Abstract
Introduction:
Glucose absorption during peritoneal dialysis (PD) is commonly assumed to occur via paracellular pathways. We recently showed that SGLT2 inhibition did not reduce glucose absorption in experimental PD, but the potential role of glucose transport into cells is still unclear. Here we sought to elucidate the effects of phlorizin, a non-selective competitive inhibitor of sodium glucose co-transporters 1 and 2 (SGLT1 and SGLT2), in an experimental rat model of PD.
Methods:
A 120-min PD dwell was performed in 12 anesthetised Sprague-Dawley rats using 1.5% glucose fluid with a fill volume of 20 mL with (n = 6) or without (n = 6) intraperitoneal phlorizin (50 mg/L). Several parameters for peritoneal water and solute transport were monitored during the treatment.
Results:
Phlorizin markedly increased the urinary excretion of glucose, lowered plasma glucose and increased plasma creatinine after PD. Median glucose diffusion capacity at 60 min was significantly lower (p < 0.05) being 196 µL/min (IQR 178–213) for phlorizin-treated animals compared to 238 µL/min (IQR 233–268) in controls. Median fractional dialysate glucose concentration at 60 min (D/D 0) was significantly higher (p < 0.05) in phlorizin-treated animals being 0.65 (IQR 0.63–0.67) compared to 0.61 (IQR 0.60–0.62) in controls. At 120 min, there was no difference in solute or water transport across the peritoneal membrane.
Conclusion:
Our findings indicate that a part of glucose absorption during the initial part of the dwell occurs via transport into peritoneal cells.
This is a visual representation of the abstract.
Introduction
Ultrafiltration (UF) during glucose-based peritoneal dialysis (PD) is dependent on a supraphysiological glucose concentration in the dialysate, leading to unwanted glucose absorption during the treatment. Several techniques have been proposed to reduce glucose absorption during glucose-based PD, such as icodextrin, 1 alternate PD fluids 2 or by altering the prescription. 3 –6
Glucose is a polar compound and is therefore impermeable across cell membranes. In the small intestine, SGLT1 transporters mediate nearly all sodium dependent glucose and galactose uptake (1 Na+ for each glucose molecule) but has only a minor role in kidney glucose handling. In contrast, SGLT2 represents the major transport pathway for glucose in the early proximal tubule where sodium and glucose (2 Na+ for each glucose) co-transport occurs against the glucose gradient to allow effective glucose reabsorption from the glomerular filtrate.
It is commonly assumed that glucose is absorbed via paracellular pathways, presumably across ‘small pores’ 7 during PD. Also, in a recent study, we found that SGLT2 inhibition did not appear to reduce glucose absorption during experimental PD in the acute setting. 8 Nevertheless, it cannot be excluded that other glucose transporters such as SGLT1 or facilitative glucose transporters (GLUT) contribute to glucose transport during PD. Indeed, several studies have shown that SGLT and GLUT subtypes are expressed in human peritoneal mesothelial cells 9 and in rat peritoneal mesothelial cells. 10 Here we used an experimental rat model of acute PD with or without administration of phlorizin, an inhibitor of sodium driven glucose co-transport via SGLT-1 and SGLT-2 to test the hypothesis that glucose transport into peritoneal cells significantly reduces glucose absorption and/or glucose diffusion capacity during experimental PD.
Methods
Experimental PD was performed in 12 adult male 9-week-old Sprague-Dawley rats (Möllegard, Lille Stensved, Denmark) having median body weight of 293 g (min–max: 257–334) with free access to food (Special Diets Services RM1(P) IRR.25 801157) and water ad libitum. The Ethics Committee for Animal Research at Lund University approved the current studies (2018-05-23, dnr 5.8.18-05699/2018), and the animals were treated in accordance with the guidelines of the National Institutes of Health for Care and Use of Laboratory animals.
