Abstract
A lanthanide trichloride system, specifically EuCl3 and YbCl3, was doped into PVA polymer film using a solution casting technique. The final composite rare earth films were described to examine their physical and structural characteristics. XRD and FT-IR investigations demonstrated significant complexity in the composite films. Thermogravimetric analysis showed that the composite films exhibited a higher residual mass percentage at 500°C compared to pure PVA. According to the UV-Vis, it was observed that pure PVA’s absorption edge and energy gap value decreased with the incorporation of Eu3+ and Yb3+ ions. The AC conductivity and dielectric constant of the composite rare earth films were analyzed. The 5 wt% (EuCl3-YbCl3) doped PVA film displayed the highest dielectric constant, followed by the 5 wt% YbCl3 and the 5 wt% EuCl3 films. The same behaviour was observed for AC electrical conductivity, which follows Jonscher’s law. Impedance data concerning the electrical modulus formalism (M*) and impedance formalism (Z*) were analyzed. The relationship between the real and the imaginary components of Z* showed an inclined spike at low frequencies and a semicircular arc at high frequencies, which was fitted to equivalent circuit models. Antibacterial activity was assessed and significantly improved against Klebsiella pneumoniae, a gram-negative bacterium.
Keywords
Introduction
Extensive research has been conducted on polymer composites because of their wide range of electrochemical devices that may utilize them, including fuel cells, sensors, solid-state batteries, electrochromic display devices, and supercapacitors. One potential way to give these materials new characteristics is by impurity doping. Rare-earth ions and suitable metal salts are being investigated as dopants because of their unique optical, magnetic, and electrical characteristics, which can enhance the electrical characteristics of different polymers. The primary advantages of incorporating metal salts into the polymer backbone include improved electrical properties, separation of ion pairs, and an increased amorphous nature. The film’s formability and desirable mechanical, thermal, and electrochemical stability are essential for making polymer composites attractive materials. These properties can often be achieved by selecting an appropriate polymer.1–6 Doping materials with rare-earth elements can change their size, morphology, and crystallographic phase, resulting in adjustable optical responses. Furthermore, rare-earth doping may control and make these materials’ electrical and catalytic performance flexible and scalable. Even in trace levels, rare-earth elements may significantly improve the characteristics of the materials, making them a popular addition or dopant.7–10 One of the most essential components of technological growth is the creation of materials doped with rare-earth elements. Comparing the rare earth elements, Europium ions reduce from trivalent to divalent more quickly. 11 Applications such as optical fiber, LEDs, laser materials, and optical signal amplification are suitable for Europium rare earth complexes.12,13 Ytterbium-containing films are gaining increasing attention due to their potential in advanced semiconductor devices. 14 Ytterbium-doped optical fibers are essential for high-power laser applications. 15 Polyvinyl alcohol (PVA) is a synthetic polymer known for its ability to absorb water and its wide range of applications. The versatility of PVA arises from its excellent chemical and physical properties, primarily due to its hydroxyl groups. The hydrogen bonding among these hydroxyl groups significantly influences the characteristics of PVA. Additionally, depending on their reactivity with the host matrix, moving ion species can be included in polymer composites to improve their physical characteristics.16–20 The primary chain of PVA degraded after rare-earth element inclusion. 21 The deterioration of the main chain causes a change in the strength of the infrared absorption bands, which in turn impacts electrical and optical conductivities due to the intensive interaction between the host and dopant.22–24 Research on rare-earth-doped polymers is still in its early stages. By using PVA as a host matrix, we created superior elastic films exhibiting excellent optical transparency. The incorporation of rare-earth elements into polymers enables the development of advanced functional materials with tailored structurel, optical, and antimicrobial properties. These enhancements make such composites attractive for use in photonics, electronics, packaging, biomedicine, and smart materials industries. This paper explores a novel investigation into the effects of Europium (Eu3+) and Ytterbium (Yb3+) ions and their interaction on electrical, structural, thermal, and optical characteristics of PVA films. Additionally, we thoroughly examined the antibacterial activity of the studied rare earth composite films.
Experimental part
Chemicals
Polyvinyl alcohol (PVA) was sourced from Alpha Chemika in India, Europium (III) chloride anhydrous (EuCl3) was obtained from ACROS in New Jersey, USA, with a purity of 99.9%, and Ytterbium (III) chloride hexahydrate (YbCl3·6H2O) was purchased from ALDRICH, also with a purity of 99.9%. The experiment used distilled water as a solvent; none of the materials needed purification.
Composite films fabrication
Mixing 95 wt% polyvinyl alcohol (PVA) with 5 wt% of Europium chloride (EuCl3), Ytterbium chloride (YbCl3), and a combination of both in a 1:1 ratio produced a series of polymeric films. The solution casting method was employed to create these films. The composites were combined with 30 mL of ionized water, which was heated to 70°C and stirred for 3 hours using a magnetic stirrer. This process ensured a uniform, clear, and transparent solution. The final stage was to transfer the solution to Petri plates and let it dry at room temperature to ensure no solvent residue remained. The resulting films were transparent and homogeneous and placed in desiccators for further study. Figure 1 illustrates a detailed step-by-step process for making the composite PVA-rare earth trichloride film. The PVA and PVA/rare earth chloride films preparation steps.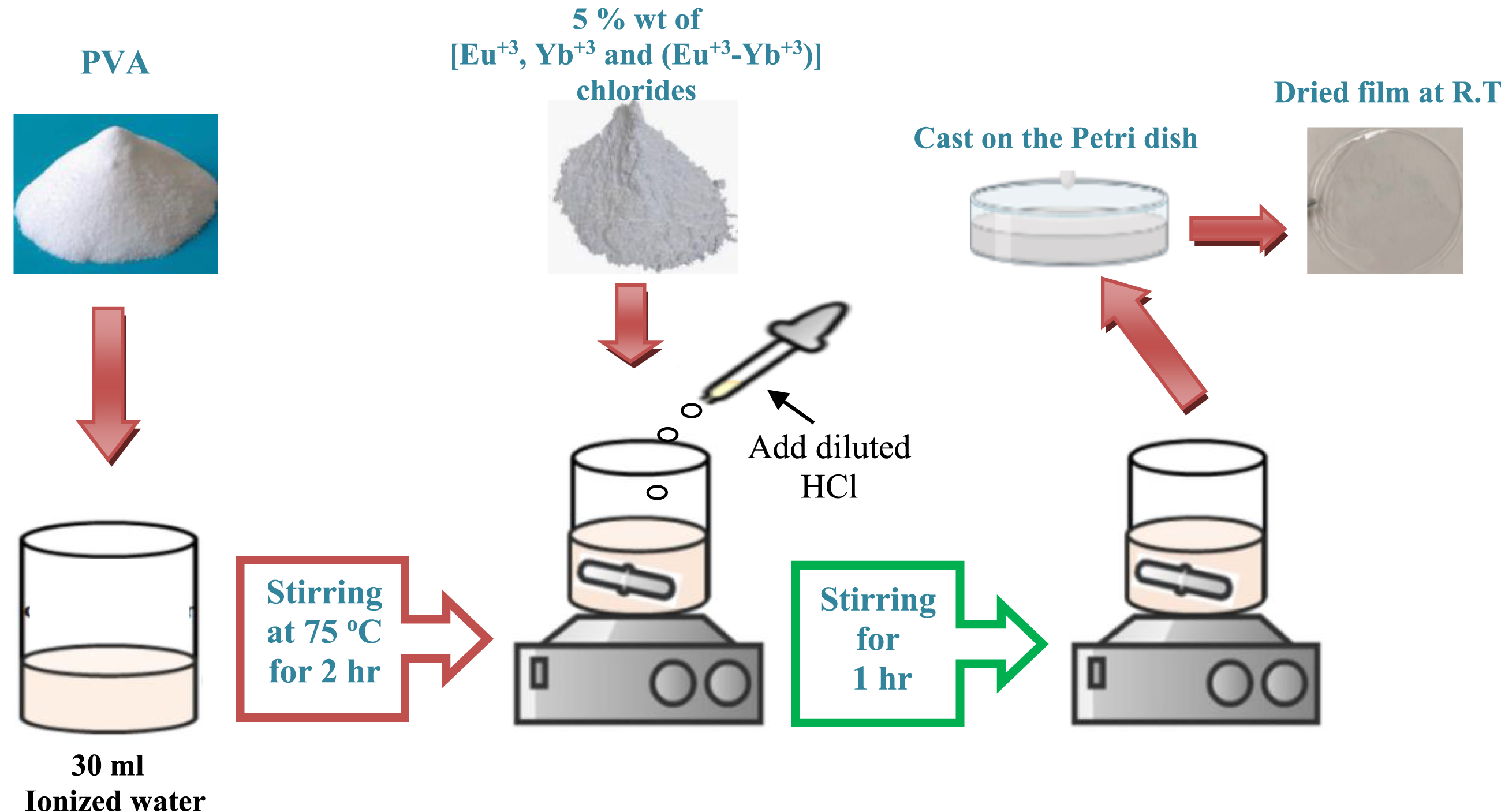
Measurements
The X'PERT-PRO-PANalytical apparatus, equipped with a Cu-Kα target (λ = 1.5406 Å), was used to gather X-ray diffraction (XRD) patterns. The 2θ range from 5° to 60° was used to capture the scans. Bruker Vertex 80 (Germany) operating in the 4000-400 cm−1 spectral band was utilized for Fourier Transform Infrared (FT-IR) measurements. Using a Shimadzu TGA-45H thermogravimetric analyzer, we investigated the thermal behavior of the produced composite films. The samples’ thermal stability was examined up to 500°C using a nitrogen flow and heating rates of 10°C min−1. A Shimadzu UV-630 UV-VIS-NIR spectrophotometer was used to record ultraviolet-visible (UV-Vis) absorption spectra within the 200 to 1000 nm wavelength range. A Novocontrol Concept 40 system was used to conduct Broadband Dielectric Spectroscopy (BDS) experiments. The antibacterial activity of the investigated samples was tested under identical conditions with a concentration of 15 mg/mL against both Gram-positive (Staphylococcus aureus, Bacillus subtilis) and Gram-negative (Escherichia coli, Klebsiella pneumoniae) bacteria.25,26 The inhibition zone was measured under visible light after 24 hours of incubation at 37°C. The agar medium was covered with the chopped samples.
Results and Discussion
Structural analysis
Figure 2 displays the X-ray diffraction (XRD) patterns of rare earth chlorides, PVA, and PVA/rare earth chloride composites. Table 1 presents the indexed (hkl) diffraction pattern for the anhydrous EuCl3 planes, which belong to the hexagonal crystal structure classified under the 6/m point group.
27
The XRD spectrum of YbCl3, as shown in Figure 2, has a monoclinic crystal structure with a 2/m point group, whereas Table 2 presents the indexed (hkl) diffraction pattern.
28
A broad peak at around 2θ = 19.87° in the XRD pattern of pure PVA, which corresponds to the (101) crystal plane, suggests that PVA is semicrystalline.29–31 One possible explanation for the semicrystalline structure of PVA is the high degree of hydrogen bonding between the hydroxyl groups along the polymer chain.
