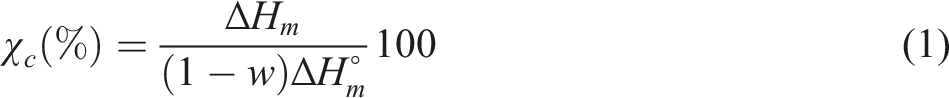Abstract
This study investigates the development of polypropylene (PP) composites reinforced with polymethylsilsesquioxane (PMSQ) microparticles at varying concentrations (0, 5, 10, and 15 wt%). The composites were produced via melt blending to evaluate the effect of PMSQ on their thermal, thermo-mechanical, and electrical properties. Differential scanning calorimetry (DSC) revealed slight changes in melting temperature, crystallization temperature, and crystallinity level with PMSQ addition. Thermogravimetric analysis (TGA) showed improved thermal stability, with residual mass increasing proportionally to PMSQ content. Dynamic mechanical analysis (DMA) indicated enhanced stiffness, as showed by a 32% increasing in storage modulus at 15 wt% PMSQ. However, the glass transition temperature remained unchanged. The thermal conductivity was improved by 20% with 10 wt% PMSQ, coupled with a tenfold increase in electrical resistivity. These results suggest that PP/PMSQ composites are promising candidates for applications requiring efficient heat dissipation and high electrical insulation, such as in electrical and electronic systems.
Keywords
Introduction
Polymers are materials that are widely present in our daily lives. They offer several advantages such as being lightweight materials with excellent formability, good corrosion resistance, high electrical resistivity, and low cost. 1 However, they have low thermal conductivities usually ranging from 0.1 to 0.5 W/(m·K). They are also electrical insulators, 2 compared to metal (copper or aluminum for example). 3 This low thermal conductivity is the reason why they are widely used as thermal insulators.
Today, miniaturization of electronic components is generated by a growing demand for increasingly powerful devices (such as processors and energy storage materials), as well as the development of hybrid and electric vehicles, as well as the emergence of new applications such as light emitting diodes (LED). In this context, the thermal properties (temperature management) of these materials become a critical issue because several of their components generate heat, leading to important temperature increases and consequently, a loss of performance in electrical and electronic components. For example, an increase of only 2°C in electronic equipment can lead to a 10% loss in its performance. 4 The dissipation of this heat is therefore essential to maintain a stable and efficient performance for electronic materials. 5 This is why improving the thermal conductivity of polymers becomes a key objective in these areas, while still preserving their properties, especially electrical insulation.
Composites are generally developed to improve the thermal conductivity of polymers. This involves incorporating heat-conducting fillers into a polymer matrix. Among the most commonly used fillers are carbon nanotubes,6–10 graphene,11,12 graphite,13,14 metallic fillers,15,16 and ceramic fillers.17,18
Improving the thermal conductivity of polymers has been the subject of a great deal of published work. According to Han et al., the thermal conductivity of the matrix reached 1.37 W/(m.K), an increase of 523%, after that 10% of carbon nanotubes (CNT) were added to polystyrene (PS). 19 By adding 5% graphene to high density polyethylene (HDPE), Tarani et al. were able to increase the matrix conductivity by 48%. 20 Zaccone et al. found that the addition of 15 wt% of graphite to polypropylene (PP) also resulted in a significant increase (350%) in thermal conductivity of 0.9 W/(m.K). 21 Similarly, Wang et al. found that adding copper rods to polyacrylate significantly increased its heat conductivity. A significant rise (1350%) was noted with a rather modest mass fraction (0.9%) of copper. 22 Zhou et al. found that when ceramic fillers (aluminum nitride, AlN) were added to PP, the thermal conductivity increased by 13%. 23
Incorporating different fillers into a thermoplastic resin may alter its properties, not withstanding their effectiveness in increasing the thermal conductivity of polymers. First, as these fillers are typically added to polymer matrices, their insulating characteristics are generally decreased.21,24,25 In addition, the viscosity of polymers can be substantially increased by these fillers, which in turn makes them more difficult to process.26,27 Not all these fillers are equally hydrophilic and some have anisotropic thermal conductivity. 28 Finding the ideal composition window is therefore of the utmost importance, as it must provide a high enough thermal conductivity while preserving the mechanical, thermo-mechanical, and electrical insulating properties of the polymer matrix, while keeping the viscosity reasonably low. Particles made of a hybrid material called polymethylsilsesquioxane (PMSQ) might be an interesting substitute.
PMSQ are hybrid spherical particles (with both inorganic and organic functionalities) belonging to the silsesquioxane family. They are synthesized via a sol–gel process in two steps: a condensation step, preceded by a hydrolysis phase of methyltrimethoxysilane (MTMS). These two steps make the reaction easier to control.
29
Figure 1 presents the general formula of PMSQ as RSiO1.5, where R can be hydrogen or an organic group such as phenyl, methyl or vinyl.30,31 The shape and size of the particles depend on the water/MTMS ratio, temperature, reaction time, and catalyst concentration, as well as the stirring level. Several authors synthesized PMSQ particles using various methods.32–35 In their work, Baatti et al. used the sol-gel method to obtain PMSQ nanoparticles with a ladder structure.
29
However, spherical microparticles were also synthesized.
36
The properties of PMSQ are similar to inorganic silica particles, featuring high thermal stability and solvent resistance, as well as flame-retardant efficiency.36,37 The presence of methyl groups on its surface makes PMSQ compatible with most hydrophobic polymers.
38
This reinforcement can be added in a hydrophobic thermoplastic matrix, without needing a surface treatment or to add a coupling agent. Moreover, the spherical shape of PMSQ particles is beneficial for achieving an appropriate viscosity.
1
Its chemical nature can maintain, or even enhance, the electrical insulating properties of the matrix. For instance, PMSQ is a widely studied filler for printed circuit substrates, due to its hybrid organic-inorganic structure, low dielectric constant, and high thermal stability, among other advantages.
