to responses from the other discipline. We obtained responses to the PDSQ from nephrologists and nurses between 23 November and 30 December 2020. We used the ‘yes’ or ‘no’ answer to define our exposure of interest by categorising patients into low-risk and high-risk groups, respectively.
In the primary analysis, we assigned a unifying index date for all patients (10 December 2020), which signified the start of the follow-up period. For the primary analysis, as imminent outcomes may be easier to predict and with limited opportunity to intervene on, we excluded patients who experienced any of the study outcomes within the first month of follow-up, including transfer to HD, death, transplantation or transfer to another facility (from 10 December 2020 to 10 January 2021). In sensitivity analysis, we included all patients who had PDSQ responses (including patients who experienced early outcomes) and used the response date of the PDSQ as the start of the follow-up for each patient instead of a unified index date. The full study timeline is shown in Figure 1.
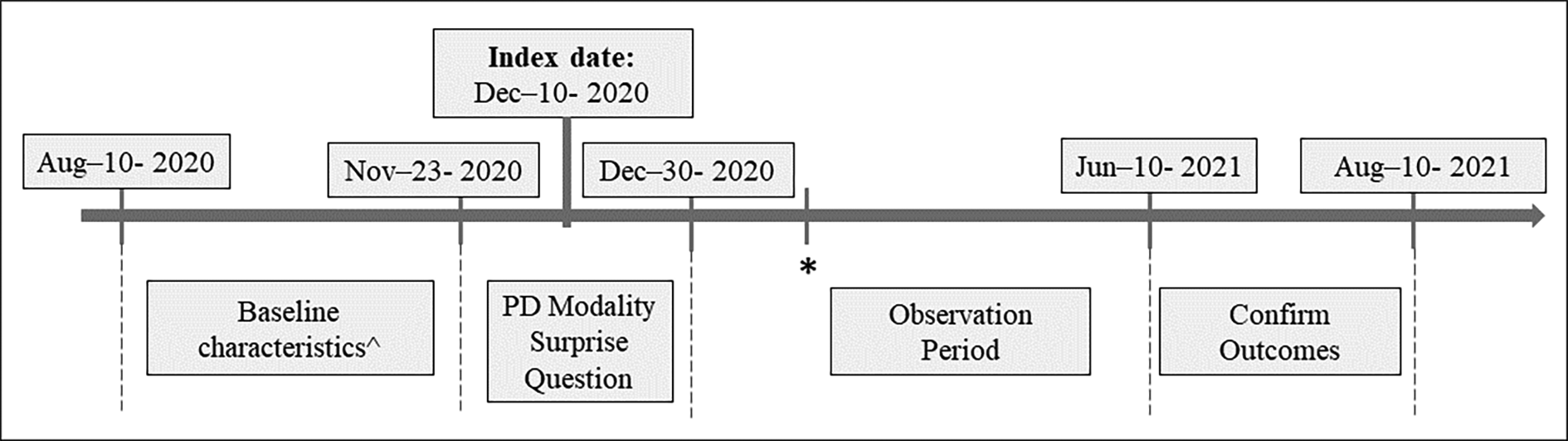
Data collection and follow-up timeline.
Study cohorts
We sought responses for all PD patients active during December 2020 (‘Target Population’). We excluded eligible patients for whom no response from the nurse or nephrologist was received. We developed three cohorts of patients: the RN-cohort included all the patients with responses from nurses, the MD-cohort included all the patients with responses from nephrologists and the RN-MD cohort included patients with responses from both the nurse and the nephrologist.
Study outcomes
The primary outcome of interest was transfer to HD. As secondary outcomes, we examined the association of the PDSQ response with death, hospitalisation and peritonitis. In addition, among patients who transferred to HD during follow-up, we evaluated whether patients identified as high risk were more likely to transfer to HD with permanent access (arteriovenous fistula/arteriovenous graft (AVF/AVG)) and whether they were more likely to transfer to home HD. Patients were followed up to the earliest of transfer to HD (the primary outcome), kidney transplant, death, discontinuation of dialysis, loss to follow-up, recovery of kidney function, transfer to a facility managed by another provider or survival on PD until 10 June 2021, the end of the 6-month follow-up period. Modality outcome data were examined until 10 August 2021 to enable confirmation of outcome designation. Transfer to HD with return to PD within 60 days was considered temporary and not censored.
Study variables
Baseline characteristics and patient and modality outcome data were obtained from the provider’s EHR system. Insurance was categorised as commercial/private, Medicaid (including managed Medicaid, self-pay and no insurance) and Medicare (including Medicare advantage). Laboratory values were the latest results recorded within 3 months before the index date for all biochemical tests, except adequacy tests for which we recorded the latest result within 6 months before the index date. Hospitalisation and peritonitis data for the baseline characteristics were observed for the 3 months before the index date. Hospitalisations included all hospital encounters including emergency department visits. Nurses’ clinical experience was collected from the organisation’s human resources database, including the number of years practicing as a PD nurse and the number of PD patients under the care of each nurse. Nephrologists’ clinical experience was collected from each nephrologist along with the PDSQ responses, including number of years in practice and number of PD patients under their care.
