Abstract
Vancomycin has been reported to cause vancomycin flushing reaction (VFR), a hypersensitivity reaction that mostly occurs after intravenous administration. The incidence of VFR in a patient receiving intraperitoneal vancomycin is rare. We report a case of a female peritoneal dialysis (PD) patient with a PD-related peritonitis who developed VFR after intraperitoneal administration of 2000 mg vancomycin. Seventy-five minutes after instillation, she developed flushing, a pruritic erythema on the upper body and swelling of the lips. Blood results revealed a vancomycin plasma concentration of 54.8 mg/L and a normal tryptase level. During a relapse of her PD-related peritonitis, vancomycin was successfully reintroduced in a 50% reduced dose. No symptoms of VFR developed, and the corresponding vancomycin plasma concentration was 33.6 mg/L. Intraperitoneal treatment was continued with 500 mg vancomycin every 2–3 days with frequently measured, adequate trough levels ranging from 15-22 mg/L. This case illustrates the risk factors for the development of VFR after intraperitoneal administration of vancomycin, namely a high and concentrated loading dose together with a low body weight, a fast peritoneal transport state and peritonitis. Reintroduction of vancomycin after occurrence of VFR is safe, but a lower loading dose or a slower instillation rate is recommended.
Keywords
Introduction
Vancomycin is an antibiotic used in peritoneal dialysis (PD) patients for infections with gram-positive microorganisms.
1
In many regions, it is more frequently used than first-generation cephalosporins, due to the increasing incidence of
VFR is also described after therapy with oral vancomycin, although far less common than after intravenous administration, since oral administration normally does not result in systemic absorption. 4 –6 VFR after intraperitoneal administration is even more uncommon, with only a few described cases in children and adults. 7 –10
Here, we report a case of an adult PD patient with a PD-related peritonitis who developed VFR after intraperitoneal administration of vancomycin.
Case description
A 33-year-old woman of 56.6 kg (body mass index 20.3) was treated with continuous ambulatory peritoneal dialysis (CAPD) for 1 year due to graft failure after a prior kidney transplantation. She had a fast small solute peritoneal membrane transport state and yet, a residual kidney function of 3–4 mL/min. She presented with a relapsing PD-related peritonitis due to
At home, 75 min after the instillation, she developed flushing and a pruritic rash of the face, neck and shoulders with swelling of the lips. She was seen at the emergency room 60 min after the start of the symptoms with stable vital signs. Her abdomen with vancomycin-loaded PD effluent was drained, and 2 mg of clemastine and 8 mg of dexamethasone were given orally. Another 120 min later, blood samples were taken for determination of vancomycin and tryptase levels.
After an observation period of 240 min in which symptoms improved, she was discharged with intraperitoneal cefazolin for 21 days. Blood results revealed a vancomycin plasma concentration of 54.8 mg/L and a tryptase level of 9.5 µg/L (normal <11.4 µg/L). The latter and the fact that she was vancomycin-naive suggested VFR instead of an IgE-mediated anaphylactic reaction.
Fourteen days after completion of therapy with cefazolin, she presented with a relapse of PD-related peritonitis. It was decided to reintroduce vancomycin with a 50% dose reduction, supported by predictions of pharmacokinetic modelling software. Therefore, she received a loading dose of 1000 mg vancomycin in 1500 mL PD solution and was clinically observed for 3 h in which no symptoms of VFR occurred. The vancomycin plasma concentration was 33.6 mg/L 120 min after intraperitoneal administration. Treatment was continued with 500 mg intraperitoneal vancomycin every 2–3 days with frequently measured, adequate trough levels ranging between 15 mg/L and 22 mg/L. The patient gave her consent for the anonymous publication of her case.
Discussion
In this case report, we present a PD patient with flushing, erythema and pruritus following intraperitoneal administration of vancomycin. She was clinically diagnosed with VFR and was successfully rechallenged with reduced doses of vancomycin.
