Abstract
Background:
Intraperitoneal (IP) vancomycin is often first-line empiric therapy for peritoneal dialysis (PD) peritonitis; however, whether dosing should be adjusted for patient-specific characteristics remains unclear. We sought to identify factors associated with the day 3 vancomycin serum level in patients receiving vancomycin for PD peritonitis.
Methods:
Retrospective single-centre adult cohort of 58 patients with PD peritonitis treated with IP vancomycin between January 2016 and May 2022. Linear regression was used to examine the association between day 3 vancomycin level and candidate predictors including age, sex, weight, glomerular filtration rate (GFR), urea and creatinine clearance (total, residual, dialysate), PD modality, peritoneal solute transfer rate and initial vancomycin dose. Logistic regression was used to evaluate the likelihood of achieving a level (≥15 mg/L) associated with these predictor variables.
Results:
A 2-g loading dose was given in 51 cases, and 38 patients (66%) had a therapeutic day 3 level. Each 5 mg/kg increase in initial vancomycin dose was associated with a 1.38 mg/L (95% confidence interval 0.52, 2.23) increase in day 3 level. Each 1 mL/min increase in GFR was associated with a 0.29 mg/L decrease (95% confidence interval 0.05, 0.52) in day 3 level. The likelihood of achieving a therapeutic level was approximately four times higher with an initial dose of ≥25 mg/kg compared to <25 mg/kg (odds ratio 3.75, 95% confidence interval 1.05, 13.46).
Conclusions:
Following an average 2-g vancomycin loading dose for suspected PD peritonitis, one-third of patients were subtherapeutic on day 3. GFR and weight-based dosing were independently associated with day 3 vancomycin level, and their consideration could improve the likelihood of achieving an early therapeutic level.
This is a visual representation of the abstract.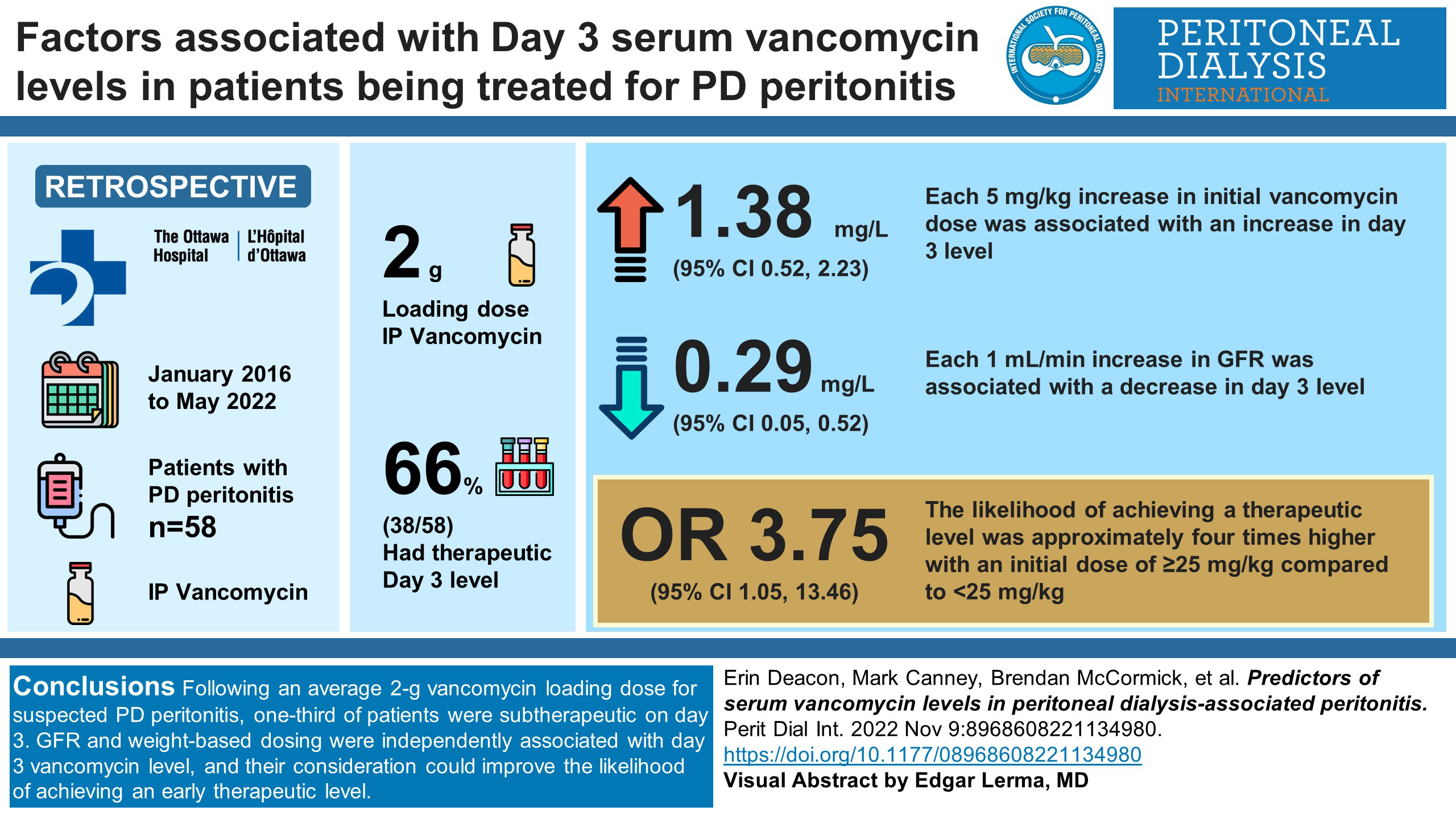
Keywords
Introduction
Peritonitis is a major complication of peritoneal dialysis (PD) and is associated with serious adverse outcomes including PD discontinuation, peritoneal membrane failure and death in severe cases. 1 –4 Depending on local experience with resistant Gram-positive organisms, intraperitoneal (IP) vancomycin is often used as first-line empiric therapy. Our unit switched to IP vancomycin as first-line empiric therapy in 2016 after observing several cefazolin treatment failures for coagulase-negative staphylococcus. 5 The choice of initial dose of vancomycin, the timing and frequency of therapeutic drug monitoring with serum vancomycin levels and the decision to repeat a treatment dose are largely based on clinical judgement, and dosing regimens vary between centres. 6 –8 The International Society for Peritoneal Dialysis (ISPD) 2016 guidelines suggest that the trough serum vancomycin level be maintained above 15 mg/L for the treatment of PD peritonitis. 9 However, optimal dosing of IP vancomycin remains unknown. Current ISPD guidelines for intermittent vancomycin dosing leave much to the discretion of the prescribing physician, suggesting 15–30 mg/kg every 5–7 days for continuous ambulatory peritoneal dialysis (CAPD) and 15 mg/kg every 4 days for automated peritoneal dialysis (APD). 10 Furthermore, the 2022 ISPD guidelines no longer indicate a target therapeutic vancomycin serum level. 10