The study was approved by an institutional review board (WCG Aspire, IRB #20203611) with the waiver of consent.
Statistical analysis
We presented baseline characteristics for categorical variables as proportions and continuous variables as mean (standard deviation) or median (Quartile [Q]1–Q3) as appropriate. We compared high- and low-risk groups in each cohort using χ 2 test for binary (Fisher’s exact test for small-sized samples) and logistic regression for continuous variables. We assumed that the data were missing at random, given the observed characteristics. Missing data were handled using multiple imputation by fully conditional specification as implemented in SAS. For the multivariable model of looking at associations between characteristics and clinician responses, we fitted a logistic regression model to each imputed data set that included any variable selected through the backward elimination algorithm, applied to each imputed data set separately and these estimates and their standard errors were combined using Rubin’s rules. For the RN-MD cohort, we compared the number of concordant and discordant responses and measured the agreement using the κ-statistic. For hospitalisation and peritonitis events, we calculated incidence rates (first event during the follow-up period) and event rates (all events during the follow-up period) by dividing the total number of patients experiencing events and the total number of events, respectively, by the total number of person-time at risk. Incidence–rate ratios and event–rate ratios were estimated using Poisson regression with an offset for person-time at risk. To compare the performance of the PDSQ tool at different time points, we performed Cox proportional hazard models to calculate prediction confusion metrics including sensitivity, specificity, positive predictive value and negative predictive value. We examined the model discrimination ability by calculating the concordance statistic (c-statistic). We also used a bootstrap method to get the bias-corrected area under the curve (AUC) (t) (SAS Macro %TIMEAUCBOOT) to perform internal validation of each predictive model. To estimate the effect of the outcome of transfer to HD by high- and low-risk groups, we first examined the association between clinicians’ responses and the first of death, transplant or transfer to HD. This association was not statistically significant in all cohorts. We then ran cause-specific and sub-distribution analyses for these outcomes. Results showed a similar lack of association. This indicated that the cause-specific analyses for the outcome of transfer to HD was sufficient to enable interpretation of the data without the need for competing risk analyses. We used Cox proportional hazard regression to estimate the unadjusted effect of the outcome of transfer to HD by high- and low-risk groups in each cohort. The p value for the test of proportional hazards assumption was all greater than 0.5, showing no evidence that the log-hazard ratio (HR) changed with follow-up time for any of the covariates that were included in the model. We used a 5% significance level for all the analyses.
All statistical analyses were performed using SAS, version 9.4 (SAS Institute, Inc, Cary, North Carolina, USA), Stata version 17.0 (Stata Statistical Software; StataCorp LLC, College Station, Texas, USA).
Results
Study flow
We identified 1275 adult patients on PD as of December 2020 and eligible to participate in the study. Nurses and nephrologists who responded provided responses for all patients under their care. PDSQ responses were obtained from 95 (out of 113) nurses and, separately, from 80 (out of 179) nephrologists. As a result, nurse responses were available for 1169 patients and nephrologist responses for 727 patients. Within 1 month after the index date, 52 patients experienced events: 13 patients transferred to HD, 20 died, 7 transferred to another facility, 1 had kidney function recovery and 11 were transplanted. For the primary analysis, patients with outcomes within 1 month after the index date were excluded (46 of patients with nurse responses and 35 patients with nephrologist responses). As such, final cohorts for the primary analysis consisted of 1123 (88%), 692 (54%) and 616 (48%) patients for the RN cohort, MD cohort and RN-MD cohorts, respectively (Figure 2).