Surgery and anaesthesia
The surgical procedure and experimental PD set-up was similar to that in our recent study on empagliflozin 8 with the difference that phlorizin was mixed into the dialysis fluid as described below instead of given as an intravenous infusion. A schematic of the experimental set-up is shown in Figure 1. The animals were firstly placed in a glass container to which a continuous supply of 5% isoflurane in air (Isoban, Abbot, Stockholm, Sweden) was administered. After induction of anaesthesia, the animal was removed from the container, and anaesthesia was maintained with 1.6% to 1.8% isoflurane in air delivered via a mask. A tracheostomy was thereafter performed after which the animals were connected to a ventilator (Ugo Basile; Biological Research Apparatus, Comerio, Italy). End-tidal pCO2 was monitored continuously using a capnograph (Capstar-100, CWE, Ardmore, Pennsylvania, USA) and was maintained between 4.8 and 5.5 kPa. Body temperature was maintained between 37.1°C and 37.3°C using a feedback-controlled heating pad. The left femoral artery was utilised for the measurement of mean arterial pressure (MAP) and heart rate and to draw blood samples (95 µL) for the measurement of creatinine, urea, glucose, electrolytes and haemoglobin (i-STAT, Abbot, Abbot Park, Illinois, USA). Access via the right femoral vein was established for infusion of a saline infusion (3 mL h−1) with creatinine (1 mg mL−1). This small amount of creatinine ensured that plasma values were higher than lower limit of detection for creatinine on the i-STAT instrument. Access to the peritoneal cavity was established percutaneously by means of a multiholed silastic catheter (Venflon, BOC Ohmeda AB, Helsingborg, Sweden) (outer diameter 1.7 mm) which was tightly secured to the skin using cyanoacrylate (Histoacryl, B. Braun Surgical, Rubi, Spain). Urine was collected in a pre-weighed vial during the course of the PD dwell (120 min) after which urinary output and glucose concentration was assessed (i-STAT). After the PD dwell, the dialysate was quickly recovered from the peritoneal cavity, firstly by extraction via the PD catheter using a syringe. Residual fluid was thereafter gently retrieved from the peritoneal cavity using pre-weighed gauze tissues. For UF determination, a residual volume of 3 mL was assumed. 11 All PD solutions were pre-warmed to 37°C before use. After the experiment, animals were euthanised by an intravenous bolus injection of potassium chloride. Directly before dialysate sampling, a 1-mL bolus of the dialysate was flushed back and forth several times.