The serum level of vancomycin during the treatment of an episode of PD peritonitis is likely dependent on a multitude of pharmacokinetic factors including peritoneal solute transfer rate, the PD prescription, residual kidney function and other patient characteristics such as weight. 11 However, there is a paucity of data on how vancomycin dosing should be adjusted to account for different pharmacokinetic factors at the individual patient level. This is important because a substantial number of patients continue to have variable vancomycin serum levels and their management necessitates frequent blood test monitoring, which inconveniences patients and adds costs to the healthcare system. Conversely, an improved understanding of the factors that contribute to vancomycin serum level could inform treatment decisions and potentially improve outcomes by helping patients to achieve target levels.
In this cohort of adult patients with PD peritonitis treated empirically with IP vancomycin, we sought to identify the relationship between patient factors and their day 3 vancomycin serum level.
Materials and methods
Design, setting and participants
This was a single-centre retrospective cohort of adult (>18 years) patients diagnosed with PD peritonitis as per the ISPD guidelines at the Ottawa Hospital from January 2016 to May 2022. Patients were included if (i) they received a loading dose of IP vancomycin for PD peritonitis, (ii) a serum vancomycin level was measured 3 days after the loading dose (the most common time frame) without an intervening additional vancomycin dose, (iii) adequacy assessment that included glomerular filtration rate (GFR), peritoneal solute transfer rate and weekly creatinine and urea clearance (total, dialysate and residual) performed within 6 months prior to the peritonitis episode. In the event of patients experiencing multiple episodes of PD peritonitis during the observation period, only the first event was included in the analysis. During the observation period, patients typically received a 2-g IP vancomycin loading dose to dwell for a minimum of 6 h (day fill added for both CAPD and APD) with repeat dosing every 2–5 days at the discretion of the treating physician based on the serum vancomycin level. Prior to June 2019, all relevant variables including patient demographics, laboratory tests, dialysis prescription and vancomycin dosing were collected from the Nephrocare® clinical information system. Beyond June 2019, the same data were obtained from the patients’ electronic health records (EPIC). The study was approved by the research ethics board of The Ottawa Hospital (approval number 20210456-01H).
Variable definitions
Pharmacokinetic factors potentially associated with day 3 vancomycin serum level were chosen based on prior literature and biological plausibility. Candidate variables included the initial vancomycin dose adjusted for weight (mg/kg), GFR (average of the creatinine and urea clearance, mL/min), age, sex, weight, height, PD modality (CAPD vs. APD), peritoneal solute transfer rate (within 6 months of peritonitis), weekly creatinine and urea clearance (total, dialysate and residual clearances in L/week). Peritoneal solute transfer rate was based on dialysate/plasma creatinine and coded during data collection as slow (0.34–0.49), slow average (0.50–0.64), fast average (0.65–0.80) or fast (0.81–1.03) 12 ; GFR was estimated using the average of the creatinine and urea clearance. Given the small patient numbers in some groups or lack of data, peritoneal solute transfer rate was grouped into three categories as average (slow average or fast average), fast or unknown. The initial vancomycin doses administered (g), date of initial (day 0) and subsequent doses were also collected. The primary outcome was the day 3 serum vancomycin level (mg/L).