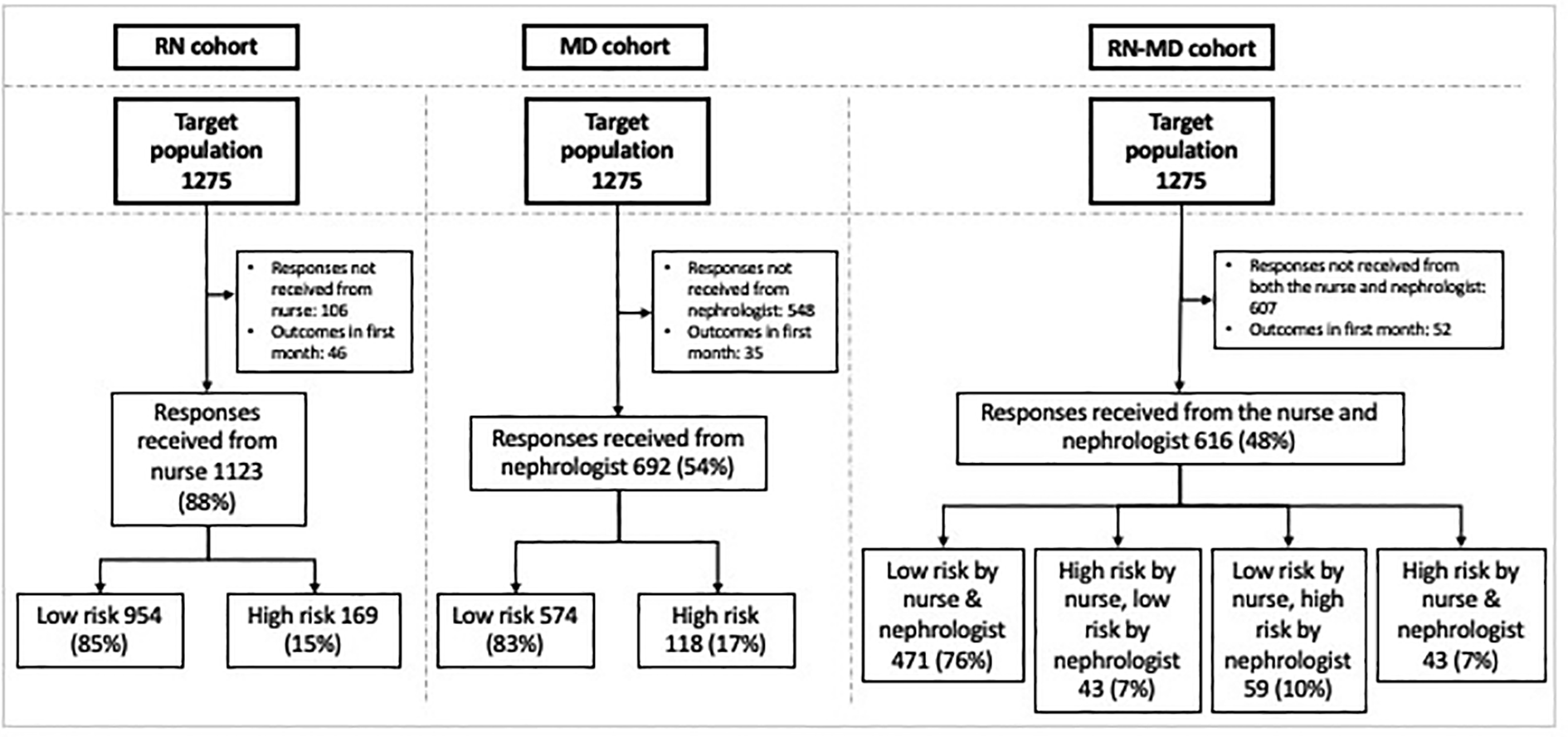
Study flow diagram.
Nurses identified 169 (15%) patients as high risk, whereas nephrologists identified 118 (17%) as high risk. In the RN-MD cohort, 43 (7%) patients were identified as high risk by the nurses and nephrologists, 59 (10%) as high risk by only the nephrologists and 43 (7%) as high risk by only the nurses.
In the RN cohort, the total follow-up time was 6341 patient-months. During the 6-month follow-up period, 47 (4.2%) patients transferred to HD, 53 (4.7%) received kidney transplants, 48 (4.2%) died, 10 (0.9%) discontinued dialysis, 1 (0.1%) recovered kidney function, 35 (3.1%) transferred to another facility and 929 (82.7%) remained on PD. The event rate of hospitalisation and peritonitis was 10 and 1.6 per 100 patient-months, respectively. Of the 47 patients who transferred to HD, 22 (47%) started HD with permanent access (AVF/AVG), and only 3 (6%) patients transferred directly to home HD. The distribution of outcomes in the other study cohorts was similar to that of the RN cohort (Online Supplemental Table 1).
Clinician characteristics and agreement between nurses and nephrologist responses
The median clinical experience for nurses and nephrologists was 4.2 (interquartile range (IQR): 1–10) years and 15 (IQR: 8–20) years, respectively. The median numbers of patients under the care of nurses and nephrologists were 13 (IQR: 8–17) and 11 (IQR: 6–15), respectively.
For 616 patients who received responses from both nurses and nephrologists, responses were concordant in 83%, with the highest agreements in the assignment to the low-risk group (76%). Results were discordant in 17% of responses (Online Supplemental Table 2). Overall agreement was fair; κ 0.36; 95% confidence interval (CI): 0.26–0.46.
