Abstract
Peritonitis is the most common complication in patients undergoing peritoneal dialysis (PD). Peritoneal dialysis-related peritonitis caused by Brucella species has been reported in only 7 patients before. Here, we report a further case of Brucella peritonitis. This patient was successfully treated with both intraperitoneal and prolonged oral antibiotics, without removal of the PD catheter. We review relevant literature and make recommendations for the diagnosis and treatment of Brucella PD-related peritonitis from the cumulative published clinical experience.
Keywords
Case Report
A 54-year-old female PD patient presented with a 1-day history of abdominal pain, nausea, vomiting, diarrhea, fatigue, anorexia, bilateral knee pain, cloudy PD effluent, and ultrafiltration decrease. Upon enquiry, she reported eating beef at a restaurant the day before the onset of symptoms and performing a single PD exchange without using her usual sanitary measures while at the restaurant.
Her past history was of end-stage kidney failure from chronic glomerulonephritis, and a history of PD for 11 years. She had no other medical history of note. Her course of PD had been rather quiet, with only 1 previous peritonitis episode due to Streptococcus salivarius 2 years earlier. Her PD regimen at the time of presentation was continuous ambulatory PD (CAPD), using a single 2-L exchange of 1.5% and 4 times 2-L exchanges of 2.5% dextrose per day.
Physical examination showed her body temperature to be 36.2°C, blood pressure 150/80 mmHg. The patient had umbilical tenderness during palpation and slight bilateral lower-limb edema. Other examination results were unremarkable. Her PD catheter exit site was normal. Her full blood count showed a white blood cell (WBC) count of 17.16 x 109/L with 86.2% neutrophils, a hemoglobin of 102 g/L, and platelet count of 168 x 109/L. Her serum biochemistry showed a serum potassium of 2.97 mmol/L, serum creatinine of 1,056 μmol/L, and electrolytes and liver function tests that were otherwise normal.
Her PD effluent was turbid, with WBC 1,950/μL with 87% neutrophils. Gram stain of the peritoneal fluid did not show any microorganisms. The patient was diagnosed as having PD-related peritonitis and received outpatient empirical antibiotic treatment with ceftazidime 1 g daily, and vancomycin 1 g daily for the first 2 days, and then 0.5 g intraperitoneally every 2 days. The effluent WBC decreased to 351, 207, and 170/ μL on the 3rd, 5th, and 7th days, respectively, with neutrophil predominance, and was slower to resolve than expected. On day 7, the PD effluent culture from the time of her admission was reported as growing Brucella spp. After involvement of the infectious disease service, we adjusted treatment to amikacin 200 mg and levofloxacin 200 mg once a day intraperitoneally, together with oral doxycycline 100 mg twice a day and rifampicin 600 mg daily. Her PD catheter was not removed, and PD was continued as usual.
On the 8th day, her blood WBC was 9.85 x 109/L with 70.1% neutrophils, her erythrocyte sedimentation rate (ESR) was 97 mm/h, her C-reactive protein (CRP) 28 mg/L, serum potassium 3.07 mmol/L, and serum creatinine 860 μmol/L. On the 6th day after modifying the antibiotic regimen, the effluent WBC decreased to 9/μL, and there was clear evidence of ultrafiltration recovery. Levofloxacin and amikacin treatments were used for a total of 3 weeks, and then modified to oral minocycline, rifampicin, and levofloxacin for a total treatment duration of 18 weeks.
The patient's progress was closely monitored during follow-up, with frequent clinical assessments as well as measurement of effluent WBC, nutritional parameters, ultrafiltration, serum potassium, liver transaminase, etc. She was reviewed every 6 weeks by the infectious disease specialists. Upon completion of treatment, the PD effluent was clear, and her effluent WBC was 4/μL. No recurrence was observed after 10 months follow-up.
