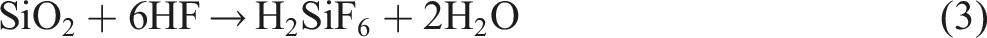Abstract
The etching of polymer surfaces has attracted considerable attention among polymer scientists and engineers owing to its wide range of applications in electronics and photovoltaic devices. In this work, the synergistic effects of HF etching and γ-irradiation at different doses (15, 25, and 35 kGy) on the structural and dielectric properties of PVA/SiO2/Ag nanocomposites are systematically investigated. The chemical structure of the nanocomposites was analysed using Fourier transform infrared spectroscopy (FTIR). X-ray diffraction (XRD) results revealed an enhancement in the amorphous regions of the nanocomposites upon irradiation at 15 and 35 kGy. Scanning electron microscopy (SEM) images revealed a porous surface morphology, with pore size strongly dependent on the applied irradiation dose. The dielectric properties were found to be highly sensitive to γ-radiation exposure. Among all samples, the HF/PVA/SiO2/Ag nanocomposite irradiated at 15 kGy exhibited the highest AC conductivity (σac), dielectric constant, and dielectric loss. Quantitatively, this sample showed a maximum concluded DC conductivity (σdc) of 1.76 × 10−6 S·cm−1, which is approximately one order of magnitude higher than that of the unirradiated composite (1.84 × 10−7 S·cm−1). This enhancement is attributed to increased amorphous content and improved charge carrier mobility, facilitating more efficient hopping conduction pathways. In contrast, the sample irradiated at 25 kGy displayed a significant reduction in electrical performance, indicating that excessive irradiation may induce radiation-induced crosslinking. These findings demonstrate that γ-irradiation is an effective and controllable approach for tailoring the structural and dielectric properties of etched polymer nanocomposites, making them promising candidates for applications in energy storage, radio-frequency devices, and optoelectronics.
Introduction
Nanocomposites present a promising avenue for a novel category of materials that synergistically combine the intrinsic characteristics of dispersed inorganic nanoparticles and host organic polymers. There has been a surge in the inclination towards nanocomposite materials that incorporate inorganic nanoparticles into polymer matrices in recent times.1–5 Due to their inherent flexibility and low weight, polymers are ideal for producing nanocomposites, which can be readily processed and molded to meet specific size and shape requirements. Hence, the limitations of manipulating nanostructured matters can be surmounted by utilizing a polymer as a host matrix to incorporate nanoparticles.
Polyvinyl alcohol (PVA) is the world’s leading product of water-soluble synthetic resins. PVA resins’ physical, chemical, and biodegradable qualities make them useful. PVA is odorless and colorless. It forms films, emulsifies, and sticks. PVA’s strong tensile strength, flexibility, and barrier qualities make it a popular thin film material. PVA is used in textiles, surgical equipment, paper, food packaging, electronics, radiation shielding, coatings, building materials, and packaging. PVA makes thin, stable nanocomposite films. 6 The backbone of its carbon chain is composed of methane carbons linked to hydroxyl (-OH) groups. 7
Silicon is a widely exploited semiconductor owing to its advantageous properties, including a large bandgap, a broad optical transmission window, a wide absorption spectrum, controllable surface roughness, and effective anti-reflection capabilities. Consequently, silicon has been widely used in photonics, 8 lithium‐ion batteries, 9 solar cells 10 and polymer composites.3,4,11
Organic-inorganic hybrid networks, such as the PVA-SiO2 system, demonstrate advantageous characteristics derived from both the inorganic and organic components, such as biocompatibility, thermal stability, chemical and corrosion resistance, rigidity, non-toxicity, elasticity, processability, flexibility, and high water permeability. 12 PVA/SiO2 recently used in various applications like solar cell, 13 sensors, 14 and food packaging. 15
The exceptional properties of silver nanoparticles (AgNPs) have been extensively explored across a wide range of applications, including antibacterial agents, 12 sensors, 16 biosensors, photocatalysis,17,18 and biomedical applications. 19 Owing to their distinctive optical, electrical, magnetic, and catalytic characteristics, AgNPs are also highly effective as functional fillers for polymer matrices. In particular, their incorporation into polymers such as PVA significantly enhances mechanical strength, thermal stability, and barrier performance. 20
The targeted removal of material from polymeric surfaces, commonly called selective etching, has garnered significant attention from polymer scientists and engineers. This is primarily due to its potential applications in electronics and photovoltaics. The primary objectives of selective etching include enhancing surface roughness of polymeric materials, modifying surface chemical composition, eliminating or dissolving low molecular weight species that migrate to the surface, and alleviating residual surface stresses. 21
Etching may change the shape of the material polymer surface and its chemical characteristics. Etching polymers before metal plating is a primary commercial use. Polymeric surfaces significantly impact metal plate adhesion and durability. Increased surface area increases metal deposition locations. Etching enhances chemical reactivity. Etching polymeric surfaces makes them hydrophilic, making surface treatments easier to apply. 22 A novel and facile polymerization-induced assembly-etching strategy was proposed for the manufacture of wrinkled zeolitic imidazolate framework-67@polypyrrole (ZIF-67@PPy) microcages using ammonium persulfate-catalyzed pyrrole polymerization and concomitant acid etching for superior electromagnetic wave absorption (EMA) applications. 23 A one-step synthesis was employed to fabricate polyaniline (PANI)-coated vanadium dioxide (VO2), in which VO2 is in situ etched by protonic acid released during aniline polymerization, forming an interconnected nanofiber network. The PANI coating enhances electrical conductivity and strengthens multipolar polarization relaxation, endowing the VO2@PANI composite with improved EMA performance. 24
Most polymeric surfaces are liquid-etched. Liquid etchants include organic solvents, oxidising acids, bases, and combinations of these. Liquid etchants roughen polymeric surfaces. Liquid etchant unsaturation and polar groups may affect a polymeric surface’s electrical state. A liquid etchant may generate a rough surface with many pits and holes by selectively dissolving a component in a heterogeneous mixture. Liquid etchants dissolve and change. Liquid etching removes surface low-molecular-weight fractions. 25 The surface modification of poly (lactic acid) (PLA) was achieved through an alkaline wet etching process to alter its properties for potential medical applications. The nano-roughness of treated 3D-printed PLA increased by 1.4 times compared to the control 3D-printed PLA. 26
In this work, porous PVA/SiO2/Ag nanocomposites were successfully fabricated via an etching technique. The etching process was carried out by introducing HF into the PVA solution in the presence of silicon oxide and silver nitrate. The prepared nanocomposites were subsequently subjected to γ-irradiation at different doses to optimize their structural and dielectric properties. The enhanced dielectrical performance observed for the nanocomposites irradiated at 15 kGy indicates that this composition is particularly promising for polymer electrolyte and energy storage applications.
Experimental Techniques
Materials
Polyvinyl alcohol (PVA) (M.wt.: 1.15.000), HF (M.wt. 20.01, 40%), and silicon oxide (M.wt. 20.01:60.08) were gotten. From Loba Chemie, India. Silver nitrate (≥99.0%, M.wt. 169.87) was acquired from Merck specialties private Limited, India. Reagents were used as received.