Baseline characteristics
In the target population, the mean patient age was 59 ± 16 years and 41% were women. The predominant races were white (55%), Asian (28%) and African American (12%). Twenty-eight per cent were Hispanic or Latinx. The majority (84%) lived with their family or partner. The median PD vintage was 20 (IQR: 9–40) months, and 92% were on automated peritoneal dialysis (APD). The median total weekly Kt/V was 2.1 (IQR: 1.9–2.4), whereas the median weekly residual Kt/V was 0.6 (IQR: 0.1–1.2). The predominant primary cause of kidney disease was diabetes (39%), followed by hypertension (22%) and GN (15%). The median Charlson Comorbidity Index (CCI) score was 4 (IQR: 2–4). Of the patients, 47 (4%) had peritonitis events within 3 months before the index date. Hospitalisation before the index date was observed in 179 (14%) patients. The distribution of baseline characteristics in individual cohorts was similar to the target population (Table 1).
Baseline characteristics.a
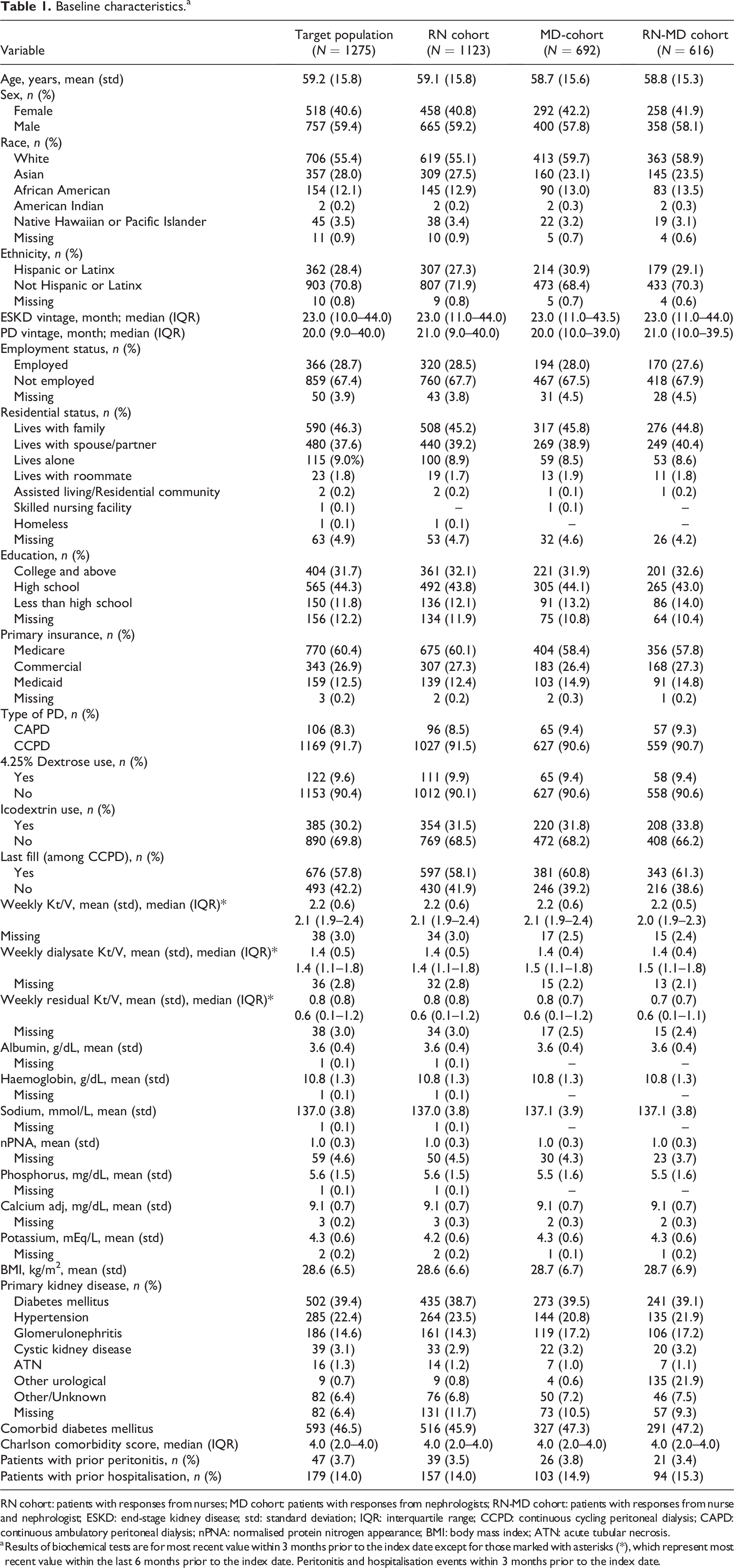
RN cohort: patients with responses from nurses; MD cohort: patients with responses from nephrologists; RN-MD cohort: patients with responses from nurse and nephrologist; ESKD: end-stage kidney disease; std: standard deviation; IQR: interquartile range; CCPD: continuous cycling peritoneal dialysis; CAPD: continuous ambulatory peritoneal dialysis; nPNA: normalised protein nitrogen appearance; BMI: body mass index; ATN: acute tubular necrosis.
a Results of biochemical tests are for most recent value within 3 months prior to the index date except for those marked with asterisks (*), which represent most recent value within the last 6 months prior to the index date. Peritonitis and hospitalisation events within 3 months prior to the index date.
