Abstract
Magnetic carbon dots (Fe3O4/N–CQDs) were synthesized via a one-step microwave-assisted method using sugarcane bagasse as the carbon precursor. The synthesized materials were thoroughly characterized via thermogravimetric analysis (TGA/DTG), Fourier transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), and transmission electron microscopy (TEM) to examine their thermal behavior, nanoscale morphology, and phase composition. These Fe3O4/N–CQDs were incorporated into acrylonitrile butadiene rubber (NBR) at varying concentrations (0–11 phr), and the resulting composites were evaluated for their structural characteristics (FTIR, SEM), thermal stability, curing behavior, mechanical performance, swelling resistance, thermal aging resistance, as well as magnetic, and dielectric properties. Among the formulations studied, the composite containing 8 phr Fe3O4/N–CQDs showed the most balanced performance, with tensile strength increasing from 2.3 to 4.2 MPa and hardness from 54 to 65 Shore A. Post-aging analysis revealed improved tensile strength and reduced swelling, attributed to enhance crosslinking. The composites demonstrated superior thermal aging resistance under accelerated aging at 110°C for 10 days. Magnetic measurements indicated soft magnetic behavior, with saturated magnetization increasing from 29.05x 10−3 emu/g (0 phr) to 91.32 × 10−3 emu/g (11 phr) and coercivity below 120 G, while dielectric and conductivity studies showed frequency-dependent permittivity and conductivity consistent with interfacial polarization and hopping mechanisms. These results demonstrate that Fe3O4/N–CQDs can simultaneously enhance the thermal, mechanical, antiaging, magnetic, and dielectric properties of NBR compounds.
Keywords
Highlights
• Magnetic nitrogen-doped carbon quantum dots (Fe3O4/N–CQDs) were successfully synthesized from sugarcane bagasse via a green, sustainable route. • Fe3O4/N–CQDs were incorporated into NBR rubber at different loadings (0–11 phr) to evaluate multifunctional performance. • TEM and SEM revealed nanoscale dispersion and morphology, and FTIR confirmed chemical interactions within the NBR matrix. • Fe3O4/N–CQDs significantly enhanced thermal aging resistance under 110°C for 10 days. • Enhanced dielectric and magnetic properties indicate strong potential for antistatic, electrostatic discharge (ESD)-safe, and electromagnetic interference (EMI) shielding applications.
Introduction
The global accumulation of agricultural waste poses significant environmental and economic challenges, as crop processing generates vast quantities of byproducts that are often discarded or burned.1,2 Sugarcane bagasse (SCB), the fibrous residue remaining after sugarcane stalks are crushed, is produced in millions of tons annually and remains largely unexploited. 3 Its high carbon content, low ash content, and abundance make it an attractive feedstock for carbon-based nanomaterials, including carbon dots (CDs).3,4 CDs are quasi-spherical, zero-dimensional nanoparticles (<10 nm) with a high surface-to-volume ratio and rich surface chemistry, including hydroxyl, carboxyl, carbonyl, and amine groups.5–8 These features enable strong interactions with host matrices and facilitate doping or hybridization with inorganic nanoparticles. 9
While CDs have been widely explored for optical applications such as sensing and imaging, recent attention has turned to their use as polymer nanofillers.10,11 Their nanoscale dimensions and reactive surfaces allow uniform dispersion within hydrophilic polymer matrices, enhancing filler–matrix adhesion. This interfacial reinforcement in compatible matrices often translates into improvements in tensile strength, elasticity, thermal stability, and chemical resistance. 10 Recent studies have demonstrated the potential of CDs as reinforcing agents in rubbers, including natural rubber, silicone rubber, and chloroprene rubber, where they promote crosslinking and impart multifunctional properties beyond conventional reinforcement.12–15
In rubber chemistry, nanoscale fillers such as nano clay, nanosilica and carbon nanotubes are commonly used to enhance mechanical and thermal properties.16,17 However, the growing interest in multifunctional rubbers has led researchers to explore alternative nanofillers, including CDs and metal or metal oxide/sulfide nanoparticles, which can impart additional functionalities.10,16,18 While CDs primarily enhance mechanical strength, elasticity, thermal stability, and interfacial adhesion; and can also contribute to dielectric behavior 19 ; metal and metal oxide nanoparticles provide complementary properties such as magnetic, dielectric, thermal, and electrical responsiveness. 16 Metal nanoparticles with specific surface functionalities are particularly attractive because they are inexpensive, easy to prepare, and can achieve effects similar to more costly nanofillers like carbon nanotubes or graphene. 16 By combining CDs with metal-based nanofillers, advanced rubber composites can be designed to simultaneously offer mechanical reinforcement and multifunctional performance.
Among metal oxides, magnetite (Fe3O4) nanoparticles are particularly attractive due to their strong magnetic response, high thermal stability, and ability to enhance dielectric and electrical properties. 20 Fe3O4 has been successfully incorporated into rubbers, including nitrile butadiene rubber (NBR), epoxidized natural rubber, and thermoplastic natural rubber, to simultaneously improve mechanical performance, thermal stability, magnetic behavior, and electrical conductivity.21–24 Despite these advantages, the combination of CDs and Fe3O4 as nitrogen-doped magnetic carbon dots (Fe3O4/N–CQDs) has not been explored in rubber composites. To expand the functional scope of CDs, recent research has investigated their doping and hybridization with inorganic nanoparticles. 9 Nitrogen doping introduces additional reactive sites on the CDs, improving their bonding with polymer chains, while attaching Fe3O4 nanoparticles to the CDs provides additional functionalities, including magnetic response, dielectric behavior, and electromagnetic wave absorption. Combining these two components into a single hybrid nanofiller offers a rational strategy to integrate nanoscale reinforcement with multifunctional properties.
Acrylonitrile–butadiene rubber (NBR) provides an ideal platform to evaluate such hybrid fillers. As a polar elastomer containing nitrile (–CN) groups, NBR exhibits excellent resistance to oils, fuels, and chemicals and shows good compatibility with polar and functional nanofillers. 25 Conventional NBR vulcanizates are widely employed in antistatic components, seals, and industrial rubber products 26 ; however, their performance is often constrained by susceptibility to thermo-oxidative aging, gradual mechanical deterioration during long-term service,26,27 and limited magnetic or dielectric response. Enhancing these properties is therefore essential to extend the functionality and reliability of NBR-based materials for next-generation multifunctional rubber components.
In this context, Fe3O4/N–CQDs are expected to address these limitations by combining the reinforcing and interfacial advantages of nitrogen-doped CDs with the magnetic and dielectric functionalities of Fe3O4. In the present study, Fe3O4/N–CQDs were synthesized via a one-step microwave-assisted method using sugarcane bagasse as a sustainable carbon precursor, with FeCl3.6H2O and FeCl2.4H2O as iron sources. The hybrid nanofillers were incorporated into vulcanized NBR, and their effects on curing characteristics, mechanical properties, swelling behavior, thermal aging resistance, and magnetic and dielectric responses were systematically investigated. By integrating biomass-derived nanomaterials into NBR, this work provides a pathway toward multifunctional polymer composites with enhanced performance and reduced environmental impact.
Substances and experimental techniques
Substances
• The sugarcane bagasse (SCB) was obtained from the Paper Industry Quena Company, Egypt. • Nitrile rubber (NBR) was bought from Bayer AG Company (Germany) with CN content of 32% and specifc gravity 1.17 ± 0.005 g/cm3. • N-cyclohexyl-2-benzothiazole sulphenamide (CBS), a light gray powder with a melting point of 95–100°C and a specific gravity of 1.27–1.31 at room temperature (25°C ± 1) provided from Sigma Aldrich. • stearic acid and Zinc oxide as activators, with specific gravities of 5.55–5.61 and 0.90-0.97 at 15°C, respectively provided from Sigma Aldrich. • The elemental sulfur as vulcanizing agent that was applied as a fine, pale yellow powder with a specific gravity of 2.04–2.06 at ambient temperature purchased from Sigma Aldrich.
Experimental techniques
Preparation of nitrogen doped carbon quantum dots
A homogeneous solution was initially formed by stirring a mixture of 30 mg SCB, 70 mg NaOH, and 2400 mg urea in 100 mL water for 30 min. The solution was then subjected to a freeze-thaw cycle (frozen overnight, defrosted the next day). Subsequently, the defrosted solution was treated with ultrasound for 2 min to ensure adequate de-clumping. For the final modification step, the solution was heated in a 700 W microwave for about 7 min. 28
Preparation of magnetite/nitrogen doped carbon quantum dots (magnetite N–CQDs)
Preparation of the magnetite N–CQDs composite was achieved through a microwave-assisted process, utilizing FeCl3⋅6H2O and FeCl2⋅4H2O as the iron sources. An aqueous solution of N–CQDs (1 g in 50 mL H2O) was subjected to 5 min of sonication. Concurrently, an iron precursor solution (50 mL) containing 9.07 g Fe3+and 5.05 g Fe2+was prepared and introduced into the N–CQDs dispersion. 1 M NaOH was used to elevate the pH to 11−12. The mixture was then heated using a 700 W microwave for 10 min. Post-synthesis, the magnetite N–CQDs product was isolated magnetically. Purification involved washing the material with deionized water and absolute ethanol. The final product was obtained after drying in an oven at 60°C. 28
Preparation of NBR compounds
Chemical substances used in NBR compounds.
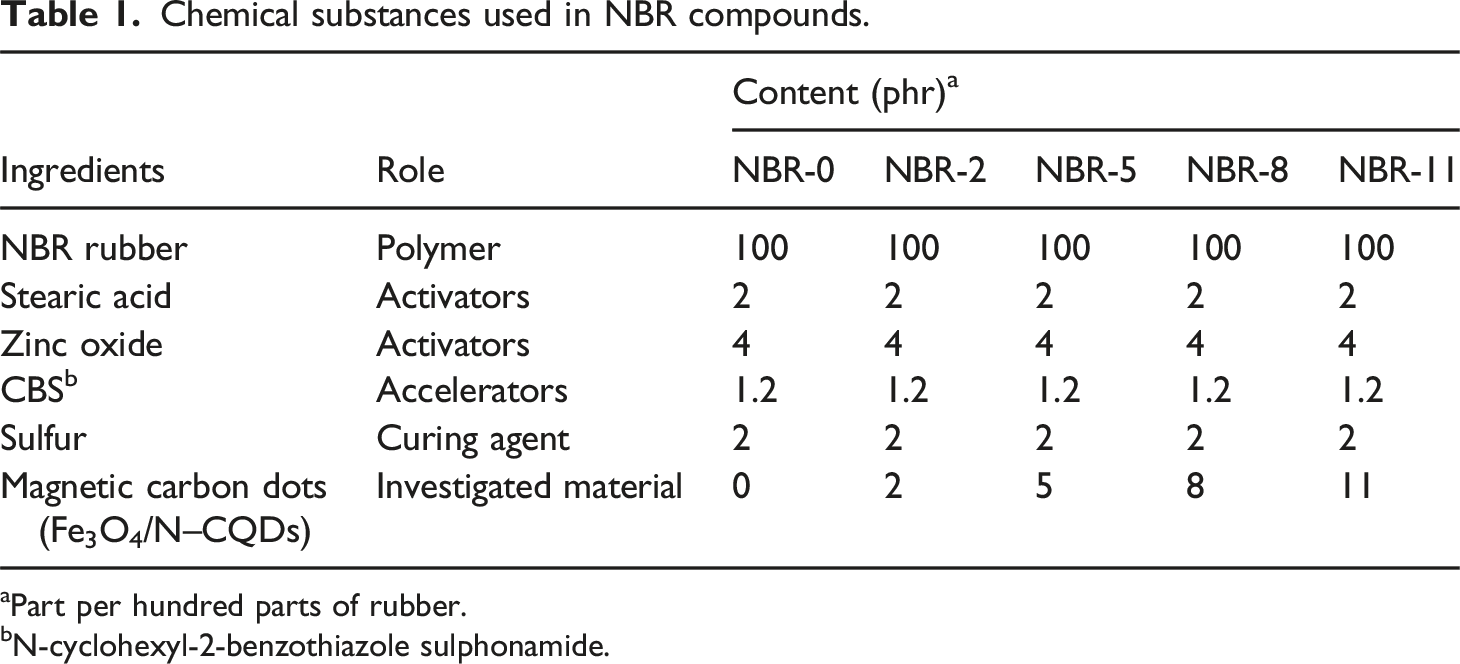
aPart per hundred parts of rubber.
bN-cyclohexyl-2-benzothiazole sulphonamide.