Statistical analysis
The association between each predictor of interest and the day 3 vancomycin serum level was evaluated in a linear regression model, first unadjusted and then multivariable adjusted. In a secondary analysis, the likelihood of achieving a therapeutic day 3 vancomycin level (≥15 mg/L) was examined in a logistic regression model that included the same predictor variables. A second logistic regression model was conducted with vancomycin dose dichotomised as ≥25 mg/kg versus <25 mg/kg for easier clinical interpretation. Due to multi-collinearity, only one metric of kidney function was included in each model. GFR was chosen for the primary analysis because of its widespread availability. In a supplementary analysis, the models were repeated with total weekly creatinine clearance as the exposure variable instead of GFR. Continuous variables are reported as mean (standard deviation (SD)) and categorical variables as count (percent). Statistical analyses were performed using SPSS for Windows commercial software. 13
Results
Patient characteristics
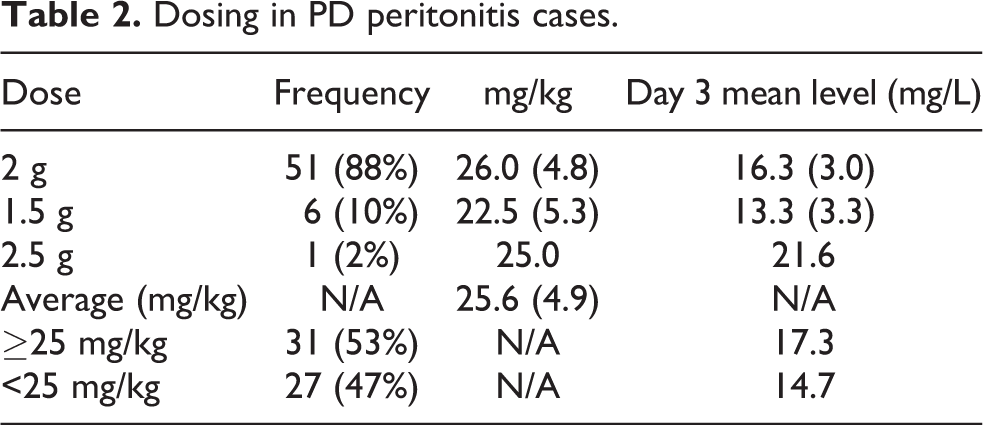
A total of 58 patients met the inclusion criteria (Figure 1). The characteristics of these patients are shown in Table 1. The mean (SD) age was 64 (14) years, the majority (72%) of cases were male and the most common cause of kidney failure was diabetic nephropathy. The mean (SD) eGFR was 4.5 (3.5) mL/min and only one patient had GFR = 0. While most characteristics were comparable between patients who achieved a day 3 vancomycin level that was greater or less than 15 mg/L, patients who had a level less than 15 mg/L had a higher mean weight, GFR and urea and creatinine clearance (total and residual components), and a greater proportion had a fast peritoneal solute transfer rate. Patients with a day 3 level less than 15 mg/L also had a lower mean vancomycin loading dose by weight. A 2-g loading dose was given in all but seven cases. The mean weight was higher in men than women. When adjusted for body weight, the mean (SD) vancomycin loading dose was 25.49 (4.89) mg/kg (Table 2). On average, women received a higher weight-based dose than men (27.67 mg/kg vs. 24.80 mg/kg, p = 0.04, Figure 2(a)).
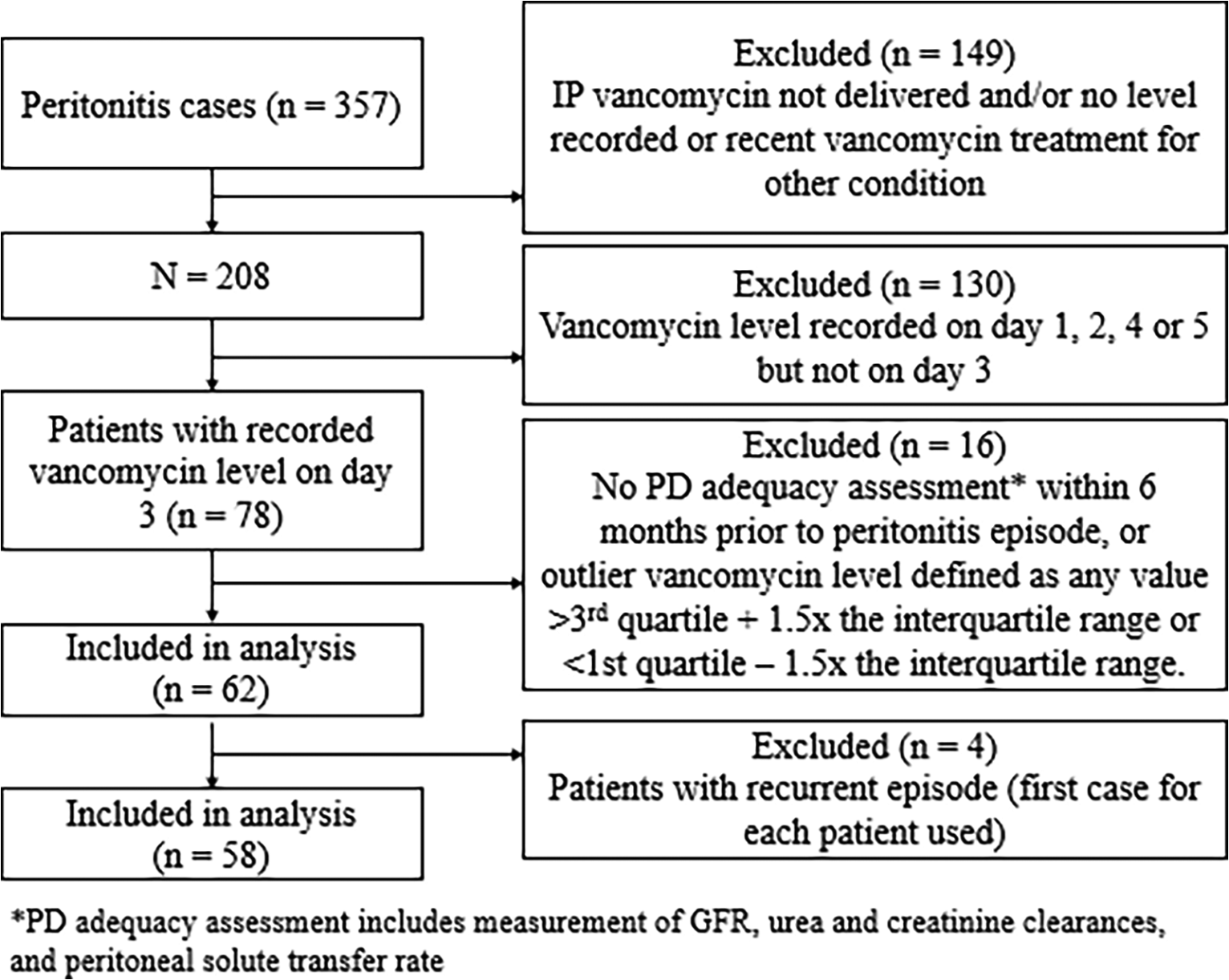
Flowchart of case ascertainment.