Patients identified as low risk were more likely to identify as Asian, be employed, be married, have higher residual kidney clearance, have a higher serum sodium and higher serum albumin. Patients identified as high risk were more likely to be male, have a higher serum phosphate, have diabetes as the primary cause of kidney disease, have atherosclerotic heart disease, have a higher Charlson comorbidity index score, have a recent episode of peritonitis or hospitalisation in the last 3 months (Online Supplemental Tables 2 and 3).
Primary outcome: Performance of the PDSQ in identifying patients at high risk of transfer to HD
In the RN cohort, 18 (11%) versus 29 (3%) patients transferred to HD in the high- versus low-risk group, respectively (HR: 3.92 (95% CI: 2.17–7.05)). In the MD cohort, 14 (12%) and 12 (2%) patients transferred to HD in the high- and low-risk groups, respectively (HR: 5.56 (95% CI: 2.65–11.67)). In the RN-MD cohort, 8 (19%), 2 (5%), 4(7%) and 12 (3%) patients transferred to HD when identified as high risk by both, high risk by the nurse only, high risk by the nephrologist only and low risk by both the nurse and nephrologist (HR: 8.56 (95% CI: 3.50–21.00), HR: 1.80 (0.40–8.03), HR: 2.92 (0.94–9.05), HR: 1.0 (Reference group)), respectively (Figure 3).
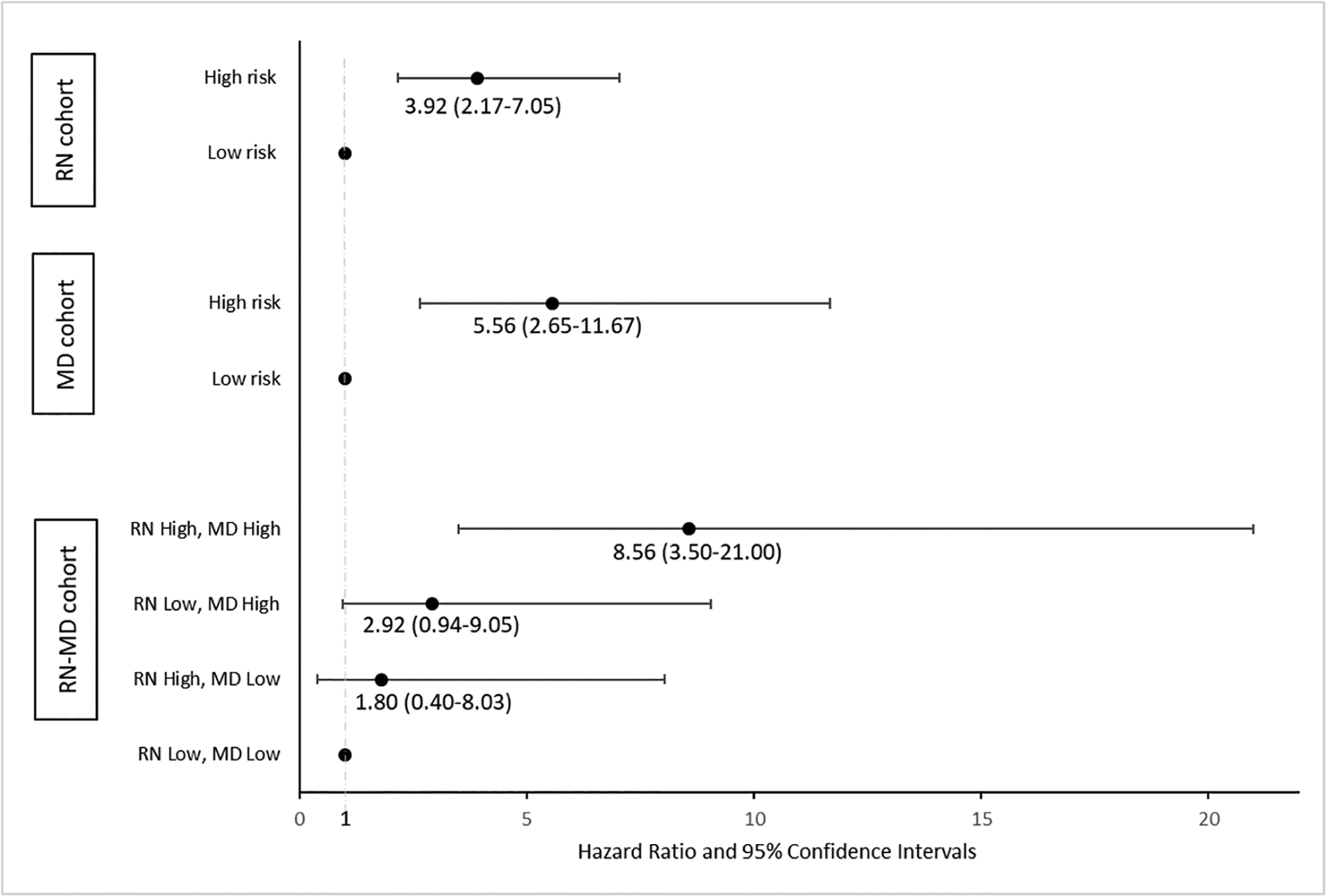
Hazard ratios for the RN, MD and RN-MD cohort.