Discussion
Brucellosis is a worldwide epidemic infectious disease, which is hyperendemic in the Mediterranean Basin, the Arabian Peninsula, India, Mexico, Central and South America, and Turkey, where the prevalence is more than 10 per 100,000 of the population (3). In China, the epidemic has risen significantly since 1996 and the average incidence rate during 2015 – 2016 was 3.81 per 100,000 of the population. There is significant regional variation, however, and the corresponding incidence rate from the northern provinces was higher at 7.7 per 100,000 (4).
Brucellosis is caused by Brucella, which can invade the body through skin and mucous membranes, the digestive tract, and the respiratory tract (5). As a zoonotic disease, the main sources of infection are sheep and cattle. The mode of transmission can be by inhalation (during slaughtering and meat packing), direct entry through skin wounds or mucous membranes (during routine animal care and dealing with excretions or products of conception), and ingestion of undercooked meat or unpasteurized dairy products, including milk, cheese, and ice cream (6). The disease is most commonly reported in male farm workers.
Systemic brucellosis can involve multiple organs and systems, such as the liver, spleen, lymph nodes, bone marrow, and so on (7). In the acute stage of brucellosis, the main manifestations include fever, fatigue, sweating, muscle and joint pain, and enlargement of the liver, spleen, and lymph nodes. In the chronic phase, joint damage can be seen. Brucella is a rare cause of peritonitis, sometimes occurring as a complication of systemic brucellosis (8 9 10 11 12-13), but also as spontaneous episodes in the setting of hepatic cirrhosis (14 15 16 17 18 19 20-21) or concurrently with neurobrucellosis in those with ventriculoperitoneal shunts (22 23-24).
Given the impaired peritoneal defenses in patients with PD, these patients have a higher risk of peritonitis than the general population (25,26). However, only 7 cases of Brucella PD-related peritonitis have been reported prior to ours: 6 cases from Turkey and 1 case from Saudi Arabia. We have summarized these cases in Tables 1 to 3. As expected, 6 of the total reported 8 cases were male, with the most frequently identified risk factors being ingestion of unpasteurized milk or cheese and direct contact with cattle and sheep.
In these cases, PD-related peritonitis due to Brucella presented in much the same way as PD-related peritonitis from other causes (Table 1). Abdominal pain and cloudy PD effluent were common, but fever and the specific clinical symptoms of brucellosis, such as fatigue and sweating, were not. There was usually an accompanying elevated WBC count, high ESR and CRP, and a high leukocyte count in PD effluent with either lymphocyte or neutrophil predominance. Peritoneal dialysis-related peritonitis due to Brucella has been reported presenting as either acute peritonitis or in a more relapsing indolent manner (27).
