Abstract
In this research, the thermal behavior and stability as well as the rheological properties of the nanocomposites containing 5 wt% multi-walled carbon nanotubes (MWCNTs) filled poly (ethylene-co-vinyl acetate) (EVA)/poly (methyl methacrylate) (PMMA) blends of different blend compositions were investigated. It was confirmed by employing the Winter-Chambon model that the physical gels were formed for the nanocomposites containing 70 wt% EVA and beyond. Hence, they behaved as shear thinning fluids throughout the angular frequency range examined. Moreover, the relaxation exponent, gel strength and fractal dimension of the critical gel were estimated to be about 0.309, 41,304 Pa.sn and 2.21, respectively. The calculation of the wetting coefficient revealed that the MWCNTs thermodynamically tended to migrate from EVA and locate inside the PMMA phase. The results obtained from the crystallization and melting rate of the materials indicated that the crystallization rate was always higher than that of the melting rate and both progressively decreased with the increase in PMMA loading for the nanocomposites. Thermogravimetric analyzer (TGA) was employed to study the thermal stability of the nanocomposites under air and nitrogen atmosphere. The highest thermal stability for air-scanned materials were found for the nanocomposites containing 85 wt% followed by 70 wt% EVA as if their thermal stabilities were even superior to that of the pristine EVA. For instance, the temperature at 50 and 70 wt% weight loss for the nanocomposite containing 85 wt% EVA was found to be 20 and 23°C higher than those of the EVA alone, respectively.
Introduction
The investigation and attempting to develop the innovative composite materials with desirable and tailor-made properties is still an exciting and vibrant topic in polymer science and technology research area. 1 In recent decades, the nanoparticles such as single- (SWCNTs) and multi-walled carbon nanotubes (MWCNTs), graphene (GN), organo-modified montmorillonite (OMMT) and polyhedral oligomeric silsesquioxane (POSS) has become a part of plastics formulations in academia and industries. The polymers incorporated with these materials could provide much better mechanical, optical, electrical, rheological, biomedical, thermal and barrier properties than those of their unfilled counterparts.2–7 Thermoplastic materials are preferred to the thermoset ones mainly because of their capabilities for high production, less hazardous chemicals, easy and swift processing, recyclability and higher environmental safety. 8 Carbon-based nanoparticles have found their place in polymer nanocomposite productions because of their excellent physico-mechanical properties. 9 The finished articles made of these nanocomposites are used in applications such as biomedical, 10 energy storage, 11 packaging, 12 tissue engineering, 13 and sensors. 14
Poly (methyl methacrylate) (PMMA) is an atactic, amorphous thermoplastic material with a variety of applications in packaging, optical and biomedical industries. This is due to its desirable optical transparency and gloss, good dimensional stability, low water absorption, excellent weathering resistance and biocompatibility as well as elevated mechanical strength. However, the material posses relatively poor thermal stability, flame resistance and electrical conductivity.15,16 Poly (ethylene-co-vinyl acetate) (EVA) with totally saturated backbone presents an outstanding ozone, weather, and heat resistance as well as great flexibility and resilience, superb processing and biological inertness property. It may also be a good candidate for substituting PVC as a halogen-free polymer in many applications.17,18 Thus, EVA could be considered as a good nominee for the modification of the PMMA shortcomings. 19
It is worth mentioning that only limited number of published works is available about the PMMA/EVA blends and their nanocomposites in literature. Bernini et al. 20 worked on the opto-thermal behavior of PMMA/EVA blend. They reported that the blend underwent a phase transition where its optical properties varied considerably. The authors suggested that the especial optical and thermal behavior of the blend made it attractive for potential application in temperature-controlled optical tools. Tewo et al. 21 studied several compositions of paraffin wax (wax)/EVA blend filled with PMMA micro-beads for possible application as investment casting pattern substance. They proposed that 40/20/40 wt% wax/EVA/PMMA blend revealed an optimum balance of properties in which the flexural strength, strain at break and needle penetration hardness were highly improved, at the cost of the augmented melt viscosity, in comparison with those of the paraffin wax itself. Inoue 22 reported that the impact strength of the PMMA/EVA blend fabricated by means of cast-polymerization is much superior to that of the melt-mixed PMMA/EVA blend. The triangle phase diagram drawn from the polymerization of 80/20/0.2 methyl methacrylate (MMA)/EVA/2,2′-azobis (isobutyronitrile) showed that the system forced into a two-phase arrangement when the polymerization continued to the level of about 2 wt% conversion at 80°C. Chen et al.23,24 studied the phase separation phenomenon during the polymerization of MMA in the presence of EVA copolymer using optical microscopy and light scattering techniques. From the cloud point curves, they found out that the ternary MMA/PMMA/EVA blend created an upper critical solution temperature (UCST), which amplified with the reduction in MMA content. The trigonal phase diagram, which was drawn based on the cloud point curves, showed that the homogeneous zone in the ternary blend was very tiny. Cheng and Chen 25 found out that the PMMA/EVA in situ blends improved the toughness of PMMA and following the addition of EVA, the fracture mechanism of the matrix was transformed from crazing to yielding. Since the EVA domains were placed near to one another, the interconnection yielding stress volumes resulted in the lengthening of elongation and toughening of PMMA. It was also reported that an abundant number of fibrils and voids were created on the fracture surfaces of the PMMA/EVA in situ blends. Therefore, the deformations that were occurred at the boundary of PMMA phase and EVA dispersed domains, avoided the particles to pull out from the PMMA at fracture and hence absorbed high amount of energy. Ying et al. 26 studied the influence of selective localization of silica (SiO2) on the mechanical properties of PMMA/EVA/SiO2 composites. They pre-dispersed the filler into either EVA or PMMA material and the other polymer was then blended. It was reported that higher SiO2 content and more spherical-shaped EVA domains brought about the optimum mechanical properties for the composite prepared by PMMA/SiO2 pre-dispersed compound. Si et al. 27 evaluated the influence of organoclay on the microstructure of polycarbonate (PC)/poly (styrene-co-acrylonitrile) (SAN), polystyrene (PS)/PMMA and PMMA/EVA blends. They reported that the domain size was appreciably decreased for the blends. The compatibilization effect was suggested to achieve via the interfacial localization of organoclay particles.
In our previous work, 28 we investigated the effect of MWCNTs content (i.e. 0, 2.5, 5 and 10 wt%) on the physical and mechanical properties of PMMA/EVA based nanocomposites containing 30 wt% EVA. The results revealed that the maximum tensile properties were obtained for the material containing 5 wt% MWCNTs. The main objective of this research was to scrutinize the influence of PMMA/EVA weight ratio in the nanocomposites having 5 wt% MWCNTs on the possibility of critical gel formation, the rheological behavior and thermal stability of the materials. Hence, the structure-property relationship was established for this far less studied system in order to tailor the properties of PMMA/EVA/MWCNTs-based nanocomposites by adjusting their compositions. It is worth mentioning that any similar report has yet to be found in the literature, to the best of the authors’ knowledge, on this subject.
Materials and methods
Materials
EVA with the trade name of EVA VS430 was procured from Hyundai Séetec (South Korea). The density, melt flow index (190°C, 2.160 kg) and vinyl acetate content of the copolymer was reported as 0.94 g/cm3, 1.7 g/10 min and 19%, respectively. PMMA with the commercial name of Acryrex CM-205 was purchased from Chei Mei Corporation (Taiwan) with the melt flow index (230°C/3.8 kg) and density of 1.8 g/10 min and 1.19 g/cm3, respectively. Its weight average molecular weight (Mw) and polydispersity index were also reported to be 79,400 g/mol and 1.73, respectively. 29 MWCNTs with the trade name of NanocylTM NC 7000 was supplied from Nanocyl S.A (Belgium). It’s mean diameter and length as well as purity were reported to be of 9.5 nm, 1.5 μm and about 90%, respectively. 30
Fabrication of the nanocomposites
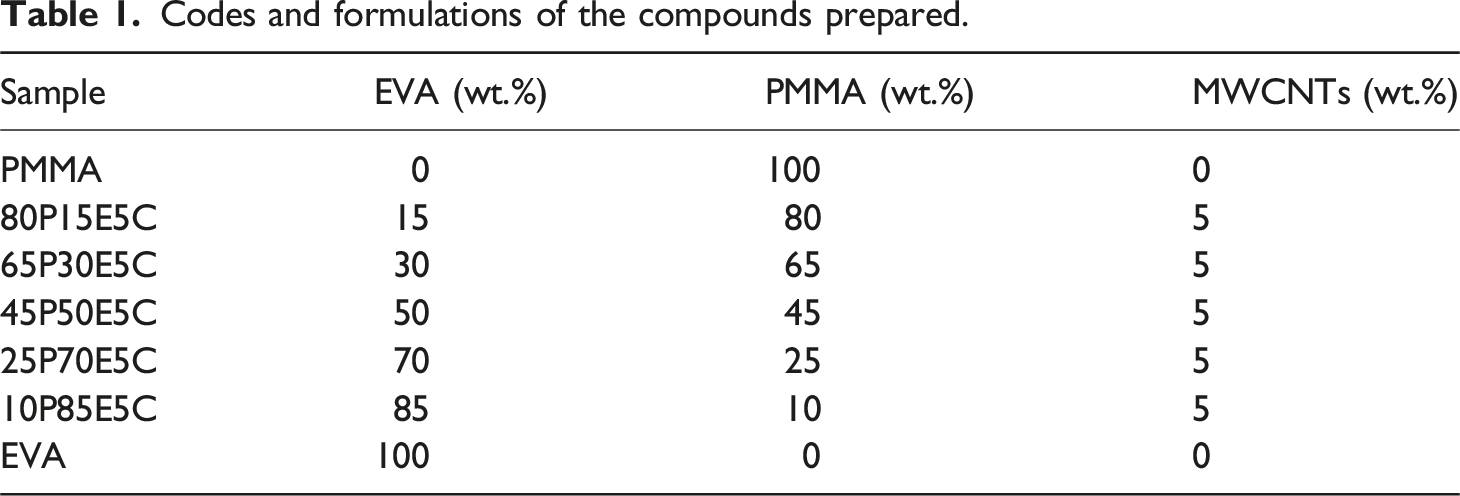
Codes and formulations of the compounds prepared.