Preparation of HF/PVA/SiO2 Ag Nanocomposites
Nanocomposite films were prepared by solution casting. Briefly, HF (5.0 mL) was added to 83 mL of distilled water, followed by the gradual addition of PVA (10 g) under continuous stirring at 60°C until complete dissolution of the polymer. Subsequently, 1.0 g of SiO2 was introduced, followed by the addition of AgNO3 (1.0 g), and the mixture was stirred to ensure homogeneity. The resulting solution was transferred into plastic falcon tubes and irradiated with γ-radiation at absorbed doses of 15, 25, and 35 kGy, using a dose rate of 0.467 kGy h−1. After irradiation, the solutions were poured into plastic Petri dishes and dried in an oven at 50°C for 24 hrs to obtain nanocomposite films. The γ-irradiation was performed using a 60Co gamma-ray source at the National Centre for Radiation Research and Technology (NCRRT) of the Egyptian Atomic Energy Authority (EAEA). Scheme 1 represent the synthesis steps of HF/PVA/SiO2/Ag nanocomposites. Preparation of HF/PVA/SiO2/Ag nanocomposites.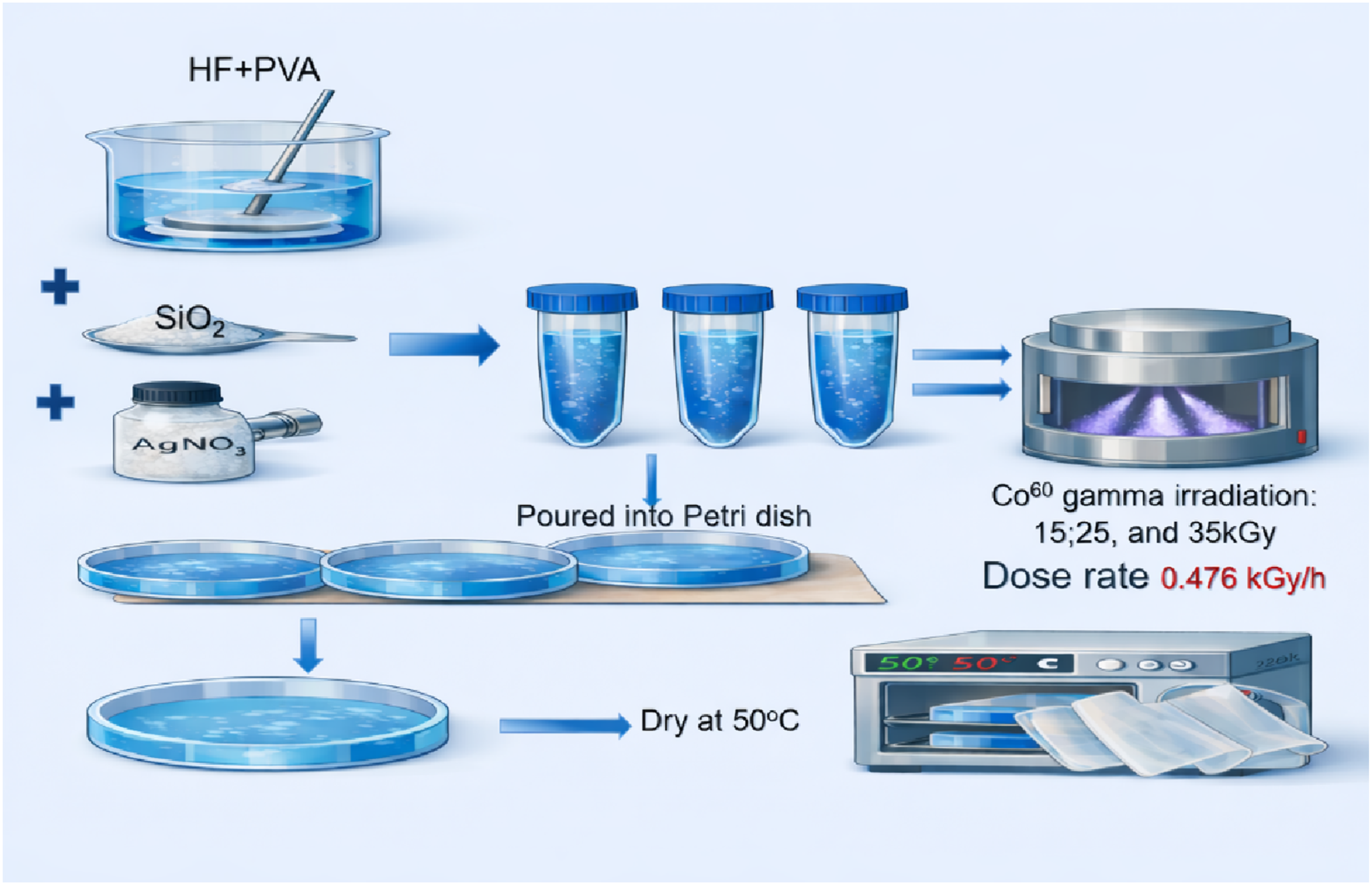
Characterization Techniques
The surface morphology of the polymer nanocomposite films was examined by scanning electron microscopy (SEM, ZEISS EVO 15, UK), and their elemental composition was analyzed using energy-dispersive X-ray spectroscopy (EDX). The chemical structure was investigated by Fourier transform infrared (FT-IR) spectroscopy in the range 400–3600 cm−1. Structural characterization was performed using X-ray diffraction (XRD) with Cu Kα radiation (λ = 1.5405 Å) on a Shimadzu diffractometer operated at 40 kV and 30 mA over a 2θ range of 4°–90°. Dielectric properties and AC conductivity were measured at room temperature using circular disk samples (1 cm diameter, ∼0.1 mm thickness) coated with silver paste on both sides to ensure good electrical contact. Measurements were conducted over a frequency range of 0.1 Hz–107 Hz using an impedance analyzer (IM-3570 RLC bridge, Japan). 27
Results and Discussion
FTIR
Infrared serves as a potent instrument for identifying functional groupings (such as vibrational bands like O-H, C-H, C = O) due to the analogous absorption frequencies of those groups across other molecules. Functional groups are structural entities in organic molecules characterized by specific atom and bond configurations. Figure 1 shows FTIR of pure PVA, unirradiated, and irradiated HF/PVA/SiO2/Ag nanocomposites. The broadband at approximately 3288 cm−1 is attributed to the stretching vibration of OH group in pure PVA, which is intramolecularly bonded. The band is observed to correspond to the asymmetric stretching vibration of CH2 at approximately 2925 cm−1.
28
The stretching mode of C=O has been assigned the peak at 1722 cm−1. The absorption peak at 1430 cm−1 has been attributed to symmetric bending of CH2. The bands at approximately 1083 cm−1 indicate the C-O elongation of carbonyl groups in the PVA backbone.
29
At 836 cm−1, the C-C stretching vibrations of the moderate absorption planar zigzag carbon backbone are observed. The swaying mode of OH is responsible for the peak at 602 cm−1. CH2 rocking is shown by the peak at 927 cm−1, whereas the peak represents (CH-OH) bending at 1322 cm−1. The band observed at 1247 cm−1 is linked to CH wagging.
