Abstract
Long-term peritoneal dialysis is associated with the development of peritoneal membrane alterations, both in morphology and function. Impaired ultrafiltration (UF) is the most important functional change, and peritoneal fibrosis is the major morphological alteration. Both are caused by the continuous exposure to dialysis solutions that are different from plasma water with regard to the buffer substance and the extremely high-glucose concentrations. Glucose has been incriminated as the major cause of long-term peritoneal membrane changes, but the precise mechanism has not been identified. We argue that glucose causes the membrane alterations by peritoneal pseudohypoxia and by the formation of advanced glycosylation end products (AGEs). After a summary of UF kinetics including the role of glucose transporters (GLUT), and a discussion on morphologic alterations, relationships between function and morphology and a survey of the pathogenesis of UF failure (UFF), it will be argued that impaired UF is partly caused by a reduction in small pore fluid transport as a consequence of AGE-related vasculopathy and – more importantly – in diminished free water transport due to pseudohypoxia, caused by increased peritoneal cellular expression of GLUT-1. The metabolism of intracellular glucose will be reviewed. This occurs in the glycolysis and in the polyol/sorbitol pathway, the latter is activated in case of a large supply. In both pathways the ratio between the reduced and oxidised form of nicotinamide dinucleotide (NADH/NAD+ ratio) will increase, especially because normal compensatory mechanisms may be impaired, and activate expression of hypoxia-inducible factor-1 (HIF-1). The latter gene activates various profibrotic factors and GLUT-1. Besides replacement of glucose as an osmotic agent, medical treatment/prevention is currently limited to tamoxifen and possibly Renin/angiotensis/aldosteron (RAA) inhibitors.
Keywords
Introduction
The continuous exposure to conventional peritoneal dialysis (PD) solutions during PD induces alterations in the peritoneal tissues that constitute the dialysis membrane. These changes concern both peritoneal morphology and function. Impaired ultrafiltration (UF), that is, insufficient fluid removal in the absence of excessive intake is the most important functional abnormality, because it often causes overhydration, which is an important cause of morbidity and death. UF starts to decline progressively after 3 years of PD when measured under standardised conditions 1,2 but does not always cause fluid overload, because this clinical condition is not only determined by dialysis-induced fluid removal but also by fluid intake and residual urine production. Therefore a reproducible definition of UFF should be based on the capability to remove a predefined amount of fluid under standardised dialysis conditions. Already in 2000 the 3 × 4 rule for UFF has been defined as a combination of net UF < 400 mL after a 4-h dwell with a dialysis solution containing 3.86/4.25% glucose. Using this definition UFF is present in about 20% of patients after 2 years PD and almost 40% after 4 years. 3
Morphologic changes in peritoneal tissues are the underlying causes of the progressive UF decline. This is illustrated in patients with encapsulating peritoneal sclerosis (EPS), in whom UFF is the only functional abnormality present in virtually all patients. 4 Impaired UF is however not specific for EPS, because it can also develop in long-term patients without clinical signs of EPS. 3 The objective of the present review is to provide a summary of the functional and morphological peritoneal alterations that can develop in long-term PD, discuss relationships between morphology and function and work out their pathogenesis. Finally, the prevention and therapeutic options will be discussed.
UF kinetics
Transcapillary ultrafiltration (TCUF) during PD occurs by pores in the microvascular wall. The amount of pores is determined by the number of perfused microvessels and is a quantitative determinant of peritoneal transport capacity. Small solute transport occurs via free diffusion through these vascular pores, meaning that their transport rates, often expressed as D/P creatinine, can be considered to reflect the number of perfused microvessels, that is, the effective peritoneal surface area (EPSA). Two sets of pores are involved in TCUF: intra-endothelial pores consisting mainly of the above discussed small pores with radii of about 40 Å and the intracellular water channel aquaporin-1 (AQP-1) with a radius <5 Å.