Characterization
The static contact angle measurements were carried out on the neat surface of compression molded sheets of PMMA and EVA. The data acquisition was accomplished at about 20°C, using the probe liquids of double distilled water (H2O) and diiodomethane (CH2I2) by means of a contact angle measuring device Krűss G10 (Germany). In each case, the measurements were carried out on minimum seven individual droplets of 6 μL volume located at different places and the average value was then calculated. DSC1 Mettler Toledo (Switzerland) was used at a cooling/heating rate of 10°C/min under a nitrogen atmosphere to evaluate the crystallization and melting behavior of the materials prepared. Each sample with the weight of 10 ± 0.2 mg was encapsulated in an aluminium closed pan and heated from −50 to 160°C at the rate mentioned above. The samples were then remained at 160°C for 5 min, then cooled to −50°C and after 5 min heated again to 160°C. The degree of crystallinity (
Results and discussion
Localization of MWCNTs
In order to realize how a material such as a (nano)filler is preferentially distributed in an immiscible blend, the particle preferred location can theoretically be predicted by calculating the wetting coefficient (ωa) value.
34
The ωa value is obtained using equation (2). This relationship, which has been suggested by Sumita,
35
only considers the thermodynamic aspects but not the kinetic features of the process.
36
For our system, the equation has been adopted as37,38;
Several methods are now available for obtaining the interfacial surface free energy of the materials (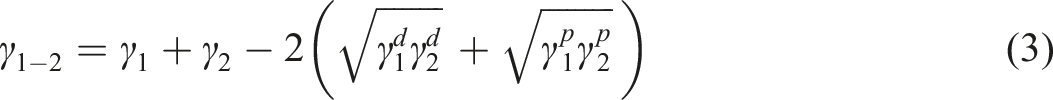
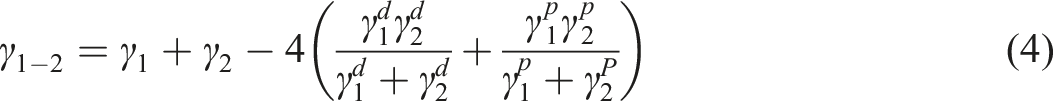
The surface free energy of the two polymers and their dispersive and polar values of the surface free energy can be found from the contact angle experiments using the following equations45,46;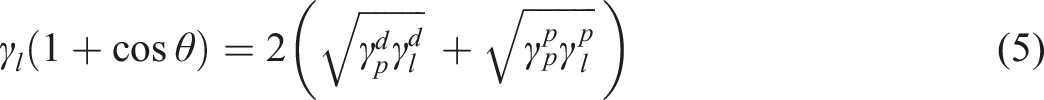


Surface free energies of the probe liquids and their contact angles on the EVA and PMMA surfaces at 20°C.

Surface free energies of the two pristine polymers and the nanofiller.
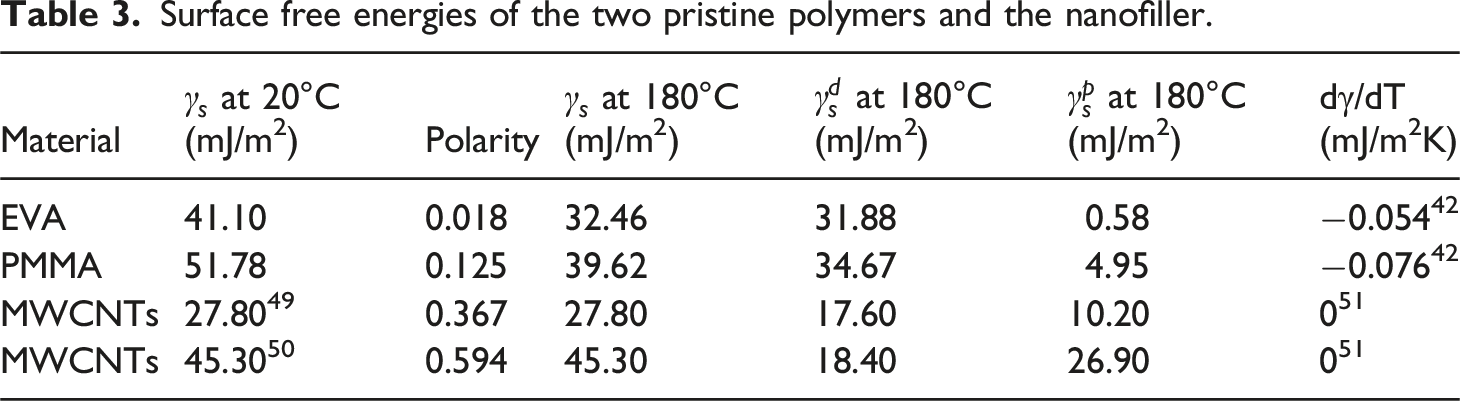
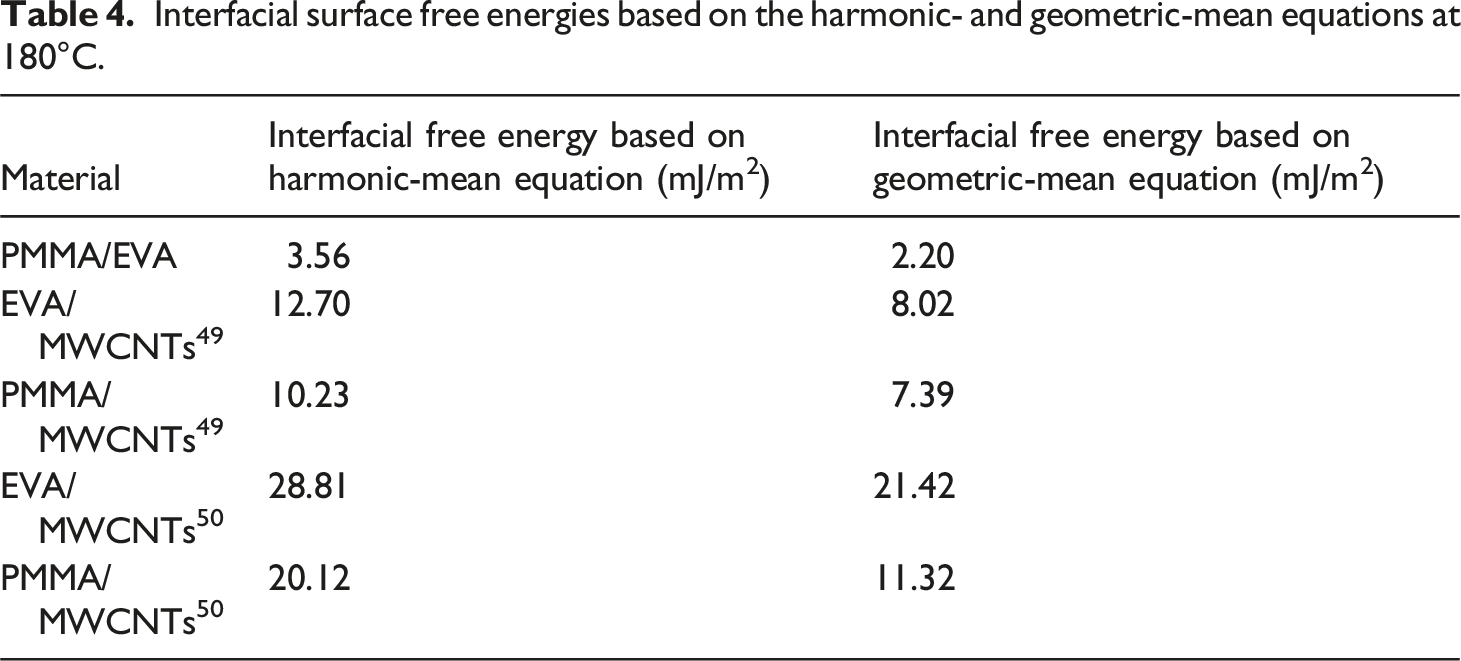
Interfacial surface free energies based on the harmonic- and geometric-mean equations at 180°C.
