Abstract
Poly (methyl methacrylate) (PMMA) and PMMA/poly (ethylene glycol) (PEG)-based composite fibers were produced using electrospinning technique with two different additives; halloysite nanotubes (HNTs) and boron phosphate (BPO4). The effect of the additives on the morphology, wettability and dye adsorption properties of electrospun fibers were investigated by Scanning Electron Microscopy (SEM), water contact angle measurement and UV-VIS spectrophotometry. The results of SEM analysis indicated that addition of PEG resulted in thinner average fiber diameter and beaded structure. Additionally, it was also observed that incorporating both HNT and BPO4 to the PMMA solution caused increased fiber diameter which might be due to increased solution viscosity. For PMMA fibers containing 3 wt% HNT, the highest average diameter and water contact angle were measured as 2204 ± 302 nm and 110.29 ± 3.9°, respectively. The produced fibers were tested for the capability to adsorb methylene blue (MB) and methyl orange (MO) from aqueous solutions. Dye adsorption capacity of the samples increased by addition of HNT and BPO4. According to adsorption capacity results, PMMA/PEG/BPO4 composite sample containing 5 wt% BPO4, with an adsorption capacity of 0.76 mg/g, was determined as adsorbent for kinetic and isotherm studies. The adsorption kinetics were well described by the pseudo-second-order model, and equilibrium data were correlated with the Langmuir model.
Highlights
• Poly (methyl methacrylate) (PMMA)-based composite fibers were produced by electrospinning process. • Average fiber diameter of PMMA fibers increased by addition of halloysite nanotubes (HNT) and boron phosphate (BPO4). • Compared with neat PMMA, thinner fibers and beaded structure were obtained by addition of poly (ethylene glycol) (PEG). • Dye removal capacity increased by addition of HNT and BPO4. • The rates of adsorption were found to comply with the pseudo-second-order kinetics. • Adsorption equilibrium data fitted well with the Langmuir model.
Introduction
Electrospinning is a straightforward, versatile, and cost-efficient technique for fabricating ultrathin polymer fibers. It is a well-known technology that was originally patented by Formhals in 1934 in order to develop continuous fibers of sizes changing from micro to nanoscale. Electrospun fibers suggest several advantages including a high surface-to-volume ratio, adjustable porosity, exceptional structural precision and the ability to control the fiber morphology and size to obtain the featured properties and functionality.1,2 Because of these advantages, electrospun fibers have been widely researched to be applied in several fields such as biomedical engineering, tissue engineering, biotextiles, regenerative medicine, optical and chemical sensors, textiles, and electronic devices.3,4 In electrospinning technique to produce fibers, polymer selection has an important role in determining fiber characteristics and their potential applications. Natural or synthetic polymers, each offering distinct advantages depending on the intended use, are both utilized. 5
Poly (methyl methacrylate) (PMMA), an amorphous polymer within the acrylate family, is known for its transparency and colorless appearance. It exhibits a glass transition temperature typically ranging from 100°C to 130°C. From a mechanical standpoint, PMMA is characterized by a relatively high Young’s modulus and low elongation at break. These properties contribute to its resistance to shattering upon failure. Moreover, PMMA is recognized as one of the most rigid thermoplastics, offering excellent scratch resistance.6,7 It is one of the most biocompatible materials, which makes it suitable for applications in fields of notably biomedical engineering as well as clinic dentistry. Because of its superior optical properties, it has a wide range of uses in electronic devices and the automobile industry.8,9 On the other hand, poly (ethylene glycol) (PEG) is a type of thermoplastic polyether compound that is also referred to as poly (ethylene oxide) (PEO) or polyoxyethylene, and it has also taken its place in electrospinning processes in recent years. Depending on its molecular weight, it can be in solid or liquid state. It is also commonly used as plasticizer to improve ductility, flexibility and reduce brittleness in polymer industry. PEG is an ideal polymer to enhance the wetting properties of electrospun fibers because of its hydrophilicity.10,11
In addition to polymer blending, the incorporation of functional additives into polymer matrices is a common approach to tailor and enhance specific material properties. Among naturally occurred inorganic nano-fillers, the halloysite nanotubes (HNTs), with high aspect ratios attract increasing attention from researchers. HNTs are promising nanofillers for a variety of applications because of their good thermal stability, high mechanical strength and biocompatibility properties. 12 Beyond their role in enhancing mechanical properties, HNTs have been shown to contribute to increase molecular adsorption. The most important advantage of HNTs as a mineral adsorbent is related to the lumen of their internal structure. HNTs have relatively lower hydroxyl group content on their surfaces, which imparts a more hydrophobic character when compared to other nanofillers.13,14 Moreover, boron-based fillers are extensively utilized as additives to improve the thermal stability at elevated temperature. BPO4, which is a very well-known compound, is an important catalyst, especially for organic reactions. These compounds exhibit high melting point (exceeding 1000°C). 15
As a result of the literature survey, it is seen that producing fibers using electrospinning method is widely studied, but usage of PMMA and its composites/blends in polymer composite fiber production by electrospinning is rare. Khanlou et al. 16 investigated the effects of polymer concentration and electrospinning parameters on the diameter of PMMA fibers produced by electrospinning. Based on the average fiber diameter measurements, it was determined that the most influential parameter was polymer concentration. Suryandari et al. 17 also produced PMMA fibers using the electrospinning technique. To control fiber morphology, structure, and diameter, various parameters such as polymer concentration, flow rate, tip-to-collector distance, and applied voltage were examined. The optimal conditions for producing PMMA fibers in their study were identified as 8% (w/v) polymer concentration, 20 kV applied voltage, a 15 cm distance between the nozzle tip and collector, and a solution flow rate of 0.004 mL/min. Additionally, Liu et al. 18 employed electrospinning to effectively fabricate PMMA nanofibers and PMMA/carbon nanotubes composite nanofibers. Their tensile stress–strain behaviour of single electrospun fiber was evaluated within the chamber of an environmental scanning electron microscope. The addition of carbon nanotubes led to notable enhancements in both Young’s modulus and tensile strength when compared to the unreinforced PMMA nanofibers.
Electrospun fibers/mats are considered as potential materials in many application areas where interactions with water are important. For instance, superhydrophobic membranes are used for oil–water separation processes, while hydrophilic variants are more favorable for tissue engineering due to their enhanced compatibility with cells. Chen et al. 19 produced PMMA/silica nanocomposite fibers containing up to 20 wt% silica by electrospinning. They found that addition of 20 wt% silica resulted in decrease of the average fiber diameter from 2.49 µm to 1.69 µm, which was attributed to significantly increasing in solution conductivity. They also observed that at 5 and 10 wt% silica, the contact angle remains nearly unchanged. A notable drop at 20 wt% is likely due to hydrophilic silica particles with silanol groups on the fiber surface. Liu et al. 20 studied the effect of solvent on wettability of PMMA membranes fabricated by electrospinning. The membranes exhibited a high water contact angle up to 153.9° and nearly zero oil contact angle. This superhydrophobic–superoleophilic surface structure was obtained by adjusting the weight ratio of binary solvent of N,N-dimethylacetamide and acetone.
Furthermore, numerous studies in the literature have demonstrated that electrospun fibers with nano/microscale structures possess remarkable potential for wastewater treatment applications. This is largely attributed to their high surface area, interconnected porous structure, surface modifications or the ability to incorporate functional additives. Koysuren and Koysuren 21 reported that the methylene blue (MB) adsorption of PMMA improved with increased surface roughness, and adsorption process was well described by the pseudo-first order model. On the other hand, Lee et al. 22 fabricated PMMA/zeolite nanofiber membrane and investigated the effect of process parameters, solution concentration and zeolite addition on the surface morphology and adsorption properties. They found that higher voltage reduced fiber diameter, while needle size and zeolite concentration had minimal effect on surface morphology. Moreover, the incorporation of zeolite into PMMA nanofibrous membranes enhanced the adsorption performance compared to pure zeolite, effectively achieving the removal of methyl orange (MO). Based on the kinetic analysis and model fitting, the adsorption behavior of the PMMA/zeolite nanofibrous membrane was best described by the pseudo-second-order kinetic model, while the equilibrium adsorption data exhibited a better correlation with the Langmuir isotherm model, indicating monolayer adsorption on a homogeneous surface of PMMA/zeolite membrane.