Characteristics of PD peritonitis cases included in study.a
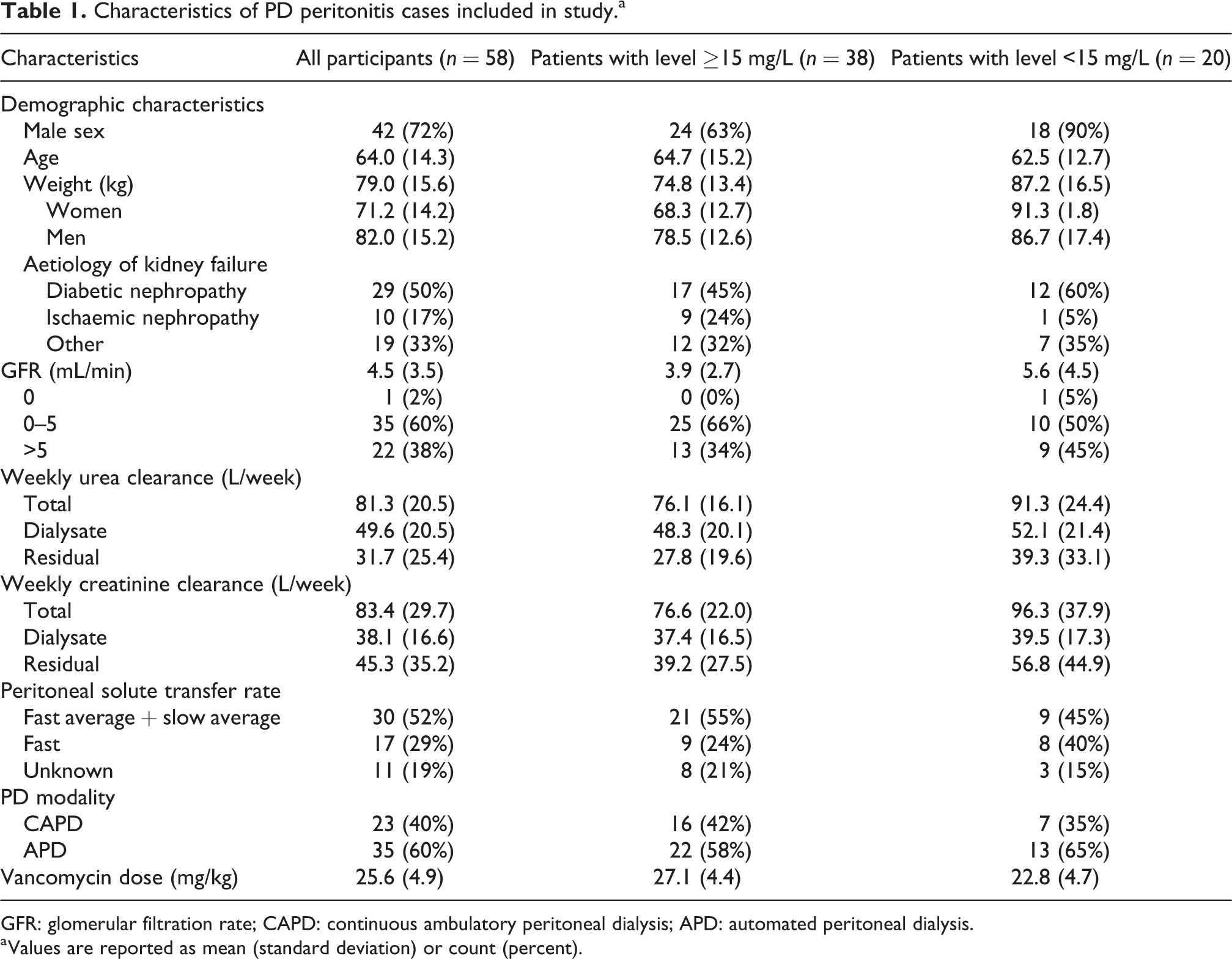
GFR: glomerular filtration rate; CAPD: continuous ambulatory peritoneal dialysis; APD: automated peritoneal dialysis.
a Values are reported as mean (standard deviation) or count (percent).