Intravascular fluid with dissolved solutes is filtered through the small pores to interstitial tissues by the hydrostatic pressure gradient, which averages 30 mmHg, but decreases from the arteriolar part to the capillaries. Due to the fall in hydrostatic pressure in the arteriolar to venous pathway, the major part of small pore fluid transport (SPFT) will occur in the arterioles. A larger EPSA will increase SPFT, as supported by the positive relationship, that has been shown between this parameter and D/P creatinine. 5
AQP-1 is present in peritoneal capillaries and venules, not in arterioles. Due to its very small radius, solutes like urea, creatinine, glucose and electrolytes cannot pass this water channel. Therefore AQP-1 allows free water transport (FWT). The quantity of AQP-1 pores is about 2% of the total number of peritoneal microcirculatory pores, making them irrelevant for hydrostatic fluid transport, but very effective for osmotic water transport. 6 During the first hour of a 3.86% glucose containing dialysis solution exchange, 40% of UF occurs through water channels. 7 A large EPSA leads to a rapid disappearance of the crystalloid osmotic pressure gradient due to enhanced absorption of glucose from the dialysis solution. This explains the negative relationship that has been found between FWT and D/P creatinine. 5
The disappearance of intraperitoneal glucose from the peritoneal cavity is faster than expected from diffusion only. 8 This suggests the possibility of glucose uptake by peritoneal cells. This uptake occurs through glucose transporters (GLUTs) present in the cell membrane. 9 Two types can be distinguished: facilitative GLUTs and sodium-linked transporters (SGLTs). GLUTs facilitate glucose diffusion through the cell membrane, glucose uptake by SGLTs is driven by that of sodium. GLUT-1 is insulin independent and is distributed ubiquitously, including cultured human mesothelial cells, where it was upregulated by glucose, but not by an equimolar concentration of mannitol. 10 Mesothelial SGLTs have also been described, but their importance is unknown. 11 Two recent studies from Sweden in an acute PD model in rats with normal kidney function showed evidently that SGLT inhibition had no effect on UF, 12 but GLUT inhibition by intraperitoneal phloretin caused an increase in UF, accompanied by a reduced glucose transport rate. 13 This dihydrochalcone flavonoid is especially present in apples. It has antioxidant properties and is mainly used for topical application on the skin. This experimental study has shown that the disappearance of glucose from peritoneal dialysate not only is dependent on diffusion to the circulation but also has a cellular component, probably mediated by GLUT-1.
Glucose-induced FWT causes a decrease in the concentration of dissolved solutes in the dialysate, which is most prominent for solutes with similar concentrations in plasma and dialysate, like Na+. This sodium sieving is especially pronounced with high-glucose concentrations in the dialysis solution and usually reaches its minimum after about 60 min. Therefore the quantity of FWT can be approximated by the determination of the D/P concentration of Na+ after 60 min or calculated using the amount of sodium removed in this period. 14 Subtraction of the amount of sodium-associated SPFT from total UF yields FWT. In this approach lymphatic absorption is neglected, but this is quantitatively unimportant during 60 min. Our group described that both FWT and SPFT decrease in long-term PD. 2 These phenomena can only be understood by knowledge of the morphological alterations that develop in long-term PD patients.
Morphologic alterations
The PD membrane is composed of the mesothelial layer and the submesothelial interstitium, in which blood and lymphatic vessels are dispersed. This interstitium consists of a ground substance containing hyaluronan and glycosaminoglycans, and a fibrous collagen network that constitutes a skeleton for the embedded structures. Adipose cells and a few fibroblasts are the predominant cell types. The mesothelial layer offers no hindrance to peritoneal solute transport and is no barrier for crystalloid osmosis. 15 Therefore the interstitial microcirculatory blood vessels have been considered the determinants of peritoneal solute and fluid transport from the circulation to the dialysate. The relationship between EPSA parameters and the number of perfused peritoneal microvessels discussed in the section on UF kinetics has only been found for immature, smooth muscle negative capillaries, not for those that are smooth muscle actin positive. 16 Vascular abnormalities can develop during the time course of PD, characterised by subendothelial hyalinosis with luminal narrowing or even obstruction. 17 This process starts from 2 years on PD and can quantitatively be characterised by the lumen/total ratio of a vessel. This abnormality is usually seen in the systemic microcirculation of patients with hypertension and/or diabetes mellitus. Deposition of advanced glycosylation end products (AGEs) is present around the microvascular wall and in the submesothelium. 18 The dialysate concentration of the AGE pentosidine increases with PD duration. 19 A relationship between AGE deposition and UFF has been described and suggests involvement of AGEs in vasculopathy. 18
Endothelial-to-mesenchymal transition (EMT) of mesothelial cells is a phenomenon that occurs in the first few years of PD. 20 It is characterised by the presence of cytokeratin in submesothelial cells and is accompanied by release of vascular endothelial growth factor. 21 This growth factor stimulates angiogenesis and causes vasodilation, which is in accordance with the presence of an increase of EPSA as found in all longitudinal studies on peritoneal solute transport. 1,2,22 The absence of longitudinal morphologic studies makes it impossible to establish a possible relationship between peritoneal EMT and the development of submesothelial fibrosis. An elegant in vitro study established different origins of mesothelial cell-derived myofibroblasts and those with a submesothelial base. 23 It showed that EMT is not directly related to submesothelial fibrosis.
