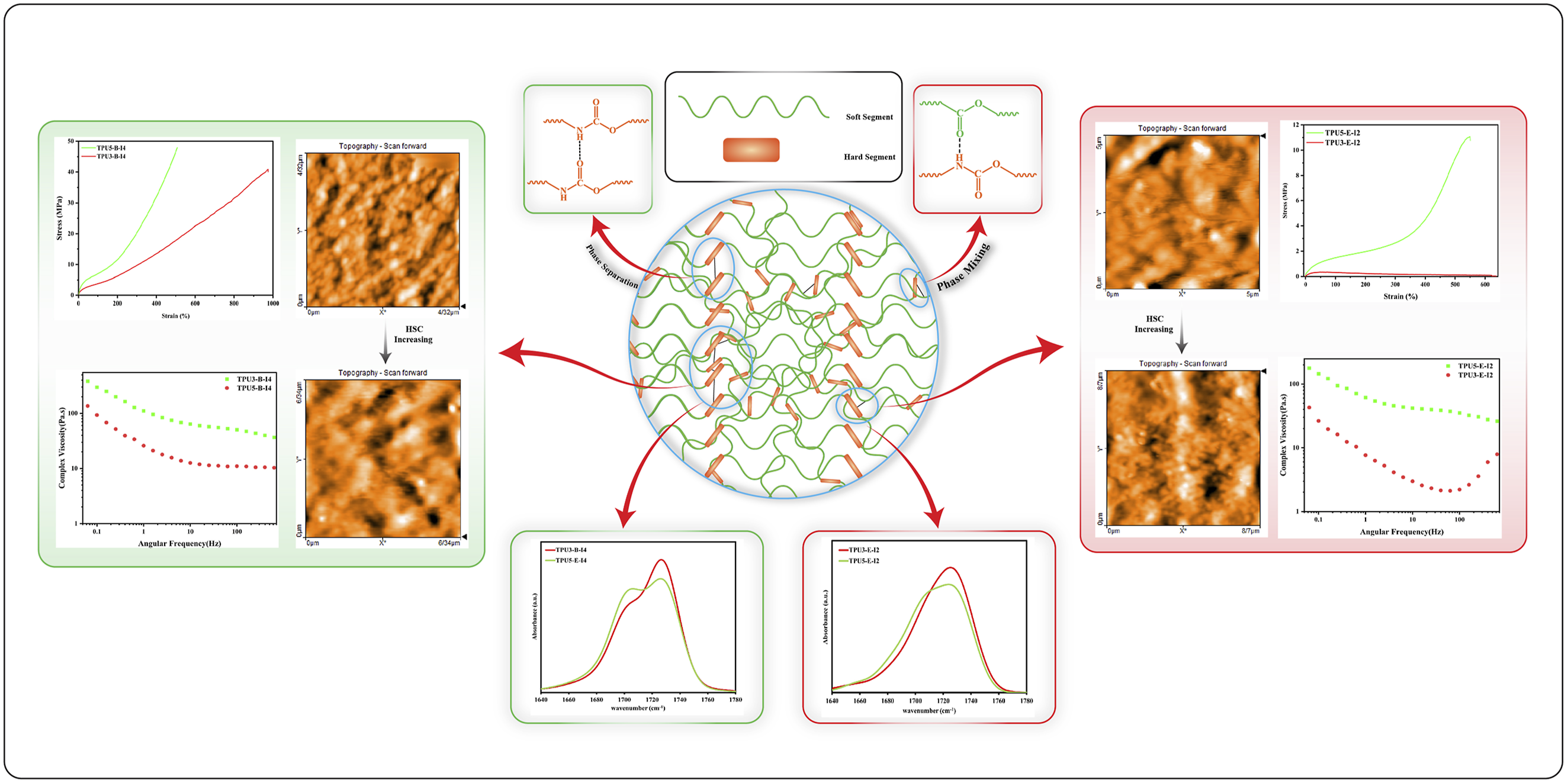
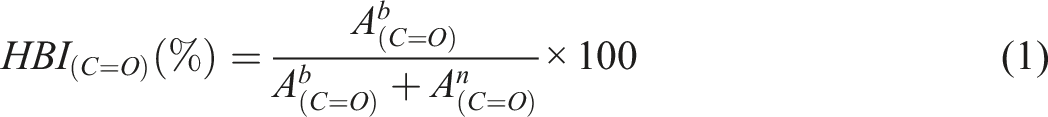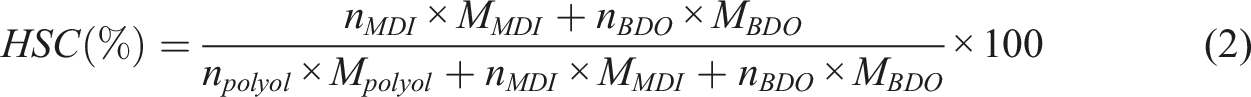Abstract
This research focuses on synthesizing thermoplastic polyurethane elastomers (TPUs) using a reactive mixing method in an internal mixer, aiming to overcome the viscosity-related challenges of traditional synthesis techniques. Two types of polyester polyols based on ethylene glycol (Polyester-E) and butane diol (Polyester-B) along with two diisocyanates (pure 4,4′-MDI and a mixture of 4,4′-MDI/2,4′-MDI), a butane diol (BDO) chain extender, and dibutyltin dilaurate (DBTDL) catalyst were used. The study examined one-step and two-step feeding methods to investigate how process parameters and reactant ratios affect TPU properties. Tensile testing and FTIR analysis revealed that higher hydrogen bonding index (HBI) improved mechanical strength. AFM images showed increased microphase separation with higher diisocyanate-to-polyol ratios, indicated by larger hard segment domains. DSC results confirmed reduced crystallinity and increased melting temperatures with higher hard segment content. Rheological analysis demonstrated that increasing hard segment content led to greater complex viscosity and storage modulus, suggesting improved rigidity and phase separation.
Keywords
Introduction
Thermoplastic polyurethane (TPU) is a highly versatile polymer that can be used in various applications with a wide range of properties.1–4 Combining different types and ratios of isocyanates and polyols allows for controlling the desired final properties.5–7 Various polyols, such as polyester polyols, polycaprolactone polyols, and polyethylene glycol-based polyether polyols, are used as the soft segment, also various diisocyanates such as 2,4-diphenylmethane diisocyanate, (2,4′ MDI), 4,4-diphenylmethane diisocyanate (4,4′MDI), and hexamethylene diisocyanate (HDI) are used as the hard segment.8–10 Polyurethanes synthesized from polyester polyols exhibit appropriate heat resistance, high tensile strength, good elasticity, suitable wear resistance, excellent photostability, and high resistance to both mineral and organic acids.11–13 Different positions of NCO groups in diisocyanates’ molecules can affect their reactivity. In 4,4′ MDI, the two NCO groups are equivalent in reactivity, but in 2,4′ MDI, the two isocyanate groups exhibit very different reactivity. In different diisocyanates the NCO groups in 4,4′ positions have higher reactivity than the NCO groups in 2,4′ positions. 14 The most common methods for producing TPU are reactive mixing processes,15,16 including reactive extrusion (REX) and internal mixing.17–19 The internal mixers are used in thermoplastic polymers melt blending with different additives and fillers. These additives and fillers include thermal and ultra violet (UV) stabilizers, micro-fillers such as carbon black or calcium carbonate, 20 and nano-fillers like nanoclays, 21 nanosilica, carbon nanofibers, 22 and nanodiamonds. This method can also be used for rubber compounding and mastication of elastomers to control their elasticity. 23
Parnell et al.
24
used a new reactive blending process to prepare poly (vinyl chloride)/thermoplastic polyurethane (PVC/TPU) blends. They used an internal mixer to blend PVC, polyester-based polyol, and a chain extender with a specified stoichiometric ratio of diisocyanate to prepare polyurethane. The results indicated that the blending these two polymers enhanced their mechanical properties. Youssefzadeh et al.
25
prepared two types of polymer nanocomposite blends based on poly (
To enhance the reaction rate and achieve more favorable mixing conditions, different catalysts are usually introduced during the mixing process in internal mixer.28–30 Thermoplastic polyurethanes are typically synthesized using two mixing routes including pre-polymerization and one-step polymerization methods.31–33 In the pre-polymerization method, the polyols and diisocyanates are mixed in the appropriate NCO/OH ratio, and the reaction proceeds under specific temperature and time conditions to yield the desired pre-polymer, then in the next step the chain extender is incorporated to the reaction mixture for producing the final polyurethane.34,35 Most thermoplastic polyurethanes are prepared using one-step bulk polymerization methods. These TPUs are industrially produced through reactive internal mixing, reaction injection molding, or reactive extrusion methods.27,36,37 In these processes, the chemical modification can lead to the changes in the polymer’s properties, including improved thermal stability and mechanical properties.38,39 The chain extender used to synthesize thermoplastic polyurethanes is typically a low molecular weight diol or diamine and, in some cases is water.40–42 The resulting polymer contains only urethane groups if the chain extender is a diol. 43 However, when a diamine chain extender is used, it also leads to the formation of urea groups, and these polymers are named as polyurethane-urea.44–46
Thermoplastic polyurethanes (TPUs) are among the most important engineering polymers. The presence of hard and soft segments in their main structure, along with specific hydrogen bonding interactions between these segments, gives these materials outstanding engineering properties. Understanding the correlation between phase morphology and final properties in TPUs through atomic force microscopy (AFM) studies is essential, as their physico-mechanical properties and rheological behavior are directly determined by their complex morphology. The polyols in this study were based on two polyester polyols: one based on the adipic acid and ethylene glycol with an amorphous morphology and the other one based on adipic acid and butane-diol with a semi-crystalline microstructure. Furthermore, two diisocyanates with different reactivities were used, which include 4,4′-diphenylmethane diisocyanate (4,4′) with high reactivity and a mixed diisocyanate consisting of 4,4′-diphenylmethane diisocyanate and 2,4′-diphenylmethane diisocyanate (4,4′-2,4′) with lower reactivity. Thermoplastic polyurethanes with different physico-mechanical properties were synthesized by combining these materials in various reactant ratios. In this study, due to the increased viscosity during polymerization, thermoplastic polyurethane elastomers were synthesized using a reactive mixing method in an internal mixer. Moreover, TPUs were synthesized using one-step and two-step methods to examine the influence of feeding order on the resulting physico-mechanical properties and rheological behavior. Based on our knowledge, no appreciable comprehensive study has been carried out on the effect of the type and ratio of reactants on the phase morphology, physico-mechanical properties, rheological behavior of thermoplastic polyurethane elastomers, and the relationship between their chemical microstructure, morphology and final properties.
The significance and advantages of this research lie in investigating the effect of changing the type of polyol and isocyanate, as well as the reactant ratios in TPU synthesis, on phase morphology, final physico-mechanical properties, rheological behavior, and the relationship between microstructure and properties in these polymers, a topic that has received limited attention in previous studies.
Experimental
Materials
Two types of polyester polyols, one based on adipic acid and ethylene glycol (polyester-EG) and the other one based on adipic acid and butane diol (polyester-BDO), both with a molecular weight of 2000 g/mole, were prepared by Rasam Polymer Nami Co. (Iran). 1,4-Butane diol (BDO), with a molecular weight of 90 g/mol as a chain extender, and Dibutyltin dilaurate (DBTDL) catalyst were kindly provided by by Rasam Polymer Nami Co. (Iran). Two types of diisocyanates were used, 4,4′-diphenylmethane diisocyanate (4,4′ MDI) and a mixture of 4,4′-diphenylmethane diisocyanate and 2,4′-diphenylmethane diisocyanate (4,4′-2,4′ MDI), both with a molecular weight of 250 g/mol, provided by Rasam Polymer Nami Co. (Iran).
Characterization methods
The Fourier transform infrared (FTIR) spectra of the samples in absorption mode were recorded in the wave number range of 400–4000 cm−1 using a FTIR spectrophotometer (Bruker, Tensor 24, Germany) with a resolution of 4 cm−1.