30
FTIR spectra of pure PVA film (a), unirradiated HF/PVA/SiO2/Ag (b), irradiated with 15 kGy (c), 25 kGy (d), and 35 kGy (e).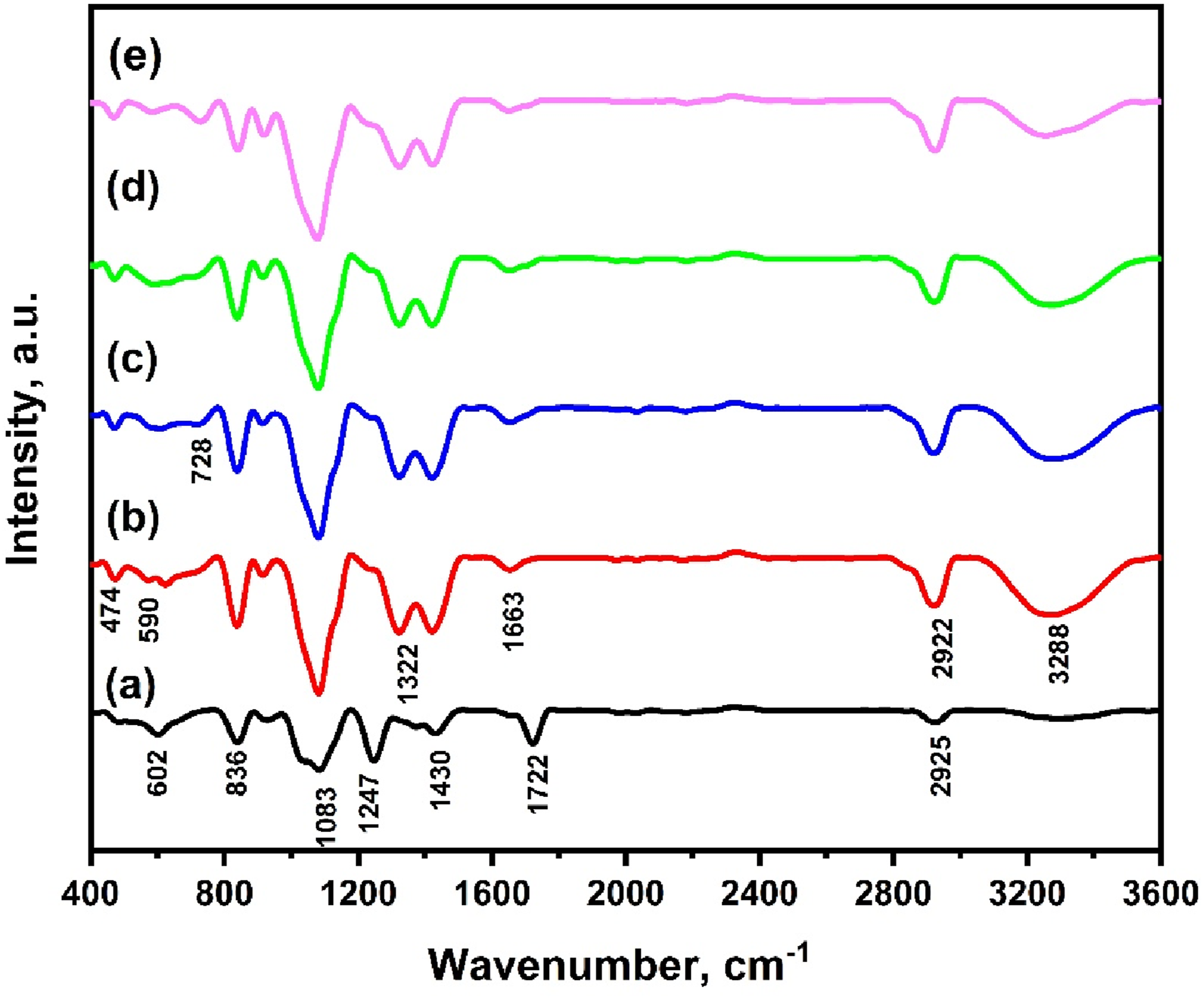
From Figure 1(b), it is noticed that many changes occurred in the spectra of HF/PVA/SiO2/Ag nanocomposites compared to pure PVA. For example, the intensities of all bands, such as OH, CH2, and C-O, have increased. Also, the intensities of the peaks at 1430 cm−1 and 1322 cm−1 increased slightly. On the other hand, the intensity of the peak found at 602 cm−1 decreased and was displaced to a higher wave number at 624 cm−1. Moreover, the peak centered at 1246 cm−1 has disappeared. In addition, new bands at 474 and 590 cm−1 were observed, which were allocated to Si-O and C-F (halo compound) stretching. 31
Moreover, the band at 1722 cm−1 (C=O) has disappeared, and a new, weak band at 1663 cm−1 indicates the formation of a C=C stretching vibration. All these changes indicate the interaction between PVA and the other components (HF, SiO2, and silver). 32 After irradiation, a new band at 728.6 cm−1 was observed, attributed to C-F stretching and possibly combined with C=C bending. Also, by increasing the irradiation dose from 25 to 35 kGy, most of the band’s intensity found at 840 cm−1 and 3288 cm−1 decreased, which may indicate an increase in amorphisation of the material and the role of radiation in the etching process.31,33
XRD
The XRD patterns of unirradiated and γ-irradiated HF/PVA/SiO2/Ag nanocomposites are presented in Figure 2. All samples exhibit a dominant broad diffraction peak centred at 2θ ≈ 19.49°, which is indexed to the (110) crystallographic plane of PVA, confirming the semi-crystalline nature of the nanocomposite system.
34
Upon γ-irradiation at absorbed doses of 15 and 35 kGy, a noticeable broadening of the main diffraction peak is observed, as evidenced by an increase in the full width at half maximum (FWHM). This behavior indicates a reduction in crystallite size and an increase in the amorphous phase content.
35
The enhancement of amorphous regions can be attributed to radiation-induced free radical formation, hydrogen abstraction, bond cleavage, and the predominance of chain scission over crosslinking at these doses.1,36 In contrast, irradiation at 25 kGy leads to a relative sharpening of the diffraction peak, indicating an increase in the crystalline phase. This behavior suggests that crosslinking becomes dominant over chain scission at this intermediate dose, thereby improving molecular ordering within the polymer matrix. The observed irradiation-induced structural evolution is expected to significantly influence the electrical behavior of the nanocomposites, with enhanced charge-transport pathways anticipated at optimal irradiation conditions, as discussed in the electrical measurements section.