Measurements and description
Fourier transforms infrared spectroscopy (FT-IR) analysis
FTIR spectra of the magnetic carbon dots were recorded using a Mattson 5000 FTIR spectrometer (Unicam, UK) employing the KBr pellet method in the range of 4000–400 cm−1. ATR-FTIR measurements of the NBR samples were carried out using a VERTEX 80 FTIR spectrometer (Bruker Corporation, Germany) equipped with a platinum diamond crystal as the internal reflection element, over the same spectral range (4000–400 cm−1).
Diffraction of X-ray powder (XRD)
X-ray diffraction (XRD) patterns were recorded using a Bruker D-8 Advance X-ray diffractometer (Germany) operated at 40 kV and 40 mA. Diffraction data were collected over a 2θ range of 5°–70° with a step size of 0.05°. Copper Kα radiation (λ = 1.5406 Å) was used to investigate the crystallinity of the samples.
Transmission electron microscope (TEM)
The JEM-2100F (JEOL, Japan) was utilized to capture TEM images at an accelerating voltage of 200 kV. The ethanol solution was put onto a carbon-coated copper grid and let to evaporate after several drops of the nanoparticle solution were diluted with one mL of ethanol.
Thermogravimetric data analysis
The instrument utilizedthe thermal gravimetric/differential thermogravimetric (TGA/DTG)was a TGA Q500 (TA Instruments, USA). It was evaluated by thermogravimetric analysis (TGA, USA). Using 0.1 mg thermo balance sensitivity, it was used to evaluate the thermal stability of powder filler and vulcanized rubber nanocomposite films. The samples were heated from 50°C to 1000°C at a rate of 10°C per minute while being exposed to nitrogen gas at a flow rate of 50 mL per minute. The differential curve was used to determine the samples weight loss at the proper temperature.
Rheology characteristics
The curing durations (Tc90, min), scorch times (Ts2, min), and cure rate indices (CRI, min−1) at 162°C were measured using the TA instrument MDR 1 (Moving Die Rheometer), USA, in accordance with ASTM D2084 to ascertain the rheometric characteristics of rubber compounds.
Scanning electron microscopy (SEM)
Observing the surface morphology of things is possible using the scanelectron microscope (SEM), a type of electron microscopy. The Quanta FEG-250 was used to take SEM images of the samples in order to analyze the surface morphology of the produced NBR composites. The samples were covered with a very thin layer of gold, ranging in thickness from 5 to 10 nm, in around 6 min. The specimensmiddle cross-section was chosen to represent the overall morphology.
Mechanical properties evaluation
The tensile behavior of the NBR vulcanizates was assessed using a universal testing machine (Zwick Z010, Germany) following the ASTM D412 standard. Dumbbell-shaped specimens with a thickness of 1 mm were precision-cut from the compression-molded sheets using a Wallace die cutter (Model S6/1/6. A). The tests were conducted at room temperature under a constant crosshead speed of 500 mm/min to evaluate tensile strength, elongation at break, and modulus characteristics.
Shore A hardness measurements were performed using a digital durometer (Bareiss, Oberdischingen, Germany) following ASTM D2240. All measurements were taken at ambient conditions to ensure consistency and comparability across samples.
Measurements of swelling
They completed the equilibrium swelling Q test parts. For a full day, each specimen weighed between 0.1 and 0.2 g in a weighing bottle immersed in toluene to attain equilibrium swelling. Following weighing, the swollen samples were dried in an oven until their weight remained consistent. The samples are weighed to determine the final weight after being free of dissolved components. Utilizing the following formulas (1), the Q% was determined.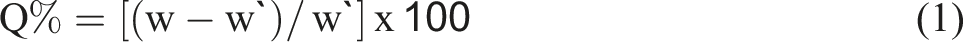
All of these tests were conducted at room temperature, and in accordance with ASTM D 471, the stated results were averaged from a minimum of five specimens.
Thermal oxidative aging
Aging of samples was carried out at 110 ± 1°C for 10 days in an air-circulating oven as ASTM D 572-04. The reported results were averaged from a minimum of five specimens.
Mechanical properties were evaluated to assess changes in tensile strength, elasticity, and hardness due to thermal-induced crosslinking or degradation. Additionally, swelling tests were repeated to determine potential changes in solvent uptake behavior, reflecting alterations in crosslinking or network integrity caused by thermal aging. This approach enabled a detailed comparison of the thermal aging response between unfilled and (Fe3O4/N–CQDs) -filled NBR matrices, providing insights into the (Fe3O4/N–CQDs) role in thermal aging resistance.
The aging factor (Ka), which measures changes in mechanical properties, was calculated from the results of the tensile test using the following 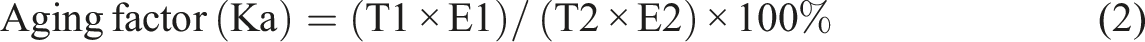
Magnetic properties
The magnetic characterization of NBR/Fe3O4/N–CQDs nanocomposites at room temperature was conducted using a Vibrating Sample Magnetometer (VSM, Lake Shore Model 7410, USA). Essential magnetic characteristics, such as saturation magnetization (Ms), remanent magnetization (Mr), and coercivity (Hc), were obtained from the hysteresis loops recorded under ambient conditions.
Dielectric spectroscopy technique
Dielectric and conductivity measurements were conducted using a high-resolution broadband impedance analyzer (Schlumberger Solartron 1260). The utilized AC electric field varied from 0.1 Hz to 1 MHz. Electromagnetic shielding was implemented on the sample holder to mitigate low-frequency noise. The impedance analyzer was interfaced with a PC using a GPIB cable IEE488 to facilitate automated measurements. Data was collected using LabVIEW. Calibration was conducted prior to sample readings to eliminate stray capacitance. The errors in the dielectric constant ε′ and the loss tangent tan δ are 1% and 3%, respectively. The sample temperature was regulated by a temperature controller equipped with a Pt 100 sensor. Desiccators loaded with silica gel maintained the samples' dryness. Subsequently, the sample was positioned in the measuring cell containing P2O5 until measurements were conducted.
Results and discussion
In this study, magnetic nitrogen-doped carbon quantum dots (Fe3O4/N–CQDs) were synthesized and characterized via FTIR, XRD, TGA, and TEM. These nanohybrids were then incorporated into an NBR matrix to develop multifunctional nanocomposites. The curing kinetics, mechanical performance, equilibrium swelling, and thermal stability of the resulting nanocomposites were investigated. Additionally, the thermal aging resistance, magnetic response, and dielectric behavior were evaluated to determine the influence of the filler on the resulting NBR composites.
Analysis of thermogravimetric data (TGA/DTG)
The thermal stability and decomposition behavior of the Fe3O4/N–CQDs were investigated by TGA/DTG analysis over the temperature range of 25–1000°C (Figure 1). The thermogram reveals a multistep degradation process characteristic of nitrogen-doped carbon quantum dots hybridized with magnetite nanoparticles. The first weight-loss stage occurs below 100°C, with a DTG maximum around 70–80°C, and is attributed to the removal of physically adsorbed moisture and residual volatile species associated with the hydrophilic surface of the carbon dots.
8
The second degradation region appears as two partially overlapping DTG peaks between approximately 90 and 245°C, corresponding to the decomposition of labile oxygen- and nitrogen-containing surface functional groups, such as hydroxyl, carboxyl, carbonyl, and amine moieties introduced during nitrogen doping and carbonization. Similar behavior has been reported for N-doped graphene quantum dots and magnetite–CQD hybrid systems.31,32 TGA of magnetite Fe3O4/N-CQDs.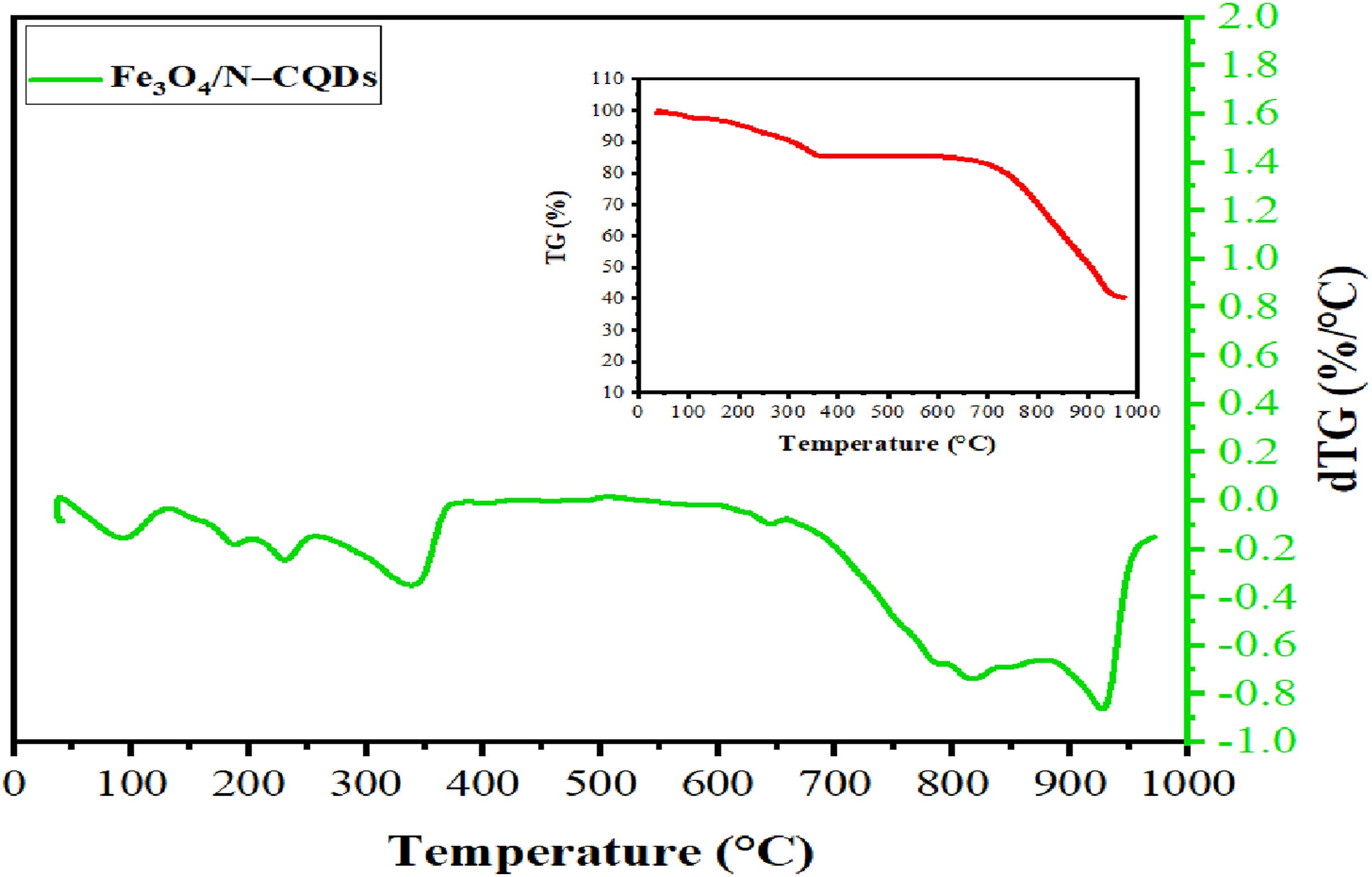
The third decomposition stage, extending from about 245 to 450°C with DTG maxima near 300 and 400°C, is attributed to the thermal degradation of more stable organic components and the partial breakdown of the carbonaceous framework of the quantum dots. This stage reflects the decomposition of condensed carbon structures and chemically bound functional groups.
A final broad weight-loss stage is observed above approximately 520°C and extends up to 980°C, which is associated with the gradual decomposition and oxidation of the carbon core, along with possible structural rearrangement or mineralization of the iron oxide phase. At these elevated temperatures, magnetite is expected to undergo partial oxidation or phase transformation toward more thermodynamically stable iron oxide phases (e.g., hematite), consistent with observations reported in the literature. 33
X-ray diffraction (XRD)
The X-ray diffraction (XRD) pattern of the Fe3O4/N–CQD nanocomposite is presented in Figure 2. A broad diffraction feature centered around 22–27° is observed, which is attributed to the presence of nitrogen-doped carbon dots. In addition, characteristic diffraction peaks appearing at 2θ ≈ 30.1°, 35.5°, 43.2°, 53.4°, 57.0°, and 62.6° correspond to the (220), (311), (400), (422), (511), and (440) crystallographic planes of cubic magnetite (Fe3O4), respectively. These reflections are consistent with the standard magnetite phase (JCPDS No. 01-076-7166), confirming the successful incorporation of Fe3O4 nanoparticles within the N–CQD structure.