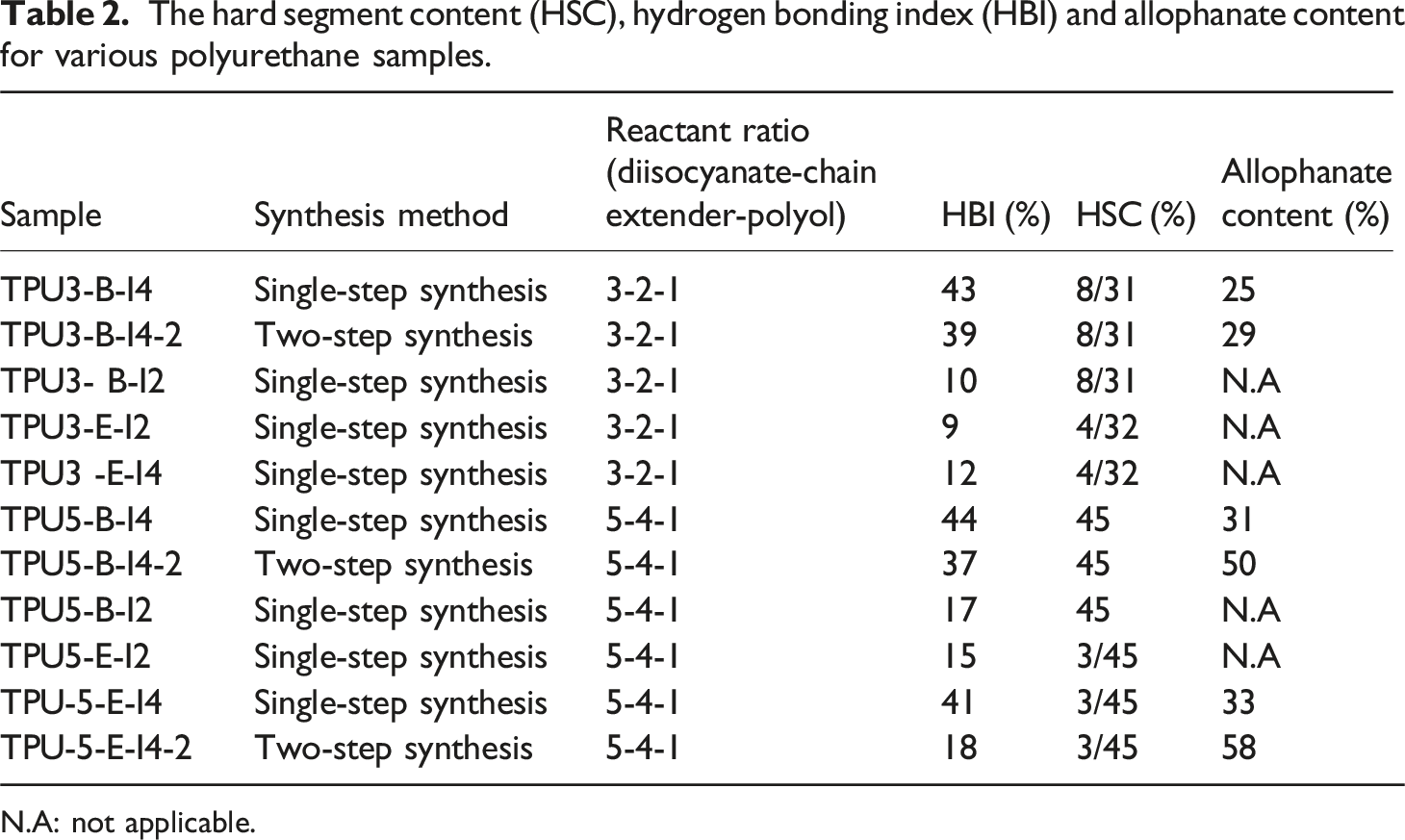
To determine the extent of hydrogen bonding in the synthesized polyurethanes, the hydrogen bonding index (HBI) was utilized. Two types of hydrogen bonds are formed in the structure of TPUs. Hydrogen bonding in polyurethanes mainly occurs between urethane groups from different chains. It can also take place between urethane linkage and ester groups of the soft segments or between urethane groups in the hard segments. Due to the overlapping of hydrogen bonding characteristic peaks in FTIR spectra for NH groups with C = O groups in the hard and soft segments of the polyurethane, calculating the hydrogen bonding index for NH groups (HBINH) is unfeasible. Therefore, the hydrogen bonding index for carbonyl groups (HBIC = O) was determined. This index is calculated based on the interaction of hydrogen in urethane NH groups with oxygen in the soft segments ester C = O groups or urethane carbonyl groups, by equation (1). In this equation,
To calculate the hard segment content (HSC) of various polyurethane samples, equation (2) was utilized, where n and M denote the molar ratio and the molecular weight of the different reactants in TPU synthesis, respectively.
48
The glass-transition temperature (Tg), melting point, and enthalpy of melting and crystallization of the synthesized samples were measured using a differential scanning calorimeter (DSC, Netzsch, F3 Maia, Germany) in the temperature range of −50–220°C, with the heating and cooling rates of 10°C/min under a nitrogen purge.
The tensile properties of the samples were examined based on ASTM D638 standard using a tensile testing machine (Zwick-Roell Z 010, Germany). The dumbbell-shaped samples were molded using a hot press machine (Brabender, Germany). The tensile tests were conducted at room temperature, with a strain rate of 10 mm/min, and all the tests were repeated at least three times. The rheological behavior of the prepared polyurethane samples was evaluated using a dynamic rheometer (MCR 301, Anton Paar, Austria) under the nitrogen atmosphere. A parallel plate geometry with a diameter of 25 mm was selected for the rheological examinations. A strain amplitude of 1% was chosen for the frequency sweep tests at a constant temperature to ensure that the measurements remained within the linear viscoelastic region. The samples were examined at 210°C temperature in the frequency ranges from 0.01 to 625 Hz.
To investigate the relationship between morphology and final properties in TPUs, and also to examine the phase morphology and quantify the phase separation between hard and soft segments, an atomic force microscopy (AFM) test (Nanosurf Mobile-S, Switzerland) was conducted.
TPU synthesis
The synthesis of TPU samples with different polyols and diisocyanates, along with DBTDL catalyst and BDO chain extender, was carried out using one-step and two-step feeding methods with various molar ratios of reactants in a Brabender internal mixer at a rotor speed of 80 r/min and a temperature of 90°C. In one step process, both polyester polyols based on ethylene glycol and butane diol was separately mixed with the respective diisocyanates include 4,4′ MDI or 4,4′-2,4′ mixed MDI along with the catalyst and butanediol as a chain extender. All the components were simultaneously fed into the internal mixer. The process continued until the internal mixer torque was kept a constant value, indicating the complete mixing and reaction of the materials. In two-step process the corresponding polyols and diisocyanates were initially mixed in the first step, then DBTDL catalyst and BDO chain extender were added to the mixture and let the reaction to continue for preparing final TPU samples.
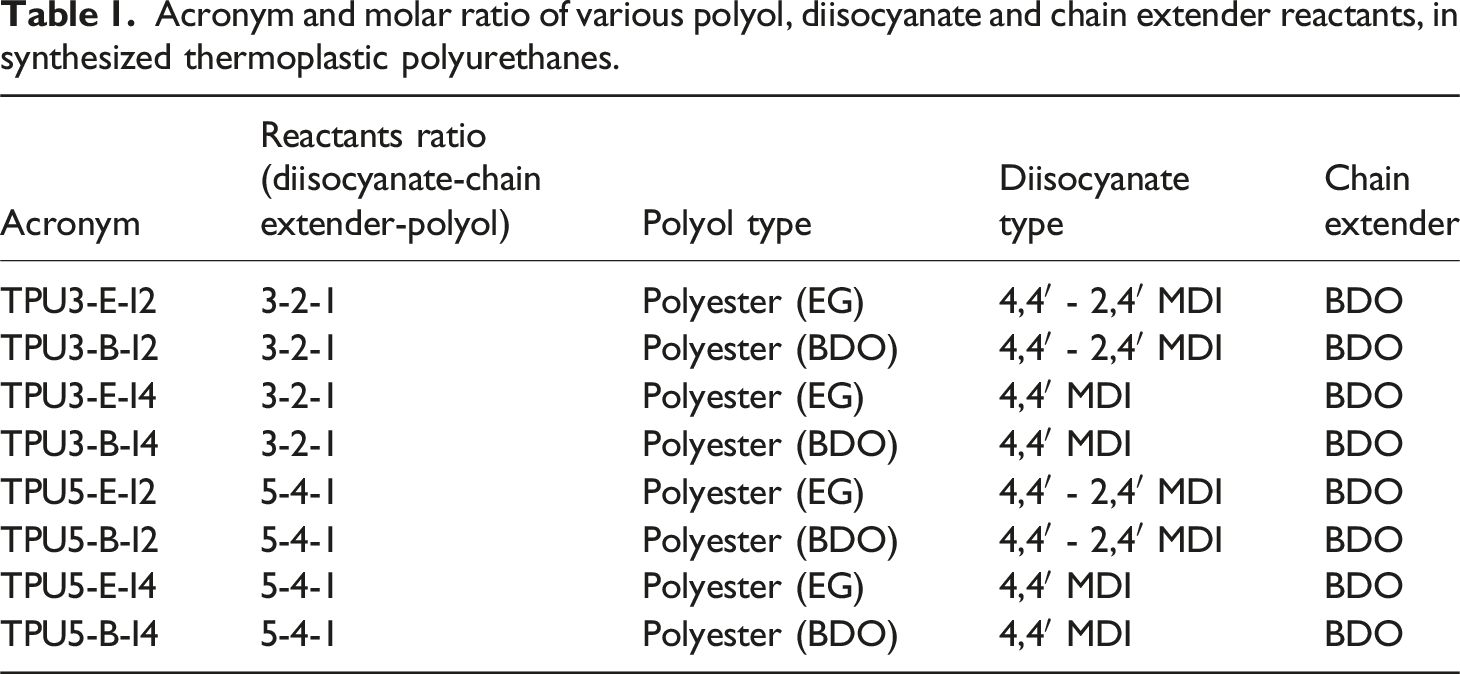
In this study, various polyurethane samples were prepared using the reactive mixing method with diisocyante-BDO-polyol molar ratios of 3-2-1 and 5-4-1. The main objective was the investigation of the effects of change in the polyol and diisocyanate type, and the reactant ratio variations on the final properties of the synthesized TPU (see Scheme 1). After removing of the synthesized polymers from internal mixer, all samples were placed in a vacuum oven at 120°C for 24 h to complete their polymerization process namely as post-curing step. The acronyms of the synthesized polyurethanes with different reactant ratios are presented in Table 1. A schematic view from synthesis of thermoplastic polyurethanes containing various reactants type and ratio in an internal mixer. Acronym and molar ratio of various polyol, diisocyanate and chain extender reactants, in synthesized thermoplastic polyurethanes.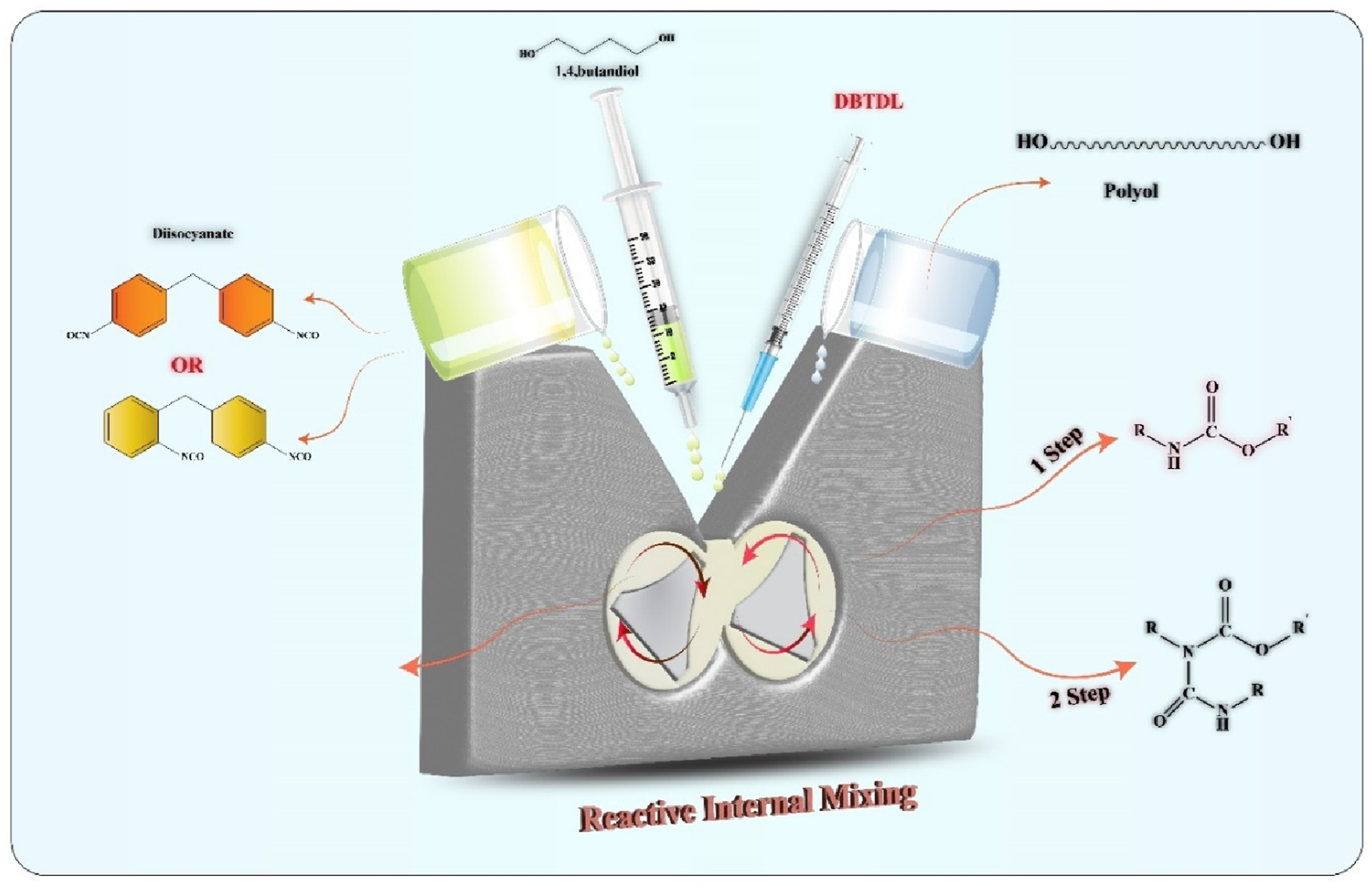
Results and discussion
FTIR spectroscopy
Figures 1 and 2 show the results of the FTIR analysis of the raw materials and the various synthesized polyurethane samples. In Figure 1, the FTIR spectra of different polyester polyols, along with the chain extender butane diol, are presented. In the spectrum of the polyester polyol based on ethylene glycol (Polyester-E), the broad band observed at a wave number of 3420 cm−1 indicates the presence of hydroxyl (OH) groups. The absorption band at 1731 cm−1 is attributed to the carbonyl (C = O) groups of the ester bonds, while the peak at 2917 cm−1 corresponds to the asymmetric stretching of CH2 groups in adipic acid. In the spectrum of the polyester polyol based on butane diol (BDO), the absorption band at 2925 cm−1 is related to the stretching of adipic acid; also, the peaks at 1731 cm−1 and 3345 cm−1 correspond to the carbonyl and hydroxyl groups, respectively. Figure 1(b) shows the spectra of the two types of diisocyanates used in the preparation of the polyurethane samples. The first diisocyanate is of the 4,4′-diphenylmethane diisocyanate (4,4′-MDI) (type I4), characterized by a distinct peak at 2288 cm−1, indicating the presence of the isocyanate group. The second type is a mixture of the 4,4′ and 2,4′ isomers of diphenylmethane diisocyanate (4,4′-MDI and 2,4′-MDI) (type I2), which also exhibits a characteristic peak at 2271 cm−1. Once these diisocyanates react with the polyol and chain extender to form thermoplastic polyurethanes, the characteristic isocyanate peak in this region disappears due to the reaction between the NCO groups of the isocyanates and the OH groups.