37
XRD for unirradiated HF/PVA/SiO2/Ag and irradiated with 15 kGy, 25 kGy, and 35 kGy of γ-ray.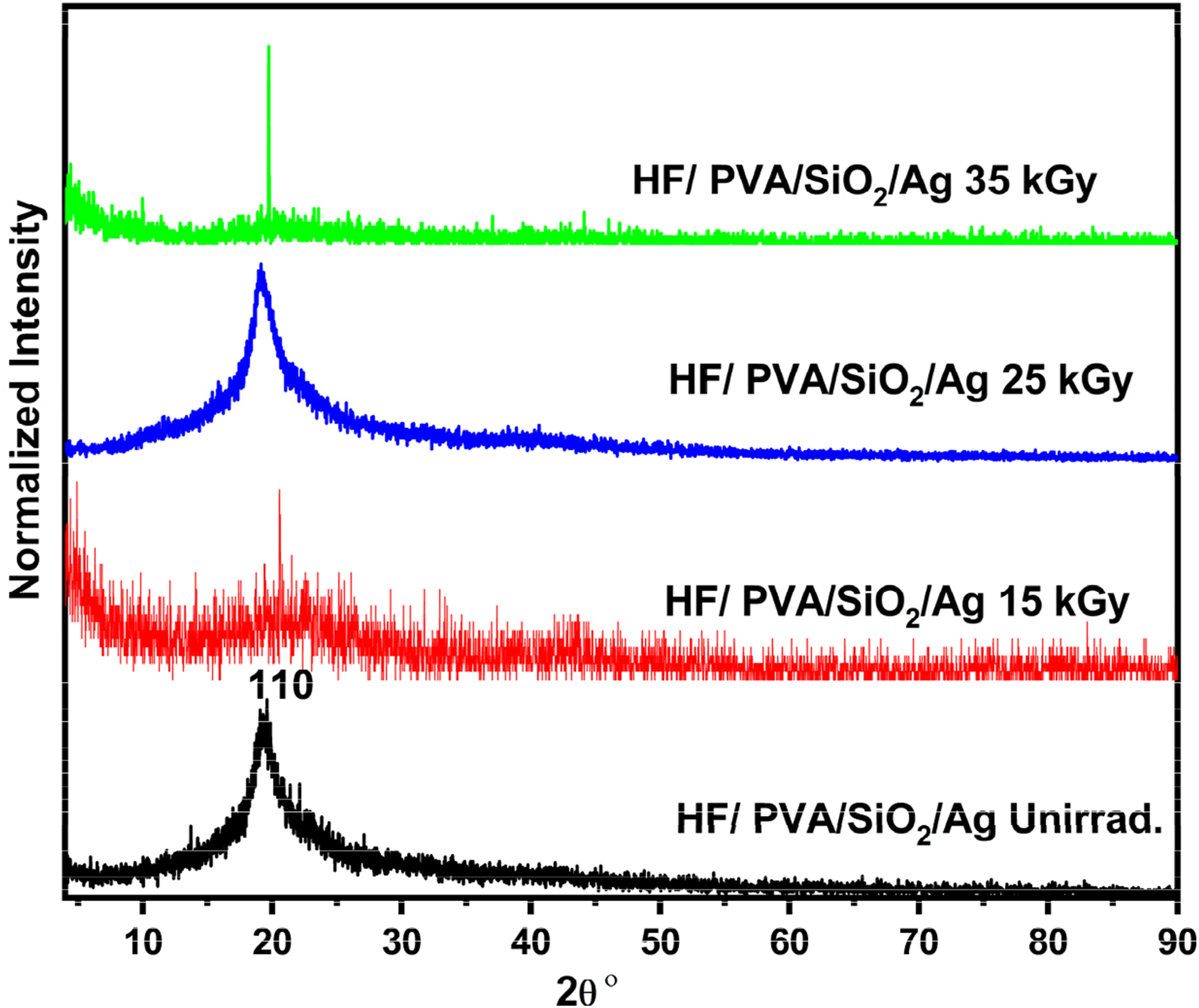
Surface Morphology
Scanning electron microscopy (SEM) is used to describe prepared nanocomposites’ structural morphology and porosity. Figure 3(a)–(d) displays the cross-sectional SEM images of HF/PVA/SiO2/Ag nanocomposite films in their unirradiated condition and subsequent to γ-irradiation at various absorbed doses. Cross-sectional SEM image for unirradiated HF/PV/ASiO2/Ag nanocomposites (a), irradiated with 15 kGy (b), 25 kGy (c), and 35 kGy (d) of gamma rays.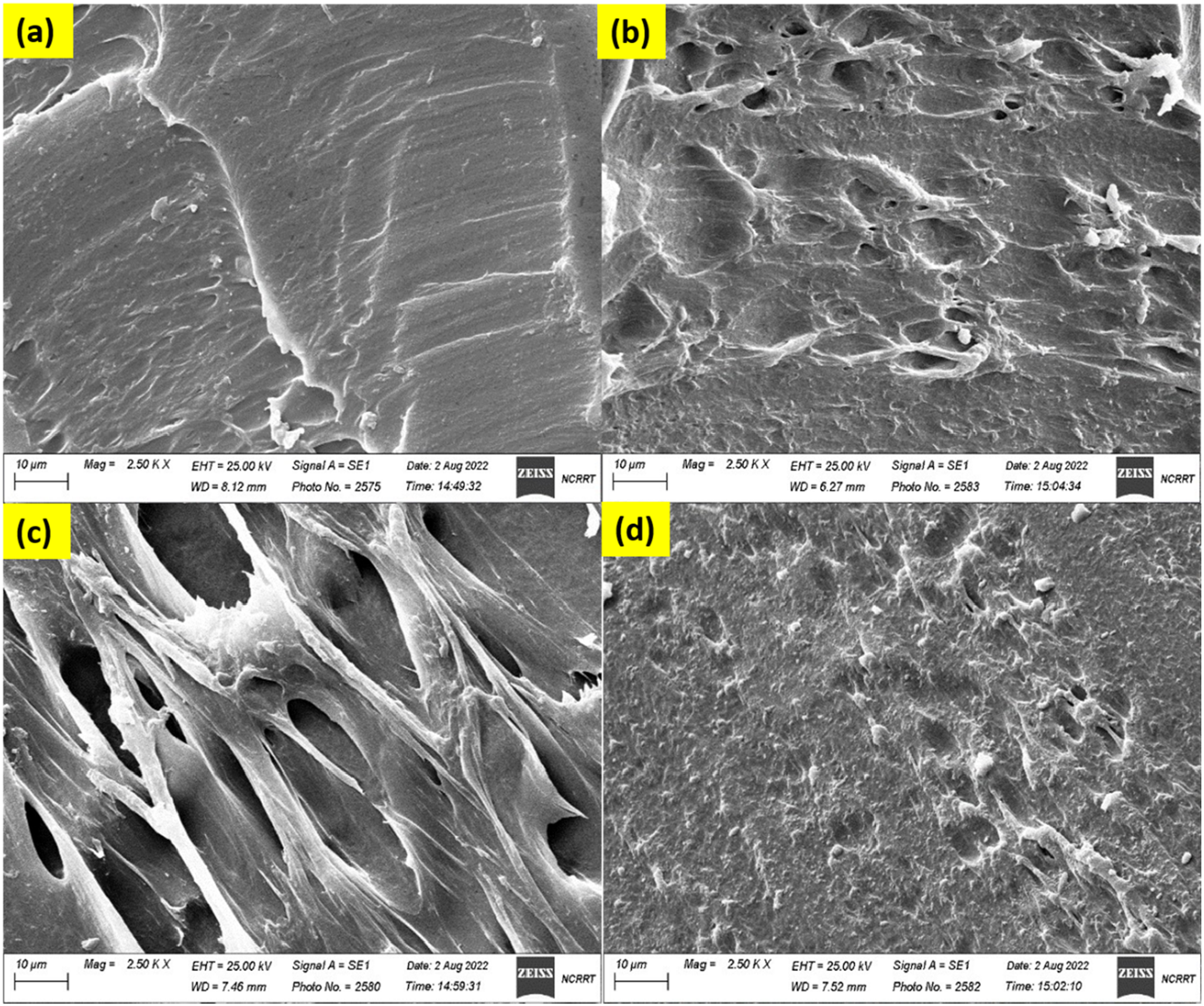
For the unirradiated sample, the polymer matrix exhibits a continuous, stiff, compact, and homogeneous structure with minimal porosity, indicating effective film formation via solution casting. The inorganic fillers are uniformly integrated within the PVA matrix without significant agglomeration, indicating effective filler dispersion. Following γ-irradiation at 15 kGy (Figure 3(b)), the surface of HF/PVA/SiO2/Ag exhibits a marked increase in roughness, along with the emergence of microscopic pores and microvoids dispersed throughout the film. This morphological change is attributed to radiation-induced free radical formation and partial disruption of polymer chains, leading to localized matrix reorganization. 1 At an absorbed dose of 25 kGy (Figure 3(c)), the HF/PVA/SiO2/Ag nanocomposite exhibits a highly porous and interconnected structure characterized by elongated voids and fibrillary elements. The increased porosity indicates significant restructuring of the polymer network induced by radiation. Increasing the irradiation dose to 35 kGy (Figure 3(d)) results in partial collapse of the porous structure and the formation of larger and irregular cavities. This behavior results from excessive radiation exposure, which induces predominant chain scission over crosslinking, thereby diminishing the film’s structural integrity and leading to a less uniform morphology.38,39
Compositional analysis of synthesized nanocomposites before and after irradiation was performed using an energy-dispersive X-ray spectrometer (EDS).
