Abstract
Halloysite nanotubes (HNTs) are fascinating carriers for the delivery of chemotherapy drugs. Surface modification of HNTs and their loading in polymer matrixes can create a delivery system with more controlled and sustained drug release. Taking this into account, in the present research, a drug delivery system was made by grafting a block copolymer of polyacrylic acid (PAA)/polyaniline (PANI) on HNT surface and incorporating copolymer-grafted HNTs into the polycaprolactone (PCL) fibers. For this purpose, PANI-b-PAA copolymer was first formed on HNT surface by grafting from strategy. Then, the copolymer-grafted HNTs and doxorubicin were loaded into PCL solution and the composite solution was processed by electrospinning. Preliminary evaluations confirmed the successful grafting of PANI-b-PAA copolymer onto HNTs. SEM and EDS analyses showed that drug loaded composite nanofibers have an average diameter of 396 nm and a uniform distribution of HNTs and doxorubicin. Drug release study revealed that composite nanofibers have less burst release and more sustained release than PCL nanofibers. Investigation of drug release mechanism by kinetics models corroborated that the drug release from composite nanofibers is mainly controlled by Fickian diffusion. Cell culture experiment verified that the composite nanofibers have higher cytotoxic effects and kill more tumor cells compared to PCL nanofibers. In summary, modifying the surface of HNT and incorporating it into PCL nanofibers can create a drug carrier with more sustained drug release and higher antitumor effects.
Highlight
- HNTs were modified with PANI-b-PAA copolymer. - Copolymer-modified HNTs and doxorubicin were loaded in PCL fibers. - Composite drug carrier showed more sustained drug release. - Composite drug carrier showed greater antitumor effects.
Introduction
Cancer is one of the most important causes of death worldwide, and cancer treatment is one of the most imperative challenges of the health system.1–3 Chemotherapy, radiotherapy, photodynamic therapy, and immune therapy are the main approaches used clinically to treat cancer.4–7 However, the insufficient efficiency of these approaches, as well as their adverse effects on non-cancerous tissues, have turned attention to new methods of cancer treatment. Chemotherapy using controlled and sustained release systems is one of the most fascinating approaches to maximize treatment efficiency and minimize the side effects of cancer therapy.8–10 These drug delivery systems gradually release their therapeutic payload and mitigate the need for frequent drug administration and markedly reduce the required drug dose. The most important aspect in drug delivery systems is designing a proper carrier.11,12
This is of particular importance for the delivery of chemotherapy drugs. These carriers must be designed to have the greatest effect on cancer cells and the least effect on non-cancerous tissues. These carries should be able to pass through biological barriers including immune cells, spleen, liver and kidney, and remain in the bloodstream for a longer time and even reach the tumor site. The size, shape, surface charge, elasticity, and hydrophilicity/hydrophobicity of the drug carriers are the most important parameters affecting their interaction with the biological environment. Controlling these parameters can prolong their blood circulation, increase their accumulation in tumor site through the enhanced permeability and retention (EPR) effect, increase their cellular uptake, and control their release profile. Nanocarriers in the range of 20-200 nm can bypass the existing biological barriers to drug delivery and those in the range of 100-200 nm can benefit from the EPR effect and accumulate at the tumor site. Non-spherical carriers like cylindrical, disk-like, oval, rod-like, and so on can remain in the blood stream for a longer time and are better absorbed by the cancer cells. The carriers with negative surface charge can circulate for a longer duration and are better in terms of systemic non-toxicity. While, the carrier with positive surface change can better uptake by the cells and are better in terms of therapeutic efficiency. Increasing the hydrophilicity of carriers reduces the adsorption of plasma proteins on their surface, decreases the phagocytosis by macrophage, and prolongs their residence time in the blood circulation, but prevents them from entering cancer cells. Therefore, by controlling the parameters of the drug carrier, the antitumor effect of the drug can be increased and its adverse effects on normal cells can be reduced.13–16
Halloysite nanotubes (HNTs) are one of the most appealing carriers for the delivery of the chemotherapy drugs as well as a wide range of other therapeutic agents.17–20 HNTs are nanoclay particles with a hollow tubular structure and a high surface area. It is possible to modify the inner and outer surfaces of HNTs and to decorate different functional groups on HNTs.21,22 These functional groups enable further surface modification and provide anchorage points for the attachment of polymer chains to HNTs. Functional groups and polymer chains on HNTs not only can control the encapsulation and release of drug but also can regulate the biodistribution and cell internalization of the drug carrier. Loading HNTs in polymeric matrixes can provide more control over drug release.23–25 Besides, after the completion of drug release, the polymer matrix can perform as a tissue engineering scaffold for the regeneration of damaged tissue.26,27 This issue is especially important when the tumor is surgically removed and then chemotherapy is done with the aim of the complete eradication of cancer cells and the prevention of tumor recurrence.
Electrospun fibers are one of the most interesting polymer matrixes for loading HNTs. These fibers facilitate drug release due to their high surface area to volume ratio and promote tissue regeneration thanks to their nanofibrous structure and similarity to the natural extracellular matrix (ECM).28–30 The incorporation of HNTs into electrospun matrixes can create composite drug delivery scaffolds with better drug release performance. Since these scaffolds provide multiple barriers against the diffusion of drug, they prolong drug release and prevent burst drug release and its adverse effects on non-cancerous tissues. Various studies have loaded HNTs into electrospun fibers and developed composite drug delivery scaffolds for a number of therapeutic agents. In this regard, Silva et al. 31 developed a drug delivery scaffold with a combination of alginate/polyvinyl alcohol (PVA) matrix and HNTs for the sustained release of cephalexin and showed that the composite scaffold had a more sustained drug release compared to drug loaded HNTs. Bulbul et al. 32 designed a drug delivery scaffold based on polycaprolactone/poly ethylene oxide (PCL/PEO) matrix and surface-modified HNTs for curcumin delivery and confirmed that the scaffold with surface-modified HNTs had higher drug encapsulation and slower drug release and caused more death in breast cancer cells. Carvalho and Coimbra 33 developed a drug delivery scaffold for the simultaneous delivery of dexamethasone and gentamicin sulfate using PLA matrix and HNTs and corroborated that the release of both drugs from the composite fibers was more sustained than that from the fibers alone. Joshi et al. 34 fabricated a composite scaffold consisting of polyurethane matrix and HNTs for the sustained delivery of chlorhexidine acetate and revealed that the composite released the drug in a more controlled manner compared to the nanofibers. Recently, Jaberifard et al. 35 made a composite scaffold including eudragit/soy protein matrix and HNTs for the simultaneous delivery of zinc oxide and allantoin and indicated that the composite fibers released both components more slowly than the fibers.
In the present research, a drug delivery scaffold was made based on polycaprolactone (PCL) electrospun fibers and surface-modified HNTs. This scaffold was loaded with a well-known chemotherapy drug, doxorubicin. Doxorubicin is an anthracycline that kills cancer cells by disrupting the function of topoisomerase II, an enzyme essential for DNA replication. Doxorubicin is commonly used as a frontline chemotherapy agent to treat a wide range of cancers. For the first time, in this research, HNTs modified with a block copolymer of polyacrylic acid (PAA) and polyaniline (PANI) were loaded into electrospun fibers. The copolymer was attached on HNT surface by a grafting from strategy. For this purpose, the HNT surface was first modified with a silane component, next a chain transfer agent was attached on the surface, and PANI-b-PAA was then formed on the surface by a combination of reversible addition fragmentation chain transfer (RAFT) and oxidative polymerizations. The copolymer-grafted HNTs and doxorubicin were loaded into PCL solution and converted into composite fibers by electrospinning.
Materials and methods
Materials
Polycaprolactone (PCL, average Mn 80,000), halloysite nanotubes (HNTs, diameter of 30-70 nm, length of 1-3 μm, pore volume of 1.26–1.34 mL/g, and specific surface of 64 m2/g), acrylic acid, (3-chloropropyl)triethoxysilane (CPTS), carbon disulfide, potassium hydroxide, xylene, ammonium persulfate (APS), hydrochloric acid, and diethyl ether were purchased from Sigma-Aldrich (USA). Aniline, sodium hydroxide, acetone, chloroform, tetrahydrofuran (THF), and ethanol were obtained from Merck (Germany). Aniline was purified by double distillation under vacuum. Azobisisobutyronitrile (AIBN) was acquired from Thermo Fisher Scientific (USA) and was recrystallized twice from methanol. Other materials were used as received.
Fabrication of PANI-b-PAA grafted HNT
To graft PANI-b-PAA on HNTs, several steps were performed: preparation of silane modified HNTs, synthesis of chain transfer agent, immobilization of CTA on HNT surface, RAFT polymerization of acrylic acid and copolymerization of aniline with acrylic acid.
To prepare silane modified HNTs, HNTs were dispersed in xylene through a probe sonicator. Then, CPTS was added to the dispersion and stirred at 80°C for 12 h. The dispersion was then filtered, the resulting precipitate was dispersed in toluene and dried in an oven.
In this research, sodium ethyl xanthate, as a chain transfer agent (CTA), was synthesized as described in detail elsewhere. 36 To synthesis CTA, briefly, ethanol was added to NaOH solution (25 M) with a volume ratio of 13:4. Then, carbon disulfide solution (10 M) was gradually added to the previous solution with a volume ratio of 10:17 and the resulting solution was continuously stirred. After 12 h, the precipitate obtained was washed successively with water and HCl and then passed through a magnesium sulfate column to remove moisture. The obtained material was dissolved in hexane, recrystallized, and finally dried in an oven.
To immobilize CTA on HNT surface, the silane modified HNTs were dispersed in CTA solution through a probe sonicator. The resulting dispersion was stirred at 80°C for 12 h and then filtered, washed with ethanol and dried in an oven.