In the RN and MD cohort, performance varied during the follow-up period. The c-statistic was 0.77 and 0.82 at 60 days and dropped over time to 0.64 and 0.65 and 0.63 and 0.68 at day 90 and day 170, respectively. Performance of the PDSQ had a high negative predictive value (>99%) but a low positive predictive value throughout the course of follow-up, indicating the best utility for excluding patients from the high-risk group. Other performance metrics in all cohorts are further described in Online Supplemental Table 5.
In the sensitivity analysis that followed each patient from the actual date of the PDSQ response (no unified index date and no excluded events), the HR of transfer to HD was 5.54 (95% CI: 3.23–9.31) and 7.21 (3.69–14.08) for the RN and MD cohorts, respectively. The c-statistic at day 60 for the RN and MD cohort was 0.82 and 0.83, respectively. At day 170, it dropped to 0.68 and 0.70 for the RN and MD cohorts. The rest of the performance metrics for the sensitivity analysis is shown in Online Supplemental Table 6.
Secondary outcomes
Among other outcomes in the RN cohort, patients identified as high risk had a higher incidence rate of death or discontinuing dialysis and a higher event rate of hospitalisation compared to the low-risk group (1.9 vs. 0.8 per 100 patient-months (incidence rate ratio (IRR) = 2.34, 95% CI: 1.34–4.09, p = 0.003) and 18.2 vs. 8.7 per 100 patient-months (IRR = 2.09, 95% CI: 1.75–2.50, p < 0.001, respectively)) – Table 2. The event rate of peritonitis and incidence rate of kidney transplant was not significantly different in the high- versus low-risk groups (p = 0.74 and p = 0.24) – Table 2.
The number and percentage of death/discontinued dialysis, hospitalisation, peritonitis and transplant events for the high-risk and low-risk groups in the follow-up period.
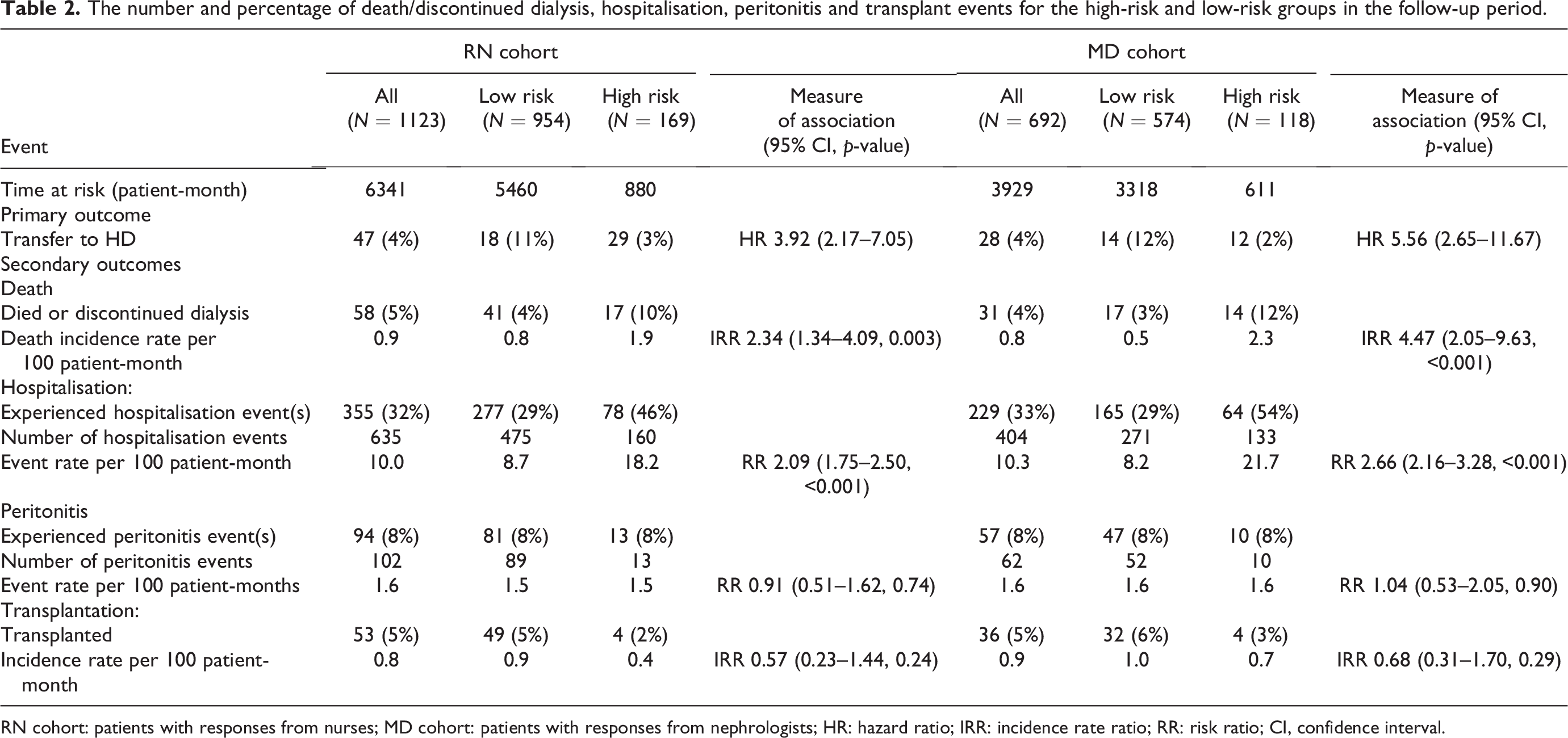
RN cohort: patients with responses from nurses; MD cohort: patients with responses from nephrologists; HR: hazard ratio; IRR: incidence rate ratio; RR: risk ratio; CI, confidence interval.
Out of the 47 transfers to HD in the RN cohort, the proportion of patients starting HD with permanent access (AVF/AVG) was similar in the high- and low-risk groups (44% vs. 48%, p = 0.80). Only 3 (6%) patients transferred directly to home HD – Online Supplemental Table 7. Findings in the MD cohort were similar – see Table 2 and Online Supplemental Table 7.
Discussion
The PDSQ identified a subset of patients to be at high risk for transfer to HD. During the follow-up period, this smaller group had a four- to six-fold higher risk of transfer to HD. In addition, patients identified as high risk had a higher rate of death or discontinuing dialysis and a higher rate of hospitalisation. Factors associated with assignment to the high-risk group included atherosclerotic heart disease, lower serum albumin, prior hospitalisation and prior peritonitis, indicating face validity of the tool.
Several prediction models exist that identify patients at risk of transfer to HD in incident PD patients. 1,2,8 These models have different definitions of transfer to HD and use different variables as predictors. 1,2,8 The leading causes of transfer to HD in PD patients are infections, PD catheter-related issues and small molecule solute or urea clearance 14,15 all of which cannot be captured prior to a patient beginning on PD. Nochaiwong 9 describes a model that predicts transfer to HD following peritonitis in PD patients, but this requires repeated dialysate leukocyte counts (on days 3–4 and day 5) which is not routinely practiced.