32
The interaction between the composites causes a drop in peak intensity when 5 wt% of EuCl3, YbCl3, and (EuCl3 -YbCl3) is added to PVA. The inclusion of EuCl3 is evidenced by a small broad peak at approximately 2θ values between 41.38° and 41.50°, which corresponds to pure EuCl3. The other characteristic diffraction peaks of EuCl3 and YbCl3 are obscured in the XRD patterns of the composite films because the rare earth elements are fully dissociated within the polymer matrix. XRD pattern of pure EuCl3, YbCl3, PVA, and PVA/rare earth chloride films. The indexed (hkl) powder diffraction pattern of EuCl3 anhydrous. The indexed (hkl) diffraction pattern of YbCl3.6H2O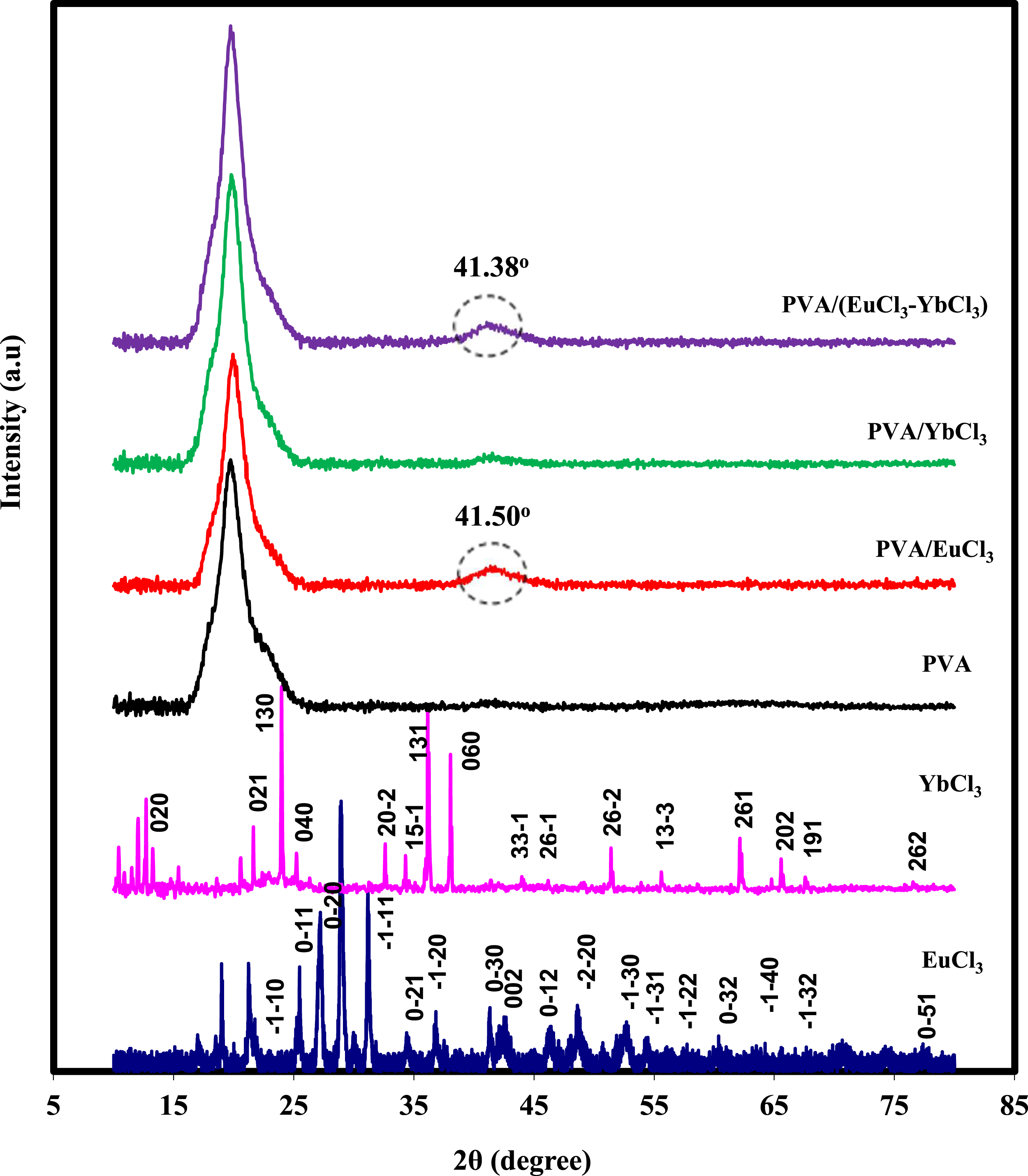
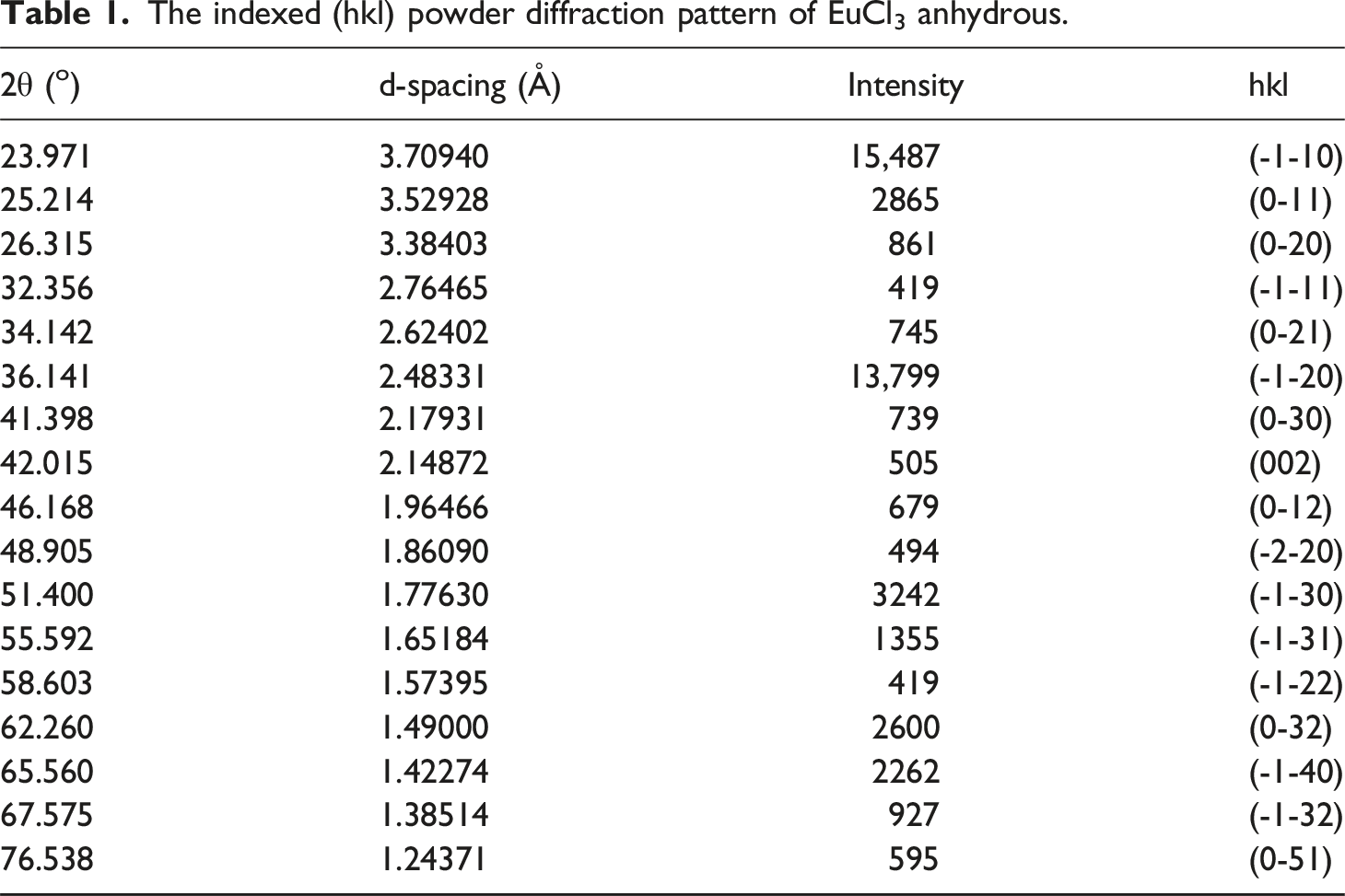
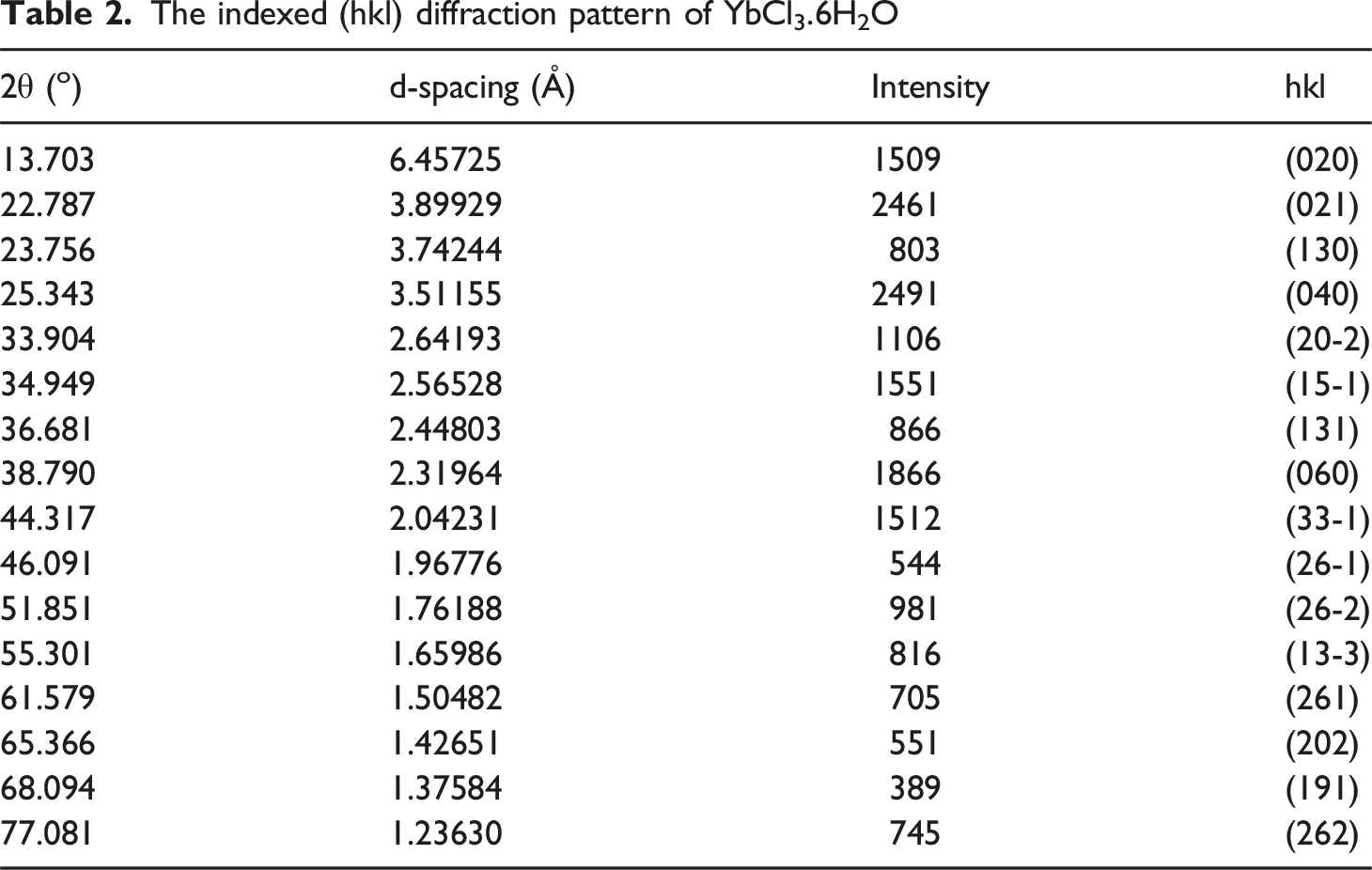
Figure 3 displays pure PVA and PVA/rare earth chloride samples’ FT-IR spectra. The FT-IR spectrum of pure PVA has a bandwidth at 3251 cm−1 corresponding to O-H stretching vibrations,33,34 whereas the -CH2 stretching band appears at 2912 cm−135–37 The C = O carbonyl stretching group corresponds to the prominent band at 1639 cm−1.38,39 The CH2 bending occurs at 1415 cm−1, while the peaks at wave numbers 1315 cm−1 and 1080 cm−1 correspond to the bending and stretching of C–H deformation and C–O stretching, respectively. C–C stretching is observed at 829 cm−1. These peaks collectively confirm the formation of pure PVA (polyvinyl alcohol). Due to the cross-linking behavior between the rare earth ions and PVA, introducing rare earth ions alters the peak intensities, particularly for the C–H, C–O, and O-H stretching bands. Also, the –CH2 stretching band (2850–2912 cm−1) weakens and becomes less distinct, reflecting reduced chain mobility. The 1350 cm−1 vibrational band peak nearly disappears after Eu3+ and Yb3+ incorporation, confirming substantial disruption of CH2-related vibrations due to reduced crystallinity and increased amorphous character in the PVA matrix. Around 850 cm−1, peaks associated with C–H and C–O/C–C skeletal vibrations show attenuation and slight shifts, attributed to altered bonding environments and heterogeneous structures formed by rare earth coordination; Nevertheless, despite these spectral modifications, the molecular structure of PVA remains unchanged. FT-IR spectra of pure PVA, and PVA/rare earth chloride films.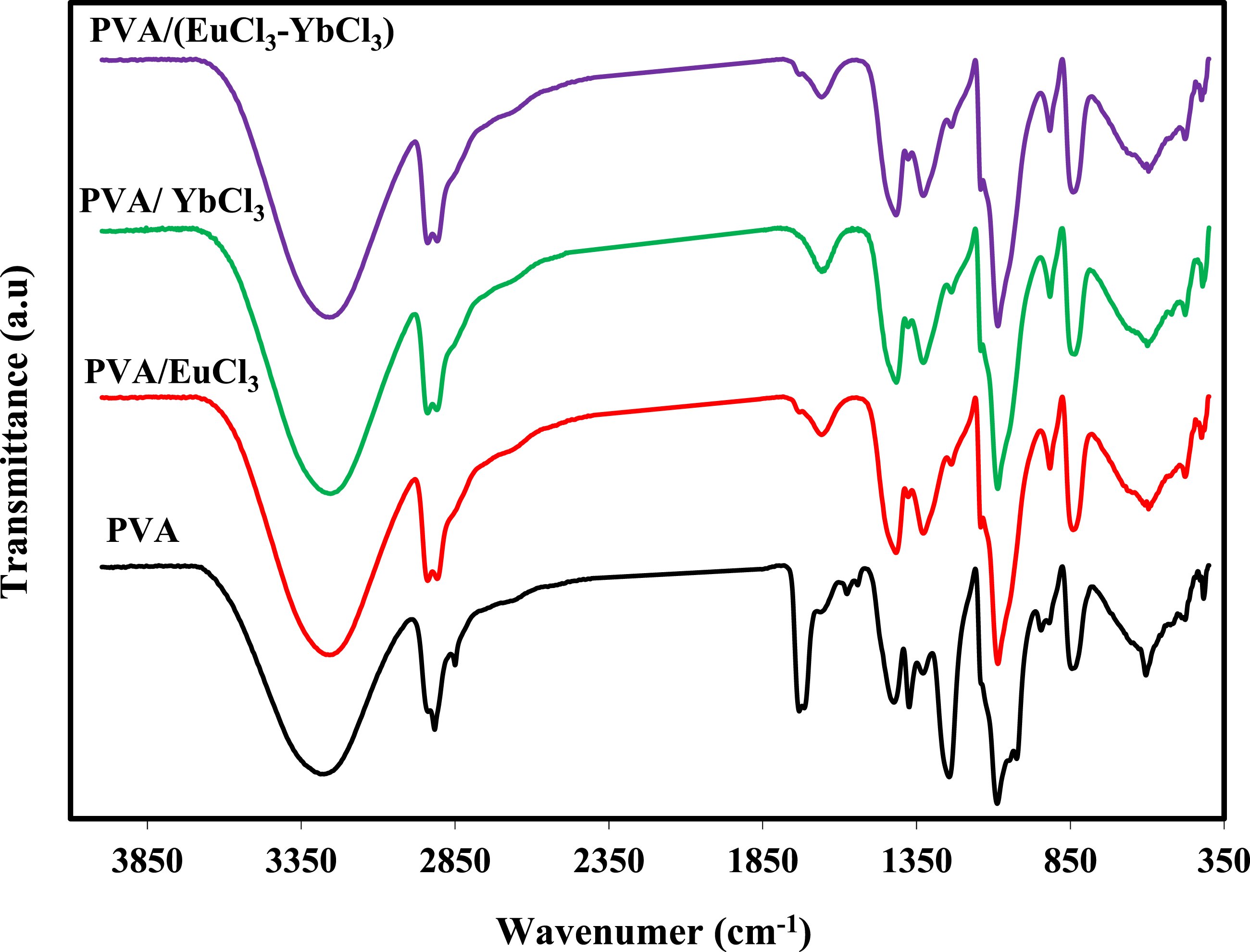