Mesothelial denudation and progressive interstitial fibrosis – often assessed as the thickness of the submesothelial zone – are the most prominent findings in long-term PD. The adipose tissue is largely replaced by myofibroblasts. These specialised fibroblasts stain positive with α-smooth muscle actin, can synthesise collagen 24 and express GLUT-1. 25
Relationships between function and morphology
The presence of fast transport rates of small solutes suggests a large vascular density, which has indeed been found for immature capillaries. 16 The increase of small solute transport rates with PD duration and the concomitant UF decrease during the first years are likely caused by vascular endothelial growth factor (VEGF) produced during EMT. Loss of mesothelial cells has never been related to functional abnormalities but causes an increased exposure of submesothelial tissue to high concentrations of glucose and glucose degradation products. These products enhance the Maillard reaction between glucose and tissue proteins, eventually leading to AGE formation. AGEs in the microvascular wall are probably important in the genesis of vasculopathy 18 and may affect hydrostatic pressure-driven SPFT by the following mechanism. Vasculopathy is characterised by the presence of multiple narrowings. Post-stenotic pressure in vasculopathy is likely reduced and will lead to reduced SPFT. Long-term exposure to conventional dialysis solutions is associated with fibrotic interstitial alterations consisting of myofibroblasts and collagen fibres. It will be argued that these alterations have no effect on solute transport but are extremely important in FWT. This importance is supported by the observation that patients with EPS have markedly reduced FWT. 3,24
Pathogenesis of long-term UFF
The continuous exposure of peritoneal tissues to extremely high-glucose concentrations is the obvious cause of the described functional and morphological alterations. This is illustrated by the observation that patients with an increase in small solute transport during long-time follow-up had a higher peritoneal glucose exposure than those with stable transport. 26 Likewise, patients who developed EPS had higher peritoneal glucose exposure compared to controls. 4 These studies do not allow to distinguish between effects of glucose itself or of glucose degradation products. Comparison between conventional dialysis solutions and the available ones with a low content of glucose degradation products and a neutral pH (L-GDP/N-pH) allows more insight in this issue. The relevant functional and morphological studies have been summarised recently. 27 Two longitudinal studies on peritoneal function comparing L-GDP/N-pH solutions with conventional ones with a follow-up exceeding 2 years have been published. 28,29 Fluid kinetics were analysed in one of these. 29 In general, the expected reduction in AGE accumulation was found, which was accompanied with less severe vasculopathy and was reflected in a better preservation of SPFT. In contrast, FWT still declined with time, pointing to the importance of glucose itself.