The PDSQ is a simple point of care question that can be used for all PD patients regardless of vintage. The burden of dialysis care and psychosocial issues often contribute to attrition 16 but are poorly captured in the EHR and not accounted for in any of the available prediction models. In contrast, the PDSQ allows a clinician to use all available conscious and unconscious information about a patient to form an assessment of the risk of transfer to HD that may not be captured in the EHR.
We assessed the PDSQ as obtained from nephrologists and from nurses to compare performance and balance it against logistics of obtaining responses. It was not possible to control response collection from the nephrologist and the nurse for a given patient to happen at the same time. To limit the possibility of clinical events occurring in the gap between responses for a given patient, we limited the window of PDSQ response collection to one month. Furthermore, the distribution of the length of time between responses from the nurse and the nephrologist for a given patient was random (as shown in Online Supplemental Figure 1), meaning that any effect would not have advantaged a group over the other. Nephrologist assessments of high risk using the PDSQ performed modestly better than the nurses in predicting transfer to HD over 6 months. This perhaps reflects additional clinical information the nephrologist had access to by caring for the patients in settings outside the dialysis centre as compared with nurses. However, nephrologists’ assessments were more challenging to obtain, and ease of response collection is an important factor when planning quality improvement interventions in dialysis centres.
The performance metrics of the PDSQ showing a high negative predictive value but a low positive predictive value is related in part to the low event rate, and these results render it most suitable for screening. The tool identifies a smaller group that can then be targeted with further workup and interventions. Even among the false positives, we showed that identified patients were at higher risk of other poor outcomes. The performance metrics showed superior performance of the PDSQ over the short-term period of 60 days and a decline of performance over time. This is an expected finding, as events take place over time that change the risk status of the patient. This finding may indicate that in practice, PDSQ may have to be repeated after 2 or 3 months to re-evaluate the risk profile. The highest performance of the PDSQ was observed in the early period in the sensitivity analysis, including patients with early outcomes. Our interpretation of this is that some patients showed features indicating eminent transfer to HD. We excluded these patients from the primary analysis as they were likely too obvious, and there was too short a period for a provider-level intervention beyond what the clinical team already do for these patients.