Although polymer composites nanofibers based on different inorganic additives have already been studied for electrospinning applications, the studies of PMMA-based composites with inorganic additives such as HNTs and BPO4, prepared by electrospinning technique are still rare. This study investigates PMMA-based electrospun nanofibers with either HNTs or BPO4, which remain underexplored, particularly for dye adsorption from water. The effects of these additives on fiber morphology, fiber diameter, dye adsorption, and wettability were evaluated. To improve flexibility and toughness, PMMA was also blended with PEG, and all fibers were characterized through morphological, adsorption, and wettability analyses.
Experimental
Materials
PMMA (Mw: 95.000 g/mol) (Altuglas V-825T) purchased from Hayim Pinhas A.Ş., Türkiye and molecular biology grade PEG (Mn: 6000 g/mol) obtained from Merck were the polymeric materials of the present work. Acetone (Sigma-Aldrich) and N,N-dimethylformamide (DMF) (Isolab Chemials) were used as solvents in the electrospinning process. HNTs with a chemical formula of Al2Si2O5(OH)4.2H2O provided from Sigma-Aldrich (685,445), and BPO4 which was synthesized by microwave technique 23 were used as additives. The length and average diameter of HNTs are 1-3 μm and 30-70 nm, respectively. 24 The average particle size of the BPO4 crystals were determined as 397 nm. 23 Before processing, HNTs and BPO4 were dried at 100°C for 5 hours. MB (Merck) and MO (Merck) were selected as dyes in this study.
Sample preparation
Formulations of the samples.

Electrospinning
The samples were prepared using an Inovenso NE 300 electrospinning machine with rotating drum collector. The optimum system parameters were determined according to our preliminary studies. The solution was transferred through a syringe at a feed rate of 0.4 mL/hr. The applied voltage, the distance between tip to collector and rotational speed of the drum collector were set as 25 kV, 15 cm and 100 rpm, respectively.
Characterization
The surface morphology of electrospun PMMA, PMMA/PEG and PMMA-based composite fibers was analyzed using a Scanning Electron Microscope (Tescan Vega 3). The Image J (Fiji) (NIH, USA) software was conducted to measure the fiber diameter. At least 100 diameter measurements were analyzed by taking three or four different parts of the fibers to determine the average fiber diameter. Viscosity measurements of the electrospinning solutions were performed with an Anton Paar Rheocompass parallel plate rheometer under 10-100 1/s shear rate range. Dynamic contact angle measurements of deionized water were analyzed using the Attension Theta Lite instrument. A 5 µL droplet was placed on the surface of the samples, and an image of the drop was captured by video camera. 