For RAFT polymerization of acrylic acid, CTA immobilized HNTs were dispersed in THF through a probe sonicator. Then, acrylic acid and initiator, AIBN, were added to the dispersion and the dispersion was continuously stirred for 70°C. After 12 h, diethyl ether was added to the mixture to precipitate the reaction product. The resulting precipitate was dried in an oven.
For the copolymerization of aniline with acrylic acid, PAA grafted NHTs were dispersed in HCl through a probe sonicator. Then, aniline and initiator, APS, were added to the dispersion and the dispersion was stirred for a while. When, the color of the reaction mixture turned green, the polymerization was finished. The resulting product was washed several times with water/ethanol mixture and centrifuged. Then, the supernatant was collected and dried in an oven. The obtained product was labeled as PANI-b-PAA grafted HNT.
Fabrication of doxorubicin loaded nanofibers
To prepare doxorubicin loaded nanofibers, PCL was dissolved in a solvent mixture of acetic acid/formic acid with a volume ratio of 90:10%v/v and a solution with a concentration of 15%w/v was obtained. Thereafter, PANI-b-PAA grafted HNTs were dispersed in PCL solution and homogenized via a probe sonicator for a few minutes. The concentration of PANI-b-PAA grafted HNTs in PCL solution was 1%w/w. Then, doxorubicin was added to the mixture and the mixture was further homogenized by stirring for 1 h. Immediately, the dispersion was transferred to a 5 mL syringe and the syringe was placed in an electrospinning setup. Electrospinning was carried out with a flow rate of 0.3 mL/min, a voltage of 21 kV, and a spinneret-collector distance of 21 cm.
Sample characterization
Attenuated total reflectance Fourier transform infrared spectroscopy
Copolymer grafting on HNT surface was studied by ATR-FTIR (Tensor 27, Bruker, USA). The ATR-FTIR was done in the range of 3500-500 cm−1, with a resolution of 1 cm−1.
Scanning electron microscopy
Morphology of copolymer-grafted HNTs and electrospun fibers was evaluated by SEM (Sigma 300
Energy dispersive X-ray spectroscopy
Presence and distribution of copolymer-grafted HNTs and doxorubicin in electrospun fibers was confirmed by EDS (Sigma 300, Zeiss, Germany).
Thermogravimetric analysis
Amount of grafted copolymer on HNTs was measured by TGA (SDTA851, Mettler Toledo, USA). TGA was performed from room temperature up to 650°C at a heating rate of 10°C/min under nitrogen atmosphere. The curves of weight and derivative of weight versus temperature were depicted.
X-ray diffraction analysis
Crystalline structure of the electrospun fibers was studied by XRD (Equniox, Inel, France). XRD was done with CuKα radiation (λ = 0.15418 nm) and XRD patterns were recorded in 2θ range of 10° to 70°.
Drug encapsulation efficiency measurement
To calculate the drug loading content and drug encapsulation efficiency, the electrospun mat was first cut into samples with dimensions of 1 cm × 1 cm. Then, the samples were dissolved in DMF, mixed with water, centrifuged and the optical absorbance of the supernatant was measured using a spectrophotometer at a wavelength of 480 nm.37–39 The optical absorbance was then converted to drug content using calibration curve of drug. The obtained value was the actual amount of loaded drug. The actual drug loading content and encapsulation efficiency were calculated using equations (1) and (2):
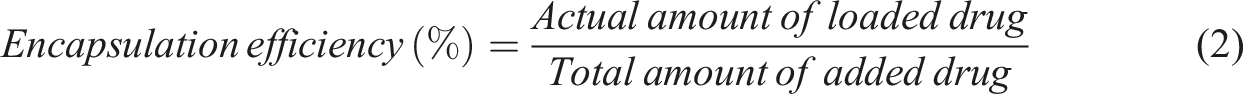
Drug release study
To study the release of doxorubicin from nanofibers, 5 mg of nanofibers was immersed in 20 mL PBS (pH = 7.4) and the samples were placed in a shaker incubator (37°C). At predesigned time intervals (1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 18, 24, 36, and 48 h), 3 mL of the release medium was collected and its optical absorbance was measured at a wavelength of 480 nm with a UV-visible spectrophotometer. Then, the tube was replenished with 3 mL of fresh PBS. The optical absorbance was converted to drug concentration by the calibration curve of doxorubicin in PBS. Cumulative drug release percentage was calculated by equation (3):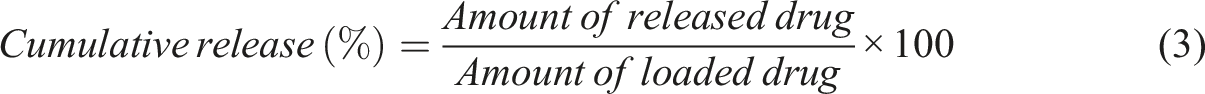
Drug release kinetics models including zero-order, first-order, Higuchi, Korsmeyer–Peppas, Weibull, and Baker–Lonsdale, the formula of the models and the definition of the model terms.

Cell culture study
The cytotoxic effect of nanofibers was examined against human breast cancer cell lines (MCF-7) and fibroblasts. For this purpose, the cells were cultured in DMEM supplemented with 10% FBS and 1% penicillin/streptomycin. After being confluent, the cells were treated with trypsin, centrifuged, counted, and suspended in cell culture media. The sterilized nanofiber samples were placed in a 48-well plate and the prepared cell suspension with a density of 10000 cells was added to each well and incubated for 4, 24, and 48 h. At each time point, the cell culture media was removed and 100 µL of MTT solution (0.5 mg/mL) was added to each well and incubated for 4 h. Then, MTT was withdrawn and 100 of DMSO was added to each well to dissolve the formazan precipitates. After 15 min, the optical absorbance of the solutions was read at 570 nm using a microplate reader. Cell viability was calculated by normalizing the optical absorbance of the sample to that of control (equation (10)).
The morphology of the cells on electrospun fibers was observed by SEM imaging. For this purpose, the sterilized nanofiber samples were placed in a 48-well plate and the cell suspension with a density of 10000 cells was added to each well and incubated for 24 h. After this period, the cell culture media was removed and the cells were washed with PBS. Then, glutaraldehyde solution was added to each well to fix the cells for 1 h. The samples were then washed with PBS and dehydrated with the gradient concentrations of ethanol. Finally, the dehydrated samples were dried in air.
Results and discussion
Fabrication and characterization of copolymer-grafted HNTs
To fabricate composite drug delivery fibers, first, surface modification of HNTs was performed and PANI-b-PAA copolymer was grafted on HNTs. The details of HNT surface modification and copolymer grafting are outlined in our previous work.
36
In brief, the HNTs initially reacted with CPTS and the chloropropyl ethoxysilane groups were formed on the surface of HNTs. Then, the resulting HNTs reacted with the pre-synthesized chain transfer agent, and the CTA was immobilized on HNT surface. Next, with the addition of acrylic acid and initiator, polyacrylic acid block was formed on the surface by RAFT polymerization mechanism. Finally, with the addition of aniline, initiator, and dopant, polyaniline block was extended along the PAA block and PANI-b-PAA copolymer was formed on the HNT surface. The grafting of copolymer on HNT was fully confirmed in our previous work, and here just a brief explanation is provided. The schematic of grafting process as well as FTIR spectrum, SEM image, TGA and DSC curves of copolymer-grafted HNTs are shown in Figure 1. Fabrication and characterization of copolymer-grafted HNTs. Schematic of PANI-b-PAA grafted HNTs (a), SEM image of PANI-b-PAA grafted HNTs (b), FTIR of PANI-b-PAA grafted HNTs (c), TGA/DTG curve of PANI-b-PAA grafted HNTs (d), and DSC curve of PANI-b-PAA grafted HNTs (e).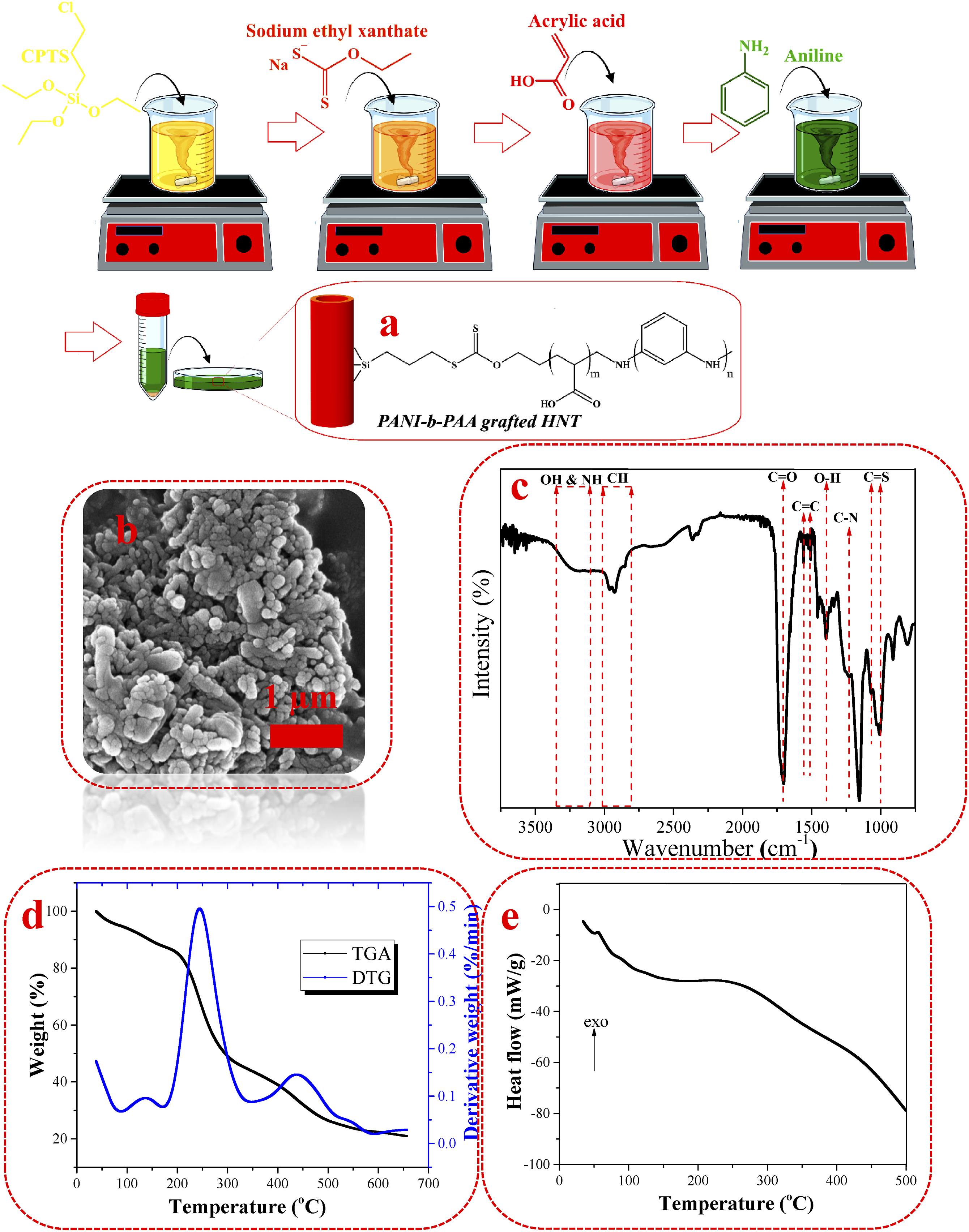
The morphology and dimension of copolymer-grafted HNTs were evaluated by SEM (Figure 1(b)). According to the SEM image of the copolymer-grafted HNTs, the HNTs preserved their cylindrical structure after surface modification. The copolymer-grafted HNTs had an average diameter and length of 40 and 106 nm, respectively. The short length of the copolymer-grafted HNTs arose from exposure to repeated sonication cycles during the surface treatment process. The short length of HNTs can promote their cell internalization and facilitate the intracellular drug release.