The rates of peritonitis were similar in the high- and low-risk groups. In our cohort, the rate of peritonitis was lower (0.180 patient-years) compared to the published literature (0.303 patient-years). 17 This is an interesting finding as peritonitis is often cited as the most common reason for transfer from PD to HD. The lack of association of a high-risk prediction of transfer to HD by PDSQ with a peritonitis event may reflect a difference in the factors that lead to these two separate clinical events.
We assessed patient readiness for transfer to HD by the presence of permanent vascular access or transfer to another home modality (home HD), and we found no difference between the high- and low-risk groups. The use of AVF/AVG at the time of transfer to HD was higher than what has been reported in cohorts from Canada 5 and Australia. 2 Only three patients transferred directly to home HD instead of in-centre HD, highlighting the need to improve transitions between home modalities for suitable patients.
Findings from this study have to be interpreted with recognition of some limitations. The study was conducted during the COVID-19 pandemic which could have potentially influenced patients’ well-being and psychosocial factors, which may in turn could have impacted patient outcomes. Provider data indicate that hospitalisation rates and transfer from PD to HD did not change in the pre- to post-pandemic years, but mortality rates increased from 0.7 to 0.9 per 100 patient months in 2019 and 2021, respectively (unpublished internal data). However, this pandemic effect was evenly distributed across all the cohorts and patient populations. Responses were collected over 1 month, which could have introduced disagreement between responses and altered predictions. However, the distribution of the nurse and nephrologist responses over time was randomly distributed, not favouring one group versus the other (Online Supplemental Figure 1). Patients were not followed after transfer to HD which could have influenced measures of secondary outcomes such as hospitalisation and death. Despite inclusion of all eligible patients from centres from multiple states across the country, generalisability may be limited by the fact that all patients were under the care of one dialysis provider. We did not investigate the causes of transfer to HD, limiting our ability to explore, for example, whether any events were amenable to interventions. Although the risk of selection bias cannot be excluded, nurses and nephrologists who participated provided responses for all their patients. Moreover, the distribution of baseline characteristics and frequency of outcomes was similar in the study groups and the target population, lowering this concern.
The strengths of the study included the prospective observational design of the study, large sample size from multiple centres across three states, unselected patients, varying dialysis vintage, low proportion of loss to follow-up and minimal missing data.
We see the PDSQ as a point-of-care screening tool to identify high-risk patients to implement targeted interventions for better utilisation of limited resources. Future studies need to examine whether the PDSQ can help target interventions to prevent patients from transferring from PD to HD when clinically appropriate. In addition, the PDSQ may help care teams improve transitions from PD to HD by allowing for vascular access and other care planning for high-risk patients. It may also offer the opportunity to ensure high-risk PD patients are aware of home HD and to allow for greater use of this modality in place of HD when transitions occur.
Conclusion
The PDSQ is a simple point of care tool which can be used by clinicians to identify patients at high risk for transfer from PD to HD over 6 months.
Supplemental material
Supplemental Material, sj-docx-1-ptd-10.1177_08968608231214143 - Predicting transfer to haemodialysis using the peritoneal dialysis surprise question
Supplemental Material, sj-docx-1-ptd-10.1177_08968608231214143 for Predicting transfer to haemodialysis using the peritoneal dialysis surprise question by Ayesha Anwaar, Sai Liu, Maria Montez-Rath, Hatsumi Neilsen, Sumi Sun, Graham Abra, Brigitte Schiller and Wael F Hussein in Peritoneal Dialysis International
Footnotes
Acknowledgement
We would like to thank all the nephrologists and nurses who participated by responding to the peritoneal dialysis surprise question.
Author contributions
Each author contributed important intellectual content during manuscript drafting or revision and agrees to be personally accountable for the individual’s own contributions and to ensure that questions pertaining to the accuracy or integrity of any portion of the work, even one in which the author was not directly involved, are appropriately investigated and resolved, including with documentation in the literature if appropriate.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AA was a grant recipient from Satellite Healthcare during the conduct of the study; HN is an employee at Satellite Healthcare; SS is an employee at Satellite Healthcare; GA is an employee at Satellite Healthcare and Baxter honoraria; BS was an employee at Satellite Healthcare during the conduct of the study, BOD Unicycive and Advisor Akebia; and WH is an employee at Satellite Healthcare and advisory board for GSK.
Ethical approval
The study was approved by an institutional review board (WCG Aspire, IRB #20203611) with waiver of consent.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported and run by Satellite Healthcare and did not receive any external funding.
Informed consent to participate
Full waiver of consent.
Informed consent to publish
NA
Trial registration
ClinicalTrials.gov Identifier: NCT04666844
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