The EDS analysis of HF/PVA/SiO2/Ag nanocomposite films, presented in Figure 4, was performed for the unirradiated sample and those subjected to γ-irradiation at different absorbed doses. The EDS spectra of the unirradiated nanocomposite confirm the successful incorporation of all constituent elements. Pronounced peaks corresponding to carbon (C, 58.9 at.%) and oxygen (O, 39.5 at.%), originating from the PVA matrix, are observed, along with silicon (Si, 0.86 at.%) from SiO2 and silver (Ag, 0.74 at.%). Minor variations in elemental composition are detected following γ-irradiation, which may influence the distribution and detectability of Ag and Si. These changes are attributed to radiation-induced crosslinking and structural rearrangements within the PVA matrix, consistent with previous reports.
40
EDX for unirradiated HF/PV/ASiO2/Ag nanocomposites (a), irradiated with 15 kGy (b), 25 kGy (c), and 35 kGy (d) of γ-rays.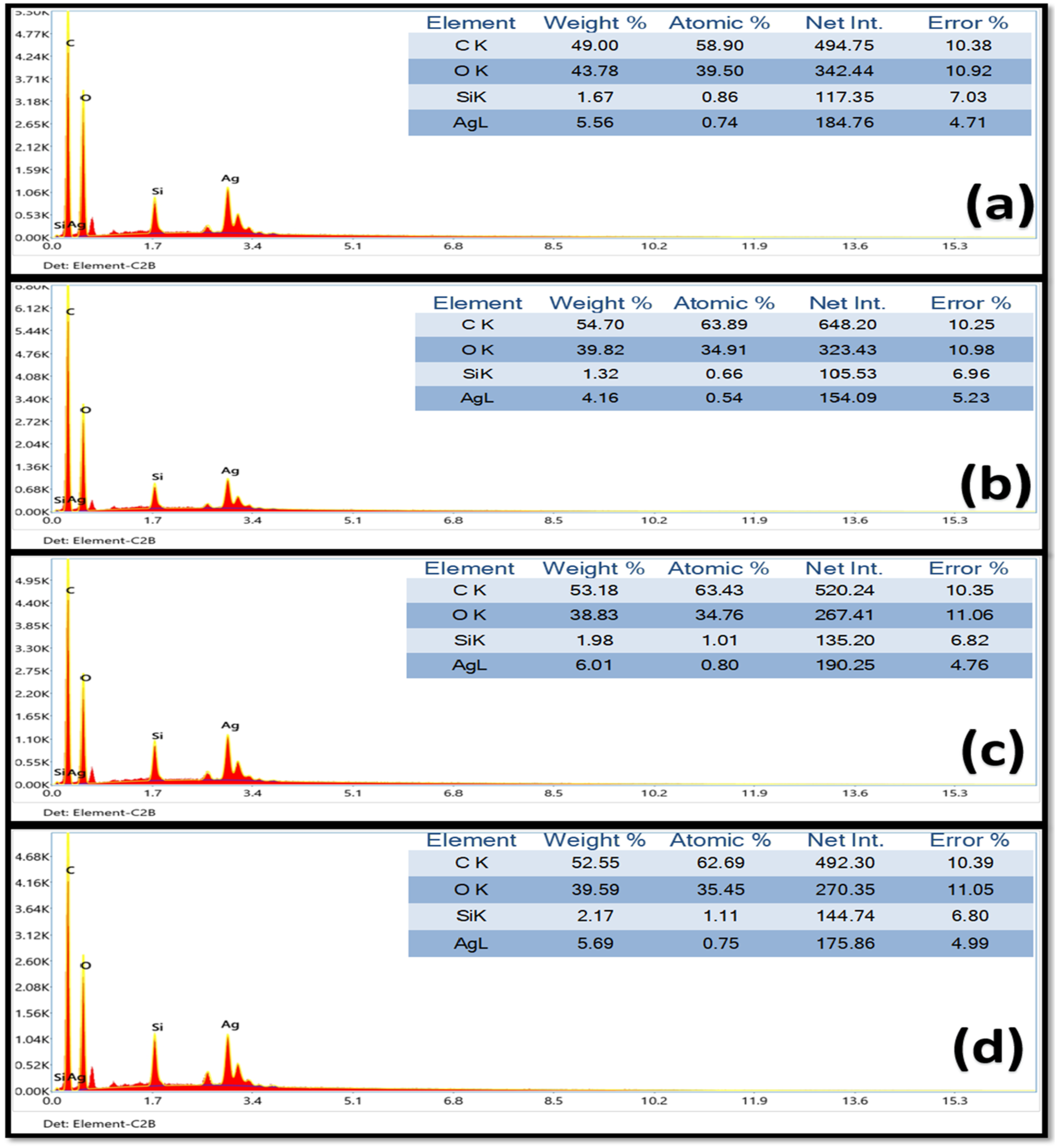
Dielectric Analysis
The complex dielectric permittivity (ε*) characterizes a material’s ability to store electrical energy when subjected to an external electric field. In polymeric materials, dielectric polarization arises from several mechanisms, including dipolar orientation, ionic migration within the polymer matrix, and charge injection from the electrodes. The complex dielectric permittivity of the prepared blends was evaluated using equation (1), which is a key parameter for understanding their electrical behavior
41
:
Figure 5 illustrates the frequency dependence of the dielectric constant (ε′) and dielectric loss (ε″) for pure PVA as well as unirradiated and γ-irradiated HF/PVA/SiO2/Ag nanocomposites at room temperature. At low frequencies, the applied electric field varies slowly, allowing different polarization mechanisms—namely electronic, ionic, dipolar (orientational), and interfacial (Maxwell–Wagner–Sillars, MWS). As a result, high ε′ values are observed in this frequency regime. With increasing frequency, dipoles and mobile charge carriers can no longer follow the rapidly alternating field, leading to the gradual suppression of polarization processes and, consequently, a decrease in ε′.