3 droplets per sample were analyzed. Dye adsorption tests for the determination of the adsorption capacity were conducted for the 10 mg electrospun samples immersed into 10 mL MB or MO aqueous solution with a concentration of 3 mg/L for 24 hours. The absorbance values were determined with a UV–visible spectrophotometer (Specord S600) at 665 nm for MB and at 464 nm for MO to measure the concentration of dyes in aqueous solution. To examine the mechanism of adsorption kinetics, 5 mg/L dye concentration was used. Isothermal studies were conducted with different dye concentrations (1-5 mg/L) and 10 mg sample. Before the adsorption tests, all the samples were dried at 100°C for 5 hours.
Results and discussion
SEM and solution viscosity analyses
The SEM micrographs and the average fiber diameter distribution results of neat PMMA and PMMA/PEG blend are given in Figure 1. It was found that when the PEG was added to the polymer solution with a weight ratio of PMMA/PEG: 80/20, the average fiber diameter decreased to 1096 nm in consistence with the results given in the literature.
25
Jafarpour et. al
25
also observed that the addition of 20 wt% PEG led to a reduction in fiber diameter from 696 nm to 472 nm. It can be attributed to the low molecular weight of PEG which reduces the solution viscosity, as a result beads formation can be also observed due to a typical formation of low viscosity solutions as seen in Figure 1(b). SEM images and fiber diameter distribution of (a) Neat PMMA, (b) PMMA/PEG.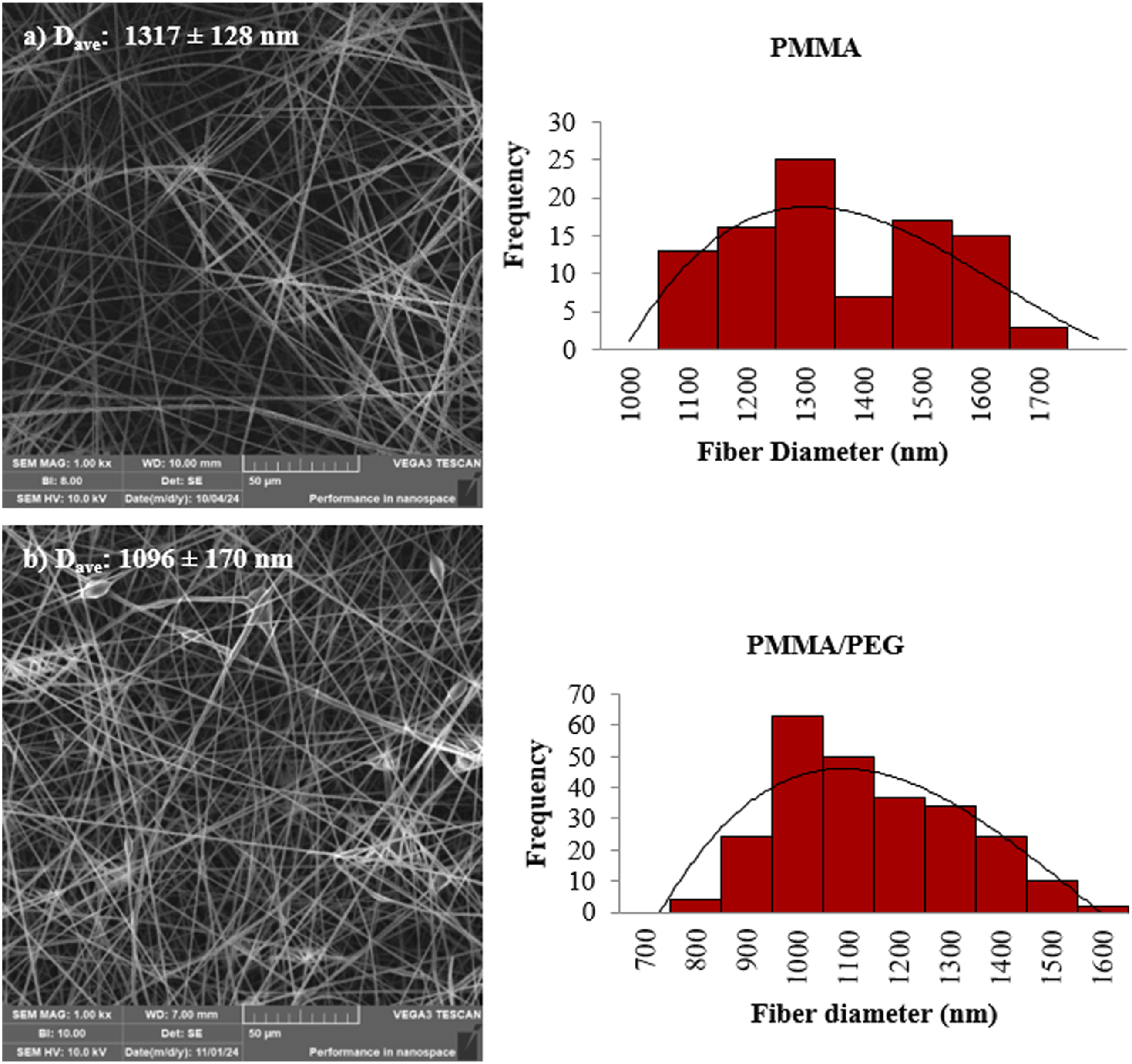
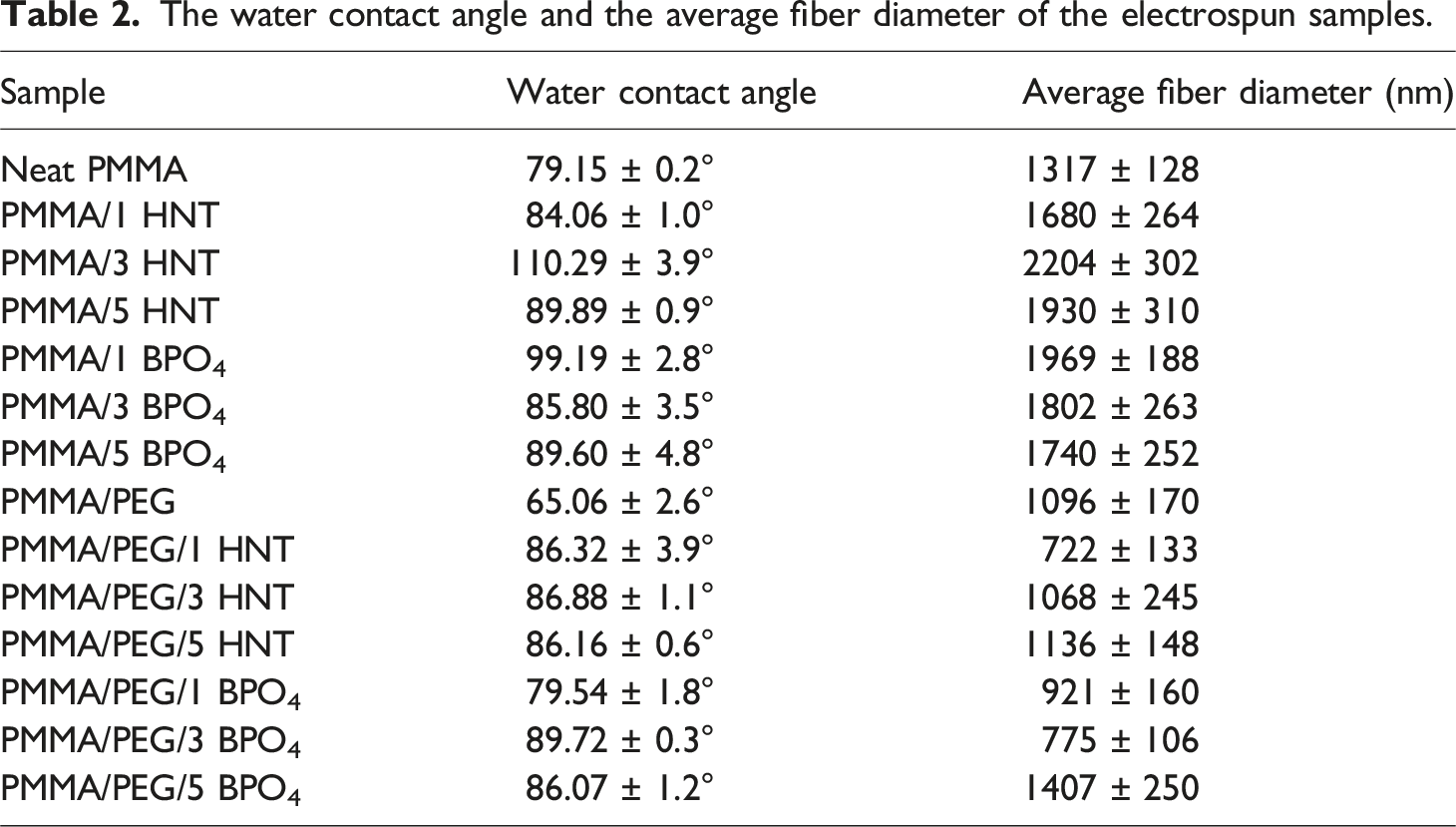
Figures 2 and 3 show the SEM micrographs and the fiber diameter distribution results of PMMA and PMMA/PEG electrospun fibers containing different concentrations of HNT and BPO4, separately. As can be understood from Figure 2 and Table 2, addition of HNT or BPO4 resulted in an increase in the average fiber diameter of neat PMMA as expected, this phenomenon can be attributed to enhanced solution viscosity due to additive addition.26,27 On the other hand, composite fibers containing HNT and BPO4 exhibited different trends in terms of average fiber diameters, considering with their standard deviations. The average diameters of the HNT-based fibers initially increased with the addition of HNT (1 and 3 wt%), followed by a subsequent decrease at higher concentrations. In contrast, the BPO4-based fibers exhibited a slightly decreasing trend in average fiber diameter with increasing BPO4 content. Moreover, considering the standard deviation of the average fiber diameter for both additives, especially at 3% and 5% weight ratio, it can be concluded that further additions do not have a significant effect on the fiber diameter. Specifically, it was observed that the samples containing 3 wt % HNT had the largest average fiber diameter. SEM images and fiber diameter distributions of PMMA/HNT and PMMA/BPO4 composite fibers with different weight percentages of HNT and BPO4 (a) PMMA/1 HNT, (b) PMMA/3 HNT, (c) PMMA/5 HNT, (d) PMMA/1 BPO4, (e) PMMA/3 BPO4, (f) PMMA/5 BPO4. SEM images and fiber diameter distributions of PMMA/PEG/HNT and PMMA/PEG/BPO4 composite fibers with different weight percentages of HNT and BPO4 (a) PMMA/PEG/1 HNT, (b) PMMA/PEG/3 HNT, (c) PMMA/PEG/5 HNT, (d) PMMA/PEG/1 BPO4, (e) PMMA/PEG/3 BPO4, (f) PMMA/PEG/5 BPO4. The water contact angle and the average fiber diameter of the electrospun samples.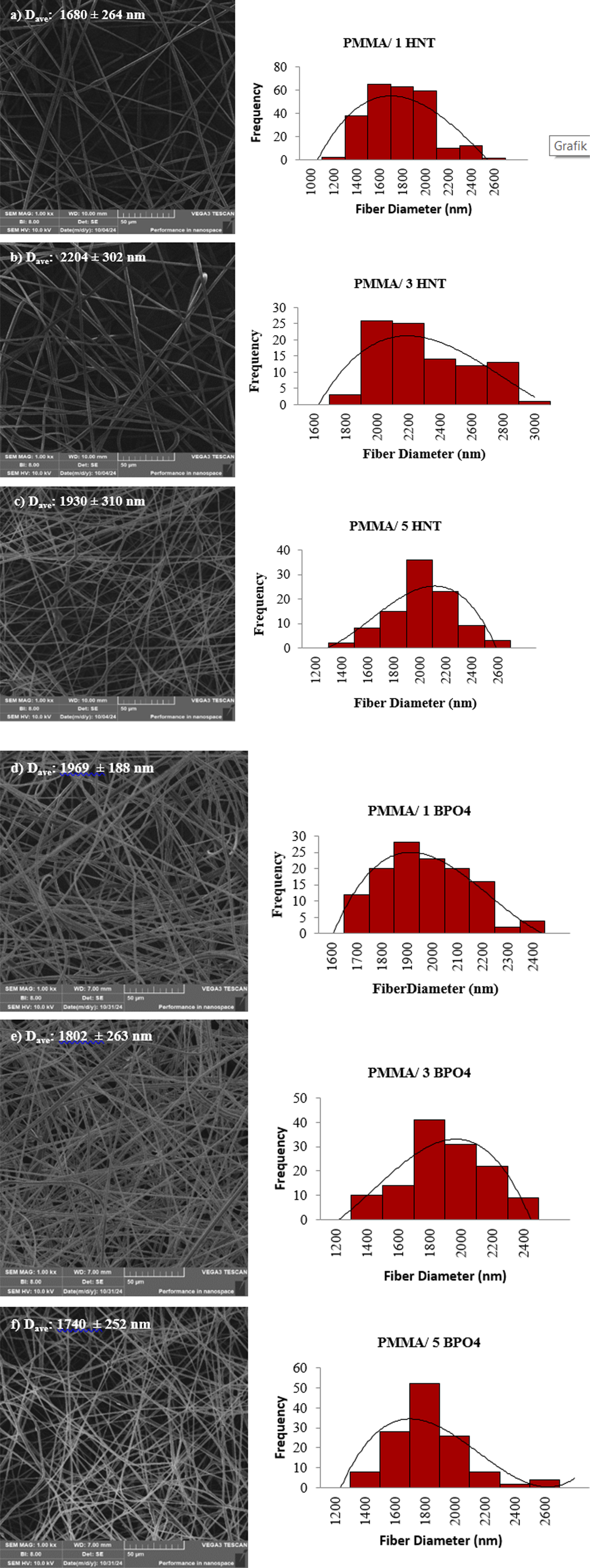