49
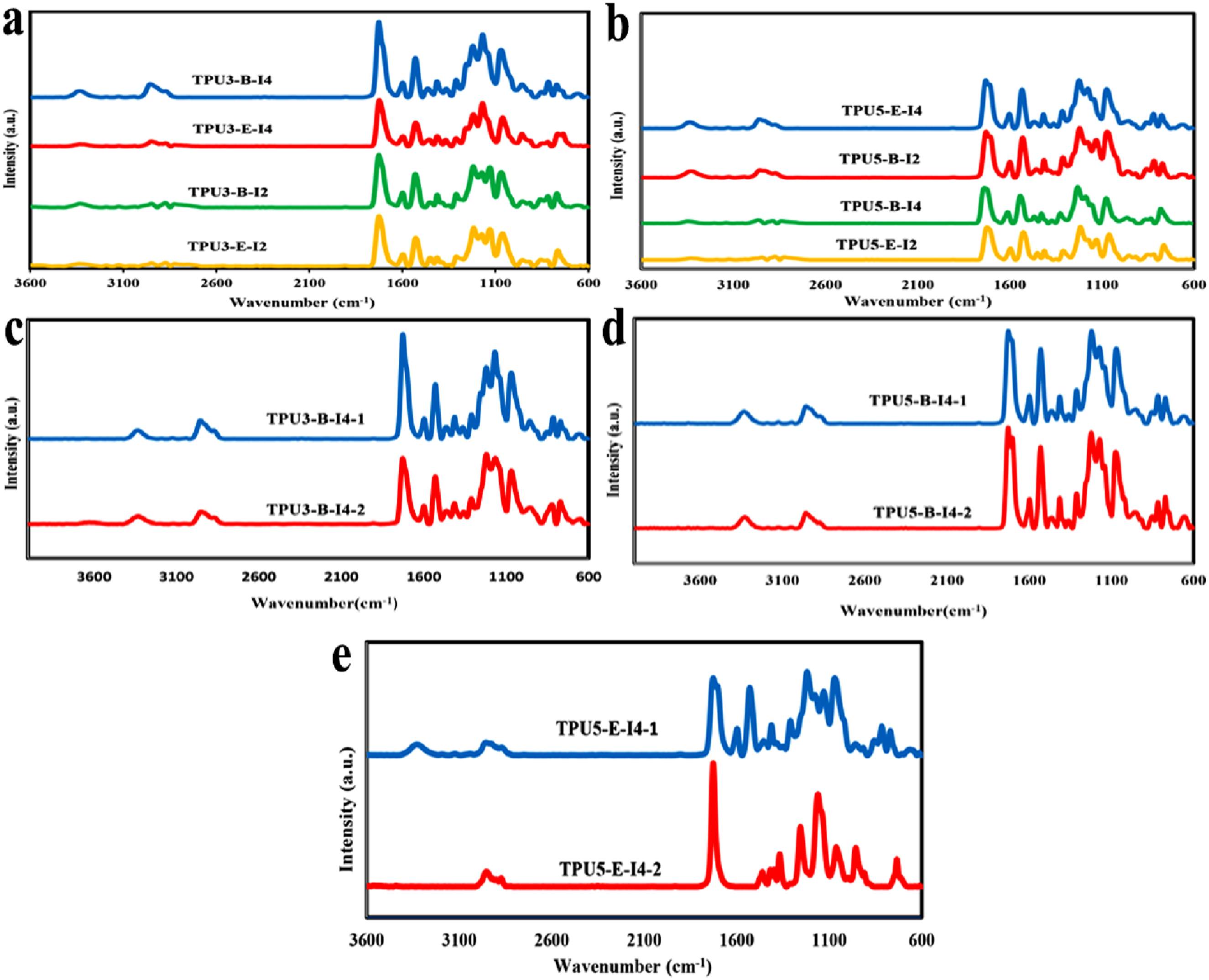
Results of the FTIR spectra for (a) Polyester polyols and butandiol and (b) diisocyanates. FTIR spectra for various synthesized thermoplastic polyurethanes with different types and ratios of reactants, (a) 3-2-1 and (b) 5-4-1. Comparison of one-step and two-step TPU synthesis methods in reactant molar ratios of (c) 3-2-1, and (d, (e) 5-4-1.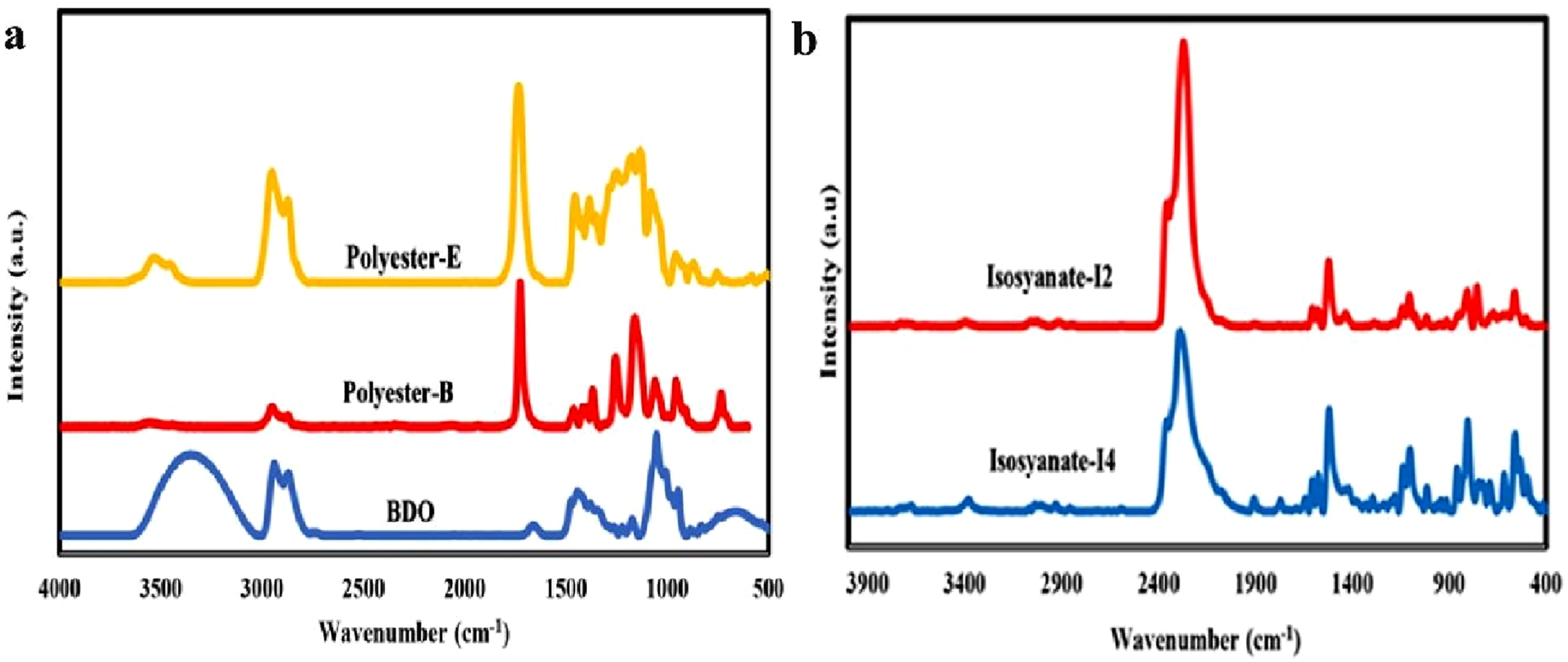
The hard segment content (HSC), hydrogen bonding index (HBI) and allophanate content for various polyurethane samples.
N.A: not applicable.
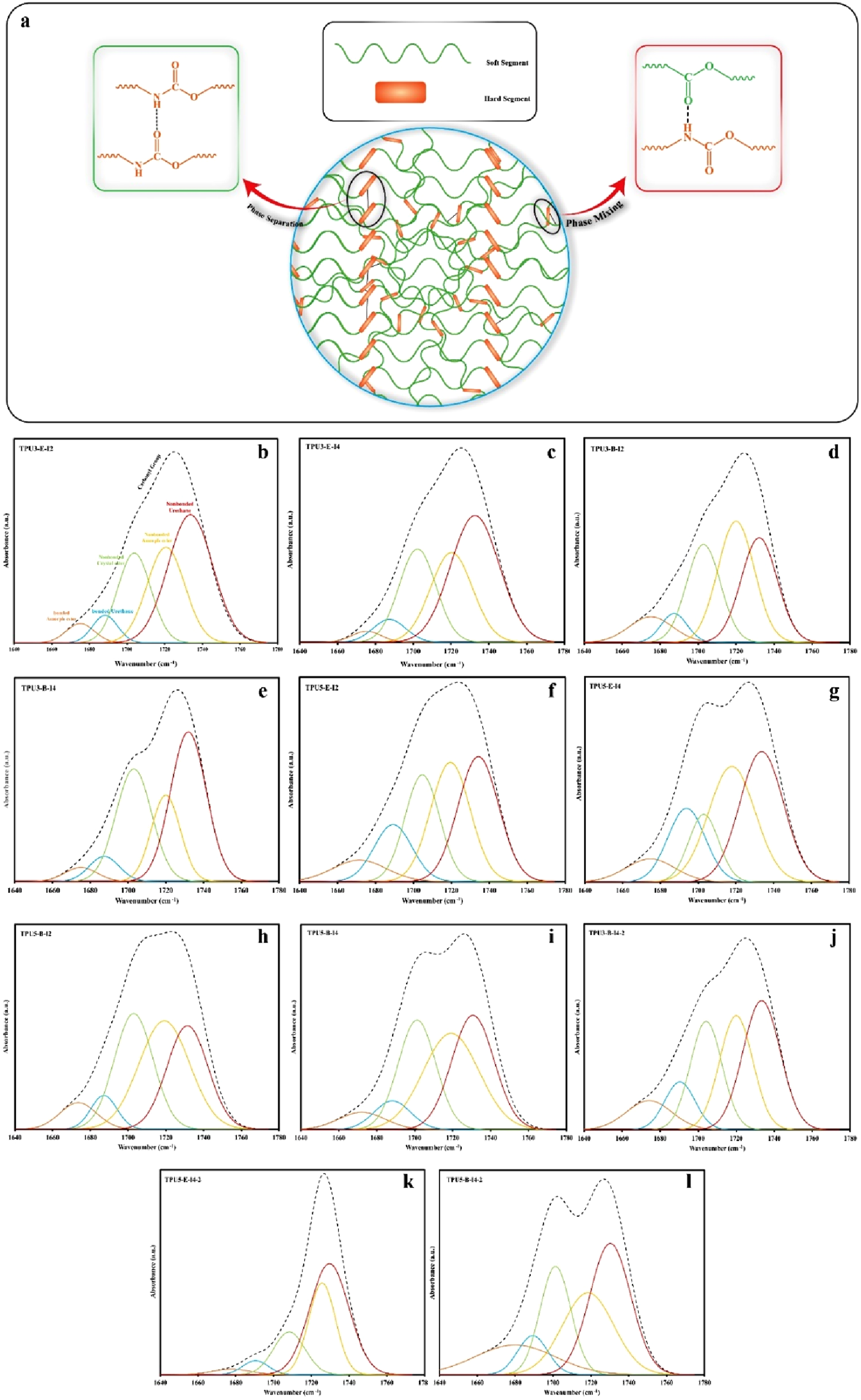
(a) a schematic view from the mechanism of microphase separation and phase mixing in TPU, and the deconvoluted FTIR results for calculating hydrogen bonding index (HBI) in various TPU samples, (b)TPU3-E-I2, (c)TPU3-E-I4, (d)TPU3-B-I2, (e)TPU3-B-I4, (f)TPU5-E-I2, (g)TPU5-E-I4, (h)TPU5-B-I2, (i) TPU5-B-I4, (j) TPU3-B-I4-2, (k) TPU5-E-I4-2 and (l) TPU5-B-I4-2.