36
Thus, based on the literature, it seems worthwhile to further study the use of PMSQ as reinforcement. Chemical structure of polymethylsesquioxane (PMSQ).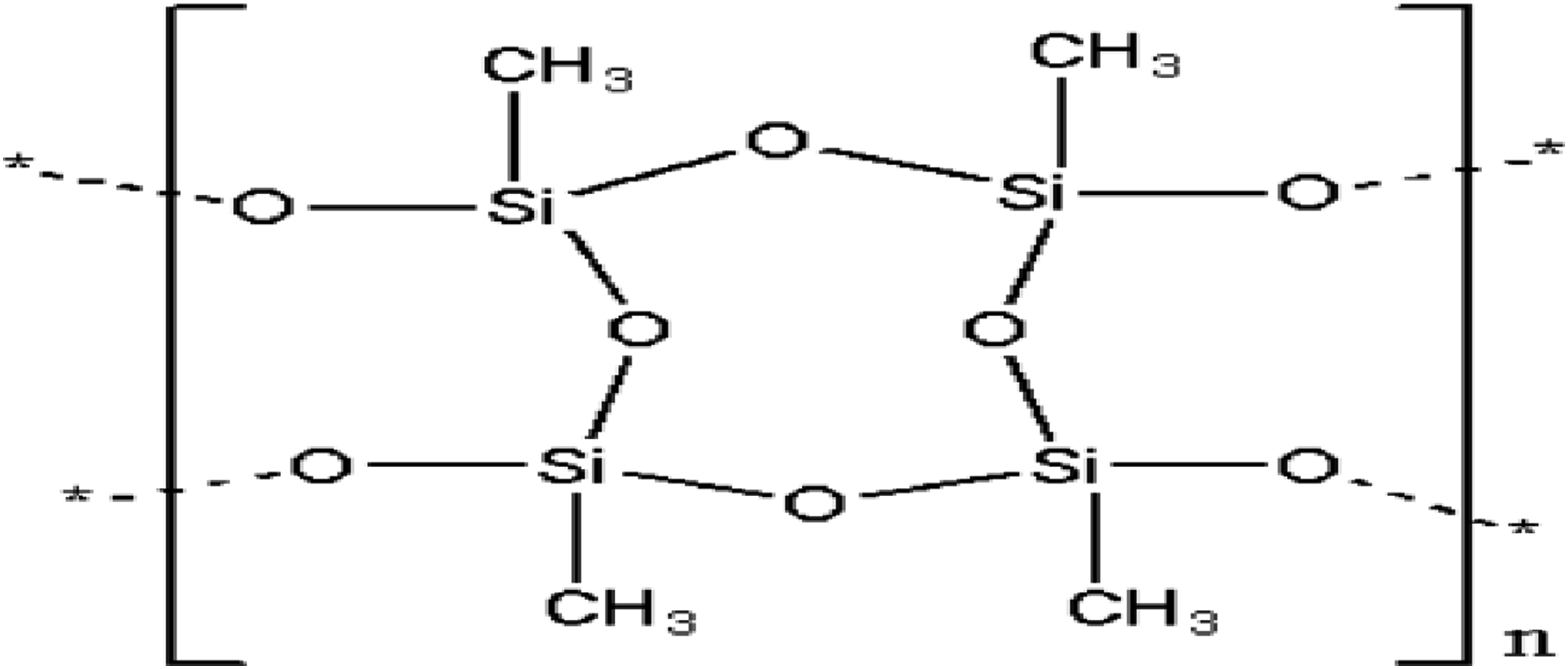
This study presents an innovative approach to enhance polymer composites by incorporating polymethylsilsesquioxane (PMSQ) microparticles into polypropylene, offering a unique solution to the longstanding challenge of improving thermal conductivity while preserving electrical insulation and processability. Unlike conventional fillers that typically compromise one or more of these critical properties, PMSQ’s distinctive hybrid organic-inorganic structure enables simultaneous enhancement of thermal performance and electrical resistance without the processing difficulties associated with other additives. The spherical morphology and inherent compatibility of PMSQ with hydrophobic polymers eliminate the need for surface treatments while maintaining favorable viscosity during manufacturing. By carefully investigating the relationship between PMSQ content and material properties, this work demonstrates significant improvements in both thermal management and mechanical performance, providing a valuable advancement.
Materials and methods
Materials
Polypropylene (PP), provided by TAIRI PRO (Taiwan), was used as the matrix. It has a density of 0.90 g/cm3, a melting temperature of 155°C, and a melt flow index of 33 g/10 min under a 2.16 kg load at 210°C. The polymethylsilsesquioxane (PMSQ, Figure 1) used in this study, consisting of spherical microparticles ranging from 2 μm to 12 μm in diameter, was purchased from Guangzhou Batai New Material Technology Co., Ltd. (China). All products were used as received.
Composite Manufacturing
The composites were prepared in an internal mixer, THERMO HAAKE, with the PolySoft Mixer RHEOMIX 3000 and Rotors roller 3000E. The mixing temperature was set at 180°C, which is above the PP melting temperature. The rotational speed and mixing time were maintained at 50 rpm and 5 min, respectively. Different formulations were carried out, namely 5, 10 and 15 wt% PMSQ. For each formulation, the matrix was melted first (5 min), before the reinforcement was added during 5 min. After the PP/PMSQ composites were prepared, the composite granules from the mixer were ground to produce smaller pellets. The machine used was a rotary knife mill from Retsch (Newtown, USA) with a RO-TAP sieve from W.S. Tyler (Mentor, USA). To characterize the composites, samples (specimens, disks, plates) were prepared using an injection molding press Haitian MARS130/II600 (Ningbo, China). The injection, screw, and die temperatures were set at 180°C, 170°C and 160°C, respectively (Figure 2). Schematic representation of the different stages of the composite fabrication process.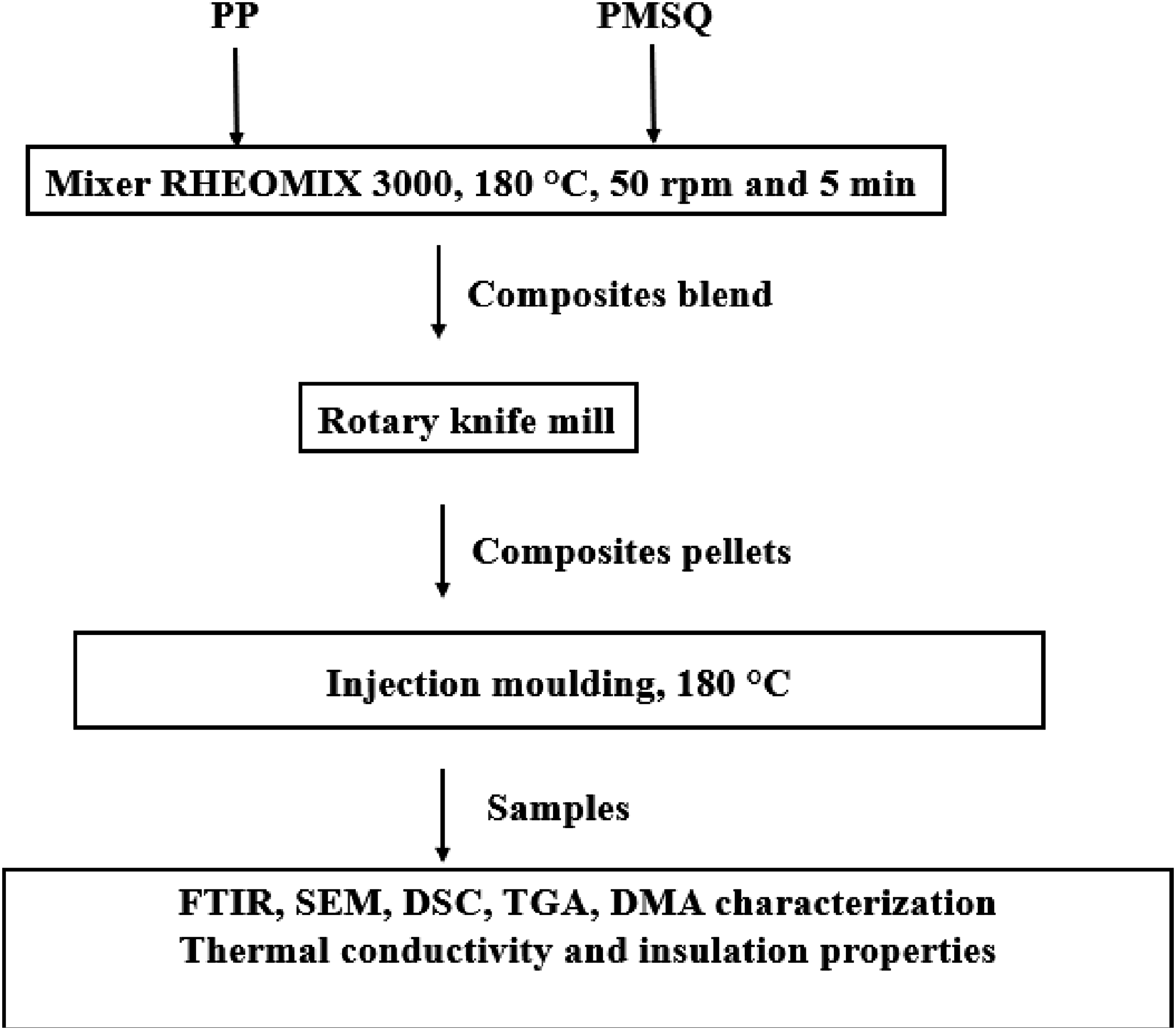
Characterizations