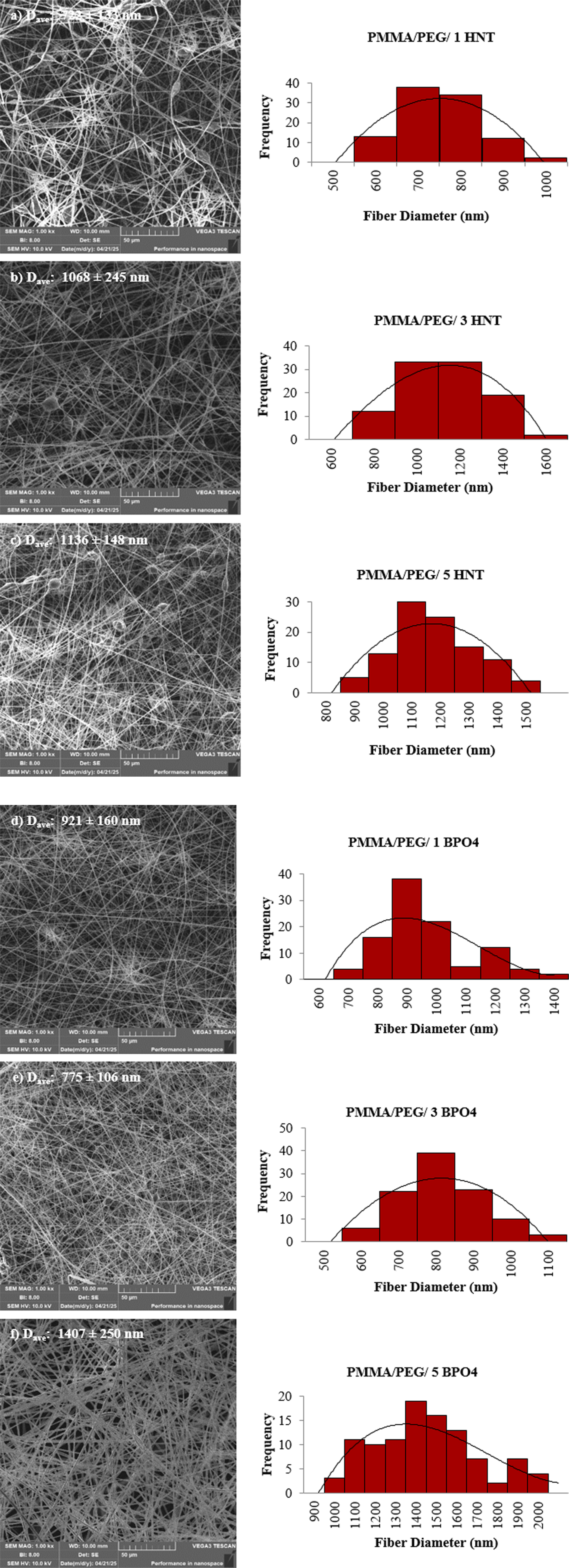
Considering Figures 2 and 3 together, it was observed that the average fiber diameters of the samples containing PEG were smaller although the same amount of HNT and BPO4 were loaded to the composites. Moreover, it can be easily seen from Figure 3 that the beaded structure (number of beads on the fibers) decreased significantly with the addition of BPO4. However, it still appeared with the addition of HNT for the PMMA/PEG samples. The average fiber diameter of the PMMA/PEG/5 BPO4 exhibited a significant increase when PMMA/PEG and composite fibers with lower BPO4 loadings were considered. Improved dispersion of smaller-sized fillers can lead to reduced electrospinning solution viscosity, resulting in finer fiber diameters. Conversely, at higher additive loadings (5 wt%), increased particle agglomeration may lead to poor dispersion and higher solution viscosity, producing thicker fibers when compared with PMMA/PEG sample.
All these morphological variations in terms of the average fiber diameters and beaded structures are thought to be a result of the variations in the electrospinning solution viscosity. The viscosity values of the selected electrospinning solutions are given in Figure 4. Viscosity values of the selected electrospinning solutions.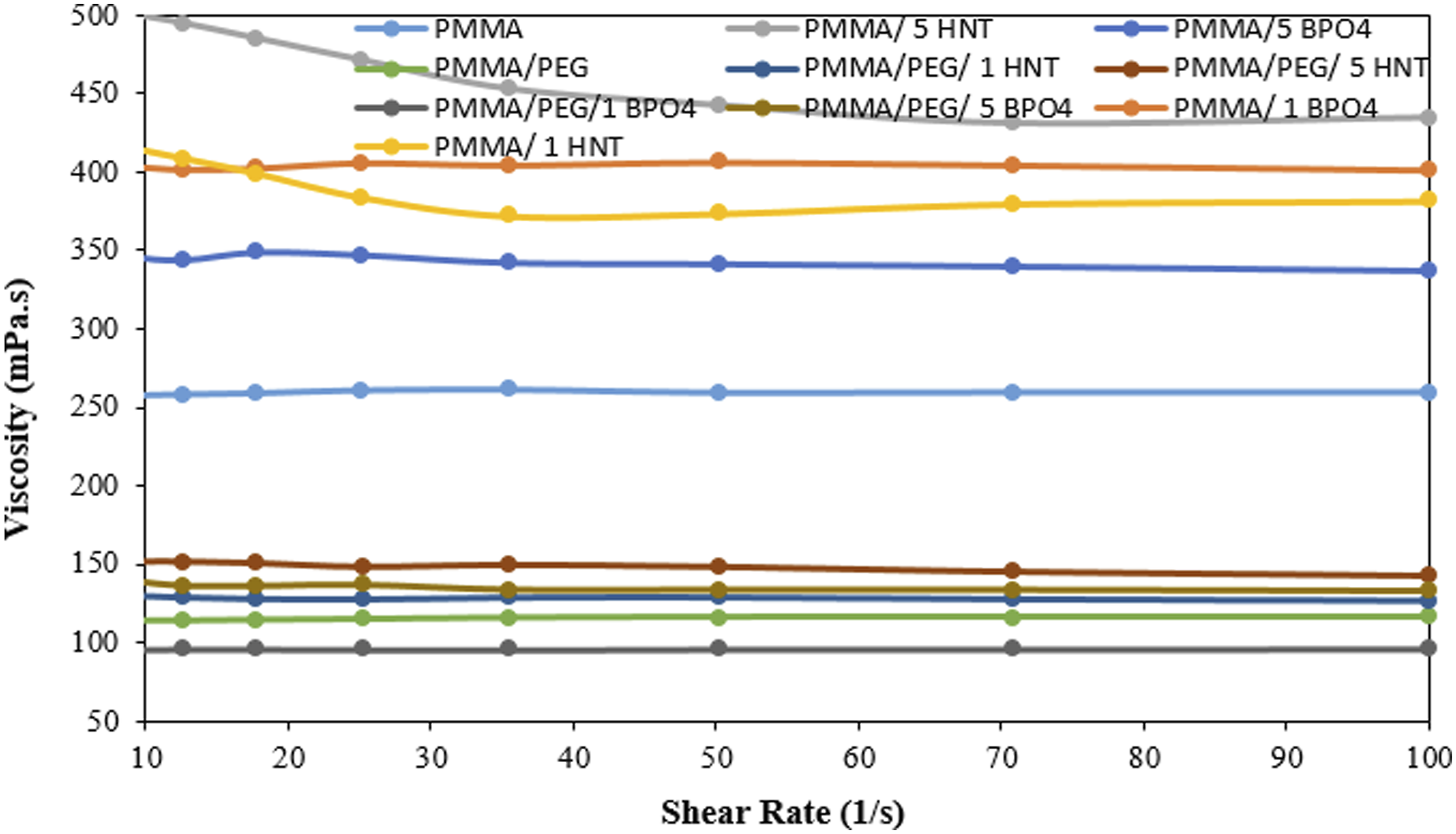
In general, the addition of fillers has been found to increase the viscosity, as confirmed by viscosity measurements. As shown in Figure 4, the incorporation of HNT and BPO4 into the PMMA matrix leads to an increase in viscosity. The viscosity of pure PMMA increased from approximately 250 MPa·s to about 450 MPa·s. Furthermore, the difference in the observed trends between these additives may be attributed to the distinct surface functionalities of the fillers and the resulting variations in their interfacial interactions with the polymer matrix, which influence chain mobility and flow behavior. Both HNT and BPO4 can immobilize PMMA chain segments at the filler interface via polar/hydrogen-bonding or acid-base interactions, thereby increasing solution/melt viscosity.28,29 However, HNT addition typically produces higher viscosity increases at comparable loadings (5 wt%) because of its hydroxylated surfaces combined with high aspect ratio and larger effective interfacial area, whereas the influence of BPO4 depends more strongly on its surface acidity, particle morphology and dispersion state. When comparing the composites containing 1 wt% of HNT and BPO4, it was observed that the BPO4 loaded solution exhibited a higher viscosity than the HNT loaded one. Consequently, the fibers produced from the BPO4 containing solution showed larger average diameters compared to those incorporating HNT.