Thermal studies
The TG and DTG curves of pure PVA and PVA/rare-earth chloride composites are shown in Figure 4, with key thermal parameters summarized in Table 3. For pure PVA, an initial weight loss of approximately 6% occurs near 90°C, attributed to the evaporation of physically adsorbed moisture. Minimal mass change is observed between 150°C and 276°C, followed by a rapid two-step decomposition above 276°C, resulting in a total mass loss of around 87%. This major degradation is associated with chain scission in the PVA backbone, initially within the amorphous regions and progressing into the crystalline domains upon melting. A residual mass of approximately 13% remains after complete thermal decomposition. In contrast, the thermal behavior of PVA films doped with 5 wt% EuCl3, YbCl3, and (EuCl3–YbCl3) exhibits significant deviations. The onset of degradation occurs earlier in all doped samples, indicating a reduction in thermal stability in the presence of rare-earth chlorides up to ∼380°C. Notably, the PVA/YbCl3 composite retains the highest residue at 500°C, suggesting improved carbonization or possible formation of thermally stable inorganic species. Coordinated water release and salt–polymer interactions likely contribute to the complexity of thermal events observed in the DTG profiles, which show overlapping endothermic and exothermic transitions between 100 and 350°C. While assigning specific degradation stages can be challenging due to the overlapping nature of these processes, the TGA data clearly demonstrate the modifying influence of rare-earth dopants on the thermal decomposition pathways of PVA.
40
(a) TG and (b) DrTGA thermograms for PVA and PVA/rare earth chlorides films. Thermogravimetric data of the PVA and PVA/rare earth composites and residual weight percentage at 500°C. Tdmax: decomposition temperature at max. peak.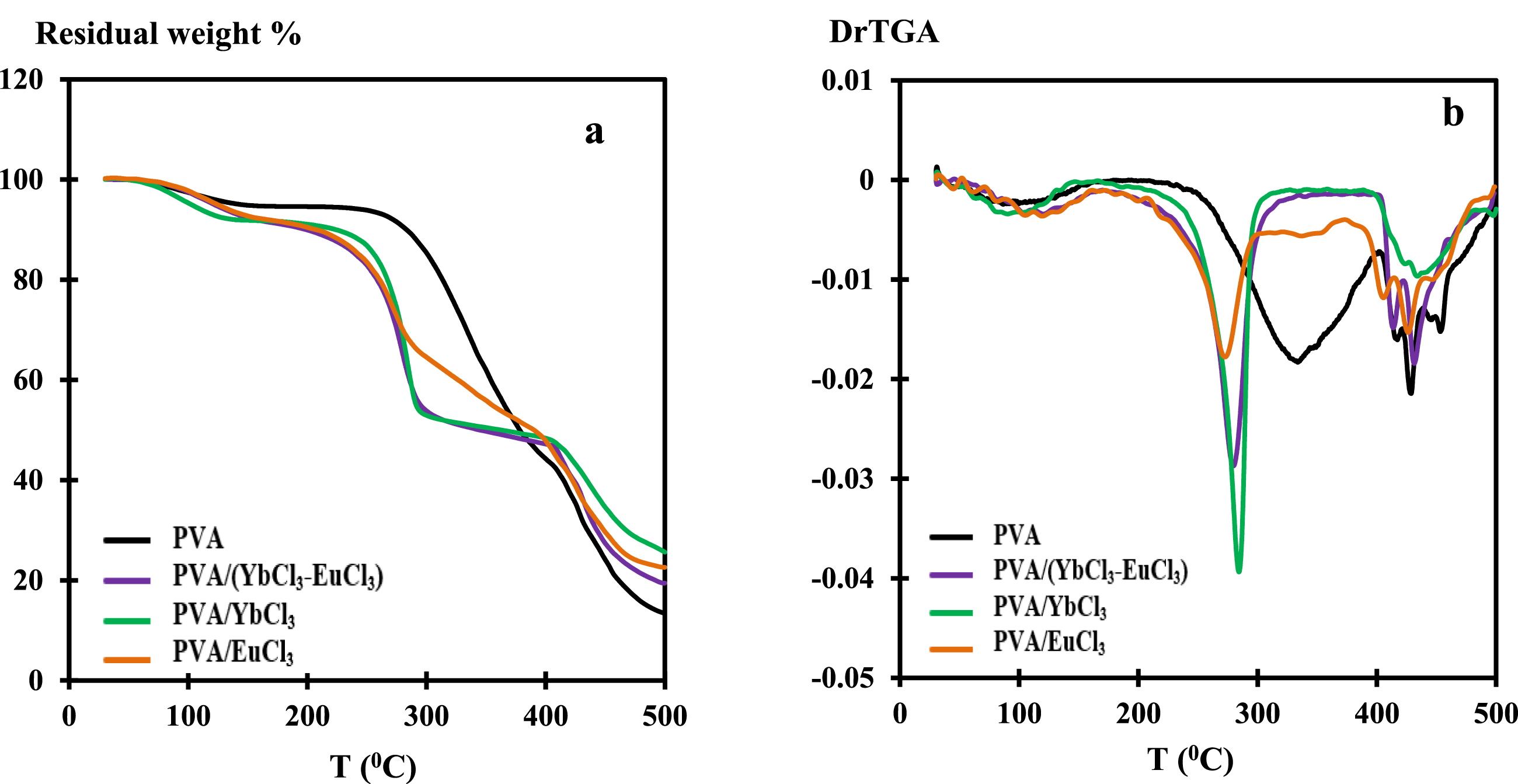
Optical analysis
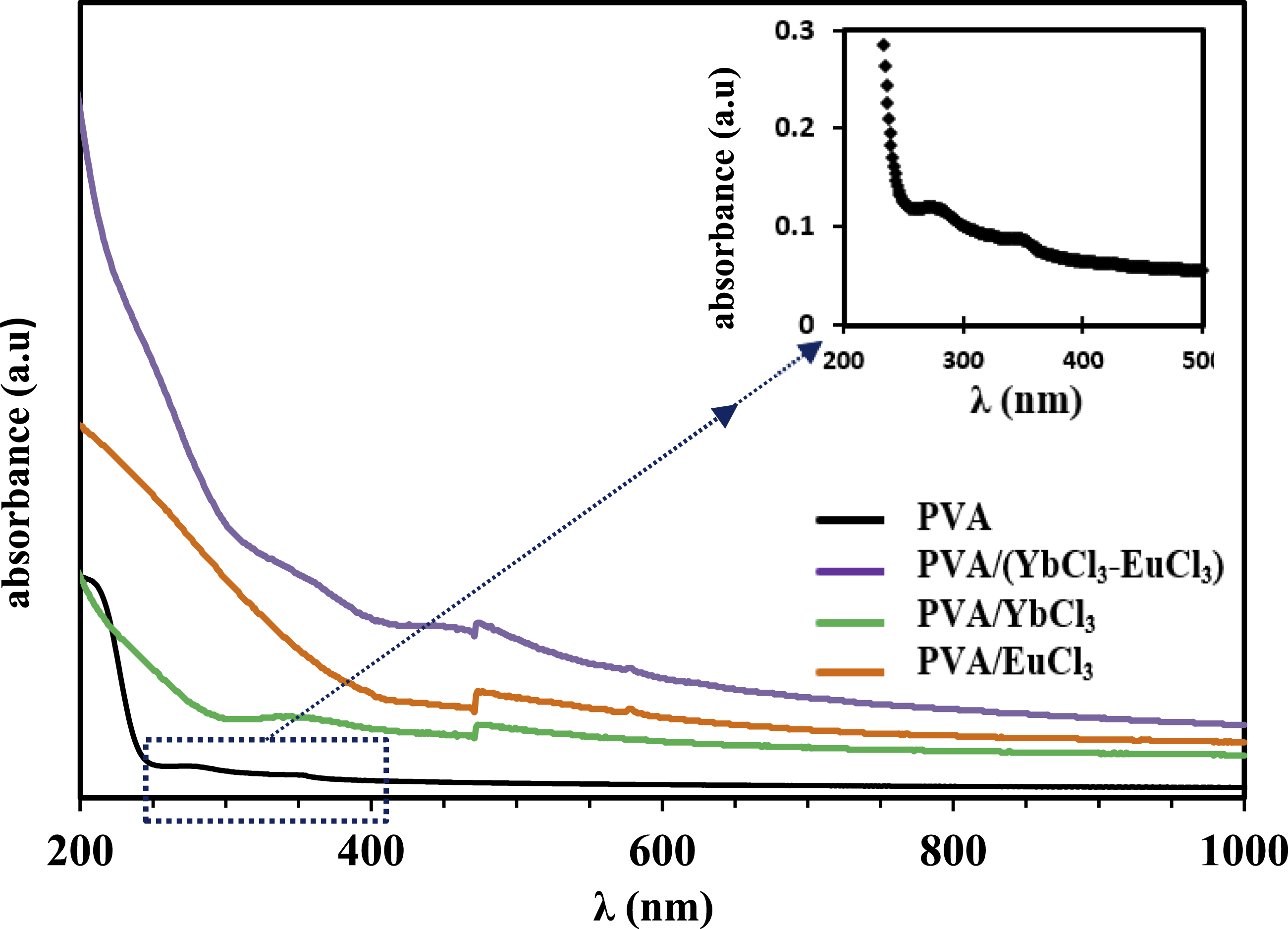