42
Dielectric constant (a) and Dielectric loss (b) dependency on frequency for pure PVA film (a), unirradiated and irradiated HF/PVA/SiO2/Ag nanocomposites.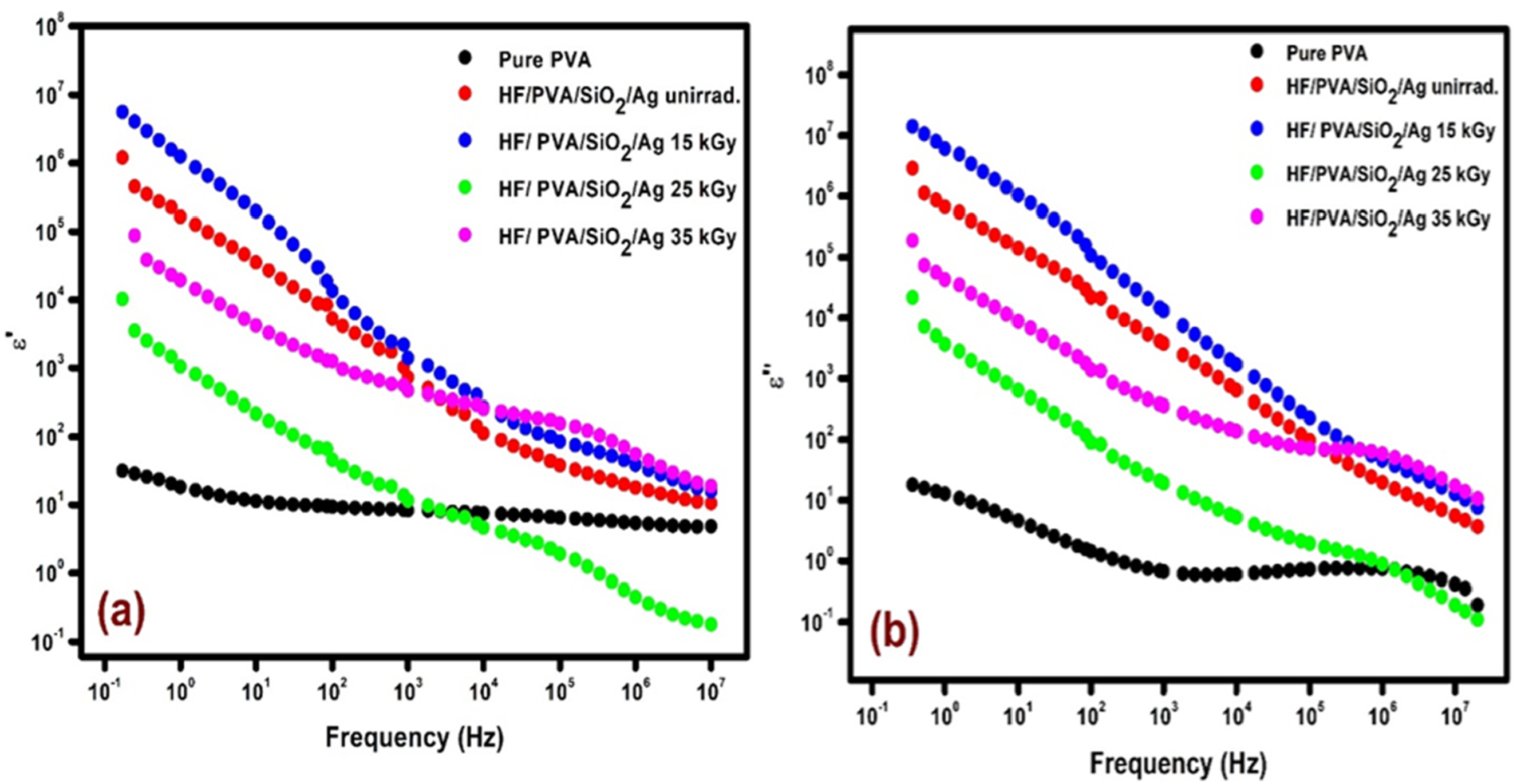
Pure PVA exhibits relatively low ε′ values across the entire frequency range due to its limited number of free charge carriers and the dominance of localized dipolar polarization associated with –OH groups along the polymer backbone.
In contrast, the ε′ value of the HF/PVA/SiO2/Ag nanocomposites reaches approximately 4.65 × 104 at 100 Hz, which is nearly five thousand times higher than that of pure PVA (ε′ ≈ 9.73) at the same frequency. This pronounced enhancement is attributed to strong interfacial (Maxwell–Wagner–Sillars) polarization arising from heterogeneous interfaces between conductive Ag species, insulating SiO2 domains, and the polymer matrix. 43
Gamma irradiation significantly influences the dielectric response of the nanocomposites. At an absorbed dose of 15 kGy, both ε′ and ε″ increase relative to the unirradiated sample. This behavior is attributed to radiation-induced chain scission and bond rearrangements within the polymer matrix, 44 thereby increasing the amorphous content and enhancing dipolar flexibility. The higher amorphous phase facilitates dipolar orientation and ion–dipole interactions, thereby improving charge storage capability.
Consequently, irradiation at 15 kGy enhances the dielectric energy storage performance of the HF/PVA/SiO2/Ag nanocomposites.
In contrast, increasing the irradiation dose to 25 kGy results in a reduction in ε′, associated with decreased amorphicity and the onset of dominant crosslinking effects. Crosslinking restricts the mobility of dipolar groups along the polymer chains, limiting their response to the alternating electric field and reducing polarization efficiency.
This dielectric behavior can be rationalized based on the combined effects of HF etching, Ag+ coordination, and γ-irradiation. In the presence of AgNO3, Ag+ ions are homogeneously coordinated by the hydroxyl groups of PVA and partially interact with silica surfaces. HF selectively attacks amorphous SiO2 through protonation of siloxane bonds followed by fluoride-induced dissolution, forming soluble hexafluorosilicate species:
Transient Ag+–F- interactions locally enhance fluoride availability near silica domains, promoting controlled and localized etching. Partial removal of SiO2 generates nanoscale voids, disrupts PVA–SiO2 hydrogen bonding, and increases the polymer free volume. As a result, stable nanoporous structures are formed, while Ag+ species become confined near pore walls, contributing to the dielectric response.
Upon γ-irradiation, these confined Ag+ species are readily reduced to metallic Ag nanoparticles by radiation-induced electrons and polymer radicals. This spatial confinement promotes uniform distribution of Ag nanoparticles along the pore walls, enhancing interfacial polarization and significantly contributing to the observed dielectric enhancement. Overall, the synergistic effects of HF-induced silica dissolution, Ag+-mediated etching modulation, and γ-irradiation-induced structural rearrangements produce a porous nanocomposite with tunable dielectric properties. At 15 kGy, chain scission dominates, increasing amorphous content and charge carrier mobility, whereas at 25 kGy, enhanced crosslinking restricts dipolar motion and reduces dielectric performance. 45
A partial recovery of ε′ at 35 kGy can be attributed to competing effects between further structural rearrangements and localized degradation, although excessive irradiation generally limits long-range dipolar motion.