Upon the addition of PEG, the viscosity was found to decrease by approximately 100 MPa.s, as expected. The decrease in viscosity upon the addition of PEG can be attributed to its plasticizing effect on the polymer matrix. PEG molecules, having flexible ether linkages and good compatibility with PMMA, tend to insert themselves between the PMMA chains, reducing intermolecular forces such as van der Waals interactions and hydrogen bonding among the polymer chains. 30 This increased free volume and chain mobility lead to a reduction in solution viscosity. When evaluating the fiber diameters, it was observed that in PMMA-PEG-based composites, the fiber diameter decreased at lower filler concentrations (1 and 3 wt%) for both types of fillers, while it increased at a higher filler concentration (5 wt%). Comparison of the viscosity values with the fiber diameters revealed that, at 5 wt% loading, the viscosity increased for both fillers; however, at 1 wt% loading, the presence of BPO4 led to a decrease in viscosity, whereas the inclusion of HNT resulted in an increase. When the filler content is very low, BPO4 particles are usually well-dispersed in the polymer solution. The strong filler–solvent interactions (due to the polar surface of BPO4) improve wetting and minimize agglomeration, thus reducing internal friction compared to systems where fillers interact strongly with each other. Moreover, the decrease in the fiber diameter at lower filler concentrations in the presence of PEG, which is also a nonionic surfactant, can also be explained with the improved dispersion of additives with lower particle size at lower concentrations of additives. Nonionic surfactants can improve the dispersion of additives and lower the particle sizes of agglomerates in polymer composites by covering the surfaces of the additives and decreasing the interparticle interactions between them. 31
Water contact angle
Surface wettability can be assessed by measuring the contact angle formed between a liquid droplet and the material surface. Surfaces exhibiting contact angles between 0° and 30° are considered highly hydrophilic, indicating strong affinity for water. As the contact angle increases towards 90°, the surface becomes less hydrophilic. Conversely, when the contact angle exceeds 90°, the material is regarded as hydrophobic, reflecting a lower tendency to interact with water. 32
Several factors influence the contact angle formed between a liquid and a solid surface. The surface tension of the liquid plays a significant role; liquids with higher surface tension typically form larger contact angles, especially on hydrophobic surfaces. 33 Surface roughness also affects wettability according to the Wenzel and Cassie-Baxter models, either enhancing or reducing the apparent contact angle depending on the nature surface. 34 The chemical composition of the surface, including the presence of functional groups or coatings, can make a surface more hydrophilic or hydrophobic, thus altering the contact angle. Additionally, the duration of contact between the liquid and the solid can lead to spreading or adsorption, affecting the angle over time.
The water contact angle of the samples and their average fiber diameters are listed in Table 2. The representative images of produced samples are also shown in Figure 5. The water contact angle for neat electrospun PMMA fibers was determined as 79.15 ± 0.2°, which implies that it is a hydrophilic material (contact angle < 90°). Although the obtained contact angle value is within the range of values stated for PMMA in the literature, there are studies with different results on whether PMMA is hydrophobic or hydrophilic.21,25,32,35 Water contact angle images of (a) PMMA, (b) PMMA/1 HNT, (c) PMMA/3 HNT, (d) PMMA/5 HNT, (e) PMMA/1 BPO4, (f) PMMA/3 BPO4, (g) PMMA/5 BPO4 (h) PMMA/PEG, (i) PMMA/PEG/1 HNT, (j) PMMA/PEG/3 HNT, (k) PMMA/PEG/5 HNT, (l) PMMA/PEG/1 BPO4 (m) PMMA/PEG/3 BPO4, (n) PMMA/PEG/5 BPO4.
The contact angles of the PMMA-based composite fibers containing HNT or BPO4 as additives, are higher than the neat PMMA which may be attributed to increased surface roughness. 35 The average fiber diameters of the electrospun PMMA fibers increased by incorporating inorganic additives, consequently increased the roughness. In addition, the highest water contact angle was measured for the sample of PMMA/3 HNT which has the largest average fiber diameter. HNTs have low hydroxyl group density on their surface, and tubular aluminosilicate structure with large specific surface area and nanotube structure which is a promising candidate for constructing rough surface in micro-and nano-scale. 36 Similar trend between average fiber diameter and contact angle was also observed for the fibers containing BPO4, compared to neat PMMA. On the other hand, the incorporation of PEG into the PMMA fibers enhanced their hydrophilicity, as evidenced by the reduced water contact angle of 65.06 ± 2.6° as shown in Figure 4(h). PEG is known for its strong hydrophilic nature, attributed to its repeating -CH2CH2O- units. This structural feature allows it to interact readily with water molecules through hydrogen bonding. 37
Furthermore, addition of HNT and BPO4 to the PMMA/PEG system resulted in increased water contact angle at the beginning of the measurement. For all the samples containing PEG, it was observed that the water droplet diffused inside the samples. In addition to its hydrophilic properties, PEG may also contribute to the improved porosity of the electrospun sample due to its surfactant effect. In Figure 6, the change of water contact angle after 10 s can be seen for the PMMA/PEG/5 BPO4 sample. Water contact angle of the PMMA/PEG/5 BPO4 sample (a) t = 0, (b) t = 10s.
Dye adsorption
The adsorption capacities of the produced fibers which are calculated from the UV-vis measurement at representative wavelengths in 3 mg/L MB and MO solutions are given in Figure 6. The adsorption capacity q (mg/g) was calculated using the following equation
21
:
It was expected that systems containing PEG will give better results in adsorbing dyes in aqueous solutions, due to the hydrophilicity of PEG. This phenomenon was observed in adsorption tests and samples containing PEG showed higher adsorption capacities when compared with those of the samples that do not contain PEG with same amount of additives. Moreover, the adsorption capacity of the samples increased as the additive amount in the composite fibers ascended. The reason for this can be the higher fiber diameter that is observed in SEM analyses (Figures 2 and 3) but it was thought that the main reason may be the increased surface roughness of the fibers at higher additive loadings which was also confirmed in contact angle measurements (Figure 5 and Table 2). The porous structure between the fibers may be enhanced by the addition of additives that may be another reason of the increased adsorption capacity. It was observed that PMMA/PEG/5 HNT system has the highest adsorption capacity for both dye types. However, it was observed that, the adsorption capacity of both PMMA and PMMA/PEG-based samples increased with increasing BPO4 concentration, that was not observed for HNT. Therefore, the PMMA/PEG/5 BPO4 sample, which exhibited the highest adsorption capacity, was selected for further kinetic and isotherm studies. On the other hand, although PMMA and PMMA/PEG-based systems had a considerable adsorption capacity at 5 wt% HNT loading for the MO, it was decided to not continue with that dye for further analyses since its removal was not as efficient as MB.
