Abstract
The copolymer precursor, poly [4-(4-vinylbenzyl)oxy)phthalonitrile]-co-methyl methacrylate (poly (VBOPN-co-MMA)), was synthesized through copolymerization of VBOPN and MMA at a temperature of 110°C. Subsequently, a single-chain polymer complex of cobalt phthalocyanine (SCP-CoPc) was formed at 150°C by intramolecular macrocyclization between the cobalt and the phthalonitrile group in poly (VBOPN-co-MMA), using excess cyclohexanol. The formation of the SCP-CoPc complex was confirmed through various spectroscopic techniques such as UV/Vis, FT-IR, 1H-NMR, and 13C-NMR. In particular, the disappearance of -CN band at 2232 cm−1 from FT-IR indicated the formation of the SCP-CoPc complex, while the presence of specific absorption bands in the UV/Vis spectrum further confirmed its creation. The electrical, dielectric, optical, and thermal properties of the SCP-CoPc complex and SCP-CoPc/ZnO 5% composites were investigated. The SCP-CoPc/ZnO 5 wt% nanocomposite exhibited improved dielectric and DC conductivity with increasing temperature. The activation energy (Ea) was found to be 0.35 eV for SCP-CoPc and 0.25 eV for SCP-CoPc/ZnO 5 wt% nanocomposite, indicating that the electrical conductivity followed the Arrhenius equation. The optical properties, including band gap, dielectric constant, and refractive index, were calculated for the SCP-CoPc/ZnO 5 wt% nanocomposite. The SCP-CoPc complex possessed a band gap of 2.89 eV, while the SCP-CoPc/ZnO 5 wt% nanocomposite had a slightly reduced band gap of 2.36 eV. The refractive indices of SCP-CoPc and SCP-CoPc/ZnO 5 wt% were also determined.
Keywords
Introduction
Single-chain nanoparticles (SCNPs) are an extremely fascinating material derived from advancements in single-chain technology in recent years. Similar to how a single polymer chain folds and compresses to form its natural structure, protein folding, SCNPs are a class of polymers that can fold into tiny nanoparticles. They serve as building blocks for the development of synthetic analogues of biomacromolecules like proteins.1–4 The synthesis methods for SCNPs are highly flexible, offering a wide range of potential applications in various fields. These applications include sensing, drug delivery, repairable materials, and catalysis.5–8
The production of SCNPs involves creating intrachain bonds between pendant groups of a polymer, either thermally or photochemically, as well as using external linker molecules. These linkages can be covalent or non-covalent, and even dynamic.9,10 Of the various methods for producing SCNPs, the coordination of functional units in the polymer chain with externally supplied metal ions is particularly intriguing. Metal ions with luminescent, magnetic, or catalytic properties can be combined with the adjustable qualities of a polymer framework.4,11
When it comes to the synthesis of SCNPs, controlled polymerization techniques that can accommodate a wide range of functional groups and monomers are crucial. These approaches enable the creation of polymers with low molecular weight and narrow distribution. Polymer chains with added functionalities can be utilized to encapsulate metals. The subsequent step involves establishing specific connections within the same chain while preventing the formation of networks between different chains.12,13 To facilitate coordination with metal centers without hindrance from the rigid backbone, the positioning of functional units along the chain needs to be considered. Additionally, factors such as ligand-to-metal ratio, precursor metal complexes, and stable metal incorporation significantly influence metal-SCNPs.12,14 Extensive research has been conducted to develop well-defined polymer precursors that carry pendant groups responsible for forming stable metal-SCP complexes. Various functional groups, including imidazole, 15 imidazolium, 16 phenanthroline,12,17 phosphine derivatives, 18 diene,9,19,20 thiole, 21 phthalonitrile10,22,23 have been extensively studied to achieve this objective.
Controlled/living radical polymerization methods like nitroxide-mediated radical polymerization (NMP), 21 reversible-addition cleavage chain transfer (RAFT) polymerization, 24 and atom transfer radical polymerization (ATRP) 23 have been employed to prepare precursor copolymers with pendant groups. Utilizing precursor copolymers or linear copolymers, researchers have successfully synthesized and characterized novel functional single-chain polymers incorporating copper phthalocyanine (CuPc), zinc phthalocyanine (ZnPc), and cobalt phthalocyanine (CoPc).10,13,22
In our work, we have employed phthalonitrile entities from the polymer precursor to create collapsed cobalt atoms in single-chain polymers. These single-chain polymer-metallophthalocyanines (SCP-MPc) are extensively studied as target molecules for light-emitting devices, dye-sensitized solar cells, and photosensitizers for photodynamic treatment due to their optical and electrical properties. SCP-MPcs belong to a category of organometallic chromophores known for their two-dimensional, 18π-electron-conjugated structure25–27
The structure of this material enabled the development of nanocomposites to improve their physicochemical properties. Fillers that conduct electricity can be added to macromolecules or polymers to give them electrical conductivity. 28 Conductive fillers commonly used to create composite materials include carbon allotrope, metal, and metal oxide nanoparticles. These fillers have been the subject of extensive research for their potential use in polymer-based electrical nanocomposites. 10 The amount and type of conductive filler used in composites affect their electrical conductivity. Filler particles create paths of conductivity in the polymer matrix, which increases the conductivity of the mixture. 29 Nanocomposites can have enhanced optical, thermal, dielectric, and electrical properties due to the interactions between the polymer matrix and the nanofiller particle. The physical and chemical qualities of nanocomposites are improved the more filler is evenly distributed in the polymer matrix. Zinc oxide nanoparticles (ZnONPs) are utilized for reinforcing polymeric materials due to their n-type semiconductor characteristics. It is a unique phenomenon in which an electron in a semiconductor or insulator material is excited to a higher energy state, leaving behind a positively charged vacancy called a hole. As a result of their mutually attractive electrostatic forces, they combine to form a stable composite entity called an exciton.30,31 ZnONPs are stable at ambient temperature and have high exciton binding energies, making them ideal for use in low-wavelength optoelectronic devices. 32
Our findings demonstrate a new way to apply dye materials in industry: the incorporation of cobalt phthalocyanine groups into SCP, followed by the synthesis of a green colored single chain polymer containing CoPc. The importance of poly (methyl methacrylate) (PMMA) to the polymer industry is well-established. This study stands out since no other reports have been found in the literature describing the synthesis of PMMA with a CoPc ring, a green color, and a dye character. Despite the fact that the tendency of phthalocyanines to adhere to one another is viewed as a detriment, phthalocyanines are utilized in a wide variety of applications. Therefore, we believe that a high-dilution reaction environment in which CoPc is made using a chain of methacrylate polymer molecules is the ideal option to address this issue. We also detail the production and characterization of a ZnONPs nanocomposite that has been doped with cobalt phthalocyanine. This work lays the groundwork for the development of high-dielectric-permittivity and optoelectronic-characteristic materials.
Materials and methods
Materials
4-vinylbenzyl chloride (VBC, Aldrich, > 90%) and methyl methacrylate (Fluka, 99% (GC)) were employed without any additional treatment. Both 2,2'-bipyridine (bpy) (Merck, 98%) and CuBr (Acros, 98%) were subjected to vacuum drying for 24 h. 2,2'-Azobis (isobutyronitrile) (AIBN, Accros, 98%) was recrystallized in methanol, and water molecules were eliminated by treating it with CaH2, followed by the addition of anhydrous MgSO4, and finally purification with benzene. 4-Chloromethyl-7-hydroxy coumarin (synthesized in our lab.) was used as is. Potassium carbonate (Merck, > 99.0%), phthalonitrile (Alfa Aesar, 98%), and nitrophthalonitrile (BLD Pharm, 99.5%) were used without further modification. Solvents such as cyclohexanol (Aldrich, > 99%) and tetrahydrofuran (THF, Aldrich, > 99.9%) were used without alteration. 1,8-diazabicyclo [5.4.0]undec-7-ene (DBU, Aldrich, 98%) was employed as a phase transition catalyst. Zinc oxide nanoparticles (ZnONPs) from Nanografi Turkey were utilized for the fabrication of single-chain polymer nanocomposites. All other solvents were used as received.
Characterization techniques
The Perkin Elmer Spectrum One Fourier-transform infrared spectrometer was used to acquire FT-IR spectra using the film on the NaCl window. The 1H-NMR bands were collected using a Bruker AVANCE III 400 MHz NMR spectrometer. For the purpose of analyzing the optical characteristics, we employed the Aurora 4000 GE-UV-NIR method. The UV-Vis readings in THF were taken with a Shimadzu 1240 min UV spectrophotometer. Using a field-emission scanning microscope (Carl Zeiss EVO MA10) equipped with a 5-20 kV SEM, images of the nanocomposite were taken. The Shimadzu TGA-50 and DSC-50 were used to measure temperatures from room temperature to 600°C and 200°C, respectively, at a heating rate of 10°C/min and a flow rate of 10 mL/min. The dielectric and electrical properties of our research component, as detailed in the composite preparation section, we followed a comprehensive procedure. Initially, we synthesized SCP-CoPc from the P(VBOPN5.6%-co-MMA) and composites of SCP-CoPc with 5 wt of ZnO. Then, we meticulously prepared our sample by taking 125 mg of the dry powder and grinding it to the desired consistency using a mortar. Subsequently, we applied a four-ton force to shape the collected powder into a pallet. Prior to launching into the experimental phase, we conducted a precise measurement of the plate’s thickness, utilizing a sensitive measuring tool. With our sample ready and measurements in place, we moved on to the setup of the impedance analyzer. Our focus was on ensuring its seamless functionality, which involved a thorough examination of the sample holder area, constructed with a gold-fixed disc, and a verification of the heater. Finally, the experimentation phase commenced, with tests being carried out at four distinct temperature settings. Through this comprehensive procedure, we were able to thoroughly assess the dielectric and electric properties of our research component, aligning with the specified methodology outlined in the composite preparation section. The materials were measured by taking readings between 100 Hz and 10 kHz with a QuadTech 7600 LRC impedance analyzer. The Fytronix Electrical Characterization device’s I-V and DC voltmeter was used to evaluate the nanocomposite.
Synthesis of 4-vinylbenzyl alcohol
The method for producing 4-vinylbenzyl alcohol was adapted from. 33 To do this, we used 0.401g and 5.59g of Bu4NCI (TEBAX, as a phase transfer reagent) to combine 10 mmol NaOH with 120 mL of water. The solution was then spiked with 50 mmol of 4-vinylbenzyl chloride (7.631g). The mixture was then reflexed for half an hour. Following cooling to room temperature, the reaction mixture was extracted twice with ethyl acetate. An entire day was spent drying the organic phase over dry MgSO4.
Synthesis of 4-((4-vinylbenzyl)oxy)phthalonitrile
The formation of VBOPN was accomplished using a technique derived from scientific literature. 10 This was achieved by adding 25 mL of distilled DMF to a round-bottom flask containing 3g of 4-vinylbenzyl alcohol and 3.85g of 4-nitrophthalonitrile. Before adding K2CO3 (33.453 mmol, 4.61g) over the course of 8 h, the solution was mixed for 30 min under N2 flow. The reaction took place at a temperature of 70°C. The product was desiccated at 40°C in a vacuum after being precipitated from methanol for 8 h.
The vibrational modes were determined through FT-IR analysis (measured in cm−1) and are as follows: The 3010-3119 band corresponds to the stretching of =C-H bonds on the aromatic ring and vinyl group. The C-C stretching observed on the aromatic ring exhibited a wavenumber of 2597 cm−1, whereas the CN stretching manifested at a wavenumber of 2230 cm−1. The vibrational mode of the C=C bonds on the vinyl group was observed at a wavenumber of 1625 cm−1, while the C-O stretching adjacent to the aryl ring was detected at a wavenumber of 1253 cm−1.
1H NMR (acetone-d6): The multiplet (m) observed in the chemical shift (δ) range of 7.91–7.34 corresponds to a total of 7 protons. One of the protons is situated in the meta position that is contiguous to the oxygen atom. Two protons are located in the ortho position, with one of them positioned between the ipso carbon adjacent to the oxygen atom and the nitrile group. The two remaining protons are situated in the ortho position, which is in close proximity to the vinyl group. A proton was observed in the range of δ = 6.55–6.65, which manifested as a doublet of doublets (dd) peak. The observed peak is attributed to the trans proton located on the vinyl group. A single peak corresponding to one proton was detected at a chemical shift of δ = 5.25. The aforementioned proton is a constituent of the aryl-CH2-O functional group. A doublet peak was detected within the chemical shift range of δ = 5.55–5.64, indicating the presence of a single proton. The observed peak is indicative of the presence of an olefinic proton located in close proximity to an aromatic ring. An additional doublet peak (d) was detected at a chemical shift of δ = 5.32–5.41, indicating the presence of a single proton. The observed peak can be attributed to the cis proton located on the vinyl group.
13C NMR (acetone-d6): chemical shift δ = 162.10, 137.67, 136.62, 136.30, 135.47, 128.98, 126.80, 120.98, 120.96, 116.72, 116.69, 116.22, 115.35, 106.59, 70.68.
Synthesis of processor copolymer MMA and VBOPN by ATRP method
In this experiment, a 10 mL round-bottomed flask was thoroughly cleaned and then purged with argon gas. The flask was then loaded with the components in a specific sequence. Firstly, 0.118 mmol of CuBr (0.0167g) was added, followed by 0.2230 mmol of 2,2'-bipyridine (0.037g), and finally, 0.116 mmol of 4-chloromethyl-7-hydroxy coumarin (0.0243g) as the initiator. After introducing 0.5 mL of dioxane, the mixture resulted in the formation of brown compounds. The flask contents were shaken for 3 min. To remove oxygen from the reaction vessel, argon gas was allowed to flow for 10 min. Subsequently, 0.41g of VBOPN powder (1.54 mmol) and 1g of methyl methacrylate (MMA) liquid (10 mmol) were added to the flask in a theoretical mathematic predicted monomer ratio. The polymerization reaction proceeded at 110°C for 24 h. The resulting material was precipitated in cold methanol, filtered, and then air-dried for 2 h at room temperature. Finally, it was vacuum dried for 24 h at 40°C. The product's properties were determined using spectroscopy techniques such as FTIR and NMR. The product was analyzed using FTIR and NMR spectroscopy.