AFM images
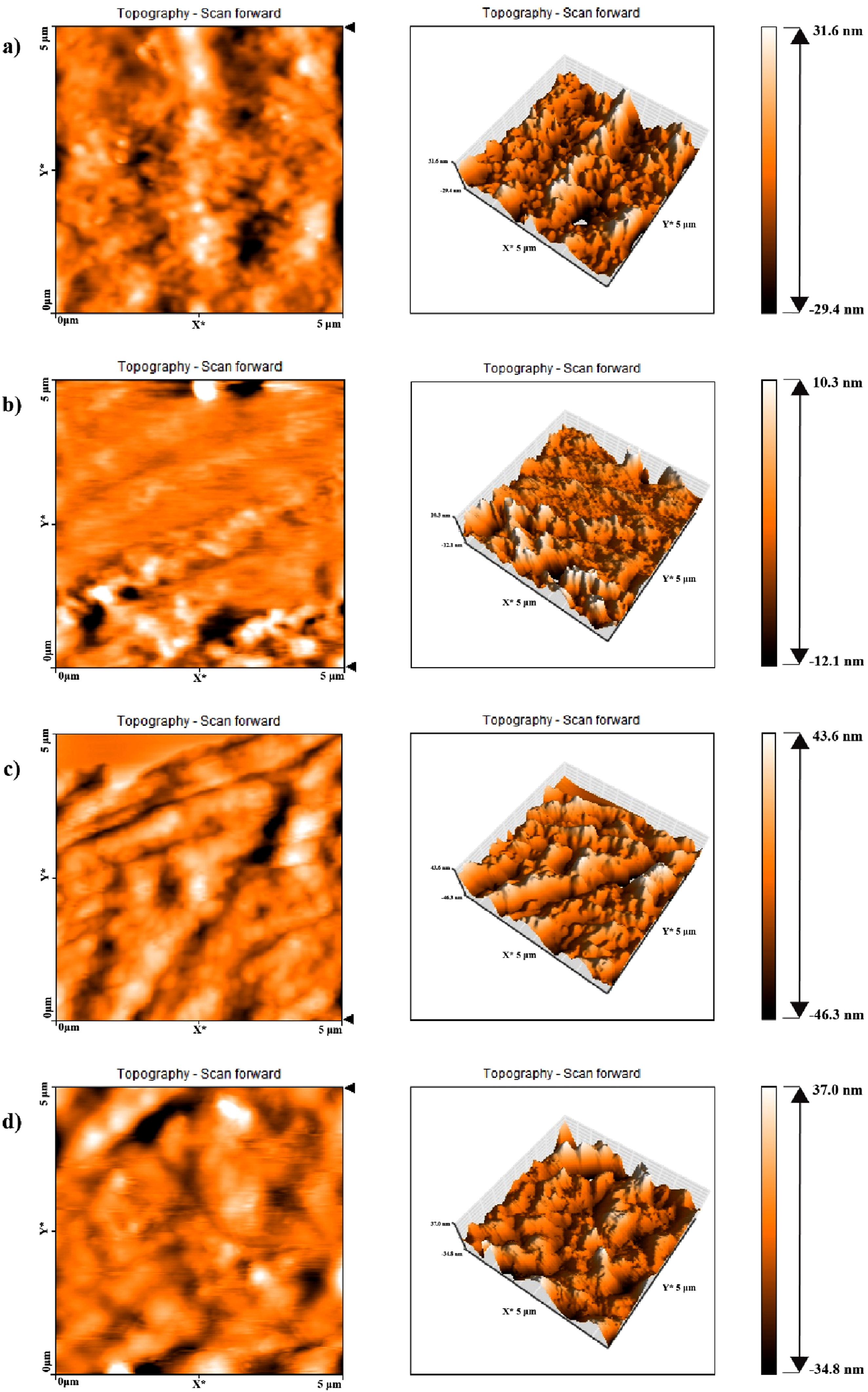
AFM images of TPUs with different HSC contents and various reactant ratios at different length scales are shown in Figures 4 and 5. The bright regions in the AFM images represent the areas corresponding to the hard segments of polyurethane, while the dark regions indicate the domains related to the soft polyol phases. Figure 4 corresponds to a reactant ratio of 3-2-1, where the hard segment content (HSC) is constant for all samples at 32 wt%. The differences between TPU samples in Figure 4(a)–(d) are related to the different type of polyols (semi-crystalline polyester-B and amorphous polyester-E), as well as the various type of diisocyanates employed (highly reactive 4,4′-MDI and less reactive diisocyanate, which is a combination of 4,4′- and 2,4′-MDI). Figure 5 similarly shows TPU samples’ AFM images with a constant hard segment content of 45.3 wt%, but with a different stoichiometric ratio of 5-4-1. In this case as well, different polyols and diisocyanates were used, which had a significant effect on the resulting phase morphology. Although the amount of hard segment is the same in the related TPU samples of 3-2-1 and 5-4-1 reactant ratios, different phase morphologies have been observed. Generally, while comparing the TPU samples with a 3-2-1 reactant ratio to those with a 5-4-1 M ratio, it is evident that the phase morphologies in the 3-2-1 TPU samples, which contain less HSC compared to the 5-4-1 samples, exhibit less microphase separation and show a dispersed-phase morphology, in which the hard segment domains are dispersed within the soft polyol phase matrix. With increasing the hard segment content in the 5-4-1 TPU samples, the phase morphology shifts to a co-continuous structure of hard and soft regions. Furthermore, when comparing the AFM images for these TPU with two different reactant ratios, it is evident that in TPU samples containing semi-crystalline polyester-B soft phases, the degree of phase separation is higher. The hard segment domains in these samples are noticeably larger than those TPUs containing amorphous polyester-E soft phases, as clearly shown in their phase morphology and topological images. This indicates that in polyurethane samples synthesized based on semi-crystalline polyester-B polyols, microphase separation occurs more readily. In other words, the formation of crystalline regions within the polyester-B soft phase and their separation from the amorphous regions, facilitate the microphase separation between the soft and hard segments in the TPU. As a result, the hard segments tend to cluster together, forming larger domains. This phase separation has led to improved mechanical properties in these samples, which is discussed in the relevant section. Another important factor affecting the degree of phase separation in the TPU samples is the type of diisocyanate used in the synthesis of polyurethane, which clearly influences the level of hydrogen bonding index (HBI) in the samples. As discussed in the FTIR analysis section, the samples prepared with the more reactive diisocyanate (4,4′-MDI, referred to as I4) exhibit a higher HBI compared to those prepared with the less reactive diisocyanate (a mixture of 2,4- and 4,4′-MDI, referred to as I2) at both reactant ratios of 3-2-1 and 5-4-1. This difference has also affected the extent of microphase separation in these samples. Specifically, all samples synthesized with the I4 diisocyanate show greater phase mixing between the hard and soft segments, which may be attributed to the higher reactivity of this diisocyanate isomer compared to the 2,4-MDI isomer, which finally leads to smaller hard phase domains in the corresponding samples. Furthermore, the topological studies of the TPUs indicate that the samples synthesized with a higher diisocyanate-to- polyol molar ratio (5-4-1 compared to 3-2-1), exhibit a lower surface roughness due to their higher hard segment content. AFM phase images and the corresponding height images of TPUs with a reactants ratio of 3-2-1, (a) TPU3-E-I2, (b) TPU3-B-I2, (c) TPU3-E-I4 and (d) TPU3-B-I4. AFM phase images and the corresponding height images of TPUs with a reactants molar ratio of 5-4-1, (a) TPU5-E-I2, (b) TPU5-B-I2, (c) TPU5-E-I4 and (d) TPU5-B-I4.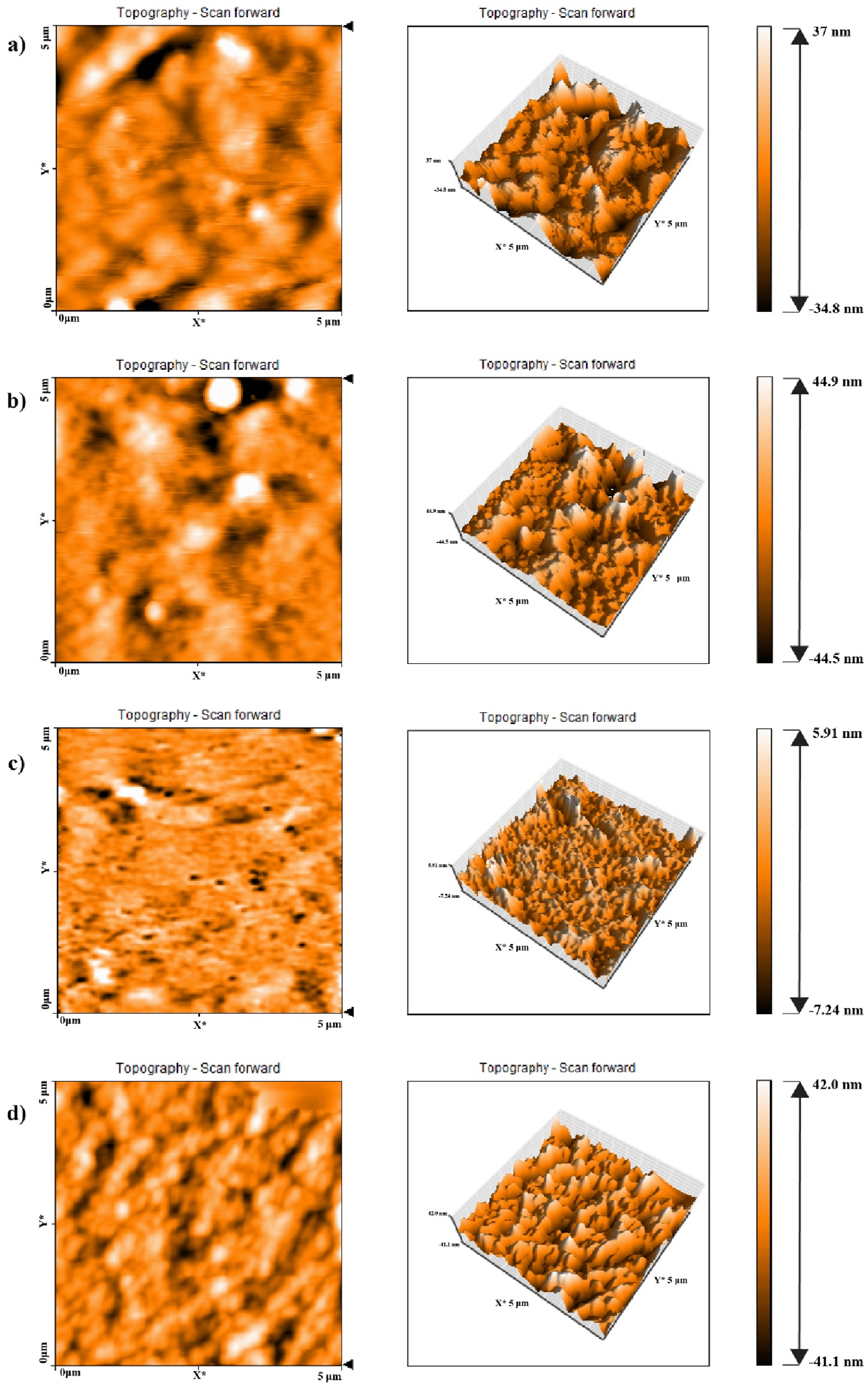
Thermal properties of TPUs
The physico-mechanical properties of the synthesized polyurethanes depend on two key parameters: the crystallinity of the soft segment and the degree of ordering in the hard segment, which arises from hydrogen bonds formed by urethane groups in the hard segment. 50 The hard and soft segments of polyurethanes undergo microphase separation, forming crystals in the soft segment. 51 In contrast, the hard segments of different chains align together, forming hard segment domains. Two phenomena can influence these properties, both resulting from hydrogen bonding interactions between different segments of the polyurethane: hard-to-hard segment interactions and hard-to-soft segment interactions. For instance, in the case of hard-to-soft interactions, hydrogen bonds form between the hard segments of the polyurethane and the ester groups of the soft segments, leading to increased phase mixing. Conversely, hard-to-hard interactions promote microphase separation, causing the hard segments to separate from the soft segments and aggregate together.
Therefore, studying the crystallinity of the soft segment is important due to its impact on the final properties of the polyurethane product.