Dosing in PD peritonitis cases.
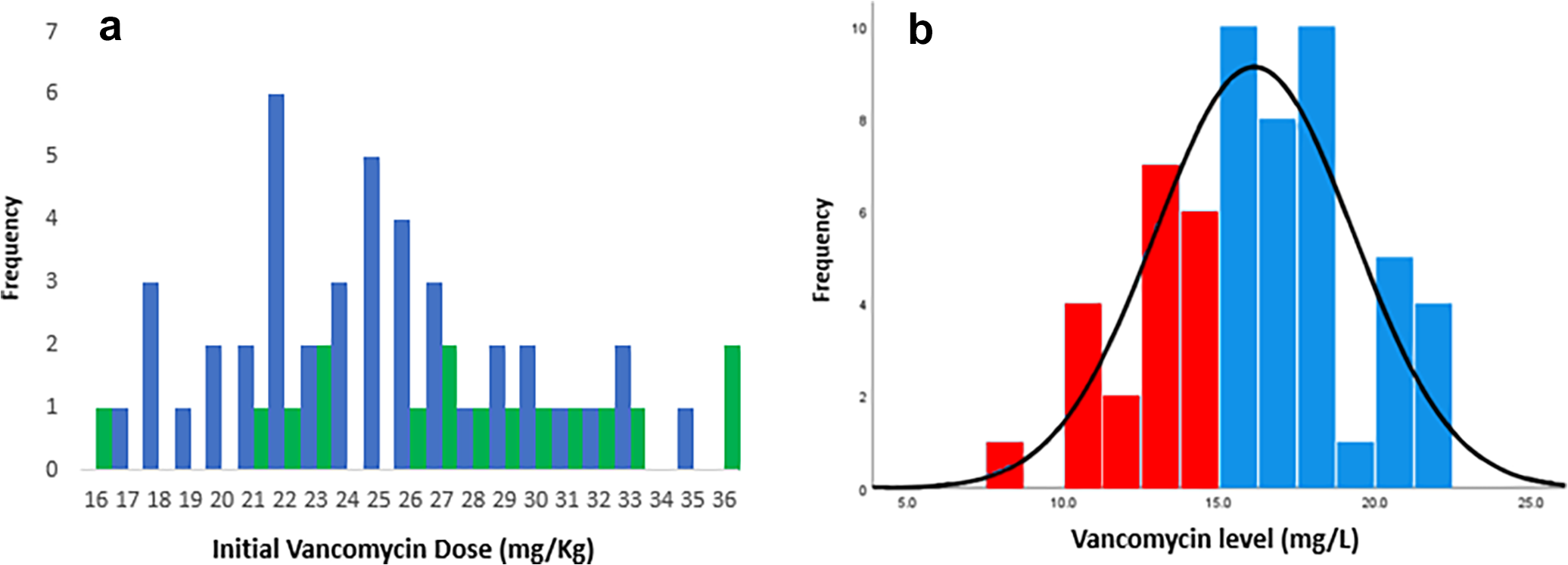
Distribution of vancomycin loading doses and day 3 levels. (a) Initial vancomycin dose (mg/kg) in men (blue) versus women (green). Dose rounded to nearest whole number. (b) Day 3 vancomycin level (mg/L); red < therapeutic level, blue = therapeutic level.
Factors associated with day 3 vancomycin serum level
Day 3 vancomycin serum level values were normally distributed with a mean (SD) of 16.13 (3.17) mg/L (Figure 2(b)). A total of 38 patients (66%) had a serum vancomycin level ≥15 mg/L on day 3; 20 patients (34%) were below this threshold.