The behavior of the samples under UV light irradiation was monitored using UV spectroscopy within the range of 200 to 1000 nm. Figure 5 illustrates the optical absorbance of pure PVA and PVA doped with EuCl3, YbCl3, and (EuCl3-YbCl3) composite samples. The data confirms that all films are transparent throughout the visible region, with absorption occurring in the UV region. The spectrum of pure PVA shows shoulder-like bands, which correspond to π→π* at 281 nm and n→π*at 345 nm transition.41–43 These bands are associated with the PVA matrix containing unsaturated bonds of the C = O and C = C types. The absorption bands observed in PVA at 281 nm disappear in the spectra of PVA samples doped with rare earth chlorides. The absence of this band suggests that the rare earth ions interact strongly with the PVA matrix in films doped with 5 wt% YbCl3 and a combination of EuCl3 and YbCl3. The doped samples also show a change in the absorbance band from 345 nm to 351 nm. Possible causes for this change in the absorption band include variations in the energy band gap, changes in crystallinity within the polymer matrix, or the creation of charge transfer complexes between the oxygen donor atoms in the OH groups of the PVA main chain and the rare earth ions. As a result, the additive filler appears to affect the hydrogen bonds in the material. Figure 6 illustrates the relation between the optical absorption coefficient, α, defined as the relative decrease in light intensity, and the photon energy hν (in eV) for all investigated samples. The absorption coefficient is calculated using Beer-Lambert’s formula.
44
Absorption spectra of the PVA and PVA/rare earth composite films. Absorption coefficient of the PVA and PVA/rare earth composite films.
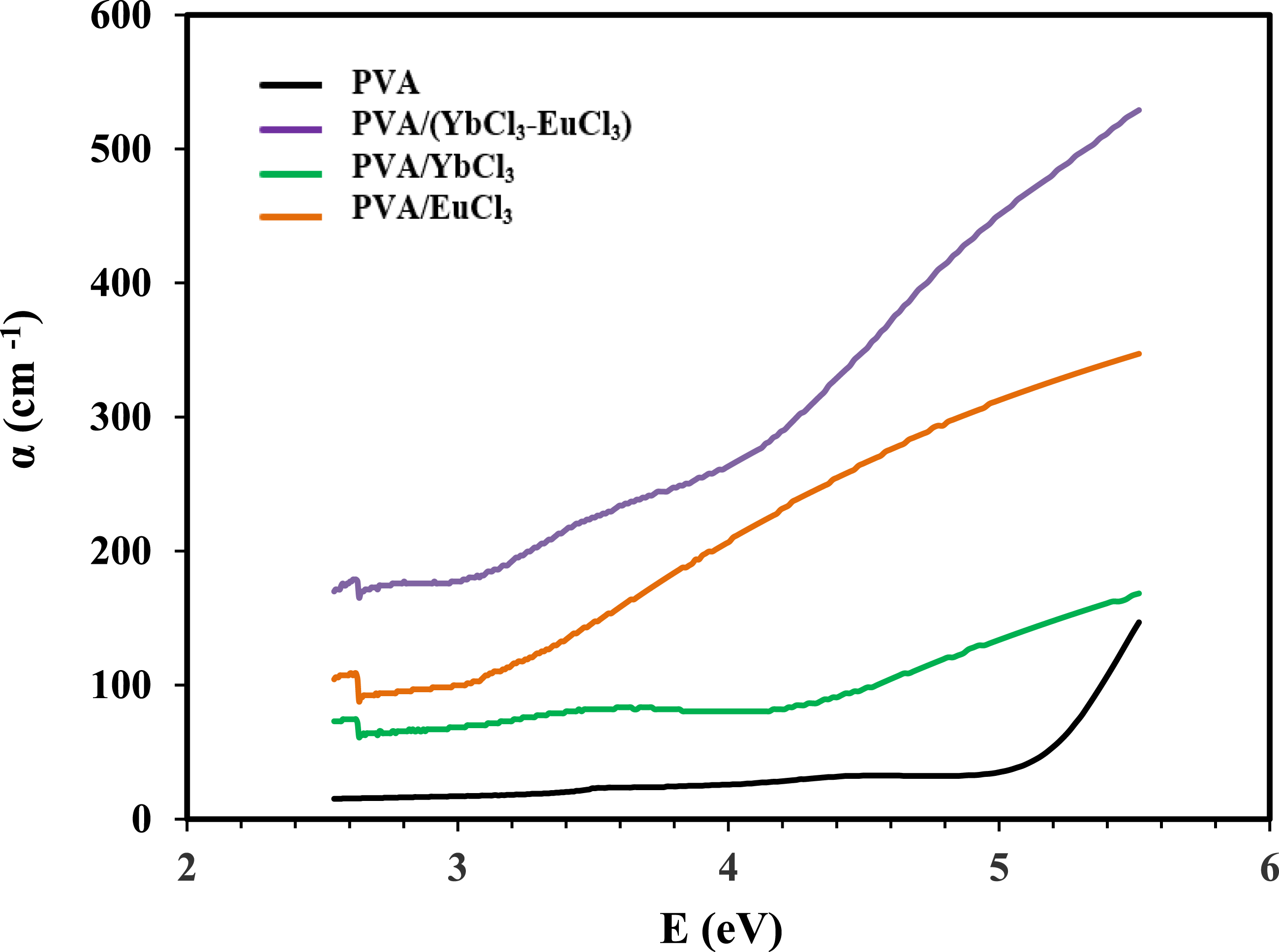
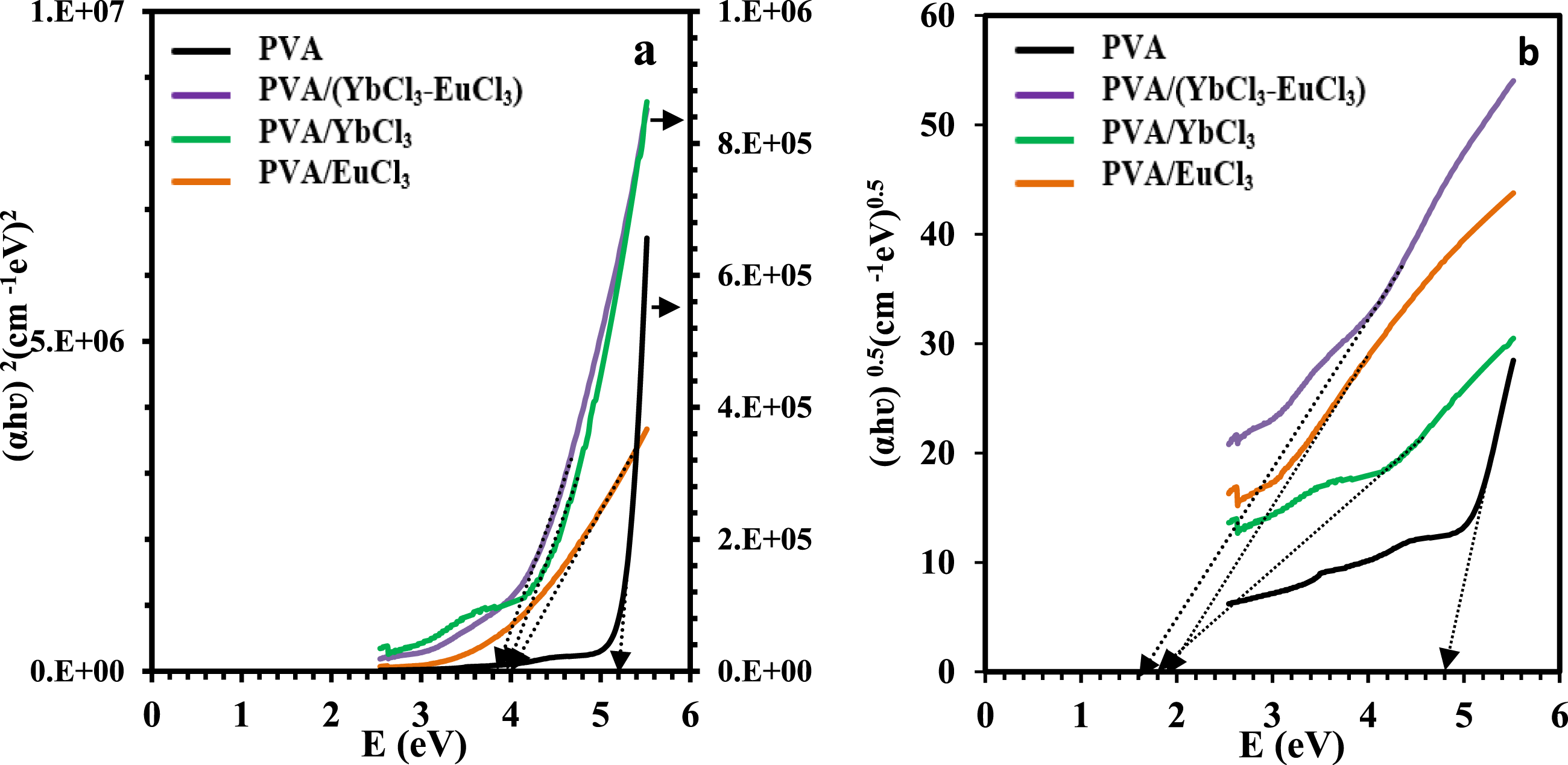
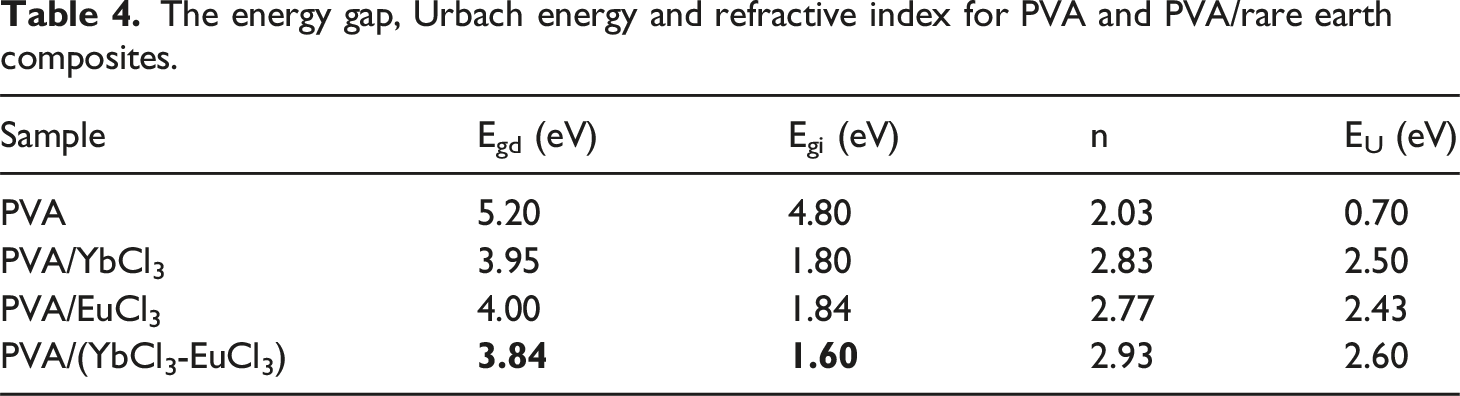
In the equation, A represents the absorbance, and d denotes the thickness of the film. It has been observed that the absorption coefficient of PVA increases when lanthanide chlorides are incorporated. This increase can be attributed to the attractive forces between the rare earth ions and the PVA chain’s hydroxyl (OH) groups. It is possible to determine the electron transition nature of all investigated films using the α values, where the band gap (Eg) of the films is related to α by Tauc’s formula45,46 (a) (αhʋ)2and (b) (αhʋ)1/2 versus photon energy of the PVA and PVA/rare earth composite films. The energy gap, Urbach energy and refractive index for PVA and PVA/rare earth composites.