The FTIR spectrum show those distinct peaks in wavenumbers (cm−1)2995-2948, 2230, 1730, 1600, 1561, 1490, 1449, 1256, 1150, 1090, 900-750
The 1H NMR spectrum recorded in acetone-d6 revealed peaks at δ = 7.0-8.05, corresponding to aromatic ring protons, and peaks at δ = 5.20-5.32, attributed to ArOCH2. Additionally, a peak at δ = 3.6 indicated the presence of CH3O- from MMA. The 13C NMR spectrum obtained in acetone-d6 exhibited peaks at δ = 161.7, 136.08, 135.27, 129.49, 127.80, 126.91, 126.76, 126.26, 126.04, 120.04, 119.78, 117.53, 115.60, 115.21, 114.92, 70.89, and 67.81.
Synthesis of SCP-CoPc
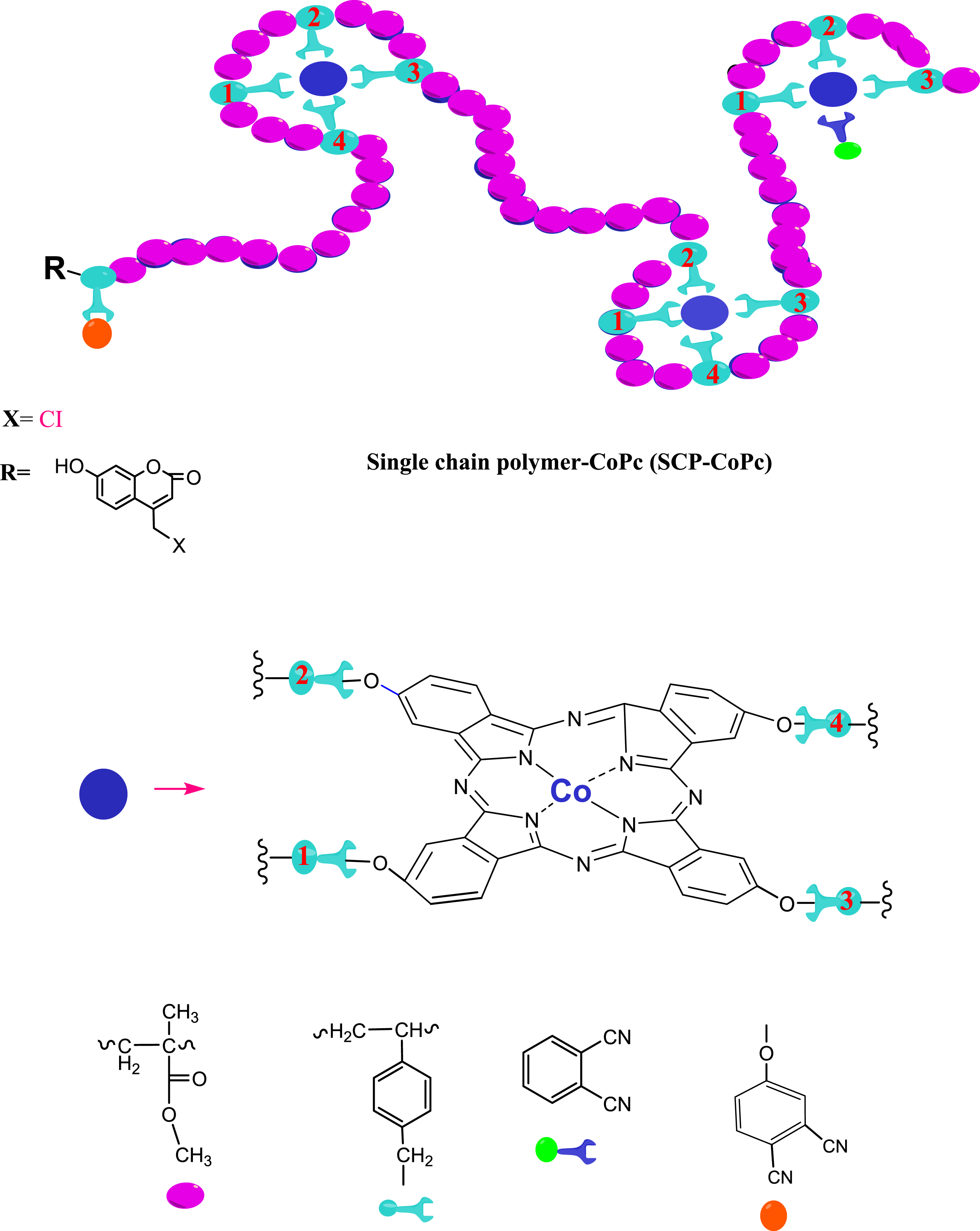
In a flask with three necks, containing 150 mL of cyclohexanol solvent, the following components were added: 305 mg of P(VBOPN5.7%-co-MMA) (0.27 mmol), 2.57 mg of Co(CH3COO)2.2H2O (0.015 mmol), and 30.9 mg of phthalonitrile (PN) (0.25 mmol). Before proceeding, the mixture was purged with argon gas. Then, it was injected with 0.049 mL of DBU (1,8-diazabicyclo [5.4.0]undec-7-ene) (0.32 mmol). The reaction was stirred for 38 h at temperatures ranging from 140 to 150°C. The completion of the reaction was indicated by the disappearing of the nitrile (CN) band in the FT-IR spectrum from 2230 cm−1 completely. At this point, the reaction was stopped, and the temperature was lowered to 25°C. The solution was allowed to cool in cold ethanol, resulting in precipitation. The obtained SCP-CoPc was then vacuum-dried.
Nanocomposite preparation
Zinc oxide nanoparticles (ZnONPs) were dissolved in THF using ultrasonication and continued stirring for 2 h to create a homogeneous dispersion. The sonicated solution was then added to the SCP-CoPc matrix solution in THF, and the entire mixture was mixed for another 2 min while being ultrasonically agitated to achieve even distribution. The SCP-CoPc and 5 wt% ZnO mixture was then injected to the cooled ethanol and vacuum-precipitated at 50°C for 24 h. The dried ingredients were then ground into a powder in a mortar for 30 min. The electrical properties of the nanocomposites were next studied by compressing them into tablets at a pressure of four tons.
Results and discussion
Characterization of precursor copolymer and SCP-CoPc
A styrene derivative containing a phthalonitrile group was successfully synthesized by performing a nucleophilic substitution reaction between 4-nitrophthalonitrile and 4-vinylbenzyl alcohol. The precursor copolymer of VBOPN and MMA was then created using the ATRP technique. The process involved heating the mixture to 110°C and using CuBr as the central metal, 2,2'-bipyridine as the ligand, and 4-chloromethyl-7-hydroxy coumarin as the initiator. Figure 1 illustrates the 1H-NMR spectral data of the aceton-d6 solvent used in the copolymer synthesis. In this spectrum, the protons with aromatic chemical shifts ranging from 7.0 to 8.05, the protons with ArOCH2- chemical shifts at 5.32 ppm (appearing as a singlet), and the protons with CH3O chemical shifts in MMA units at 3.6 ppm were the most prominent features. These chemical composition changes are indicated by arrows in Figure 1. The composition of the copolymer was determined by comparing the integral height of the CH3O signal at 3.6 ppm (characteristic of MMA) with that of the ArOCH2 signal at 5.32 ppm in the VBOPN components. The ratio between these signals provided the solution, revealing a molecular concentration of 5.7% VBOPN units within the copolymer composition. In the cyclotautomerization process of P(VBOPN 5.7%-co-MMA) to produce green SCP-CoPc, Co(CH3COO)2.2H2O, excess phthalonitrile, and cyclohexanol were employed. It was observed that when phthalonitrile was omitted, the yield decreased and insoluble impurities formed. To prevent the occurrence of chain coupling or crosslinks and achieve the production of CoPc on the SCP, an excess amount of phthalonitrile, unrelated to the synthesis of phthalocyanines, was added to the reaction flask. The key to successful rephrasing was the use of a highly diluted medium and an excessive quantity of phthalonitrile during the production of SCP-CoPc. This approach effectively limited the intermolecular coupling processes of SCP-CoPCs, which otherwise resulted in the formation of difficult to remove contaminants. 1H and 13CNMR spectrum of precursor copolymer.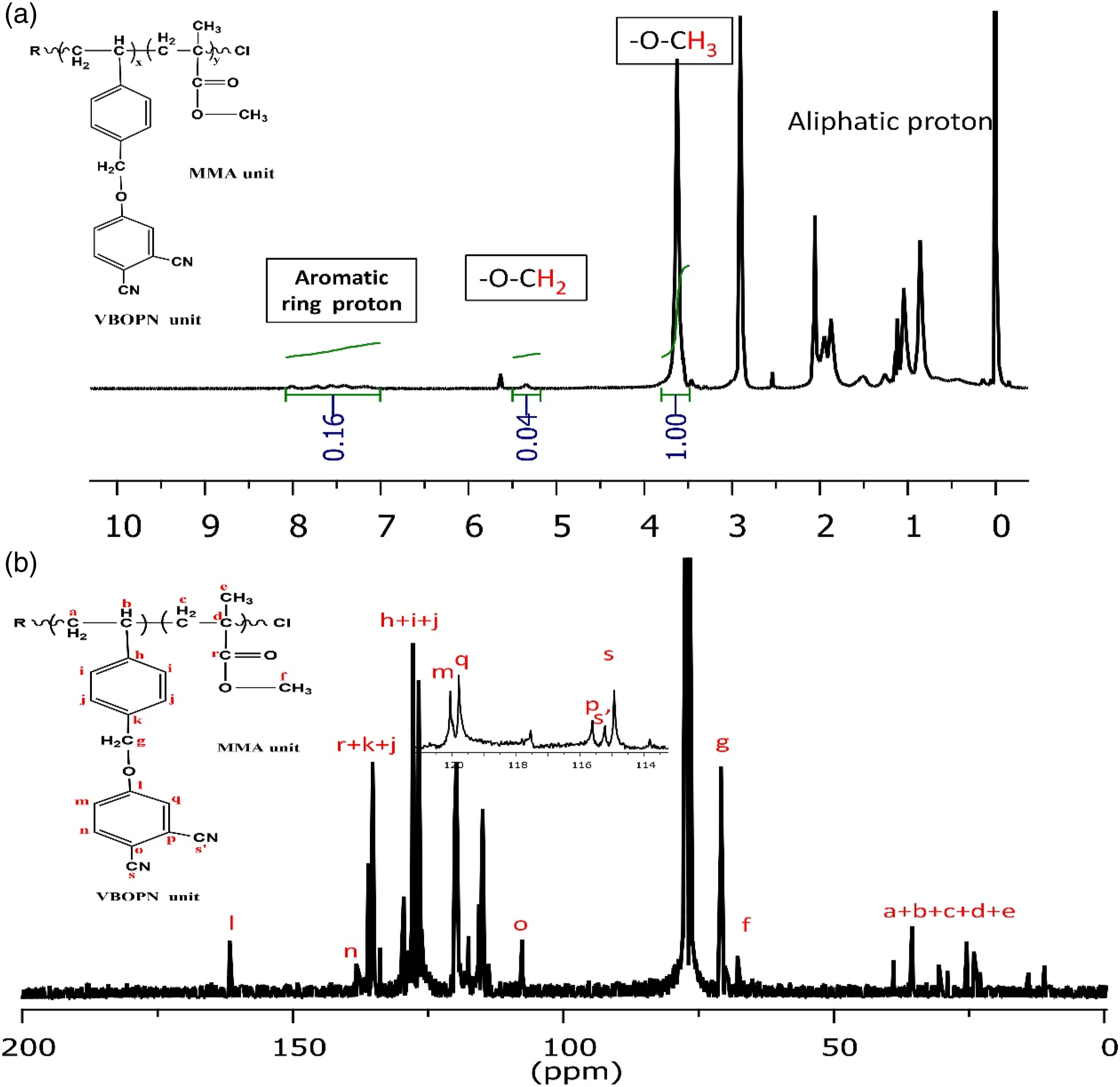