The infrared spectra of PMSQ powder, neat PP and its composites were obtained by a Fourier transform infrared spectrometer (FTIR) in transmittance mode with a resolution of 8 cm−1 and 64 scans in the range of 4000 to 400 cm−1 at room temperature. The tests were carried out using Shimadzu IR Tracer-100 (Japan). X-ray diffraction (XRD) was performed on the PMSQ powder to determine its crystalline structure. The analysis was conducted using a Bruker (USA) D8 diffractometer with copper Kα radiation (λ = 1.54 Å). The measurement was carried out over a two-theta range from 0° to 80°.
Thermogravimetric analysis (TGA) of the PMSQ powder, neat PP and its composites were performed on a TA Instruments Q50 device (New Castle, USA). About 25 mg were heated from room temperature to 600°C at 10°C/min under a nitrogen atmosphere.
A morphological study was conducted to examine the dispersion state of PMSQ particles inside the PP matrix. The analysis was performed using a JEOL JSM-840 scanning electron microscope (SEM) operating at 15 kV. The observations were carried out on the fractured surface of the samples after impact testing.
Differential scanning calorimetry (DSC) was performed to determine the melting temperature, glass transition temperature, melting and crystallization enthalpies, as well as the crystallinity degree of the composites. The analyses were conducted using a Q20 analyzer from TA Instruments (USA). About 15 mg were heated from −20 to 195°C at 10°C/min under a nitrogen atmosphere. To erase their thermal history, an isothermal step was set at 195°C for 1 minute. The samples were then cooled from 195°C to −20°C. This cycle was repeated three times in an inert atmosphere, and the reported data were obtained from the third heating and cooling cycle. The crystallinity degree of the samples was calculated from the melting enthalpy during the third heating cycle using the following equation
39
:
Dynamic mechanical analysis (DMA) tests were performed using a DMA Q800 v20.24 Build 43 instrument (TA Instruments, New Castle, USA) in the tensile configuration following ASTM D 5026. The samples from injection molding (35 cm × 12.6 cm × 0.2 cm) were analyzed over a temperature range from −50°C to 120°C with a heating rate of 5°C/min. The frequency was set at 1 Hz under a strain of 3%.
A custom-designed thermal conductivity analyzer was used to evaluate the thermal conductivity (k) of samples measuring 50 mm × 50 mm following ASTM E1225. The measurement device consists of two plates, upper and lower, maintained respectively at 33°C and 13°C resulting in a temperature difference (ΔT) of 20°C relative to the average room temperature of 23°C. Each sample was placed between thin aluminum sheets to minimize thermal contact resistance and subjected to a weight of 7.2 kg to ensure optimal contact between the components. The thermal conductivity was determined using Fourier’s law equation as
41
:
Surface resistivity and conductivity analyses of the neat PP and PP+10% PMSQ composites were performed at room temperature using a Keithley (USA) 6517-B electrometer coupled with a 8009-resistivity test fixture following ASTM D257. Square plate samples (10 × 10 mm2) with a thickness of 3 mm were prepared for each test. The applied voltage was fixed at 100 V during the measurements.