52
Similarly, investigating the endothermic enthalpy related to the ordering of the hard segment is also crucial. For this reason, the thermal behavior of the raw materials forming the soft segment and the final synthesized polyurethane products were analyzed using DSC, and the results were presented in Figures 6–10. DSC thermograms of (a) the cooling stage of Polyester-E and Polyester-B, (b) the second heating stage of Polyester-E and Polyester-B. (a) The schematic view of the formation of polyurethane chain structures during heating and cooling processes. The first heating stage DSC thermograms of various TPU samples, (b) with a reactant ratio of 3-2-1, (c) with a reactant ratio of 5-4-1 (d), comparison of some TPU samples with a reactant ratio of 3-2-1 in one-step and two-step synthesis methods, and (e, f) comparison of some TPU samples with a reactant ratio of 5-4-1 in one-step and two-step synthesis methods. The second heating and cooling stages in DSC thermograms of various TPU samples, (a, b) second heating and cooling stages with a reactant ratio of 3-2-1, (c, d) comparison of the second heating and cooling stages thermograms for some TPU samples with a reactant ratio of 3-2-1 prepared by one-step and two-step synthesis methods. The second heating and cooling stages in DSC thermograms of various synthesized polyurethane samples using different types of diisocyanates and polyols. (a, b) the second heating and cooling stages with a reactant ratio of 5-4-1, (c, d) comparison of the second heating and cooling stages thermograms for TPU5-E-I4 samples in one-step and two-step synthesis methods, and (e, f) comparison of second heating and cooling stages thermograms for TPU5-B-I4 samples in one-step and two-step synthesis methods. DSC thermograms of the third heating stage after 48 hours aging at room temperature for some selected polyurethane samples synthesized with reactant ratios of 3-2-1 and 5-4-1.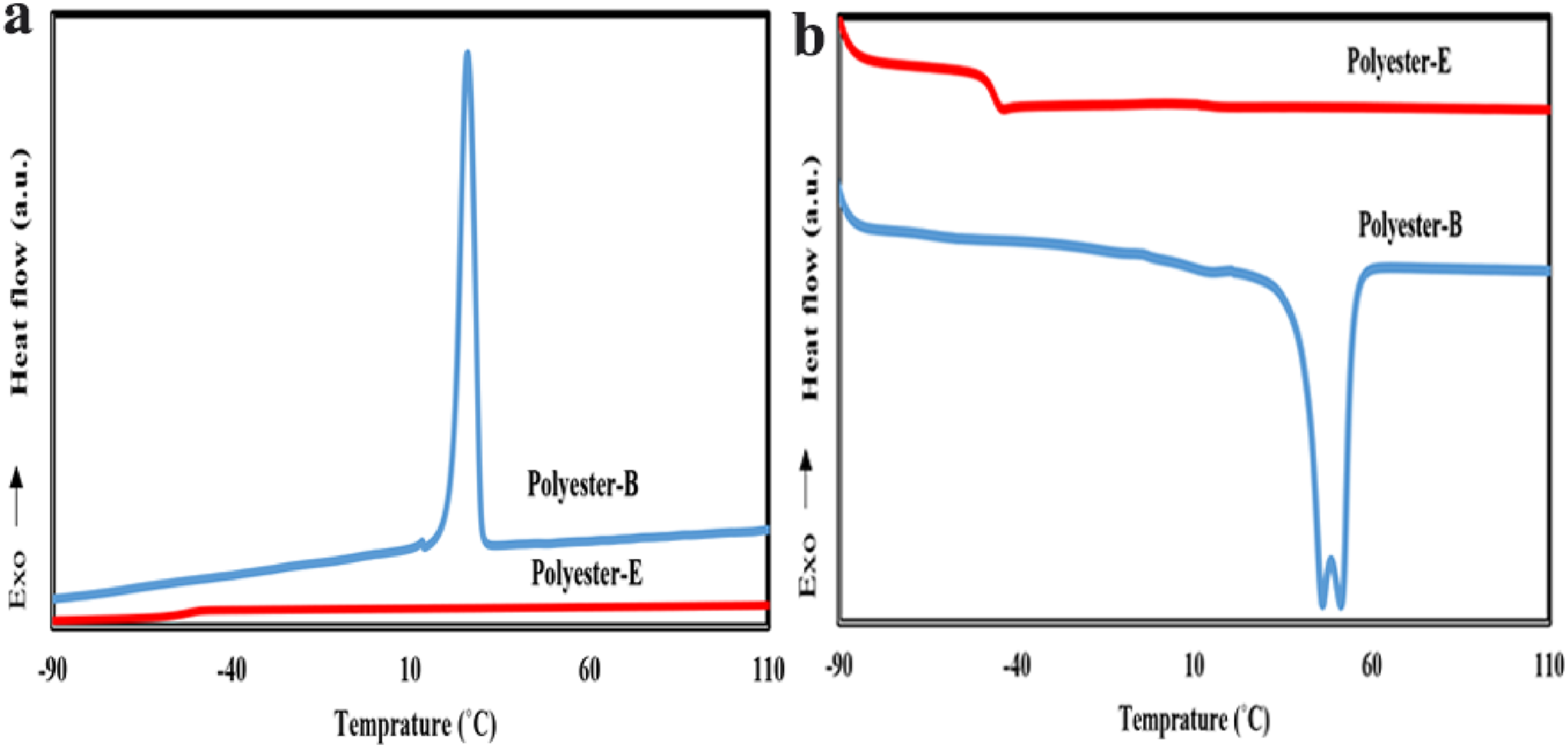
Figure 5 shows that the synthesized polyester based on butane diol has a completely crystalline structure in the second heating and cooling curves. In contrast, the polyester based on ethylene glycol is entirely amorphous. Specifically, in the second heating curve, a melting peak of 50–60°C is observed for the crystallinity of the polyester-B. In contrast, the polyester-E only exhibits a glass transition temperature (Tg) around −40°C. Therefore, the synthesized polyurethanes based on the polyester-B are expected to have a crystalline structure in the soft segment. In contrast, those based on the polyester-E will have an amorphous soft segment, leading to different properties in these two types of polyurethanes. To investigate the effect of the crystallinity of the soft phase on the crystalline structure of the resulting polyurethanes, various polyurethanes synthesized from these two types of polyesters with different diisocyanates were studied. The results of their thermal analysis during cooling and heating stages are presented in Figures 7–9.
Figures 7 and 8 show the first heating, cooling, and second heating stages of DSC thermograms for the synthesized polyurethanes. These polyurethanes were derived from a 3-2-1 reactant ratio using polyester-B and polyester-E polyols, as well as different diisocyanates including 4,4′ MDI (type I4) and 4,4’ −2,4′ mixed MDI (type I2). All samples synthesized using polyester-E exhibit no crystallinity in the soft segment region during either heating or cooling. These amorphous samples display only a single glass transition temperature (Tg). Upon precise examination of the results of polyurethane samples synthesized from polyester-E, it is evident that they have a higher Tg than the corresponding polyester-B. This increase in Tg is attributed to the anchoring effects of the hard segments on the soft segments, which even can influence the crystallinity of the soft phase (in case of semi-crytalline polyester-B) and shift the Tg to higher temperatures. For example, in sample TPU3-E-I4, the Tg has shifted to the range of 0 to 10°C, whereas in the original polyester-E, this value was −40°C. A similar increase in Tg is observed in other samples. In contrast, polyurethane samples synthesized using polyester-B exhibit crystalline regions. For instance, in TPU3-B-I4 and TPU3-B-I2 samples, certain crystallinity in the soft segment is noticeable, while this crystallinity isn’t observed in other samples.
The endothermic enthalpy associated with hydrogen bond dissociation is discussed in the second part of the thermal analysis. As observed, the ordering of the hard segments is evident in all samples at temperatures above 150°C during their first heating. However, when comparing the second heating stage (Figure 8) with the first heating curves (Figure 7), the ordering related to hydrogen bonds deteriorates, and the corresponding endothermic peaks disappear. It should be noted that the microphase separation between hard and soft segments in polyurethanes is a time-consuming process, which affects the crystallization of the soft segments and the hydrogen bond-induced ordering in the hard segments. During thermal analysis tests, there is no enough time for complete microphase separation, and consequently for the soft segments’ crystallization, or the hard segments’ ordering. Therefore, the corresponding peaks are observed more broadly at lower temperatures. Another notable aspect in these graphs is the influence of the diisocyanate type on the thermal properties of the synthesized TPU samples. As observed in the FTIR test, hydrogen bonding (HBIC = O) is higher in TPU samples containing 4,4′ MDI diisocyanate compared to the other diisocyante. These hydrogen bonds are associated with interactions between hard-hard and hard-soft segments, and their effects are also clearly observed in the DSC results. The interactions between the hard and soft segments reduce the crystallinity of the soft phase. This reduction may be attributed to the following factors: the anchoring effects of the hard segments on the mobility of the soft segment chains, and the hydrogen bonding interactions between the hard and soft segments. As a result, the crystallinity of the soft segments decreases, and the crystallization temperature of polyurethanes shifts to higher temperatures.
In the hard segment regions, the ordering related to hydrogen bonds is more pronounced in the sample containing diisocyanate (I4) with higher activity, which exhibits a higher degree of hydrogen bonding (HBI). Conversely, in samples synthesized with the lower activity mixed diisocyanate (I2), the hydrogen bonding index is lower, resulting in reduced ordering in the hard segments and a corresponding decrease in endothermic enthalpy. These changes ultimately affect the final properties of the TPU products.
To investigate the effect of changing the reactant ratio on the final properties, the polyurethane samples with a 5-4-1 composition and a higher diisocyanate to polyol ratio were synthesized. As shown in Figure 9, increasing the diisocyanate ratio in the polyurethane formulation leads to an increase in the intensity of the peaks associated with ordering of the polyurethane’s hard segments. This observation is entirely consistent with the Fourier transform test results. As the amount of hard segments increases, the HBI also rises, resulting in greater ordering of the hard segments. A similar discussion regarding the index, as applied to the samples with a 3-2-1 reactant ratio, can also be extended to this composition. Samples synthesized with the more reactive diisocyanate (I4) exhibit a higher ordering of hard segments than those with less reactive I2. Additionally, the endothermic enthalpy related to hydrogen bond deterioration is greater. Thus, the final product properties are more favorable. Another critical aspect examined in the DSC analysis is the effect of the feeding order during polyurethane synthesis on its thermal properties. As shown in Figures 8(c) and (d) and 9 (c)–(f), the samples with the reactant ratios of 3-2-1 and 5-4-1 were synthesized by polyester-B using both one-step and two-step feeding order in internal mixer and their DSC thermograms were analyzed. As observed, the TPU samples synthesized in one-step process exhibit greater ordering of the hard segments. In the two-step feeding sequence, the hard segment consists of a prepolymer with excess NCO. The excess NCO in the system forms allophanate groups, reducing hydrogen bonding. This effect is reflected in the decreased enthalpy of hard segment ordering observed in the DSC tests. The allophanate groups behave differently from urethane groups; they are thermally reversible chemical bonds, meaning they can break and reform as the temperature increases. Therefore, during the two-step polyurethane synthesis in an internal mixer, allophanate groups form instead of some urethane groups due to the chain’s reaction between excess diisocyanate and the pre-existing urethane groups. This reduction in the number of urethane groups and their associated hydrogen bonds is evident in both 3-2-1 and 5-4-1 reactant ratios, as shown in Figures 8 and 9.
To investigate the effect of aging time on the microphase separation behavior and the complete formation of soft segment crystals and hard segment ordering of TPUs, the DSC thermal analysis was performed on the selected polyurethane samples (TPU3-B-I4, TPU5-B-I4). This analysis was conducted on the samples during the third heating stage after 48 h aging at room temperature, and the results are presented in Figure 10. The results show that in these samples, compared to the second heating stage, the completion of the microphase separation process leads to several changes. These include an increase in the ordering of the hard segments, the reformation of hydrogen bonds, stronger interactions between hard segments, and enhanced soft segment crystallinity.
Melting enthalpy of the soft segments (ΔHs), disordering enthalpy of hard segments (ΔHh), melting temperature of the soft segment (Tms), ordering temperature of the hard segment (Th), and glass transition temperature (Tg) for various polyurethane samples synthesized with different compositions.
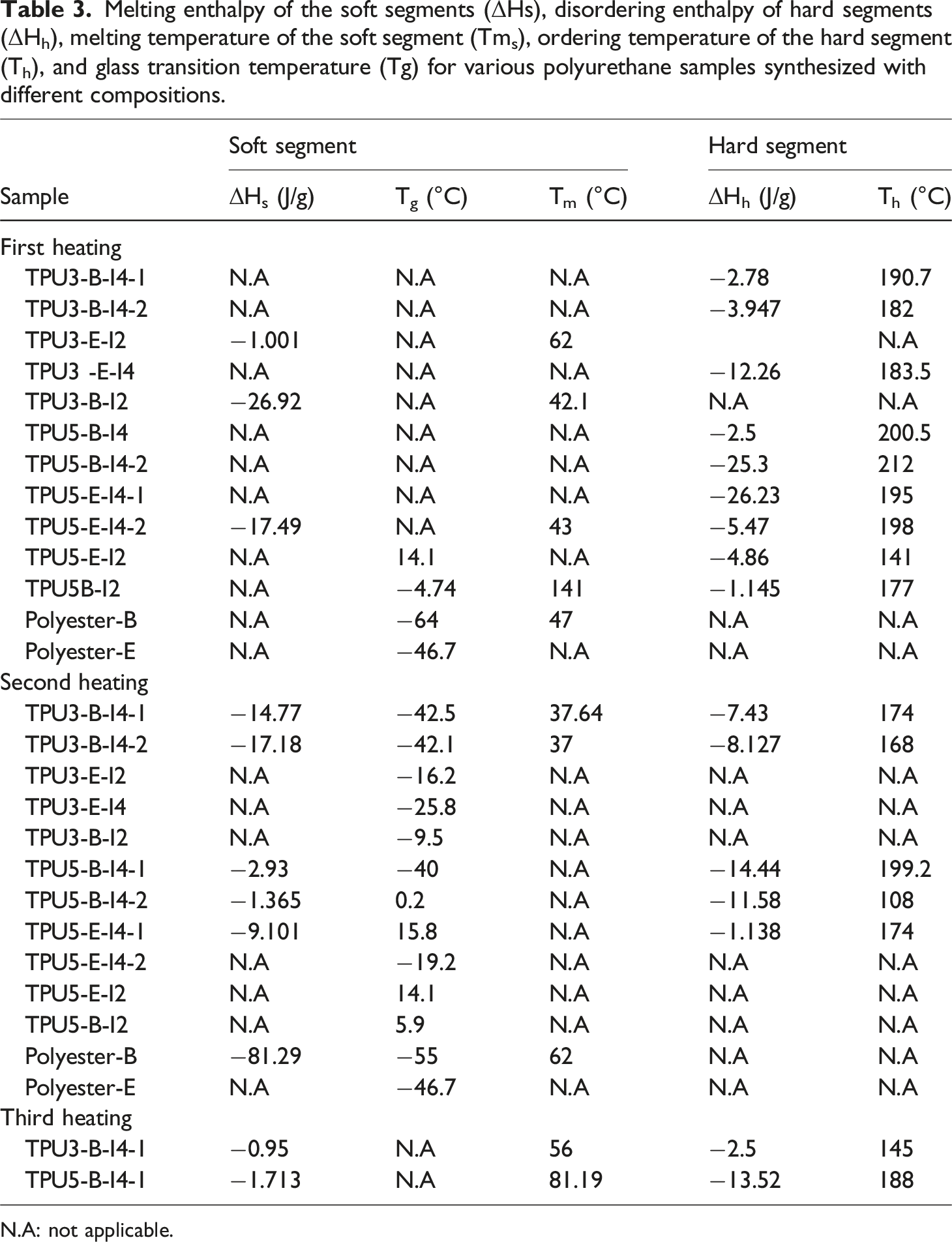
N.A: not applicable.