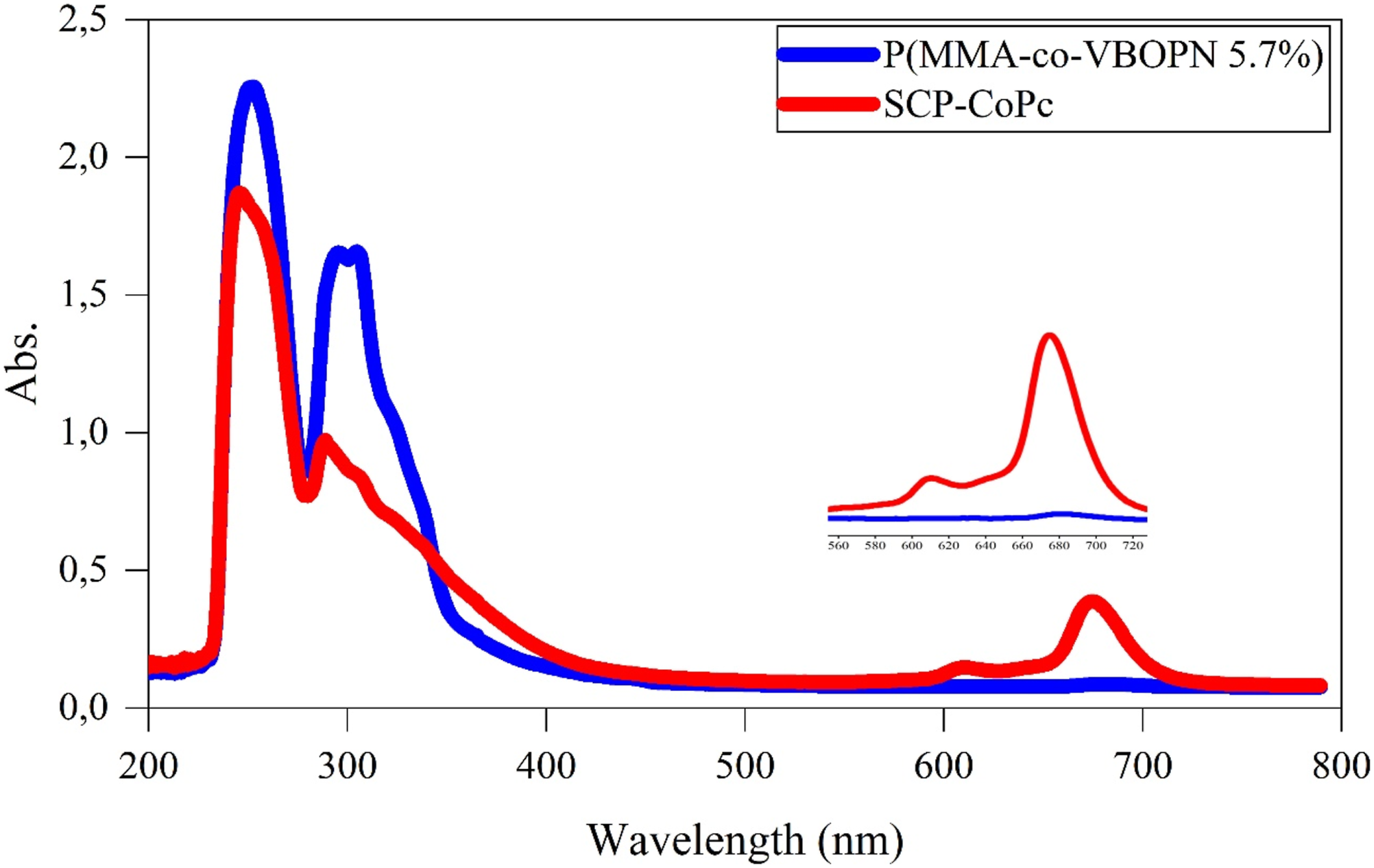
The reaction was allowed to proceed for a duration of 38 h, resulting in the formation of a transparent green solution. This implies that no insoluble particles were present in the solution medium. To eliminate cyclohexanol and any additional reagents, the solution was precipitated by adding excess cooled methanol. The resulting SCP-CoPc was then filtered, dried, and subsequently re-dissolved in dichloromethane. It was once again precipitated in cooled methanol and subjected to vacuum drying for an entire day. Figures 15 and 16 illustrate the mechanisms involved in the transformation of the precursor copolymer into SCP-CoPc and provide a representation of the polymer chain in the form of a diagram, respectively. FT-IR spectra of (red) precursor copolymer and (blue) SCP-CoPc. UV–Vis spectrum of red SCP-CoPc and blue copolymer (solvent: THF).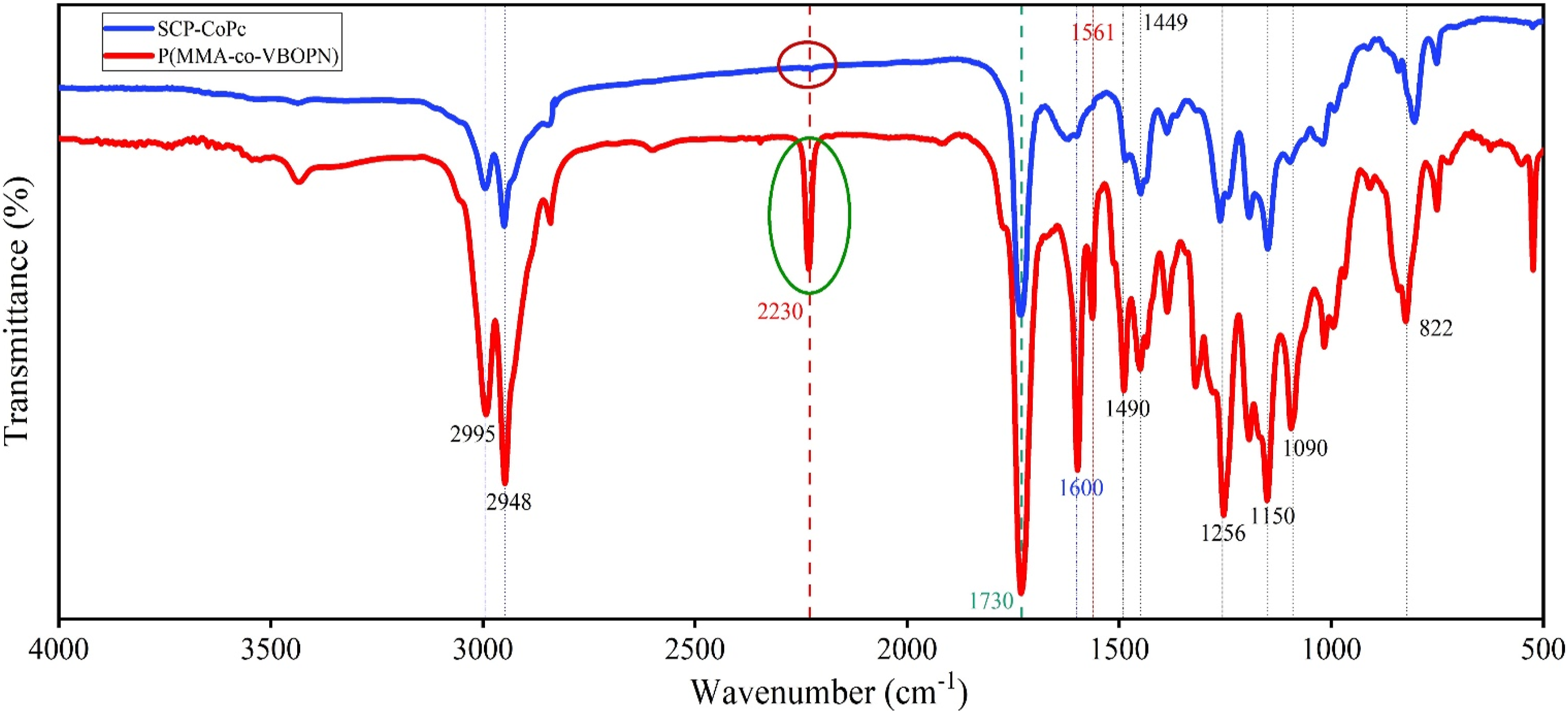
The FT-IR spectra of the precursor copolymer and the SCP-CoPc were compared in order to offer convincing evidence for the formation of SCP-CoPc. The results of the FTIR spectral analysis (Figure 2) and the bands are summarized in Table 1. It clearly shown in Figure 2 that the copolymer contain carbonyl group belong to the MMA entity and nitrile groups present in the VBOPN unit. The sharp CN and C=O bands stretched at 2230 and 1730 cm−1 respectively in the copolymer. This demonstrate that the complete absence of the 2230 cm−1 band stretching, which corresponds to CN resultant formation of SCP-CoPc. (a) SEM images and (b) EDX data of ZnONPs. FT-IR evaluation for copolymers of P(VBOPN-co-MMA) and SCP-CoPc of P(VBOPN-co-MMA).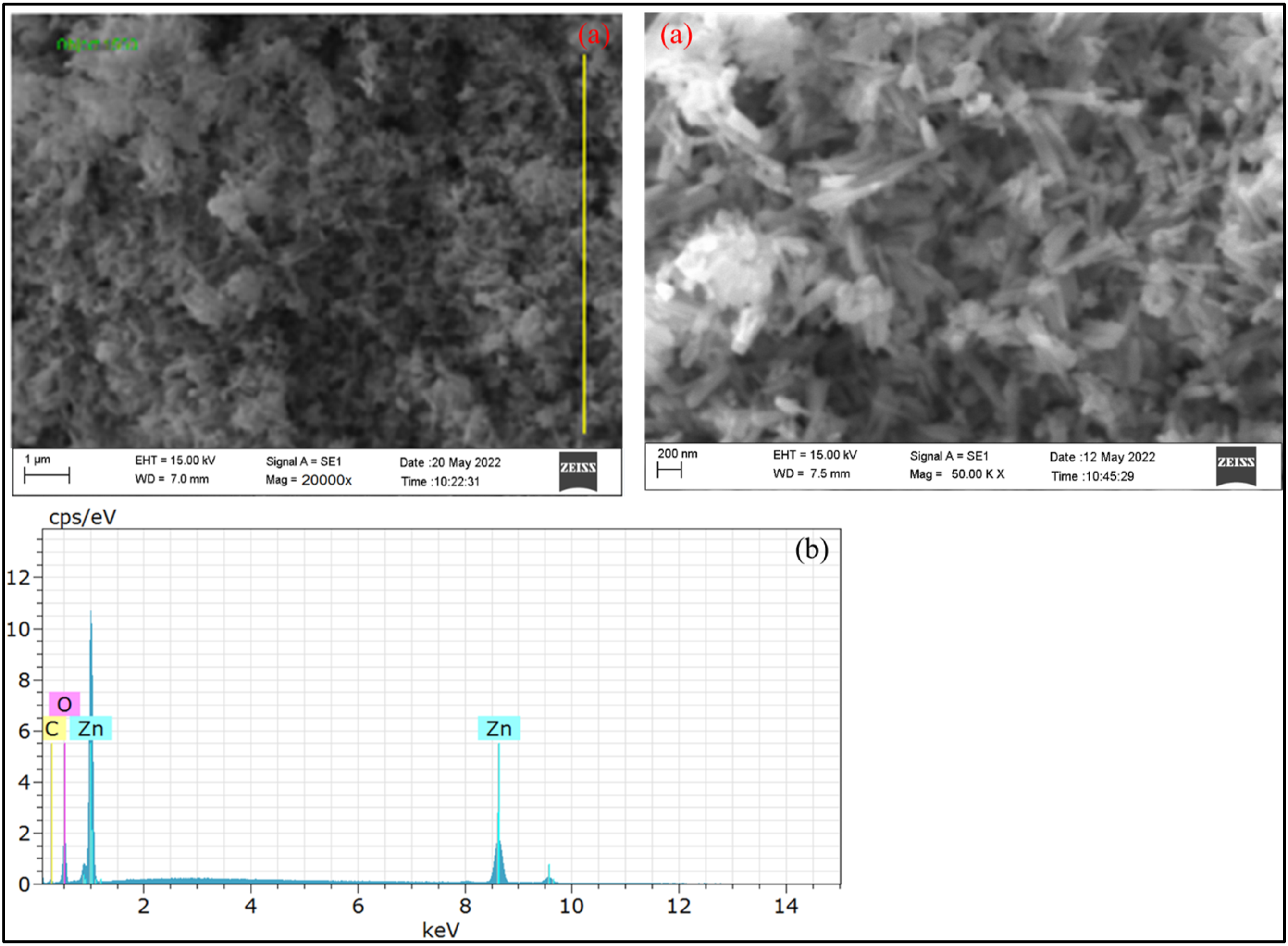
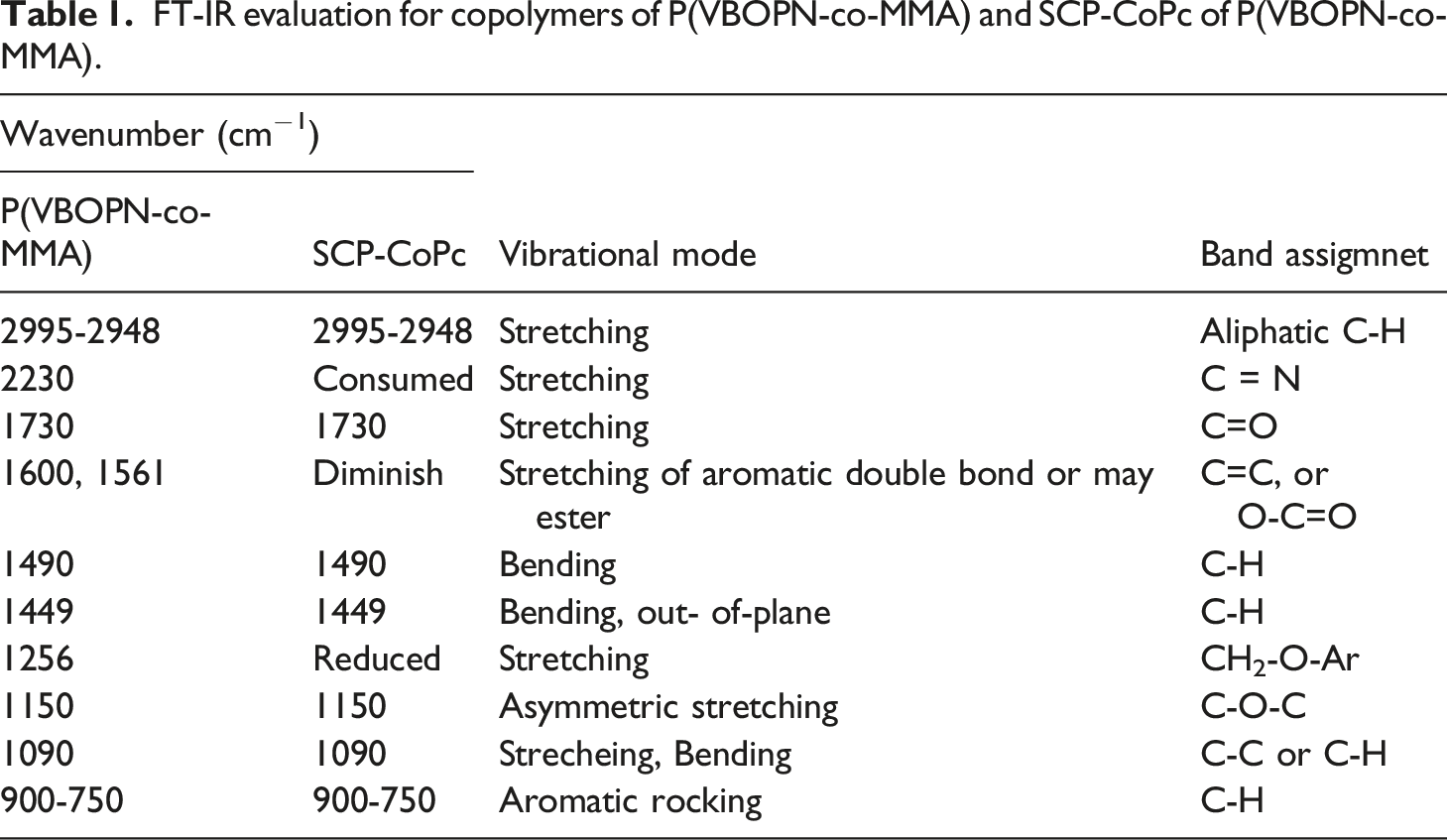
As depicted in Figure 2, at the 2230 cm−1 region, the distinct peak associated with the nitrile group has been completely consumed. This phenomenon occurs as a result of the interaction between the nitrile groups within the precursor linear copolymer and cobalt ions under the specified reaction conditions in the synthesis of SCP-CoPc.10,22,23 Additionally, the formation of phthalocyanine through cyclotautomerization has led to the diminishment of other peaks, notably those at 1600 and 1561 cm−1, which are attributed to the C=C bonds in the aromatic ring present in VBOPN. During the formation of SCP-CoPc, adjacent aromatic C=C bonds can be significantly affected by a number of effects. As a result of cyclotetramerization and the formation of cyclic structures, the electronic distribution in the aromatic ring is disturbed and electronic effects occur as a result. The participation of cyano groups in cobalt ion binding and the formation of cyclic structures can change the polarization and resonance properties of C=C bonds. The steric effects occur as a result of the constraints imposed by the cyclic structure and potentially affect the rotation and flexibility of the C=C bonds in adjacent aromatic rings. In addition, chemical bonding changes as coordination bonds form between cyano groups and cobalt ions and as cyclotetramerization progresses; as a result, changes occur in the chemical environment of the aromatic rings, which ultimately affect the vibrational modes. As a result of their role in coordination and cyclotetramerization, cyano groups also produce inductive and resonant effects that change electron density and distribution, thereby affecting the vibrational properties of C=C bonds in adjacent aromatic rings. It is worth noting that several other peaks remain unchanged in both P(VBOPN-co-MMA) and SCP-CoPc. This is expected since these bands correspond to stretching and vibration modes that remain relatively stable, such as CH2- and CH3- groups, as well as C=O bonds. A comprehensive summary of band assignments and vibration modes is provided in Table 1.