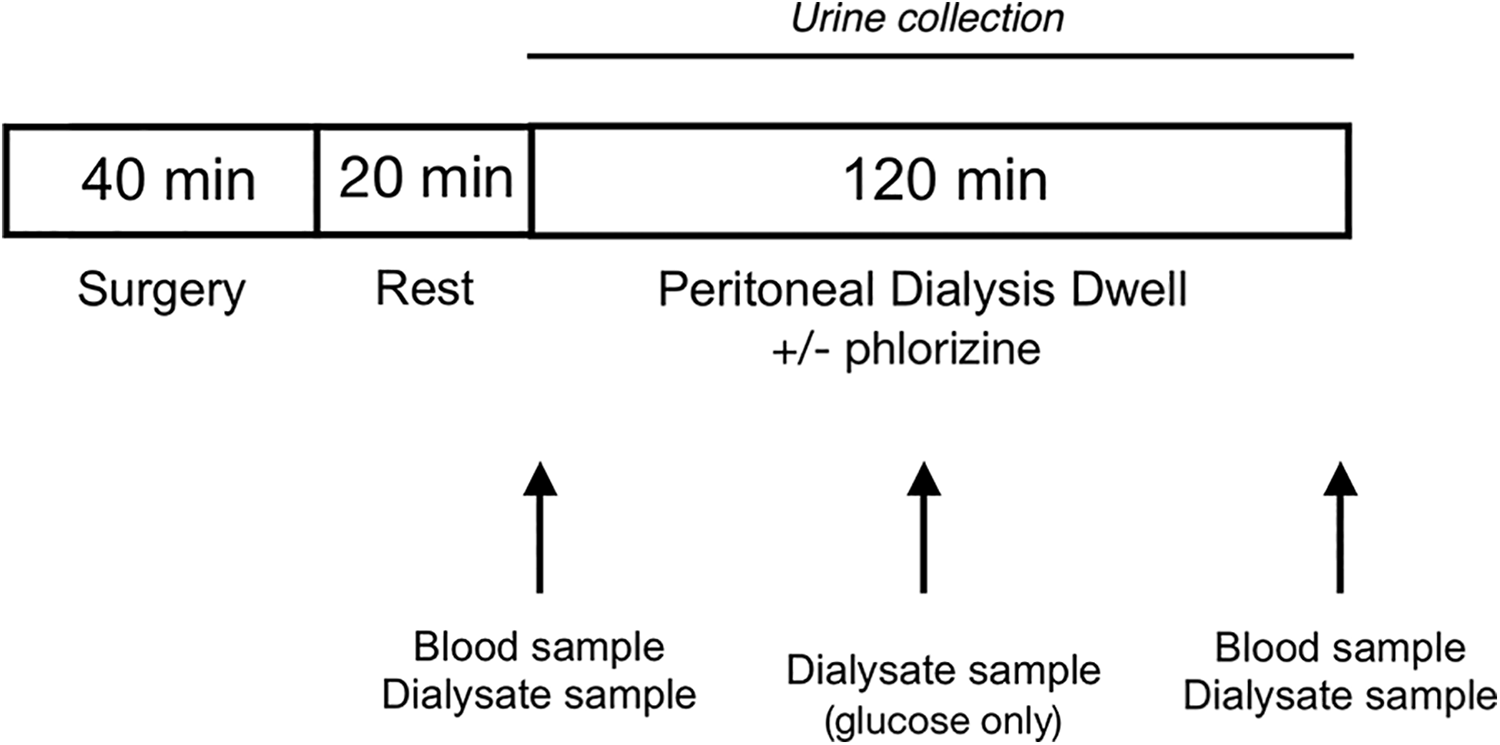
Schematic of the experimental set-up. PD with 1.5% glucose fluid with (n = 6) or without (n = 6) phlorizin was performed in anesthetised Sprague-Dawley rats using a fill volume of 20 mL. Sampling of the dialysate occurred at baseline and at 0 min, 60 min and 120 min. Blood samples were taken before and after PD. PD: peritoneal dialysis.
Experimental protocol
In the intervention group (n = 6), phlorizin dihydrate from apple wood (Merck, Darmstadt, Germany) was mixed with 1.5% PD fluid (BicaVera, Fresenius Medical Care, Frankfurt, Germany) to achieve a concentration of 50 mg/L. Sham groups received plain 1.5% glucose fluid (n = 6). Samples from the dialysate were collected before and immediately after filling of PD fluid and after 60 min and 120 min. After 120 min dwell time, the dialysate was drained after which a post-dialysis blood sample was collected. Plasma-to-dialysate clearances were calculated as the average solute removal rate (mmol/min) divided by the mean plasma concentration (mmol/L). Dialysate-to-plasma clearances were calculated as the mean solute absorption rate (mmol/min) divided by the average dialysate concentration (mmol/L). Isovolumetric and isocratic diffusion capacities were calculated as described previously. 8 A modified version of the three-pore model by Rippe 7 was used to analyse water and solute transport during PD. The parameters used (adapted to the rat) are shown in Supplemental Table 1.
Statistical methods
Statistical calculations were performed using R for mac version 4.1.1. Statistical differences between groups were assessed using a Wilcoxon–Mann–Whitney test (wilcox.test function). p Values below 0.05 were considered significant. Unless otherwise stated, values are given as median (IQR).
Results
Systemic effects of intraperitoneal phlorizin
As shown in Table 1, intraperitoneal phlorizin (50 mg/L) resulted in systemic effects in the form of marked glucosuria, increased urinary output and lowered plasma glucose. The difference in plasma sodium before and after dialysis (before–after) was 0.5 mmol/L (0–1.0) in sham animals compared to 2.5 mmol/L (2.0–3.8) in phlorizin-treated animals (p < 0.05). Chloride concentrations were similarly elevated in phlorizin-treated animals (p < 0.05), whereas there was no difference in total CO2 concentrations after dialysis (data not shown).
Effects of phlorizin on urinary output and glucose excretion, plasma creatinine and plasma glucose.