Crystallization temperature (Tc), onset temperature of crystallization (Tonset) and the time required to achieve 50% crystallinity (t50%) and 90% crystallinity (t90%) for various polyurethane samples.
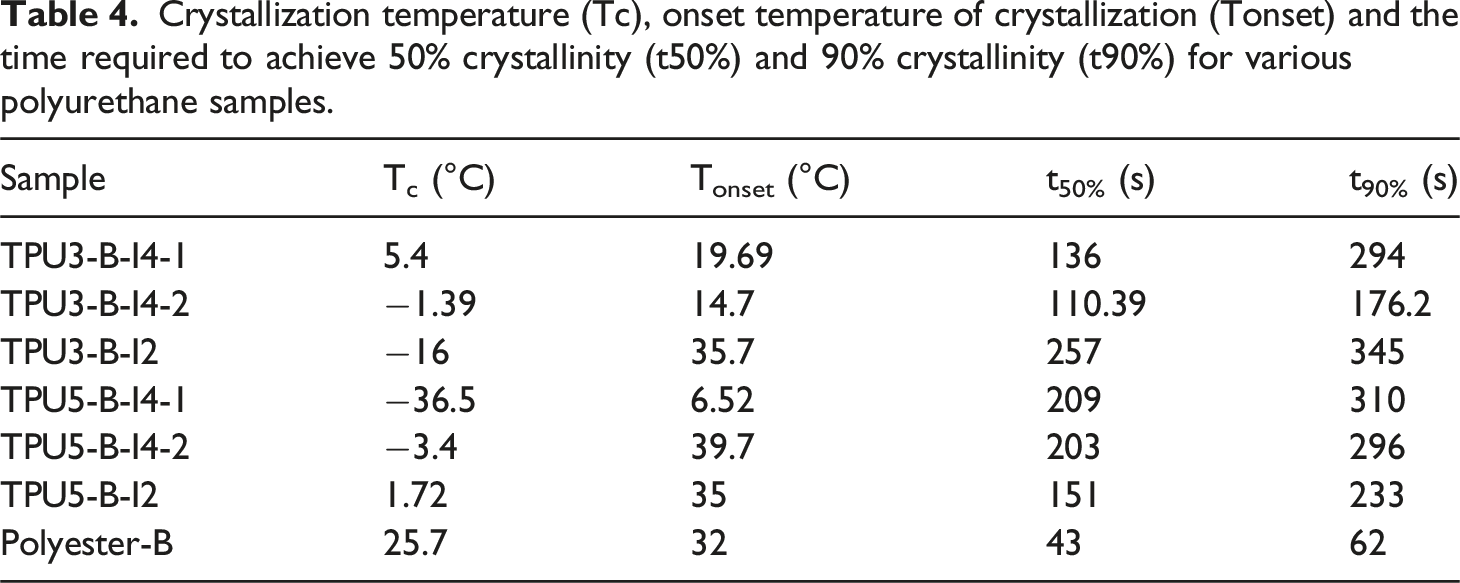
Figure 11 presents the changes in relative crystallinity of the synthesized polyurethanes with respect to temperature and time to investigate the crystallization process of the synthesized polyurethanes in more detail. These plots can be used to determine the exact crystallization onset temperature and time and the crystallization rate for different polyurethane samples. As shown in Figure 11, crystallization begins as the temperature decreases. For example, in the polyurethane sample synthesized with polyester-B polyol, a comparison between the one-step and two-step feeding methods reveals that, in the two-step method, crystals form later and at a lower temperature compared to the one-step method. This suggests that crystallization occurs more effectively in the one-step synthesis method. Additionally, when comparing the crystallization rates of these samples, it is evident that the time required to achieve 50% crystallization (t50%) is shorter in the one-step method than in the two-step method. This indicates that the one-step method has a faster crystallization rate. A comparison of the crystallization onset temperatures and crystallization rates for different polyurethane samples is provided in Table 4. The relative crystallinity changes vs temperature and time for various polyurethane samples synthesized using different types of diisocyanates, chain extenders, and polyols: (a, b) relative crystallinity changes with temperature and time for samples with reactant ratios of 3-2-1 and 5-4-1, (c, d) comparison of the relative crystallinity changes vs temperature and crystallization time for the samples with a reactant ratio of 3-2-1 in one-step and two-step synthesis methods, (e, f) comparison of the relative crystallinity changes versus temperature and time for samples with a reactant ratio of 5-4-1 in one-step and two-step synthesis methods.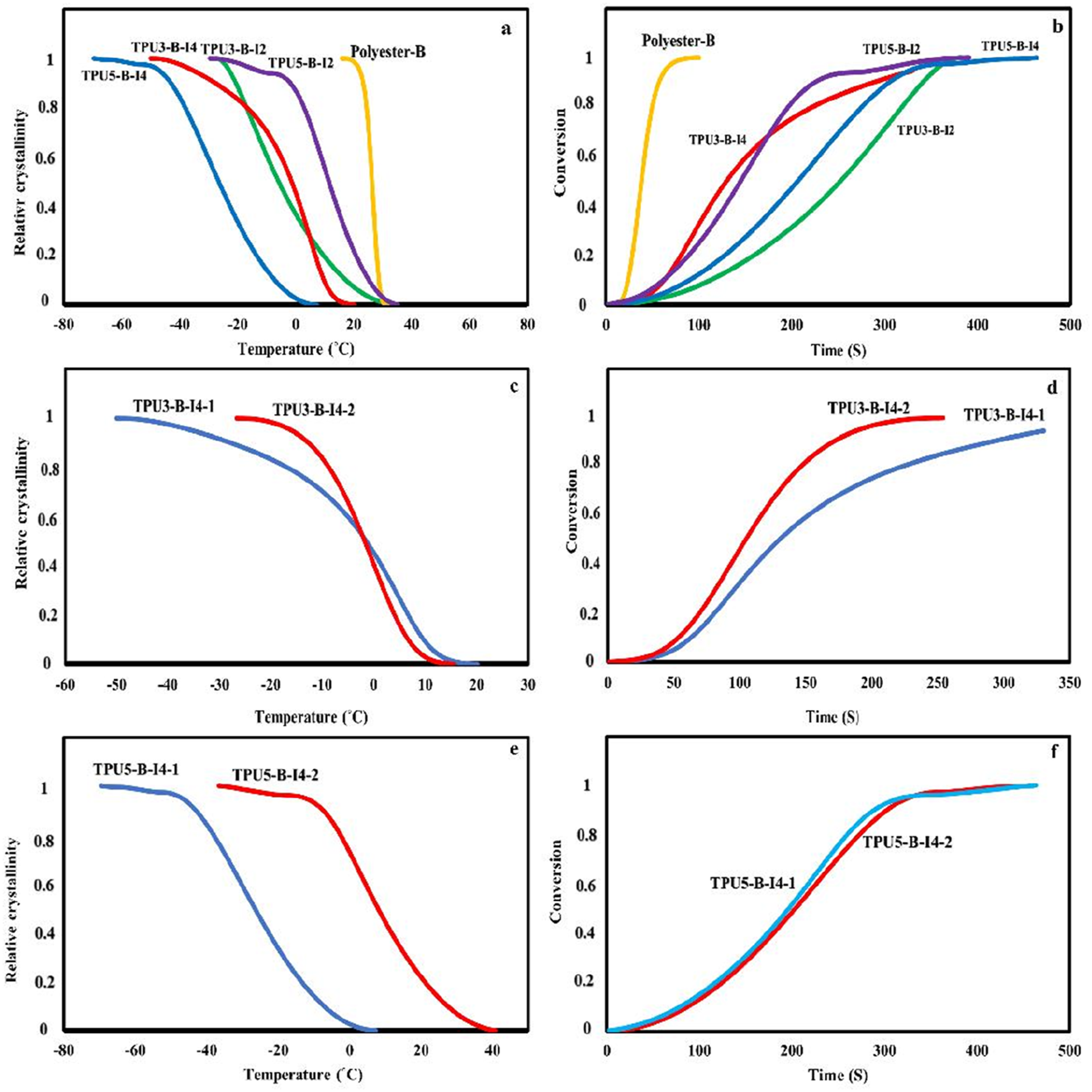
Mechanical properties of polyurethanes
Several factors are essential to consider when assessing the mechanical properties of polyurethanes prepared from various polyols and diisocyanates. The main factors include the soft segment’s crystalline behavior, the hydrogen bonding index (HBI), the degree of microphase separation between hard and soft phases, the hard segments content (HSC), and the extent of reactivity of different diisocyanates. Figure 12 illustrates the results of the mechanical properties evaluation for several polyurethanes, displaying stress-strain curves for four samples created from different polyols and diisocyanates, using reactant ratios of 3-2-1 and 5-4-1 for the corresponding samples. These curves reveal three distinct regions including elastic deformation, plastic deformation, and the strain hardening area. Tensile stress-strain curves for various polyurethane samples synthesized with different types and ratios of reactants at room temperature. These diagrams compare, (a, b) the reactant ratios of 3-2-1 and 5-4-1, (c) the various feeding order (one-step and two-step methods) with the reactant ratio of 3-2-1, (d, e) the various feeding order (one-step and two-step methods) with the reactant ratio of 5-4-1 containing different polyols (polyester-E and polyester-B).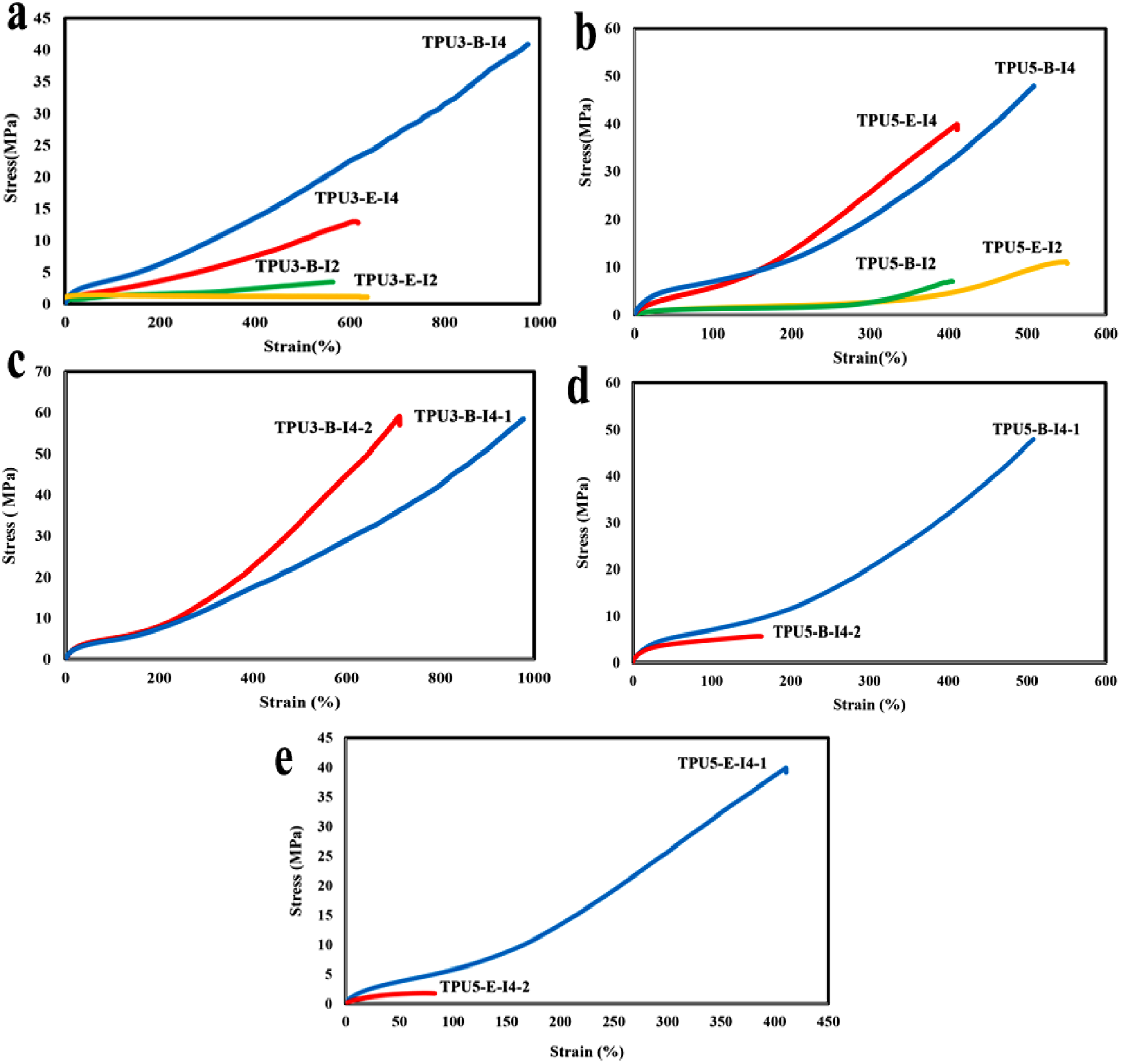
The tensile mechanical properties for various thermoplastic polyurethane samples.