The wetting coefficients attained by harmonic- and geometric-mean equations at 180°C.

X-ray diffraction analysis
The XRD spectra of the parent polymers and prepared materials are shown in Figure 1. On the one hand, double important reflections were unveiled by the semi-crystalline copolymer, which could be observed at about 2θ of 21.3 and 23.4°C.
59
The former is assigned to the plane (110) and the latter to that of (200), which are both related to the polyethylene orthorhombic unit cell.
60
On the other hand, PMMA revealed no distinct reflections but only a broad halo diffraction, reflecting that PMMA was an amorphous polymeric material.
61
The results showed that the reflection peaks become visible at almost the same locations, similar to those of the unfilled EVA, for the nanocomposites prepared. The appearance of the reflections at similar places and the lack of the formation of new peaks revealed that MWCNTs was unable to modify the crystalline structure of the semi-crystalline component of the nanocomposites. In addition, no visible reflections could be seen at the 2θ of around 26 (stronger peak) and 44° (weaker peak), which were assigned to the graphite (002) and (100) planes of the well-ordered concentric tubes of the nanofiller.
62
This might be attributed to the good dispersion of the nanofiller particles throughout the polymers. XRD diffractions of the parent polymers and the nanocomposites.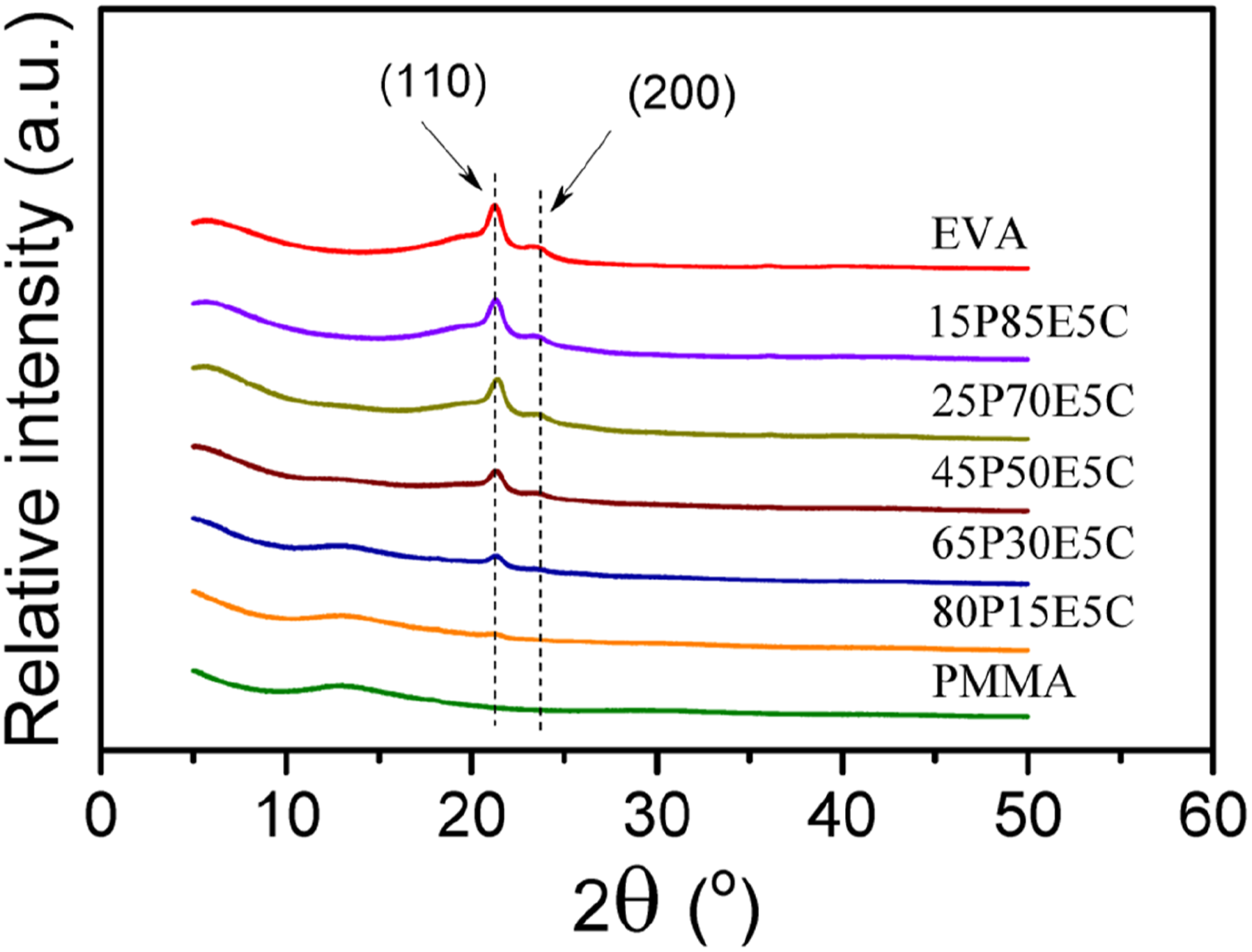
Crystallization and melting behavior
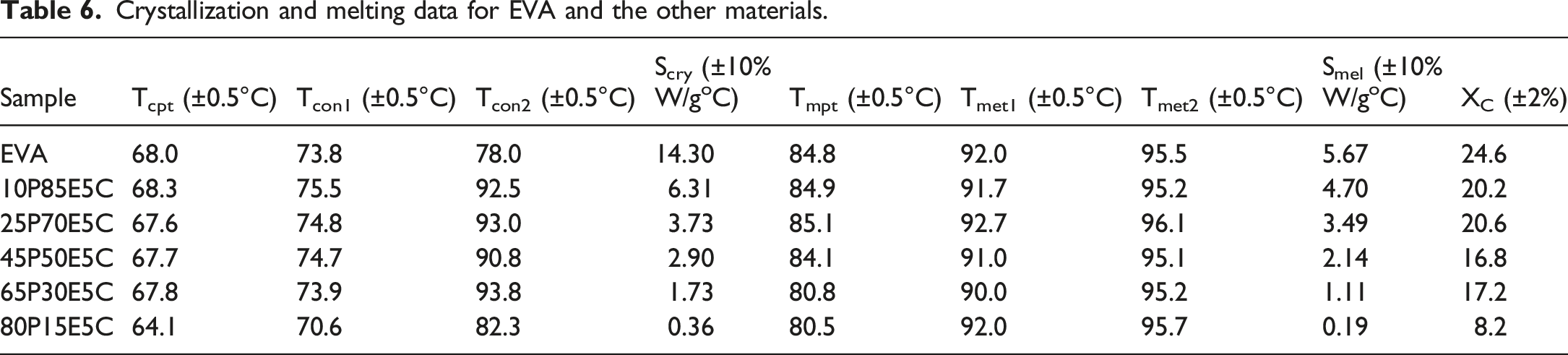
The crystallization and melting response for the crystallizable materials, obtained by DSC, are shown in Figure 2(a) and (b), respectively. All the thermograms were scrutinized to determine the crystallization onset temperature (Tcon), crystallization peak temperature (Tcpt), melting peak temperature (Tmpt), melting end temperature (Tmet) and XC as well as the rate of crystallization (Scry) and rate of melting (Smel) of the materials. The data obtained are tabulated in Table 6. Both Tcon and Tmet were found using two different methods. In one method, the desired temperature was determined by drawing two tangents at the high-temperature zone of the crystallization and/or melting thermogram and the baseline at which these lines intersected each other at one point (i.e. Tcon1 and Tmet1). The other method was derived from drawing a line along to the baseline until it intersected the thermogram at one point. The intersection point showed the temperature at which the crystallization curve was deviated from the baseline because the crystallization process just initiated (Tcon2) and/or the melting curve reached the baseline (Tmet2), revealing the temperature at which the last trace of crystals (i.e. the thickest lamella) were disappeared. Moreover, Scry and Smel were obtained by calculating the slope of the linear section of the crystallization and melting thermograms at their high-temperature zones, respectively.