Glucose-induced peritoneal alterations
Long-term PD is functionally characterised by reduced FWT and morphologically by submesothelial interstitial fibrosis. This suggests a common aetiology. The interstitial fibrosis consists of myofibroblasts that synthesise extracellular matrix components and collagen fibres, especially collagen-1. Mesothelial origin of these cells is unlikely, 23 but transdifferentiation of adipocytes into myofibroblasts is a possibility. Chronic inflammation has been postulated as the trigger for these fibrotic alterations, but this is not very plausible as argued previously. 30 In recent years hypoxia has gained attention as initiator of fibrotic processes, because it activates the transcription factor hypoxia-inducible factor-1 (HIF-1). 31 For instance, hypoxia is considered an important player in the progression of chronic kidney disease, also because HIF-1 is stimulated by angiotensin-2. Involvement of HIF-1 in the morphologic alterations of long-term PD is an attractive possibility, because it stimulates the various factors that are involved in peritoneal remodelling, like VEGF, transforming factor β (TGFβ), plasminogen activator inhibitor-1 (PAI-1) and connective tissue growth factor (CTGF); moreover it increases the expression of GLUT-1. 31
Ischaemia of peritoneal tissues could be due to vasculopathy, but this is not very likely, because vasculopathy is not present at the start of PD and takes time to develop. Glucose-induced pseudohypoxia similar to diabetes mellitus is an attractive possibility, because this has been described as a mechanism to explain the vasodilation induced by hyperglycaemia in patients with diabetes. 32 The mechanism is as follows: Hypoxia generally leads to vasodilation as a compensatory mechanism. At the cellular level this is mediated by nicotinamide dinucleotide (NAD), which is present in the cytosol in a reduced form (NADH) and an oxidised form (NAD+). Oxygen is required for the oxidation of NADH to NAD+. Therefore hypoxia causes an increased intracellular NADH/NAD+ ratio. A great cellular glucose load also increases the NADH/NAD+ ratio, which explains the term pseudohypoxia.
Degradation of the ingested glucose in PD and how this affects the NADH/NAD+ ratio has recently been described in detail. 30,33,34 In brief, the intracellular NADH/NAD+ ratio increases in both the glycolysis and the polyol/sorbitol pathway. The effects may be augmented by the use of lactate-based solutions which may inhibit or reverse the lactate dehydrogenase reaction, and kidney failure mediated mitochondrial dysfunction. The various pathways are shown in Figure 1.
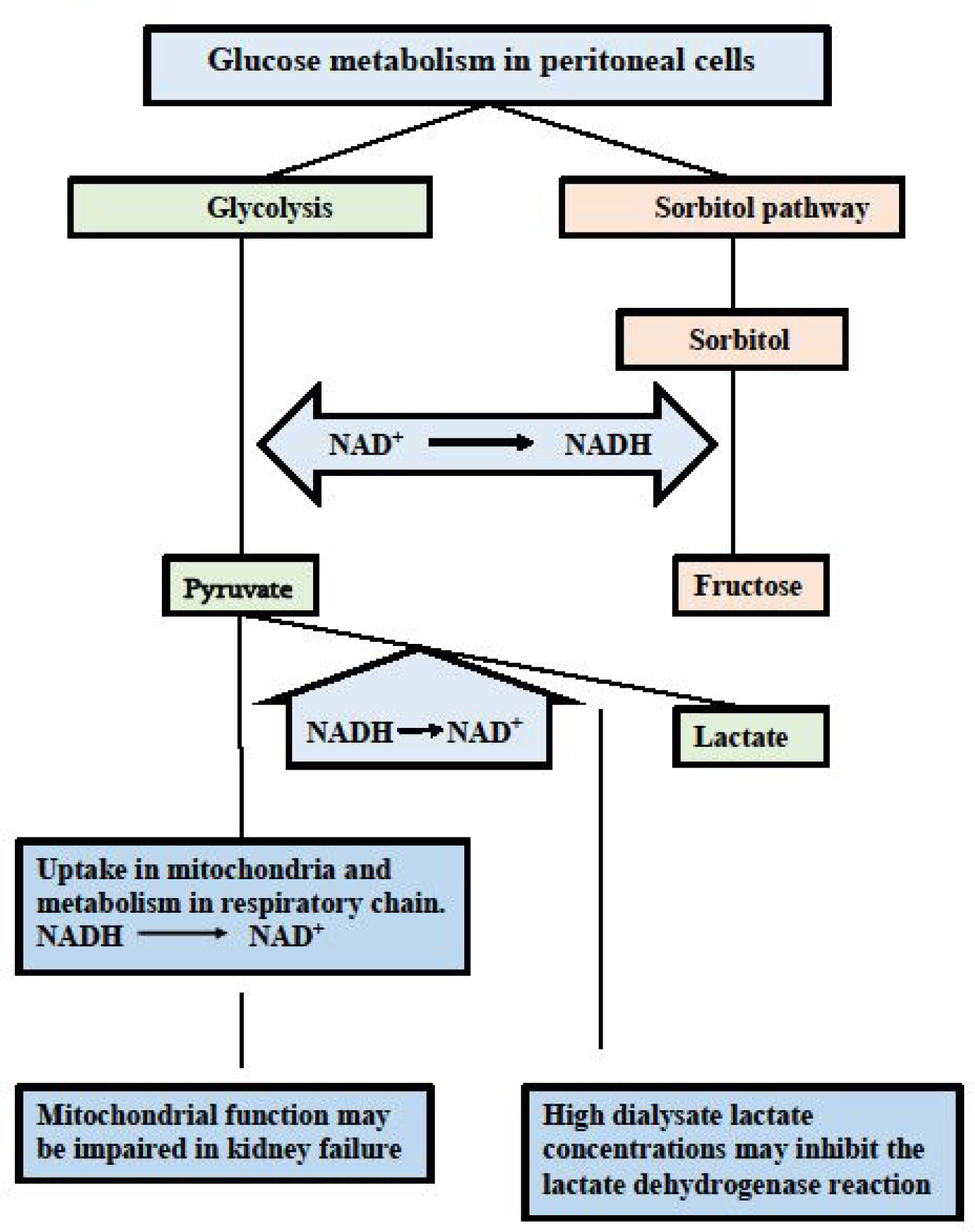
Glucose metabolism in peritoneal cells. Intracellular glucose can be degraded in the glycolysis and in the polyol/sorbitol pathway. NAD is involved in by the reduction of NAD+ to NADH. Pyruvate is the end product in the glycolysis. Oxidation of the formed NADH occurs by metabolism of pyruvate in the mitochondria and conversion into lactate. These normal compensatory mechanisms may be impaired due to mitochondrial dysfunction and to the use of lactate as buffer in the dialysis solution. Fructose is the end product of the polyol/sorbitol pathway without any compensatory mechanism to oxidise NADH. The resulting increased NADH/NAD+ ratio is an indicator of cellular hypoxia. Taken from Krediet. 34