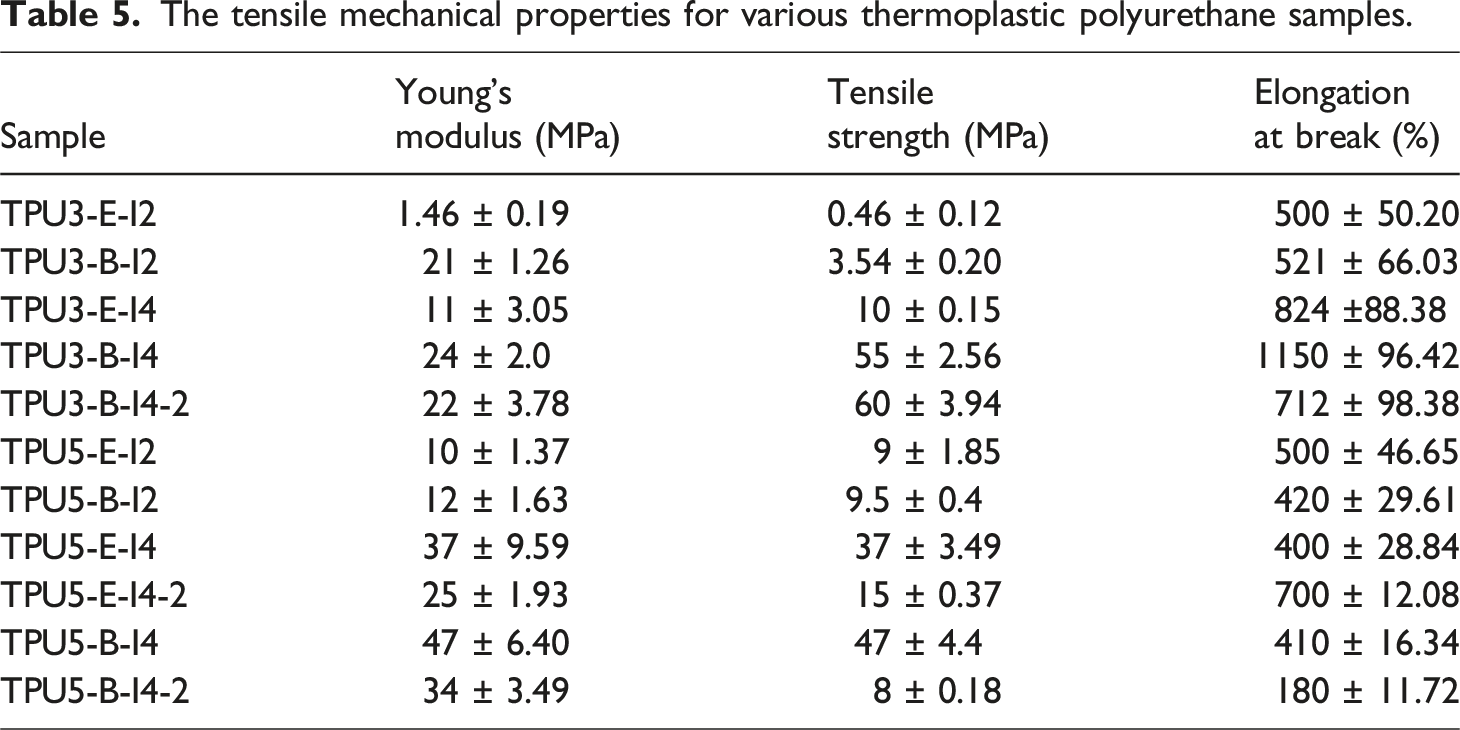
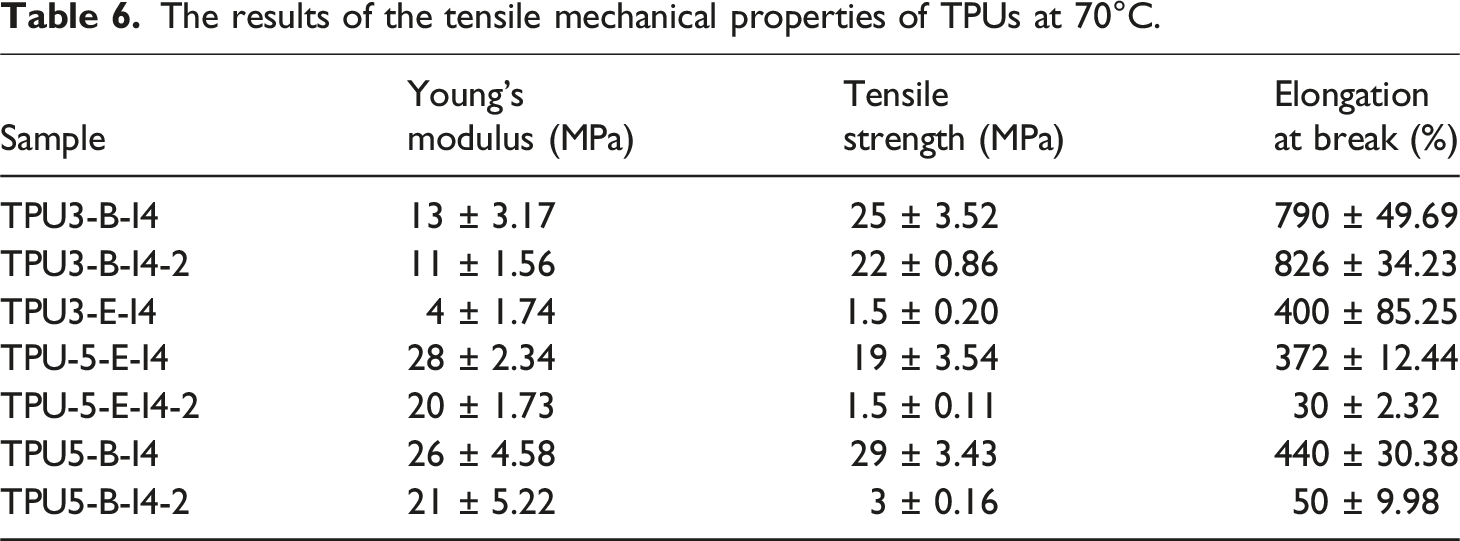
The mechanical properties of polyurethane mainly depend on the chains molecular weight, crystallinity of the soft segment, the hard segment content, and the degree of hydrogen bonding between hard to hard and hard to soft segments. To eliminate the influence of soft segment crystallinity during mechanical testing and focus on the effects of the hard segment content alongside hydrogen bonding index, the tensile tests were performed at temperature above the soft segment’s melting point, precisely at 70°C. This temperature was selected because it surpasses all polyurethane samples’ melting points. The results of this test can be found in Figure 13. When the sample TPU3-E-I4 is compared with the sample TPU3-B-I4, although the crystallinity of the soft phase in the crystalline polyester has been vanished at this temperature, the mechanical strength of the samples synthesized with polyester-B polyol is still higher than that of the sample synthesized with polyester-E polyol. This may be attributed to the higher HBI in the hard phase of the sample TPU3-B-I4 compared to the sample TPU3-E-I4, which has increased in its mechanical strength. This observation is also obvious when comparing the TPU samples with 5-4-1 reactant ratio, where the mechanical properties are similar. This similarity is due to the comparable proportions of the hard segment and hydrogen bonding in this reactant ratio. As the crystallinity of the soft phase in sample TPU5-B-I4 has been vanished in this temperature, its mechanical properties at the moment closely match the TPU5-E-I4 sample, which features an amorphous soft phase, owing to their comparable HBI values. Tensile stress-strain curves at 70°C for the selected samples of polyurethane synthesized with various types and ratios of reactants, (a) comparison of the 3-2-1 and 5-4-1 reactant ratios and (b, c, d) comparison of one-step and two-step synthesis methods with 3-2-1 and 5-4-1 reactant ratios.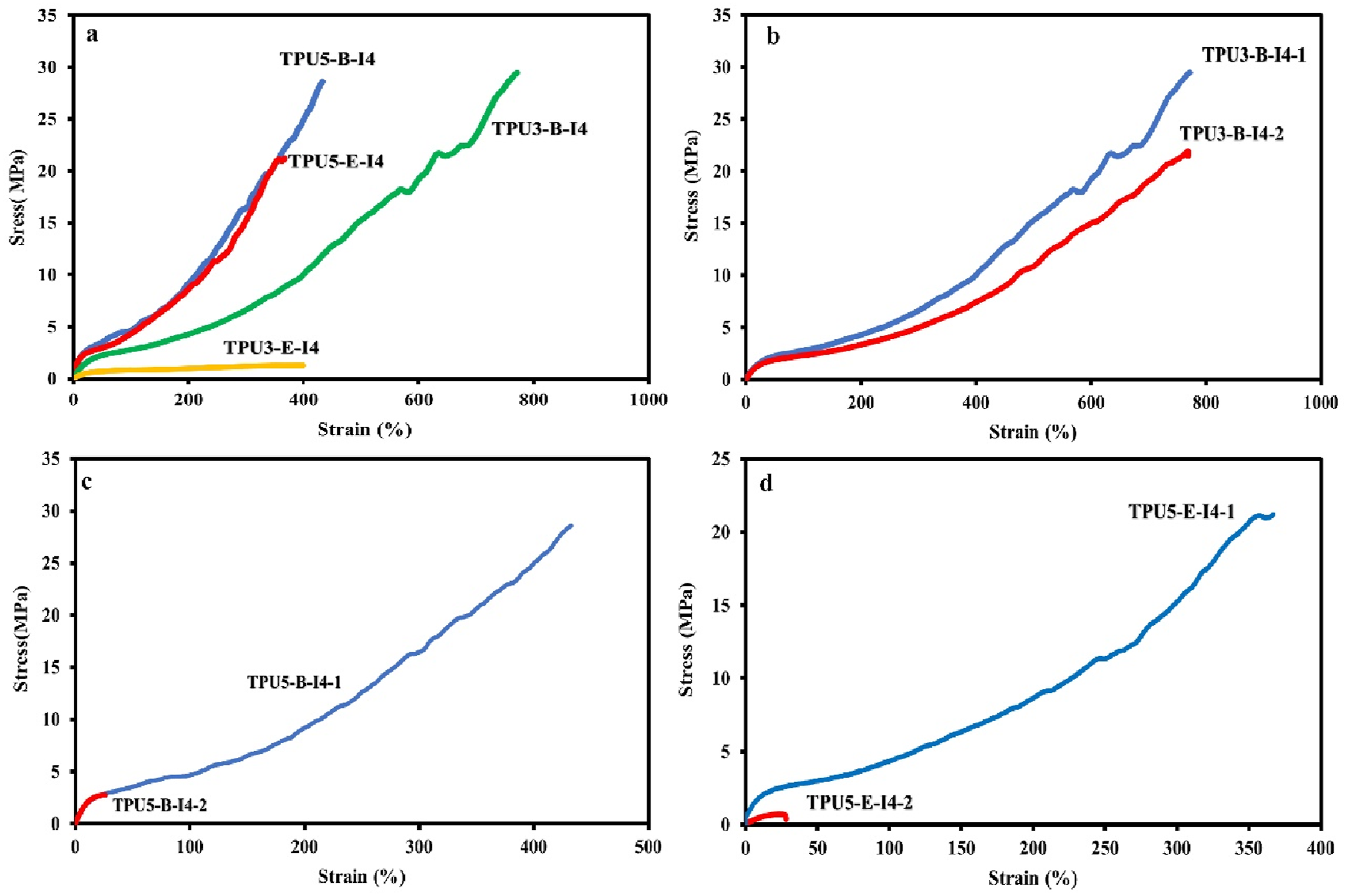
The results of the tensile mechanical properties of TPUs at 70°C.
Rheological behavior of TPUs
Rheological analyses were conducted to examine the synthesized polyurethanes’ melt behavior accurately. For this purpose, all samples were evaluated at a temperature above the disordering temperature of the hard segments. DSC the rmal analysis studies indicated that at temperatures above 200°C, the hard segment’s orderings in all samples were deteriorated; therefore, 210°C was selected as the reference temperature for comparison. Also, the rheological evaluations were performed using a frequency sweep test at a constant temperature at a strain level of 1%, which falls within the linear viscoelastic region.