63
The results indicated that with the increase in PMMA content, at the constant amount of MWCNTs (i.e. 5 wt%), Tcpt, Tcon1, Tmpt remained nearly untouched except for the highest PMMA loading at which the values reduced a few degree of Celsius. The values of Tmet1 and Tmet2 are also relatively constant. It was observed that Tcon2 is much higher for the nanocomposites than that of EVA. This evidently indicated that the addition of PMMA and MWCNTs increased the heterogeneous nucleation sites and thus encouraged the crystallization process to be started at higher temperatures than that of EVA itself. This could be continued until reaching the nanocomposites containing up to 65 wt% PMMA beyond which Tcon2 reduced but the value still remained about 4°C higher than that of EVA. The crystallization ability of the EVA domains is decreased with the increase in PMMA loading because the crystallizable domains are being more separated from each other, and as the PMMA continuous phase has already become solidified before starting the EVA crystallization process, the movement of EVA chains to the crystallization front is highly prevented. Therefore, the Xc value is gradually reduced with the increase in PMMA content. The values of Scry and Smel are steadily reduced with the addition of PMMA in the nanocomposites, implying that the rate of crystallization and melting both decreased with the increase in PMMA loading. This could be attributed to the dilution effect and imposing restrictions on EVA molecular mobility by PMMA. However, the rate of crystallization was larger than that for the melting process in each case, reflecting the initial part of crystallization process was taken place faster than the final part of melting process. (a) Crystallization and (b) melting thermograms of EVA and the nanocomposites. Crystallization and melting data for EVA and the other materials.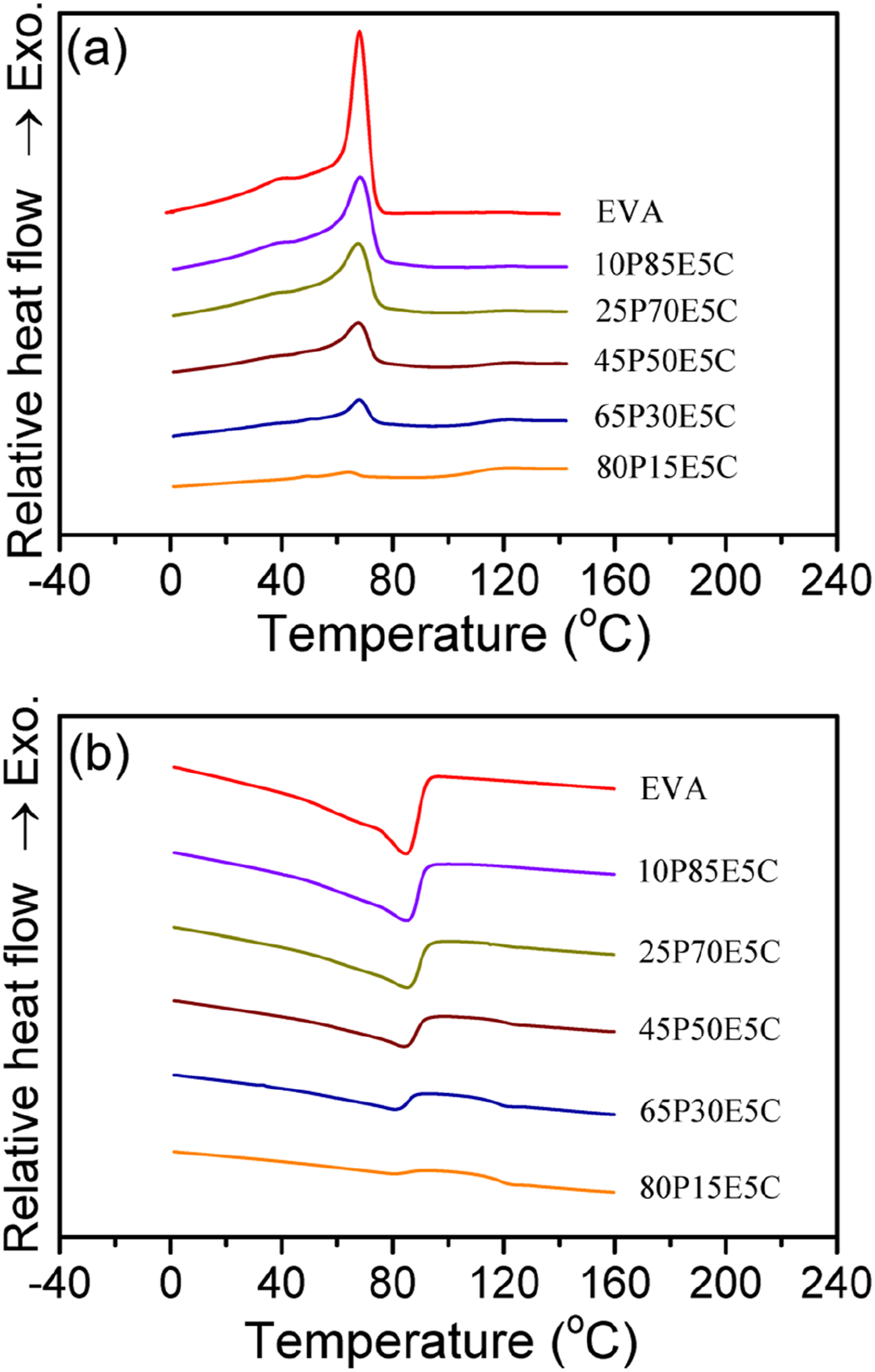