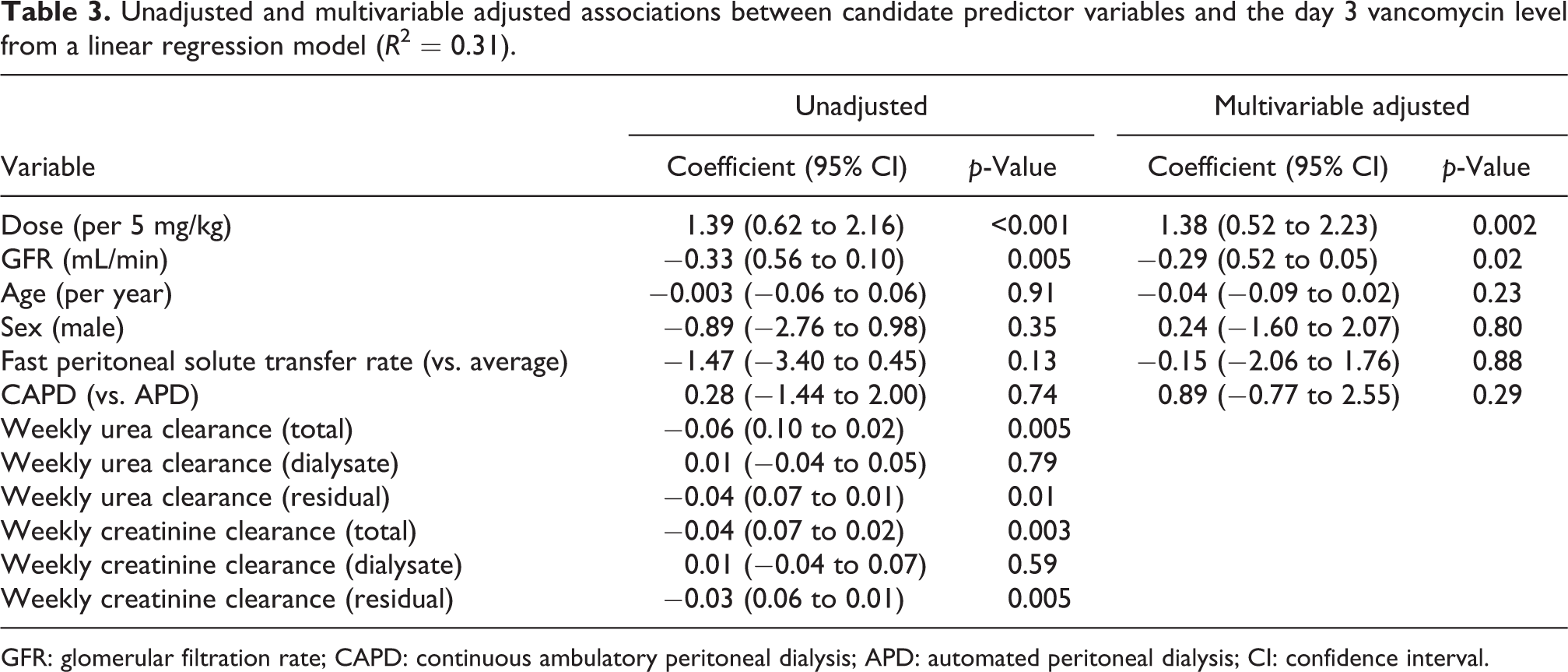
In an unadjusted analysis, higher vancomycin dose (mg/kg) was significantly associated with a higher day 3 vancomycin level. Higher eGFR, weekly urea clearance (total), weekly urea clearance (residual), weekly creatinine clearance (total) and weekly creatinine clearance (residual) were significantly associated with a lower day 3 vancomycin level (Table 3). Although not statistically significant, a lower day 3 vancomycin level was also observed for those with fast peritoneal solute transfer rate (compared to slow average and fast average combined). PD modality, sex, age and the dialysis component of urea and creatinine clearance were not found to be associated with day 3 vancomycin level.
Unadjusted and multivariable adjusted associations between candidate predictor variables and the day 3 vancomycin level from a linear regression model (R 2 = 0.31).
GFR: glomerular filtration rate; CAPD: continuous ambulatory peritoneal dialysis; APD: automated peritoneal dialysis; CI: confidence interval.
In a multivariable linear regression model, the initial dose of vancomycin and GFR were found to be independently associated with the day 3 serum vancomycin level (Table 3). Each 5 mg/kg increase in the initial loading dose was associated with a 1.38 mg/L increase (95% confidence interval 0.52–2.23, p = 0.002) in day 3 vancomycin level. Each 1 mL/min increase in GFR was associated with a 0.29 mg/L decrease (95% confidence interval 0.05–0.52, p = 0.02) in day 3 vancomycin level. PD modality, sex, age and peritoneal solute transfer rate were not found to be independently associated with day 3 vancomycin level. Similar correlations were observed in a multivariable regression using weekly creatinine clearance instead of GFR (Table S1).
Likelihood of achieving a therapeutic day 3 serum vancomycin level
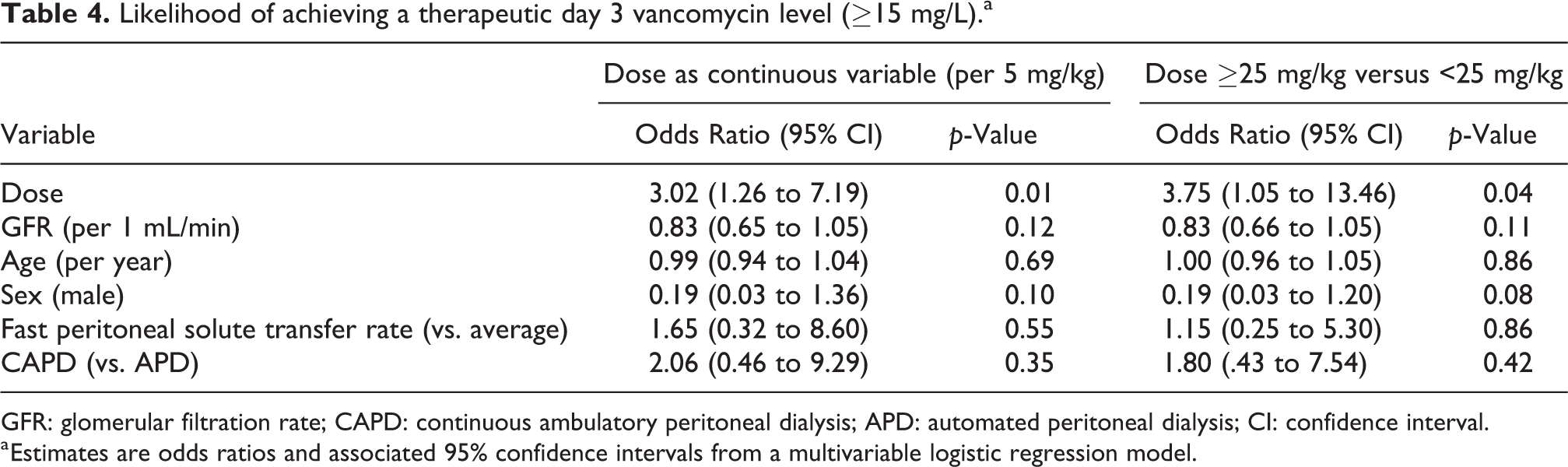
The factors associated with achieving a day 3 serum vancomycin level ≥15 mg/L are shown in Table 4. Each 5 mg/kg increase in initial dose was associated with a threefold increase in the likelihood of achieving a therapeutic level (odds ratio 3.02, 95% confidence interval 1.26–7.19, p = 0.01). The odds of achieving a therapeutic level were almost four times greater in those who received a loading dose ≥25 mg/kg versus <25 mg/kg (odds ratio 3.75, 95% confidence interval 1.05–13.46, p = 0.04). Similar outcomes were observed in a logistic analysis where weekly creatinine clearance (total) was used instead of GFR (Table S2).