When Figure 7 is examined, it can be seen that the highest adsorption capacities obtained are around 0.8 mg/g. In the literature, however, Koysuren and Koysuren
21
reported adsorption capacities ranging from 5 to 7 mg/g in their study, where adsorption tests were carried out using electrospun PMMA having different molecular weight. The adsorption capacity of electrospun samples for methylene blue and methyl orange.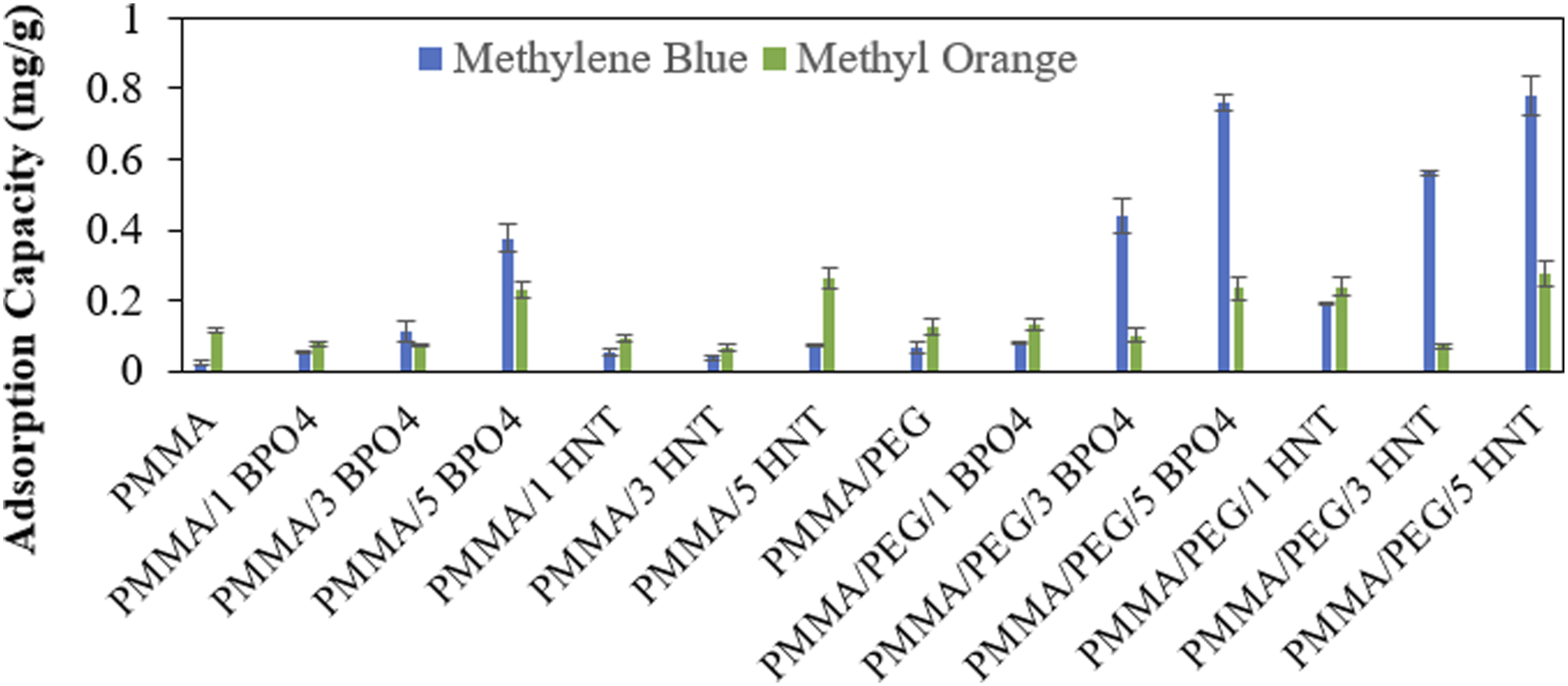
Adsorption kinetics
Figure 8 illustrates the effect of contact time on adsorption capacity of PMMA/PEG/5 BPO4 composite fibers for methylene blue dye solution. The adsorption capacity change was higher during the first 2 hours and then started gradually decreasing until it plateaued. This could be attributed to the porous structure with large internal surface area of the fibrous composite that facilitates dye adsorption. Thereafter, only a slight improvement in dye removal was observed after 6 hours. Effect of contact time on adsorption capacity of PMMA/PEG/5 BPO4 composite fibers.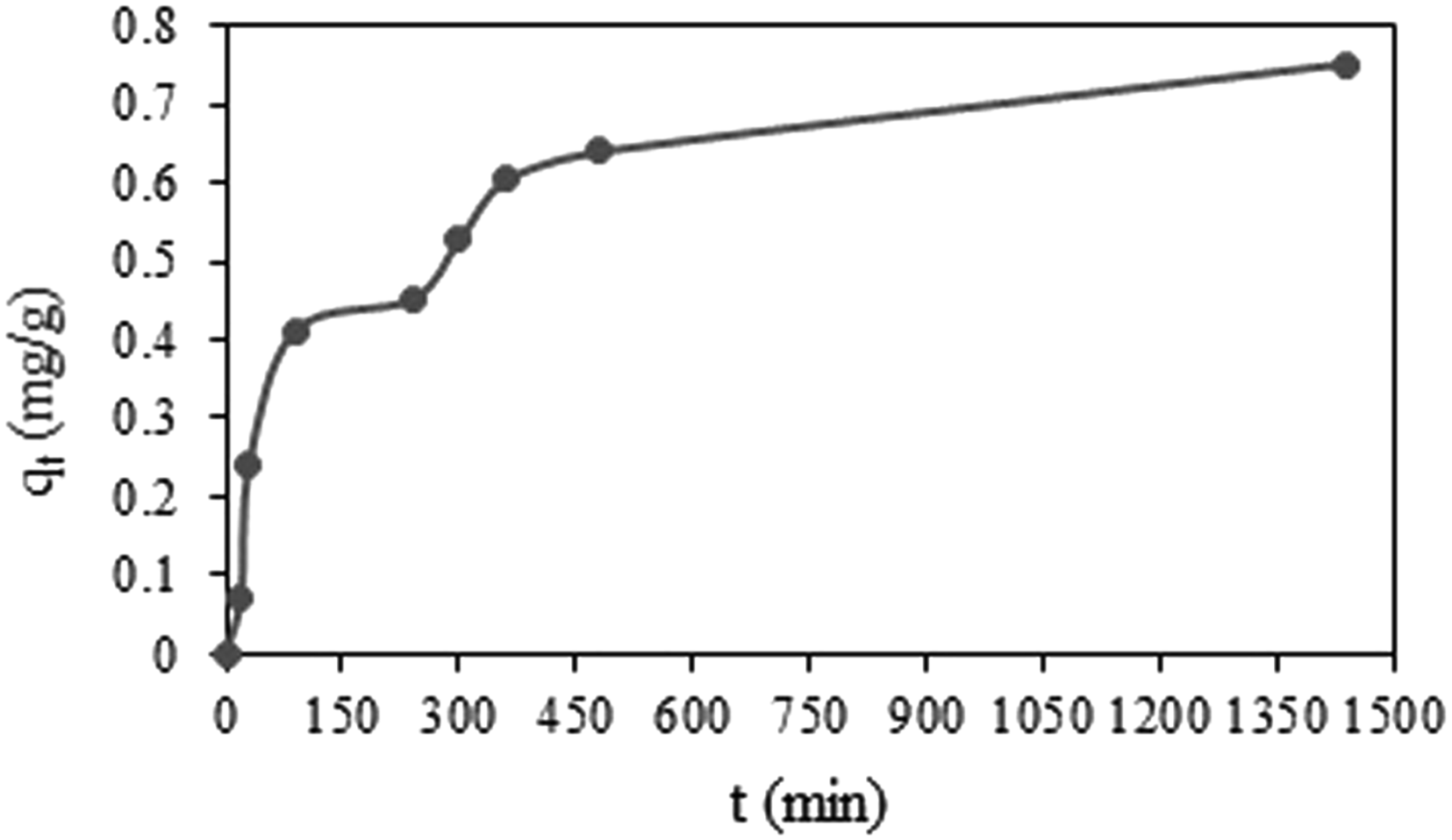
To analyze the mechanism and determine the rate-limiting step of the overall adsorption process, the adsorption behavior is evaluated using three kinetic models: the pseudo-first- order model, the pseudo-second-order model, and the intra-particle diffusion model.