From Figure 5, the dielectric loss (ε″) decreases with increasing frequency for all specimens. The higher ε″ observed at lower frequencies can be attributed to mobile charges within the polymer matrix. At higher frequencies, the rapid oscillation of the applied electric field limits ion migration along the field direction, thereby reducing polarization contributions. 42
The elevated ε″ values of the nanocomposite irradiated at 15 kGy are further ascribed to the enhanced chain mobility of the polymer matrix. This increased mobility arises from a higher amorphous phase content, as confirmed by XRD analysis. 36
An important characteristic of polymeric nanocomposites is their AC conductivity (σac), which underpins their applicability in electrical and electronic devices. The σac of the nanocomposite was calculated using equation (4)
46
Figure 6 presents logarithmic plots of the σac dependency on frequency for pure PVA, unirradiated, and irradiated nanocomposites. It has been shown that all samples’ σac increase with frequency. This effect arises because an external electric field causes charge carriers to move more freely. In addition, releasing trapped charges is responsible for the change in alternating current conductivity.
47
The σac dependency on frequency contains three different regions: low-frequency, intermediate, and high-frequency dispersion regions. AC conductivity dependency on frequency for pure PVA film (a), unirradiated and irradiated HF/PVA/SiO2/Ag nanocomposites.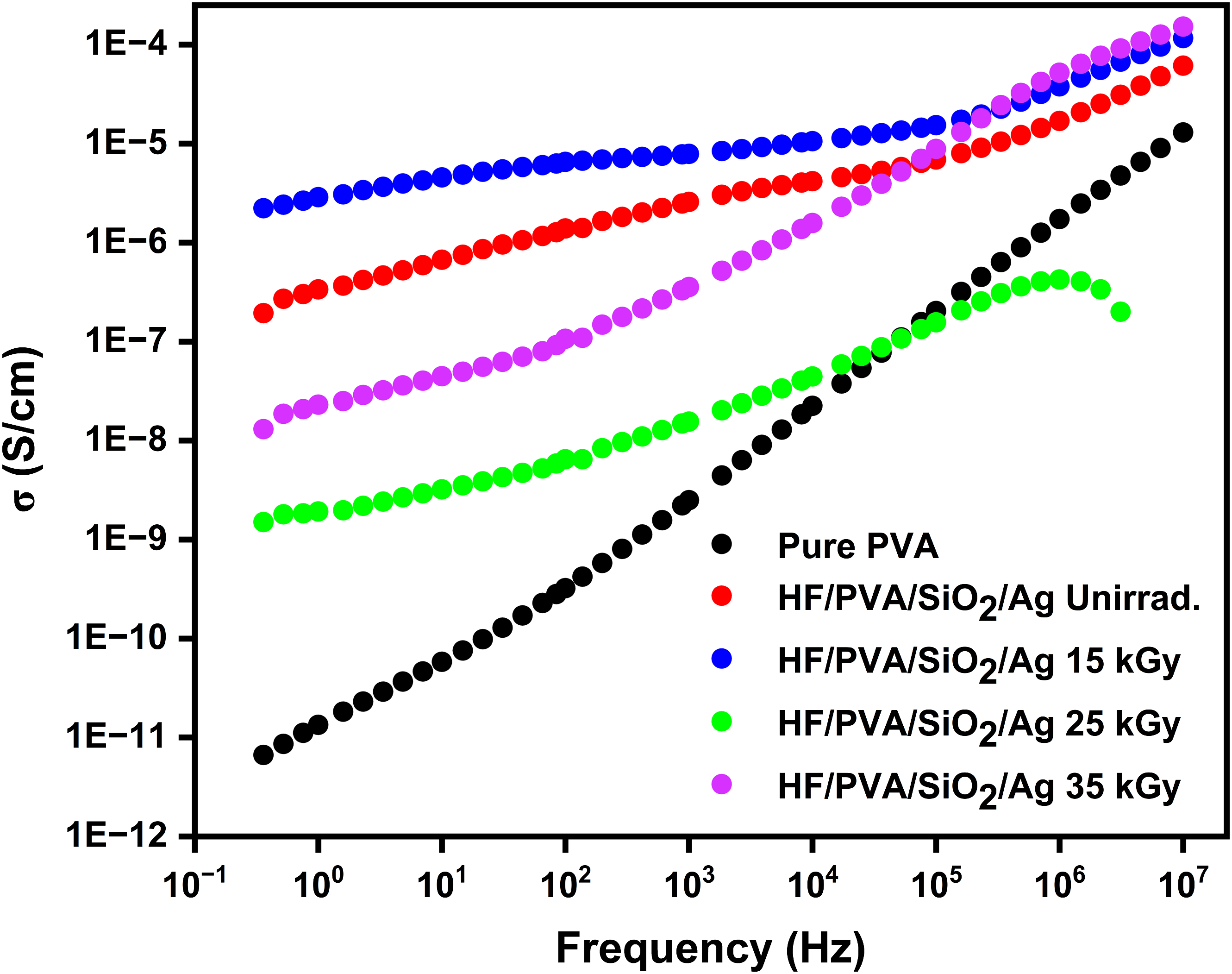
The high-frequency range corresponds to the collective relaxation of mobile ions due to Coulombic interactions between charge carriers. The intermediate region is attributed to ion mobility via a hopping mechanism, driven by long-distance diffusion. The low-frequency domain is ascribed to polarization of space charge at the interface of electrolyte-electrode, which is attributable to the electrode blocking. 36
Jonscher’s power law governs the behavior of AC conductivity, as follows:
Fitting parameters from Jonscher’s power law σdc, A and n for different samples.