The refractive index (n) is crucial in fabricating optical devices, coatings, and anti-reflection applications. Several empirical relationships, including those proposed by Dimitrov et al.,47,48 can help determine the high-frequency refractive index based on the material’s band gap.49,50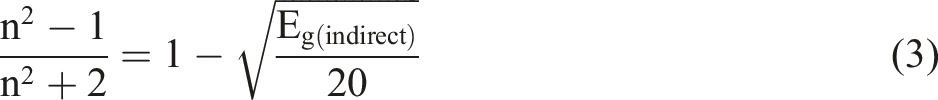
Table 4 presents the refractive index values of PVA and PVA/rare earth composites. Composite samples have more significant refractive indices than pure PVA. The increasing values of the refractive index observed in the rare-earth-doped PVA films suggests enhanced optical reflectivity, which can be attributed to improved structural packing and an increased density of charge carriers arising from the coordination of Eu3+ and Yb3+ ions within the polymer matrix. The incorporation of these rare-earth ions induces significant modifications in the electronic structure of the host polymer, as evidenced by the concurrent reduction in optical band gap and elevation in refractive index. This inverse relationship is indicative of increased electronic polarizability and stronger light–matter interactions, both of which contribute to the enhanced optical response of the composite films. Such modulation of refractive index is particularly advantageous for integrated photonic applications, including polymer-based optical waveguides, wherein effective light confinement requires a core material with a refractive index sufficiently higher than that of the surrounding cladding to support total internal reflection.
To gain insight into the degree of structural disorder and the presence of defect states in the polymer composites, the Urbach energy (EU) was calculated. This was done using the Urbach empirical relation
20

EU represents the width of the exponential absorption edge in the UV–Vis spectrum, which arises due to localized states in the bandgap. The values of EU were determined from the reciprocals of the slopes of the linear regions in the plots shown in Figure 8, and the corresponding results are summarized in Table 4 pure PVA exhibited a relatively low Urbach energy of 0.7 eV, indicating a more ordered structure with fewer electronic defects. Upon doping with rare-earth ions (Yb3+, Eu3+, and their combination), the EU values increased significantly, reaching up to 2.60 eV in the PVA/(YbCl3–EuCl3) composite. This substantial increase suggests that the introduction of rare-earth dopants induces greater structural disorder and creates additional localized tail states. These tail states facilitate sub-bandgap optical transitions, contributing to the narrowing of the optical band gap and enhancing light absorption in the visible region. The broadening of the Urbach tail also correlates with increased polarizability and refractive index, indicating stronger light–matter interaction due to rare-earth incorporation. Overall, the Urbach tail analysis confirms that rare-earth doping alters the electronic structure of PVA, enhancing its optical activity through increased disorder and density of localized states. The relation between ln(α) and photon energy (hʋ) of the PVA and PVA/rare earth composite films.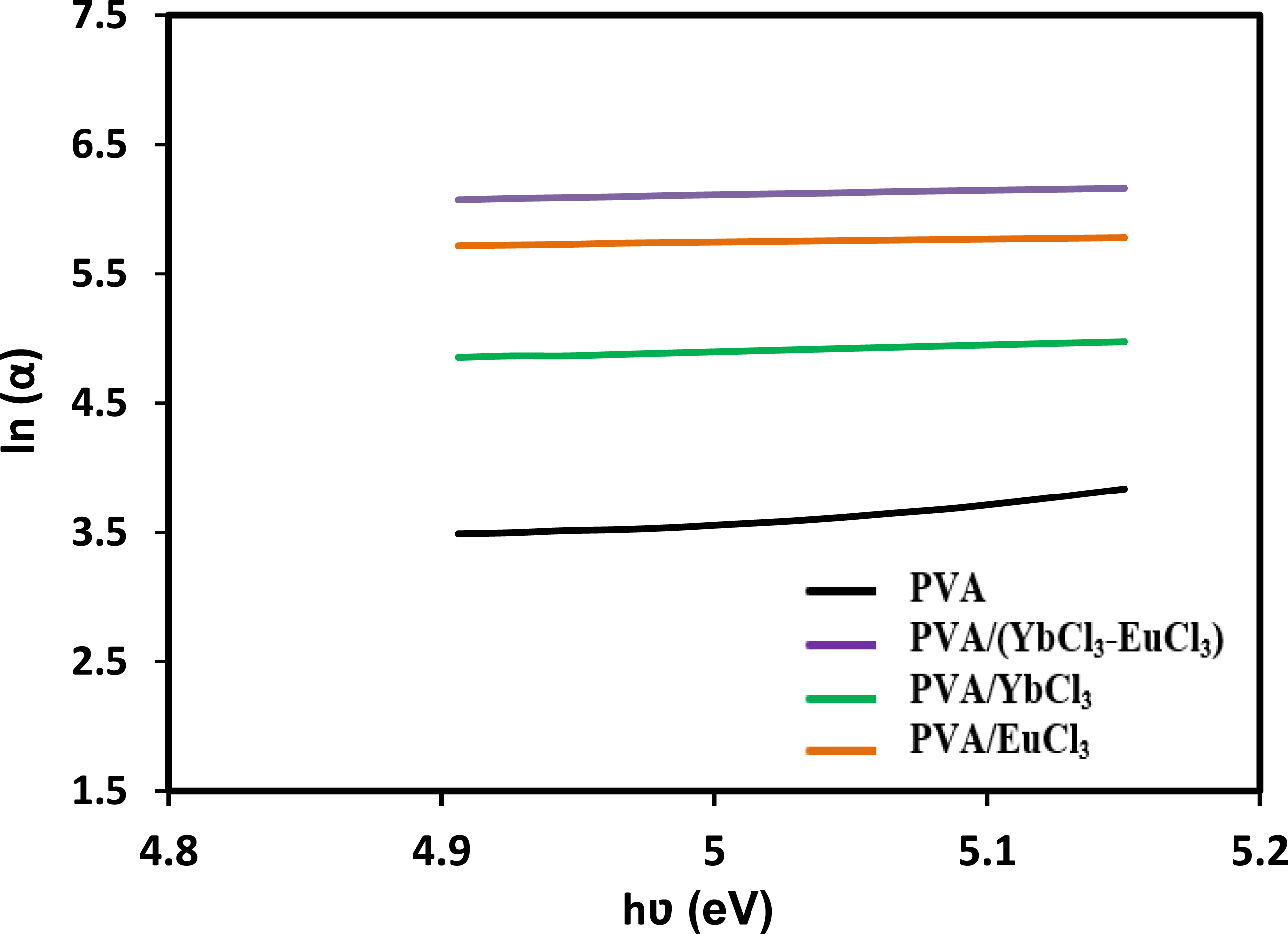
Electrical analysis
Dielectric constant
Figure 9 illustrates the variation of the real part (ε′) and the imaginary part (ε′′) of the dielectric constant as a function of frequency for pure PVA, and PVA doped with 5 wt% of EuCl3, YbCl3, and a composite of EuCl3-YbCl3 at room temperature. It is evident from Figure 9(a) that ε′ drops down with increasing frequency. This behavior indicates that polar materials start with a high initial value of ε51 and the value of ε′ begins to fall as the AC field frequency increases. Electrical relaxation mechanisms are responsible for this change. A rapid rise in the dielectric constant with frequency in the low-frequency domain indicates space charge polarization. The relative importance of space charge and total polarization diminishes with increasing frequency. The dielectric constant values increase in the following order: pure PVA has the lowest dielectric constant, followed by 5 wt% EuCl3, YbCl3, and finally, the (EuCl3-YbCl3) combination, which exhibits the highest dielectric constant. From low frequency up to 10 kHz, the dielectric constant varies between Eu3+ and Yb3+ as the frequency increases. Creating a continuous network of ions is responsible for the rise in the dielectric constant of produced films.
52
The increasing dielectric constant across the EuCl3, YbCl3, and mixed-chloride films can be explained by the effective polarizability of the incorporated rare-earth ions and their chloride complexes. Although detailed polarizability data for EuCl3 and YbCl3 in polymer matrices are not available, literature on rare-earth ions in other hosts (e.g., glass) indicates strong electronic polarizability of rare earth species.
53
Spectroscopic studies likewise show that coordination and ion pairing in chloride complexes (e.g., EuCl3) are significant, which supports the idea of a strongly polarizable network.