Results and Discussion
Characterization of the PMSQ Powder
Figure 3 shows the FTIR spectrum of PMSQ. The absorption bands at 763 and 852 cm−1 are associated to the out-of-plane vibrations of methyl group in Si-CH3. While the symmetric and asymmetric Si-O-Si vibrations are observed at 1002 and 1100 cm−1, respectively. These two bands are due to the Si-O-Si stretching and their presence indicates its ladder structure.29,36 The band at 1271 cm−1 is related to the Si-CH3 stretching vibrations, while the C-H vibrations of the methyl groups appear at 2974 cm−1. FTIR spectrum of the PMSQ powder.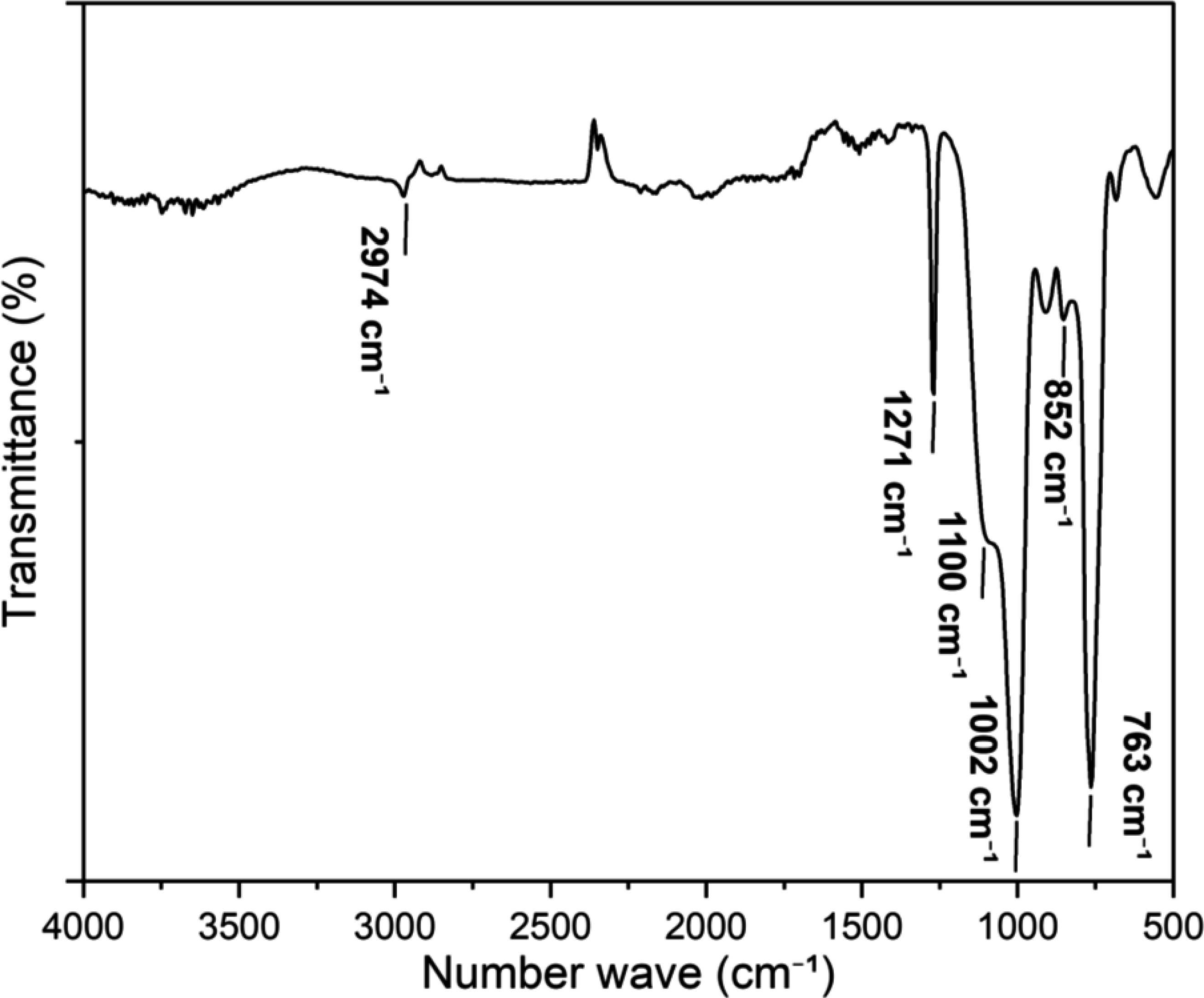
Figure 4 shows the XRD pattern of PMSQ. The lattice spacing is calculated using Bragg’s law as
42
: XRD spectrum of the PMSQ powder.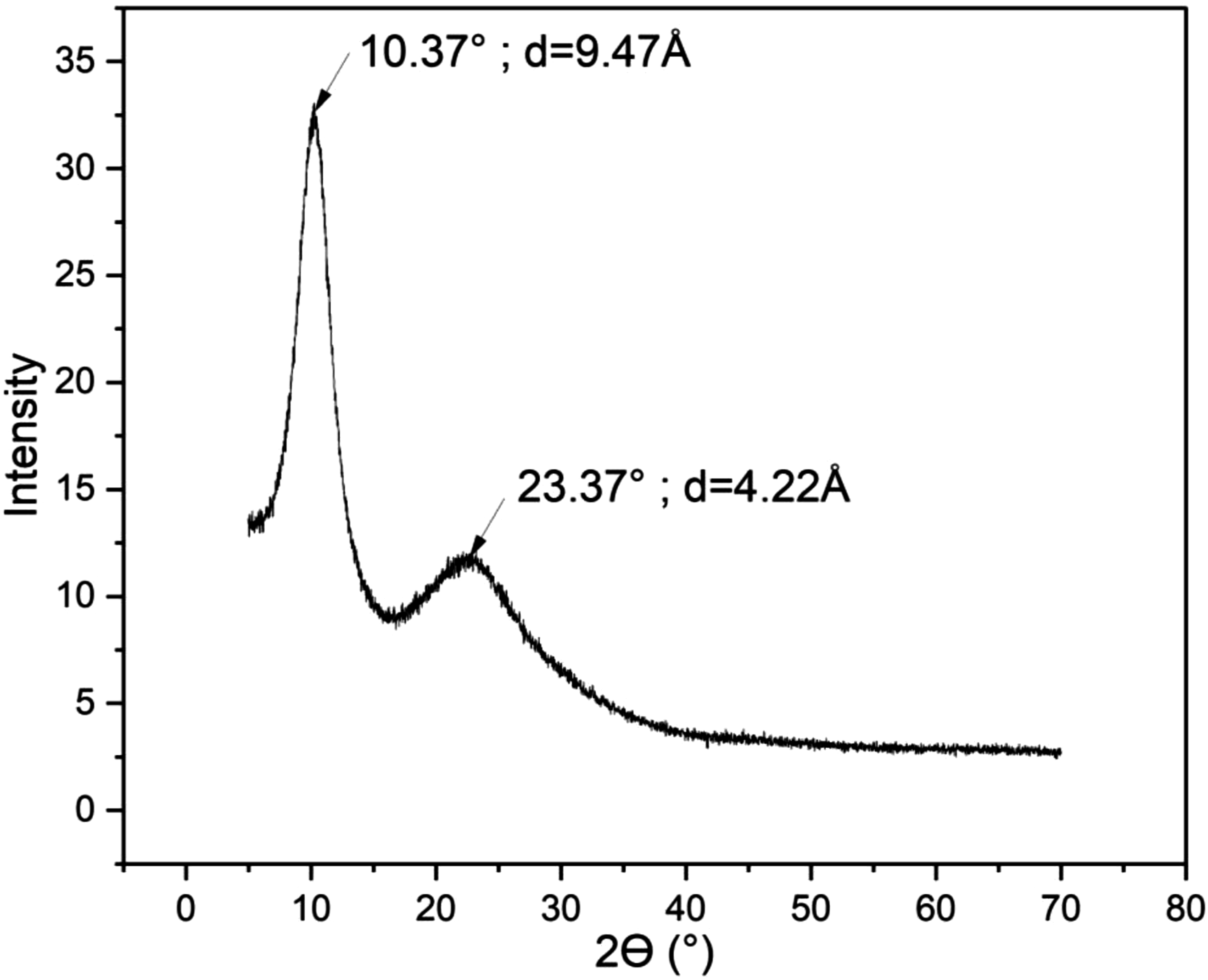
The XRD pattern of PMSQ reveals the presence of two distinct peaks, a confirmation of its crystalline structure. The first intense peak (2θ = 10.37°) corresponds to a spacing of 9.47 Å which is associated with the intermolecular distance between two PMSQ chains; i.e. the width of the molecular motif of PMSQ. However, a second less intense peak is observed at 23.37° and corresponds to a spacing of 4.22 Å. The latter can be associated with the thickness of the PMSQ particles. The presence of these two peaks confirms the ladder structure of PMSQ observed above. 36
Figure 5 shows the TGA and DTG (derivative thermogravimetric) curves of the PMSQ powder performed under a nitrogen (N2) atmosphere. The TGA curve indicates three decomposition stages. The first two stages occur between 200°C and 350°C and correspond to a mass loss of 2.6%. These transitions are attributed to the decomposition of residual silanol and methoxy groups. The third stage is observed at 350°C and 600°C and corresponds to the decomposition of methyl groups on the surface of the PMSQ.
