Abstract
Background:
Little is known about the impact of haemodialysis (HD) and peritoneal dialysis (PD) on health-related quality of life (HRQoL). We compared HRQoL between conventional in-centre HD and home-based PD in 1082 newly diagnosed kidney failure patients.
Methods:
This was an open-label, randomised, non-inferiority trial of adult patients with a diagnosis of end-stage kidney disease (estimated glomerular filtration rate ≤ 15 mL/min/1.73 m2) requiring maintenance dialysis from 36 sites in China randomised 1:1 to receive PD or conventional in-centre HD. The primary outcome was the ‘Burden of Kidney Disease’ assessed using the Kidney Disease Quality of Life-Short Form (KDQoL-SF) survey over 48 weeks and the main secondary outcomes were the remaining scales of KDQoL-SF and all-cause mortality. The effect of PD versus HD on the primary outcome was compared by their geometric mean (GM) ratio, and non-inferiority was defined by the lower bound of a one-sided 95% confidence interval (CI) >0.9.
Results:
A total of 725 subjects completed the trial per protocol (395 PD and 330 HD, mean age 49.8 (standard deviation (SD) 14.4) years, 41.4% women). For the primary outcome, the mean (SD) change in ‘Burden of Kidney Disease’ over 48 weeks was 2.61 (1.27) in PD group and 2.58 (1.35) in HD group, and the GM ratio (95% CI) was 1.059 (0.908–1.234), exceeding the limit for non-inferiority. For the secondary outcomes, the PD and HD groups were similar in all scales. There were 17 and 31 deaths in PD and HD groups, respectively. Patients receiving PD had more adverse events, adverse event leading to hospitalisation and serious adverse events compared to those allocated to HD, but adverse events leading to death and discontinuation of the trial were not different between PD and HD.
Conclusions:
In this trial, PD may be non-inferior to HD on the ‘Burden of Kidney Disease’ among Chinese kidney failure patients who are of younger age and have lower comorbidity after 48 weeks’ follow-up.
This is a visual representation of the abstract.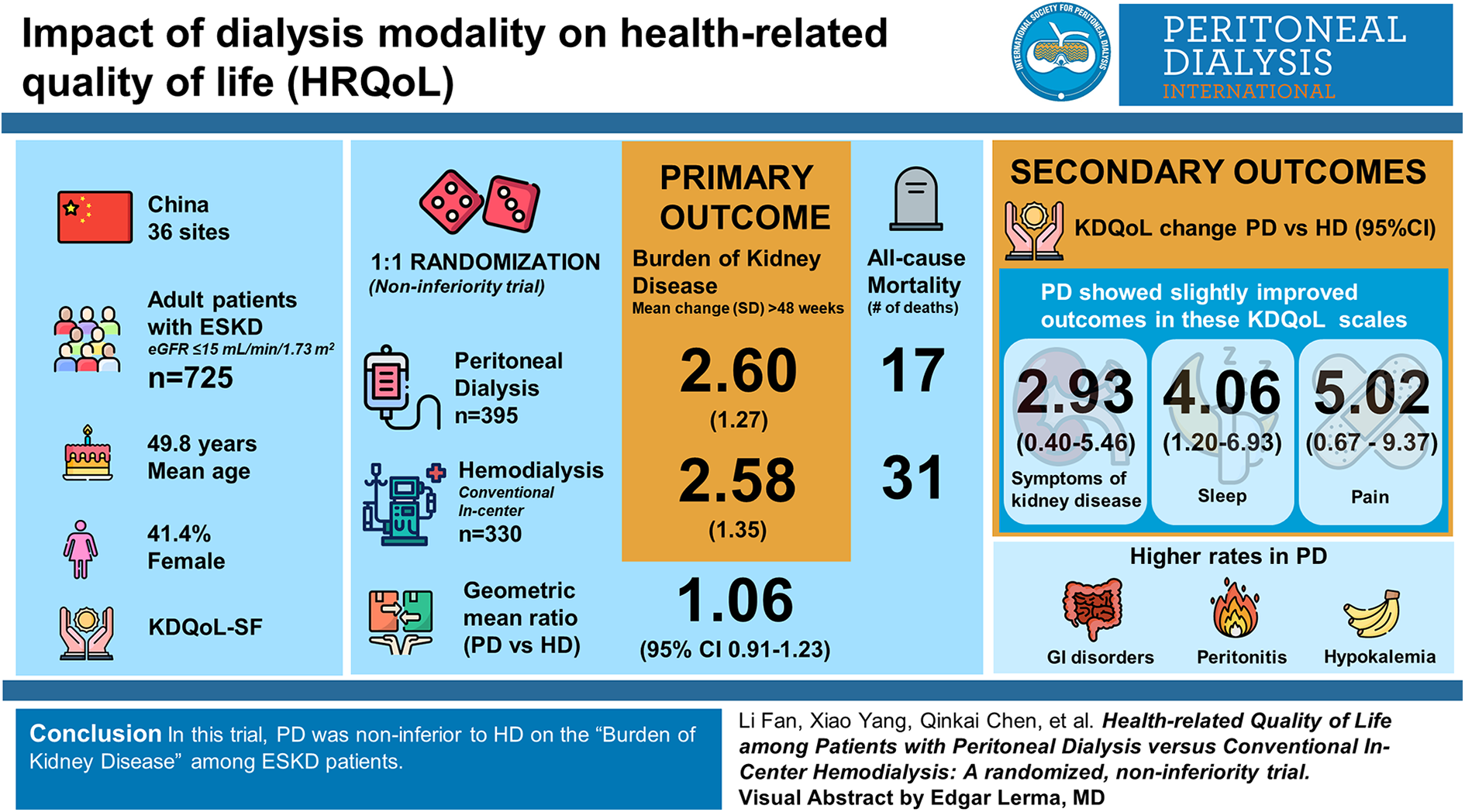
Background
Kidney failure is an important cause of patient life-years lost and reduced health-related quality of life (HRQoL). 1,2 Globally, dialysis is the most common kidney replacement therapy. Approximately 89% of dialysis patients receive haemodialysis (HD) and the remainder receive peritoneal dialysis (PD). 3,4 The proportional use of HD versus PD varies widely by health jurisdiction. For instance, more than 97% of dialysis patients in Japan are treated with HD, but in Jalisco of Mexico, more than 50% are treated with PD. 5
With the increase in life expectancy, patient-centred outcomes are receiving more attention. Kidney failure patients can prolong their life by receiving dialysis, but an important question remains to be answered: What is the impact of HD versus PD on their HRQoL? To date, almost all studies comparing clinical and patient-centred outcomes between PD and HD have been observational in design, 6,7 with some studies 8 –11 suggesting that mortality risks are comparable but that patient-reported outcomes may be better with PD. However, overall, the results from these observational studies have been mixed. 7,10 –12 As a home-based dialysis, while it is conceivable that patients receiving PD are more able to maintain active work and social interactions than patients receiving conventional in-centre HD, 13 –15 this finding is yet to be confirmed by randomised trials, given that the choice of HD or PD is made at least in part by the patient. We are aware of only two randomised controlled trials that have addressed this issue and the results of one trial may no longer be applicable to contemporary practice dialysis. 8 The other was insufficiently powered due to poor recruitment and patients’ non-acceptance of allocation to PD or HD. 16
Beyond HRQoL, the findings from this type of study are also useful from a health economics and service access perspective. For most healthcare systems, including those in the United States, Australia, Indonesia and China, PD is less expensive than HD. 4,5,17 Therefore, a greater utilisation of PD might afford more kidney failure patients to be treated, especially in middle- or low-income countries and those living in rural or remote areas, as is the case for nearly half of the Chinese population.