54
Despite the slightly larger ionic radius of Eu3+, Yb3+ exhibits higher effective polarizability within the polymer environment due to its greater charge density, stronger coordination with PVA, and larger distortion of the surrounding electron cloud. These features increase Maxwell–Wagner polarization at low frequencies, resulting in ε′(PVA/YbCl3) exceeding ε′(PVA/EuCl3). In the mixed EuCl3–YbCl3 system, ions with different polarizabilities and coordination strengths coexist, promoting the formation of a more continuous ionic network and further amplifying space-charge polarization, which explains the highest dielectric constant observed among all samples. The trend of ε′′ with frequency variation is comparable to ε′ with frequency, as shown in Figure 9(b). However, 5 wt% (EuCl3-YbCl3) exhibits the highest ε′′across the entire frequency range. Frequency dependence of (a) real part (ε′) and (b) imaginary part (ε′′) of dielectric constant for PVA and PVA/rare earth composite films at room temperature.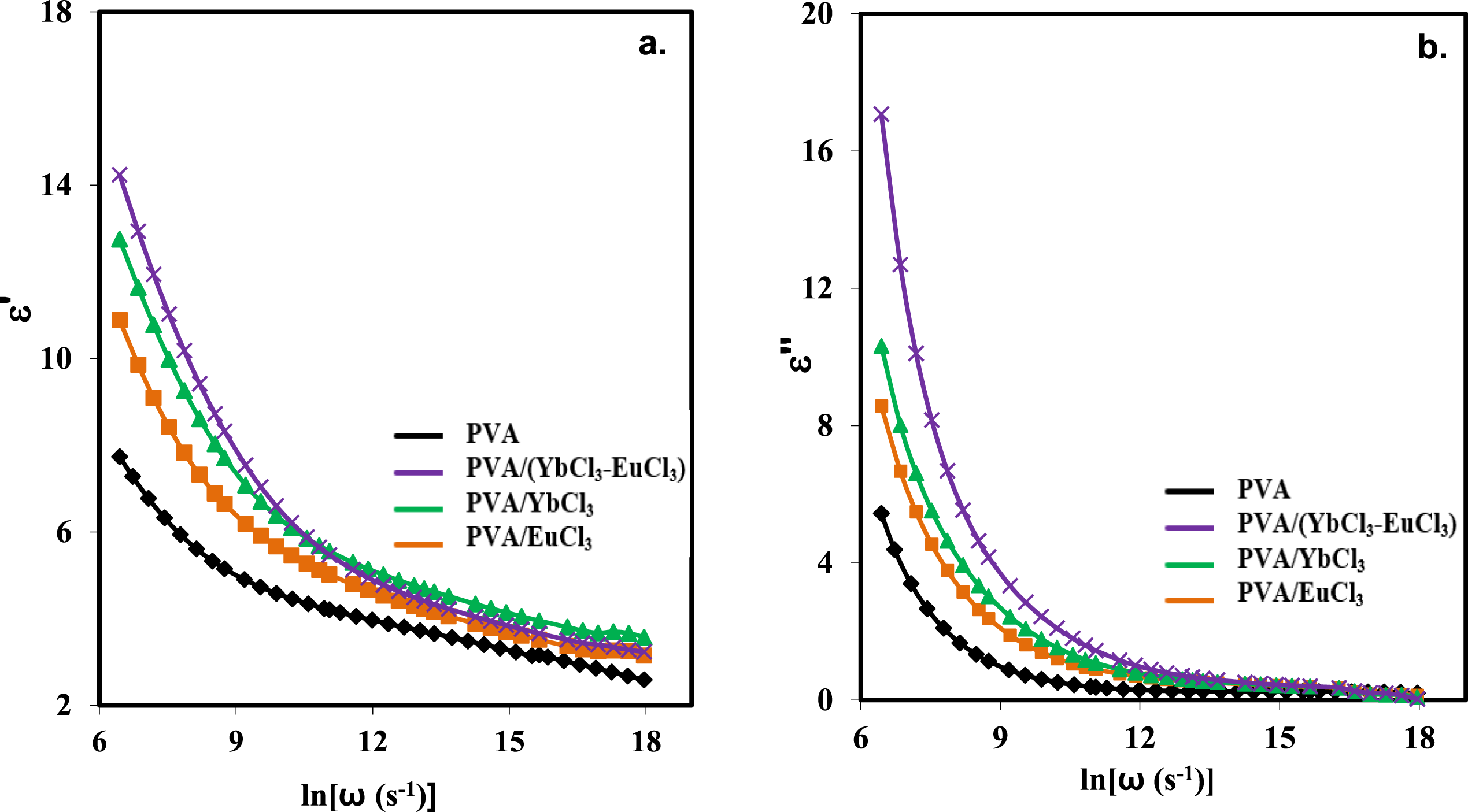
Electrical conductivity
Figure 10 shows the logarithmic plot of the electrical conductivity (σ) versus angular frequency (ω) at room temperature for pure PVA, and PVA doped with 5 wt% of EuCl3, YbCl3, and (EuCl3-YbCl3) composites. Figure 10 shows that conductivity grows linearly with frequency in the dispersion region, which is the high-frequency range. This indicates that charge carriers are more mobile at higher frequencies.
55
Generally, universal dispersion relaxation (UDR) models frequency-dependent conductivity.
56
Dependence of ln (σac) on frequency for PVA and PVA/rare earth composites at room temperature.
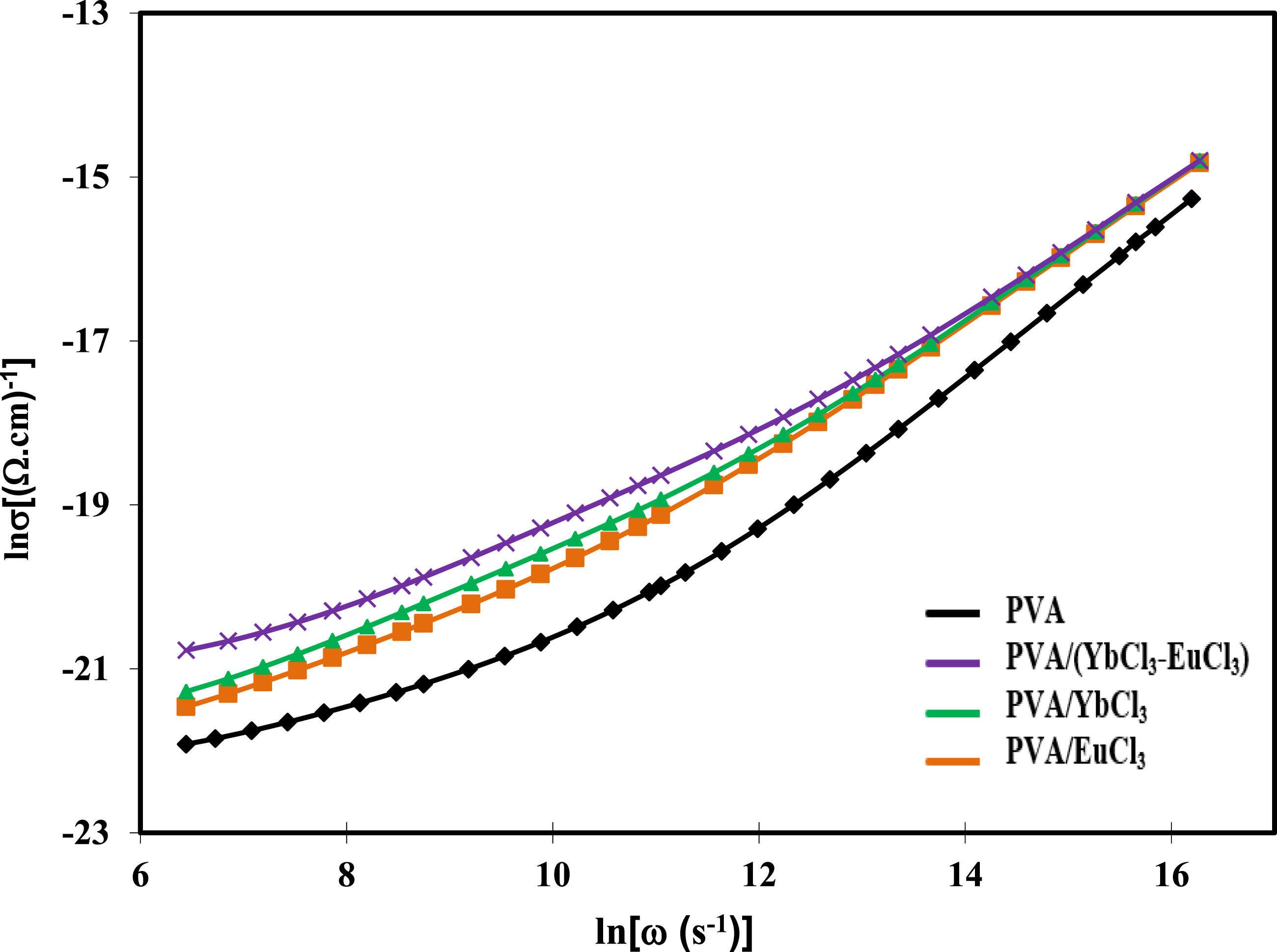
The dc conductivity of the sample is represented by σdc; the angular frequency is denoted by ω the temperature-dependent constant AT, which determines the intensity of polarizability, and the power law exponent, s, is dependent on both temperature and frequency. An exponent s represents the strength of the interaction between mobile ions and their environment. The Jonscher region, which includes several hopping models and experiments, yielded s values between 0 and 1. The transport mechanism is a thermally stimulated hopping process between two locations separated by an energy barrier.57,58 By comparing composite samples to a resistor-capacitor (RC) equivalent circuit, where capacitance occurs between conductive filler clusters, Zhang et al. 59 were able to explain the frequency dependence of σac in terms of polarization effects. They found that the conductivity of these composites remains constant up to a specific frequency. Below this frequency, the AC conductivity-frequency curve shows a plateau because the capacitor contribution becomes dominating beyond this threshold. The conductivity of composites is improved as the frequency rises because more current flows through each capacitor. According to Jonscher’s power law, this pattern suggests that a hopping-type process governs the electrical conduction of materials. For every composite film analyzed, the value of σac grows as the frequency increases. The strengthening of the weak connections between the polymer and rare earth chloride has also been linked to the strong coupling seen across the grain boundaries. 60 The change in hopping behavior from long-range to short-range distances is responsible for increased conductivity with higher frequency. The sample with the highest conductivity is the one with 5 wt% (EuCl3-YbCl3), followed by the one with 5 wt% YbCl3. Pure PVA polymer, on the other hand, had the lowest conductivity. This is because amorphous sections are more common than crystalline ones, making it easier for induced ions to flow over short distances, resulting in higher ac conductivity (σac) values.
Conductivity Relaxation
The complex electric modulus M* was employed to investigate the conductivity relaxation phenomenon. Eliminating the electrode effect is a formalist benefit.