The formation of copolymer on HNTs was confirmed by FTIR (Figure 1(c)). According to the FTIR spectrum of copolymer-grafted HNTs, the peak in the range of 3310-3348 cm−1 was ascribed to the overlap of OH and NH stretching, and the peak in the range of 2833-3027 cm−1 was attributed to CH stretching. The peaks emerging at 1710, 1558, 1508, 1460, 1400, 1240, and 1150 cm−1 were assigned to C=O stretching, C=C stretching of quinoid group, C=C stretching of benzenoid group, C-H bending, O-H bending, C-N stretching, and C-O stretching, respectively. In addition, the peaks appearing at 1063 and 1006 cm−1 were attributed to C=S stretching. The appearance of the aforesaid peaks corroborated the formation of PANI-b-PAA on HNT surface. 41
The amount of the grafted copolymer on HNTs was calculated based on the TGA curve (Figure 1(d)). In TGA curve, the weight loss in the range of 90°C–180°C was related to the removal of dopant molecules and decomposition of ethyl xanthate group, two weight losses in the ranges of 180°C–340°C and 340°C–512°C were attributed to the decomposition of copolymer, and the weight loss in the range of 512°C–580°C was assigned to the decomposition of ethoxy silane groups. The total weight loss of the copolymer decomposition was 59% and was considered proportional to the amount of the grafted copolymer.
The thermal transitions of the grafted copolymer on the HNT surface were investigated by differential scanning calorimetry (DSC). DSC heating curve of PANI-b-PAA grafted HNTs is displayed in Figure 1(e). In DSC curve, two endothermic phenomena can be observed. The first one in the range of 40°C–56°C, corresponding to the loss of water, and the second one in the range of 56°C–89°C, corresponding to the glass transitions of PANI and PAA. In addition, a broad exothermic peak in the range of 186 to 315°C was seen, which can be attributed to the degradation and crosslinking of PANI and PAA. 42
GPC was done to provide information on the average molecular weight and polydispersity index (PDI) of PANI-b-PAA grafted onto HNT surface. For this purpose, the copolymer was cleaved from the HNT surface. GPC showed that the Mn, Mw and PDI of PANI-b-PAA grafted onto the HNT surface was 8710 g/mol, 10840 g/mol and 1.24, respectively. The low molecular weight confirmed that the chain lengths of the copolymer formed on the HNT surface were short. In addition, the low PDI corroborated that the copolymer chains were uniform in length.
SEM images of electrospun fibers
After grafting the copolymer on HNT surface, the copolymer-grafted HNTs and doxorubicin were dispersed in PCL solution and the dispersion was electrospun (Figure 2 (a)). The morphology of the electrospun fibers was observed by a scanning electron microscope. SEM micrographs and fiber diameter distribution histograms of PCL, PCL/HYB, and PCL/HYB/DOX electrospun fibers are shown in Figure 2(b). Fabrication and morphological characterization of electrospun fibers. Schematic of fabrication process of drug loaded fibers (a), SEM images and fiber diameter histograms of electrospun fibers (b).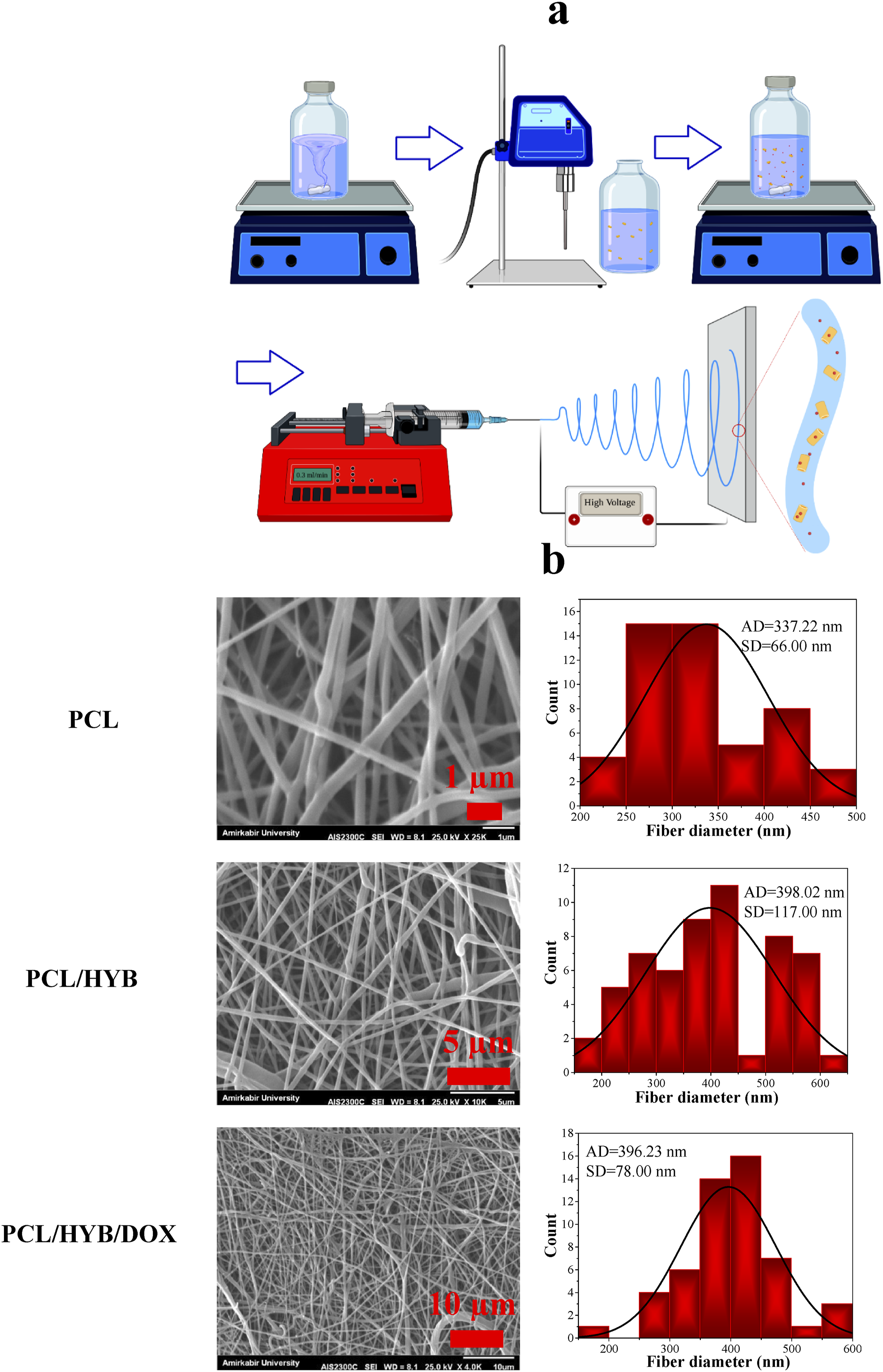
PCL is one of the most attractive polymers to produce electrospun fibers due to its solubility in various solvents and tunable properties. 43 By controlling solution and process parameters, defect-free PCL fibers with a wide range of diameters and surface porosity can be produced. As can be seen in SEM micrographs, the PCL electrospun fibers are almost defect-free and homogenous. Only in some regions, evidence of fiber merging can be observed, which can be explained by the presence of residual solvent in the fibers. Acetic acid and formic acids are solvent with high boiling points and their evaporation occur at higher temperature compared to the common solvents used for PCL electrospinning, such as chloroform. Therefore, it is probable that when the fibers are placed on the collector, some amount of solvent is still left in them, causing the fibers coalescence. The surface of PCL fibers was completely smooth, whereas when copolymer-grafted HNTs were added to the composition, some protrusions appeared on the surface and the surface smoothness was slightly reduced. The appearance of protrusions on the surface of electrospun fibers can be explained by the presence of HNTs in fibers. However, there was no evidence of HNT agglomeration in the fibers most likely due to the decoration of PANI-b-PAA chains on the HNT surface. The presence of PANI-b-PAA copolymer on the HNT surface can prevent HNTs from binding to each other and forming large agglomerates. Moreover, the presence of PANI-b-PAA copolymer on HNT surface improve the compatibility between hydrophilic HNTs and hydrophobic PCL matrix, leading to better dispersion of HNTs in polymer matrix.