This randomised, non-inferiority trial aimed to evaluate HRQoL among kidney failure patients receiving home-based PD versus conventional in-centre HD. The primary outcome was change in ‘Burden of Kidney Disease’ from baseline to 48 weeks post-randomisation, assessed using the Kidney Disease Quality of Life-Short Form (KDQoL-SF). Secondary outcomes were changes in remaining scales of HRQoL, all-cause mortality and other relevant clinical outcomes including adverse events.
Methods
Study design
The study was a prospective, randomised, parallel, open-label, multicentre, non-inferiority trial. The trial protocol was approved by the ethics committees of all participating centres. The conduct of the trial conformed with the Good Clinical Practice guidelines and the principles of the Declaration of Helsinki. All patients provided written informed consent before undergoing any trial procedures. The trial was overseen by an external data and safety monitoring board and a steering committee.
The trial was conducted in two phases. In phase I, the primary outcome was all-cause mortality, and secondary outcomes included HRQoL measures with a planned maximum duration of 5 years after randomisation of the first subject to either HD or PD from 1 June 2011. At a planned interim analysis by the data monitoring and safety board, the mortality rate in the trial was found to be only 0.03 per patient-year, lower than the rate of 0.15 per patient-year that was used to calculate sample size, with a conditional power of only 0.5 for the primary outcome. In the absence of any predefined stopping rules, the board expressed concern that the original design may be futile. As a result, the steering committee recommended terminating phase I and initiating phase II. Phase II was the same as phase I in terms of the inclusion and exclusion criteria, study procedures and data collection protocols, except that (1) HRQoL became the primary end point and total mortality became the secondary end point and (2) eight new study sites were added to increase the recruitment of patients. Eligible patients in phase II were randomised to either HD or PD from 9 May 2014 to 1 July 2016 and followed up until 29 September 2017.
Participants
Participants from 36 sites in China were screened. Adult patients (age ≥ 18 years) were eligible for participation if they had been recently diagnosed with kidney failure (defined as estimated glomerular filtration rate ≤ 15 mL/min/1.73 m2), anticipated to initiate maintenance dialysis within 10 weeks after enrolment, expected to remain on dialysis for at least 48 weeks, were able to complete the standardised pre-dialysis education programme and home-based PD training programme, able to attend HD clinics as required by the protocol and able to understand and voluntarily sign the informed consent form. Key exclusion criteria included patients who were human immunodeficiency virus positive; not eligible for either PD or HD, as judged by the investigator; already receiving maintenance dialysis for more than 4 weeks; diagnosed with an active infection or other conditions that the investigator determined may have jeopardised their ability to receive either dialysis modality; had previously received kidney transplantation or other ongoing immunosuppressive therapy and were anticipated to have a life expectancy of <48 weeks (full inclusion and exclusion criteria were available in the Online Supplemental Material).
Randomisation and masking
Eligible patients who gave their consent were randomly assigned to receive either PD or conventional in-centre HD at the end of the screening period in a 1:1 ratio per study site by a centralised randomisation system. The randomisation code and scheme were provided by an interactive voice response system and generated by a trial statistician who was blinded to trial implementation. Enrolled patients were informed of their allocation by investigators at each site. Due to the nature of the intervention, this was an open-label trial, and patients and investigators were aware of allocation.
Procedures
After randomisation, patients were provided with up to 8 weeks for preparation to start dialysis, including implantation of a PD catheter for those randomised to PD and placement of a native arteriovenous fistula or other permanent vascular access for those randomised to HD. All patients were followed every 4 weeks for the first three visits and every 12 weeks for the remaining three visits, with a total follow-up period of 48 weeks.
Patients randomly assigned to PD were treated with continuous ambulatory PD (96.5% of patients) or automated PD (3.5% of patients). Patients received three to five manual PD exchanges (1.0–2.5 L dialysate at each exchange) or three to five automated PD cycles (1.0–2.5 L at each cycle) per night with a day dwell of 2 L per day at home. Those randomised to HD received three to four sessions (4–4.5 h of dialysis per session) of conventional in-centre HD per week. The prescriptions were adjusted to maintain weekly total Kt/Vurea ≥1.7 per week in PD and single-pool Kt/Vurea ≥1.2 per dialysis session in HD. Dialysis cost of patients was covered by the National Medical insurance, regardless of whether the patient participates in this study.
HRQoL was evaluated using the Chinese version of the KDQoL-SF™ (version 1.3) 18 –20 at the first day of dialysis initiation and 48 weeks after dialysis initiation (details of the KDQoL-SF survey are described in the Online Supplemental Methods).