The pseudo-first-order and pseudo-second-order models can be expressed as the following equations
38
: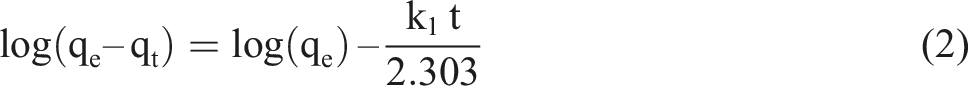


Kinetic plots for three models are given in Figure 9. The calculated values of parameters k1, k2, kp and qe and the correlation coefficients of the plots were tabulated in Table 3. Based on the comparison of correlation coefficient values, the pseudo-second-order model provided a more accurate representation of the adsorption process. In addition, the adsorption capacity value calculated according to the pseudo-second-order model, which closely aligns with the experimentally obtained one, also supports this result. Kinetic plots of (a) the pseudo-first-order model (b) pseudo-second-order model (c) intra-particle diffusion model. Kinetic parameters for MB on PMMA/PEG/5 BPO4.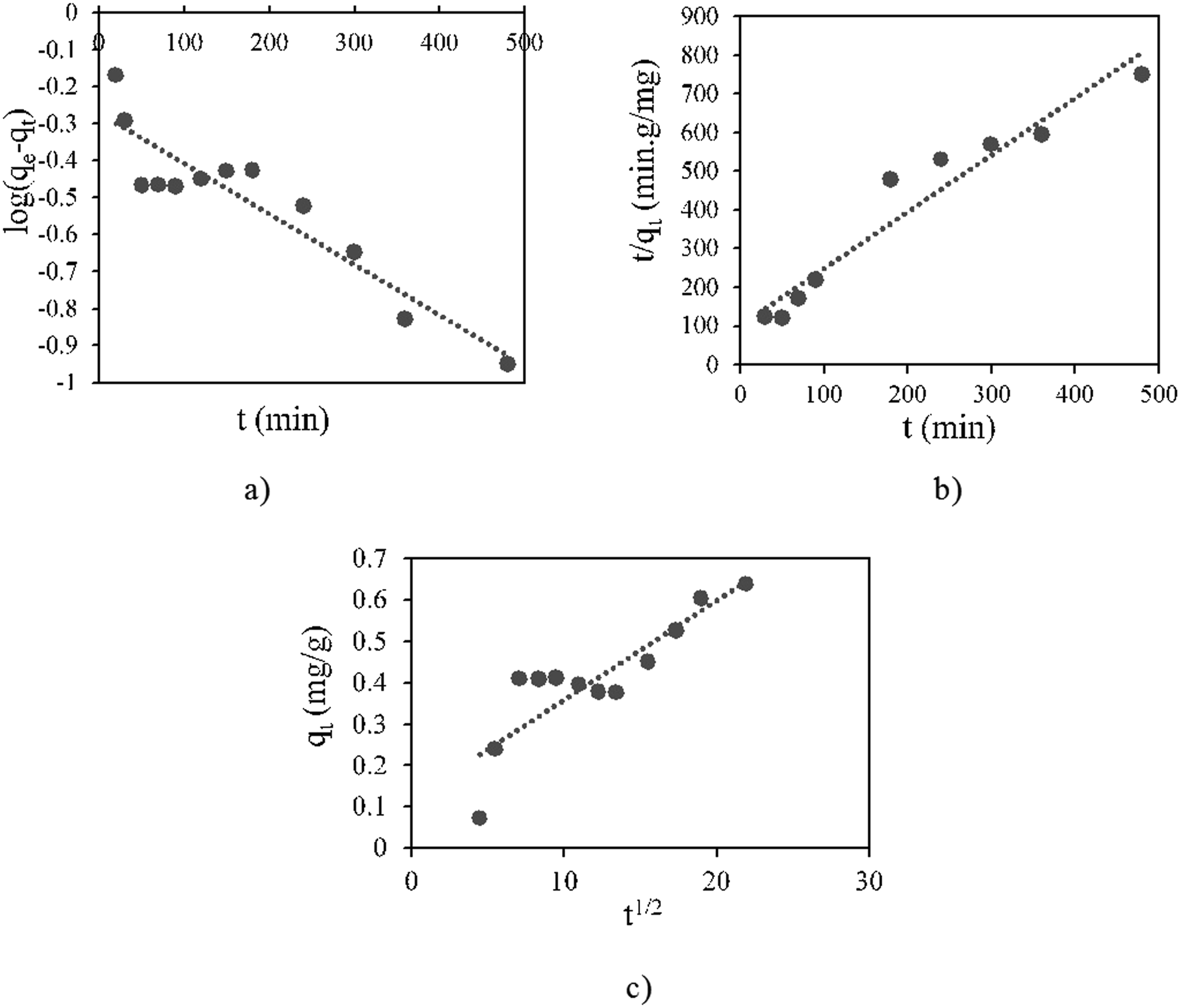

The conformity of the kinetic data to the pseudo-second-order model suggests that the rate-limiting step is primarily controlled by chemisorption, involving valence forces through the sharing or exchange of electrons between the adsorbent and adsorbate molecules. This implies that the adsorption process is governed by chemical interactions rather than simple physical diffusion.
Adsorption isotherms
Adsorption isotherm models are generally used for designing an adsorption mechanism. The well-known Langmuir isotherm model was applied to investigate the equilibrium isotherms for the adsorption of MB by electrospun PMMA/PEG/5 BPO4 fibers (Figure 10). Isotherm plot of Langmuir model.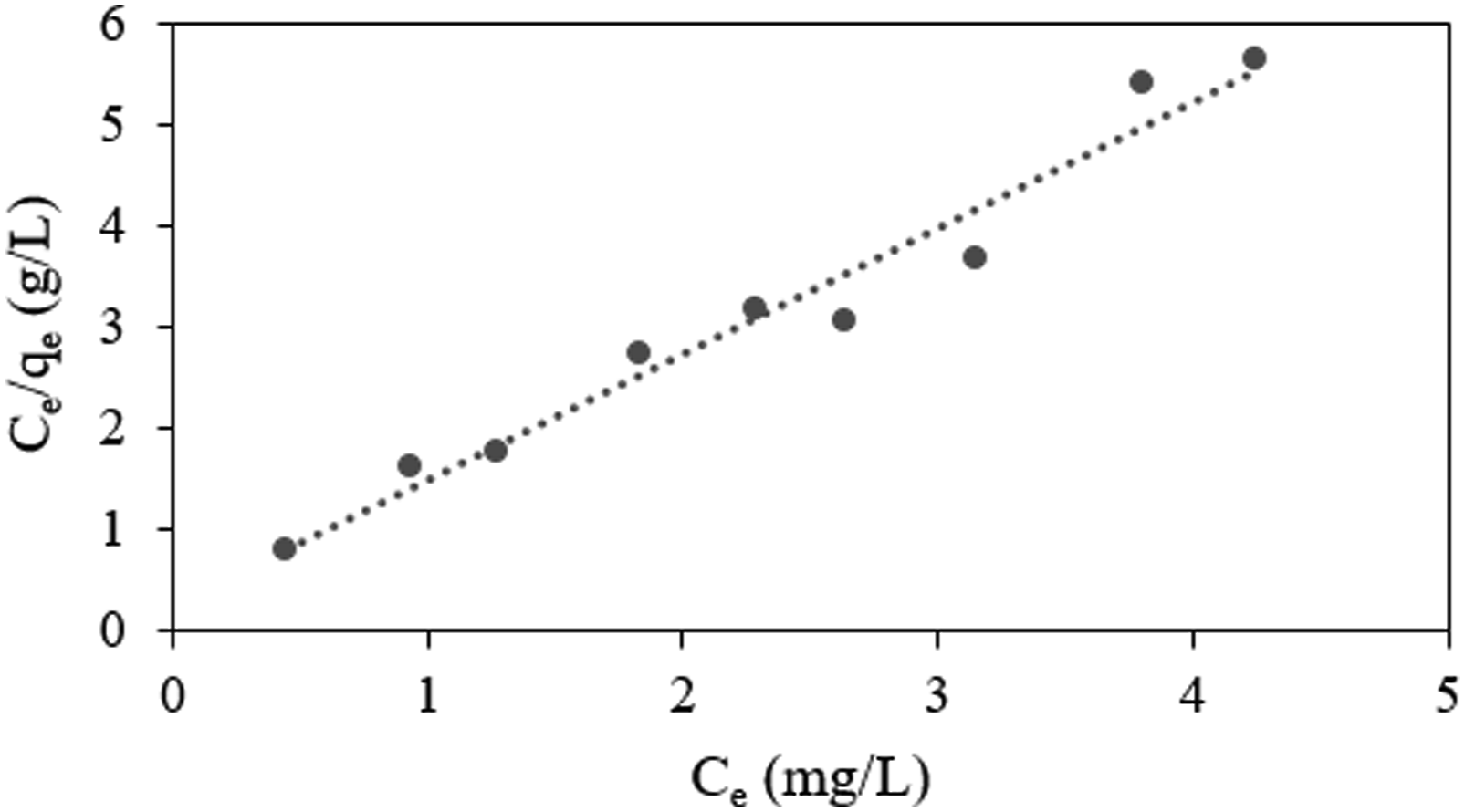

Isotherm parameters of MB on PMMA/PEG/5 BPO4.