Likelihood of achieving a therapeutic day 3 vancomycin level (≥15 mg/L).a
GFR: glomerular filtration rate; CAPD: continuous ambulatory peritoneal dialysis; APD: automated peritoneal dialysis; CI: confidence interval.
a Estimates are odds ratios and associated 95% confidence intervals from a multivariable logistic regression model.
Discussion
Vancomycin dosing schedules for PD peritonitis remain poorly studied and vary widely between centres. 6 –8 At our centre, most patients received a 2-g loading dose with subsequent levels and dosing in 2–5 days at the discretion of the treating physician based on clinical judgement, clinic hours and the patient’s ability to return for sampling. In the present study, most patients received their first level 3 days following almost universal dosing of 2 g, and 34% of cases had a subtherapeutic vancomycin level (<15 mg/L). Each 5 mg/kg increase in initial vancomycin dose was associated with a 1.4 mg/L increase in the day 3 vancomycin level, whereas each mL/min increase in GFR was associated with a decrease in the level by 0.29 mg/L. Providing a loading dose of 25 mg/kg or greater was associated with a 3.75-fold increase in the likelihood of achieving a therapeutic level. Taken together, these findings suggest that the incorporation of weight-based dosing and consideration of GFR, especially in patients with substantial residual kidney function, may help refine initial PD peritonitis treatment with vancomycin.
The notion that residual kidney function should be considered in vancomycin dosing was supported in the 2010 iteration of the ISPD guidelines, which suggested that for patients with significant residual function (GFR > 5mL/min), antibiotic dosing may need to be adjusted accordingly. 14 However, suggestions to modify dosing according to GFR were removed from the 2016 update based on limited evidence. 9 The significance of residual kidney function for vancomycin dosing has varied between studies. For example, a study of 263 Gram-positive or culture-negative PD peritonitis episodes treated with either cefazolin (74%) or vancomycin (23%) found that patients with higher creatinine clearance (>5 or >0–5 mL/min) had higher odds of treatment failure compared to anuric patients (0 mL/min). 6 These findings are consistent with a recent Peritoneal Dialysis Outcomes and Practice Patterns Study (PDOPPS) prospective cohort study of 954 Gram-positive or culture-negative PD peritonitis episodes, which found that higher residual kidney function was associated with higher odds of relapse or recurrent peritonitis. 15 These studies did not directly examine antibiotic levels in relation to residual kidney function, so lower levels in association with increased kidney function and higher odds of treatment failure can only be speculated. Contrary to the previous two studies, a study by Ma et al. of 61 Gram-positive or culture-negative PD peritonitis episodes found that residual kidney function was not associated with suboptimal day 5 serum vancomycin levels (n = 61). In that study, the residual kidney creatinine clearance was only 14.2 L/week/1.73 m2; almost 40% of participants were anuric. Although not statistically significant, anuria (defined as <100 mL urine output in 24 h) was associated with lower odds of short-term adverse outcomes in PD peritonitis. 7 The discrepancies in the significance of residual kidney function in the above studies may be related to different sample sizes, definition of anuria and measurement of residual kidney function. On balance, our results and those of the above studies suggest that patients at the upper range of residual kidney function may require higher (mg/kg) doses of vancomycin or shorter dosing intervals.
In our cohort where most patients received an initial 2-g dose, men received a lower weight-adjusted vancomycin dose (mg/kg) than women. Interestingly, however, men did not have a statistically significantly lower day 3 vancomycin level than women, showing the importance of other factors than just dose (mg/kg) that contribute to the day 3 level.