Outcomes
The primary outcome was the difference in change in ‘Burden of Kidney Disease’ score from the KDQoL-SF (range 0–100, a higher score indicating a lower burden of kidney disease) over 48 weeks between PD and HD (four items of ‘Burden of Kidney Disease’ are shown in the Online Supplemental Methods). The main secondary outcomes were the difference in changes in the remaining scales of the KDQoL-SF over 48 weeks between PD and HD and all-cause mortality. Other secondary outcomes included change in dialysis modality, kidney transplantation, cause of death, change in residual kidney function as evaluated by 24-h urine volume and serum haemoglobin, albumin and phosphate levels. Safety was evaluated by comparing percentage of patients in each arm with adverse events, including abnormal laboratory test findings with any clinical significance.
Statistical analysis
The final sample size determination for the primary outcome was made before the commencement of phase II, accounting for patients from phase I with complete measures of the ‘Burden of Kidney Disease’ from the KDQoL-SF at both baseline and 48 weeks. We estimated that a total of 754 patients would provide a power of 0.801 to determine that PD is not inferior to HD with a one-sided α = 0.05 and a non-inferiority margin of −10%, given the following assumptions: The true geometric mean (GM) ratio of the log-transformed score of the ‘Burden of Kidney Disease’ scale for PD versus HD (calculated as the ratio of the GM of the burden of kidney score over 48 weeks in PD divided by the GM of the score over 48 weeks in HD) was 1.0 and the standard error was 0.63. Non-inferiority would be claimed if the lower bound of a one-sided 95% confidence interval (CI) of GM ratio was >0.9.
The primary outcome was assessed using a pooled per-protocol (PP) analytical population of phase I and phase II, due to the non-inferiority nature of the study hypothesis. A sensitivity analysis was performed in a pooled intention-to-treat (ITT) analytical population and phase I and phase II ITT and PP populations. The PP population was defined as those participants who received a randomly assigned dialysis modality, did not permanently change their dialysis modality and completed the measures of ‘Burden of Kidney Disease’ at both baseline and at 48 ± 2 weeks. The treatment effect of PD versus HD was compared by their GM ratio and calculated as the exponentiation value of (ln (PD change from baseline to 48 weeks) − ln (HD change from baseline to 48 weeks)). Non-inferiority was defined by the lower bound of a one-sided 95% CI of GM ratio >0.9. Since the Burden of Kidney Disease scores at both baseline and 48weeks were linear distribution (Online Supplemental Figures 11 and 12), a linear mixed-effect model was used to evaluate the effect between the PD and HD groups, with study site as a random effect and baseline ‘Burden of Kidney Disease’ scores included in the model as covariates; the p values and 95% CIs were computed based on effect estimates and standard errors were from the linear-mixed model. Data imputation was not performed for missing data in the main analysis.