61
One way to express the complex dielectric modulus is as the inverse of the complex dielectric permittivity.62–65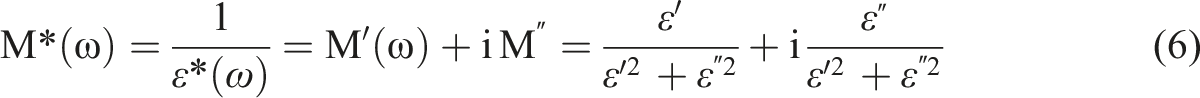
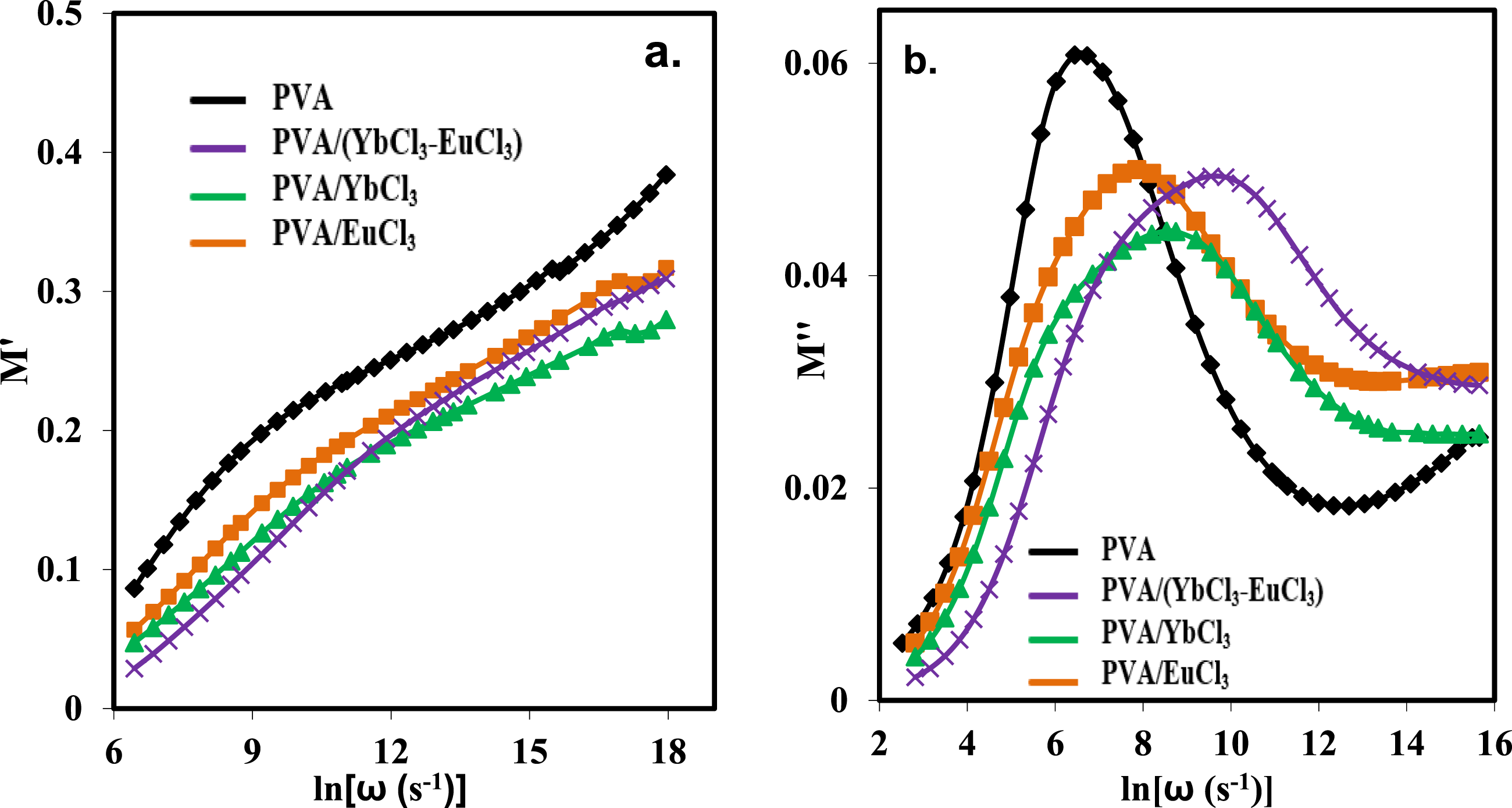
The dielectric modulus has real and imaginary components, denoted as M′ and M'', respectively. Figure 11 shows that M′ and M″ vary with frequency at room temperature for all samples examined. The electrode polarization effect is so strong at low frequencies that M′ is almost zero. According to Figure 11(a), the M′ achieves its maximum saturation value at very high frequencies. Because dipoles can’t reorient themselves fast enough to keep up with the rapid reversal of the electric field at these high frequencies, the value of M′ becomes less relevant.
66
Ionic conductivity is seen by the S-shaped curve.
67
All of the samples examined exhibit frequency-dependent variations in the imaginary component of the dielectric modulus, M″, as seen in Figure 11(b). A bell-shaped peak, indicative of a relaxation transition, is present in all composite films. Where charge carriers are active due to long-range hopping in the frequency band below the peak maximum. Conversely, carriers are limited to moving short distances due to probable good confinement at frequencies higher than the peak maximum.68–71 The presence of clearly defined broad peaks with noticeable asymmetry indicates the type of relaxation processes seen in Figure 11(b). Ionic conductors are the most probable candidates for these samples since a relaxation peak appears in the M″ curves.
72
When comparing the doped films to the pure PVA, the maximum value of the peak drops suggests that the fillers play a role in the relaxing process. The curve is asymmetric because of the distribution of relaxation times and a departure from ideal Debye-type behavior, and the M″ peaks are broad.
73
You may see the relaxation region either above or below the maximum M″. The low-frequency region to the left of the peak is linked to the DC conductivity that arises from a repetitive hopping process. Charge carriers can travel great distances in this process. However, the relaxation polarization process is associated with the high-frequency region to the right of the peak. This area represents the frequency range where ions may only move small distances inside their potential wells, as seen by the region.
74
The reversible movement of ions across a small region causes the AC conductivity peak on the right side. With an increase in frequency, this peak frequency signifies a change from long-range to short-range mobility. Adding Eu3+ and Yb3+ ions creates new interfacial areas, which shortens the relaxation period as the typical relaxation peak moves to the high-frequency side. The hopping type of conduction mechanism is confirmed by the enhanced shift in the 5 wt% (EuCl3-YbCl3) sample, which suggests that the generated charge carriers can pass via these new interfacial areas. To further verify the nature of the relaxation process in the pure and doped films, the stretch exponent parameter (β) was evaluated. The β values were calculated using the following relation.75,76 The frequency dependence of (a) real part (M′) and (b) imaginary part (M′′) of dielectric modulus for PVA and PVA/rare earth composites at room temperature.
Complex impedance analysis
Several formalisms, including complex electric modulus (M*) and complex impedance (Z*), have been investigated to interpret the dielectric spectra.
56
When the capacitive and/or resistive analysis is dominated by localized relaxation, the M* formula is valid; however, if long-range conduction is dominant, the Z* formula is more practical,
16
we can calculate the complex impedance: The Nyquist plots for the impedance spectra of (a) pure PVA, (b) PVA/EuCl3, (c) PVA/YbCl3, and (d) PVA/EuCl3-YbCl3 composites at room temperature.
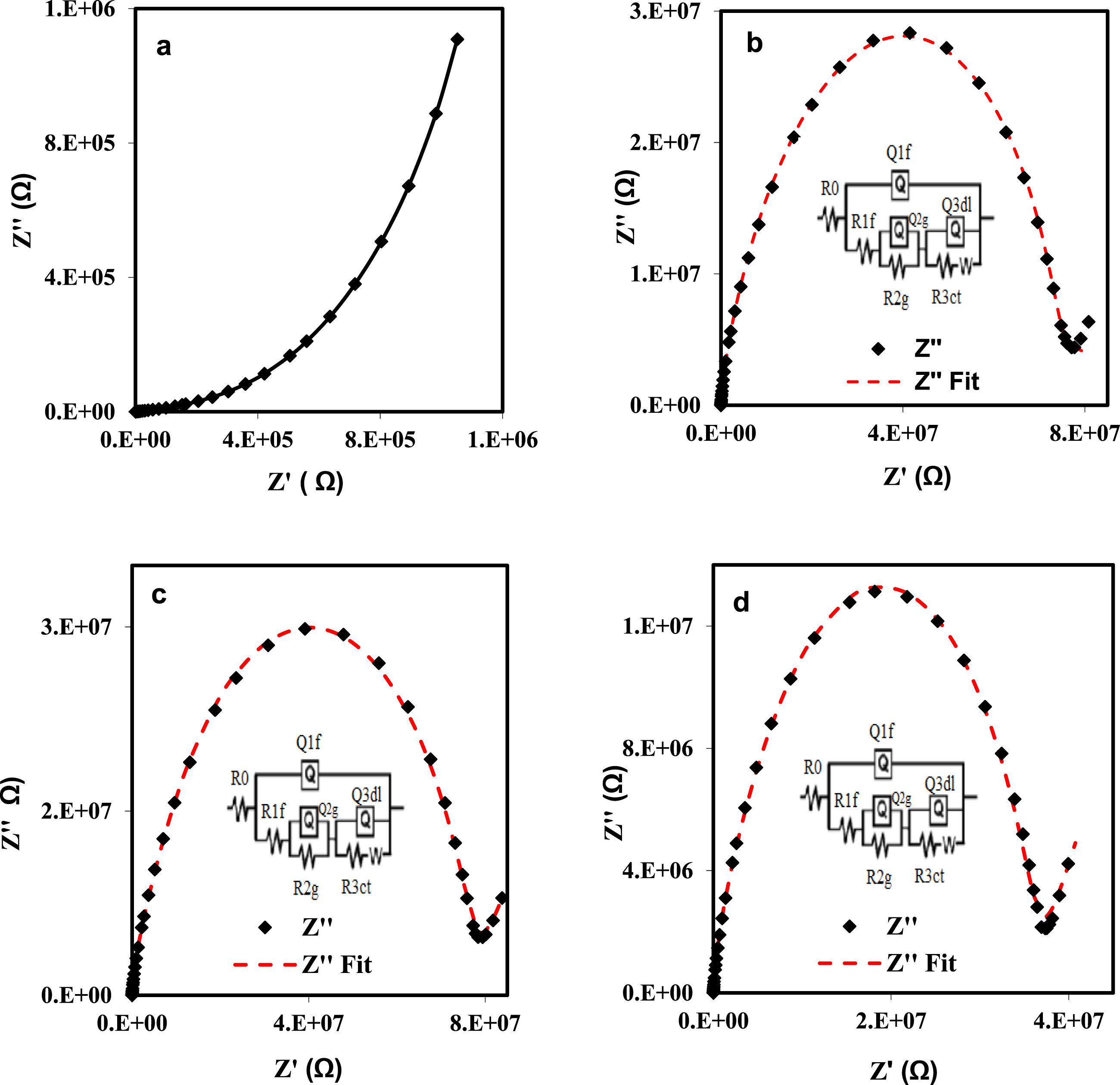

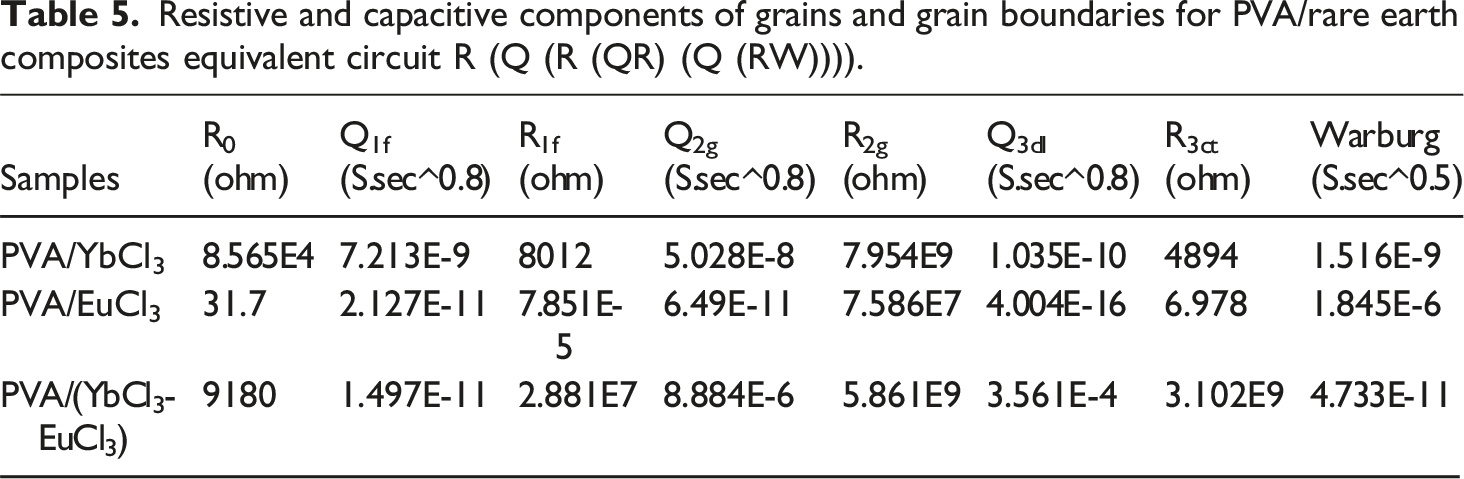
Resistive and capacitive components of grains and grain boundaries for PVA/rare earth composites equivalent circuit R (Q (R (QR) (Q (RW)))).