8
This result is in agreement with previous report.34–36 XRD of magnetite Fe3O4/N-CQDs.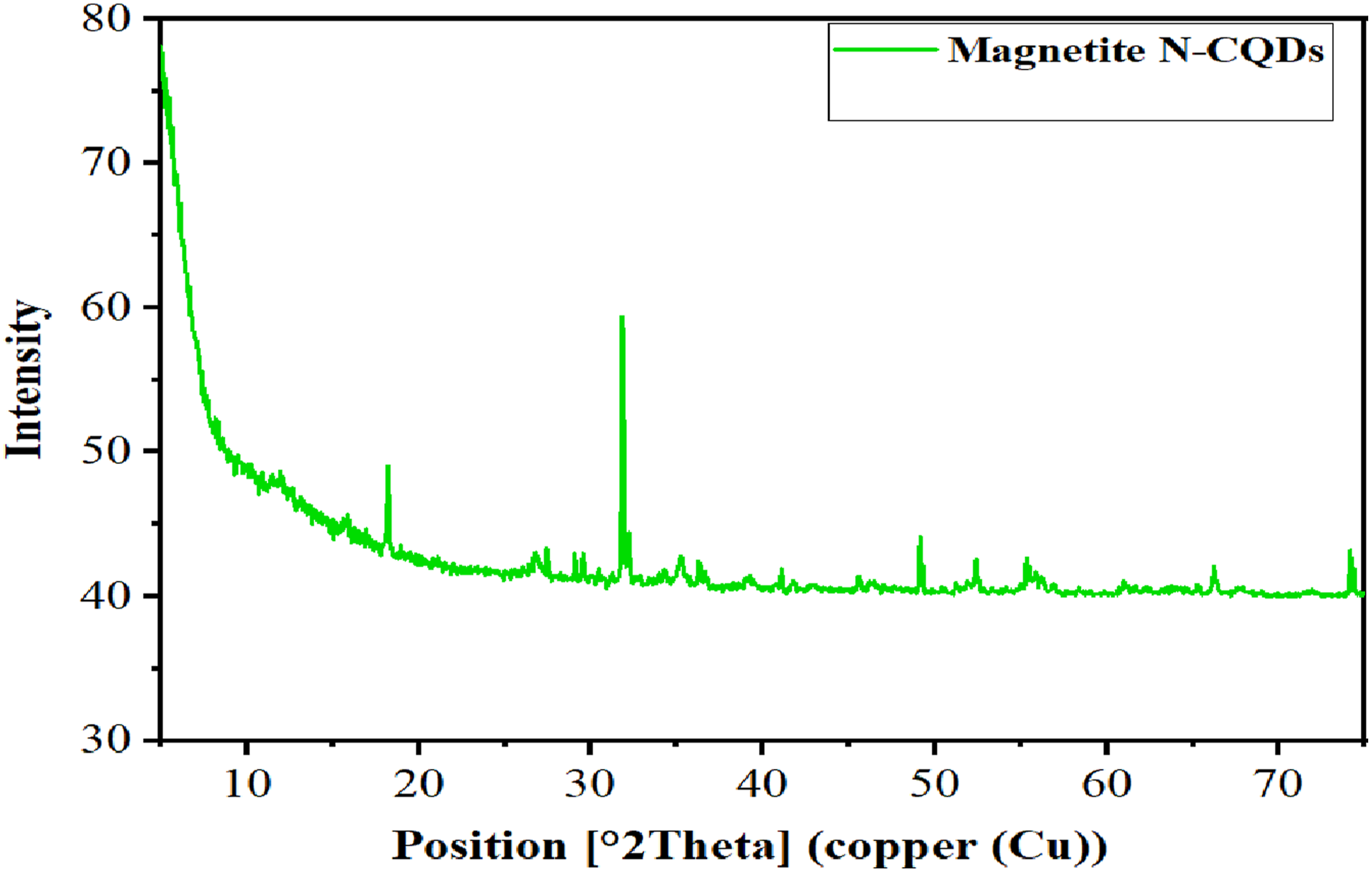
Infrared fourier transforms spectroscopy (FTIR)
The FTIR spectrum of the magnetite N–CQDs is shown in Figure 3. A broad absorption band observed between 3191 cm−1 and 3330 cm−1 is assigned to O–H and N–H stretching vibrations, indicating the presence of multiple hydroxyl and amine functional groups on the NCQD surface.
37
The absorption band at 1621 cm−1 is attributed to C = O stretching vibrations, whereas the band at 1409 cm−1 corresponds to C = C stretching of the carbon framework. The band at 1064 cm−1 is associated with O–C = O stretching, confirming the presence of carboxylate groups. The absorption at 865 cm−1 is assigned to C–N stretching, providing evidence of nitrogen incorporation into the carbon dots. Finally, the characteristic absorption band at 655 cm−1 corresponds to Fe–O stretching vibrations, confirming the successful incorporation of magnetite (Fe3O4) within the N–CQD structure.8,38 FTIR of magnetite Fe3O4/N-CQDs.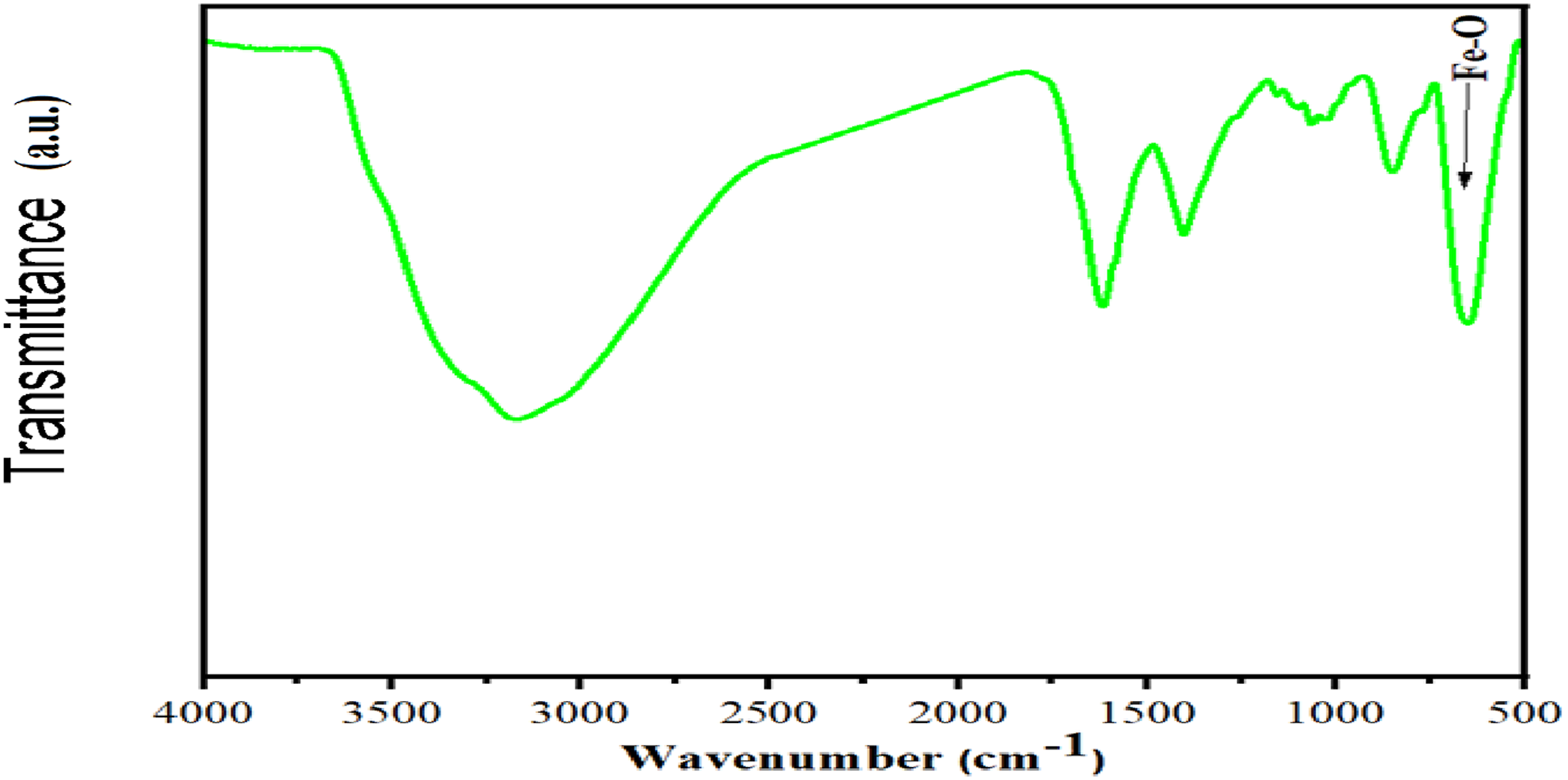
Morphological studies by using transmission electron microscopy (TEM)
Typical TEM images and diameter distributions of N-CQDs and Fe3O4/N-CQDs are shown in Figure 4. The TEM image of N-CQDs reveals uniformly spherical dots with diameters ranging from approximately 2.35 to 2.90 nm indicating good monodispersity and the absence of aggregation. These observations are consistent with previously reported HRTEM results, which showed spherical, homogeneously monodispersed CQDs with no noticeable aggregation and particle sizes in the range of 7–16 nm.
39
In contrast, for the Fe3O4/N-CQDs nanomaterial, the CQDs embedded between the platelets have diameters of about 0.74 to 0.97 nm, while the platelets themselves aggregate to sizes between 5.77 and 8.00 nm. Due to the small size and high dispersion of the N-CQDs, individual N-CQDs were not distinctly visible within the Fe3O4/N-CQDs sample. These observations, together with the FTIR and XRD results, confirm the successful synthesis of the Fe3O4/N-CQDs.
8
TEM image of N-CQDs and Fe3O4/N-CQDs.
Rheological studies
Rheological properties of the prepared NBR vulcanizates.
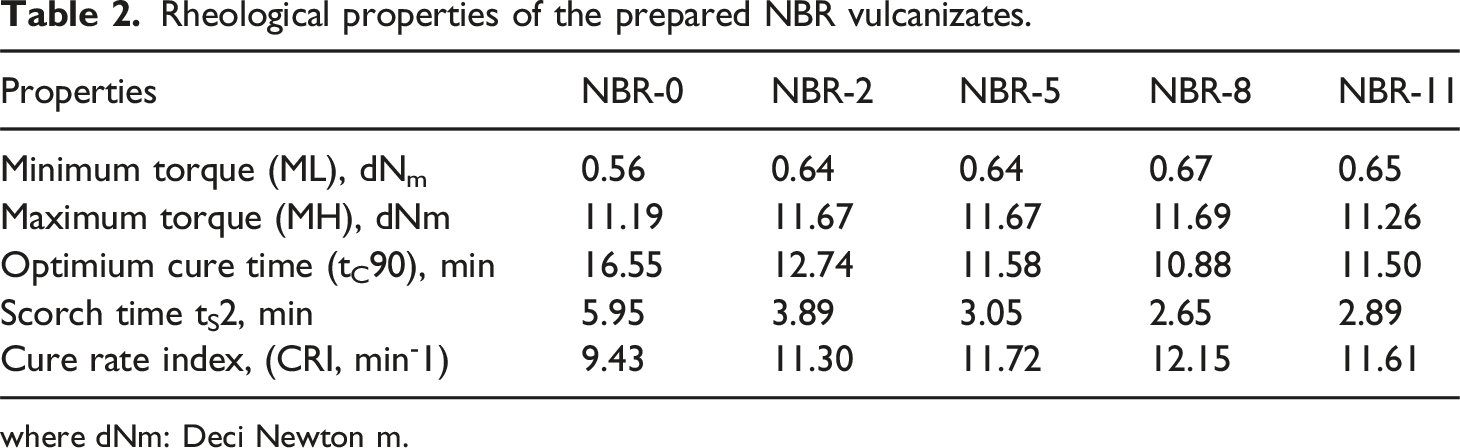
where dNm: Deci Newton m.