Published literature describing similar cases of VFR after intraperitoneal administration of vancomycin are scarce, outdated or incomplete, with no reported cases in adults in the past 30 years. 9,10 An overview of this literature is summarised in Table 1. Search for literature was conducted in PubMed using the term ‘vancomycin flushing reaction’ or its synonyms or abbreviations, in combination with ‘intraperitoneal’. Relevant articles were also searched for additional literature by cross-referencing. Symptoms of VFR in published literature were associated with a fast instillation rate and rapid increase in blood concentration, a large administered vancomycin dose and the presence of an inflamed peritoneum. 7,9,10
Described cases of vancomycin flushing reaction after intraperitoneal vancomycin.
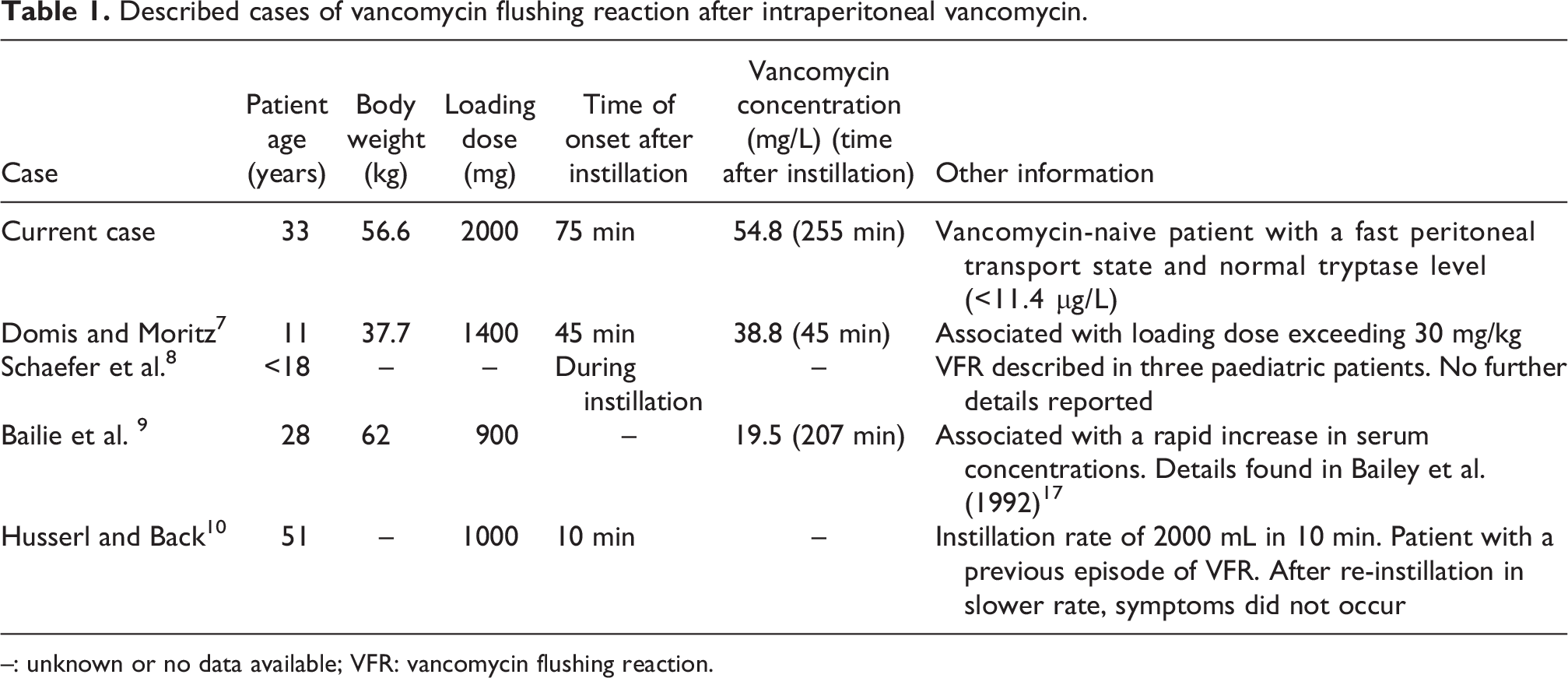
–: unknown or no data available; VFR: vancomycin flushing reaction.