Rheological measurements
The plots of G′ and η* with ω for PMMA/EVA/MWCNTs nanocomposites of different compositions and of the pristine polymers are shown in Figure 3(a) and (b). The figures showed that the values of both parameters increased with the addition of the nanofiller, in the terminal region, especially for the nanocomposites having 65 wt% PMMA. The growth of the nanofiller network structures within a material could generally suppress the long-range macromolecular chain motions and consequently enhance the two quantity values.
64
The parent polymers and the nanocomposites containing up to 50 wt% EVA acted analogous to the Newtonian fluids at low ω region followed by shear thinning at high ω region. However, those of containing 70 and 85 wt% EVA behaved as shear thinning fluids in the whole ω range examined. Therefore, the two materials could possibly form the gel-like structures at these particular compositions. The plot of (a) G′ and (b) η* versus ω for the pristine polymers and the nanocomposites at 180°C.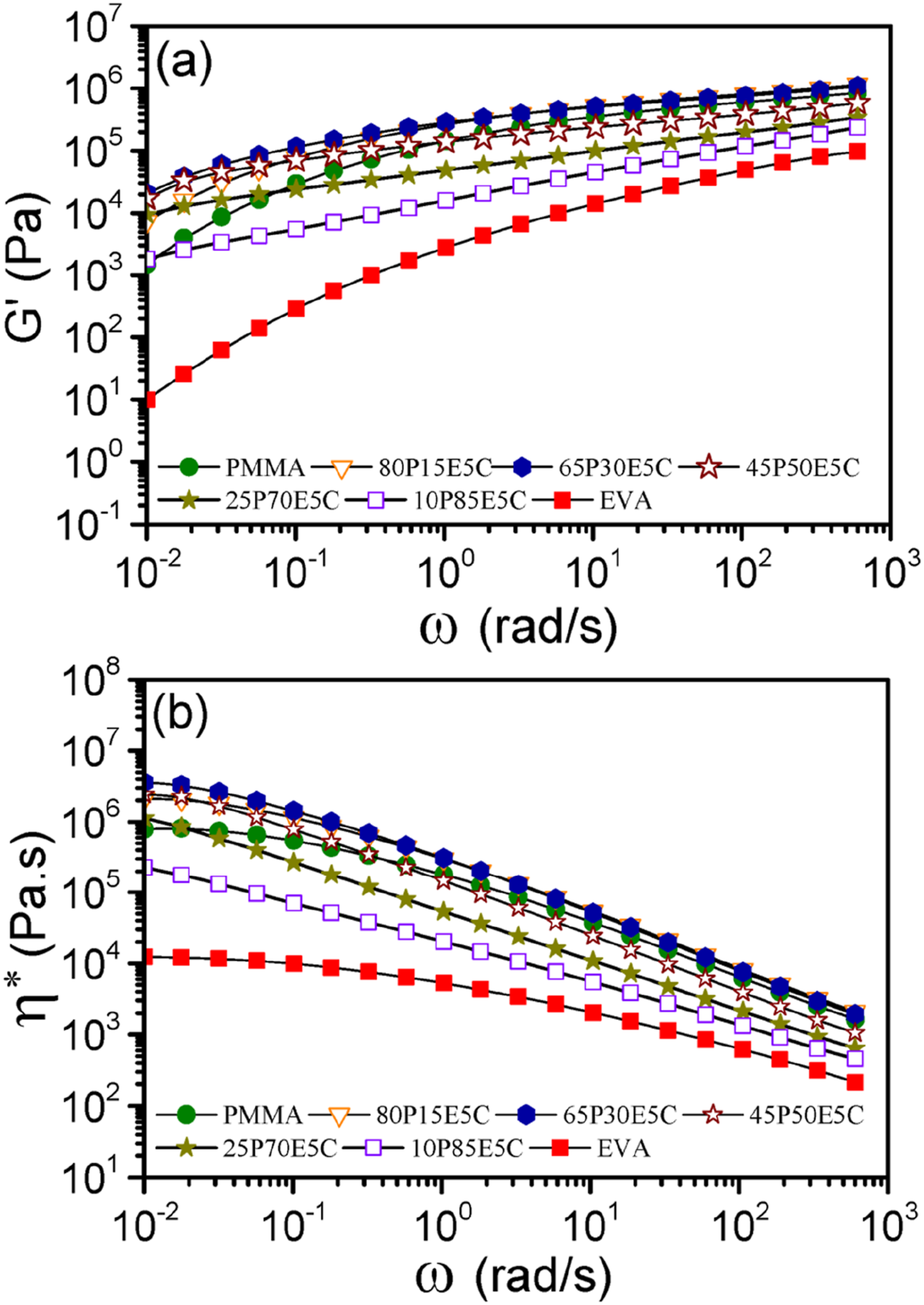
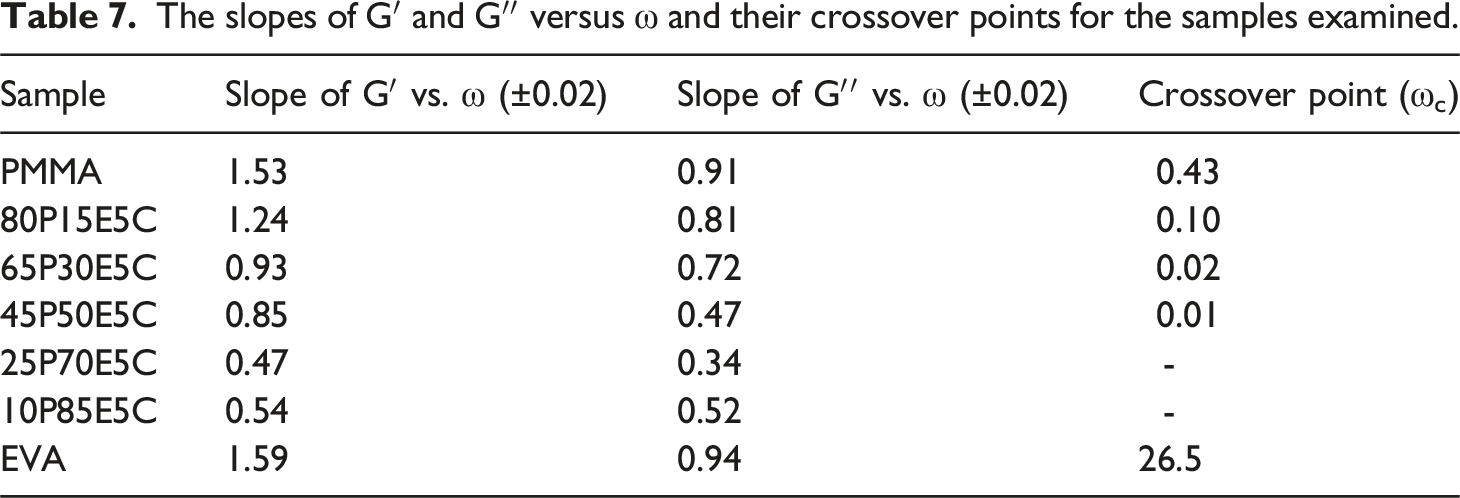
The slopes of G′ and G′′ versus ω and their crossover points for the samples examined.
The plot of tanδ against ω for the parent polymers and the nanocomposites is shown in Figure 4. At the terminal zone the elasticity of the materials, especially the parent polymers, were lower because more freedom was available for the molecular chains to move and relax than that of high ω region. The elasticity extent of the nanocomposites increased with the increase in EVA content until 70 wt% at low ω region and tanδ value stayed relatively constant for 25P70E5C and 10P85E5C throughout the ω range studied. However, at medium to high ω range, the damping characteristics of the