30
It is associated with a mass loss of 7.6%. Thus, a total mass loss of only 10.2% is observed up to 600°C, indicating good thermal stability of the PMSQ. In other words, the high residual mass (89.8%) beyond 600°C may confirm the formation of a stable silica-rich inorganic residue, indicating the good thermal stability of PMSQ for high-temperature applications. TGA and DTG curves of the PMSQ powder.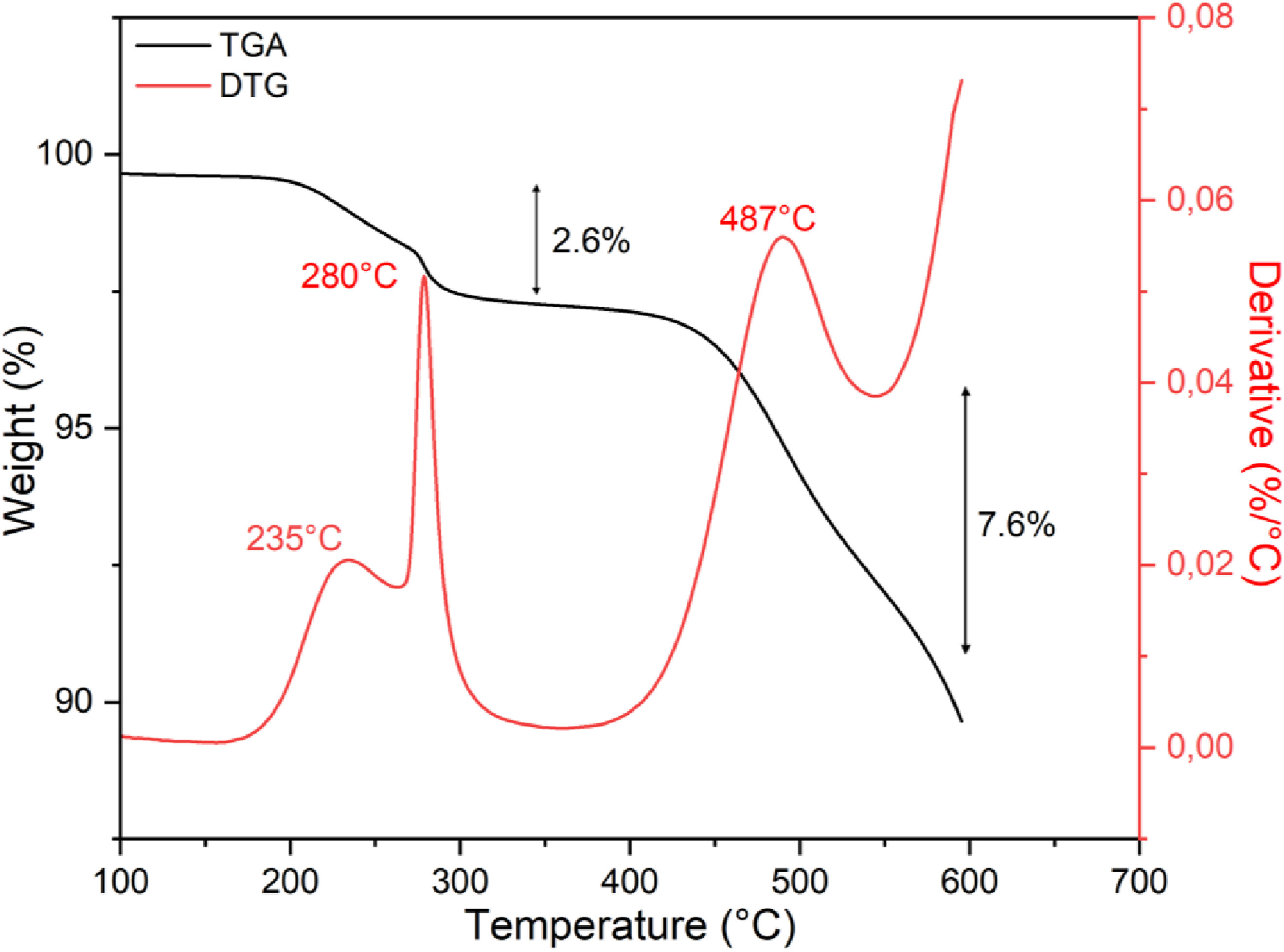
The DTG curve exhibits two peaks (235°C and 280°C), which can be attributed to the decomposition of residual silanol and methoxy groups, while the main decomposition peak around 480°C can be related to the degradation of methyl groups (Figure 5).
Evaluation of the Manufactured composites
Morphological studies
The FTIR spectra of neat PP and its composites are shown in Figure 6. Neat PP exhibits nine characteristic bands, including those at 972, 999, and 1165 cm−1, which are attributed to the vibrational and oscillatory movements of C-C bonds. The two bands at 1377 and 1458 cm−1 are associated with the symmetric and asymmetric C-H vibrations of CH3, respectively. The symmetric and asymmetric vibrations of -CH2 are linked to the bands at 2866 and 2916 cm−1. In contrast, the bands at 2835 and 2951 cm−1 correspond to the symmetric and asymmetric vibrations of -CH3, respectively.