The minimum torque (ML), which reflects the viscosity and stiffness of the uncured rubber compound, shows a slight increase with the incorporation of Fe3O4/N-CQDs compared to neat NBR. This increase indicates enhanced filler–rubber interactions and restricted chain mobility due to the presence of well-dispersed nanofillers. 40 Similar behavior has been reported in carboxylated XNBR latex reinforced with carbon dots (CDs), where a decrease in the tan δ peak height (tan δmax) was observed with increasing CD concentration. 15 The reduction in tan δmax reflects decreased polymer chain mobility during the dynamic transition process and is also attributed to strong polymer–filler interactions. In the present study, the highest ML value is observed for NBR-8, suggesting the formation of an interconnected filler–rubber network, whereas the slight reduction in ML at NBR-11 is likely due to partial aggregation of Fe3O4/N-CQDs, which reduces the effective interaction area between filler and polymer chains. The maximum torque (MH), which is directly related to the stiffness and crosslink density of the vulcanized rubber, increases progressively with increasing Fe3O4/N-CQDs content and reaches a maximum value of 11.69 dNm for NBR-8. This enhancement indicates improved crosslink density and reinforcement efficiency, arising from strong interfacial interactions between N-CQDs and the NBR matrix. The slight decrease in MH observed for NBR-11 is attributed to excessive filler loading causing aggregation, which hinders stress transfer and crosslink formation.
The scorch time (tS2), representing the processing safety of the rubber compound, decreases significantly with increasing Fe3O4/N-CQDs loading. Compared with neat NBR, tS2 decreases from 5.95 min to 2.65 min at NBR-8, indicating a faster onset of vulcanization. This behavior suggests that Fe3O4/N-CQDs act as cure activators, likely due to the presence of nitrogen-containing functional groups on the N-CQDs surface and the catalytic activity of Fe3O4, which facilitate sulfur crosslinking reactions.41,42 In contrast to previously reported DVCR/N-CDs systems, where the t10 time was initially longer due to acidic surface groups requiring MgO to neutralize them before rapid vulcanization could proceed, in the Fe3O4/N-CQDs/NBR system the acidic groups interact with Fe3O4 and do not inhibit curing. 43 As a result, the vulcanization starts faster without an introduction period. At higher filler loading (NBR-11), a slight increase in tS2 is observed, which may be attributed to partial nanofiller aggregation that limits the accessibility of active functional groups and reduces curing efficiency. Similarly, the optimum cure time (tC90), defined as the time required to reach 90% of the final crosslink density, decreases markedly with increasing Fe3O4/N-CQDs content, from 16.55 min for neat NBR to 10.88 min for NBR-8, confirming accelerated curing kinetics. This behavior is consistent with the same mechanism observed for tS2. A slight increase in tC90 beyond 8 phr follows the same trend as tS2, attributed to restricted polymer chain mobility and reduced availability of reactive sites due to excessive nanofiller loading.
The cure rate index (CRI), which quantitatively describes the curing speed, increases significantly with Fe3O4/N-CQDs addition and reaches a maximum value of 12.15 min−1 at NBR-8. This trend confirms the enhanced vulcanization rate due the presence of nitrogen-containing functional groups on the N-CQDs surface and the catalytic activity of Fe3O4. The decrease in CRI at NBR-11 further supports the negative effect of excessive filler loading on curing efficiency.
From the rheological results, it has been demonstrated that Fe3O4/N-CQDs significantly influence the curing behavior of NBR by enhancing filler–rubber interactions, accelerating cure kinetics, and increasing crosslink density. However, beyond an optimal filler loading of 8 phr, the benefits diminish due to filler aggregation and restricted chain mobility.
Fourier transforms infrared spectroscopy for NBR and NBR/(Fe3O4/N-CQDs)
Figure 5 shows the FTIR of NBR and NBR/(Fe3O4/N-CQDs) composites. As observed from this figure that The FTIR spectrum of vulcanized NBR displayed the characteristic absorptions at ∼2237 cm−1 (C≡N stretching), 2850–2919 cm−1 (aliphatic C–H stretching), and CH2 bending at ∼1440 and 1357 cm−1, consistent with previous assignments.44,45 Upon incorporation of (Fe3O4/N-CQDs), two notable spectral changes are observed. A new absorption band appears at 1596 cm−1, which is attributed to the asymmetric stretching vibration of carboxylate (–COO-) groups on the nitrogen-doped carbon dots.
46
In addition, a distinct band at approximately 696 cm−1 corresponds to the Fe–O stretching vibration of magnetite, confirming the presence of Fe3O4 within the composite.
8
Moreover, the broadening of the 3200–3500 cm−1 regions indicates O–H/N–H stretching from CQD surface functionalities.
37
These modifications confirm the successful incorporation of (Fe3O4/N-CQDs) into the NBR matrix and suggest interfacial interactions between the polymer chains and the functional groups of the carbon dots. FTIR of NBR and NBR containing Fe3O4/N-CQDs.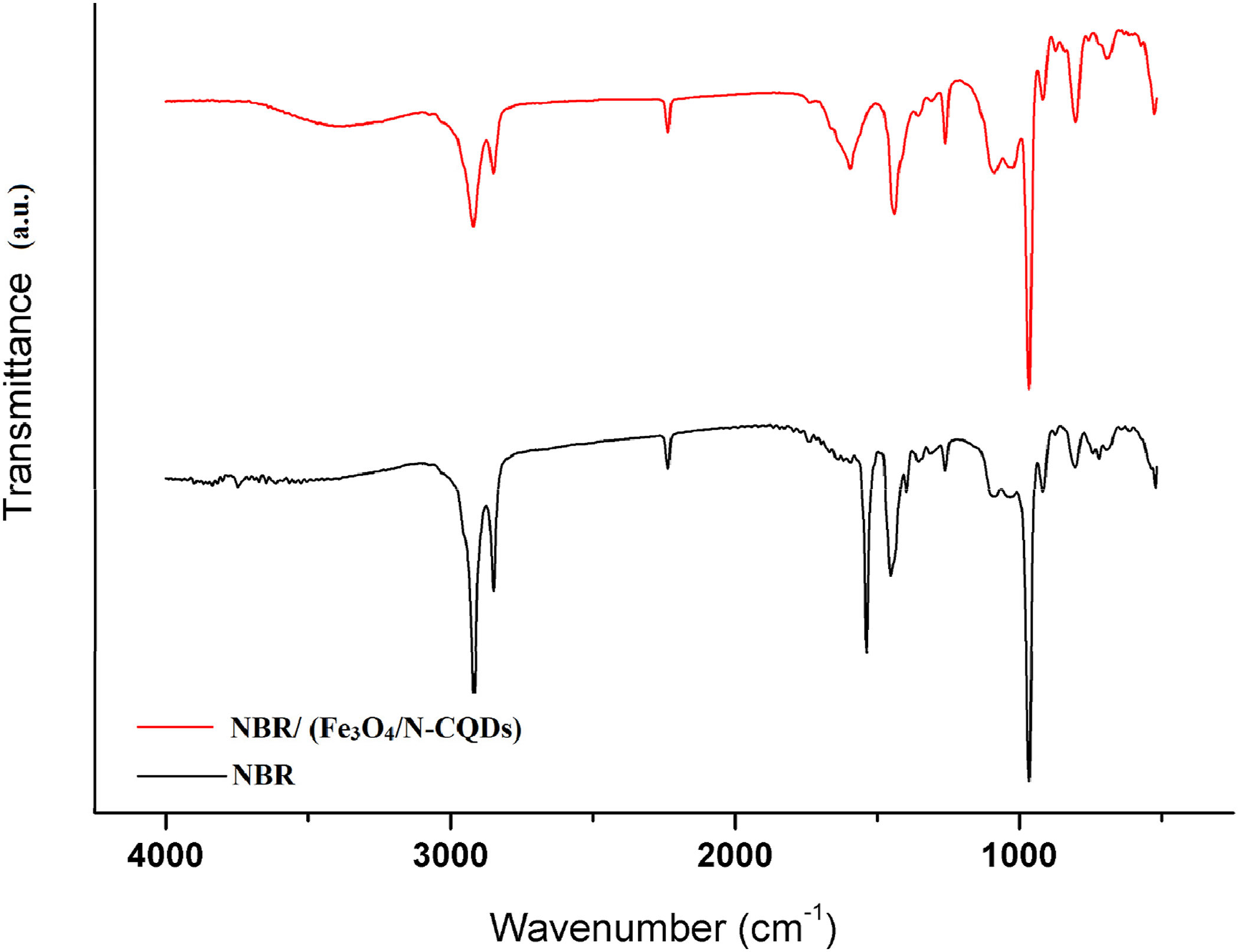
Based on the FTIR results, multiple interfacial interactions are involved in the bonding between the NBR matrix and the Fe3O4/N-CQDs hybrid filler. The polar functional groups on the surface of the CQDs (–OH, –NH2, and –COOH) interact with the nitrile (–C≡N) groups of NBR through hydrogen bonding and dipole–dipole interactions, enhancing the interfacial affinity between the filler and the rubber matrix.46,47
Under vulcanization conditions, the elevated temperature and curing agents can induce partial deprotonation of surface carboxylic groups on the CQDs, enabling coordination with Fe3O4 nanoparticles. 46 This coordination is supported by the appearance of a new band at 1596 cm−1, assigned to the asymmetric stretching of carboxylate (–COO-) groups, and by the shift of the Fe–O stretching vibration from 655 cm−1 in pristine Fe3O4/N-CQDs to 696 cm−1 in the composite. Coordination between the CQDs and Fe3O4 stabilizes the hybrid filler structure, which in turn reinforces the physical interactions at the NBR–filler interface, improving adhesion and dispersion of the hybrid nanofiller. In addition, the flexible NBR chains can wrap around the Fe3O4/N-CQDs, providing mechanical interlocking and further enhancing the overall structural integrity of the composite.
Mechanical properties of NBR vulcanizates reinforced with magnetic carbon dots (Fe3O4/N-CQDs)
Mechanical properties of NBR vulcanizates loaded with different concentrations of magnetic carbon dot (Fe3O4/N-CQD).
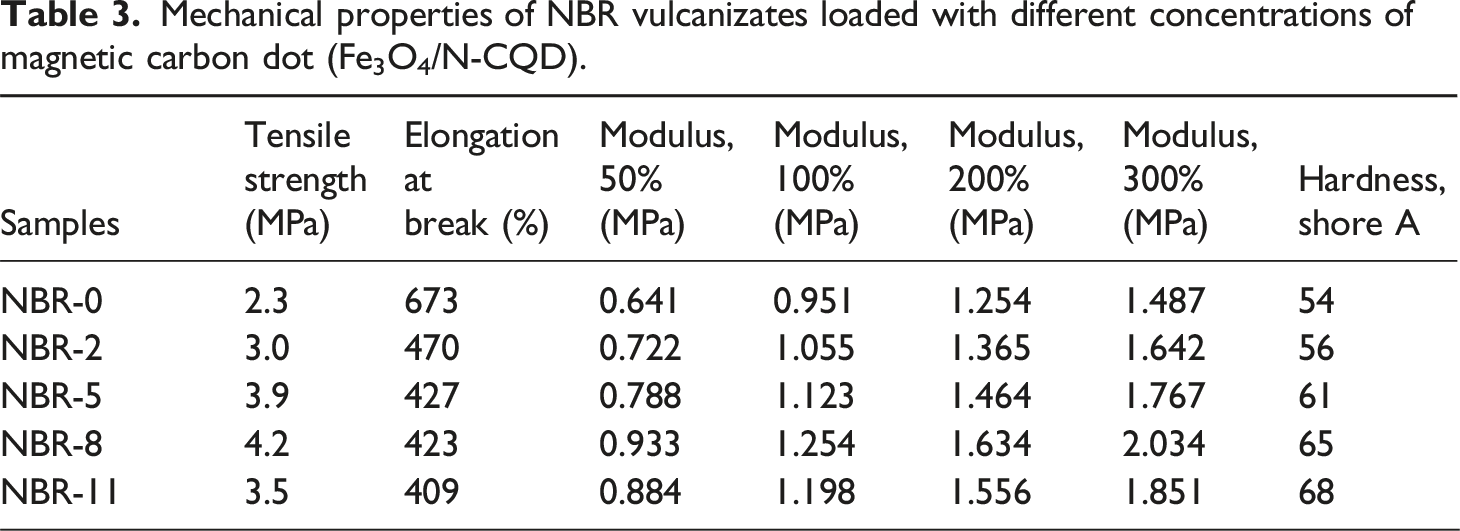
Elongation at break shows a decreasing trend with increasing Fe3O4/N-CQD content. The reduction in elongation reflects the increased rigidity of the vulcanizates due to filler incorporation, which restricts polymer chain mobility. Consistent behavior has been reported in polymer nanocomposites reinforced with graphene oxide nanosheets (GOn) and ferromagnetic iron oxide (Fe3O4) nanoparticles, where increasing filler content led to reduced elongation due to constrained chain motion and increased stiffness in PVDF/SBR-based hybrid systems. 40
These results indicate that moderate loading of Fe3O4/N-CQDs (up to 8 phr) provides optimal reinforcement, improving tensile strength and modulus. Excessive filler loading leads to agglomeration, reduced polymer–filler interactions, and limited stress transfer, which diminish the mechanical benefits.