materials were changed relative to one another. This could mainly be attributed to the fact that the nano-sized MWCNTs were played their roles to impede the long-range motions with length scales more than an entanglement of polymeric structure and not the short-range dynamic motions of the macromolecular chains.
67
The plots of tanδ versus ω for the parent polymers and their nanocomposites at 180°C.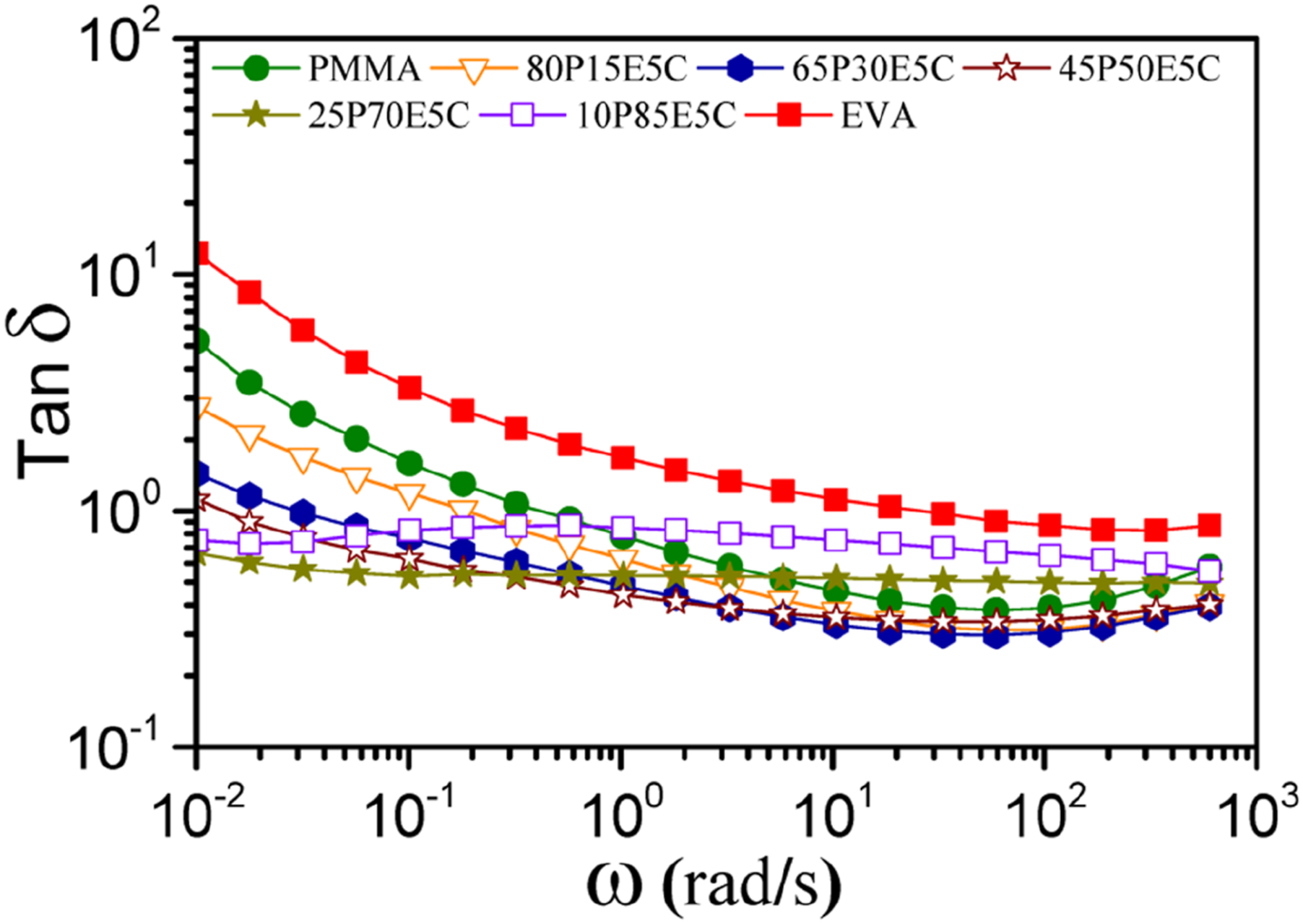
The van Gurp-Palmen plot, which is created by plotting the phase angle (δ) against the complex shear modulus (G*(ω)),
68
is a very susceptible tool for the identification of the composition, macromolecular structure and relaxation behavior of polymers and their nanocomposites.69,70 The constructed van Gurp-Palmen plot for the system studied is depicted in Figure 5. The results indicated that δ for the parents polymers was close to 90° in lower G*(ω) values and also it could be seen that δ decreased with increasing in EVA concentration up to 70 wt% at constant nanofiller content. The flow behavior of the materials, except for 25P70E5C and 10P85E5C, was similar to that of a viscoelastic liquid at low ω region. However, the compositions containing 70 and 85 wt% EVA showed a significant reduction in their phase angle to below 45°, reflecting they could both behave as viscoelastic solids. The van Gurp-Palmen plots for the parent polymers and their nanocomposites at 180°C.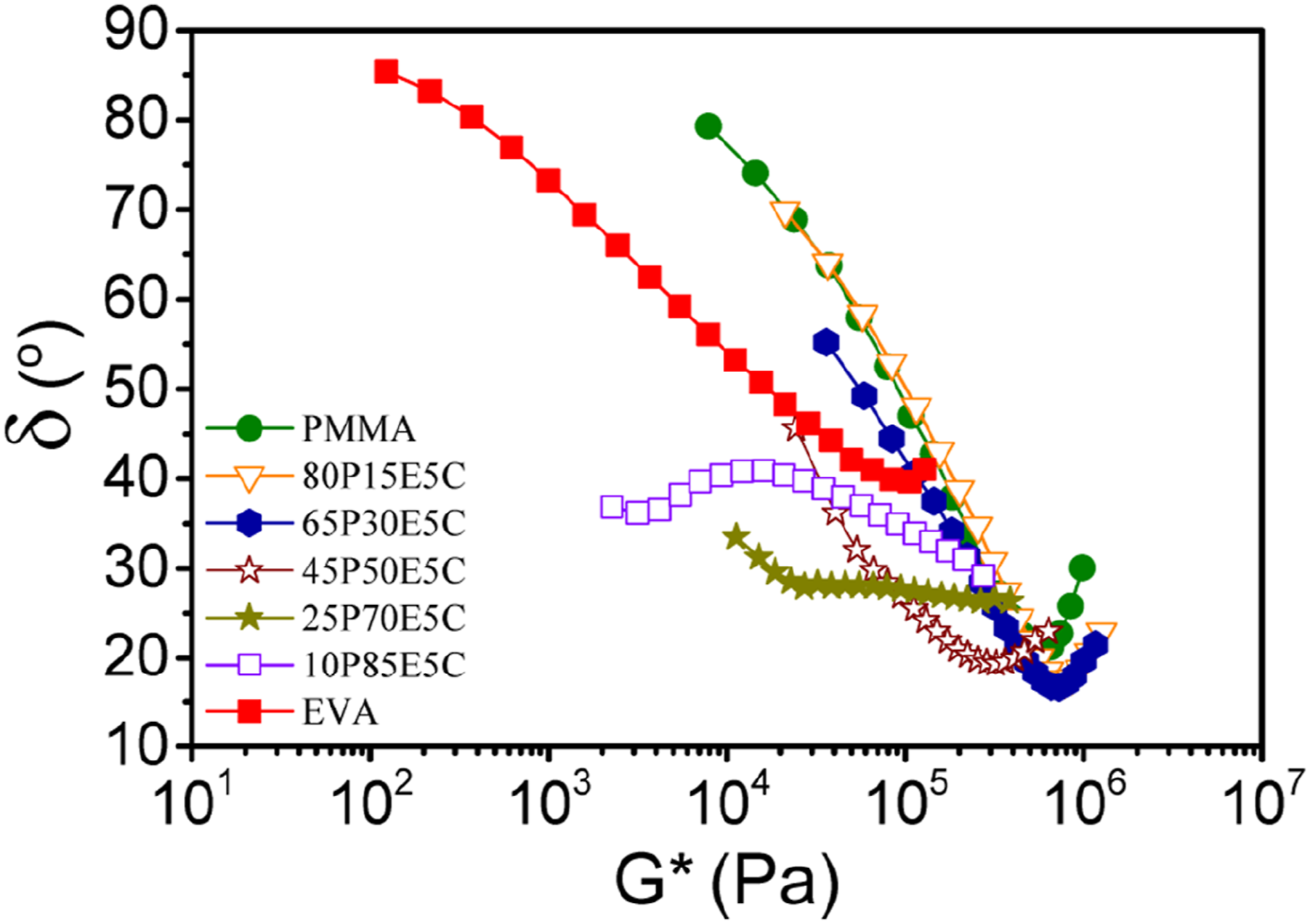
The Winter-Chambon71,72 method was utilized to elucidate whether the physical gelation was taken place for the nanocomposites or not. They proposed that the stress relaxation modulus (G(t)) of a critical gel could be designated by a power-law relationship which illustrated the self-similar or fractal formation of clusters at the gel point73,74; The plots of tanδ versus EVA content for several selected ω values. The critical gel point is shown by an arrow.