Figure 3 illustrates the measurement and depiction of the UV-Vis absorption of the SCP-CoPc system in THF. The spectrum of SCP-CoPc exhibits two prominent absorption bands, namely the Q and B bands. The Q band, falling within the visible range of 600-700 nm, is characterized by clear absorptions ranging from 603 to 670 nm. These absorptions correspond to the π-π* transition, originating from the HOMO to the LUMO of the Pc ring. The presence of the Q-band in the UV-Vis spectra of CoPc synthesis provides significant evidence for SCP-CoPc formation, resulting from the intramolecular cyclotautomerization of P(VBOPN5.7%-co-MMA) with Co(CH3COO)2.2H2O. Furthermore, distinctive absorption bands known as the B band were observed in the wavelength range of 300-400 nm, indicating a lower LUMO energy level. The obtained spectrum data clearly demonstrate the successful utilization of targeted CoPc structures with electronic absorption spectra containing single-chain polymer-metallophthalocyanines. This information is supported by references.10,13,22,23 DSC curves of (a) precursor copolymer, (b) SCP-CoPc and (c) SCP-CoPc/ZnO 5 wt% nanocomposite.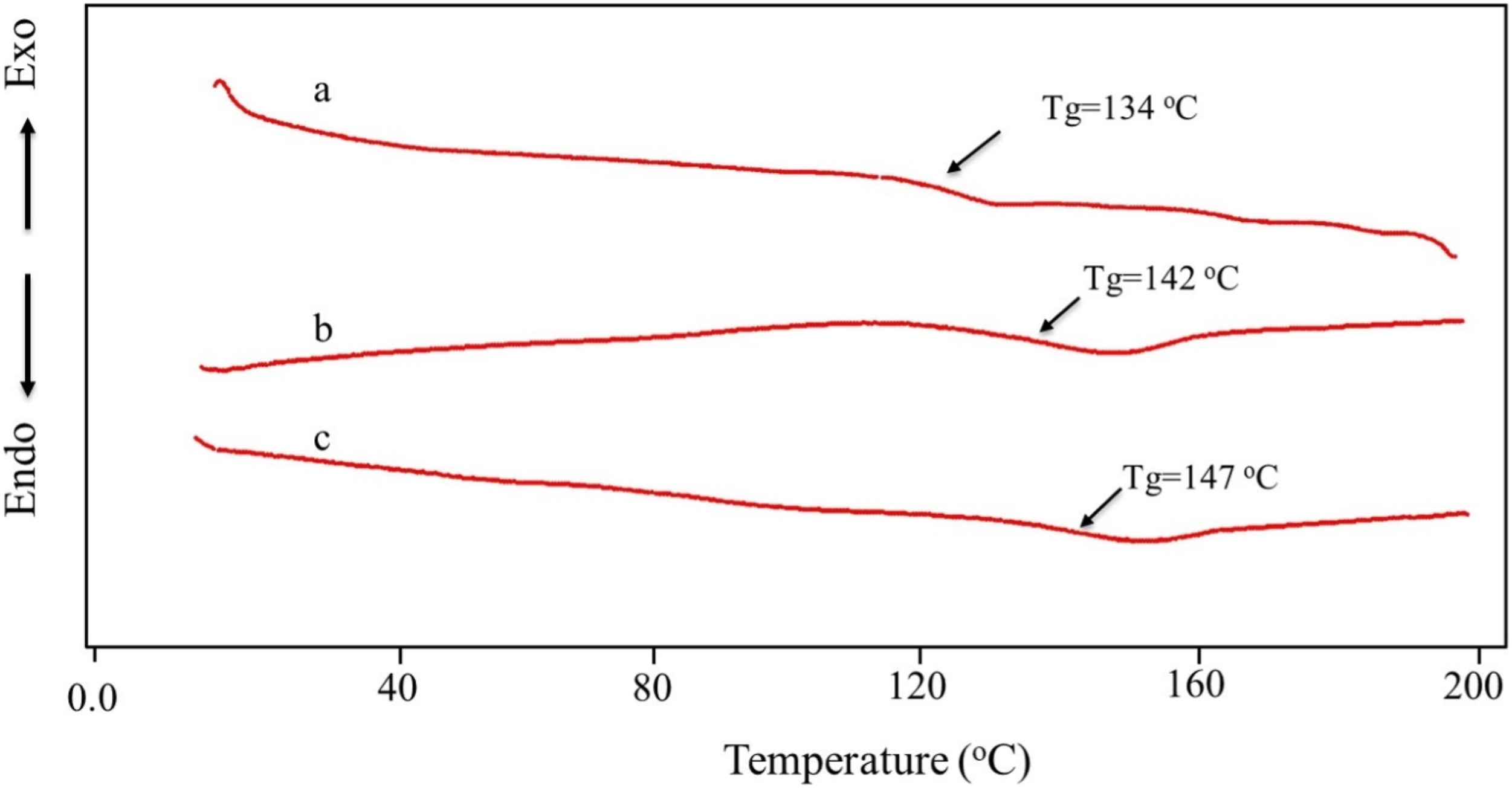
Scanning electron microscopy and energy dispersive X-ray analysis of zinc oxide nanoparticles
Scanning electron microscopy (SEM) and energy dispersive X-ray analysis were conducted to examine the morphology of zinc oxide nanoparticles, as depicted in Figure 4(a). The images in Figure 4(a) clearly illustrate the even dispersion of ZnONPs throughout the sample. The SEM micrograph of ZnONPs reveals the characteristic morphology of ZnONPs characterized by uniform distribution of rod-like structures spanning a range from nanometers to microns in size. Furthermore, we conducted an examination using energy-dispersive X-ray (EDX) spectroscopy to delve into the chemical composition. Figure 4(b) illustrates the EDX spectrum of ZnONPs, which reveals distinct signals indicating the presence of zinc, oxygen, and some other element, with atomic weight percentages of 67.0%, 24%, and 8.9%, respectively. These weight percentages are sequentially represented as 74%, 6.59%, and 1.83%. TGA curves of copolymer and A plot of log10 [1 – (1 –α)/T2] versus 1000/T of (VBOPN5.7%-co-MMA), SCP-ZnPc and SCP-ZnPc/5 wt% ZnO nanocomposite heated under nitrogen flow obtained at heating rates of 10.0°C/min.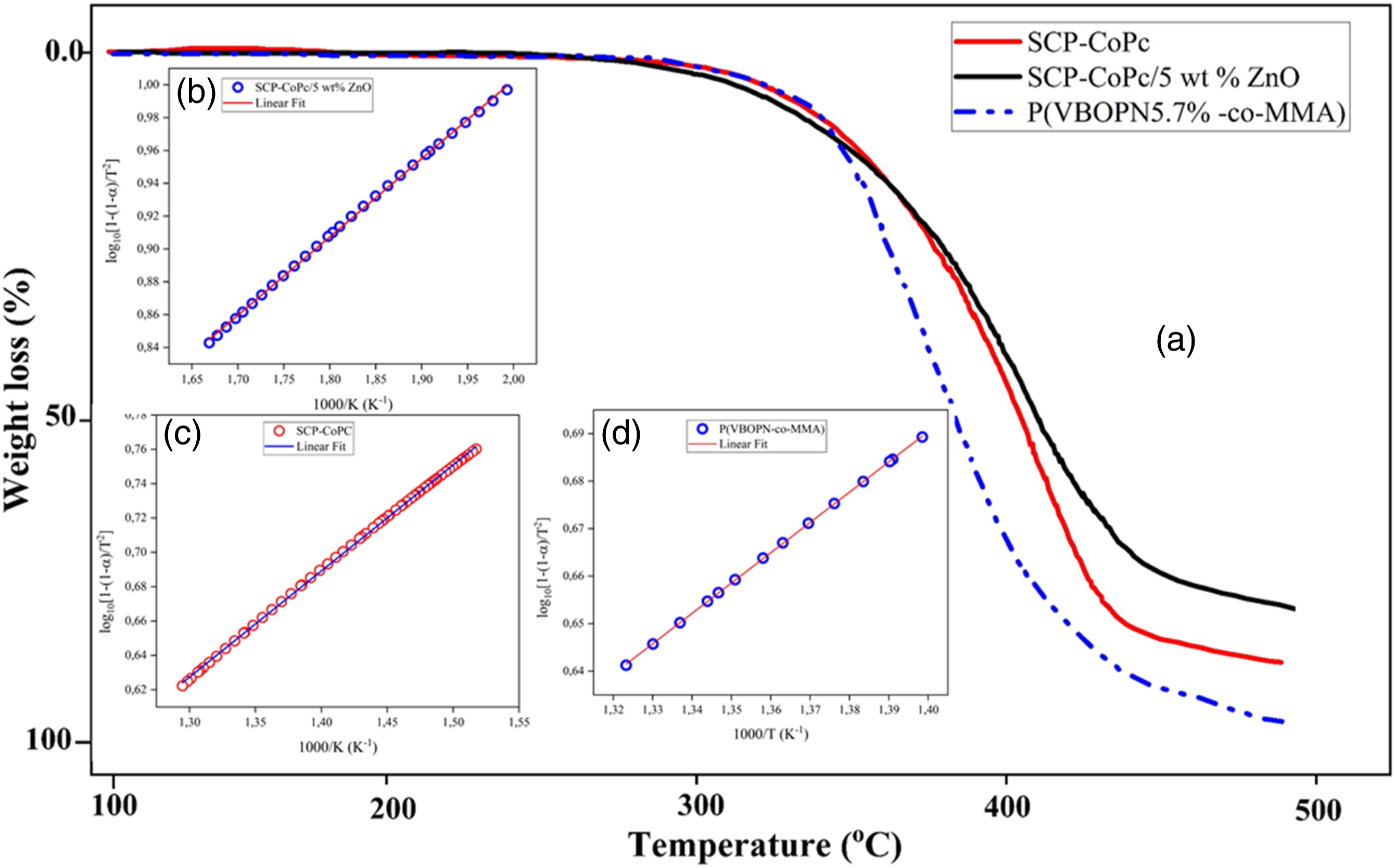
Thermal properties of materials
Figure 5 presents the glass transition temperatures (Tg) of three different materials: (a)P(VBOPN5.7%-co-MMA), (b)SCP-CoPc, and (c) SCP-CoPc/ZnONPs 5 wt%. Tg values were determined using data obtained from DSC thermograms. Based on the data, P (MMA-co-VBOPN5.7%) has a Tg of 134°C. The Tg of PMMA, on the other hand, is 105°C.The presence of VBOPN units in the precursor copolymer chains led to a reduction in free volume and an increase in Tg, which hindered chain movement to some extent. The DSC analysis showed that SCP-CoPc has a Tg of 142°C, which is 12° higher than that of P (MMA-co-VBOPN5.7%). This behavior indicates the elimination of phthalocyanine ring developments in the polymer chains, resulting in decreased chain entropy and enhanced uniformity. The investigation of the impact of zinc oxide nanoparticles (ZnONPs) on Tg revealed interesting results. The addition of ZnONPs to the SCP-CoPc matrix slightly increased its Tg. The DSC results indicated that the Tg of the nanocomposite reached 147°C. The high surface area of SCP-CoPc may explain the increase in Tg when exposed to ZnONPs. Therefore, it is reasonable to assume that interfacial interactions between the polymer matrix and the filler material also contribute to the Tg increase. Previous research has demonstrated that polymer nanocomposites containing ZnO can experience an increase in Tg when there are sufficient interactions between the polymer matrix and the fillers. The position of the additives relative to the interfaces plays a crucial role in affecting the glass transition temperature of polymer composites. It becomes more pronounced near strongly adsorbing surfaces but decreases in concentration near free surfaces. However, it can increase significantly in concentration near free surfaces.