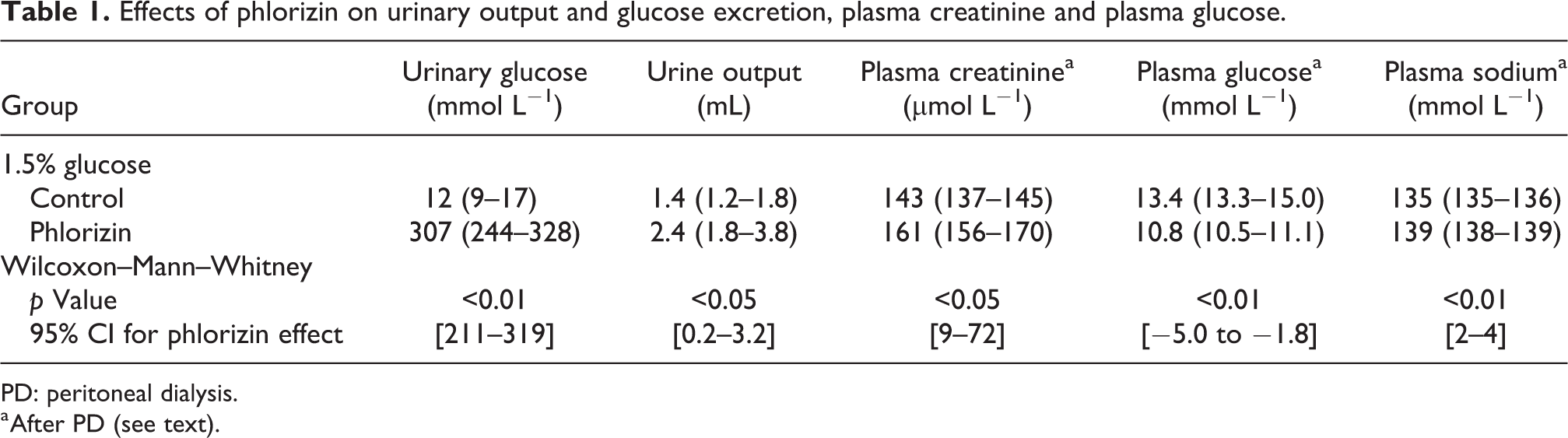
PD: peritoneal dialysis.
a After PD (see text).
Effects on glucose transport parameters
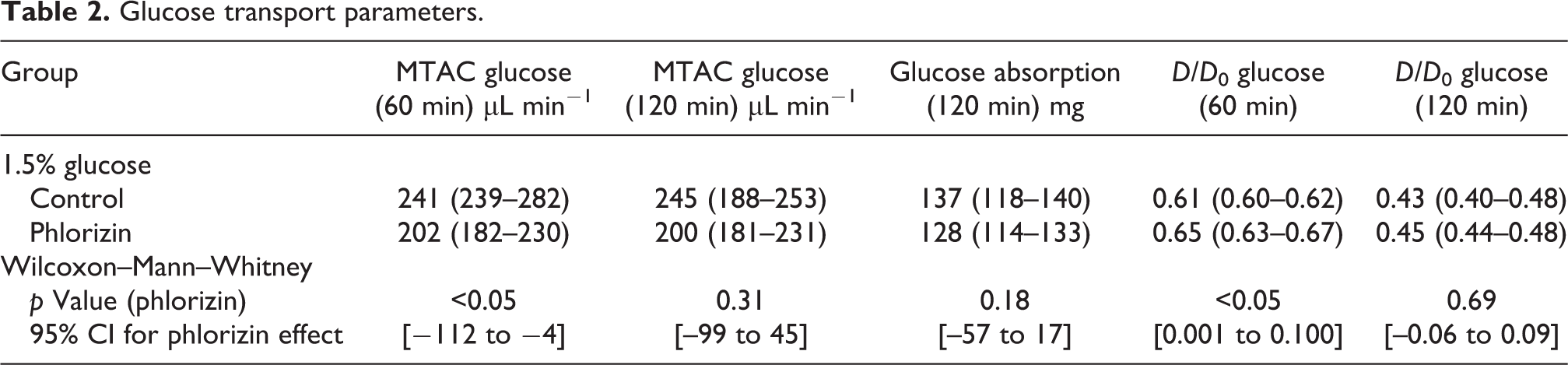
Fractional dialysate (D/D 0) glucose concentration at 60 min was significantly higher in phlorizin-treated animals, whereas there was no difference at 120 min, nor any other glucose transport parameters (Table 2). Isocratic diffusion capacities (MTACiso) calculated for the 60 min glucose concentration was 241 µL/min (IQR 239–282) for sham animals compared to 202 µL/min (IQR 182–230) for phlorizin-treated animals (p < 0.05), indicating a phlorizin-induced reduction of glucose MTACiso of −112 to −4 µL/min (95% confidence interval). This is in contrast to 120 min MTACiso which did not differ significantly between groups (Table 2).
Glucose transport parameters.