The average diameter of PCL, PCL/HYB, and PCL/HYB/DOX fibers was found to be 337.22 nm, 398.02, and 396.23 nm, respectively. When copolymer-grafted HNTs were added to PCL, the average fiber diameter increased (
When doxorubicin was added to the composition, the average fiber diameter decreased, however the decrease was not statistically significant (
Moreover, according to the fiber diameter histograms, the addition of copolymer-grafted HNTs broadened the fiber diameter distribution, whereas the addition of doxorubicin narrowed the fiber diameter distribution. The later can be ascribed to the increase in electrical conductivity of electrospinning solution.
The porosity percentage and average pore diameter of the electrospun fibers was measured from SEM micrographs using image processing software. The porosity of PCL, PCL/HYB, and PCL/HYB/DOX electrospun fibers was found to be 44.66%, 45.87%, and 48.86%, respectively. The average pore size of PCL, PCL/HYB, and PCL/HYB/DOX electrospun fibers was found to be 0.77 µm, 0.90 µm and 1.03 µm, respectively. The higher porosity and the larger pore size of PCL/HYB can be attributed to the presence of amine groups on the HNT surface. The amine groups become positively charged in aqueous media, and the repulsive forces between the positively charged amine groups prevent the tight packing of the electrospun fibers. Once the drug was added to the composition, the porosity and pore size further increased because the drug molecules also carry amine groups. The higher porosity and the larger pore size can facilitate the drug diffusion from electrospun fibers.
EDS results of electrospun fibers
EDS spectra of PCL, PCL/HYB, and PCL/HYB/DOX electrospun fibers are shown in Figure 3. In the EDS spectrum of PCL fibers (Figure 3 (a)), the peaks appearing at 0.28, 0.52, and 1.49 keV are characteristic of carbon, oxygen, and aluminum, respectively. In the EDS spectra of PCL/HYB, in addition to the above mentioned peaks, new peaks appeared at 0.39, 1.75, and 2.31 keV. The peaks appearing are characteristic of nitrogen, silicon, and sulfur, respectively. The characteristic peaks of silicone and sulfur arose from ethoxysilane and ethyl xanthate, respectively, confirming the successful surface modification of HNTs. Since nitrogen is exclusively present in polyaniline, the appearance of nitrogen peak corroborates the successful grafting of copolymer on HNT surface. EDS results of electrospun fibers. EDS spectrum, magnified nitrogen peak, and EDS table (a), EDS map of carbon, nitrogen, and silicon of PCL, PCL/HYB, and PCL/HYB/DOX fibers.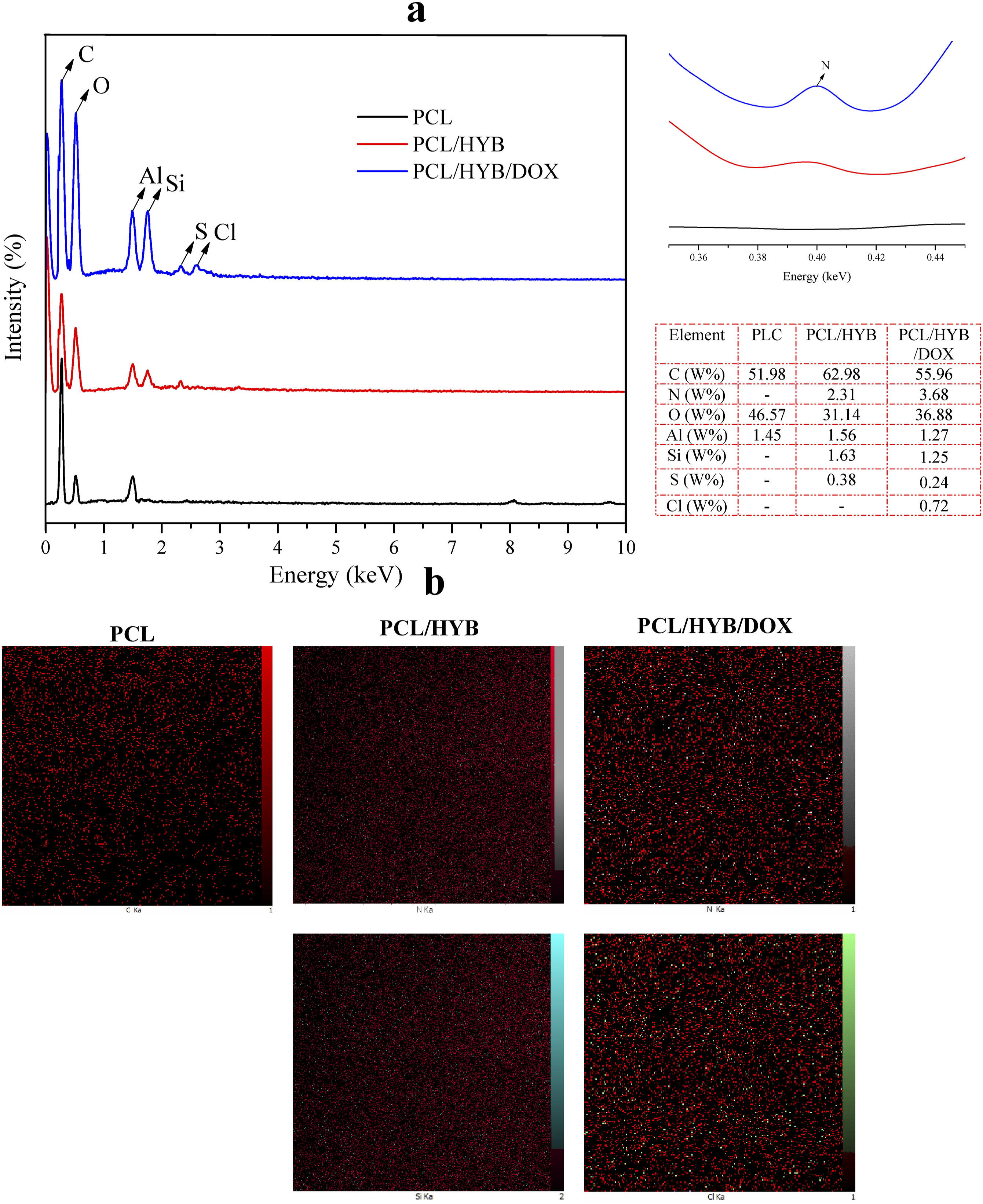
The peaks appearing in EDS spectrum of PCL/HYB/DOX fibers were similar to those of PCL/HYB fibers. However, a new peak appeared at 2.62 keV, attributing to the chlorine element. The appearance of the characteristic peak of Cl corroborates the successful loading of the drug into electrospun fibers. Moreover, according to the quantitative data of EDS, the nitrogen weight percentage of PCL/HYB/DOX was higher than that of PCL/HYB, also indicating the loading of doxorubicin in fibers. For better understanding, EDS maps of different elements are represented (Figure 3 (b)). EDS map of silicone, sulfur, and nitrogen confirmed that copolymer-grafted HNTs and doxorubicin were uniformly dispersed in PCL matrix.
FTIR results of electrospun fibers
FTIR spectra of PCL, PCL/HYB, and PCL/HYB/DOX electrospun fibers are shown in Figure 4. For pure PCL fibers, the peaks appearing at 1720, 1243, 1169 and 1050 cm−1 can be attributed to C=O stretching, C-O-C asymmetric stretching, C-O-C symmetric stretching and C-O stretching respectively.