34
Variation of dielectric constant, ε’, dielectric loss factor, ε’’, tanδ with frequency for SCP-CoPc and SCP-CoPc/ZnO 5 wt% nanocomposite at different temperature.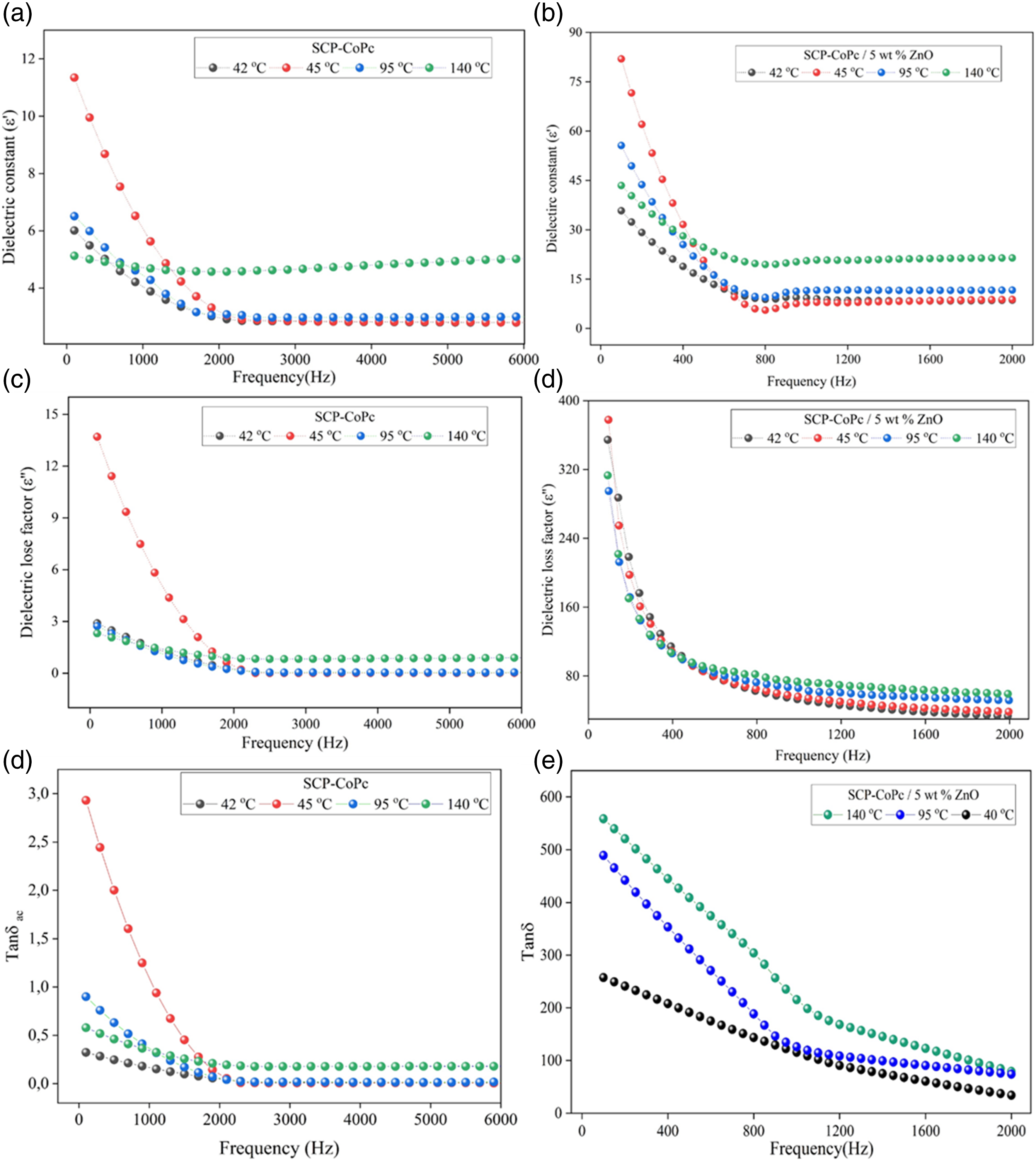
Figure 6 Jonscher’s power law applied on the (a)SCP-CoPc (b)SCP-CoPc/5 wt% ZnONPs at 140°C.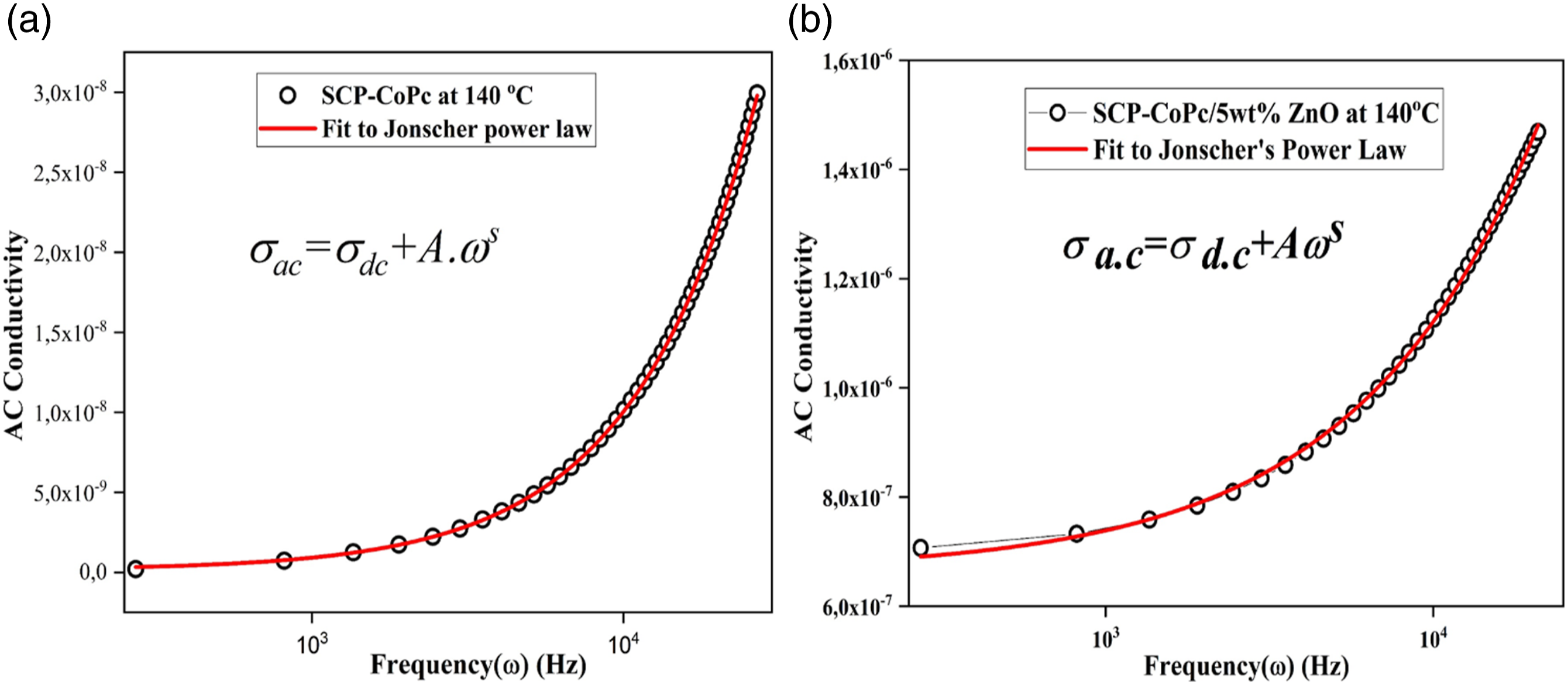
The final products obtained from the thermal decomposition process include heat, carbon monoxide, water vapor, and carbon dioxide. However, the presence of P(VBOPN5.7%-co-MM) in significant quantities indicates that only a small number of the previously mentioned links were present. The high phase of thermal decomposition occurs at around 400°C and results in radical transfer to the unsaturated chain ends. Subsequently, cleavage occurs due to random bond breakage, which takes place after 400°C. SCP-CoPc exhibits increased thermal stability, with higher temperatures required for peak weight loss in both stages. This remains true even with the addition of ZnO nanoparticles at a weight percentage of 5% to form SCP-CoPc/ZnO. Although the addition of ZnO nanoparticles does not significantly affect the decomposition temperature, it reduces the percentage of weight loss, likely due to the higher nanoparticle concentration. The thermal stability of SCP-CoPc undergoes changes with the introduction of nanoparticles. The SCP-CoPc/ZnO 5% weight sample shows lower thermal stability compared to pure SCP-CoPc when heated to 338°C. This indicates a moderate impact of ZnO on the thermal stability and deterioration rate of SCP-CoPc. The SCP-CoPc/ZnO 5 wt % weight sample loses 12% of its original weight when heated to 500°C. The presence of fillers like ZnO can either accelerate or slow down the decomposition rate of polymer matrices under heat. In our research, the inclusion of ZnO in polymer composites leads to a slower thermal degradation of SCP-CoPc after reaching 338°C. Various hypotheses have been proposed in research papers to explain the improved thermal characteristics of polymer nanocomposites. 35 One such hypothesis is the efficient heat sink theory, which suggests that ZnO nanoparticles limit the degradation of nanocomposites by dissipating more heat compared to the polymer matrices. Consequently, 11.5% of the residue can be attributed to ZnO, while the remaining residue consists of carbonized compounds and other materials. Table 1 provides a summary of the findings obtained from the TGA curves of the polymers and nanocomposites.
The assessment of kinetic parameters relating to solid-state reactions involving sample weight loss has been extensively documented in the literature.
36
In this study, the kinetic parameter, including activation energy, was evaluated for copolymer (VBOPN-5.7%-co-MMA), SCP-CoPc, and SCP-CoPc with 5 wt% ZnO, employing a non-isothermal kinetic equation originally proposed by Coats et al.
37
This equation is represented as: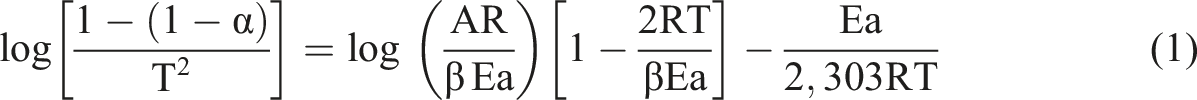

W0 is the initial weight, W is the weight at time t, Wf is the final weight E is the activation energy of reaction, R is the gas constant, β is the heating rate in K/min. The order of reaction is assume as 1st, A is frequency factor.
The decomposition characteristics of polymer, SCP-CoPc and SCP-CoPc/5 wt% ZnONPs and activation energy calculated by coats-redfern.
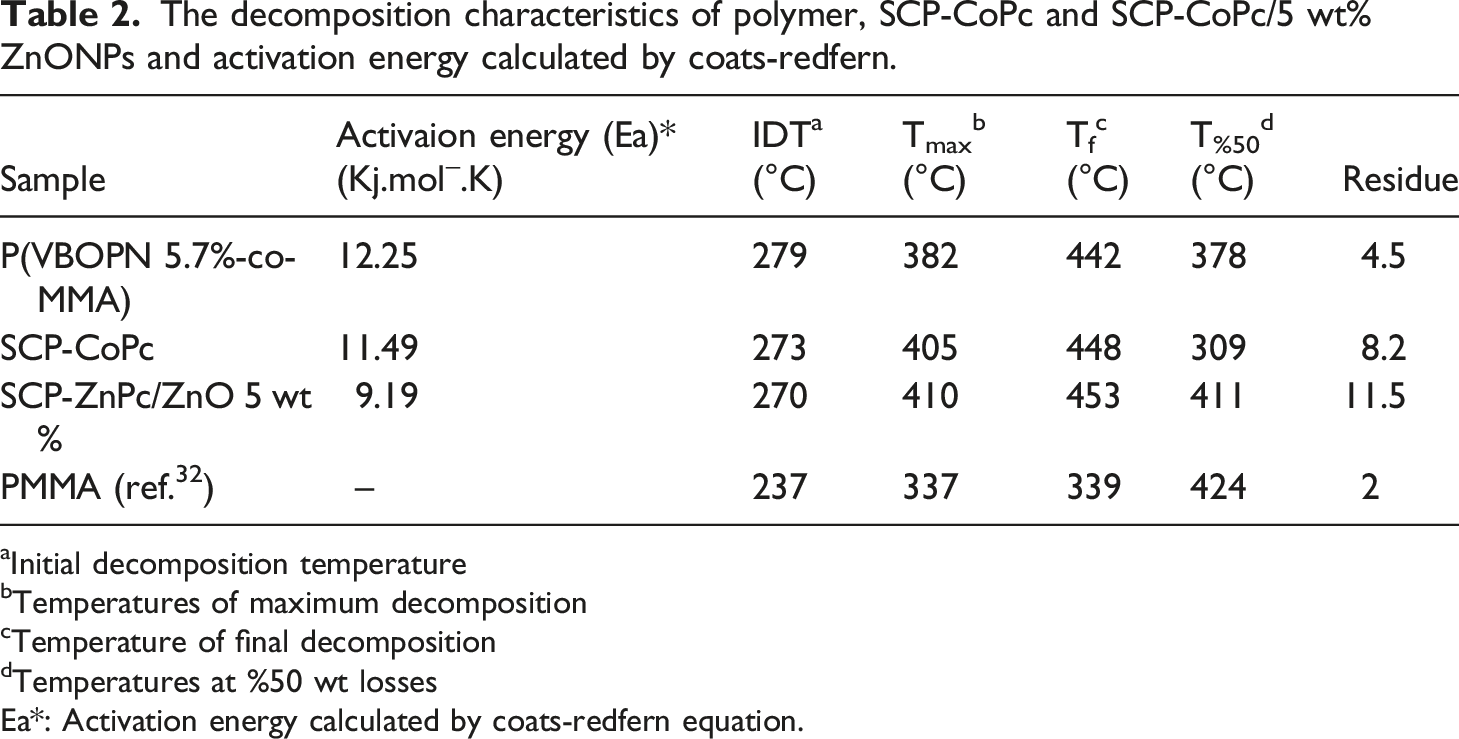
aInitial decomposition temperature
bTemperatures of maximum decomposition
cTemperature of final decomposition
dTemperatures at %50 wt losses
Ea*: Activation energy calculated by coats-redfern equation.
Dielectrical measurements of materials
Dielectric measurements are often used to investigate the electrical characteristics of materials such as polymers and metallophthalocyanine (MPc) compounds. These measurements offer important insights into the materials' charge transport, polarization behavior, and dielectric response.