Our patient had a vancomycin plasma concentration of 54.8 mg/L after a dwell time of only 135 min, whereas duration of the dwell is associated with systemic vancomycin exposure and the time to therapeutic plasma concentration. 11 The blood sample was taken 120 min after drainage of intraperitoneal vancomycin, and the herein measured vancomycin plasma concentration was much higher than reported in literature. 11 –13 In 10 patients with PD-related peritonitis, the mean peak plasma concentration after a 6-h dwell of 2000 mg intraperitoneal vancomycin was 39.7 ± 8.1 mg/L. 12 In this study, mean plasma concentrations decreased to 35.0 ± 7.5 mg/L 2 h after ending the dwell and exchange with a vancomycin-free PD fluid. This suggests that the vancomycin plasma concentration in our patient must have been even higher at time of drainage of the PD fluid, also considering her low but preserved residual kidney function.
Several factors may have contributed to the high vancomycin plasma concentration and the occurrence of VFR in our patient. First, she received a loading dose of 2000 mg vancomycin which was not adjusted for her low body weight and, therefore, low absolute volume of distribution. A recent update of the International Society for Peritoneal Dialysis guideline recommends an intermittent dose of 15–30 mg/kg vancomycin every 5–7 days for CAPD patients but also states that optimal dosing of intraperitoneal vancomycin is unknown. 1 Our patient received an intraperitoneal vancomycin dose of 35.3 mg/kg. Second, our patient had a fast small solute peritoneal membrane transport state. Because of the increased transport of solutes, the osmotic gradient disappears more quickly and this may have contributed to a rapid diffusion of vancomycin across the peritoneal membrane. This was probably further promoted by an increased permeability and capillary blood flow during peritonitis and the high vancomycin concentration in the PD solution (1333 mg/L vancomycin). Moreover, the bioavailability of vancomycin in patients on CAPD with peritonitis is increased compared with patients without peritonitis (70–91% vs. 50%). 13
As tryptase is elevated within minutes, peaks after 1–2 h and gradually returns to normal within 6–24 h after an IgE-mediated anaphylactic reaction, it is an important parameter to measure shortly after start of hypersensitivity symptoms. 14 However, it must be noted that tryptase may be elevated in non-immune-mediated hypersensitivity reactions as well. Therefore, this marker should be used in combination with other clinical characteristics to distinguish between a real type I allergy and VFR. 15
VFR is a clinical diagnose in which vancomycin can be safely reintroduced under close clinical observation. It should be considered to administer a loading dose not exceeding 30 mg/kg and to use modelling software for estimation of a patient’s pharmacokinetic parameters. In our case, rechallenge of vancomycin in 50% reduced dosages resulted in a VFR-free treatment with adequate trough levels. In addition, antihistamines should be considered as pre-treatment, as they have been shown to be protective against the occurrence of VFR. 16
Conclusion
VFR is a rare complication after intraperitoneal administration of vancomycin. Risk factors are a high and concentrated loading dose, a low body weight resulting in a low volume of distribution, a fast peritoneal transport state and peritonitis. Nephrologists and dialysis nurses should be aware of this reaction and understand its pathophysiology. It is safe to reintroduce vancomycin under close clinical observation after an episode of VFR. However, dose reductions or a slower instillation rate is recommended, depending on the patient’s risk factors.
Footnotes
Acknowledgement
None.
Author contributions
JEM wrote the first draft and the revised version of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the (revised) manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
University Medical Centre Utrecht does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent to participate
Not applicable. The patient received routine care.
Informed consent to publish
Written informed consent was obtained from the patient for their anonymised information to be published in this article.