Hardness of NBR/Fe3O4/N-CQD vulcanizates
The hardness of NBR vulcanizates increases steadily with Fe3O4/N-CQD loading, from 54 Shore A for neat NBR to 68 Shore A at 11 phr (Table 3). This continuous increase reflects the addition of rigid nanofillers, which restrict local polymer chain mobility and enhance resistance to surface deformation. 48
It was also noticed that this trend differs from the modulus and tensile strength, which showed an increase up to 8 phr and slightly decrease at higher loadings. This divergence can be explained by the fact that hardness is primarily a measure of local surface rigidity, whereas modulus and tensile strength depend on efficient stress transfer across the bulk material. 49 At high filler content, partial aggregation of Fe3O4/N-CQDs reduces bulk stress transfer, lowering modulus and tensile strength, but the surface stiffness, and thus hardness, continues to rise due to the overall filler content.
Swelling behavior of NBR/Fe3O4/N-CQD vulcanizates
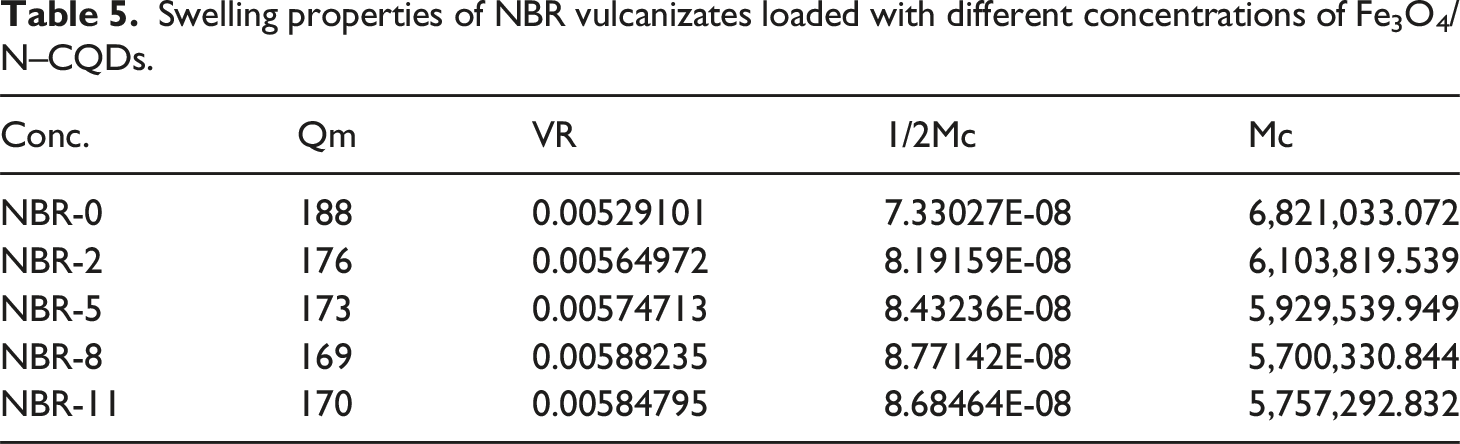
Equilibrium swelling of NBR vulcanizates loaded with different concentrations of magnetic carbon dot (Fe3O4/N-CQD).

The crosslinking density of NBR and NBR/Fe3O4/N–CQDs nanocomposites was quantitatively determined from equilibrium swelling measurements in toluene for 24 h at 25 ± 1°C and calculated using the Flory–Rehner equation.
12
The crosslink density (v) and the molecular weight (g/mol) between crosslinks (Mc) were calculated as follows:
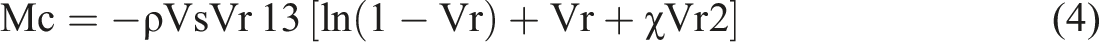
Swelling properties of NBR vulcanizates loaded with different concentrations of Fe3O4/N–CQDs.
SEM images of NBR samples
Figure 6(a)–(c) shows the fracture surface morphology of pristine NBR, NBR/(Fe3O4/N-CQDs) at 8 phr, and NBR/(Fe3O4/N-CQDs) at 11 phr at low and higher magnifications. The SEM micrograph of pristine NBR (Figure 6(a)) shows a relatively smooth surface without obvious defects, except for a few asperity pits and original processed textures, which is typical of unfilled rubber matrices and indicates limited resistance to crack propagation.46,53 SEM of (a and b) NBR, (c and d) NBR/(Fe3O4/N-CQDs) (8 phr)and (e and f) NBR/(Fe3O4/N-CQDs) (11 phr) at low and high magnification.
With the incorporation of 8 phr Fe3O4/N-CQDs (Figure 6(b)), the fracture surface becomes significantly rougher and more irregular. This behavior is analogous to that reported for NR reinforced with hybrid CDs/MoS2 fillers, where the introduction of a low filler content promoted a more uniform mixed-filler system. 46
At higher filler loading (11 phr, Figure 6(c)), localized platelet-like structures and micro-scale agglomerates are observed on the fracture surface, particularly at higher magnification. This behavior closely resembles that reported in CDs/MoS2-reinforced NR at elevated filler contents, where excessive filler loading led to deteriorated dispersion and aggregation. 46 This result is consistent with the slight decline in mechanical performance observed at higher filler concentrations.
Thermal analysis
Figure 7(a) and (b) presents the TGA and DTG curves of neat NBR and NBR filled with Fe3O4/N-CQDs. Both samples exhibit a single dominant thermal degradation step, which is characteristic of NBR and is mainly associated with the decomposition of the polybutadiene backbone and acrylonitrile segments. Compared with neat NBR, the Fe3O4/N-CQDs–filled NBR shows a noticeable shift of the main degradation region toward higher temperatures, indicating a delay in the thermal decomposition process. This improvement in thermal stability can be attributed to the presence of thermally stable inorganic Fe3O4 nanoparticles and the strong interfacial interactions between N-CQDs and the NBR matrix. Similar behavior has been reported in PVDF/SBR nanocomposites reinforced with Fe3O4 and graphene oxide, where the incorporation of inorganic nanofillers increased the onset degradation temperature and shifted the maximum degradation temperature (Tmax) to higher values due to their barrier effect and high intrinsic thermal stability.37,40 TGA (a) and DTG (b) curves of neat NBR and NBR filled with Fe3O4/N-CQDs.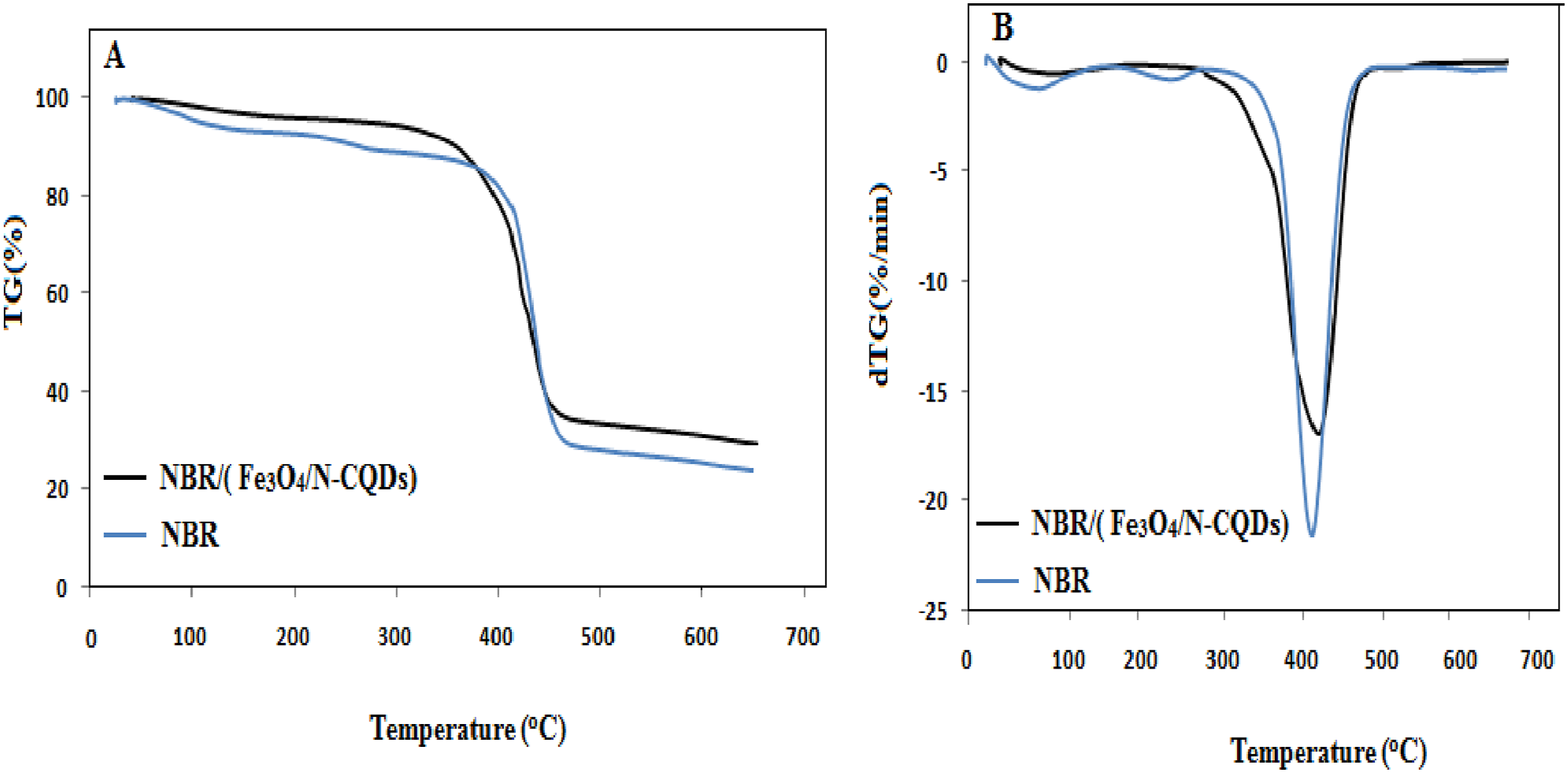
The DTG curves further support this observation, as the maximum degradation rate of the composite shifts slightly toward higher temperatures compared to neat NBR, confirming a slower degradation kinetics. This trend is consistent with previous reports on Fe3O4-based polymer nanocomposites.40,53
In addition, the char yield of the Fe3O4/N-CQDs/NBR composite (∼30%) is significantly higher than that of neat NBR (∼24%). The increased residue content is attributed to the inorganic Fe3O4 component and the carbonaceous structure of the CQDs, which promote char formation and enhance thermal resistance at elevated temperatures. Similar increases in char yield have been observed in Fe3O4@GO-reinforced polymer systems.37,40
Thermal aging resistance