The dielectric properties of a standard material can be characterized by various parameters, including the dielectric constant (ε'), the dielectric loss factor (ε''), and the ac conductivity (σac). To evaluate the dielectric behavior, capacitance measurement results are combined with other statistical measures. The complex permittivity can be expressed using the following equation
38
:
The real part of the complex permittivity, denoted as ε', represents the dielectric constant of the material, while the imaginary part, denoted as ε'', reflects its energy dissipation characteristics. The electrical and dielectric properties are often described using the concept of complex permittivity (ε*). This complex permittivity comprises the dielectric constant (ε'), which measures the stored energy when an external electric field is applied. equation (4) provides a method to calculate the dielectric constant (ε') at different temperatures, using capacitance values measured in the strong accumulation region. This calculation is based on the relation form,
39
which is further clarified as follows:

From assimilating all above equations, it allow to determine the dielectric properties of single-chain polymer cobalt phthalocyanines and its zinc oxide nanocomposites
Figure 9 Plots of the variation of absorbance (A), Transmittance (T%), reflectance (R), and extinction coefficient (k) versus wavelength of SCP-CoPc and SCP-CoPc/ZnO 5 wt%.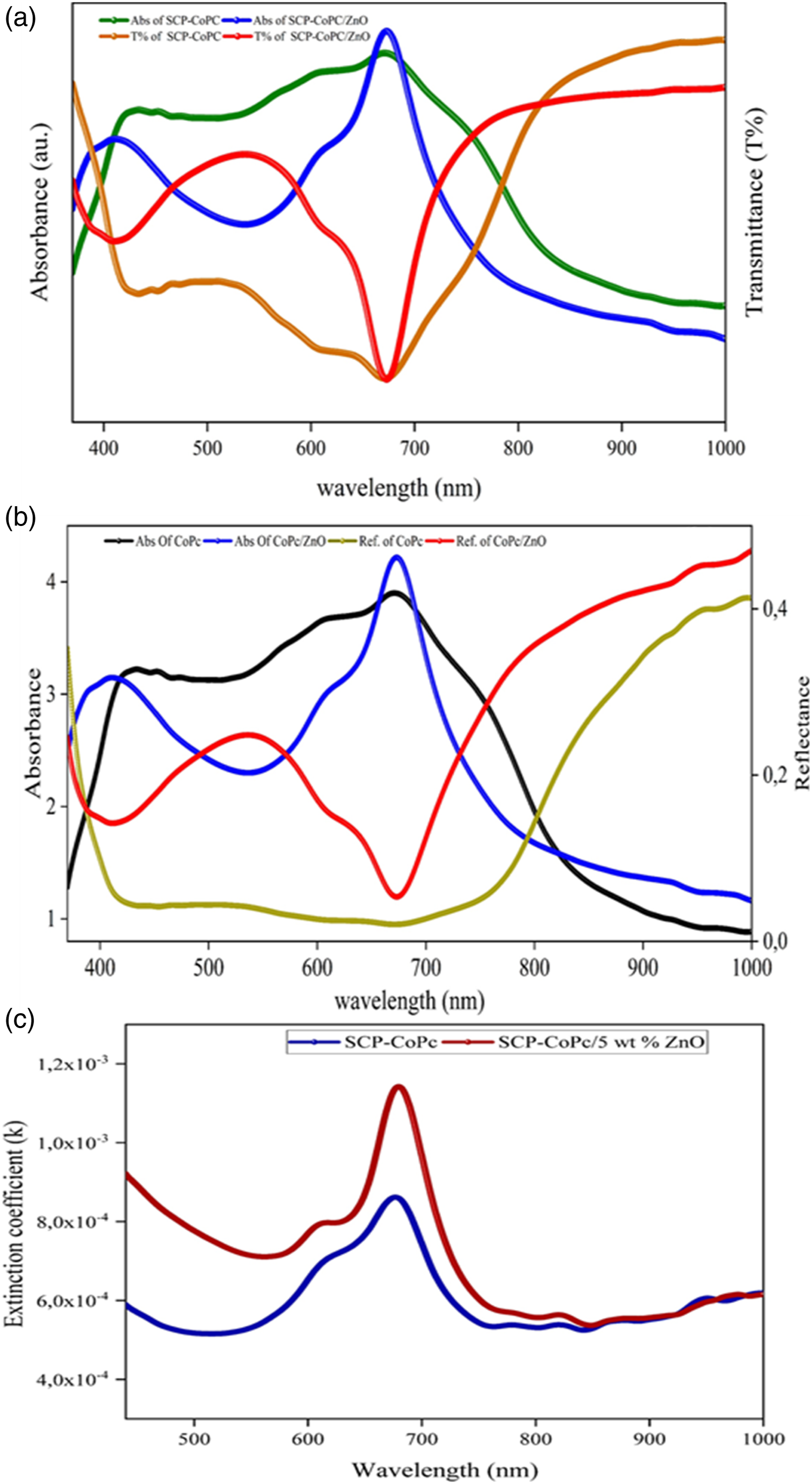
Figure 7 illustrates the effects of adding zinc oxide (ZnO) nanoparticle filler to the single chain cobalt complexed polymer (SCP-CoPc). The plot includes values of ε' (real part of complex permittivity), ε'' (imaginary part of complex permittivity), and Tanδ (dielectric loss factor) for both SCP-CoPc and SCP-CoPc/ZnO 5 wt% composite. The measurements were taken across frequencies ranging from 100 Hz to 6 kHz and at different set temperatures. The dielectric parameters exhibit a rapid drop-off in values as the frequency increases below 1 kHz. This behavior can be attributed to the combined effects of the Maxwell-Wagner effect “alternating current in phase with the applied potential” and the increased mobility of charge carriers at lower frequencies. It is essential to evaluate the ε' and ε'' values of the pure SCP-CoPc material before analyzing the composite systems. Figure 6(a) displays the ε' value of SCP-CoPc at 1 kHz for varying temperatures (42, 45, 95, and 140°C), while Figure 8(d) shows the corresponding values for SCP-CoPc/ZnO 5 wt%. Figure 6(b) and (c) represent the SCP-CoPc, while Figure 7(e) and (f) depict the SCP-CoPc/ZnO nanocomposite systems. Notably, the dielectric loss factor (ε'') and relative permittivity tan values are significantly lower for the pure SCP-CoPc material. All three measures of relative permittivity (ε', ε'', and tan) show a slight increase with rising temperature. This can be attributed to the alignment of dipoles in the direction of the electric field, as the free volume expands and the rigid structure of the material relaxes. Consequently, molecular expansion occurs, leading to an increase in electronic and interfacial polarization. This phenomenon results in higher ε' and ε'' values for the dielectric material.41–44 Plots of (a)Refractive index (n), (b)optical dielectric real (εr), (c) reflectance % (R%) and (d) optical conductivity(σ) of real part versus wavelength of SCP-CoPc and SCP-CoPc/ZnO 5 wt%.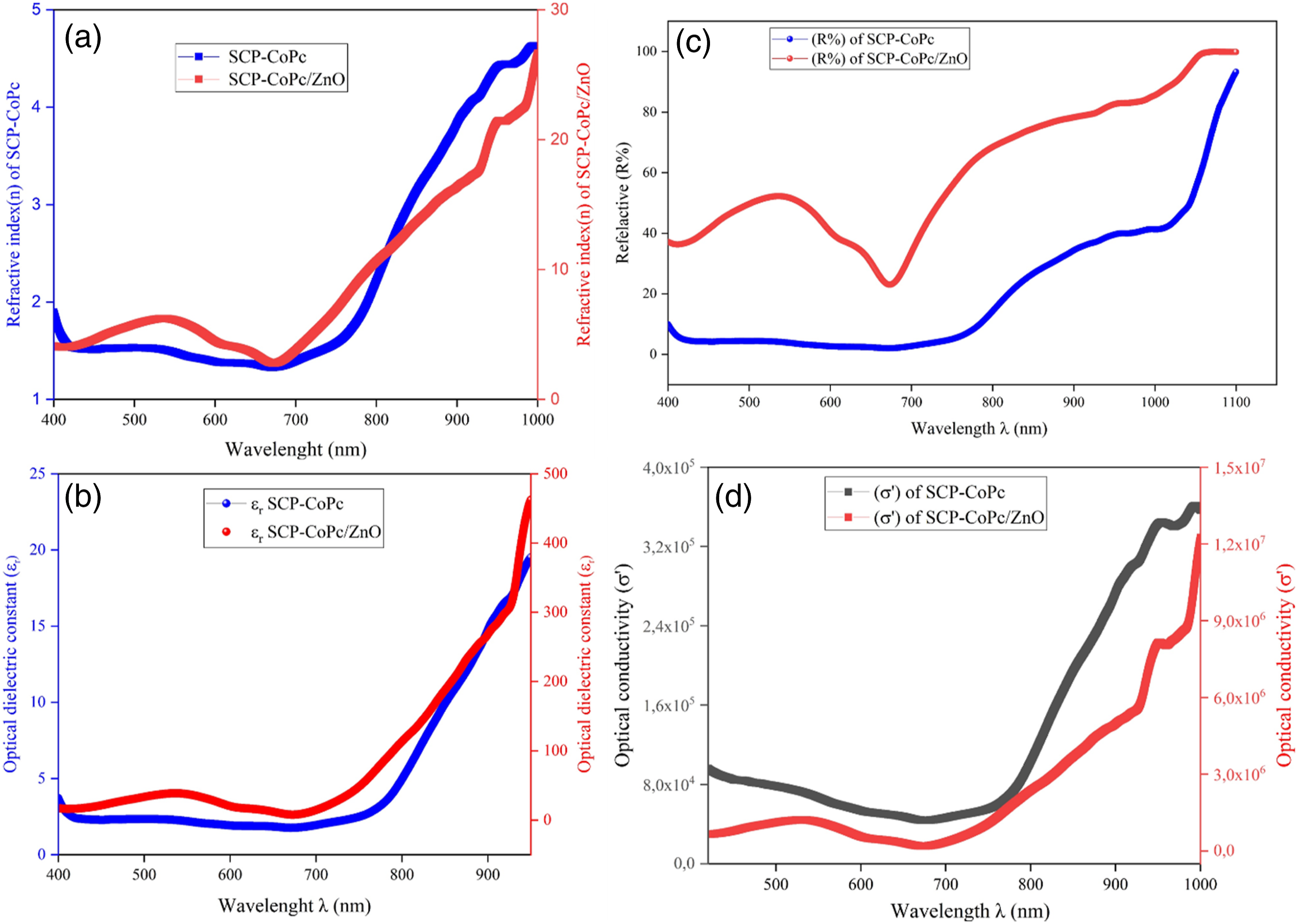
On the contrary, the dielectric properties of SCP-CoPc were significantly enhanced when combined with ZnO nanoparticles at a concentration of 5 wt%. This improvement was evident in the dielectric constant, as shown in Figure 7(d). The calculated dielectric constant of the nanocomposite, regardless of temperature, ranged from 12 to 20 at 1 kHz. The interaction between SCP-CoPc chains and ZnO nanoparticles increased the number of dipoles aligned in parallel within the composite. The distribution of conductive fillers such as ZnO has an impact on the dielectric performance of the material due to the migration and deposition of charge carriers at the interfaces between the matrix and the fillers. Figure 8 illustrates the frequency dependence of the dielectric loss factor (ε'') in the nanocomposite. As expected, the nanocomposite with 5 wt% ZnO exhibited a significantly higher ε'' compared to pure SCP-CoPc. Specifically, at 413 K and 1 kHz, the SCP-CoPc/ZnO 5 wt% nanocomposite had a value of 400. This substantial increase in dielectric loss can be attributed to the improved dispersion of ZnO nanoparticles within the composite and the stronger connections both between and within ZnO nanoparticle clusters. Dielectric constant plots of materials may not overlap at higher frequencies due to distinct polarization mechanisms, relaxation processes, ionic conductivities, intermolecular interactions, dispersive behavior, and boundary effects. These factors result in diverse responses to applied electric fields, causing variations in dielectric constant behavior among materials as frequency increases. Due to our material consist of saturated chain of polymer as well as the maycrocyle of CoPc. Various atom present in the compound led behaved irregularly with frequency. 45
The polarizability of SCP-CoPc and the SCP-CoPc/5 wt% ZnO nanocomposite has been thoroughly investigated, and an analysis of their alternative conductivity concerning applied angular frequency (ω) has been conducted using Jonscher’s power law. This analysis is effectively captured by equation (6):30,31,46
Figure 8 illustrates the variation of Jonscher’s power law with respect to the applied angular frequency for both SCP-CoPc and SCP-CoPc/5 wt% ZnO at a constant temperature of 140°C. The parameters associated with this law, such as the degree of correlation between mobile ions and the lattice (s) and the strength of polarizability (A), are determined by performing linear curve fitting on the aforementioned plots, considering the slope and intercept as key indicators. The strength of polarizabilities (A) for both SCP-CoPc and SCP-CoPc/5 wt% ZnO aligns with the findings from the dielectric analysis. According to Jonscher's equation, when s is less than or equal to 1, 45 the result implies a rapid hopping motion attributed to translational movement. Typically, in the context of ionic conductors, the s value can range between 1 and 0.5, indicating the presence of optimal long-range pathways and diffusion-constrained hopping. 47 In our investigation, we have determined that the degree of correlation between the lattice and mobile ions (s) of SCP-CoPc is one and SCP-CoPc/5 wt% ZnO is 0.78. This result aligns hopping as the dominant mechanism. 39 The value of σac and σdc for SCP-CoPc and SCP- are (1.45x10−10, 8x10−14) S/cm while for CoPc/5 wt% ZnO are (6, 64 × 10−7 and 7.8 × 10−11) S/cm.