Clinical and other data that are in favour of peritoneal pseudohypoxia
Cells in peritoneal effluent of non-infected PD patients express many genes. The majority of these is upregulated and increases with the duration of PD. 35 This upregulation with PD duration was most marked for CD24, a heavily glycosylated gene that is upregulated during hypoxia. 36 CD24 expression showed a relationship not only with PD duration but also with collagen-1 and 6 genes and various extracellular matrix genes. 35 These data suggest involvement of CD24 in long-term peritoneal alterations, of which EPS is the most severe one. The abundance of factors involved in the sclerotic process includes TGFβ, CTGF and PAI-1. All are upregulated by HIF-1. Also the concomitant decrease in FWT in EPS and long-term PD 3,24 can be explained by a HIF-1 induced increase of GLUT-1 expression in peritoneal myofibroblasts. Such increase leads to a decreased glucose concentration at the microvascular endothelial cells that express AQP-1. 33
Hypoxia induces the production of nitric oxide for compensatory vasodilation. Long-term PD patients show indeed a higher activity of endothelial nitric oxide synthase in peritoneal tissue compared to short-term controls. 37 This was accompanied by upregulation of VEGF. No cause for nitric oxide activation could be identified in that study suggesting that pseudohypoxia might have been involved.
Oxidative stress with the production of free radicals has been described in chronic kidney failure, haemodialysis (HD) and PD patients. Plasma malondialdehyde concentrations – a marker of oxidative stress – were higher in PD than in HD patients, suggesting an effect of exposure to dialysis solutions, in particular glucose or glucose degradation products. 38 Furthermore, PD solutions induce free radical production by endothelial-like cells in vitro. Although seemingly paradoxical, hypoxia is associated with free radical production, as has been described in many conditions including end-stage kidney disease. 39 The marked increase in mitochondrial NADH caused by compensatory anaerobic glycolysis is the most likely mechanism. Glucose-induced pseudohypoxia is especially characterised by high intracellular NADH concentrations, making this condition a very likely explanation for oxidative stress in PD patients.
It can be concluded that glucose-induced pseudohypoxia is the common denominator for the above discussed peritoneal alterations in long-term PD. These include pathophysiological characteristics, like induction of endothelial nitric oxide and oxidative stress, but also the discussed morphological and functional changes, that consist of EMT of mesothelial cells, submesothelial and interstitial fibrosis, angiogenesis and rarely EPS in long-term patients. UFF, due to impaired FWT, is the most important functional complication and an important determinant of PD survival. The mechanisms for ultrafiltration failure are summarized in Figure 2.