Effects on UF and sodium transport
The UF rate was not significantly different in phlorizin-treated animals compared to controls (Table 3). Sodium clearance was not significantly increased in phlorizin-treated animals (p = 0.06) with a median value of 21 µL min−1 compared to 13 µL min−1 in controls which both corresponded to about 80% of the UF rate in their respective groups (Table 3). In terms of UF efficacy, control animals had a UF volume of 17 mL per gram glucose absorbed (IQR 13–20) compared phlorizin-treated animals having 24 mL UF per gram glucose absorbed (IQR 16–30) with no difference between groups (95% CI 19 to −11).
Sodium and osmotic water transport parameters.
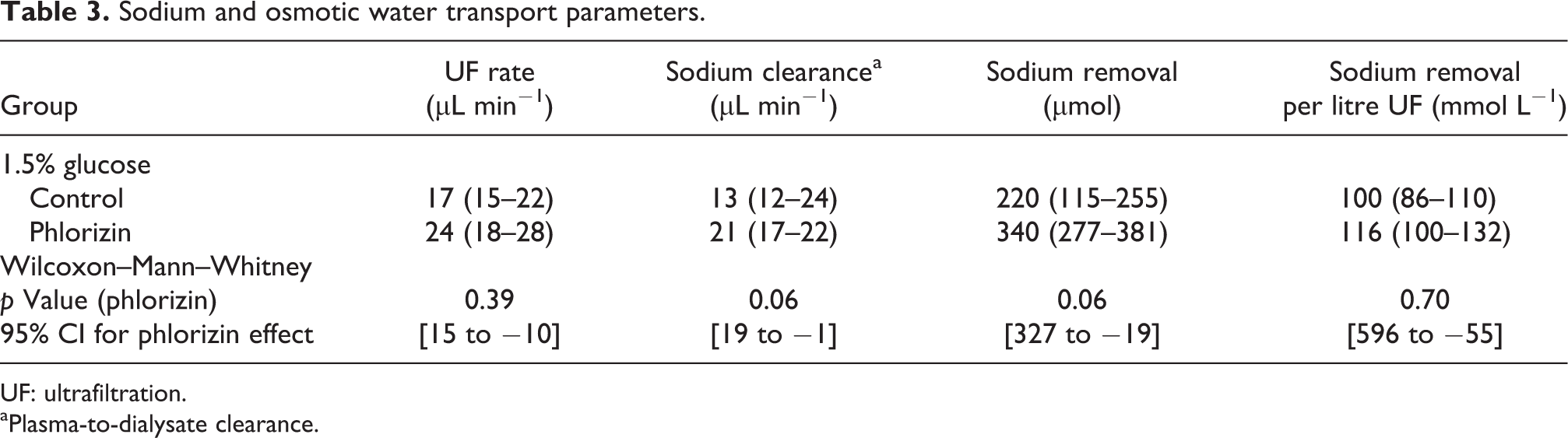
UF: ultrafiltration.
aPlasma-to-dialysate clearance.
Three-pore model analysis
The finding of a lower diffusion capacity for glucose at 60 min was replicated using the three-pore model (Supplemental Table 2). There were no differences in water transport parameters in terms of hydraulic conductance or osmotic conductance to glucose. Shown in Figure 2 is a forest plot showing 95% confidence intervals for small solute diffusion capacities (MTAC), UF rate and sodium clearance. The urea MTAC was 400 µL min−1 (IQR 363–527) in control animals compared to 339 µL min−1 (IQR 305–367) in phlorizin-treated animals (p = 0.09). The creatinine MTAC was 201 µL min−1 (IQR 136–237) in control animals compared to 168 µL min−1 (IQR 149–203) in phlorizin-treated animals (p = 0.59). For potassium, the MTAC was 450 µL min−1 (IQR 422–498) in control and 442 µL min−1 (IQR 387–471) in the phlorizin group (p = 0.70).