Secondary outcomes were assessed using a pooled ITT framework, and sensitivity analysis was performed in the pooled PP, phase I PP and phase II ITT and PP populations. The treatment effect of PD versus HD on the remaining scales of the KDQoL was modelled using a generalised linear model to compare the difference between groups in changes in scale values from baseline to 48 weeks and was presented as the difference in change in the KDQoL scores between PD and HD. Non-parametric Wilcoxon rank-sum testing was used to compare the difference in change in the ‘Change in Health’ rating between PD and HD due to the discrete distribution of the data. Multiple comparisons were adjusted using Benjamini–Hochberg method in remaining KDQoL scores included in secondary outcome. Mortality, dialysis modality change and kidney transplantation rates, as well as the changes of 24-h urine volume, haemoglobin, serum albumin and phosphate, were described and did not compared the difference between PD and HD based on protocol. All analyses were performed using R project for Windows (version 3.4.2).
Results
A total of 6157 eligible participants were screened and 1082 patients were randomised. Four thousand six hundred and sixty participants refused to participate in this study because they refused the randomisation, and 115 participants withdrew consent after randomisation due to refusal to accept the randomised allocation. In phase I, 235 (142 PD and 93 HD) of the 414 randomised participants, and in phase II, 490 (253 PD and 237 HD) of the 668 randomised participants, had complete measures on the ‘Burden of Kidney Disease’ at both baseline and 48 weeks. Thus, 725 participants were available for the PP analysis of the primary outcome (Figure 1). There were no differences in baseline characteristics of patients who withdrew from the trial due to either refusal of randomisation, lost follow-up, or protocol deviation after randomisation in phase I and phase II (Online Supplemental Tables 1 and 2). Baseline characteristics of the study population were balanced between the PD and HD arms (Table 1 and Online Supplemental Tables 3 to 8). Of note, the mean (standard deviation (SD)) age was 49.8 (14.4) years, 41.4% were female, 16.3% had a history of cardiovascular disease and 27.8% had diabetes.
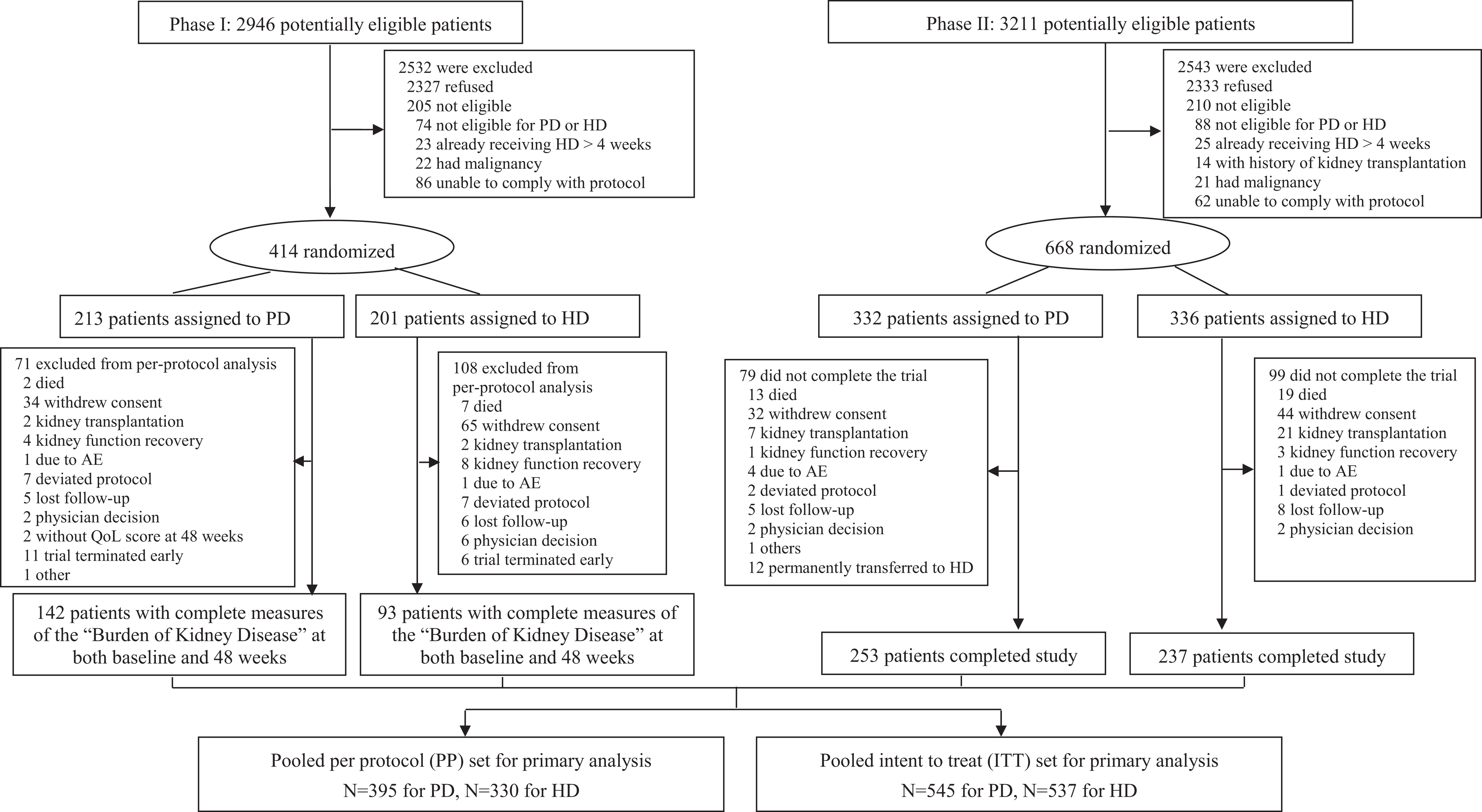
Screening, randomisation and follow-up.
Baseline patient characteristics by treatment group for pooled PP and ITT populations.a
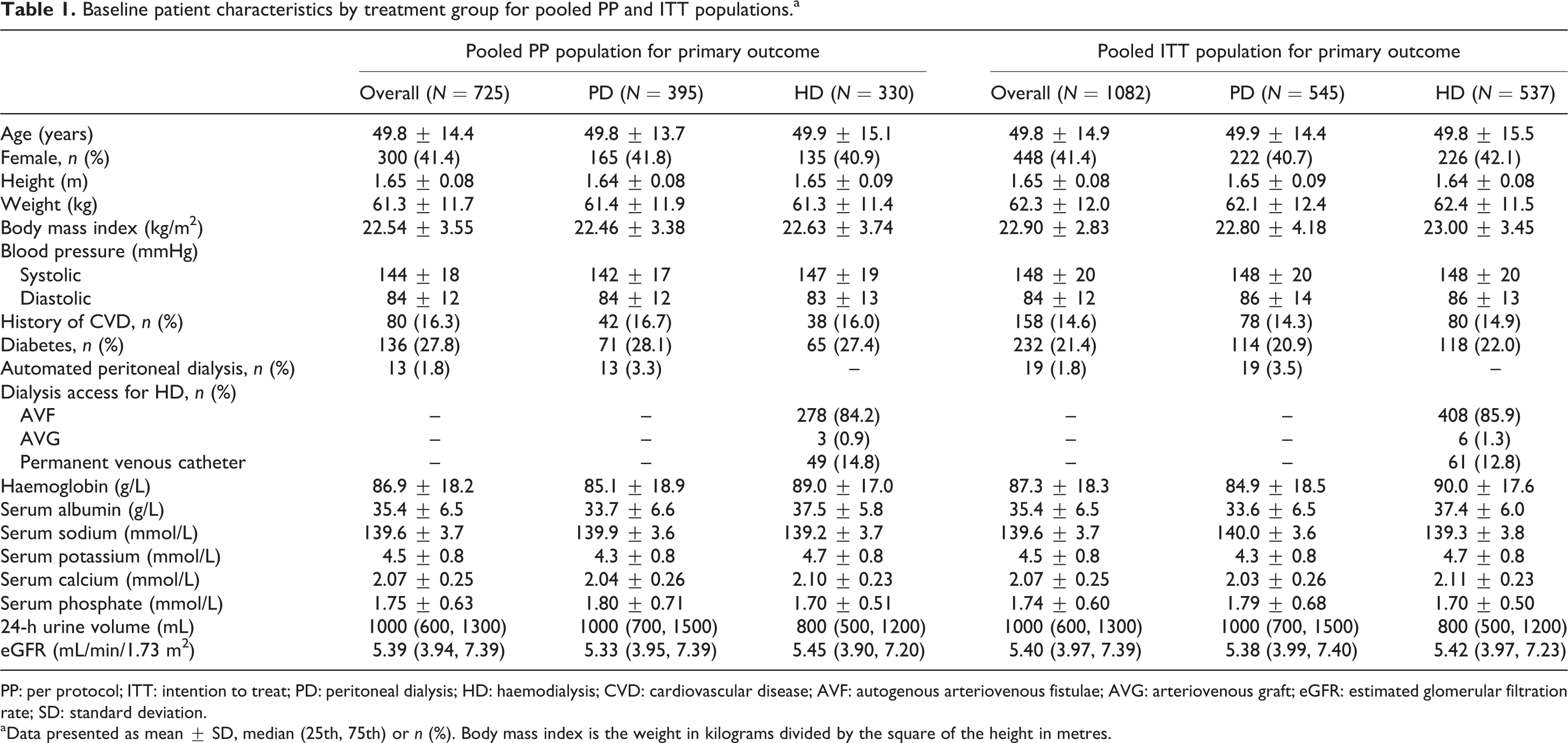
PP: per protocol; ITT: intention to treat; PD: peritoneal dialysis; HD: haemodialysis; CVD: cardiovascular disease; AVF: autogenous arteriovenous fistulae; AVG: arteriovenous graft; eGFR: estimated glomerular filtration rate; SD: standard deviation.
a Data presented as mean ± SD, median (25th, 75th) or n (%). Body mass index is the weight in kilograms divided by the square of the height in metres.