The favorability of an adsorption process can be evaluated using a dimensionless constant referred to as the separation factor or equilibrium parameter (RL). This parameter is determined through the application of the following equation
14
; Equilibrium parameters (RL) for the adsorption of MB.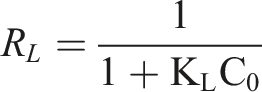
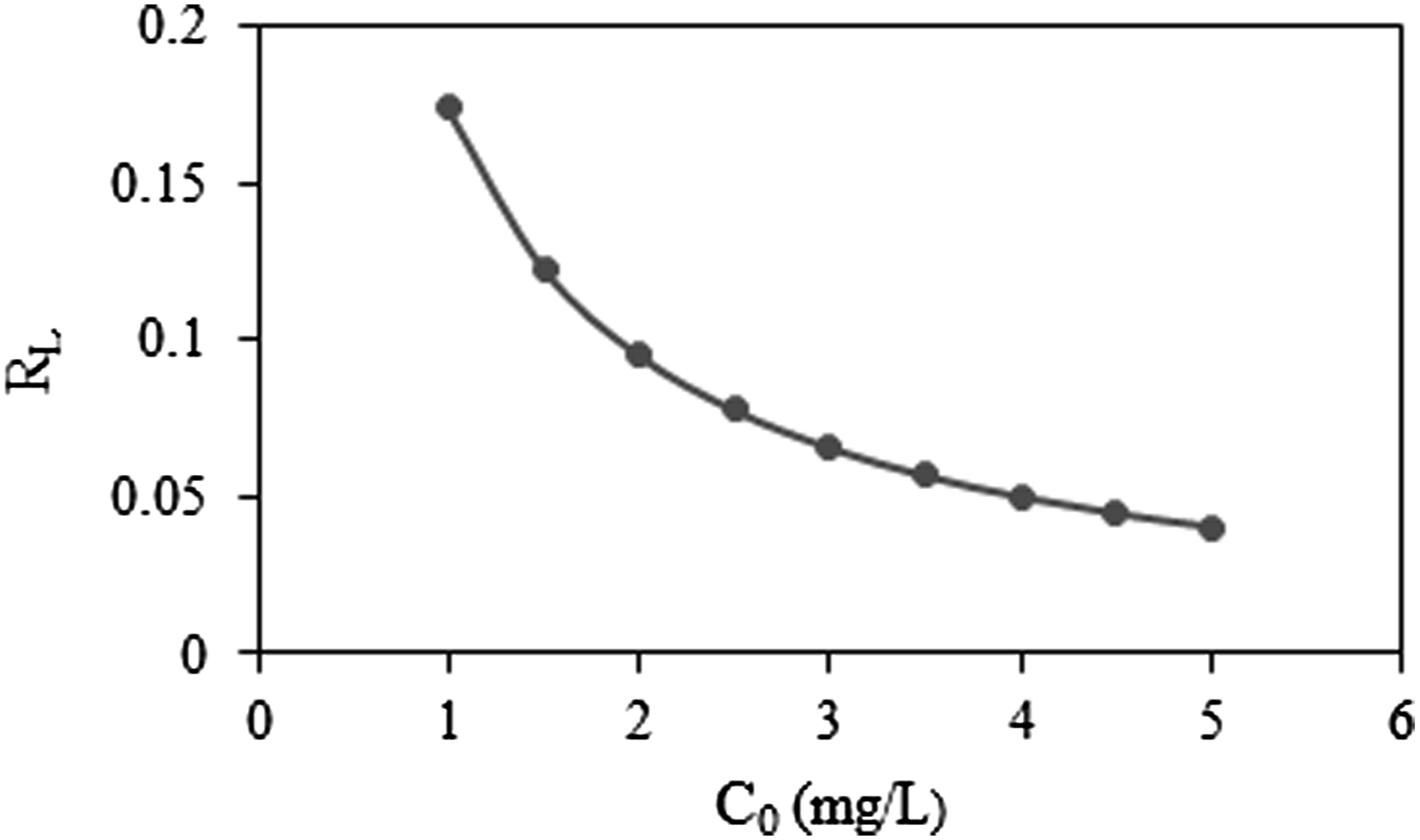
The correlation coefficient (R2) indicates that the adsorption equilibrium data of MB onto PMMA/PEG/5 BPO4 fibers align closely with the Langmuir isotherm model. This outcome implies that the adsorption occurs at distinct, uniform sites on the adsorbent, leading to the formation of a monolayer of MB molecules on its surface. 41 Moreover, the finding that the adsorption mechanism follows monolayer adsorption as described by the Langmuir model aligns with the observed increase in adsorption capacity associated with enhanced surface roughness.
Conclusions
In this study, PMMA and PMMA/PEG-based electrospun composite fibers were successfully fabricated using electrospinning method. Acetone/DMF with a weight ratio of 1/3 was used as a solvent system. To enhance the properties of the produced fibers, HNT and BPO4 were added to the PMMA and PMM/PEG polymer solutions. SEM analyses demonstrated that addition of HNT and BPO4 into the PMMA matrix led to an increase in the average fiber diameter. Upon incorporation into the PMMA/PEG polymer system, both types of additives resulted in a reduction in the average fiber diameter at 1 wt % and 3 wt% loadings compared to the neat PMMA/PEG. However, at a higher concentration of 5 wt %, the average fiber diameter increased. Water contact angle measurements showed that the neat PMMA fibers have hydrophilic properties and the hydrophilicity enhanced by incorporating PEG to the system. The highest water contact angle was observed in PMMA composite fibers containing 3 wt% HNT, which also exhibited the largest average fiber diameter. Dye adsorption capacities of the electrospun samples from the aqueous solutions were studied using two different dyes: MB (anionic) and MO (cationic). The findings demonstrated that the samples possessed an enhanced affinity for MB adsorption. Composite fibers containing BPO4 revealed higher adsorption capacity for both PMMA and PMMA/PEG matrices. Consequently, the PMMA/PEG/5 BPO4 was selected for further adsorption isothermal and kinetic studies. Equilibrium data was well fitted with the Langmuir model. The adsorption rates were found to follow pseudo-second-order kinetic behavior, exhibiting a strong correlation with the model.
Footnotes
Acknowledgements
The authors would like to thank Dr Cemal Merih ŞENGÖNÜL and Dr Salih ERTAN from Atılım University for their support regarding the water contact angle and UV-Vis spectrophotometer measurements. We would also thank Miss Toprak ÇAĞLAR for her help during the SEM analysis.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