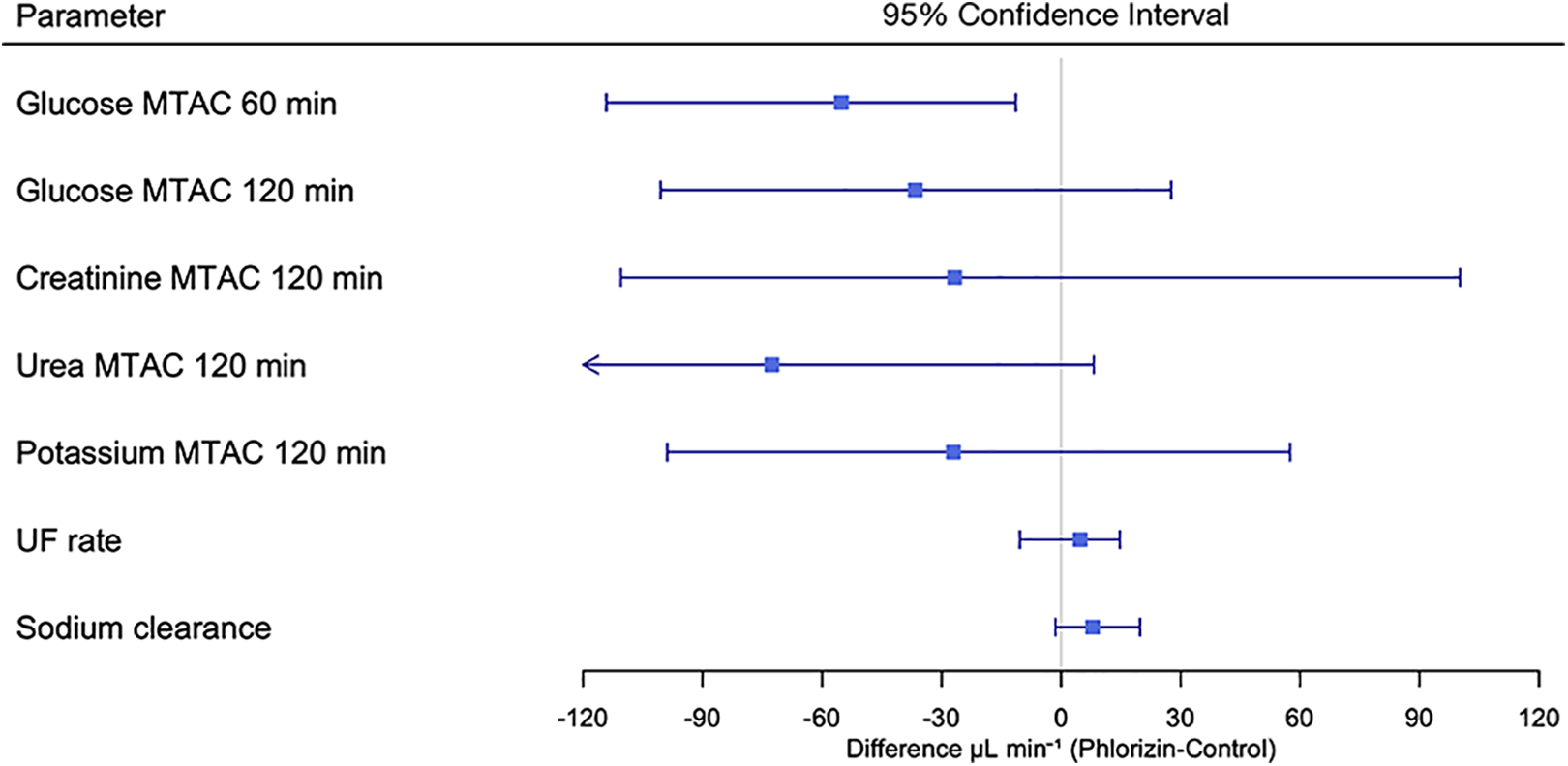
Forest plot showing 95% confidence intervals of the differences between phlorizin and control groups in small solute diffusion capacities (obtained using the three-pore model), the UF rate and sodium clearance. UF: ultrafiltration.
Discussion
The main finding in this study is that the glucose diffusion capacity during the initial part of the PD dwell was reduced by dual SGLT1/SGLT2 inhibition by phlorizin, indicating that part of the glucose is absorbed into peritoneal cells via sodium–glucose co-transporters. The reason that the glucose sparing effect of phlorizin occurred only during the initial 60 min of the treatment may simply be due to the fact that intraperitoneal levels of phlorizin will be lower during the latter part of the dwell due to diffusion of the relatively small phlorizin molecule (∼436 Da) into the circulation. The lack of an effect on glucose absorption using SGTL2 inhibition in our previous paper, 8 and the fact that phlorizin does not affect facilitative glucose transporters, 12 would suggest that the present effects are due to the inhibition of SGLT1.