44
(a) IR spectra of pure PP, PMSQ and PP-PMSQ composites and (b) IR spectra of the samples in the characteristic region of PMSQ.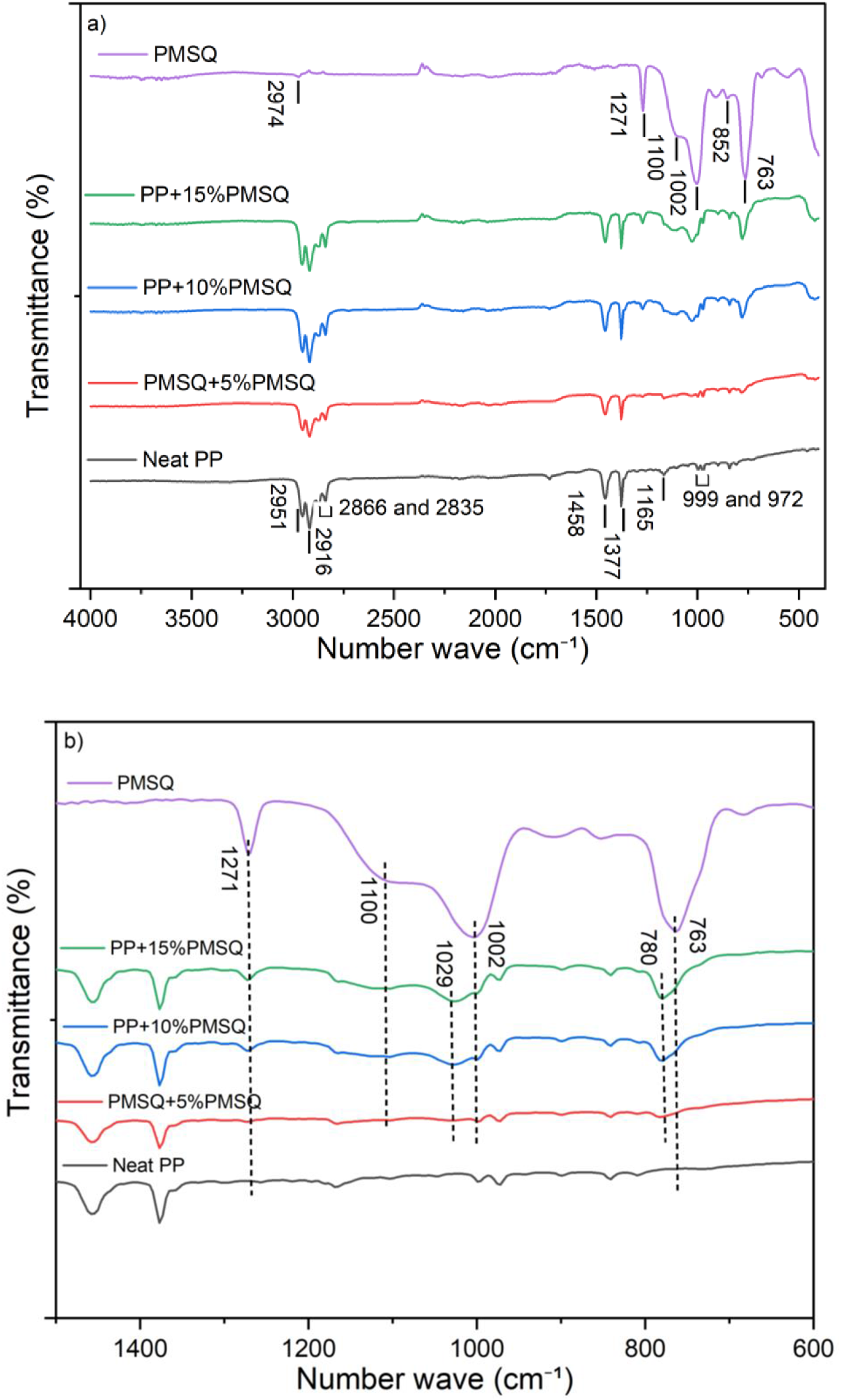
The FTIR analysis of PMSQ reveals the presence of several characteristic bands. The asymmetric vibrations of C-H bonds in methyl groups are observed at 2974 cm−1. The band at 1271 cm−1 is characteristic of the stretching vibrations of Si-CH3. The two bands at 1100 and 1002 cm−1 are attributed to the asymmetric and symmetric stretching vibrations of Si-O-Si, respectively. The bands at 852 and 763 cm−1 are associated to the out-of-plane vibrations of methyl group in Si-CH3.31,44
Between 1500 and 600 cm−1, the intensity of the bands of PMSQ microparticles increases as a function of their mass concentration in the composite’s spectra. The stretching vibration band Si-CH3, the asymmetric and symmetric bands vibration of Si-O-Si and the band of out-of-plane vibration of methyl group of Si-CH3 are all present in the IR spectra of the composites. However, a clear variation of the vibration frequencies of Si-O-Si and Si-CH3 were noted. The symmetric stretching vibration Si-O-Si and the vibration out-of-plane Si-CH3 at 1002 cm−1 and 763 cm−1, respectively in the PMSQ spectrum changed to 1029 cm−1 and 780 cm−1 in the IR spectrum of the composites. These observations could be related to changes in the chemical environment of silicon atoms, resulting from interactions between PMSQ and PP due to their compatibility. 45
Figure 7 presents SEM images of the neat PP and its composites. First, the neat PP shows a homogeneous structure without any defect, void, and cracks indicating that good processing conditions were used. Then, the other images reveal a good dispersion of PMSQ particles within the PP matrix. They also show an increase in the number (density) of PMSQ particles in the matrix with increasing content. The good dispersion of PMSQ particles may be the result of good compatibility between PP and PMSQ, probably due to their similar hydrophobic nature.46–48 However, voids can be observed in the composites, and this phenomenon becomes more pronounced at higher filler contents. SEM images of the fractured specimen: neat PP and its composites.