General Information and Clinical Findings in Reported Cases of Brucella PD-Related Peritonitis
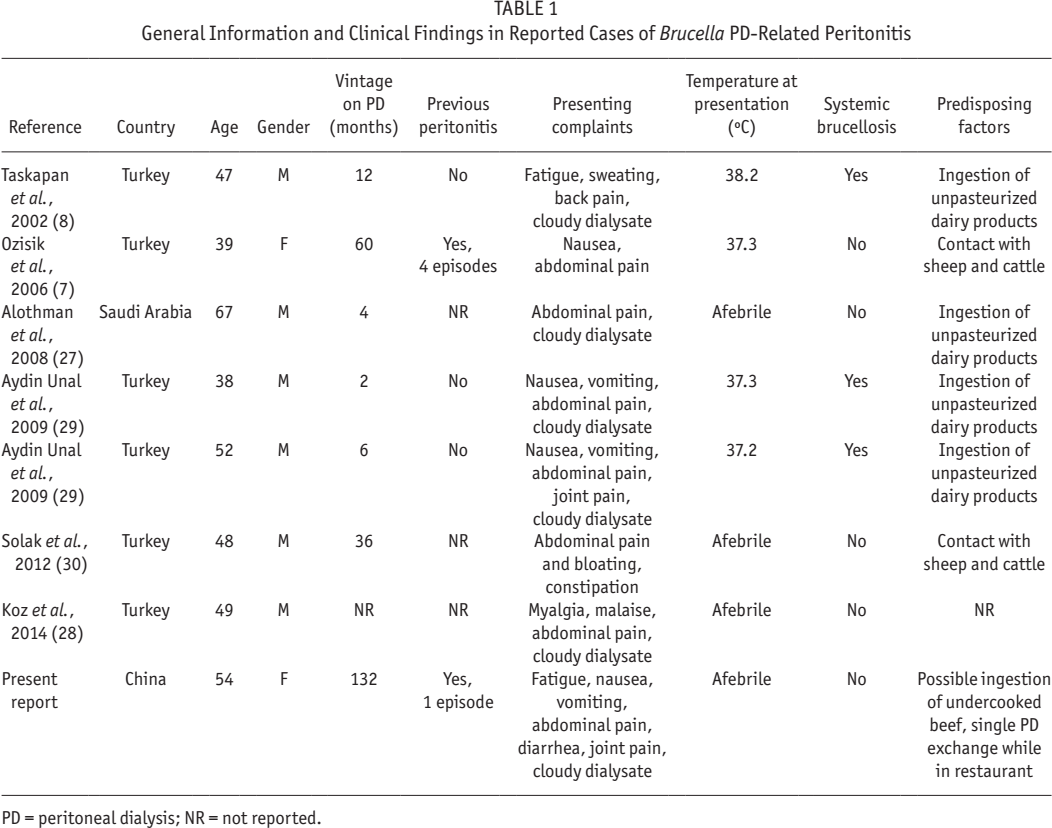
PD = peritoneal dialysis; NR = not reported.
There are 5 teaching points arising from our case. First, PD-related Brucella peritonitis can and does occur in the absence of systemic brucellosis. If Brucella growth is seen in both blood and effluent cultures, the microorganisms might have spread from the circulation to the peritoneum via the hematogenous route (8). Our patient's blood culture was negative, however. In cases such as ours, the isolated PD-related peritonitis is surmised to arise from direct entry of the bacteria though the PD catheter (7).
Second, infections due to Brucella can occur in patients without obvious environmental risk factors. In China, PD is widely used in rural settings, which obviously exposes patients to transmission of the bacteria from infected animals. However, the entire general population is susceptible to Brucella. In urban settings, exposure is most likely to occur from under-cooked meat and unpasteurized dairy products. In our patient, Brucella is surmised to have been transmitted from contaminated ingredients in her restaurant meal. Of note, freezing, smoking, drying, and pickling do not kill the bacteria that cause brucellosis, and routine restaurant precautions other than thorough cooking are not protective.
Third, the diagnosis of Brucella PD-related peritonitis is often delayed. Brucella grows slowly in culture medium, and identification of the bacteria usually requires extended culture duration (see Table 2). In addition, some of the new expanded spectrum cephalosporins show reasonable activity against Brucella and are an increasingly common part of empirical regimens for PD-related peritonitis. In the case of our patient, empirical antibiotics resulted in a partial response to treatment, which delayed our search for a less conventional cause and impeded our recognition of peritonitis from an atypical organism. One possible way of expediting diagnosis might be the use of Brucella immunocapture–agglutination tests on PD effluent, which are generally positive and might be preemptively ordered if the clinician's index of suspicions were high enough. Enzyme-linked immunosorbent assay (ELISA) tests for serum Brucella immunoglobulin M (IgM) and G (IgG) antibodies have also been used, although there is not enough experience to make a recommendation.