We did not find that faster peritoneal solute transfer rate assessed prior to the episode of peritonitis was associated with lower day 3 vancomycin serum level. It has been shown that vancomycin is more rapidly absorbed by an inflamed peritoneum, resulting in initially higher serum levels perhaps mimicking a fast peritoneal solute transfer rate membrane in patients with more severe peritonitis regardless of baseline transport status on the peritoneal equilibration test. 16 –18
Vancomycin serum trough levels have also been shown to vary with dialytic clearance by PD modality, where clearance of vancomycin may be slightly increased in APD (30% relative to total plasma clearance) compared to CAPD (20–25% relative to total plasma clearance). 11 A review by Lam et al. suggested that this effect may be attributable to the increased cycling that occurs during APD. 11 However, these studies are limited to non-infected patients, and APD studies are limited to intravenous vancomycin dosing. 11 In contrast, other studies have demonstrated that PD modality does not impact the clearance of other IP antibiotics (cefazolin, tobramycin, ceftazidime, gentamycin and rifampicin) during PD peritonitis. 6,19 In our study, although we identified a small increase in day 3 levels for patients on CAPD versus APD, this result was not statistically significant. In our programme, patients are treated with incremental PD such that CAPD and APD prescriptions vary significantly between patients, both in terms of volume of dialysate and dwell times. This variation in dialysis prescription may have impaired our ability to detect a difference if one truly exists.
It is important to note that the association between vancomycin serum levels and PD peritonitis outcome remains somewhat unclear. In one retrospective cohort study, 23% of patients with a serum vancomycin level >15 mg/L did not have a PD effluent level target of 4 mg/L (the Minimum Inhibitory Concentration (MIC) for some Gram-positive organisms). 20 Furthermore, vancomycin serum levels (<12, 12–24 and >24 mg/L) were not predictive of patient outcomes in another study. 21 In contrast, a more recent retrospective study found that patients who experienced adverse outcomes had significantly lower day 5 trough serum vancomycin level (8.4 vs. 12.5 mg/L). 7 These results are in agreement with those of another study in which the 4-week mean trough vancomycin levels were significantly higher in patients who were relapse-free (13.7 vs. 7.8 mg/L). 22 It is also important to note that although we have shown a much higher odds of achieving a therapeutic serum vancomycin level using a dose of greater than 25 mg/kg, there are theoretical concerns about kidney toxicity with very high serum levels. 23 The measured day 3 levels reflect a trough, while the peak level is not known. It is also possible that patients with greater residual kidney function achieve initially therapeutic vancomycin levels but experience a more rapid decrease in levels. Based on these inconsistencies in the literature and potential concerns about the effect of peak vancomycin level on residual kidney function, it has been suggested that smaller, more frequent doses of vancomycin may be preferable. 20 Future studies with monitoring of peak vancomycin levels and subsequent changes in residual kidney function are necessary to explore these concerns.
Our study has a number of limitations inherent to its retrospective design. Vancomycin levels were drawn on day 3 of treatment but not necessarily at 72 h from first dose. The decision to check a level on day 3 was at the discretion of the treating physician, which may have introduced a selection bias. The measurement of all patient variables had to have occurred within 6 months prior to the peritonitis episode, however variables may still change over the time, and the peritoneal solute transfer rate may be very different in an infected versus non-infected state. Almost all patients in our study had residual kidney function, assessed by GFR and not urine volume which may limit generalisability. The sample size was relatively small which limited our capacity to test multiple candidate predictor variables and contributed to wide error bounds for some estimates. Some data were missing for peritoneal transport rate. Although we selected variables based on biological plausibility, there remains the potential for measured or unmeasured confounding, for example, related to the relationship between peritoneal membrane surface area, weight and vancomycin absorption. While we identified some factors which may help explain some of the variation in day 3 vancomycin level, 69% of the variance in levels remains attributable to unknown factors.
Conclusion
Our current IP vancomycin dosing regimen resulted in subtherapeutic levels by day 3 in one-third of cases. Weight-adjusted dosing and residual kidney function were found to be independent predictors of vancomycin level, and an initial dose ≥25 mg/kg increased the likelihood of achieving a therapeutic level, suggesting that physicians should consider weight-based dosing for vancomycin during PD peritonitis and incorporate residual kidney function into treatment decisions. At our centre, we will be moving to a weight-based protocol instead of fixed dosing, with consideration of higher initial loading doses and shorter monitoring/dosing intervals (i.e. 2 days instead of 3, 4 or 5) for patients with substantial residual kidney function. Some questions remain unanswered including the potential for nephrotoxicity from supra-therapeutic vancomycin levels in the first 48 h of treatment, particularly in individuals with low body mass who receive a standard loading dose. Future prospective studies with structured capture of vancomycin levels are needed to address these questions and refine our approach to antimicrobial therapy for PD peritonitis.
Supplemental material
Supplemental Material, sj-pdf-1-ptd-10.1177_08968608221134980 - Predictors of serum vancomycin levels in peritoneal dialysis-associated peritonitis
Supplemental Material, sj-pdf-1-ptd-10.1177_08968608221134980 for Predictors of serum vancomycin levels in peritoneal dialysis-associated peritonitis by Erin Deacon, Mark Canney, Brendan McCormick, Pierre Brown, Mohan Biyani and Deborah Zimmerman in Peritoneal Dialysis International
Footnotes
Acknowledgement
The authors thank Justine Davis for her administrative support throughout this project.
Author contributions
DZ conceived the study. DZ and ED were involved in gaining ethical approval and researching literature. DZ, ED and MD were involved in protocol development and in data analysis. ED wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethics approval for this study was obtained from the research ethics board of The Ottawa Hospital (approval number 20210456-01H).
Funding
The authors disclosed receipt of the following financial support for the research, authorship and publication of this article: This article was supported by Jones Family Foundation.
Informed consent
Informed consent was not required for this retrospective chart review.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