Optical characterization of SCP-CoPc and SCP-CoPc/5 wt %ZnO nanocomposite
The refractive index (n) plays a crucial role in the practical application of optical materials across various industries. It is an important characteristic that defines the optical properties of solid substances, such as pure polymers or composites. In this study, the optical properties of the SCP-CoPc and SCP-CoPc/ZnO 5 wt% nanocomposite tablets were examined using spectrophotometric reflectance (R) measurements within the 400-1000 nm wavelength range. Figure 9(a)–(c) display the variations of optical parameters (including absorbance, reflectance, and transmission, extinction coefficient (k)) with respect to wavelength. The SCP-CoPc film exhibited typical absorptions in the Q band, ranging from 603 to 670 nm, which were consistent with the exciton of electrons observed in the liquid form using UV-visible spectroscopy. The reflectance of SCP-CoPc and SCP-CoPc/ZnO decreased as the incident photon wavelength increased, stabilizing at around 700 nm. Subsequently, there was a rapid increase in reflectance with further wavelength increase. Similarly, the transmittance showed a progressive rise until 700 nm, followed by a sharp surge. These changes can be attributed to the increase in packing density resulting from the intermolecular interaction between cobalt ions and SCP, SCP-CoPc, and SCP-CoPc with ZnO particles. The extinction coefficient of SCP-CoPc/5 wt% ZnO surpasses that of SCP-CoPc across all wavelengths. This occurs because the incorporation of ZnO in SCP-CoPc/5 wt% ZnO elevates the material's refractive index. As a result, the material reflects a greater amount of light, leading to a reduction in the quantity of absorbed light. This behaviors also observed in refractive index of both materials. (a) and (b) dc conductivity versus temperature for SCP-CoPc and SCP-CoPc/ZnO 5 wt% nanocomposite, (c) ac conductivity versus frequency at four different temperatures of SCP-CoPc/ZnO 5 wt% and (d) Voltage-current (V-I) of the SCP-CoPc/ZnO 5 wt% composite showing the dependence of dc current on the applied potential at 25°C.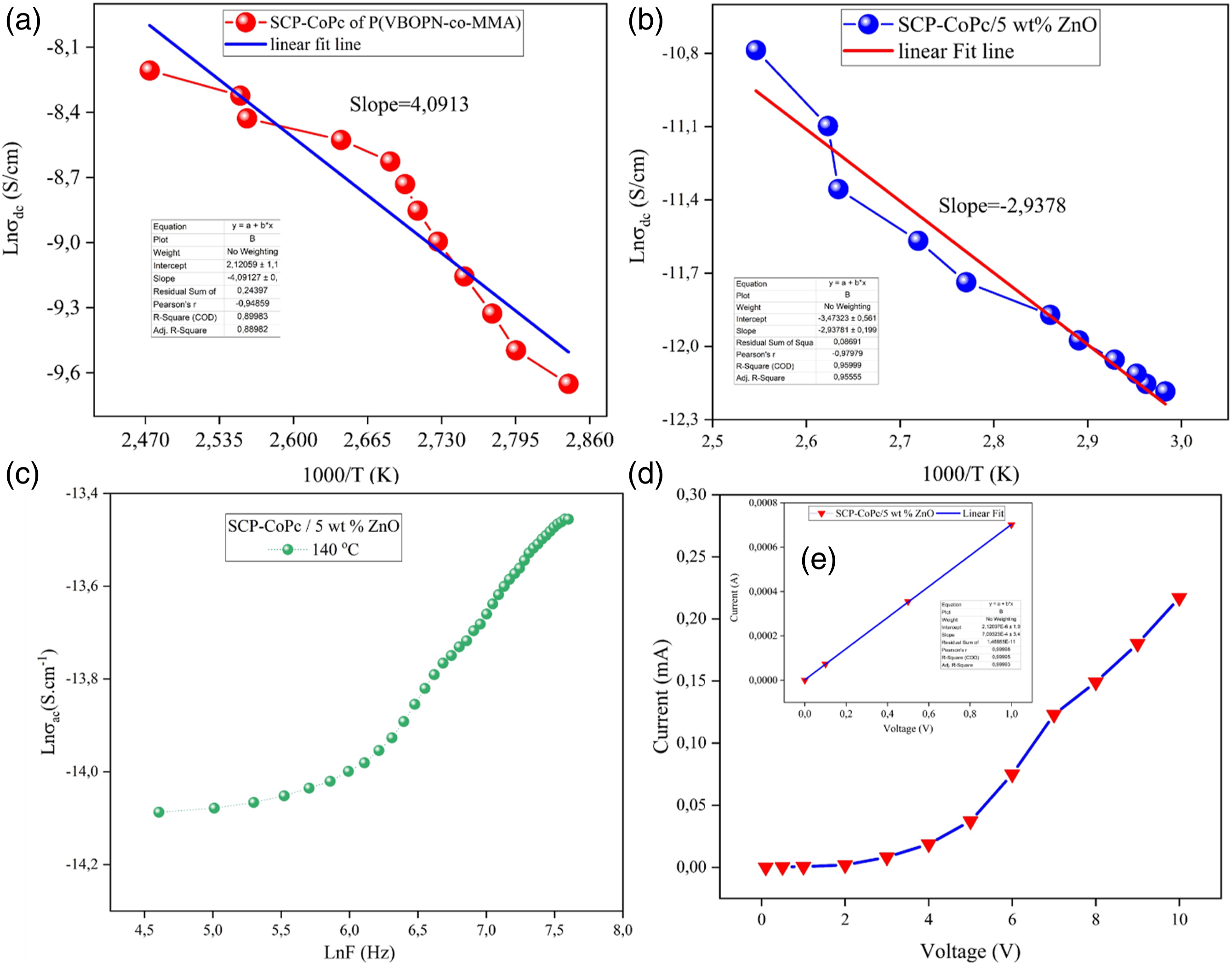
Studying changes in optical properties, such as refractive index (n) and dielectric constant, is crucial for understanding the density of absorbing centers, including impurity absorption, excitation transition, and defects in the crystal lattice. This investigation is particularly valuable during the development of samples. To estimate complex optical properties like permittivity and conductivity, the following equations can be employed.10,48

Optical parameters include (n, εr, R%, and σ) of SCP-CoPc and SCP-CoPc/ZnO 5 wt% versus wavelength have been plotted in (Figure 10(a)–(d) The dependence of 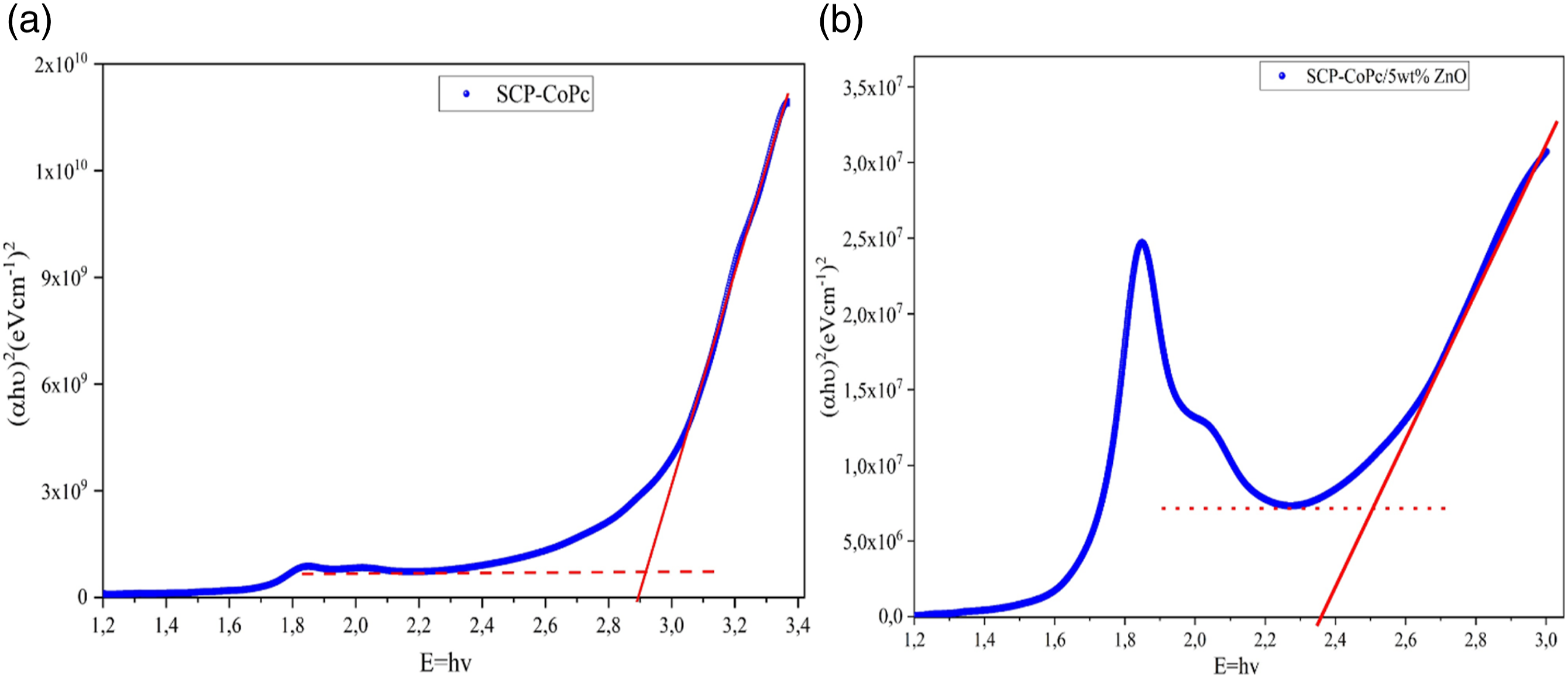
Electrical characterization of polymer nanocomposite
Figure 11(d) depicts the relationship between current and voltage for the SCP-CoPc/ZnO 5 wt% composite at room temperature. This composite exhibits semiconductor behavior across a wide temperature range. The potential difference (V) in this study is directly proportional to the applied current (A) and the 5 wt% ZnONPs content of the polymer matrix. Also, the resistance have been calculated for SCP-CoPc/5 wt% ZnONPs by linear curve and the resistivity of nanocomposites is determined by equation (9) as follows (a) Urbach tail of SCP-CoPc (b) Urbach tail of SCP-CoPc/5 wt% ZnO.
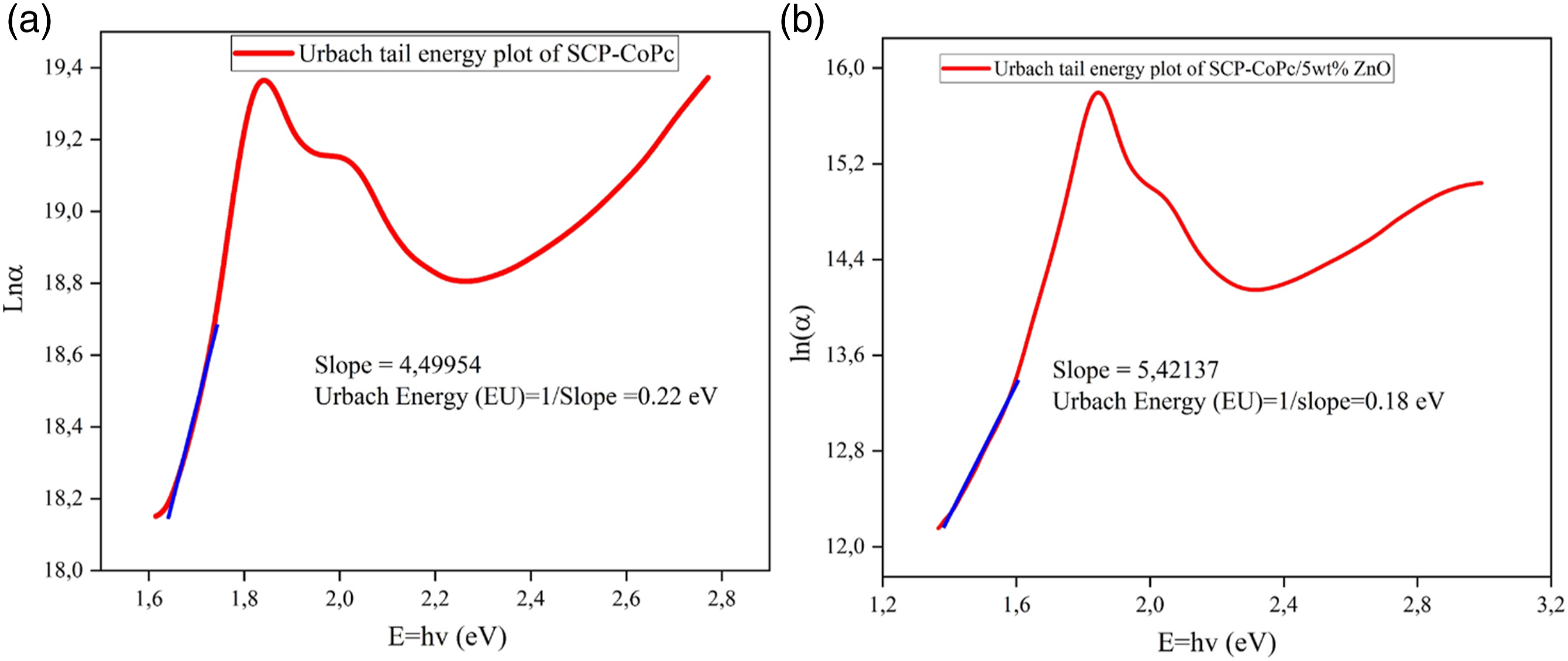
Figure 9(c) illustrates the frequency (lnF)-dependent ac conductivity (lnac) for the SCP-CoPc/ZnO 5 wt% composite. At 140°C, polarization align with range 0-1 that is 0.38 for the SCP-CoPc/ZnO 5 wt% composite. 39 The connection between dc conductivity and temperature is shown in Figure 11(a) and (b), spanning a temperature range of 363 K to 413 K. Both SCP-CoPc and nanostructured SCP-CoPc/ZnO 5 wt% exhibit an Arrhenius plot of lnσdc versus 1000/T. Figure 13(b) demonstrates that the nanocomposites have higher dc conductivity values compared to the SCP-CoPc, as determined by equation (6). The concentration of ZnONPs directly affects the dc conductivity of pure SCP-CoPc, resulting in increased conductivity. Different sections of the composite's conductivity profile are associated with different conduction mechanisms. When the temperature of the material rises, more charge carriers can overcome the activation energy barrier and participate in the conduction process, leading to enhanced electrical conductivity.
The Arrhenius equation (equation (10)) illustrates the correlation between temperature and direct current electrical conductivity (σdc), as shown in reference
45
: Refractive index dispersion plot of (a) SCP-CoPc and (b) SCP-CoPc/5 wt% ZnONPs using Wemple–DiDomenico model, (c) SCP-CoPc and (b) SCP-CoPc/5 wt% ZnONPs using Sellmeier model.
The discovery of varying activation energies within the temperature range of 363 K to 413 K suggests the presence of a donor level in the system. Figure 12(a) and (b) depict the activation energy of both SCP-CoPc and SCP-CoPc/ZnO 5 wt% nanocomposite. The temperature falls within the range of the composite's glass transition temperature (Tg). It has been observed that as the Tg temperature of a polymer is approached, the free volume also increases. The increased conductivity at higher temperatures can be attributed to the expansion of the composite caused by temperature, potentially leading to an increase in free volume. This facilitates the movement of charge carriers into the sample volume, resulting in higher current and enhanced conductivity with rising temperature. As a consequence, the activation energy is reduced, as less work is required for charge carriers to move between locations due to the larger free volume. 30 However, as the temperature continues to rise, the majority of charge carriers participate in the conduction of electricity by surpassing the activation energy barrier. 49
The term “band gap" refers to the difference in energy levels between the highest occupied energy level, known as the valence band, and the lowest unoccupied energy level, known as the conduction band, within the electronic band structure of a specific material. It plays a crucial role in determining the electrical and optical behavior of a substance. Through the analysis of photonic absorption values obtained from UV-visible spectrum tests, it is possible to determine the optical band gaps corresponding to direct transitions. The concept of a direct energy gap describes the interaction between the energy of a photon and an electron located in the valence band. This interaction facilitates the transition of an electron from the valence band to the conduction band. equation (11) is employed to determine the magnitude of this transition.