Thermal aging refers to the progressive degradation of polymeric materials resulting from prolonged exposure to elevated temperatures, often in the presence of oxygen. In elastomers such as nitrile butadiene rubber (NBR), which contain unsaturated bonds, thermal-oxidative aging can induce chain scission, oxidative reactions, and undesirable changes in crosslink structure. These processes typically lead to deterioration in mechanical properties, reduced elasticity, and ultimately premature material failure. Consequently, improving the resistance of rubber composites to thermal aging is critical for extending their service life. 12,53
Mechanical properties after aging
The thermal aging behavior of NBR compounds was evaluated by aging the samples at 110 ± 1°C for 10 days. The variations in tensile strength, elongation at break, and hardness after aging are presented in Figure 8(a)–(c). The aging behavior of Fe3O4/N-CQD–filled NBR was systematically compared with that of unfilled NBR (NBR-0). For neat NBR, a significant deterioration in mechanical properties was observed after aging. The tensile strength decreased from 2.3 MPa to 1.5 MPa, corresponding to a loss of approximately 35% of its initial strength, which can be attributed to thermal oxidative degradation and chain scission. In contrast, all Fe3O4/N-CQD–filled NBR samples exhibited markedly improved tensile strength retention after aging. It was also noticed that the NBR-8 sample showed the highest tensile strength after aging, reaching 5.9 MPa, indicating superior resistance to thermal aging compared to unfilled NBR. The improved aging resistance of Fe3O4/N-CQD–filled NBR can be attributed to several synergistic effects. The hybrid nanofillers act as effective barriers against thermal and oxidative degradation by restricting oxygen diffusion and limiting polymer chain mobility. In addition, carbon dots are known to possess radical-scavenging capability, which can suppress thermo-oxidative chain scission by neutralizing free radicals generated during aging, thereby retarding degradation of the rubber network.12,53,54 Physico-mechanical properties of NBR vulcanizates before and after aging: (a) tensile strength; (b) elongation at break; (c) Hardness.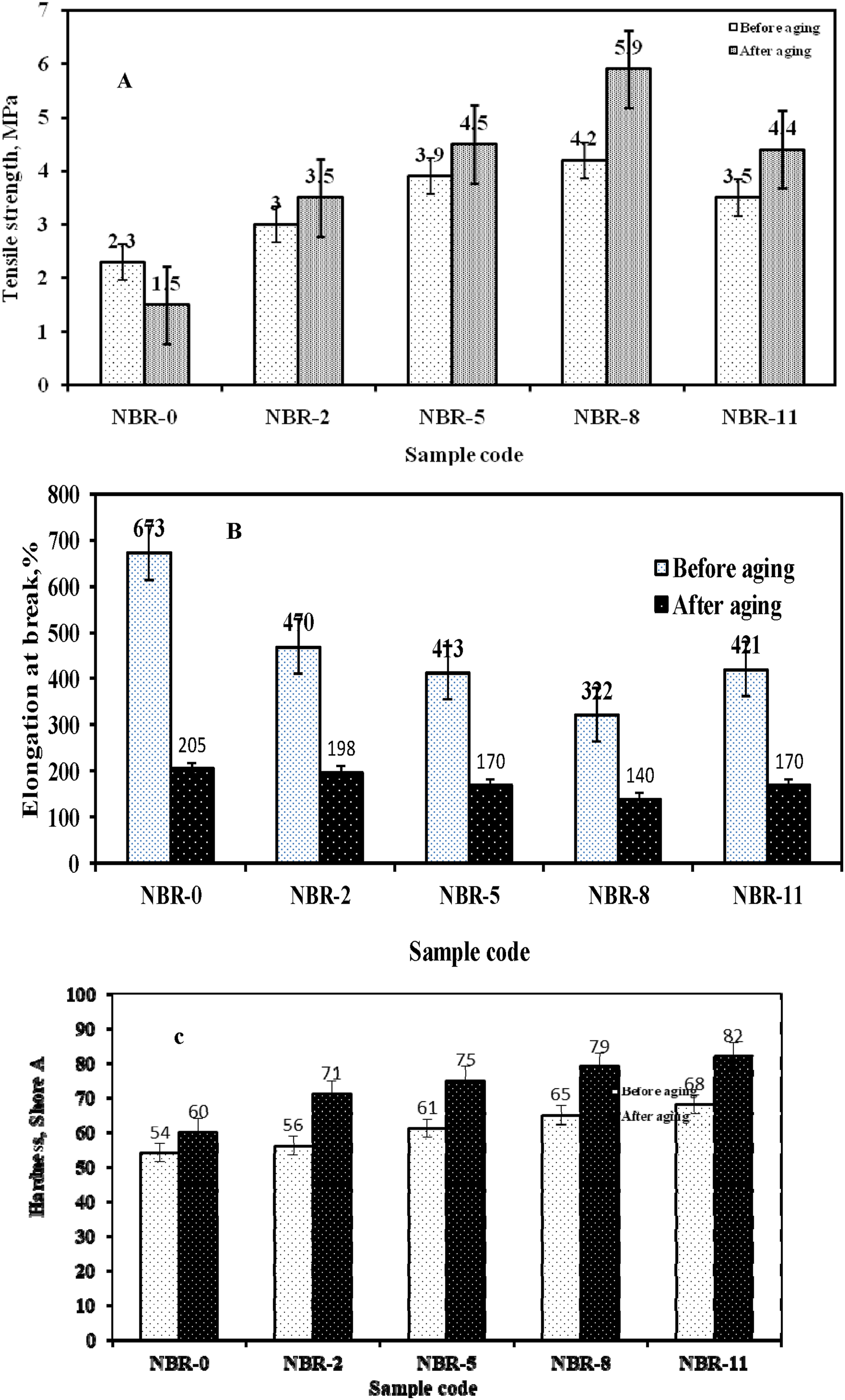
Elongation at break decreased for all samples after aging, reflecting increased stiffness caused by degradation of polysulfidic cross-links and the subsequent post-curing process. 52 For NBR-0, elongation at break dropped sharply from 673% to 205%, indicating a severe loss of elasticity due to dominant oxidative degradation. Hardness measurements further support these observations, as the hardness of all samples increased after aging. This increase is primarily attributed to the transformation of thermally unstable polysulfide cross-links into shorter mono- and disulfide cross-links, which are more resistant to heat and oxidation and result in a stiffer and more compact rubber network. 52 In addition, the polar functional groups on Fe3O4/N-CQDs (–OH, –COOH, –C = O) can form hydrogen bonds with oxygenated species generated in the aged NBR. These hydrogen bonds act as temporary physical crosslinks that restrict chain mobility and complement the chemical network, further stabilizing the rubber structure. 52 That is why Fe3O4/N-CQD–filled NBR samples exhibited higher Shore A hardness values than neat NBR.
From these results it was found that the incorporation of Fe3O4/N-CQDs significantly enhances the thermal aging resistance of NBR by improving tensile strength retention, maintaining reasonable ductility, and increasing hardness.
The aging factor
The product of tensile strength and elongation at break is commonly used as an indicator of a material’s ability to absorb mechanical energy during deformation prior to fracture.
55
Based on this concept, the aging factor (Ka) of the NBR vulcanizates was calculated to evaluate the overall retention of mechanical performance after thermal aging, and the results are represented in Figure 9. Values of aging factor after thermal oxidative aging.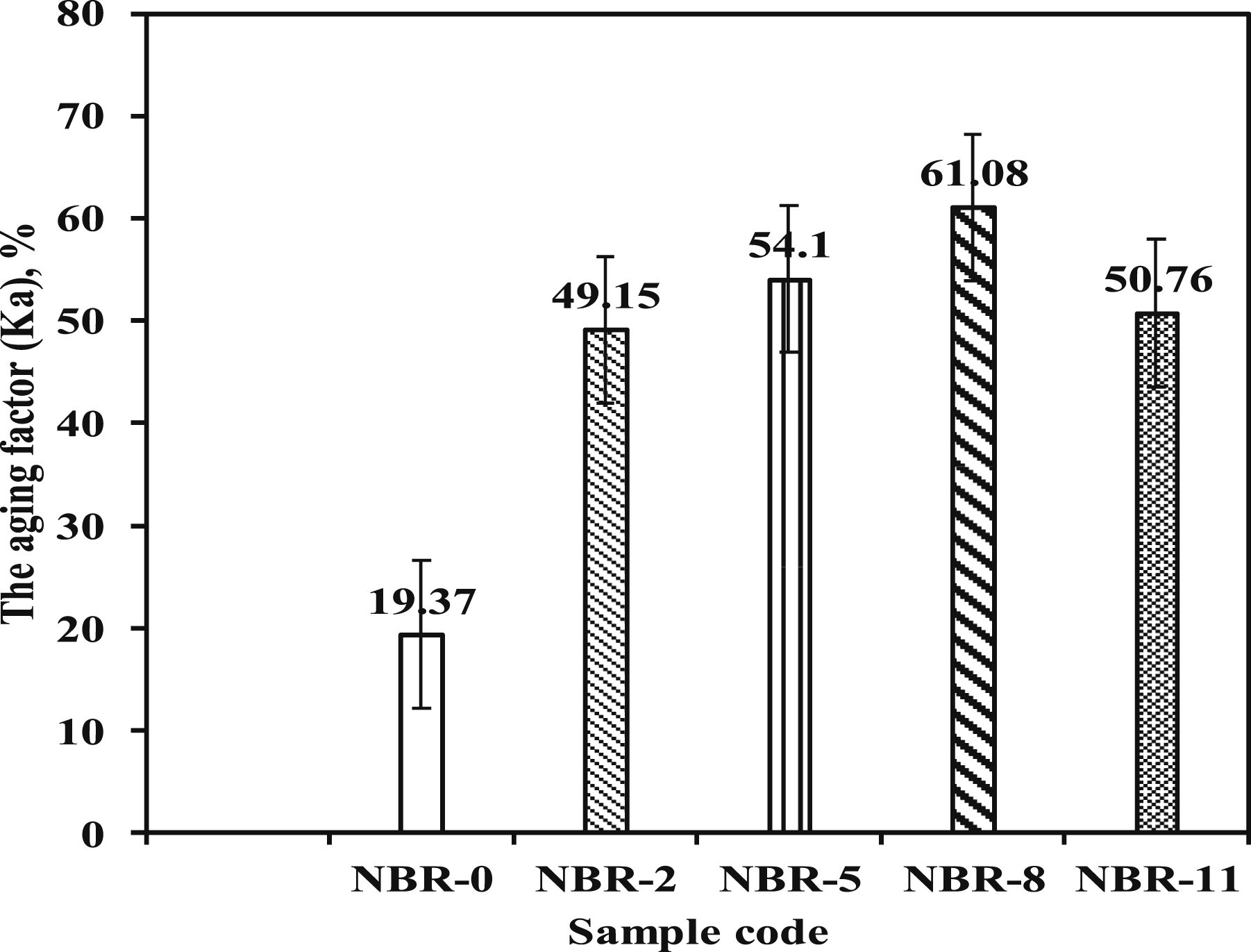
As shown in Figure 9, the aging factor of all NBR compounds increased after aging, indicating structural changes in the rubber network during prolonged thermal exposure. The incorporation of Fe3O4/N-CQDs significantly enhanced the aging factor compared to unfilled NBR. With increasing filler content, Ka increased gradually and reached a maximum at 8 phr, after which a slight decrease was observed at higher loading levels. For neat NBR (NBR-0), the aging factor increased from 19.37 before aging to 61.08 after 10 days of aging. It was also noticed that the NBR filled with 8 phr Fe3O4/N-CQDs exhibited the highest aging factor after aging, demonstrating superior retention of mechanical energy absorption capability. 55
The higher Ka values observed for Fe3O4/N-CQD–filled NBR indicate improved resistance to thermal aging. This enhancement is attributed to the synergistic effects of the hybrid nanofillers, including improved interfacial interactions, restriction of polymer chain mobility, and suppression of thermo-oxidative degradation. Although thermal aging generally leads to deterioration of rubber mechanical properties, the presence of CQDs helps maintain higher tensile strength and elongation at break, resulting in a higher aging factor compared to unfilled NBR.