The mean (SD) change in the ‘Burden of Kidney Disease’ score over 48 weeks was 2.61 (1.27) in the PD group and 2.58 (1.35) in the HD group, in the pooled PP population. The difference (95% CI) in ‘Burden of Kidney Disease’ from baseline to 48 weeks was 0.03 (−3.29, 3.35) between the PD and HD groups. The GM ratio (95% CI) was 1.059 (0.908, 1.234), exceeding the limit for non-inferiority. Similar results were seen in the pooled ITT, phase I and phase II ITT and PP populations (Table 2 and Online Supplemental Table 9).
Non-inferiority test for difference between PD and HD in the changes in primary outcome over 48 weeks for pooled PP populations.a
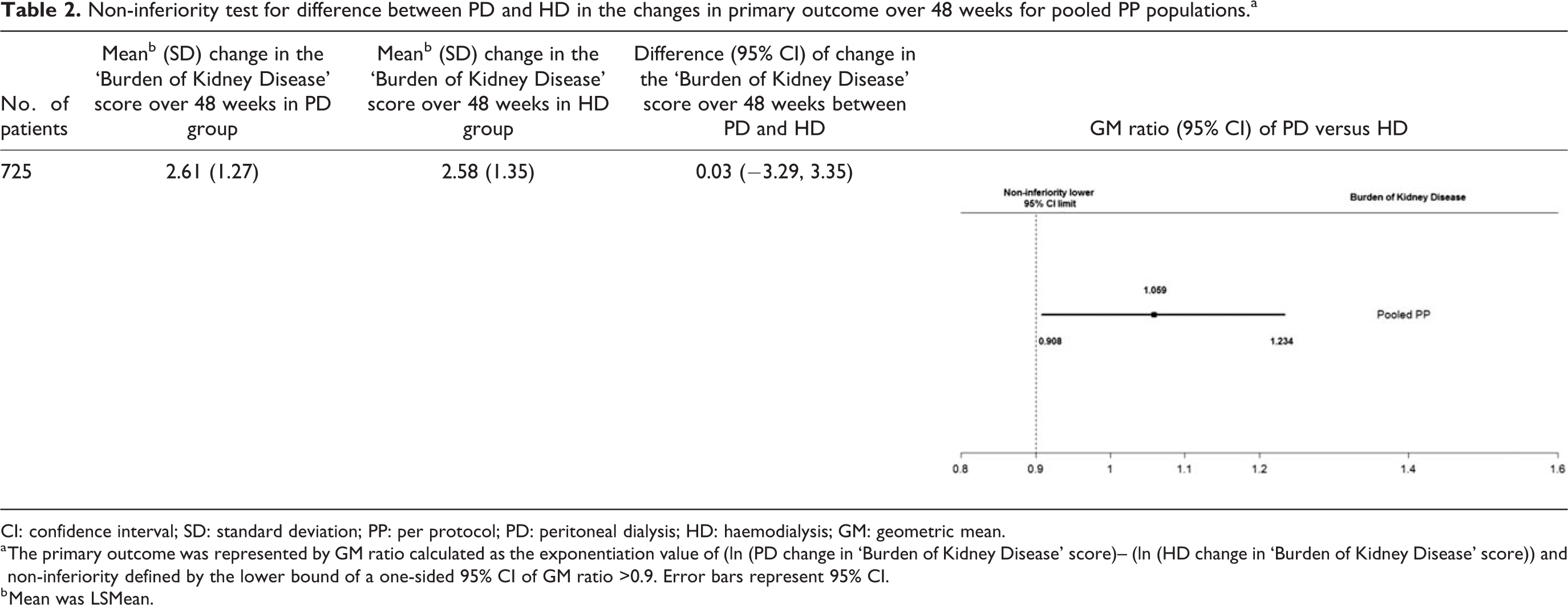
CI: confidence interval; SD: standard deviation; PP: per protocol; PD: peritoneal dialysis; HD: haemodialysis; GM: geometric mean.
a The primary outcome was represented by GM ratio calculated as the exponentiation value of (ln (PD change in ‘Burden of Kidney Disease’ score)– (ln (HD change in ‘Burden of Kidney Disease’ score)) and non-inferiority defined by the lower bound of a one-sided 95% CI of GM ratio >0.9. Error bars represent 95% CI.
b Mean was LSMean.
For the secondary outcomes, a total of 1082 pooled patients were available for the ITT analysis. With over 48 weeks of follow-up, the study found no differences in all scales (Figure 2(a)). Similar results were seen in the pooled PP and phase II ITT and PP populations (Figure 2(b) and Online Supplemental Figures 1 to 3). There were 17 (3.1%) deaths in the PD group and 31 (5.8%) deaths in the HD group (Online Supplemental Table 10). The most common causes of death were cerebrovascular and cardiovascular disease, affecting 15 (6 PD, 9 HD) and 9 patients (1 PD, 8 HD), respectively (Table 3).