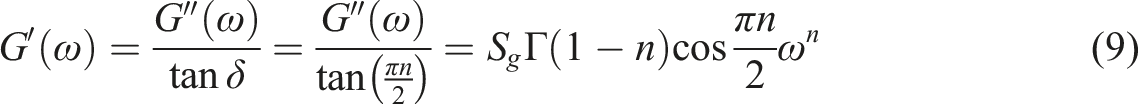
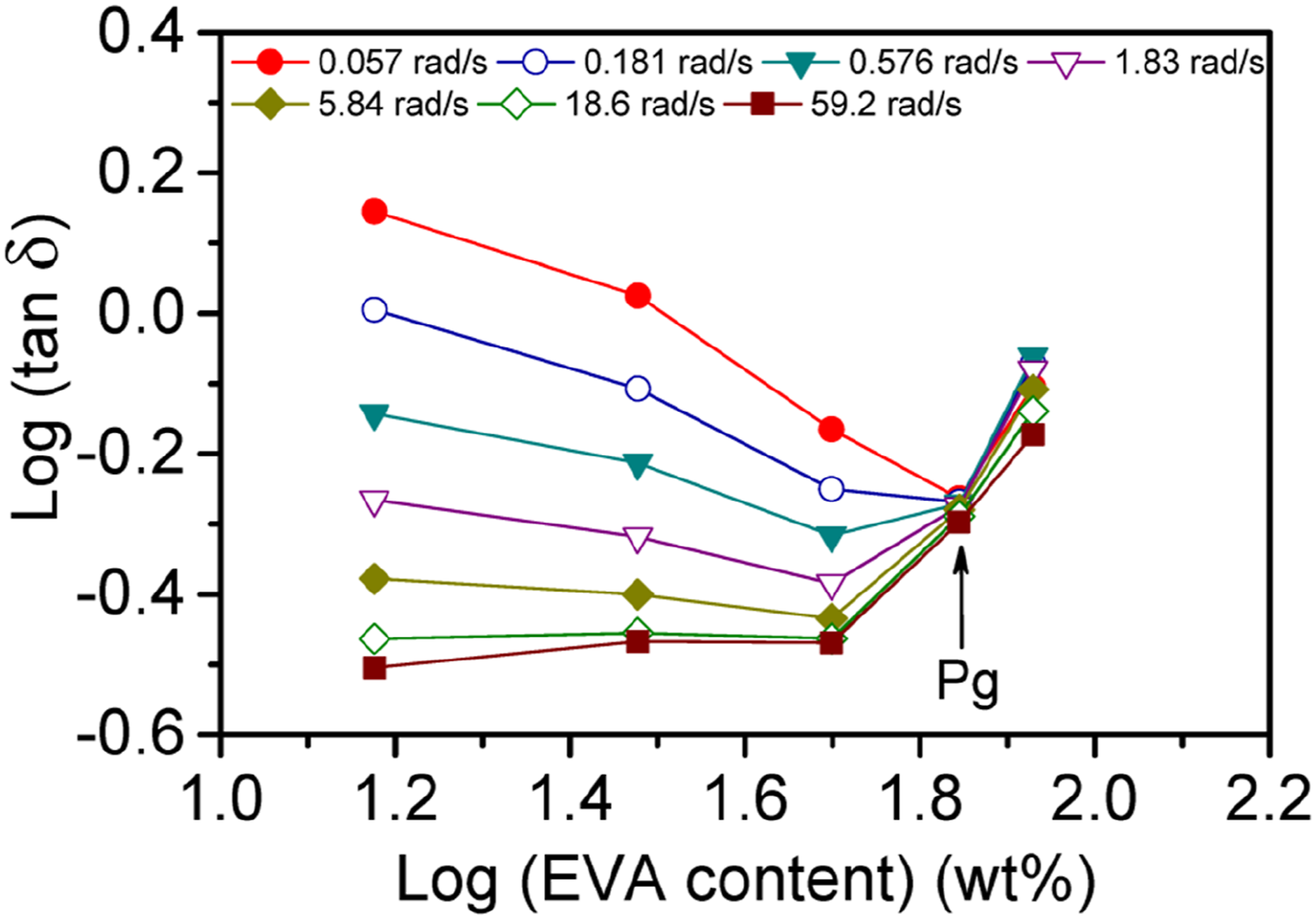
The quantity of tanδ at the meeting point could be employed to determine the The plots of G′ and G″/tan (πn/2) versus EVA content for several selected ω values. The former and the later values are indicated by the solid and open symbols, respectively and the critical gel point is shown by an arrow.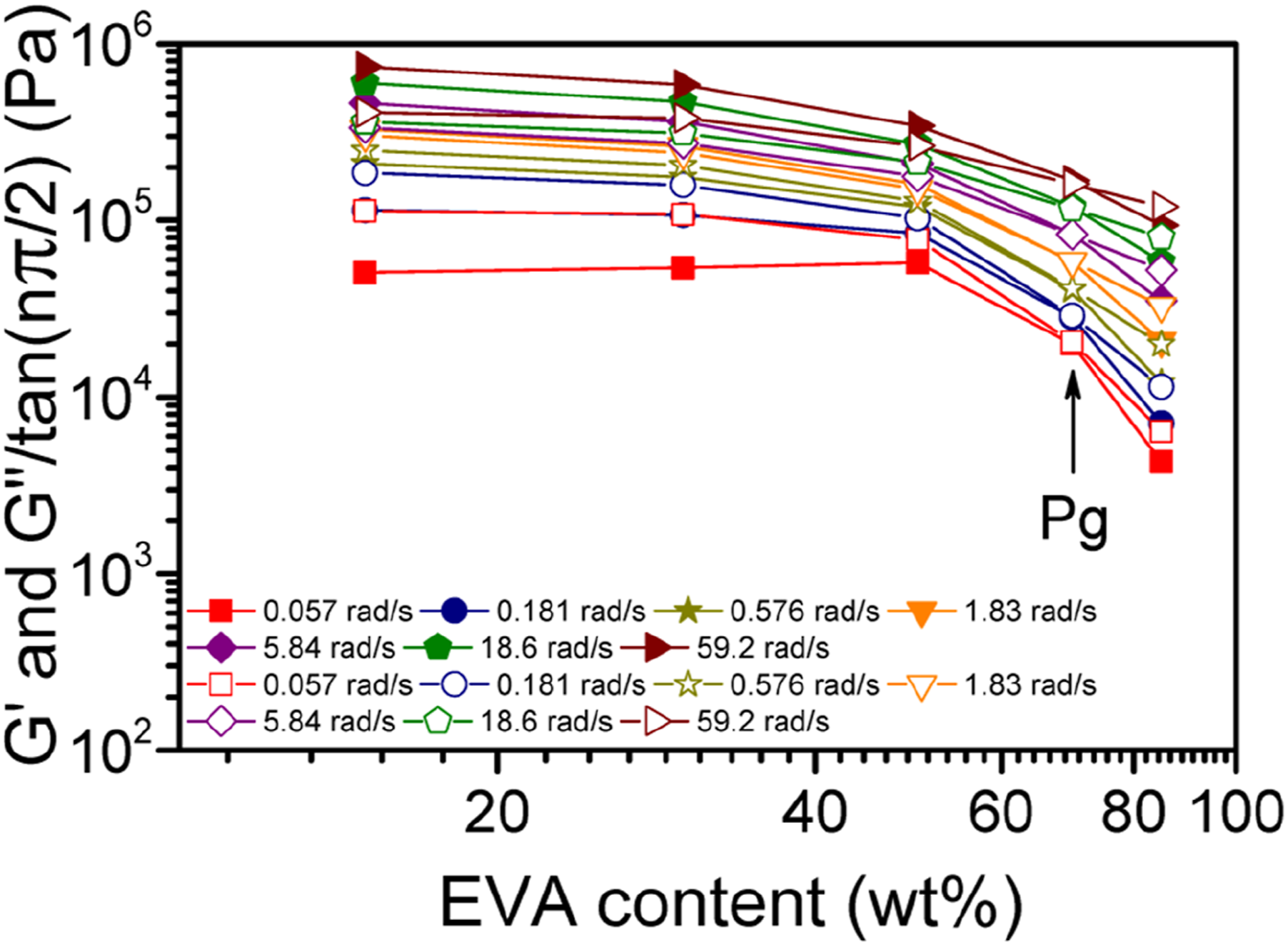
Excluded volume screening is generally occurred after the development of chemical or physical gels. Near the point at which a gel is formed, as far as to the screening effect is related, the system behaves analogous to a molten state.
78
The statistical self-similarity of a polymeric material is generally demonstrated by a fractal dimension (df). Presuming that the excluded and hydrodynamic interactions are totally screened at Pg, the value of df could be calculated from
79
;
Thermal stability
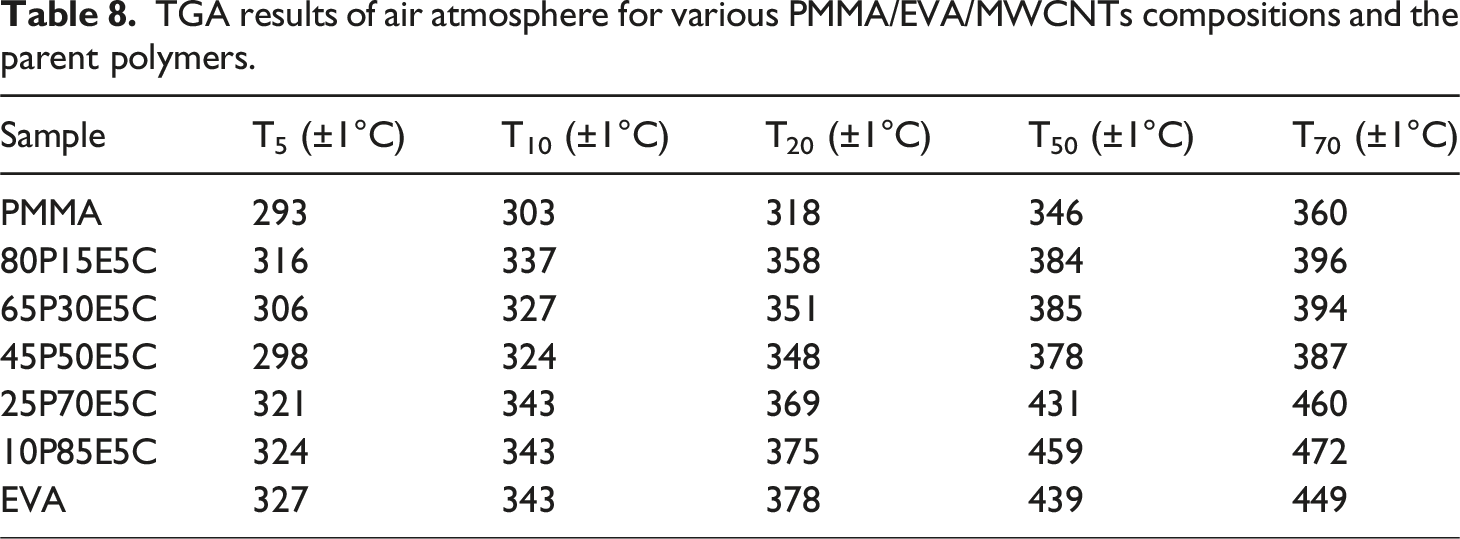
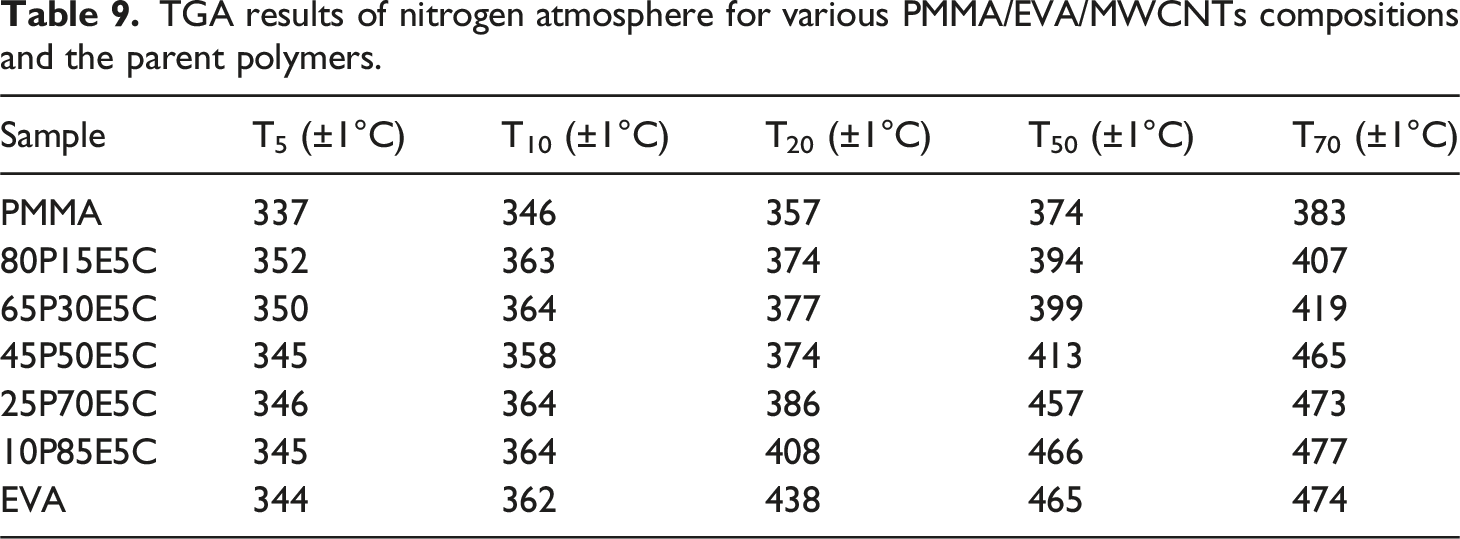
TGA decomposition behavior of PMMA/EVA/MWCNTs nanocomposites along with the parent polymers in air and nitrogen atmospheres are shown in Figure 8 (a) and (b), respectively. The temperature at 5 wt% (T5), 10 wt% (T10), 20 wt% (T20), 50 wt% (T50) and 70 wt% (T70) weight loss were determined for both atmospheres to examine the thermal stability of different compositions. The corresponding TGA results are gathered in Tables 8 and 9 for air and nitrogen, respectively. The degradation of PMMA was taken place in one
81
and that for EVA in two stages
82
in both atmospheres. PMMA has been reported to decompose in nitrogen due to the random C-C bond scissions of the macromolecular backbone. However, the polymer is degraded because of depolymerization as well as C-C bond breakage of PMMA chains in air.