Swelling properties after aging
The effect of thermal-oxidative aging on the swelling ratio (Q%) of NBR composites before and after aging is presented in Figure 10. For neat NBR (NBR-0), Q% increased from 188% to 194% after aging at 110°C, indicating the predominance of chain-scission processes that progressively weaken the rubber network under thermal-oxidative conditions. Equilibrium swelling of NBR vulcanizates before and after aging.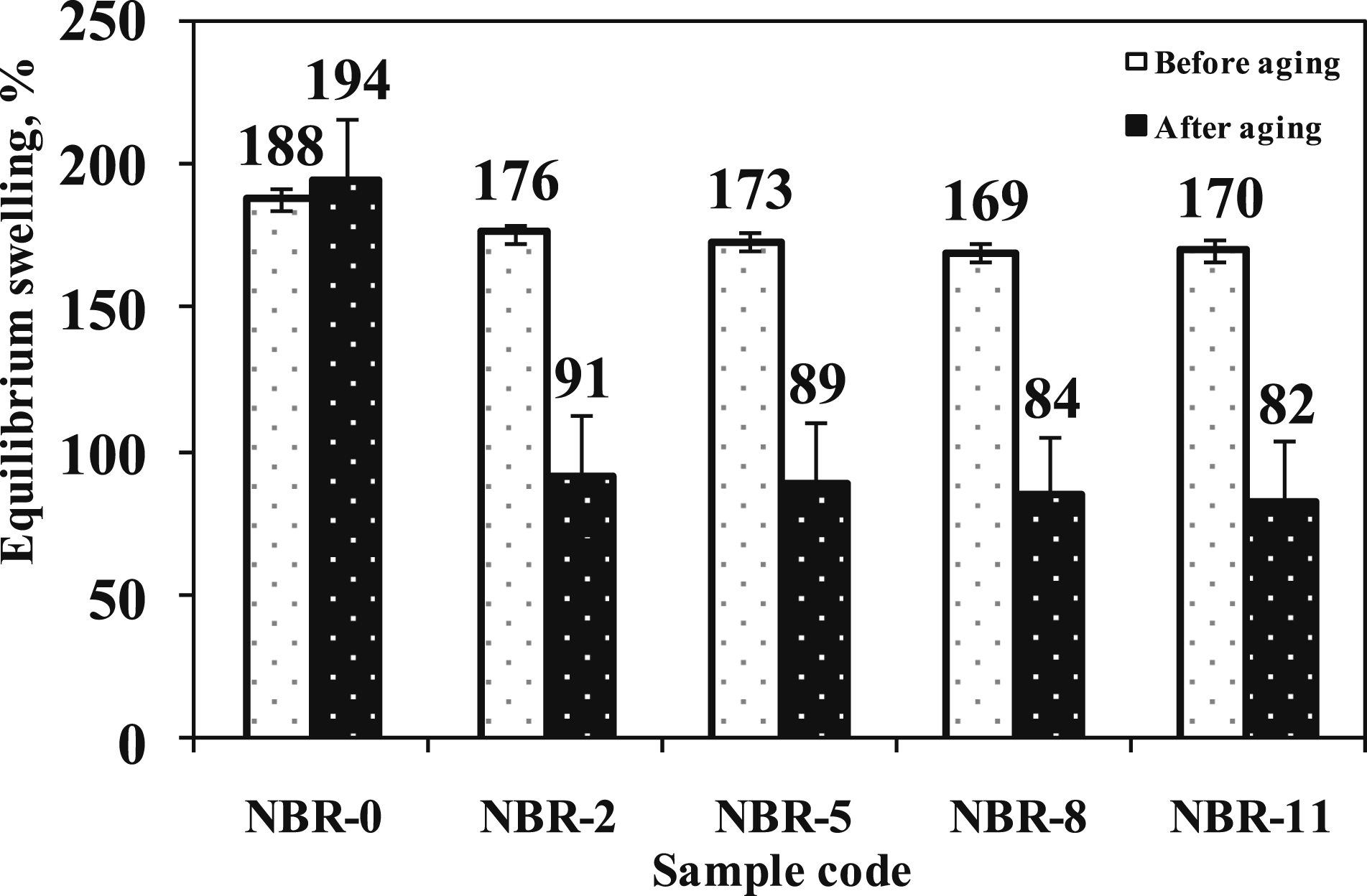
In contrast, the (Fe3O4/N-CQD)-filled composite (NBR-8) exhibited a pronounced decrease in Q%, from 180% before aging to 153% after aging. This reduction suggests that the nano-(Fe3O4/N-CQD) hybrid acts as an anti-aging additive by promoting post-curing and additional crosslink formation during aging, resulting in a denser and more stable network structure.12,53–55 Consequently, this network resists solvent uptake, resulting in the marked decrease in swelling observed in the aged composites.
Magnetic properties
The room-temperature magnetic characterization of NBR/Fe3O4/N–CQDs nanocomposites is presented in Figure 11. It exhibits a clear dependence on filler loading, as evidenced by the hysteresis loops and the summarized magnetic properties. Saturation magnetization (Ms) increases from 29.05x 10−3 emu/g at 0 phr to 91.32 × 10−3 emu/g at 11 phr, indicating the appropriate addition of magnetic fillers and an enhancement in magnetic response. This aligns with existing knowledge regarding Fe3O4-based polymer nanocomposites.
56
The coercivity (Hci) values exhibit a non-monotonic trend. The pristine matrix exhibits a notably high coercivity of 3546.3 G, likely attributed to residual magnetic impurities or anisotropy inherent to the matrix. The filled samples exhibit soft magnetic behavior, characterized by a Hci below 120 G, consistent with the effects of nanoscale magnetite dispersion.
57
The remnant magnetization (Mr) and squareness ratio (Mr/Ms) remain low across all samples, indicating super paramagnetic-like properties suitable for applications requiring rapid magnetic reversibility and minimal residual magnetization.
58
The more pronounced hysteresis loops observed with 11 phr loading indicate stronger magnetic interactions and a greater magnetic moment density. Slight loop asymmetries and negative coercivity values may result from internal stress or measurement errors. The findings suggest that NBR/Fe3O4/N–CQDs composites are suitable for flexible magnetic and magneto-responsive applications, and EMI shielding.
59
Magnetization curves of NBR/Fe3O4/N–CQDs nanocomposites.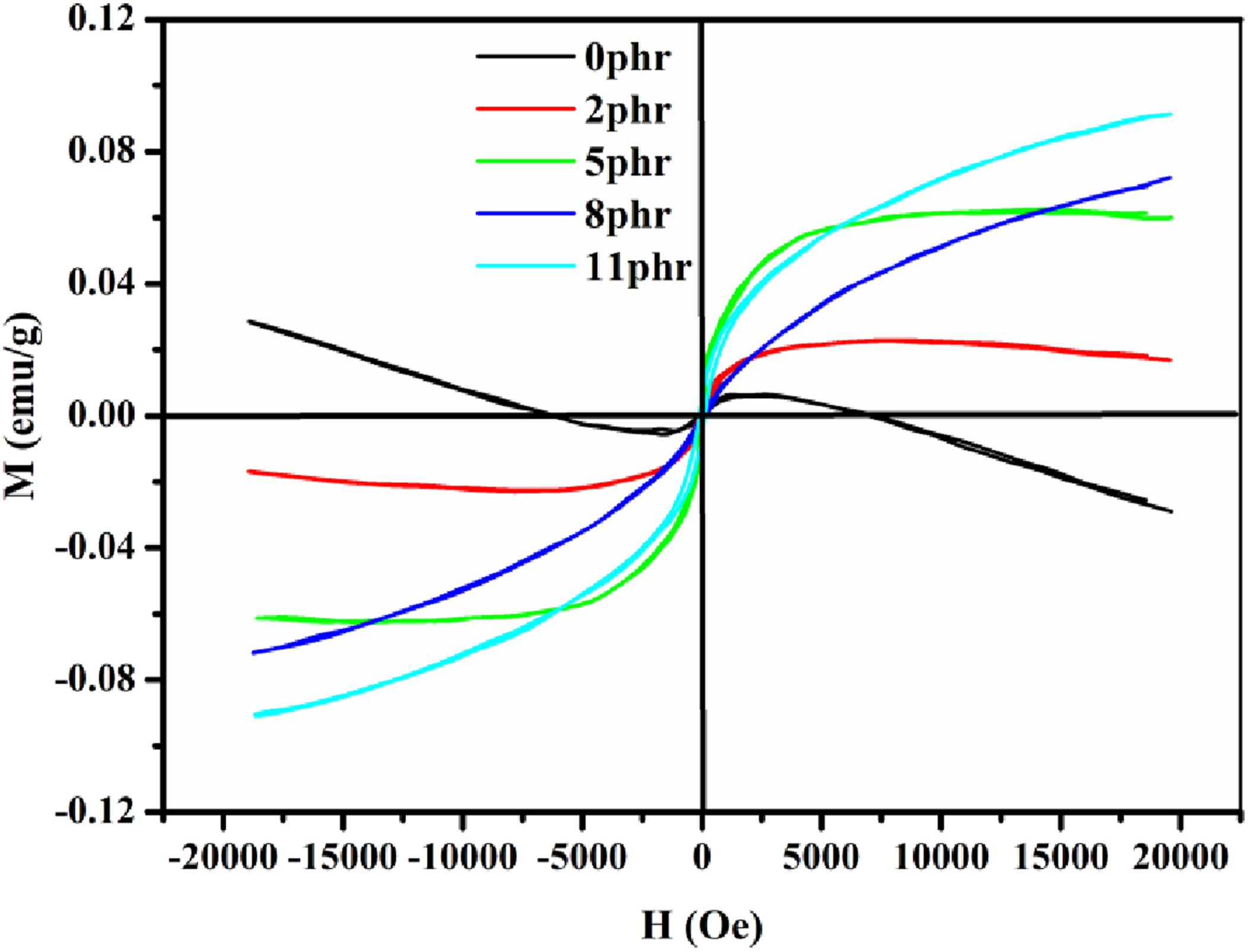
Dielectric properties
The incorporation of Fe3O4/N-CQDs into a rubber matrix significantly alters the composite’s dielectric and conduction properties, as evidenced by the frequency-dependent results presented in Figure 12(a)–(d). The hybrid filler, composed of magnetite nanoparticles (Fe3O4) encased in nitrogen-doped carbon quantum dots (N-CQDs), exhibits a distinctive combination of magnetic, conductive, and polar attributes that interact with the polymer matrix at various levels. Frequency-dependent dielectric properties of NBR rubber filled with Fe3O4/N-CQDs at varying concentrations (0,2, 5, 8, and 11 phr), measured at 25°C over the range 0.1 Hz to 1 MHz. (a) Real part of permittivity (ε′), (b) dielectric loss (ε″), (c) loss tangent (tanδ), and (d) Cole–Cole plot of electric modulus (M″ vs M′).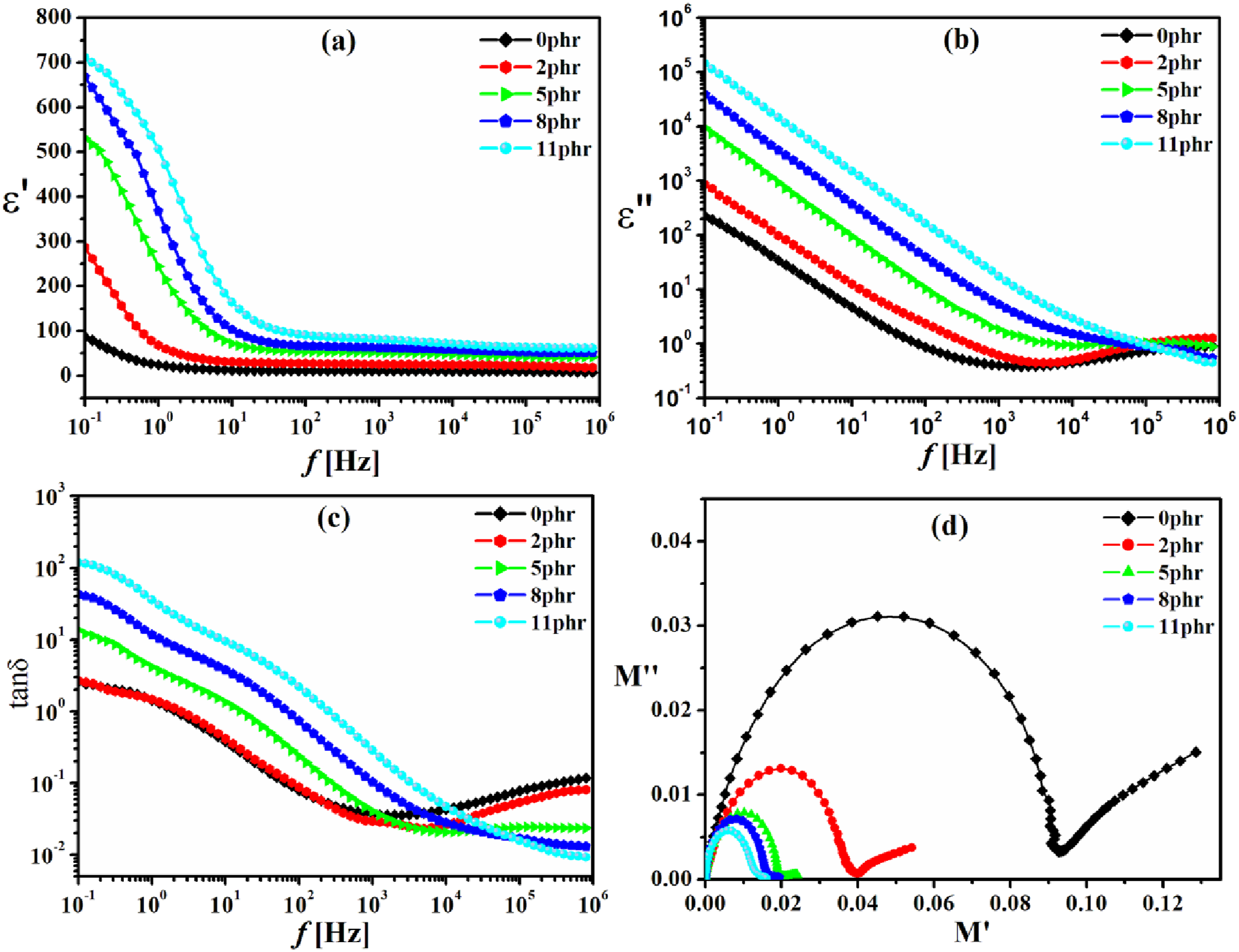
The real part of permittivity (ε′) signifies a material’s ability to retain electrical energy when subjected to an electric field. Figure 12(a) demonstrates that ε′ reduces with rising frequency as the loading of Fe3O4/N-CQDs increases, owing to dipolar orientation lag at higher frequencies. 60 The considerable rise in ε′ at low frequencies, with increased Fe3O4/N-CQDs loading, is attributed to interfacial polarization, which occurs when the conductivity and permittivity of the rubber matrix and Fe3O4/N-CQDs are distinctive. The accumulation of charges at the interfaces is referred to as the Maxwell–Wagner–Sillars (MWS) effect.61,62 The N-CQDs incorporate supplementary polar functional groups, including –NH and –OH, which enhance the number of dipolar sites and improve the dielectric response. The nanoscale arrangement of Fe3O4/N-CQDs improves the strength of microcapacitive networks, therefore elevating ε′. The observed saturation and loss in ε′ at high frequencies indicate that the polarization mechanisms are unable to sufficiently adapt to the rapid oscillations of the electric field, resulting in a decrease in permittivity.