Laboratory Findings in Reported Cases of Brucella PD-Related Peritonitis
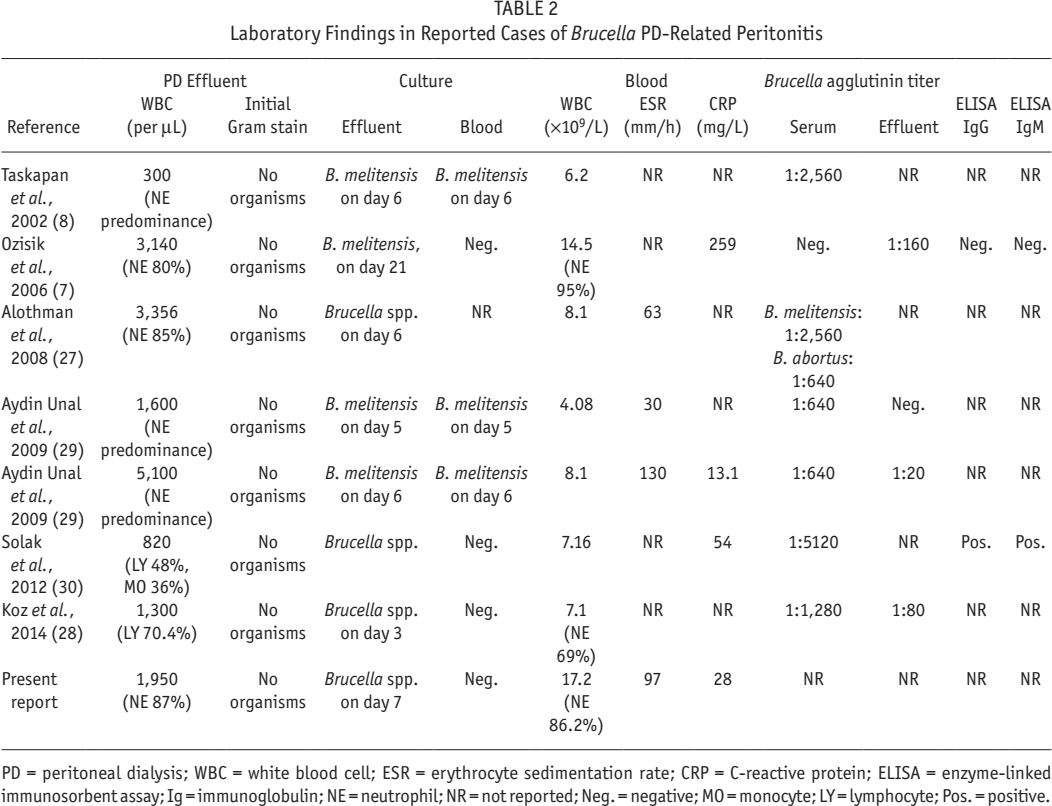
PD = peritoneal dialysis; WBC = white blood cell; ESR = erythrocyte sedimentation rate; CRP = C-reactive protein; ELISA = enzyme-linked immunosorbent assay; Ig = immunoglobulin; NE = neutrophil; NR = not reported; Neg. = negative; MO = monocyte; LY = lymphocyte; Pos. = positive.
Fourth, best results are seen when antimicrobial treatment is administered early, in combination therapy, with adequate dosing, and for several months (see Table 3). In the case of our patient with PD-related peritonitis, we administered antibiotics intraperitoneally for 3 weeks according to the ISPD guidelines, as would be done for any PD-related peritonitis. Logically, intraperitoneal antibiotics in the early stage will facilitate control of the local infection, and reduce the colonization or formation of PD-catheter biofilm, thereby preventing recurrence (28,31). Of note, we also administered standard treatment for systemic brucellosis, with oral administration of minocycline, rifampicin, and levofloxacin for a total of 18 weeks. It goes without saying that all decisions should be made with the input of infectious disease or microbiology specialists.