In contrast to the Nyquist plot of impedance, the Cole-Cole plot (M″ vs M′) better differentiates relaxation effects caused by grains (conductive areas) and grain boundaries (resistive regions) in materials.
85
As shown in Figure 13, the complex modulus graphs for the pure PVA and the PVA samples doped with 5 wt% of EuCl3, YbCl3, (EuCl3 -YbCl3) are shown at room temperature. The electrode polarization, grain, interface, and grain boundary all impact the one semicircle. The widening of the peak indicates a dispersion in the relaxation, characterized by a constant relaxation time distribution. The dielectric relaxation process is also clearly demonstrated by the peaks in M′ and M″.86–88 A departure from Debye relaxation and the short-range movement of charge carriers is shown by the non-coincidence of the peaks corresponding to M″ and Z″. The cole-cole diagram of pure PVA and PVA/rare earth composites at room temperature.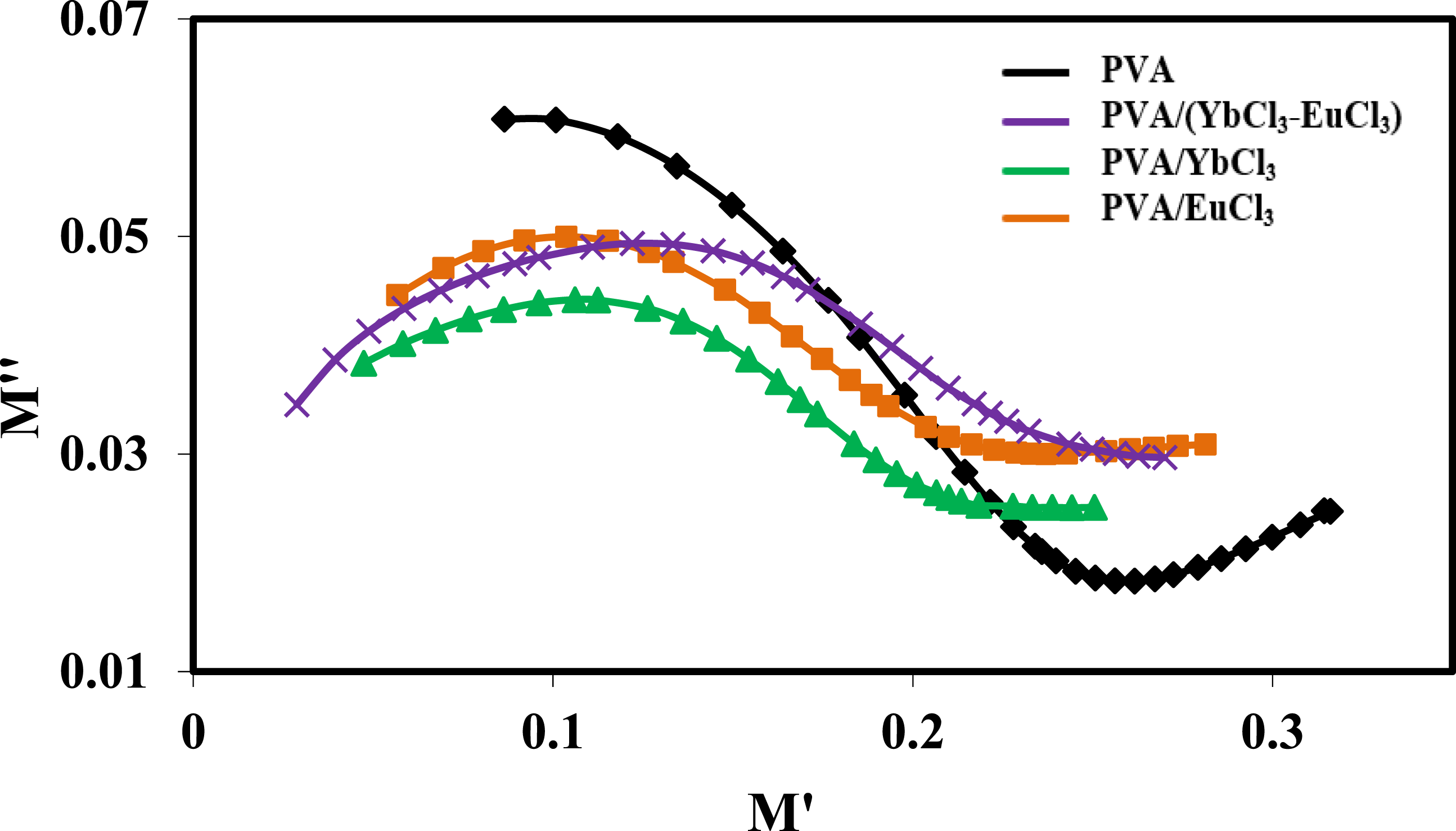
Antibacterial activity
Zone of Inhibition (mm) for PVA/rare earth composites against various bacterial strains.
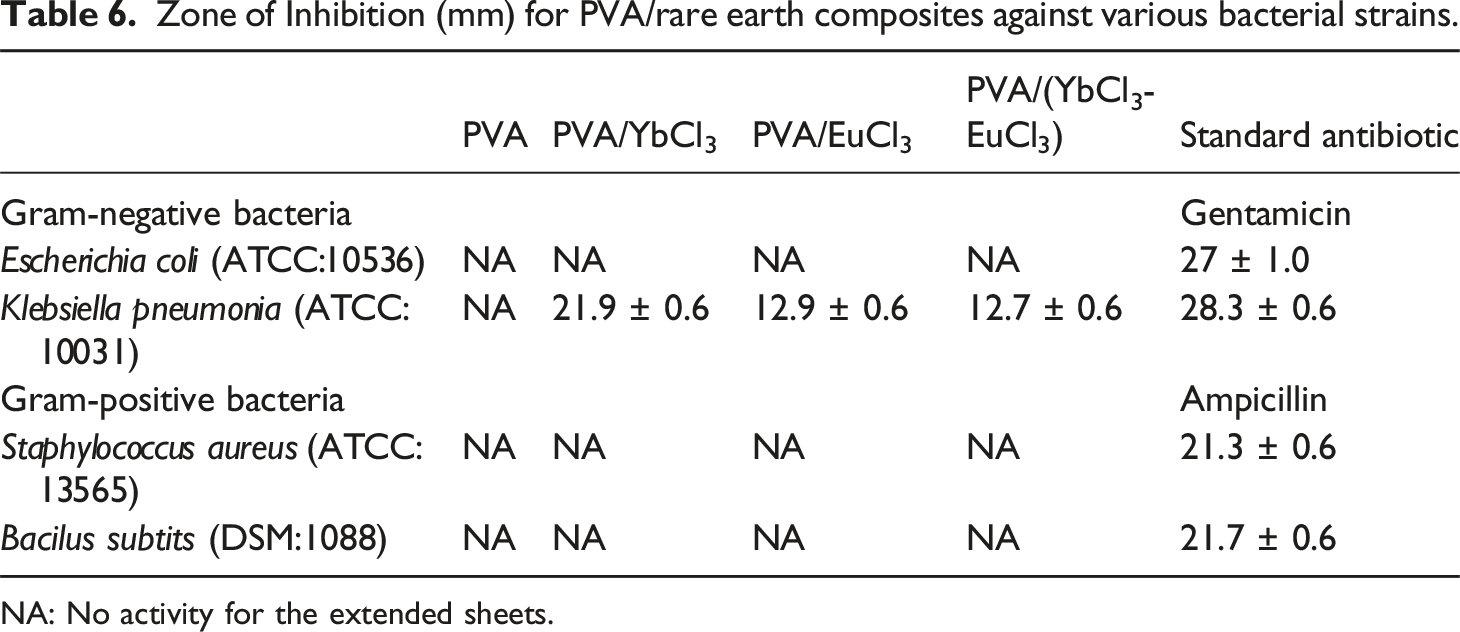
NA: No activity for the extended sheets.
The enhanced antimicrobial activity against Klebsiella pneumoniae is mainly linked to its structural and physiological features, which make it more sensitive to rare-earth-ion–loaded PVA films. As a Gram-negative bacterium with a thin peptidoglycan layer and a highly negative lipopolysaccharide-rich outer membrane, Klebsiella pneumoniae readily interacts with the positively charged Eu3+/Yb3+ ions released from the composite, promoting membrane destabilization, increased permeability, and leakage of intracellular contents. Rare-earth ions can also induce reactive oxygen species (ROS) formation and disrupt key metabolic pathways, to which Klebsiella pneumoniae is particularly susceptible due to its high membrane fluidity and relatively weak oxidative defenses compared with other Gram-negative and Gram-positive microorganisms. The outer membrane also acts as a selective barrier, and the passive diffusion of antimicrobial agents depends on its composition, including the presence and structure of porin channels, which differ from those in Escherichia coli. Variations in porin expression or structure, as well as the interactions between the polymeric film and bacterial surface, can affect the uptake, distribution, and overall efficacy of the antimicrobial agent.89–91 These combined effects—electrostatic interactions, ROS generation, membrane destabilization, and membrane-structure-dependent uptake—explain the comparatively stronger inhibition observed against Klebsiella pneumoniae. This understanding may guide the potential biomedical and pharmaceutical applications of such composite materials.
Conclusion
Composite films were synthesized by mixing PVA and 5 wt % of EuCl3, YbCl3, and (EuCl3 -YbCl3) by solution casting technique. The complexation of rare earth ions in the PVA polymer matrix was confirmed by the results obtained from the XRD structural parameters and FT-IR spectra. An investigation was conducted on the thermal behavior of composite films. The composite containing 5 wt % YbCl3 had the highest residual percentage at 500°C among all composites, indicating enhanced thermal resistance compared to the other formulations. UV–Vis study showed that the absorbance depends on the content of Eu3+ and Yb3+ ions. Both direct and indirect electronic transitions are allowed. The optical band gap values of PVA/rare earth composites are smaller than those of pure PVA. A rise from 2.03 to 2.87 was seen in the refractive index. The PVA/(YbCl3–EuCl3) composite film exhibited the highest refractive index (n = 2.93), corresponding to the lowest indirect band gap (1.60 eV) and highest Urbach energy, indicating a synergistic effect of dual-ion doping that enhances optical density through increased structural disorder and charge localization. A broad frequency range of 10−1 to 107 Hz at room temperature was used to study the AC electrical conductivity of the composite films. It is demonstrated that the electrical conductivity of the composites follows Jonscher’s law. The conductivity is shown to be lowered for 5 wt% EuCl3, while it is maximized for 5 wt% (EuCl3 -YbCl3). The enhanced electrical conductivity observed in the doped PVA composites correlates with the reduced optical band gap and increased Urbach energy, indicating that greater structural disorder and localized states facilitate both charge transport and optical absorption. The complex M* electric modulus and the complex Z* impedance formalisms were used to examine the experimental dielectric data. By analyzing the impedance components Z′ and Z″, the equivalent electrical circuit of the composite films was determined. The charge carrier’s short-range movement also causes relaxation, and the current system follows the non-Deby relaxation. Antibacterial studies showed selective activity, with the PVA/YbCl3 composite exhibiting the highest inhibition zone against Klebsiella pneumoniae, suggesting potential biomedical or antimicrobial packaging applications.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Science, Technology & Innovation Funding Authority (STDF) provides open-access funding in cooperation with the Egyptian Knowledge Bank (EKB). There was no specific funding for this study from any funding entity in the public, commercial, or not-for-profit sectors.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