Figure 12(b) illustrates the imaginary component of permittivity (ε″), which quantifies the energy dissipated via conduction and relaxation processes. It is obvious that the values of ε″ decreases as the frequency increases. However, as the frequency increases, the time available for dipoles to reposition is insufficient. Consequently, their impact on dielectric loss ε″ diminishes, resulting in a reduction of ε″. 60 On the other hand, the rubber composites with higher Fe3O4/N-CQDs content exhibited increased ε″ values, attributable to interfacial polarization and possibly hopping conduction among adjacent Fe3O4/N-CQDs. The Fe3O4component may generate localized magnetic and conductive regions that facilitate the movement of charge carriers in the presence of an electric field. This increases the dielectric loss, particularly in composites with extensive filler networks.63,64 In heterogeneous systems like rubber composites, charge carriers accumulate at interfaces due to differences in conductivity and permittivity across phases. This accumulation contributes to ε″. At elevated frequencies, the carriers cannot migrate or aggregate effectively, hence hindering interfacial polarization and further reducing ε″. 65
The loss tangent (tanδ), shown in Figure 12(c), indicates the efficiency of energy dissipation relative to energy storage. For all samples, tanδ decreases with increasing frequency, consistent with the observation that dielectric losses diminish at higher frequencies. 60 tanδ exhibits considerable values at low frequencies due to ohmic conduction losses and electrode polarization effects. However, it subsequently reaches a minimum in the mid-frequency range, specifically between 103 and 104 Hz. 66 The observed increase at elevated frequencies may suggest the onset of additional relaxation processes or the high-frequency limit of the primary relaxation. 52 In addition tanδ increases with the increasing concentration of Fe3O4/N-CQDs at low frequencies. The incorporation of Fe3O4/N-CQDs enhances energy dissipation, attributed to several factors: the restricted mobility of polymer chains at filler surfaces, increased friction between these surfaces, and the presence of polar and conductive sites that facilitate polymer relaxation. The broadening of tanδ peaks with increasing Fe3O4/N-CQDs content indicates a variety of relaxation times, suggesting complex interactions between the filler and the matrix. 67
The electric modulus formalism employed in Figure 12(d), where M* = 1/ε*, is highly effective for examining materials with elevated conductivity, as it eliminates electrode polarization artifacts and emphasizes bulk relaxation phenomena. 68 Figure 12(d) shows a Cole–Cole plot of electric modulus, with M″ on the y-axis and M′ on the x-axis. This model emphasizes relaxation dynamics by reducing the effects of electrode polarization. The semicircular arcs you see are common in dielectric relaxation processes. The 0phr sample has the broadest and most symmetrical arc. The arcs get thinner and more twisted as the content of Fe3O4/N-CQDs increases. Moreover, the diameter of the semi-circles decreased with increasing Fe3O4/N-CQDs content. This behavior is attributed to the increased conductivity of the rubber composites resulting from a rise in charge carriers. However, there are a lot of relaxation centers, such as filler-matrix interfaces, polar groups, and conductive domains. This leads to a large range of relaxation times and more variety. 69
Electrical conductivity
The frequency dependence of alternating current conductivity (σac) can be explained by Jonscher’s power law equation (5);
60
(a) The frequency dependence of ac NBR rubber filled with Fe3O4/N-CQDs at varying concentrations (0,2, 5, 8, and 11 phr), measured at 25°C and (b) Varation of conductivity with Fe3O4/N-CQDs loading.
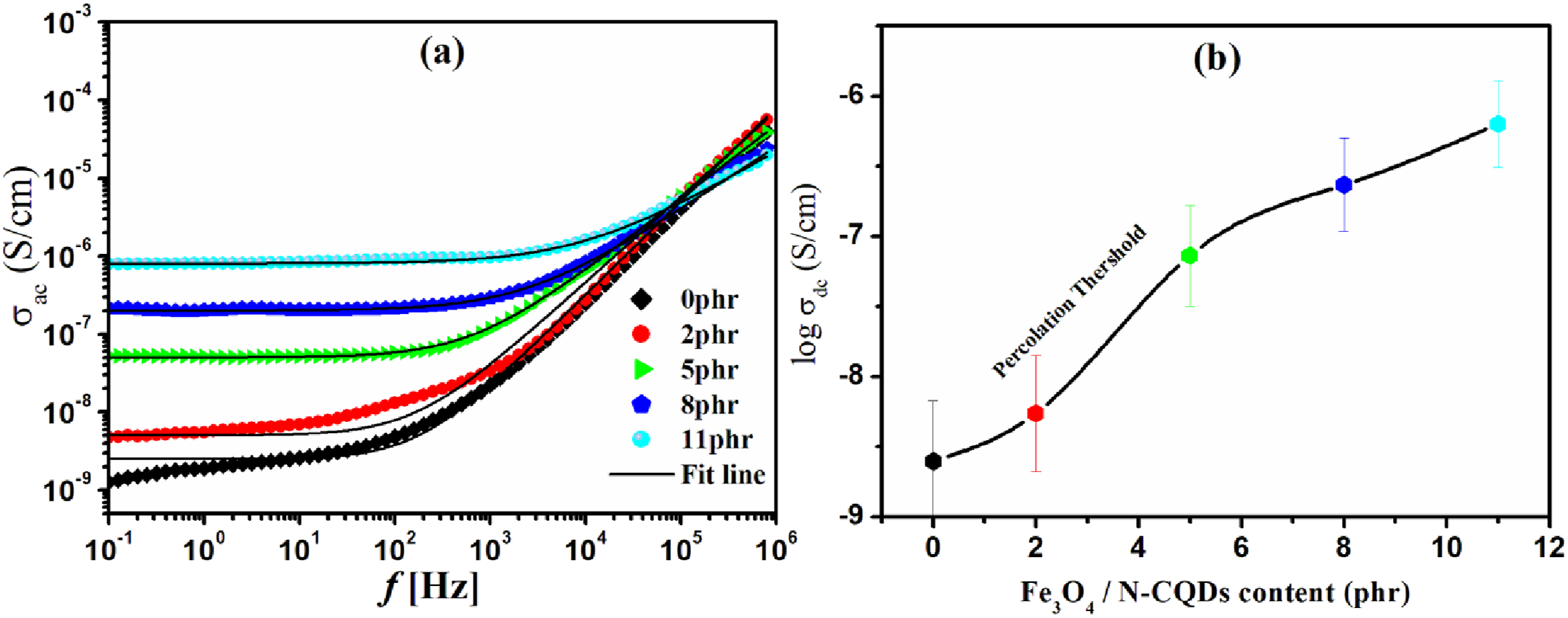
Figure 13(b) shows how the DC conductivity (σdc) changes with Fe3O4/N-CQD loading. As the concentration of Fe3O4/N-CQD doping rose, a significant rise in conductivity was noted, indicated by the percolation threshold. The direct current conductivity (σdc) values of the Fe3O4/N-CQD composites are found between 3 × 10−9 and 6 × 10−7 S/cm, placing these materials in the semiconducting to weakly conductive category. The defined conductivity range is advantageous for applications requiring antistatic characteristics or safeguarding against electrostatic discharge (ESD). Materials with σdc values under 10−5 S/cm are classified as dissipative, allowing them to neutralize static charges effectively while inhibiting significant current flow. The conductivity measurements demonstrate that the Fe3O4/N-CQD composites effectively reduce electrostatic buildup on surfaces. This characteristic makes them suitable for applications in packaging sensitive electronic components, antistatic coatings, and environments intended to be ESD-safe. The relatively low conductivity suggests that these composites pose minimal risk of short-circuiting in electronic assemblies, while permitting regulated charge dissipation. The observed frequency-dependent behavior, marked by reduced conductivity at lower frequencies due to electrode polarization (EP), supports the presence of a hopping conduction mechanism.70,71 The observed behavior corresponds with the disordered structure and nanoscale interfaces in the composite, which facilitate localized charge transport. 72 The capacity to modify σdc through compositional or structural alterations offers increased adaptability for tailoring these materials for specific electrostatic control applications.
Conclusion
In this study, a multifunctional Fe3O4/N–CQD hybrid nanofiller was successfully synthesized from sugarcane bagasse using a rapid microwave-assisted method. FTIR and XRD analyses confirmed the formation of Fe3O4 and nitrogen-doped carbon dots, while SEM revealed the nanoscale morphology of the hybrid filler. The Fe3O4/N–CQDs were incorporated into an NBR matrix at loadings ranging from 0 to 11 phr. Rheological results showed that the nanofiller significantly influenced the curing behavior of NBR by accelerating cure kinetics and increasing crosslink density, with optimal effects at 8 phr; higher loadings reduced benefits due to filler aggregation and restricted polymer chain mobility. FTIR analysis further confirmed successful incorporation of the filler into the NBR matrix.
Thermogravimetric analysis indicated enhanced thermal stability for Fe3O4/N–CQD-filled NBR, as the main degradation region shifted to higher temperatures, attributed to thermally stable Fe3O4 nanoparticles and strong interfacial interactions with N–CQDs. Mechanical testing demonstrated that moderate filler loading improved tensile strength and modulus, while excessive loading led to agglomeration and reduced stress transfer, as confirmed by SEM. Thermal aging studies showed improved tensile strength retention, maintained ductility, and increased hardness. Magnetic measurements revealed soft magnetic behavior, with saturation magnetization increasing from 29.05x 10−3 to 91.32 × 10−3 emu/g and coercivity below 120 G. Dielectric and conductivity analyses indicated frequency-dependent permittivity and hopping conduction governed by interfacial polarization.
Based on these results, Fe3O4/N–CQDs derived from agricultural waste can act as an effective multifunctional nanofiller, simultaneously enhancing curing behavior, mechanical performance, thermal stability, aging resistance, magnetic properties, and dielectric behavior of NBR.
Footnotes
Acknowledgements
The author appreciates the National Research Centre for supporting.
Author contributions
Hebat-Allah S. Tohamy: Investigation & Methodology, Software, Conceptualization, Validation, Formal Analysis, Writing – Original Draft, and Writing – Review & Editing. Heba Kandil: Methodology, Data Curation, Investigation, Formal Analysis, Validation, Writing – Original Draft and Writing – Review & Editing. Azza A. Ward: Data Curation, Validation, Investigation, Visualization, Formal Analysis, and Writing – Review & Editing. Doaa E. El Nashar: Supervision, Resources, Conceptualization, Investigation, Formal Analysis, Writing – Original Draft, and Writing – Review & Editing.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Data Availability Statement
All data is presented in the figures, graphics and tables in the context of the manuscript.