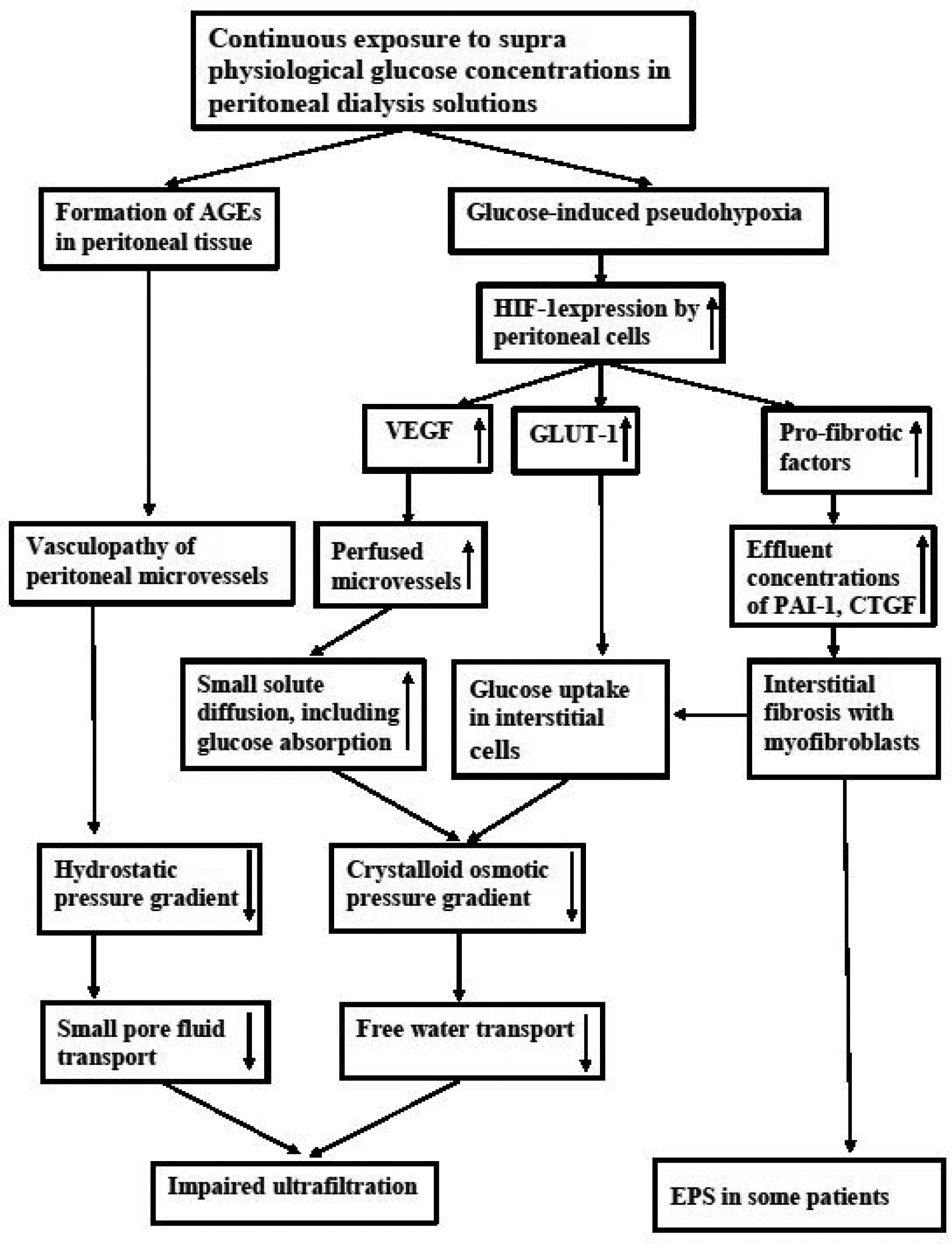
A schematic representation of the mechanisms involved in the peritoneal handling of glucose in dialysis solutions leading to AGE formation and pseudohypoxia, which finally results in ultrafiltration impairment, increased small solute transfer rates and peritoneal fibrotic alterations with the development of EPS in some patients.