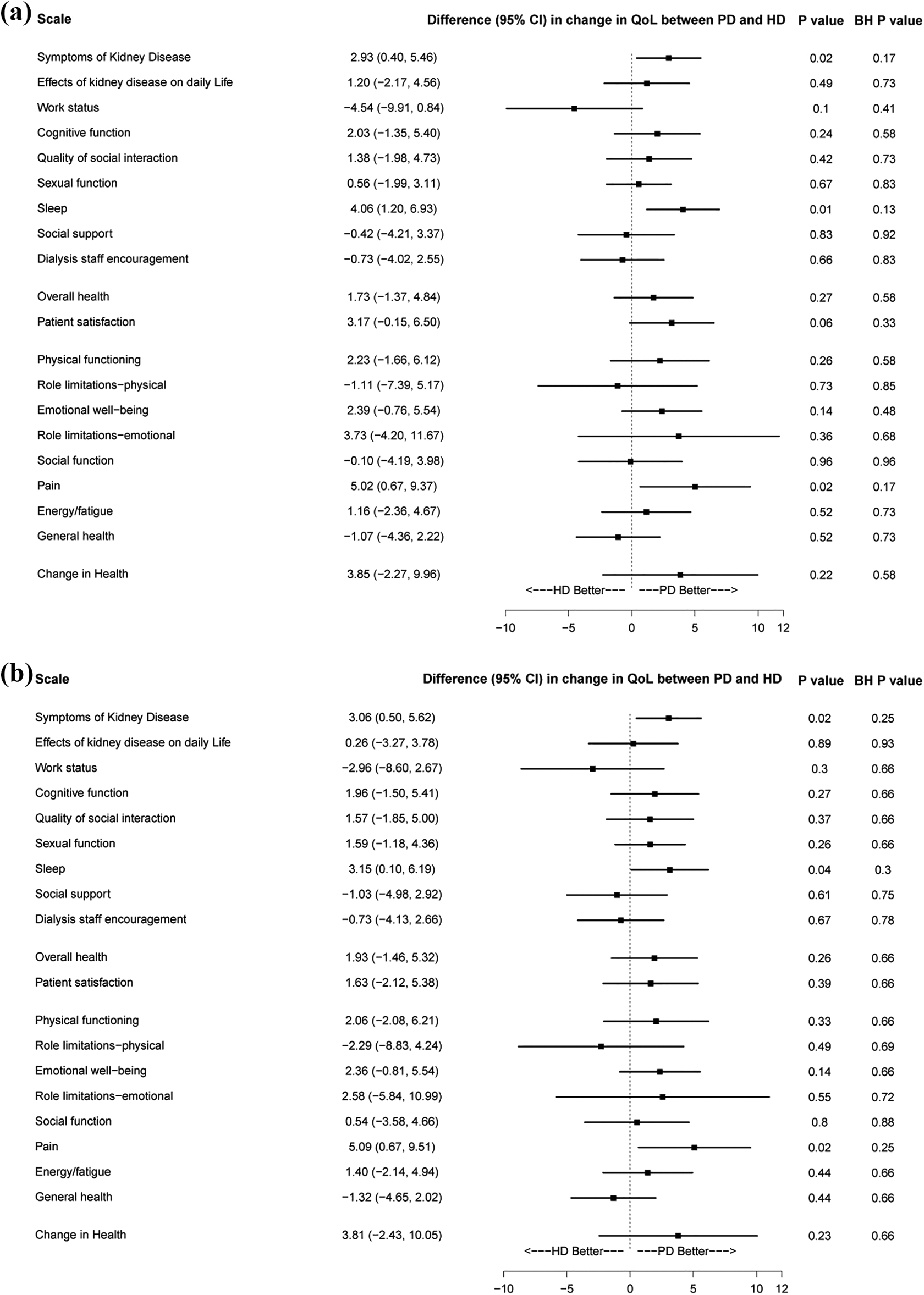
Secondary outcomes as defined by the difference in change in KDQoL score between PD and HD groups in pooled ITT (a) and pooled PP (b) populations. Change in KDQoL score was calculated as KDQoL score at 48 weeks minus KDQoL score at baseline. The difference in change between PD and HD was calculated as the KDQoL score of the PD group minus the KDQoL score of the HD group. The difference in change between PD and HD was compared using a generalised linear model. Benjamini–Hochberg method was used to adjusted multiple comparisons of p values. PP: per protocol; ITT: intention to treat; PD: peritoneal dialysis; HD: haemodialysis; KDQoL: Kidney Disease Quality of Life; BH p value: Benjamini–Hochberg adjusted p value.
Comparison of adverse events between PD and HD among pooled ITT and PP populations.
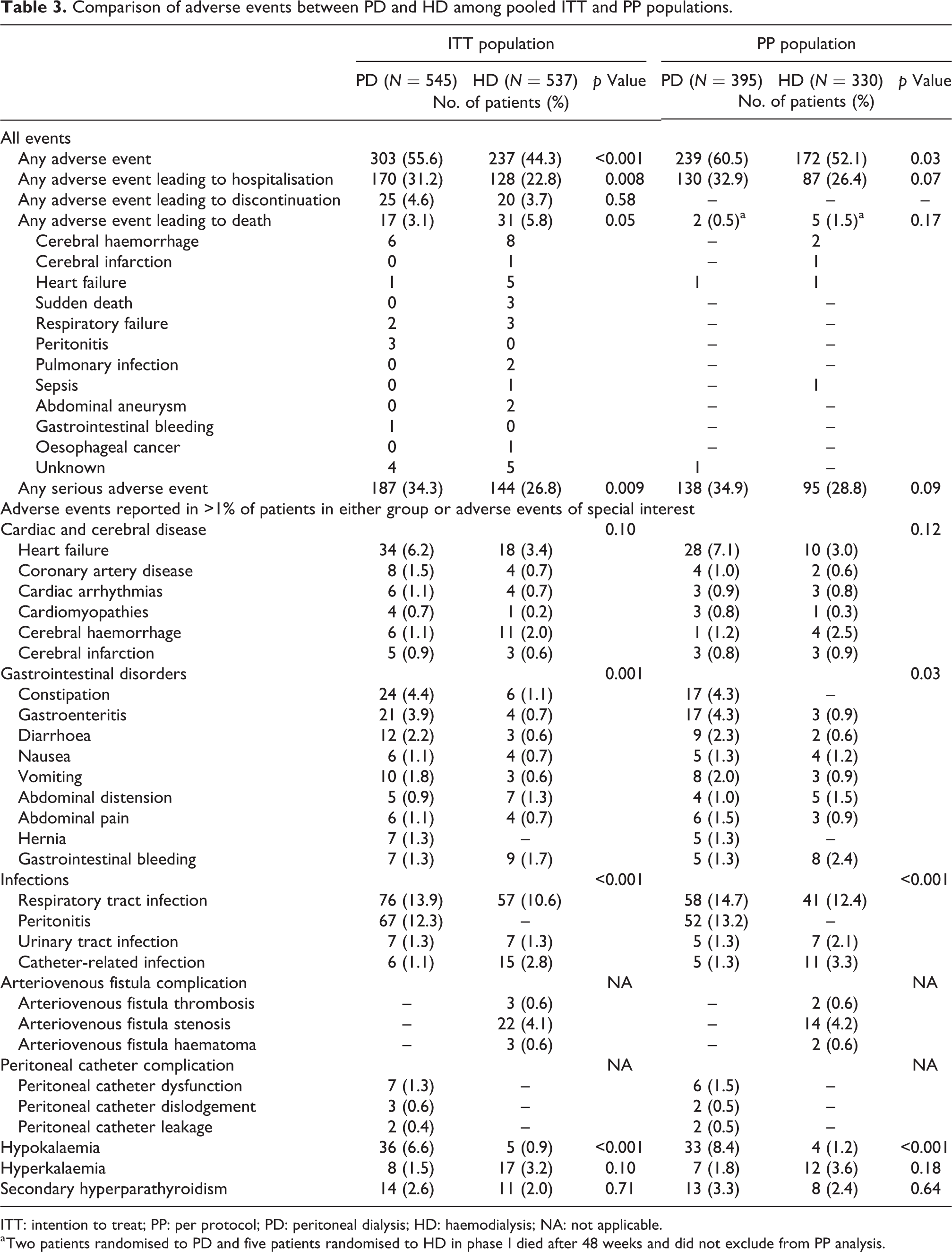
ITT: intention to treat; PP: per protocol; PD: peritoneal dialysis; HD: haemodialysis; NA: not applicable.