48

The Urbach energy (Eu) is a crucial concept in solid-state physics and materials science. It quantifies the width of the tail of localized states within the band gap of semiconductors or insulators, determining the rate at which the absorption of photons increases near the band gap energy. This parameter, usually expressed in (eV), offers valuable insights into the distribution and density of these localized states, making it a fundamental tool for understanding and characterizing the optical properties of these materials. Crystalline materials are exhibiting broad Urbach tail at the absorption edge due to coupling of excitons and phonons.
30
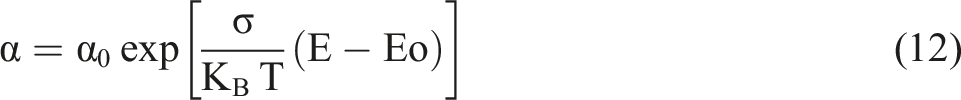
Practically, using the absorption coefficient (a) and the absolute temperature (T), the Urbach energy (Eu) and the onset of exponential absorption tail (E0) can be deduced using the following empirical relation
30
:

Taking natural logarithm on both sides to the above equation.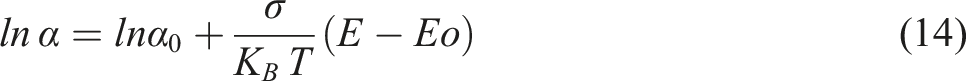
Figure 13(a) and (b) represents the Urbach tails of SCP-CoPc and SCP-CoPc/5 wt% ZnO, respectively. Here, we have taken straight line fitting to deduce the slope. The reciprocal of obtained slopes gives rise to the Urbach energies for SCP-CoPc and SCP-CoPc/5 wt% ZnO. From the slope Eu of the SCP-CoPc and SCP-CoPc/5 wt% ZnO are given as 0.22 eV and 0.18 eV respectively. Conversion of precursor copolymer to SCP-CoPc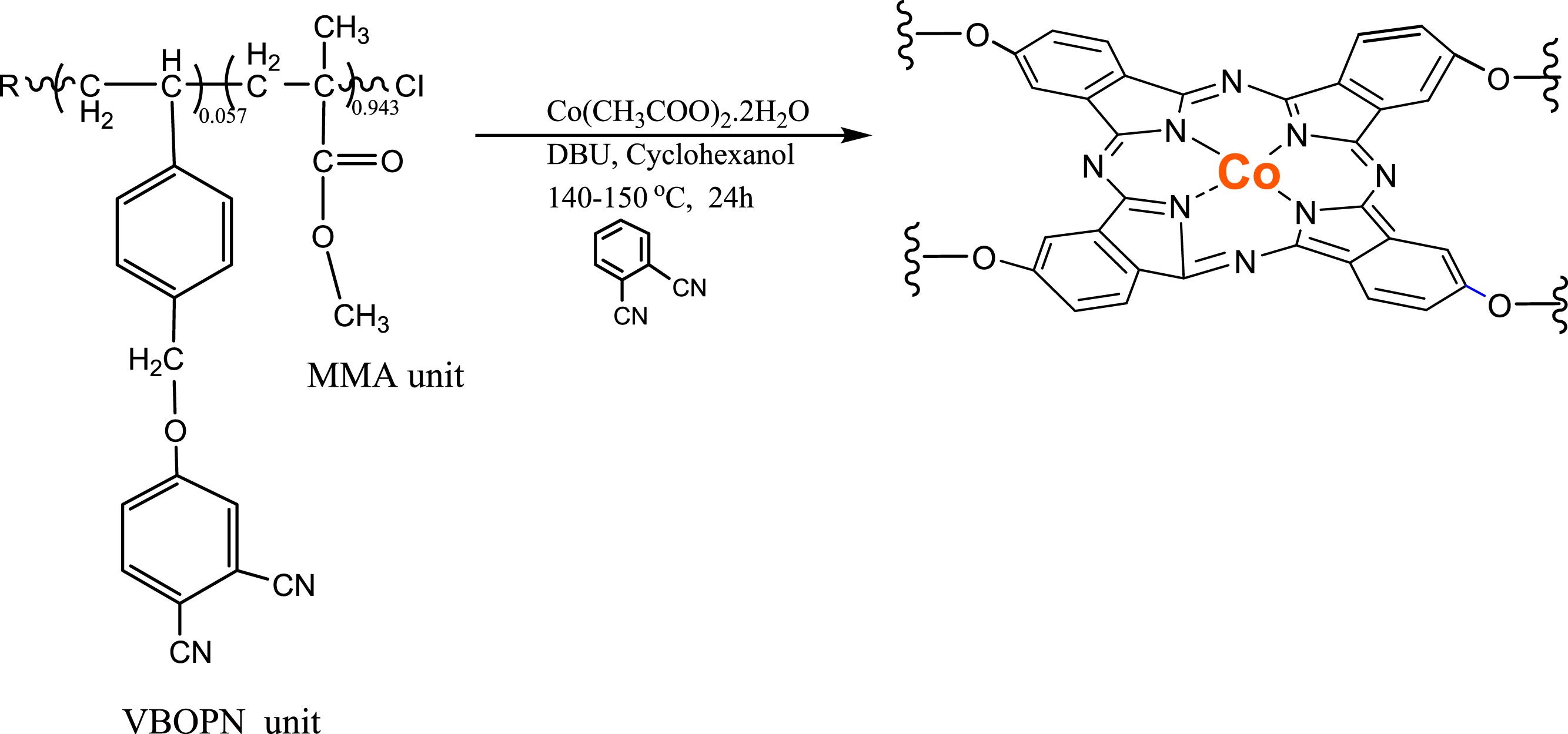
Refractive index dispersion
To examine how the refractive index behaves below the energy band gap in SCP-CoPc and SCP-CoPc/5 wt% ZnO, we utilize the Wemple-DiDomenico Single Oscillator model, a concept introduced by Wemple and DiDomenico in 1971.
50
This model consists of inter band transition of electrons as a single oscillator within the brillouin zone, which behaves as an individual oscillator and recognizes that the valence electrons of atoms contribute to one such oscillator. The relationship between the refractive index and photon energy below the inter band absorption edge (hv/Eg) is defined as.
46

In Figure 14(a) and (b), we illustrate the refractive index dispersion below the energy band gap for SCP-CoPc and SCP-CoPc/5 wt% ZnO. The determination of the single oscillator energy (Eo) and the dispersion energy (Ed.) involves fitting the experimental data with straight lines, as outlined in the expressions below: The formation of single chain polymer bearing cobalt phthalocyanines reprint from.
10
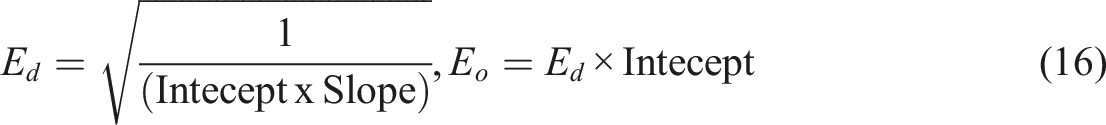
The notable trend here is the decrease in dispersion parameters, particularly the single oscillator energy (E0). Generally, the single oscillator energy is often considered synonymous with cohesive energy. 21 Therefore, SCP-CoPc/5 wt% ZnO displays a lower value for this parameter compared to pure SCP-CoPc. Single oscillator energy (Eo) and the dispersion energy (Ed.) for SCP-CoPc are 3.27 eV and 0.68 eV and for SCP-CoPc/5 wt% ZnO are 2.01 eV and 1.01 eV respectively. Single oscillator energy (Eo) are in approximate with band gap energy.
Sellmeier dispersion relation
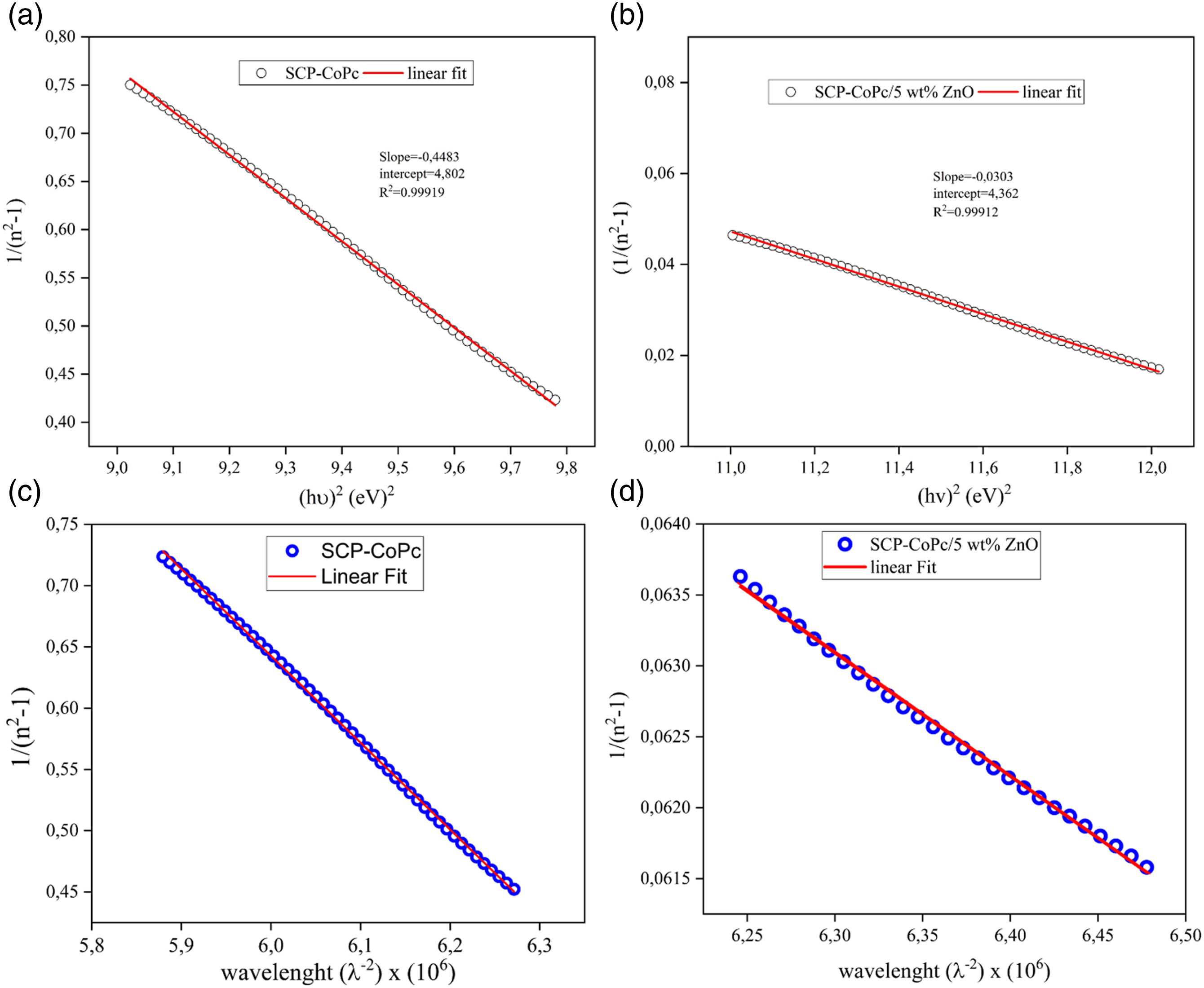
A mathematical model, known as the Sellmeier dispersion relation, is used to characterize how the refractive index of a material changes as a function of the wavelength of light passing through it. It is especially helpful in the fields of optics and photonics for studying the interaction of light with various materials. It can be expressed as.
30


The dispersion parameters such as S0, n0 and λ0 are obtained from the slope and intercept of (n2-1)−1 versus λ−2 plots for SCP-CoPc and SCP-CoPc/5 wt% ZnONPs. Figure 14(c) and (d) shows the Sellmeier plots for SCP-CoPc and SCP-CoPc/5 wt% ZnONPs. The obtained parameters S0, n0 and λ0 are 0.709, 4,898 and 6,906 belong to the SCP-CoPc correspondingly. In addition, the dispersion parameters for SCP-CoPc/5 wt% ZnONPs such S0, n0 and λ0 are 0.0087, 0.118 and 13.55 respectively. From this one can observe that on SCP-CoPc/5 wt% ZnONPs, the dispersive parameters are decreasing. The material exhibiting the lowest average oscillator strength (S0) indicates the highest level of ZnONPs integration within SCP-CoPc. Which the This assertion is supported by the chemical bonding principle described in reference, 23 which states that atoms are more likely to form favorable bonds when they are of various types as opposed to when they are of the same type. In the process of bonding, bond energies diminish until all available valences are filled up.
Conclusion
The formation of CoPc rings on a single chain of PMMA led to the development of a new green-colored variant of PMMA. This discovery is of great importance to polymer chemists as it demonstrates the dye-like characteristics of PMMA, with notable absorption at 603 and 670 nm in the UV-Vis region. The presence of cobalt phthalocyanine in single-chain polymers resulted in their solubility in various organic solvents, confirming that the prepared SCP-CoPc was a soluble polymer without cross-linked chains. Furthermore, the addition of ZnO nanoparticles to the SCP-CoPc enhanced its thermal stability, as evidenced by thermogravimetric analysis. The nanocomposite remained stable even at high temperatures, exhibiting a higher glass transition temperature (Tg) compared to pure SCP-CoPc and the copolymer. The dielectric measurements demonstrated frequency-dependent patterns, with the real dielectric constant (ε') decreasing with frequency due to the higher NG content causing interface polarization. The optical properties analysis revealed that both SCP-CoPc and SCP-CoPc/ZnO nanocomposite displayed semiconducting properties, as indicated by their optical band gaps and optical constants (refractive index, real part of optical dielectric constant). The DC conductivity measurements yielded activation energy values lower than the energies of the optical band gaps, suggesting the potential for new types of capacitance in technological devices through the incorporation of zinc oxide nanoparticles, which reduced the forbidden energy.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