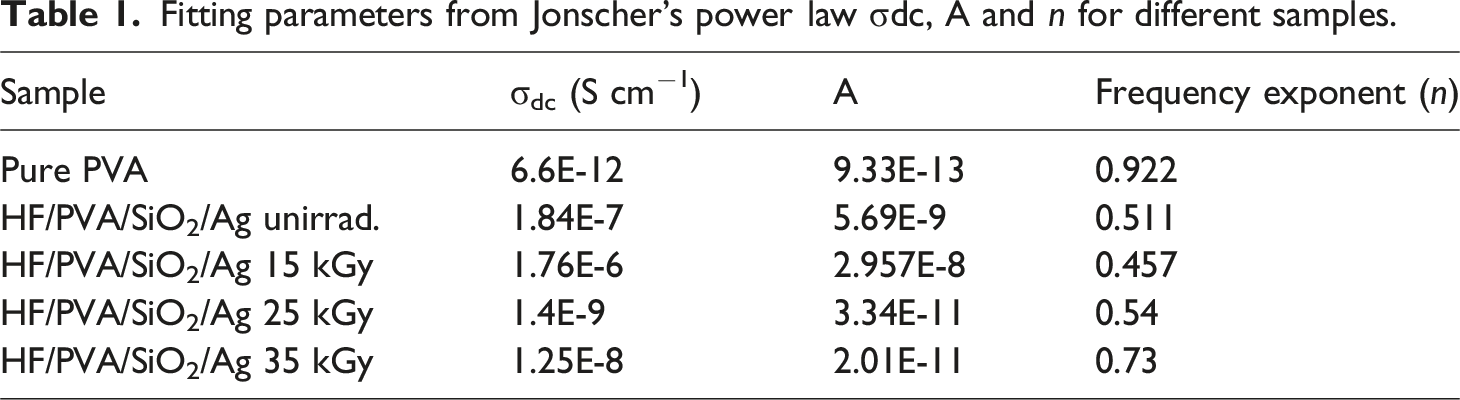
The dielectric modulus provides a more comprehensive understanding of charge transport processes, including relaxation mechanisms and ion dynamics, as they vary with frequency. The electric modulus (M∗) may be computed by taking the reciprocal of the complex relative permittivity (ε∗), as expressed in equation (6)
50
:


Figure 7 depicts the relationship between frequency and M′ and M″ for pure PVA and unirradiated and irradiated HF/PVA/SiO2/Ag nanocomposites at room temperature. Real part of dielectrical constant (a) and imaginary part dielectrical constant (b) dependency on frequency for pure PVA film (a), unirradiated and irradiated HF/PVA/SiO2/Ag nanocomposites.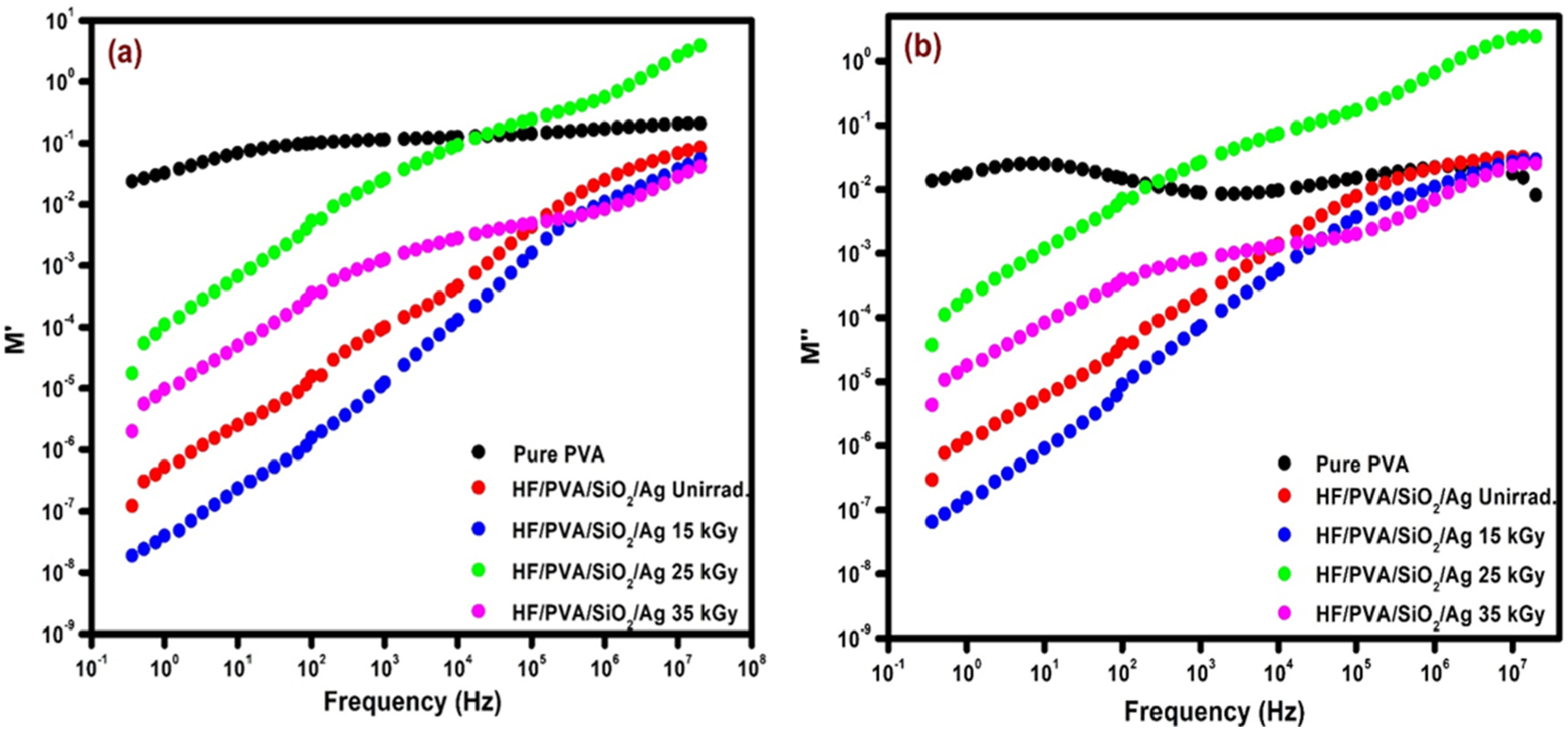
It is evident that both M′ and M″ increase as the frequency rises, which indicates a contrasting behavior when compared to the patterns of ε′ and ε″.
Figure 7 also shows that M′ and M″ follow the typical pattern observed in various disordered materials. The observation of a lower M′ value for pure PVA at lower frequencies than ε′ indicates inhibition of electrode polarisation. It has been observed that Mʹ increases in frequency and attains a maximal saturation value at high frequencies, resulting from a short range of charge-carrier mobility. 52 Due to the contemporaneous rise in the real component of permittivity, the observed pattern shows a reduction in Mʹ for HF/PVA/SiO2/Ag nanocomposites. 53 The system irradiated with 15 kGy exhibits a lower M″ value than the unirradiated ones. Both have lower values than pure PVA, potentially attributable to increased charge-carrier mobility. 54 The M″ values exhibit a notable decrease at lower frequencies across all samples, suggesting the effective elimination of electrode polarisation. The observed increase in M″ with increasing frequency can be attributed to the distribution of relaxation across a range of frequencies. 54 Compared to other samples, the nanocomposite irradiated with 25 kGy has the highest M″, which is consistent with its lowest conductivity, as described earlier. The lack of a relaxation peak indicates an experimental frequency constraint, perhaps due to the increased ion density and the enhanced mobility of charge carriers in both irradiated and unirradiated systems. 54
Conclusion
Porous HF/PVA/SiO2/Ag nanocomposites were effectively synthesized using the drop-casting method. Gamma irradiation induced significant, dose-dependent structural alterations in the nanocomposites. Irradiation at 15 and 35 kGy led to an elevated amorphous phase, attributed to the dominance of chain-scission mechanisms over crosslinking at these dosages. Irradiation at 25 kGy decreased amorphosity, indicating the prevalence of radiation-induced crosslinking and improved molecular organization. The dielectric response of the nanocomposites was strongly governed by the applied irradiation dose. The sample irradiated to 15 kGy exhibited the greatest enhancement in dielectric performance, with the highest σac, dielectric constant, and dielectric loss among all investigated compositions. Also, this sample achieved the maximum σdc of 1.76 × 10−6 S·cm−1, reflecting improved charge transport. This improvement is mainly linked to radiation-induced chain scission, which boosts charge-carrier mobility. In contrast, the prevalence of crosslinking at 25 kGy constrained dipolar motion, leading to diminished dielectric characteristics; hence, this sample displayed the greatest electric modulus values owing to its lowest conductivity. These findings indicate that γ-irradiation is a potent and manageable method for modifying the structural and dielectric characteristics of HF-etched polymer nanocomposites. The nanocomposite irradiated at 15 kGy is determined to be the best composition for energy-storage applications based on the integrated structural, dielectric, and modulus evaluations.