43
In PCL/HYB and PCL/HYB/DOX fibers, similar peaks were observed as in PCL fibers due to the high contribution of PCL in the fibers. However, when the spectra of PCL/HYB and PCL/HYB/DOX fibers were magnified, some changes were detectable. C=O peak shifted from 1722 cm−1 in PCL/HYB fibers to 1720 cm−1 in PCL/HYB/DOX fibers. The shift of the carbonyl peak to lower wavelengths in PCL/HYB/DOX fibers compared to PCL/HYB fibers corroborated the formation of hydrogen bonds between copolymer-grafted HNTs and doxorubicin. These interactions can be formed between the drug molecules with the hydroxyl groups of HNT and the functional groups of the copolymer. Besides, the intensity of the C=C stretching of polyaniline at 1508 cm−1 and 1558 cm−1 was reduced, which confirmed the formation of π-π interactions between the quinoid and benzenoid groups of PANI and doxorubicin ring. Moreover, the OH stretching peak in the range of 3410-3460 cm−1 broadened and shifted to lower wavelength, confirming the formation of hydrogen bonds between the copolymer-grafted HNTs and the drug molecules. The sift of the C=O peak to lower wavelengths, the decrease in the intensity of the C=C peak, and the broadening of the OH stretching peak in the spectrum of PCL/HYB/DOX fibers corroborated the interaction between the doxorubicin molecules and the copolymer grafted on HNTs. FTIR spectra of PCL, PCL/HYB, and PCL/HYB/DOX electrospun fibers. The PCL/HYB and PCL/HYB/DOX spectra are magnified at the positions of the OH, C=O, and C = CC=C stretching peaks.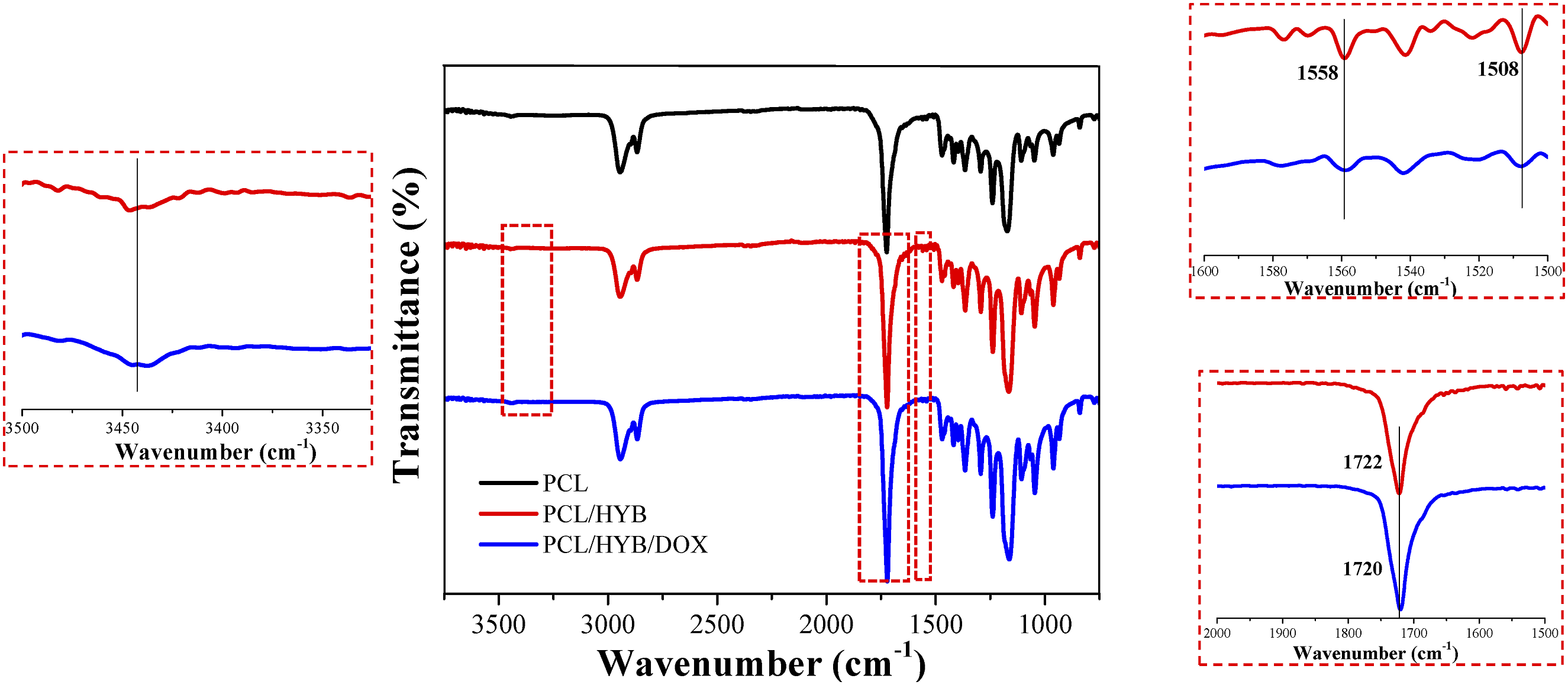
XRD results of electrospun fibers
XRD patterns of PCL, PCL/HYB, and PCL/HYB/DOX electrospun fibers are shown in Figure 5. As can be seen, pure PCL shows a broad bump in the range of 20-30°, corroborating that the PCL fibers are almost amorphous. If the PCL fibers are semi-crystalline, two crystalline peaks are observed at about 21 and 23°.
44
The amorphousness of the fibers formed in this study may be due to the application of high voltage during electrospinning. XRD spectra of PCL, PCL/HYB, and PCL/HYB/DOX electrospun fibers.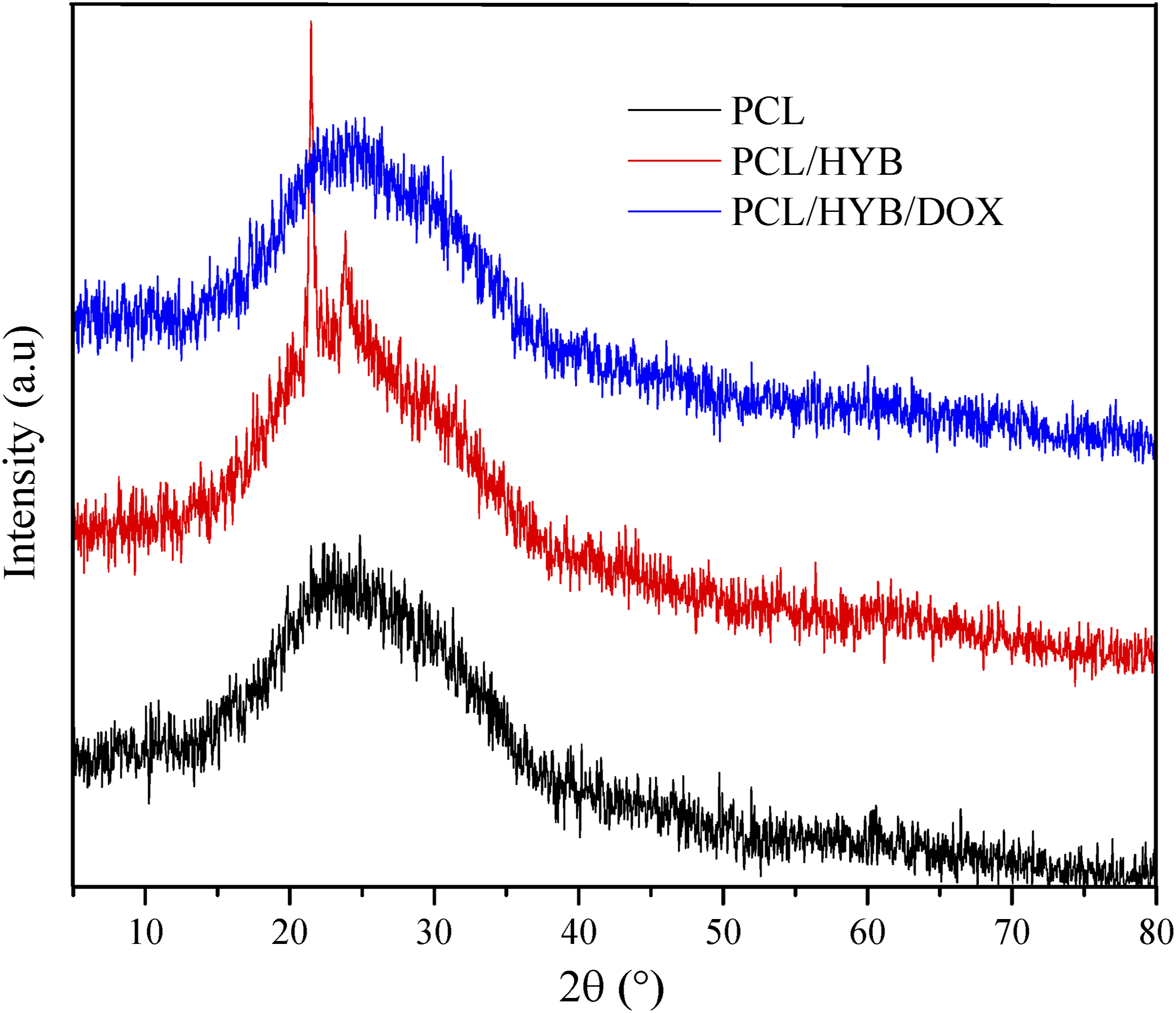
The XRD pattern of PCL/HYB fibers was almost similar to that of PCL fibers, only two sharp peaks appeared at 21.55° and 23.77°. These peaks can be attributed to copolymer-grafted HNTs. It should be noted that according to our previous work, PANI-b-PAA grafted HNTs have several crystalline peaks. When the copolymer-grafted HNTs were incorporated into the PCL matrix, few crystalline peaks appeared, confirming that nanotubes were well surrounded by polymer matrix.
When doxorubicin was added to the formulation, the crystalline peaks of HNTs disappeared most likely due to the interaction of HNTs with drug molecules. Moreover, the crystalline peaks of doxorubicin were not observed in XRD spectrum, corroborating the molecular dispersion of drug in PCL matrix. When the drug crystalline peaks do not appear in the XRD spectrum, it means that the drug lost its crystalline structure and is probably dispersed at the molecular level in the polymer matrix. This subject has also been addressed by other studies. For instance, Bulbul et al. 32 reported that in curcumin-loaded PCL/PEO nanofibers, the reduction or absence of curcumin crystalline peaks is related to the good dispersion of drug molecules in the polymer matrix. Ravikumar et al. 45 showed that when curcumin was incorporated into cellulose acetate phthalate matrix, the drug crystalline peaks were not seen in the XRD spectrum of the fibers, which was attributed to the molecular dispersion of the drug in the polymer matrix.