Thermal studies
Figure 8 shows the DSC curves of the neat PP and its composites. The melting temperature, crystallization temperature, enthalpy of fusion, enthalpy of crystallisation, and degree of crystallinity during the third cycle are listed in Table 1 for comparison. DSC curves of the neat PP and its composites. Summary of the thermal analysis data for the neat PP and its composites.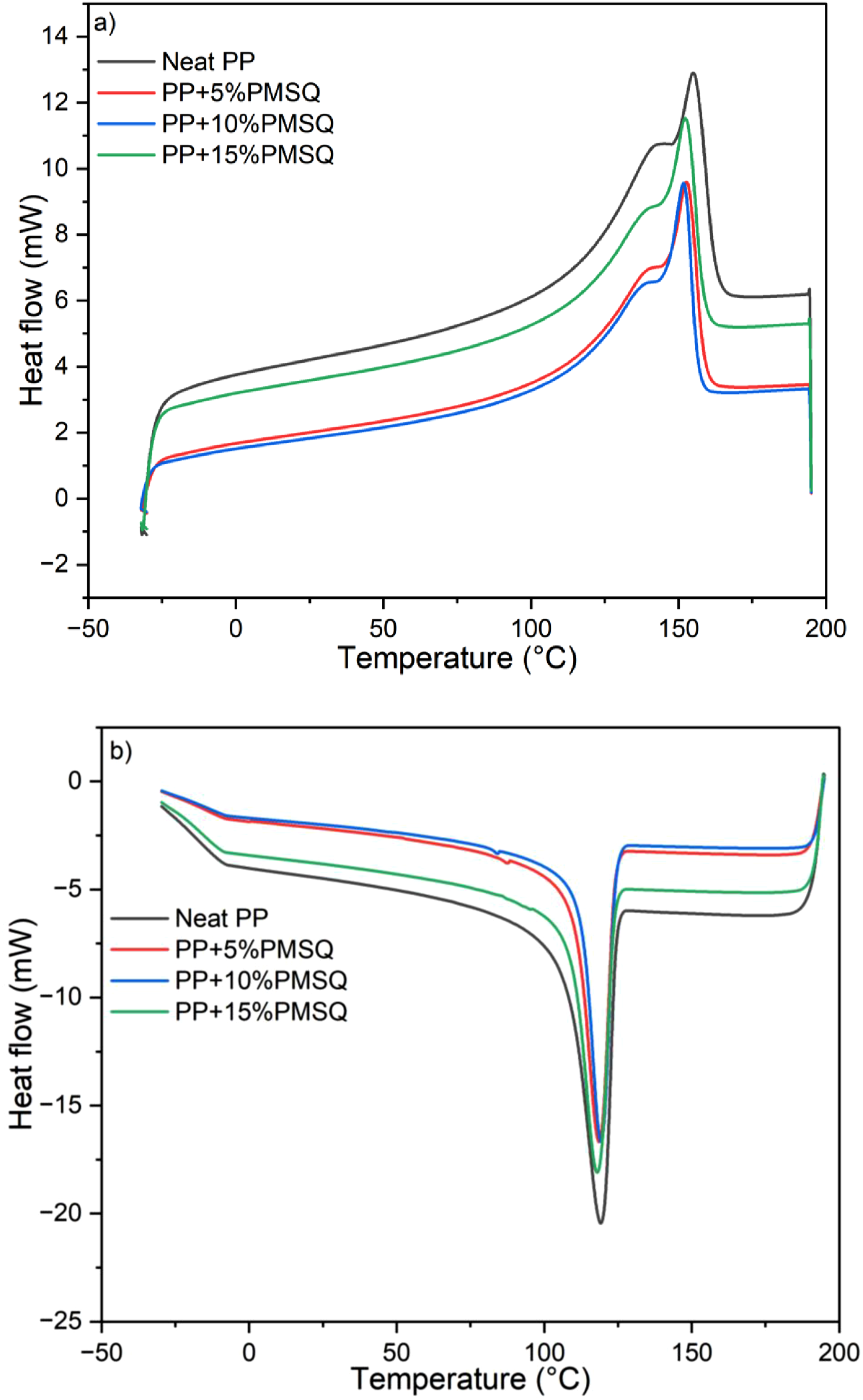
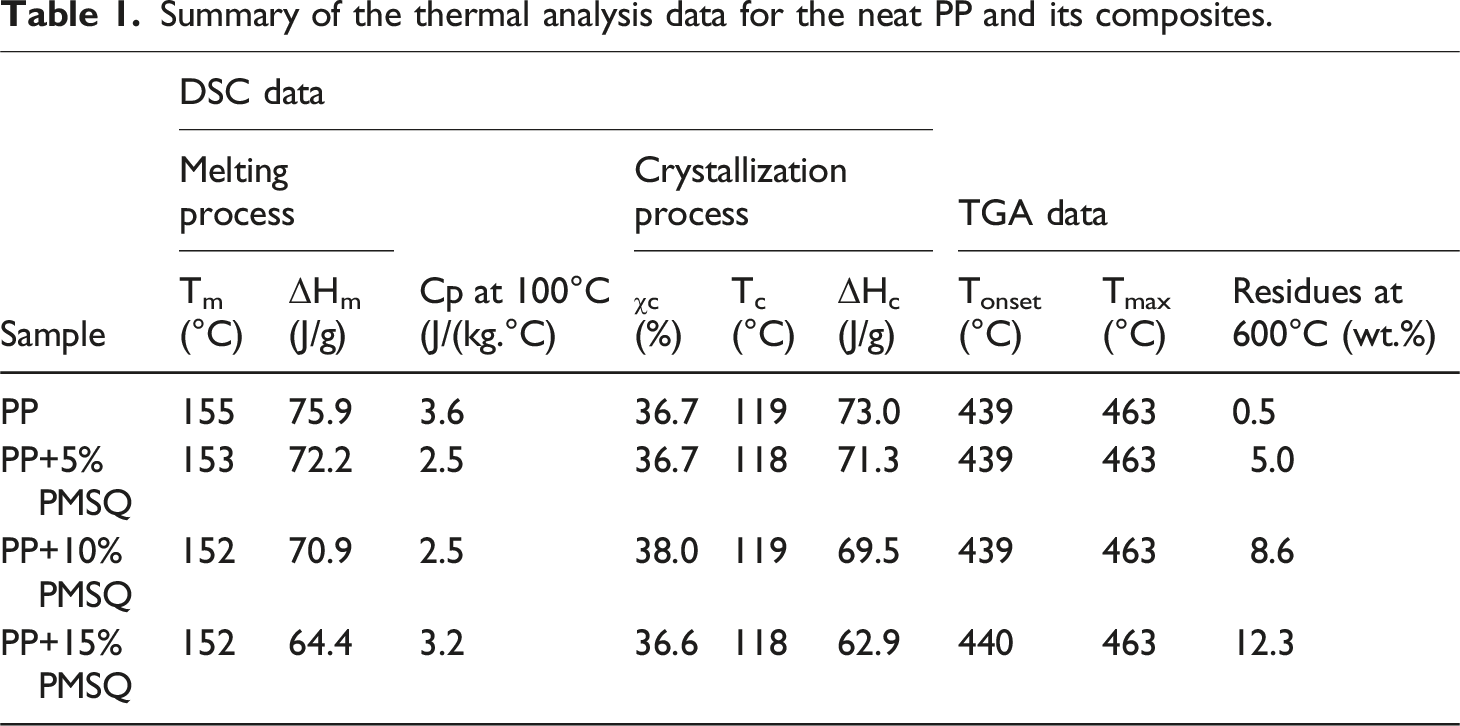
It can be seen that two endothermic peaks are present in the DSC curves (Figure 8(a)). The peak at 140°C is associated with a partial recrystallization of the amorphous phase induced by the cooling cycle, while the peak at 155°C corresponds to the melting temperature of PP. 49 It can also be noted that the melting temperature of PP (Tm = 155°C) is slightly affected by the addition of PMSQ particles, which would suggest that the size of the matrix crystals remain unchanged. 50 Furthermore, this slight decrease in melting temperature may be attributed to the heterogeneous nucleation effect of PMSQ particles, promoting the formation of a higher number of imperfect or less ordered crystals. These imperfect crystals require less thermal energy to melt, resulting in a reduced overall melting temperature of the composites. 51 This result was also observed in the literature by Hassan et al., after adding glass fiber in PP. 52 Generally, both the amount of nucleating agent and the mobility of the polymer chains affect the crystallinity rate when a filler is added to a thermoplastic matrix. 53 In this study, the PMSQ addition slightly affects the degree of crystallinity (χc) of the matrix. The most important increase in crystallinity (1.3%) is obtained by adding 10% PMSQ. This result can be attributed to the nucleating effect of PMSQ microparticles in the PP matrix, 45 leading to a slight increase in the degree of crystallization of the matrix. Similarly, the incorporation of titanium nanoparticles into PP was also reported to enhance crystallinity. Bendaoud et al. and Sun et al. observed increases of 4% and 4.52% in the crystallinity of PP by incorporating 7.5 wt% and 5 wt% of titanium nanoparticles respectively, which was attributed to the accelerating effect of nano titanium dioxide particles (TiO2) on the PP crystals and to the restriction of the polymer chains mobility.40,54 In addition, DSC results revealed that the crystallization temperature of PP is slightly reduced by the addition of PMSQ. With the addition of 15 wt% PMSQ, the crystallization temperature decreased from 119.1°C for neat PP to 117.8°C (Figure 8(b)).