UF is crucial for PD patients in order to maintain euvolemia. The mechanisms behind ultrafiltration insufficiency (UFI) 13 were recently reviewed by Krediet, 14 and it was hypothesised that glucose uptake into cells may play a role in acquired UFI together with other processes that reduce peritoneal water permeability (fibrosis and vasculopathy) and osmotic conductance (reduced aquaporin 1 (AQP1) expression). Indeed, in a recent study 15 on a large number of PD patients, Morelle et al. showed that a common variant of AQP1 is associated with decreased UF and increased risk of death or technique failure. Furthermore, a recent genome-wide association study of patients treated with PD suggests a possible genetic contribution to inter-individual variability in small solute diffusion capacities. 16
Expectedly, intraperitoneal administration of phlorizin had very similar systemic effects to that of SGLT2 inhibitors such as empagliflozin 8 or ipragliflozin, 17 acting as an effective osmotic diuretic. Thus, in the present experiments, phlorizin increased urine output almost two-fold and increased plasma sodium and chloride concentrations indicating potentiated free water clearance in the kidney. 17 The diffusion capacities calculated at 120 min were nearly identical to those obtained at 60 min indicating the absence of a Waniewski effect 18 in this experimental model of PD. This may be due to the fact that the rats are naïve to PD, and therefore do not respond in the same manner to PD fluid compared to PD patients.
The present study has several limitations. Firstly, its acute nature means that chronic effects cannot be studied. For example, in a recent study, Balzer and colleagues showed beneficial effects of SGLT2 inhibition in a mouse model of chronic PD treatment. 19 It is well-known that the inflammatory state in the peritoneal tissues in freshly started PD patients elicits higher diffusion capacities, which may, in the present experiments have partially masked the effect due to phlorizin. Furthermore, the non-selective nature of phlorizin means that we cannot determine if the observed effects on glucose diffusion are due to inhibition of a single glucose channel or if several transporters are involved. Also, the anaesthetic agent used (isoflurane) may induce changes in glucose metabolism as is exemplified by the hyperglycaemia present already at baseline, which is caused by the anaesthesia. 8 Regardless of phlorizin administration, three-pore model estimations of hydraulic conductance and osmotic conductance to glucose were similar, but somewhat higher compared to our previous study. 8 These differences may, in part, be due to the fact that residual volumes were not taken into account in the present study, which may cause differences in volumetric assessments. Intraperitoneal administration of phlorizin was chosen since this is more realistic in a clinical application; however, the use of intravenous administration as in our previous study 8 could have clarified whether the lack of an effect in the latter part of the dwell was a result of phlorizin diffusing from the peritoneal cavity. Lastly, the small sample size of only six animals in each group is a major limitation of this study since effect sizes will need to be very large in order to give significant results. Indeed, 95% confidence intervals for several non-significant parameters (e.g. UF and sodium removal) were clearly asymmetric in favour of a phlorizin effect.
Our data showed that phlorizin have effects on glucose diffusion capacity during the initial part of the dwell during experimental PD in Sprague-Dawley rats. Although there were no significant effects at 120 min, confidence intervals and median values showed similar trends compared to 60 min values, suggesting that the effect size may have been too small to allow detection in such a small number of animals. Further research could be focused on SGLT1 inhibition or inhibitors of facilitative glucose transporters in rats. In contrast to SGLT2 inhibitors, such agents have the advantage that they do not cause activation of tubuloglomerular feedback and lowering of the glomerular filtration rate which could be potentially harmful in PD patients with low residual kidney function.
Supplemental material
Supplemental Material, sj-docx-1-ptd-10.1177_08968608221080170 - Dual SGLT1/SGLT2 inhibitor phlorizin reduces glucose transport in experimental peritoneal dialysis
Supplemental Material, sj-docx-1-ptd-10.1177_08968608221080170 for Dual SGLT1/SGLT2 inhibitor phlorizin reduces glucose transport in experimental peritoneal dialysis by Giedre Martus, Karin Bergling and Carl M Öberg in Peritoneal Dialysis International
Footnotes
Acknowledgements
None.
Author contributions
GM and CAÖ researched literature and conceived the study. GM and CAÖ was involved in protocol development and data analysis. CAÖ wrote the first draft of the manuscript. KB and CAÖ performed three-pore model simulations. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from Ethics Committee for Animal Research at Lund University.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant by the Thure Carlsson foundation and Region Skåne (Medical Training and Research Agreement [Avtal om Läkarutbildning och Forskning; ALF]).
Informed consent
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