Drug release study
Before studying the drug release, the actual drug loading content and drug encapsulation efficiency of the PCL/DOX and PCL/HYB/DOX fibers were calculated as detailed in previously. The actual drug loading content of PCL/DOX and PCL/HYB/DOX was 3.00% and 3.85%, respectively. Also, the drug encapsulation efficiency of PCL/DOX and PCL/HYB/DOX was 75.05% and 96.23%, respectively. The higher actual loading content and the greater encapsulation efficiency of the PCL/HYB/DOX fibers can be explained by the role of NHTs and grafted copolymer in improving drug loading. The tubular structure and high surface area of HNTs provide a good space for placing drug molecules. Besides, the grafted copolymer on HNTs makes physical interaction with drug molecules and better preserves them in the drug carrier. The increase in drug encapsulation efficiency of electrospun fibers in the presence of halloysite nanotubes has been confirmed by other researchers. Bulbul et al. 32 reported that when HNTs were added to PCL/PEO matrix, the encapsulation efficiency of curcumin increased significantly, and the surface modification of HNTs further improved the encapsulation efficiency.
Doxorubicin standard curve and cumulative drug release curves of PCL/DOX and PCL/HYB/DOX are shown in Figure 6. a and b. As can be seen in the Figure, both samples had a burst release in the first 3 hours, however the amount of their burst release was different. The burst release of PCL/HYB/DOX was lower than that of PCL/DOX ( Doxorubicin calibration curve (a) and doxorubicin release curve (b). * indicates 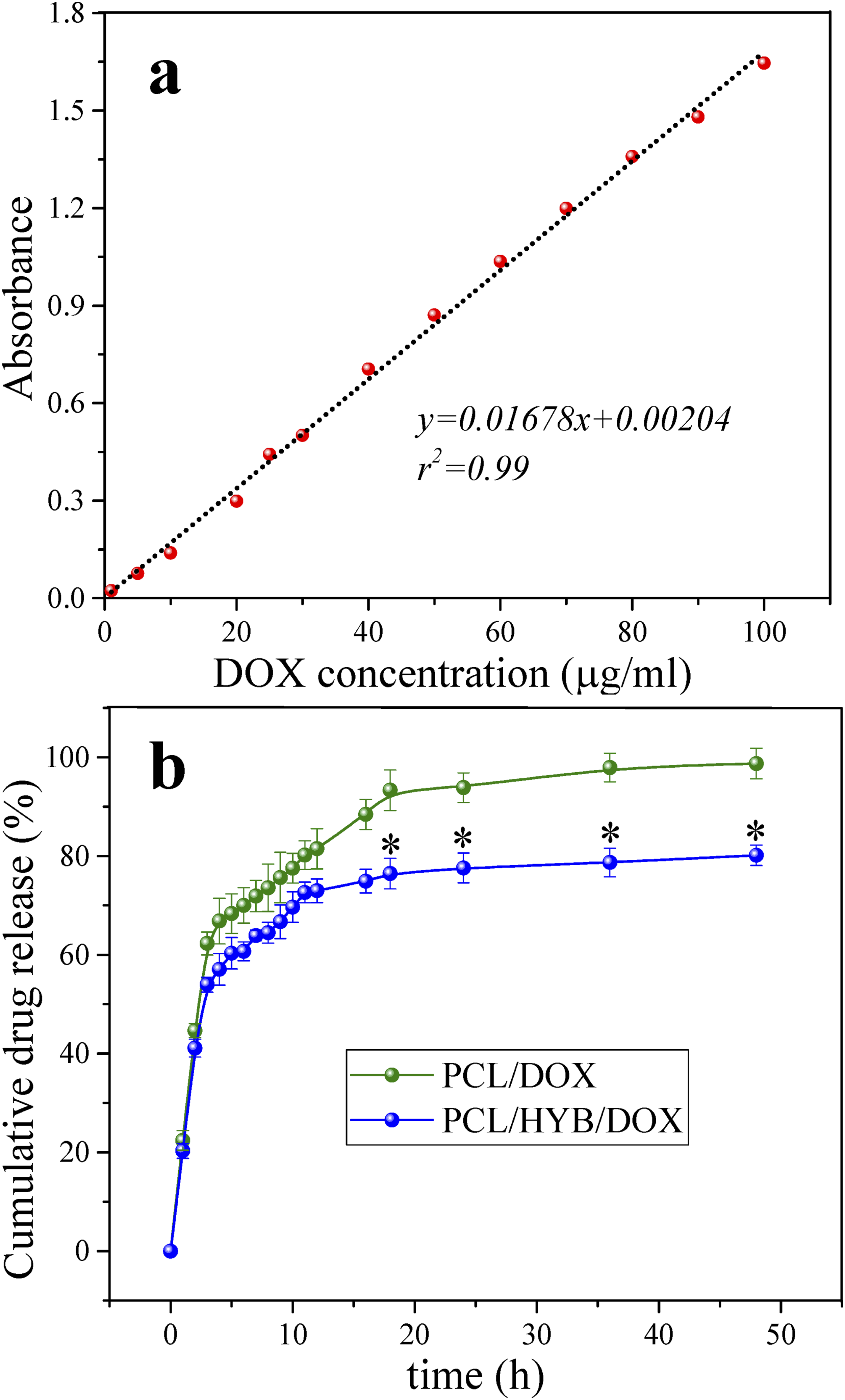
After the initial burst release, the drug release became gradual and sustained. Similar to the initial release, in this release stage, the amount of released drug was lower in PCL/HYB/DOX fibers compared to PCL/DOX fibers. This trend can be elucidated by the more impeded drug release and further drug-carrier interaction in PCL/HYB/DOX fibers. In addition to the effect of HNTs themselves, the copolymer grafted on HNTs can also affect the drug release behavior. The presence of PANI-b-PAA copolymer onto HNTs can provide a further barrier to drug diffusion and make the drug release more gradual. At the same time, PAA portion of the copolymer can increase the hydrophilicity and water uptake, leading to easier drug diffusion; however, this effect is limited to the HNT surface and does not affect the overall hydrophilicity of the fibers. Electrostatic interactions of drug molecules with the amine groups of polyaniline and the carboxyl groups of polyacrylic acid can also reduce the amount of drug released and slow down the drug release process. Of course, it should be noted that the molecular weight of the copolymer chains formed at the HNT surface is low and as a result the chains are short. Increasing the molecular weight of the formed chains can enhance their effect in improving the dispersion of nanotubes and prolonging the drug release.
After 16 h, the difference in drug release between the two groups became statistically significant. The amount of drug released from PCL/HYB/DOX fibers at 18 h, 24 h, 36 h, and 48 h was significantly lower than that of PCL/DOX fibers (
The amount of released drug after 48 h in PCL/DOX and PCL/HYB/DOX fibers reached 98.30% and 80.22%, respectively. Indeed, after 48 h, in PCL/HYB/DOX fibers, total amount of drug was released. However, in PCL/HYB/DOX fibers, a fraction of drug still remained in the carrier. This fraction can be attributed to dug molecules which are entrapped in HNTs.
Wang et al. 11 loaded doxorubicin into electrospun fibers based on PLGA and attapulgite, a clay mineral. Drug release study showed that the initial drug release from fibers was about 5% and reached about 10% after 400 h. The lower drug release from these fibers compared to PCL/HYB/DOX fibers can be attributed to the larger fiber diameter as well as the higher crystallinity of the fibers. The larger fiber diameter and higher crystallinity impede the diffusion of drug molecules and prolong their release. Dai et al. 46 incorporated doxorubicin into PLA/pearl powder fibers. In fibers with 3% pearl powder, the amount of drug released in the first 3 h was 40% and the drug release was completed within 24 hours. The faster drug release of these fibers compared to PCL/HYB/DOX fibers can be explained by the high hydrophilicity of the pearl powder. The high hydrophilicity of the carrier can facilitate water absorption and increase the plasticity of the polymer matrix, leading to enhanced drug release. Wang et al. 47 loaded doxorubicin into core-shell fibers. The core was composed of drug molecules and the shell was composed of a blend of poly (lactide-co-caprolactone) and gelatin. The drug release was about 20% in the first hour and reached about 40% after 400 h. The slower drug release from these fibers compared to PCL/HYB/DOX fibers was due to their core-shell structure, in which the drug molecules were entrapped in a thick layer of polymer.