83
The first degradation stage of EVA, is related to the elimination of acetate side groups as acetic acid, which is also known as deacetylation, leading to generate an unsaturated macromolecular chains or polyene in both atmospheres. The following step is attributed to the allylic chain scission of the polyene in an inert atmosphere and its aromatization into a char in an oxidative atmosphere. The char could then be oxidized into carbon dioxide above 500°C and thus the complete decomposition of EVA might eventually take place.84–86 The two pristine polymers were found to experience total decomposition as if little or no chars could leave behind in both atmospheres. However, the thermal degradation behaviors of the materials under air are significantly different from those of established under nitrogen atmosphere. The samples scanned under air, mostly because of the auto-oxidation reaction, exhibited lower degradation temperatures in comparison with those examined under nitrogen atmosphere.
87
It was revealed that the thermal stability of the nanocomposites, both in air and nitrogen, was higher than that of the pristine PMMA as well as their unfilled counterparts (data not shown here). Therefore, the filled materials experienced higher thermal stabilities in regard with all characteristics. This could be assigned to the dispersion state of the nanofiller and its high aspect ratio, which created both the labyrinth effects, strong physical adsorption of the macromolecular chains on the individual surface of the nanofiller particles and perhaps the role of each MWCNT as a radical scavenger function.87–89 These could postpone the emission of small gaseous substances trapped into the nanocomposites and shift the degradation of the filled materials to higher temperatures. Figure 8(a) reveals that not only the highest thermal stability belongs to 10P85E5C and then 25P70E5C, but also their thermal stability at high temperatures is even better than that of EVA itself. For instance, T50 and T70 of 10P85E5C are 20 and 23°C superior to those of EVA, respectively. This was in agreement with the existence of a substantial amount of MWCNTs in EVA phase of the nanocomposites. However, this was not the case for Figure 8(b) where it was clear that the thermal stability of the nanocomposites was strongly related to the extent of EVA in each composition. Moreover, the nanofiller was found to be more advantageous in increasing the thermal stability of the nanocomposites under oxidative substance such as air than that of nitrogen. TGA decomposition curves of different PMMA/EVA/MWCNTs nanocomposites and the parent polymers (a) under air atmosphere and (b) under nitrogen atmosphere. TGA results of air atmosphere for various PMMA/EVA/MWCNTs compositions and the parent polymers. TGA results of nitrogen atmosphere for various PMMA/EVA/MWCNTs compositions and the parent polymers.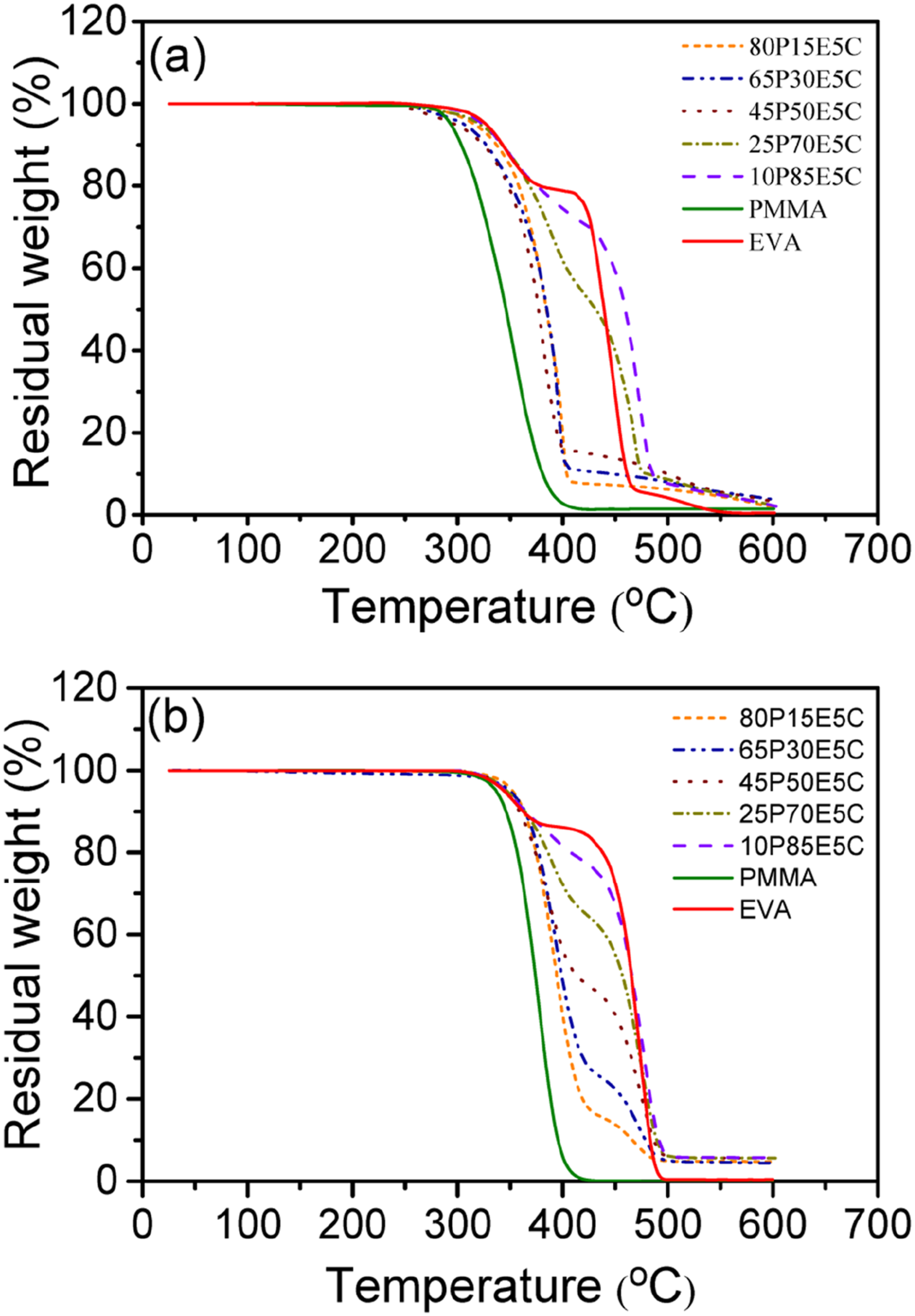
Conclusions
First, the PMMA/EVA/MWCNTs nanocomposites including 5 wt% MWCNTs and different polymer weight ratios were prepared through the mixing in the molten state. The preferred localization of MWCNTs were estimated by the determination of the surface free energy and then wetting coefficient. The results indicated that the nanofiller would thermodynamically prefer to migrate from EVA phase and accommodate into the PMMA phase. However, it was found that MWCNTs were located in both phases and a major amount of the nanofiller yet remained within the EVA domains. The rheological examinations pointed out that the nanocomposites containing 70 and 85 wt% EVA behaved as shear thinning materials throughout the entire ω range studied and no crossover point was found between the G′ and G″ of the two compositions. Using the Winter-Chambon method, the formation of a physical critical gel was verified in the nanocomposites containing 70 wt% EVA. The calculated fractal dimension value of the physical gel revealed that the system could be regarded as the one in which the excluded volume interactions were greatly screened. The crystallization and melting thermographs analysis indicated that the rate of crystallization and melting decreased with the increase in PMMA, reflecting PMMA imposed restrictions not only on the formation of crystalline domains of EVA phase but also on their melting. The TGA results showed that the thermal stability behaviors of the materials were significantly different in air and nitrogen atmosphere. The thermal stability of the materials were generally higher in nitrogen than that of in air atmosphere, however, the benefit of MWCNTs in putting off the degradation process was more influential in air than for nitrogen atmosphere. Now it is the time to raise the limitations of the present work. Studies on the influence of mixing sequences and mixing time on the nanofiller localization, morphology, thermal stability, flow behavior and other physical and mechanical properties of the nanocomposites should be accomplished for further analysis of the behavior and performance of the system. In this vein, performing additional characterization experiments such as transmission electron microscopy (TEM), atomic force microscopy (AFM) and dynamic mechanical thermal analysis (DMTA) could also provide some other beneficial information about this novel system.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported by Iran Polymer and Petrochemical Institute (Grant No. 31761207).