55
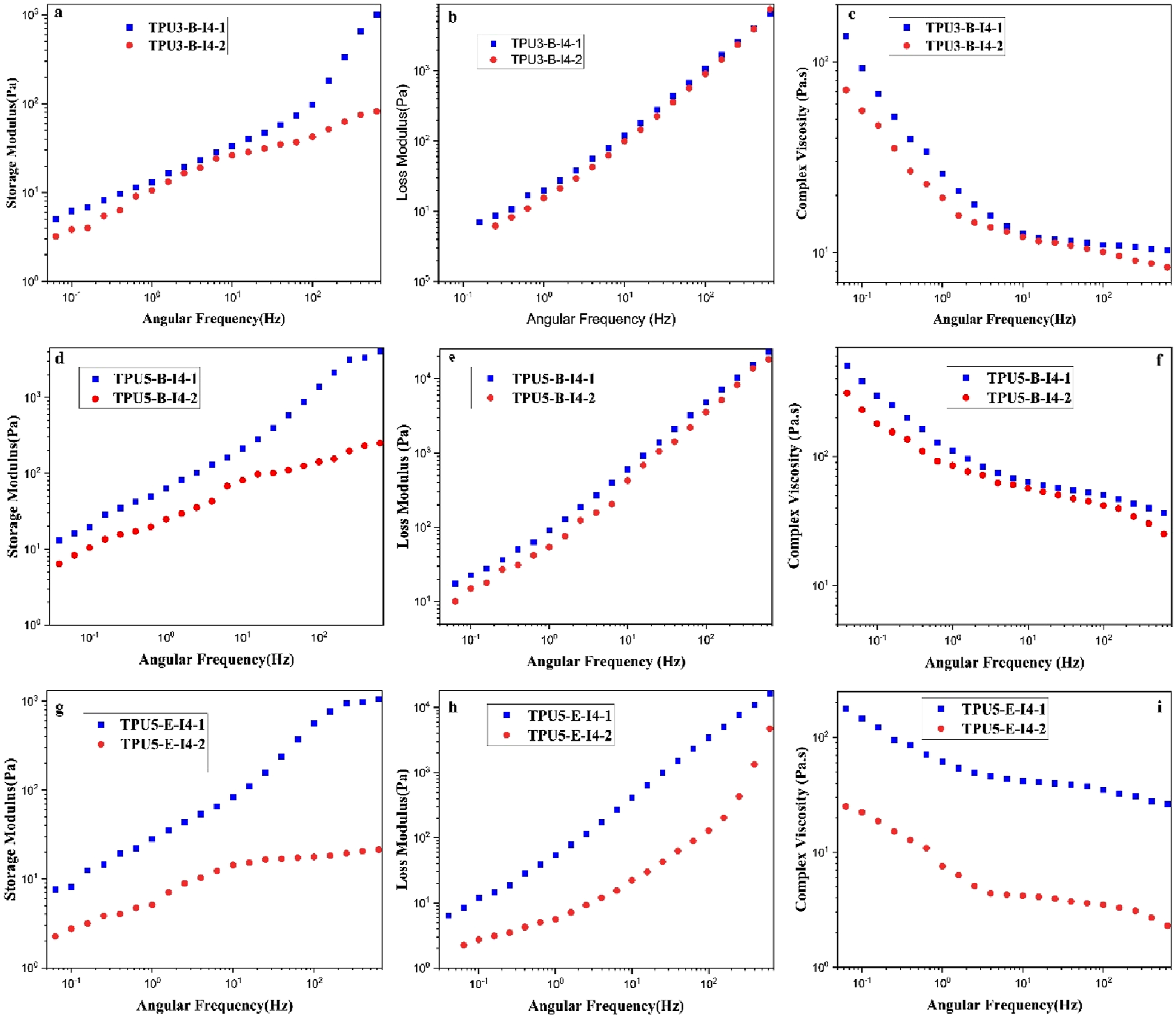
Figure 14 reports the results of the storage modulus (G′), loss modulus (G″), and complex viscosity (η*) for different samples. Rheological tests were conducted on the selected polyurethane samples that exhibited superior mechanical properties. As observed, the storage modulus, loss modulus, and complex viscosity are higher in TPU samples with the reactant ratio of 5-4-1 compared to the samples with the reactant ratio of 3-2-1. Furthermore, in polyurethane samples synthesized based on polyester-B polyol, due to the higher reactivity of polyester-B polyol with the corresponding diisocyanates compared to those based on polyester-E polyol, it seems that the polyurethane chains have grown more extensively, achieving a higher molecular weight. Consequently, these samples exhibit higher storage and loss moduli, and complex viscosity. Two important factors influence the rheological behavior of synthesized polyurethanes: a higher reactant ratio, which leads to an increase in storage modulus (G′), loss modulus (G″), and complex viscosity (η*); and changing the type of polyol from polyester-E to polyester-B, which enhances their reactivity with diisocyanates, increases chain length, and ultimately results in higher values of G′, G″, and η*. The comparison of the rheological behavior of the samples synthesized with different reactant ratios using the one-step and two-step methods is presented in Figure 15. The values of G′, G″, and η* are higher in the samples synthesized via the one-step method. In the samples where the ratio of diisocyanate to polyol is higher (the reactant ratio 5-4-1), the two-step synthesis method results in a significantly excess amount of diisocyanate in the reaction system. A portion of –NCO groups of this excess amount of diisocyanates probably reacts with the –OH groups of polyol, while the remainder –NCO groups reacts with the urethane groups of the TPU pre-polymer as a side reaction, leading to the formation of allophanate groups. This subject inhibits the proper growth of the polyurethane chains and restricts the proper growth of the molecular weight in the resulting polymer.
56
Consequently, the rheological properties of the samples decline accordingly. In the formation of allophanate groups, the excess diisocyanate is consumed in the side reactions instead of forming different pre-polymer chains. This prevents the reaction of –NCO end prepolymer with the chain extender in the next step, which would otherwise increase the length of the polyurethane chains. As a result, shorter chain lengths are formed, and the complex viscosity becomes approximately 10 times lower than that of the one-step synthesis method. Ultimately, the inadequate growth of the polyurethane chains leads to a decline in the mechanical properties. Rheological test results of various thermoplastic polyurethane samples synthesized with different types and ratios of reactants; (a) variation of G” vs frequency for reactant ratios of 3-2-1 and 5-4-1, (b) variation of G' vs frequency for the reactant ratios of 3-2-1 and 5-4-1 and (c) variation of η* vs frequency for the reactant ratios of 3-2-1 and 5-4-1. Rheological test results of various thermoplastic polyurethane samples synthesized with different types and ratios of reactants, (a, b, c) comparison of G′, G″, and η*variations vs frequency for the reactant ratio of 3-2-1 in one-step and two-step synthesis methods using polyester-B polyol, (d, e, f) comparison of G′, G″, and η*variations vs frequency for the reactant ratio of 5-4-1 in one-step and two-step synthesis methods using polyester-B polyol and (g, h, i) comparison of G′, G″, and η*variations versus frequency for the reactant ratio of 5-4-1 in one-step and two-step synthesis methods using polyester-E polyol.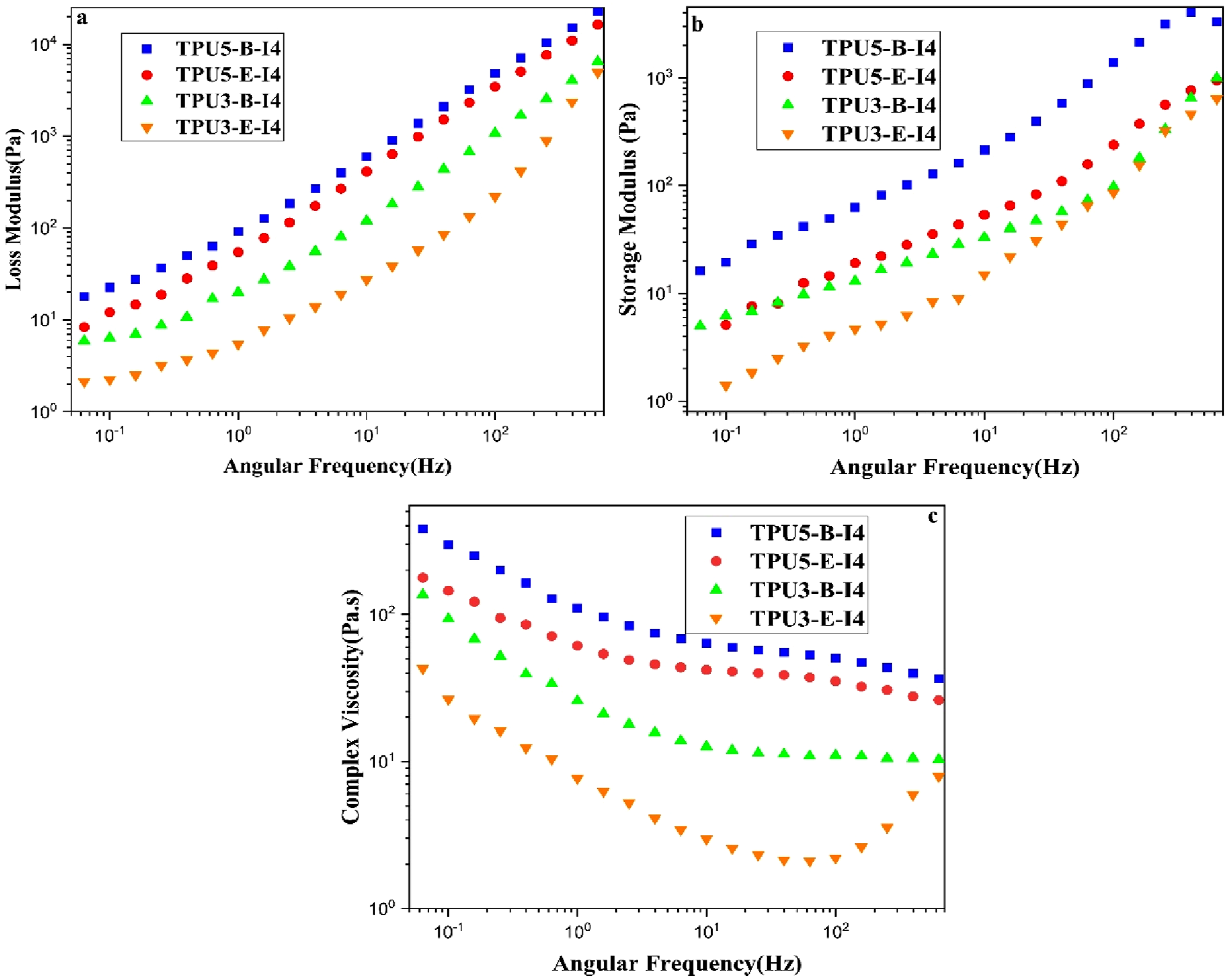
Conclusion
Thermoplastic polyurethane elastomers can be synthesized through one-step or two-step (prepolymer) techniques using the reactive mixing method. Alternatively, changing the ratio of the reactants is one of the main factors influence the final properties of polyurethane, particularly its mechanical properties. Altering the ratio of hard to soft segments in this polymer changes the hydrogen bonds and between hard-hard and hard-soft segments, which significantly affect the phase morphology, physico-mechanical properties and rheological behavior of the synthesized TPU samples. In this research different thermoplastic polyurethane samples were synthesized based on methylene diphenyl diisocyanate (MDI) and polyester polyol using an internal mixer via the one-step and two-step methods. The results indicated that as the reactivity of diisocyanate groups in different isomers increased, the amount of hydrogen bonding in the resulting polyurethanes also increased. In the most TPU samples synthesized with 4,4′-MDI, due to its higher reactivity with hydroxyl groups compared to the other isomer (2,4′-MDI), the HBIC = O also increased. Furthermore, in two-step method, since the diisocyanate was introduced in excess value relative to the polyol, the excess diisocyanate was prone to side reactions, such as reacting with urethane groups of the prepolymer and forming reversible allophanate groups. The formation of allophanate groups in the two-step synthesis method influenced the HBIC = O and other physico-mechanical properties of the synthesized polyurethanes, as observed in the DSC test results. The interactions between the hard and soft segments reduced the crystallinity of the soft phase. These factors ultimately lead to a decrease in the soft phase’s crystallinity. Consequently, the degree of ordering in the hard segment and the corresponding endothermic enthalpy decrease, influencing the final product properties. Once the ratio of diisocyanate in the polyurethane formulation was increased, the intensity of the peaks related to the ordering of the hard segment also increased, which was consistent with the results of HBIC = O in the FTIR analysis. To investigate the crystallization process of the synthesized polyurethanes in more detail, the changes in relative crystallinity of the TPU samples with temperature and time were also examined. In the polyurethane sample synthesized with polyester-B polyol, it was observed that in the two-step method, its crystals formed later and at a lower temperature, indicating more effective crystallinity in the one-step synthesis method. The tensile test results revealed that the polyurethane samples prepared from polyester-B polyol and diisocyanate type I4 (with higher reactivity) showed superior crystallinity in the soft segment, along with improved mechanical properties such as increased Young’s modulus and mechanical strength, compared to those synthesized from polyester-E polyol. AFM images revealed that different phase morphologies have been formed in TPU samples with 3-2-1 and 5-4-1 reactant ratios. Generally, when comparing TPU samples with a 3-2-1 reactant ratio to those with a 5-4-1 ratio, it is observed that the phase morphologies in the 3-2-1 samples, which have lower hard segment content compared to the 5-4-1 one, exhibited less microphase separation. Instead, they displayed a dispersed phase morphology, where hard segments were dispersed within the soft-phase polyol matrix. In contrast, with the increase in hard segment content in 5-4-1 samples, the phase morphology transitions into a co-continuous structure with interconnected domains of hard and soft phases were observed. In this study, the rheological behavior of samples synthesized with different reagent ratios using both one-step and two-step methods was compared. In the samples synthesized via the one-step method, the values of G′, G″, and η* were higher. In TPU samples with 5-4-1 reactant ratio, when a two-step synthesis method was used, the excess diisocyanate in the reaction system was too high. As a result, a portion of it reacted with the polyol, while the remaining excess NCO groups interacted with the urethane groups of the pre-polymer, forming allophanate groups. This might hinder the proper growth of the polyurethane chains and could limit the increasing of the molecular weight of the resulting polymer. Consequently, the rheological properties of the samples also decline accordingly. In two-step method, excess NCO groups don’t contribute in increasing the polyurethane chain length. Instead, it solely undergoes side reactions, leading to the formation of allophanate groups. As a result, the chains become shorter, and the complex viscosity of the TPUs become lower than the TPU samples synthesized with one-step method. Ultimately, the inadequate growth of polyurethane chains will lead to a decline in mechanical properties.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.