Presenting the results of the drug release kinetic model, including the coefficient of determination, release constant (K), and release power (
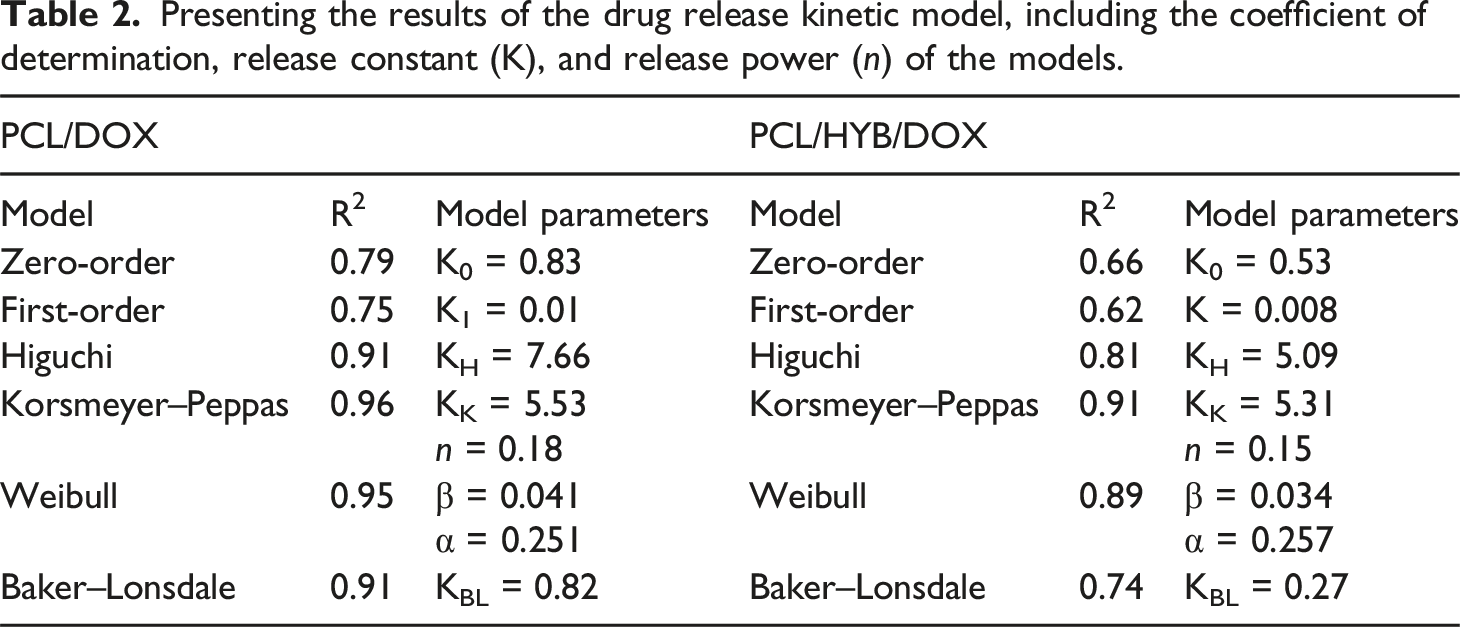
According to the values of the coefficient of determination, Korsmeyer–Peppas was the best model to fit the experimental data. Because the drug delivery system designed here is heterogeneous and multicomponent, the Korsmeyer–Peppas model can predict the release behavior better than other models. Korsmeyer–Peppas is a semi-empirical models, in which in addition to diffusion, other mechanisms like swelling and relaxation of polymer chains are considered. Therefore, it can better describe the release of doxorubicin from multicomponent and complex carriers composed of PCL fibers and copolymer-grafted HNTs.
The Korsmeyer–Peppas exponent for PCL/DOX and PCL/HYB/DOX fibers was 0.18 and 0.15, respectively. Korsmeyer–Peppas exponent lower than 0.45 offers that the drug release is mainly controlled by Fickian diffusion. Moreover, according to the results, the release constant decreases with the addition of HNTs to the polymer matrix, corroborating the slower and more prolonged drug release from PCL/HYB/DOX fibers.
Based on the coefficient of determination values, the Weibull model was the second best model for fitting the release data after the Korsmeyer-Peppas model. Because, the designed drug delivery carrier is complex, the Weibull model, which is an empirical equation, can fit the drug release data well. In this model, when β < 1, the drug release is largely mediated by Fickian diffusion. Here, β was less than one for both PCL/DOX and PCL/HYB/DOX fibers, indicating that the drug release is diffusion-controlled.
The suppression of burst release and the reduction of release rate by incorporating HNTs into electrospun fibers have been reported by other researches. Joshi et al., 34 reported that the release of chlorhexidine acetate from PU/halloysite fibers was slower than that from PU fibers. The total amount of drug in PU fibers was released after 48 h, whereas less than half of drug in PU/halloysite fibers was released during this period.
However, in a research performed by Carvalho and Coimbra, 33 an opposite trend was observed. In this research, gentamicin sulfate and dexamethasone loaded in PLA/halloysite fibers were released faster than those loaded in PLA fibers. To justify this trend, it was stated that although PLA/halloysite fibers created more barriers against drug diffusion, the diameter of PLA/halloysite fibers was smaller than PLA fibers, leading to greater surface area and faster drug release.
Cell culture results
MTT assay was performed for normal and cancer cells. L929 (Figure 7) and MCF7 (Figure 8) were selected as normal and cancer cell lines, respectively, and MTT assay was done after 4 h, 24 h, and 48 h of cell seeding. Cell viability was calculated by normalizing the optical absorbance of the sample to that of control. According to the obtained results, the cell viability of PCL and PCL/HYB nanofibers was above 90% for both cell types, confirming the nontoxic and biocompatible nature of these fibers. It is worth mentioning that the cell viability percentages of PCL/HYB nanofibers were higher than those of PCL nanofibers. Concerning fibroblast, at t = 24 h, the cell viability of PCL/HYB fibers was significantly higher than that of PCL fibers ( Cell culture results with L929. MTT results and SEM images of cells. Cell culture results with MCF7. MTT results and SEM images of cells.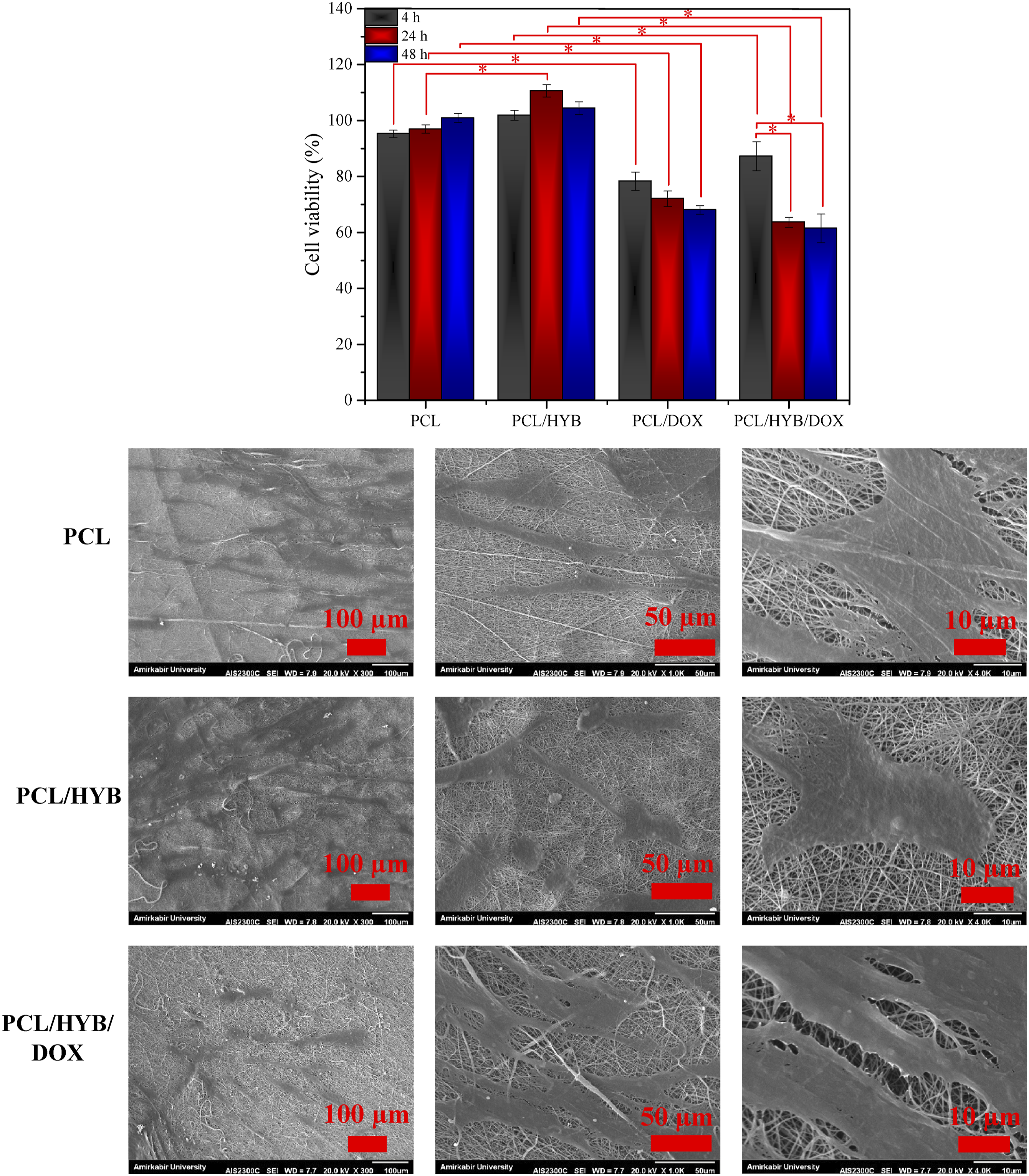
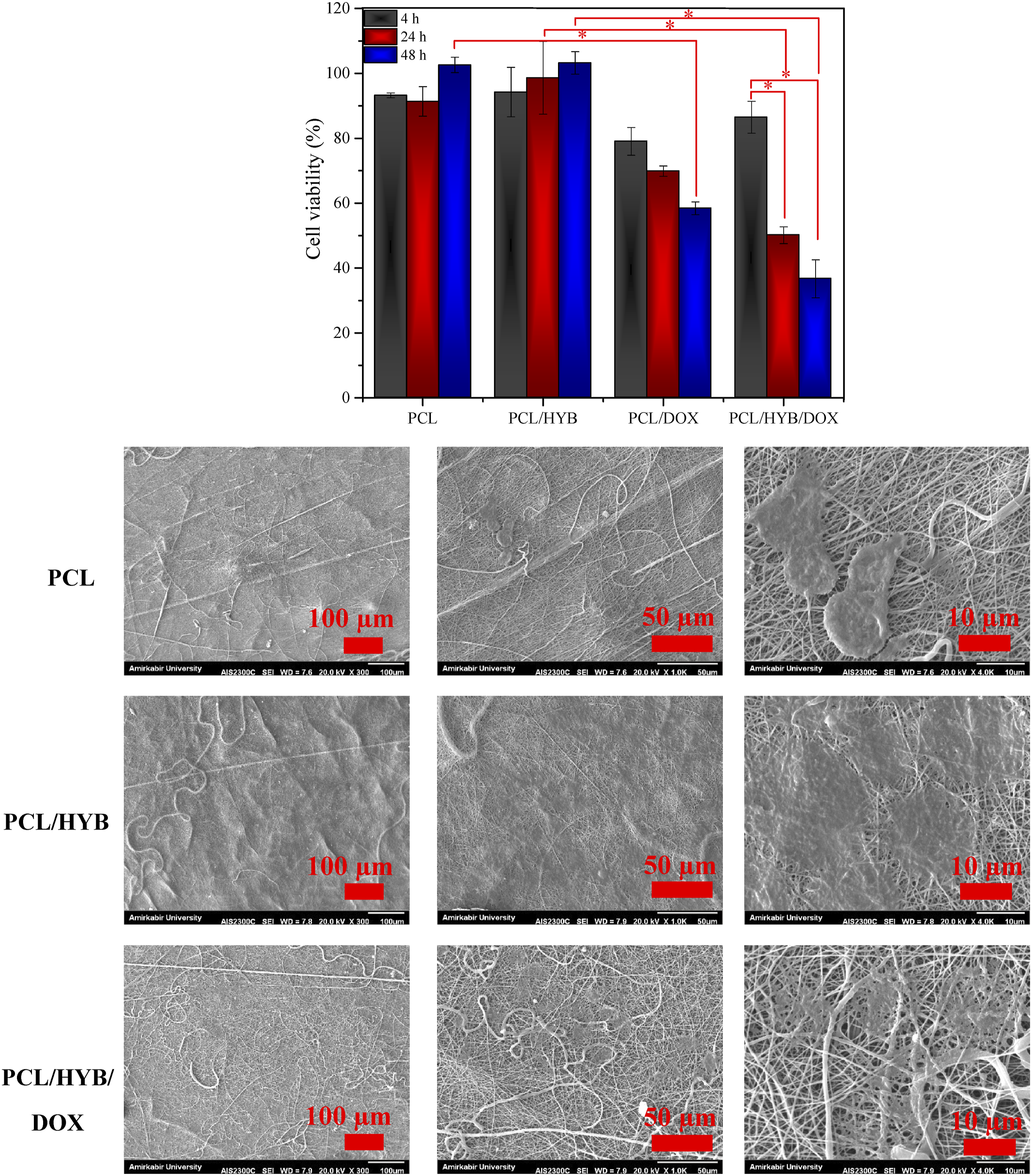
Unlike PCL and PCL/HYB fibers, the cell viability of PCL/DOX and PCL/HYB/DOX fibers was less than 90%, indicating the toxic effects of theses samples on cells. Concerning fibroblast, at t = 4 h, 24 h and 48 h, the cell viability of PCL/DOX fibers was significantly lower than that of PCL fibers (
Regarding MCF7, at t = 48 h, the cell viability of PCL/DOX fibers was considerably lower than that of PCL fibers (
Doxorubicin is an anthracycline chemotherapy drug that induces its anticancer effect through two mechanisms. In the first mechanism, doxorubicin intercalates with DNA molecules and block the function of topoisomerase-2, leading to disruption of DNA replication. In the second mechanism, doxorubicin interacts with intracellular oxygen and produces reactive oxygen species, thereby causing damage to cellular components.50,51
Over time, cell viability of PCL/DOX and PCL/HYB/DOX fibers decreased. In PCL/DOX fibers, the decrease in cell viability from 4 to 48 h, was not statistically significant (
According to the obtained results, it can be said that in the presence of PCL/HYB/DOX fibers, only after 24 h, half of the cancer cell population was eradicated. The remarkable cytotoxic effect of PCL/HYB/DOX fibers can be explained by the positive effects of HNTs on the loading and release of doxorubicin. The hollow structure and the high surface area of HNTs, as well as the presence of copolymer on HNT surface, allow loading a large amount of drug molecules into the electrospun fibers.
HNT has a tubular structure and nano-sized dimensions, which facilitate its penetration into plasma membrane and the release of its therapeutic payload in the cytoplasm. The polyacrylic block on HNTs improves the dispersion of HNTs in cell culture media and prevents their sedimentation, and the polyaniline block of grafted copolymer on HNTs enhances their penetration into cells. Moreover, when the chemotherapy drug is incorporated into HNTs, the efflux pumps cannot remove the drug from the cells and the drug accumulation in cells increases. 52