a Two patients randomised to PD and five patients randomised to HD in phase I died after 48 weeks and did not exclude from PP analysis.
There were 14 (1.3%) PD patients who permanently transferred to HD, and no HD patients were transferred to PD (Online Supplemental Table 10). Nine patients in the PD group and 23 patients in the HD group were undergoing kidney transplantation (Online Supplemental Table 10 and Online Supplemental Figure 4). During follow-up, 24-h urine volume gradually decreased, haemoglobin and serum albumin increased for both HD and PD patients and phosphate increased in HD patients but decreased in PD patients (Online Supplemental Figure 5). Similar trends were seen in pooled PP and phase I and phase II ITT and PP populations (Online Supplemental Figures 6 to 10).
There were 303 (55.6%) patients who reported adverse events in the PD group, and 237 (44.3%) in the HD group (p < 0.001). Adverse events leading to hospitalisation and serious adverse events were higher in the PD group compared to the HD group (p = 0.008 and 0.009, respectively), but adverse events leading to death and discontinuation of the trial were not different between HD and PD (Table 3; adverse events in phase I and phase II shown in Online Supplemental Tables 11 and 12). Cardiac and cerebral disease, hyperkalaemia and secondary hyperparathyroidism were not different between groups, but patients in the PD group had more gastrointestinal (GI) disorders, infection-related adverse events and hypokalaemia.
Discussion
To the best of our knowledge, this is by far the largest randomised, non-inferiority trial to compare HRQoL between conventional in-centre HD and home-based PD among patients with newly diagnosed kidney failure. It addresses an important evidence gap regarding the effect of dialysis modality on HRQoL in kidney failure patients.
The primary outcome of this trial is the ‘Burden of Kidney Disease’ scale of the KDQoL-SF, which assesses perceptions of frustration and interference of kidney disease in one’s life. The result of the trial showed that there is no significant difference in this scale between PD and HD. The difference of change in the ‘Burden of Kidney Disease’ between PD and HD was very small and was clinically meaningless. Despite being a home dialysis modality, and presumably more onerous in terms of work to both patients and their caregivers, PD was not inferior to HD in terms of perceived burden of disease and care. Our findings help to clarify the uncertainty from observational studies on this topic. There have been several systematic reviews summarising the effect of dialysis modality on patient-centred outcomes from the body of published literature to date. A meta-analysis showed that PD had higher health utility versus HD (effect size 0.03, 95% CI 0.00–0.06, p = 0.08). 10 One study observed superiority for PD versus HD for the ‘Effect of Kidney Disease’ scale of the KDQoL, 11 but others did not produce convincing conclusions, 12 or were published 10 years ago. 21 –23
For secondary outcomes, although remaining scales of KDQoL-SF were not significantly different between PD and HD after adjustment of multiple comparisons, our trial demonstrated PD may improve scores of ‘Symptoms of Kidney Disease’, ‘Sleep’ and ‘Pain’. The ‘Symptoms of Kidney Disease’ scale is axiomatically an important outcome, assessing symptom control in kidney failure in terms of issues relevant to patients on dialysis including soreness of muscles, pain, cramps during dialysis, bruising, itchy skin, shortness of breath, dizziness, lack of appetite, excessive thirst, numbness in hands or feet, nausea and problems with dialysis access. After dialysis initiation, symptoms of kidney disease were improved in both PD and HD, but PD slightly improved symptoms of kidney disease at 48 weeks, which may be related to preserved residual kidney function and better uremic toxin clearance in PD. The second scale assesses sleep quality, which is reduced for a large proportion of, if not most, dialysis patients. 24 It is also an important outcome, associated with increased mortality, and a leading priority for patients who are more likely to change dialysis modality for the prospect of better sleep rather than the prospect of lengthened life. 25,26 The last scale assesses the degree of bodily pain and the impact of pain on a patient’s normal work during the past 4 weeks. Pain is a common complaint in dialysis patients with a prevalence of 40–60% 27 and is associated with depression, sleep disturbances and an increased risk of mortality. 28,29 Further study should be done to assess the impact of dialysis modality on these three scales.
We noted that more patients in the HD group underwent kidney transplantation compared to those in the PD group, but the cause of this difference is not clear. Similar findings were also reported in the China Kidney Disease Network (CK-NET) 2015 annual data report, such that 79.35% of kidney transplantation candidates received HD, while 13.3% of them were treated with PD. 30 We also noted that there were 14 (1.3%) PD patients who permanently transferred to HD due to peritonitis and complications which may affect the quality of life of patients. We found that patients randomised to PD reported more adverse events and serious adverse events compared to those randomised to HD, but adverse events leading to death were lower than HD, although the difference was borderline. Adverse events leading to discontinuation of the trial were similar between the two groups. Patients in the PD group had more trouble in GI system, peritonitis and hypokalaemia compared to HD, indicating the need for attention and effort to reduce GI symptoms and prevent peritonitis in PD patients in order to improve their quality of life and long-term outcome.
Findings from our trial provide critically needed evidence to inform the clinical discussions between patients and providers concerning dialysis modality selection. Guidelines recommend that modality selection should be a shared decision between practitioners and patients, 31 although a well-acknowledged vulnerability of the process is whether information is framed in a balanced way when options are presented to patients. 32 Practitioners may have disparate and fixed opinions about patient suitability for PD or HD, 33 and this would seem that a significant proportion of modality decisions are still directive. Our current trial provides objective evidence from a randomised controlled trial, and hopefully patients and providers can be better informed about the pros and cons of PD and HD and make an informed choice about what is the best option when considering patients’ preference and experience.