Treatment and Outcomes in Reported Cases of Brucella Peritonitis in PD
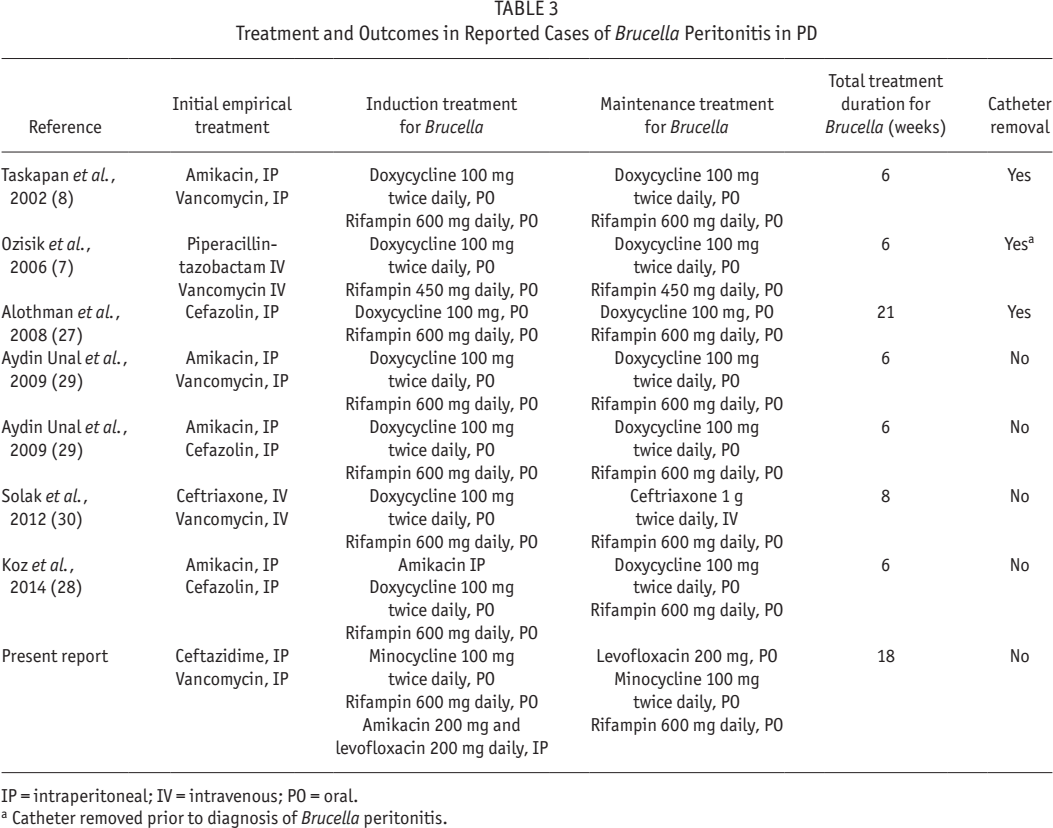
IP = intraperitoneal; IV = intravenous; PO = oral.
Catheter removed prior to diagnosis of Brucella peritonitis.
Finally, catheter removal is clearly not necessary for every case of Brucella PD-related peritonitis. In the earlier literature, catheters were often removed due to ineffective initial treatment or recurrence of peritonitis (7,27). In our case, treatment was effective without catheter removal, as was the case for all the other more recently reported cases (8,28–30). This experience suggests that catheter removal should be reserved for patients with severe local manifestations, and disease that is unresponsive or recurrent despite optimal treatment with IP antibiotics combined with a prolonged course of appropriate oral antibiotics.
In summary, Brucella PD-related peritonitis is rare but associated with excellent outcomes if appropriately treated. Clinicians should have a low index of suspicion in those with risk factors, especially if there is a delayed or incomplete response to empirical antibiotics. Diagnosis may require prolonged culture of PD effluent, or the use of serological tests. In our opinion, the keys to good outcomes are initial IP antibiotics followed by a suitably prolonged course of oral antibiotics, as guided by infectious disease or microbiological specialists. Removal of the PD catheter should be reserved for severe, unresponsive, or recurrent disease.
Footnotes
Huiping Zhao has received a research grant from Baxter. Mark R. Marshall is a fully employed by Baxter Healthcare Asia (Pty) Ltd. The other authors have no financial conflicts of interest to declare.