According to the MTT results of PCL/HYB/DOX fibers, the cell viability decreased from 4 h to 24 and from 24 h to 48 h, as more drug was released and a higher cytotoxic effect occurred over time. The initial cytotoxic effect of nanofibers comes from the doxorubicin molecules that are not trapped in HNTs. These molecules quickly diffuse from the fibers, enter the cells, and induce their cytotoxic effects. For subsequent effects, two scenario are possible. The first possible scenario is that the drug-loaded HNTs can diffuse out of the fibers, enter the cells, and release the drug into the cells. The second case is that drug molecules trapped in HNTs can diffuse from HNTs into the space of the fibers, penetrate outside the PCL fibers, release into the cell culture media, and finally penetrate into the cells.
Since the MTT assay was performed for both normal and cancer cells, the toxic effect of the carriers on normal and cancer cells could be compared. According to the MTT results, after 48 h, the PCL/DOX carrier caused a 33% reduction in the normal cell population and a 41% reduction in the tumor cell population. PCL/HYB/DOX carrier, after 24 h, led to a 37% and 50% reduction in the normal and cancer cell populations, respectively. After 48 h, it caused a 41% and 64% reduction in the normal and cancer cell population, respectively.
Therefore, it can be said that the toxicity of PCL/HYB/DOX fibers against cancer cells was greater than that against normal cells. A less cytotoxic effect against normal cells compared to tumor cell is beneficial for a drug delivery carrier for cancer treatment, as it ensures that normal tissues are not severely damaged. 51 The prepared carriers, especially PCL/HYB/DOX, can perform their anticancer function while minimizing adverse effects on non-cancerous tissues.
SEM images of MCF7 and L929 cultured on electrospun fibers for 24 h are shown in Figures 7 and 8. According to the SEM images of MCF7, the cells adhered and spread on both PCL and PCL/HYB nanofibers, however to different extents. The cell adhesion and spreading were higher on PCL/HYB compared to PCL nanofibers. Besides, the number of cells on PCL/HYB fibers was higher than on PCL fibers. As can be perceived from the high resolution images, cells made a better interaction with PCL/HYB fibers, firmly adhered to the fibers, developed their lamellipodia and filopodia, and even communicated with neighboring cells. The cell number was dramatically reduced on PCL/HYB/DOX fibers compared to PCL/HYB fibers due to the cytotoxic effect of doxorubicin. From the high resolution image of PCL/HYB/DOX fibers, it can be observed that the cells have lost their integrity and become fragmented. The reduction of cell number as well as the fragmentation of cell body corroborates that PCL/HYB/DOX fibers are capable of eradicating cancer cells.
According to the SEM images of L929, the cell proliferation was moderately higher on PCL/HYB fibers compared to PCL fibers. As previously mentioned, PCL/HYB fibers provide a much better milieu for cell residence and activity, leading to improved cell proliferation. The number of cells was significantly reduced on drug loaded fibers compared to their drug free counterpart. Besides, the cell-cell connections were damaged in drug loaded fibers.
To better clarify the effect of nanofibers on cell proliferation, the percentage of surface covered by cells was measured with image processing software. The surface area covered by the MCF7 was found to be 46.49%, 59.25%, and 50.44% for PCL, PCL/HYB, and PCL/HYB/DOX fibers, respectively. The surface area covered by the L929 was found to be 56.22%, 61.14%, and 54.30% for PCL, PCL/HYB, and PCL/HYB/DOX fibers, respectively.
Conclusion
Due to the relative ineffectiveness of conventional chemotherapy and its adverse effects on non-cancerous tissues, much attention has been devoted to nanocarriers for the delivery of chemotherapy drugs. Halloysite nanotubes by the virtue of high surface area and tubular structure are one of the most popular carries for cancer drug delivery. Surface modification of NHTs can increase their blood circulation, prevent their phagocytosis, and improve their endocytosis. Considering this, in the present study, HNT has been modified with a block copolymer of polyacrylic acid and polyaniline. The acrylic block of copolymer improves the dispersion of HNTs in biological fluids, and the aniline block of copolymer facilitates its penetration into cancer cells. Besides, to further control drug release, copolymer-grafted HNTs were included in PCL nanofibers. The nanofibers not only prolong the drug release but also act as scaffold for the regeneration of damaged tissue. Preliminary analyses confirmed that the copolymer was successfully grafted on the HNTs, and the copolymer-grafted HNTs had an average diameter and length of 40 and 106 nm, respectively. SEM images showed that the drug-containing composite fibers had an average fiber and pore diameter of 396 nm and 1 µm, respectively. EDS results confirmed that the copolymer-grafted HNTs and drug molecules were uniformly dispersed in PCL fibers. Drug encapsulation efficiency measurement revealed doxorubicin encapsulation efficiency of 75 and 96% for PCL and composite nanofibers, respectively. Drug release study showed burst release of 63 and 53% for PCL and composite nanofibers, respectively. Fitting the drug release data with different kinetics models confirmed the best fit with the Korsmeyer–Peppas model, and the rate constant of this model corroborated the diffusion-controlled nature of drug release. Cell culture tests with two groups of cells, L929 and MCF7, showed a higher cytotoxic effect of composite nanofibers and attributed this effect to the higher drug loading, more gradual drug release, and positive role of HNTs in cellular internalization of drug molecules.
Footnotes
Acknowledgements
Not applicable
Author contributions
Shahabaldin Jafarzadeh is the main researcher of the work and writer of the manuscript. Vahid Haddadi-Asl is supervisor of the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
Data will be available on reasonable request.