Medical treatment and prevention
Primary prevention of the consequences of pseudohypoxia consists of replacing the high-glucose concentrations in the dialysis solutions by other osmotic agents. Possible modifications of osmotic agents aimed at the various possibilities to achieve this have recently been discussed. 34 None of these alternatives are currently available for clinical PD. The following section will therefore focus on possibilities of secondary prevention and treatment by drugs that may be relevant for preservation of peritoneal membrane integrity during routine PD. The following division was made: (1) drugs that interfere with the NADH/NAD+ ratio, (2) drugs that inhibit GLUTs and (3) drugs that inhibit HIF-1 activation. These are summarised in Table 1.
Drugs aimed at secondary prevention of peritoneal membrane alterations.

PD: peritoneal dialysis; UF: ultrafiltration; GLUT: glucose transporter; TGFβ: transforming factor β; NAD: nicotinamide dinucleotide; EPS: encapsulating peritoneal sclerosis.
Drugs that interfere with the NADH/NAD+ ratio
The NADH/NAD+ ratio is reduced by inhibition of the polyol/sorbitol pathway. This is possible with aldose reductase inhibitors that block the conversion of sorbitol to fructose. Currently these inhibitors are not available for human use, because of severe toxicity. The aldose reductase inhibitor zopolrestat has been investigated in a long-term peritoneal exposure model in rats, where it decreased peritoneal fibrosis and angiogenesis. 40
Another way to interfere with the NADH/NAD+ ratio is the use of nicotinamide mononucleotide, which is a precursor of NAD+. It is promoted as an anti-ageing agent, especially in Japan. Ongoing studies in humans have been summarised recently, but no study focused on its effects in PD. 41
Drugs that inhibit GLUTs
Sodium glucose transport (SGLT) inhibitors have become very popular, especially type 2 (SGLT-2). Besides lowering of plasma glucose concentrations in patients with type 2 diabetes, they reduce blood pressure and have been found beneficial in patients with heart failure and with chronic kidney disease. Uptake of sodium in cells is the driving force for glucose transport from extra to intracellular, 9 as occurs for instance in the proximal tubule. In the absence of extensive sodium transport in PD, an important contribution of these transporters to glucose uptake in peritoneal cells is unlikely, although their presence has been shown in peritoneal mesothelial cells. This has been confirmed by the absence of an effect on UF of the SGLT-1 and SGLT-2 inhibitor phlorizin in an acute rat model, 12 although its application was associated with a reduced peritoneal absorption of glucose. 42 However, the systemic use of SGLT-2 inhibitors in PD patients may have beneficial effects, for instance on urine production and cardio-protection, but currently no clinical data are available. In contrast, the GLUT inhibitor phloretin caused improved UF and reduced glucose absorption in the same model, as discussed earlier. 13 These effects are in agreement with the presence of GLUT-1 in murine and human peritoneal mesothelial cells. 11 No data are available on the use of phloretin in human PD.
Drugs that inhibit HIF-1 activation
The following drugs will be discussed: tamoxifen, rapamycin and drugs that interfere with RAAS. Tamoxifen has anti-estrogenic properties that explain its use in mama carcinoma but also has anti-fibrotic effects, illustrated by its well-known use in retroperitoneal fibrosis. Also its application in EPS patients has given favourable results. 43 However, no results have been published on its prophylactic use in long-term PD patients. Recently the mechanism of the tamoxifen effect has been found to be dependent on HIF-1 inhibition, leading to regulation of collagen cross-linking and thereby of the extracellular matrix. 44
It is known from transplant surgery that rapamycin/sirolimus is associated with delayed wound healing. This effect is caused by inhibition of mTOR, a protein kinase that is upregulated by oxidative stress. 45 The mTOR inhibition was associated with decreased production of TGFβ by cardiac myocytes. As discussed above (pseudo)hypoxia can be associated with oxidative stress. A possible beneficial effect of rapamycin/sirolimus in PD patients with EPS is likely, but has only been described in case reports. 46 However, possible beneficial effects of rapamycin/sirolimus will have to be weighed against its well-known side effects.