The most important strength of our trial is that it is a randomised controlled trial conducted to evaluate patient-centred outcomes by dialysis modality, and it has the largest number of randomisations and a successful execution. This kind of trial is challenging to conduct because most kidney failure patients prefer to make their own treatment decision rather than agree to be randomised to PD or HD. One of two published randomised controlled trials was terminated due to challenges in patient recruitment. 16
Our trial is limited by a high number of screening failures and dropouts, mostly due to refusing randomisation, which raises concern for potential selection bias. To address this concern, we have compared the baseline characteristics of those included versus those excluded, and those who completed the trial versus those who refused or dropped out. The results showed baseline characteristics were not different. We have also conducted a series of sensitivity analyses. However, we could not exclude the influence of participants’ social and personal factors which could not be captured in baseline data on analysis. In addition, a total of 115 participants who withdrew consent after randomisation may not be truly randomised. We also could not exclude the bias due to unblinded outcome assessment, missing outcome data and open-label design, as well as the bias raised by the funder involved in the study design, although the funder has no role in study execution, data collection, analysis or interpretation. In terms of generalisability, our trial was performed in 36 centres across China, including a mixture of both large urban and smaller rural areas. As such, there is a reasonable generalisability of our findings to Chinese kidney failure patients. However, caution is needed when generalising our findings to other countries with different health service systems, cultures, lifestyles or different patient characteristics. In particular, automated PD was not common and receiving HD three times a week may not be available due to limited resources, which were relevant for less advanced countries. All HD were in-centre HD, icodextrin was not available and patients who urgent initiated dialysis were excluded. Our trial population was younger with a mean age of <50 years, 27.8% with diabetes, less than 20% of patients had a history of cardiovascular disease and had good residual kidney function. In addition, there was no haemodiafiltration used in the trial, which may have a corresponding negative effect on the HD arm. Second, our primary end point of Burden of Kidney Disease consisted of only four items of KDQoL-SF questionnaire and could not comprehensively evaluate the impact of dialysis modality on remaining scales of HRQoL. Third, baseline HRQoL was measured after 8 weeks of preparation to start dialysis and may exclude the impact of dialysis access creation and preparation on ‘Burden of Kidney Disease’. HRQoL score was only assessed at baseline and at 48 weeks, therefore we might have lost the information if the patients dropped out during follow-up. In addition, HRQoL score was missing due to adverse event, death, transferred from PD to HD and kidney transplantation and may exclude the impact of adverse event on ‘Burden of Kidney Disease’. Finally, our trial only had 1 year of follow-up after dialysis inception, and longer term differences between PD and HD on HRQoL could not be evaluated.
In conclusion, this randomised controlled trial demonstrated that PD may be non-inferior to HD on the ‘Burden of Kidney Disease’ among Chinese kidney failure patients who are of younger age and have lower comorbidity after 48 weeks’ follow-up. Further studies with longer follow-up are needed to evaluate the impact of PD versus HD on patient survival and longer term HRQoL.
Supplemental material
Supplemental Material, sj-docx-1-ptd-10.1177_08968608221088638 - Burden of kidney disease among patients with peritoneal dialysis versus conventional in-centre haemodialysis: A randomised, non-inferiority trial
Supplemental Material, sj-docx-1-ptd-10.1177_08968608221088638 for Burden of kidney disease among patients with peritoneal dialysis versus conventional in-centre haemodialysis: A randomised, non-inferiority trial by Li Fan, Xiao Yang, Qinkai Chen, Hao Zhang, Jianqin Wang, Menghua Chen, Hui Peng, Zhaohui Ni, Jianxin Wan, Hongtao Yang, Yun Li, Li Wang, Ai Peng, Hongli Lin, Jinyuan Zhang, Huaying Shen, Fei Xiong, Yongcheng He, Yan Zha, Minyan Xie, Jundong Jiao, Gengru Jiang, Xunhuan Zheng, Jun Xiao, Rong Rong, Jiaqi Qian and Xueqing Yu in Peritoneal Dialysis International
Footnotes
Acknowledgement
The authors would like to thank the participating patients and their families, clinicians and trial investigators.
Author contributions
XQY conceived the study and contributed to study design. XQY and JQQ were chief investigators and LF, XY, QKC, HZ, JQW, MHC, HP, ZHN, JXW, HTY, YL, LW, AP, HLL, JYZ, HYS, FX, YCH, YZ, MYX, JDJ, GRJ, XHZ, JX and RR contributed to local implementation of the trial, screening, enrolment and patient follow-up and data collection. LF and XQY drafted and revised the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Data sharing
All individual participant data are available (including data dictionaries). Individual participant data that underline the results reported in this article (text, tables, figures and appendices), after de-identification, will be shared. Study protocol and the statistical analysis plan are available as Supplemental Material. Sharing data will be available starting when the article is published and ending at 36 months following the publication of this article and can be used for individual participant data meta-analysis and other analysis. Investigators whose proposed use of the data has been approved by an independent review committee will have access to use the sharing data. Proposals for sharing data should be sent to
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: XQY has received grant/research support from National Natural Science Foundation of China, Baxter, Wanbang Biopharmaceuticals, AstraZeneca and GSK and consulting fees and lecture fee from Baxter, Wanbang Biopharmaceuticals, Fresenius, Fresenius Kabi and Kyowa Kirin. All other authors declare no support from any organisation for the submitted work, no financial relationships with any organisations that might have an interest in the submitted work in the previous 3 years and no other potential conflict of interest relevant to this article.
Ethical approval
The trial protocol was approved by the ethics committees of all participating centres. The ethical approval for this study of leading site was obtained from the Medical Ethical Committee of The First Affiliated Hospital, Sun Yat-sen University (approval number: [2011]153 and [2014]109).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This trial was funded by Baxter Healthcare Corporation. The sponsor of the study was involved in the study design, but no role in study execution, data collection, analysis or data interpretation. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Informed consent to participate
Written informed consent was obtained from all subjects before the study.
Informed consent to publish
Written informed consent was obtained from the patients for their anonymised information to be published in this article.
Supplemental material
Supplemental material for this article is available online.
Trial registration
Clinical Trials.gov number: NCT01413074, NCT02378350.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