The notion that angiotensin-2 can activate HIF-1, the similarity between the microvascular changes in diabetic nephropathy and those of the peritoneal microvessels in long-term PD 47 and the favourable effects of RAAS inhibition on the progression of diabetic nephropathy were driving forces for the analysis of possible effects of RAAS inhibition in PD patients. A single centre study in which 66 incident PD patients were included with a follow-up of at least 2 years and regular measurement of peritoneal function showed that the 36 patients that had received ACE inhibitors or angiotensin-2 receptor blockers for at least 20% of their follow-up time had stable small solute transport rates up to 4 years follow-up. In contrast, the 30 control patients showed the well-known increase of small solute transport rate after 3 years of PD. 48 These results on the time course of small solute transport were subsequently confirmed in a comparison of 217 incident PD patients that were included and followed in the Netherlands Cooperative study on the adequacy of dialysis cohort. The120 with ACE inhibitors or angiotensin-2 receptor blockers showed no increase during follow-up, in contrast to the controls. 49 The analysis of UF in the 66 patients of the first study showed no differences between the two groups in net UF, FWT or its contribution to UF. However, the 36 patients with RAAS inhibition had a larger decrease in SPFT than the 30 controls, 48 possibly caused by a decreased filtration pressure. These results suggest that RAAS inhibition has no direct effect on pseudohypoxia but may inhibit the formation of new capillaries. A few other small clinical studies have been published on effects of RAAS inhibition in PD patients, but these were either cross-sectional or had a follow-up of 1 year only. It can be concluded that more and larger studies are required on possible effects of RAAS inhibition in PD patients.
The review of the discussed options for secondary prevention of long-term peritoneal alterations shows that the possibilities are limited to drugs that inhibit fibrosis and have acceptable side effects, like tamoxifen. More studies are required on SGLT-2 inhibitors and RAAS inhibitors. The discouraging results of medical treatment/prevention up to now underline the need for replacement of high-glucose concentrations in dialysis solutions by combinations of other osmotic agents.
Summary and conclusions
Impairment of UF capacity is the most frequent functional abnormality of long-term PD. The reduced UF concerns both SPFT mainly driven by the hydrostatic pressure gradient and FWT through AQP-1, which is exclusively dependent on the osmotic pressure gradient. The former mainly occurs in the arteriolar part of the microcirculation, and the latter in the venular segment. Assessment of peritoneal sodium transport allows to distinguish between the two routes. The dialysate glucose concentration and consequently FWT decreases during a dialysis dwell due to absorption, which is partly by diffusion but also by uptake in peritoneal cells through channels in the cell membrane. These GLUTs consist of facilitative GLUTs and sodium glucose transporters (SGLT). Experimental studies have made it likely that GLUTs are most important in PD. Deposition of AGEs, an increased extracellular matrix with replacement of adipocytes with fibroblasts, collagen deposition and vasculopathy are major morphological abnormalities in long-term PD. Both SPFT and FWT decrease in long-term PD. Glucose-induced AGE formation probably explains the vasculopathy leading to reduced SPFT, glucose-induced pseudohypoxia – defined as an increased intracellular NADH/NAD+ ratio – is the culprit of the FWT reduction and the interstitial fibrotic abnormalities. A high nicotinamide adenine dinucleotide ratio upregulates HIF-1, which stimulates TGFβ, VEGF and GLUT-1 expression. Pseudohypoxia leading to high intracellular NADH concentrations also explains the nitric oxide production and the oxidative stress that have been described in PD. The primary prevention of the consequences of glucose-induced peritoneal AGE formation and pseudohypoxia obviously consists of total or partial replacement of glucose as osmotic agent. Most potential drugs for secondary prevention cannot be given to humans or are in an experimental phase. Currently only effects of tamoxifen or sirolimus/rapamycin and RAAS inhibitors have been described, although mainly in a limited number of patients.
It can be concluded that more insight is present in the mechanisms of glucose toxicity as the cause of functional and morphological peritoneal damage in long-term PD patients than previously renowned. This and the overwhelming data on all advantages of PD as continuous dialysis treatment for the first 3–4 years should urge the providers of commercial dialysis solutions to increase their efforts to develop dialysis solutions that are (almost) free of glucose and do not cause long-term complications. Combinations of various osmotic agents each in a low dose are probably the best approach to achieve this goal.
Footnotes
Acknowledgements
None.
Author contributions
Raymond T Krediet and Alena Parikova contributed similarly to this review.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
No ethical approval required for this review.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
None.