The melting enthalpy also decreases when the microparticles were added to the matrix. Consequently, less energy is required to melt the composites compared to the neat matrix (75.9 J/g). The highest enthalpy decrease is 15% (64.4 J/g), which is achieved by adding 15% of reinforcement.
Heat capacity is a very important parameter as it measures the amount of heat required to raise the temperature of one unit of mass by one degree Celsius. The effect of adding PMSQ microparticles on the heat capacity (Cp) of PP is shown in Figure 9 and the values are reported in Table 1. It is clear that the presence of PMSQ led to lower heat capacity of the matrix. This result can be attributed to the low heat capacity of PMSQ itself, approximatively 1.5 J/(kg.°C),
55
and increased density of the composite limiting the mobility of the polymer chains induced by the presence of PMSQ.
56
Weidenfeller et al. obtained similar results by incorporating talc microparticles and copper rods into PP. The authors observed a decrease in heat capacity as the talc and copper content increased, while the thermal conductivity increased with filler addition. For example, adding 15 wt% of talc and copper led to specific heat capacities of 1.35 J/(kg.K) and 1.10 J/(kg.K) respectively, compared to 1.58 J/(kg.K) for the neat matrix. This corresponds to reductions of 15% and 30%.
2
The effect of PMSQ particles on the thermal conductivity of PP is discussed below. Heat capacity (Cp) of the neat PP and its composites.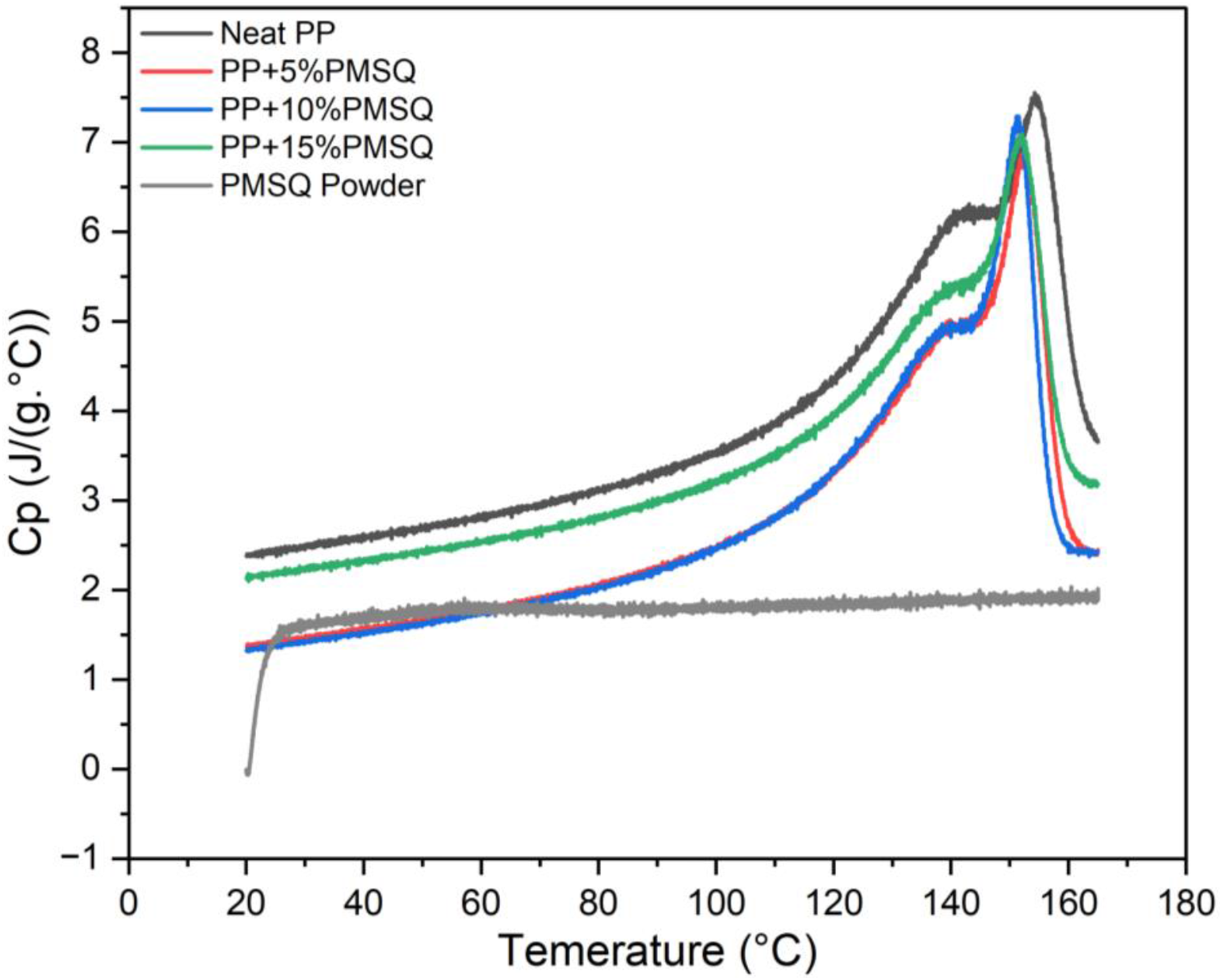
The results of the thermogravimetric analysis carried out on PP and its composites are reported in Figure 10, along with their first DTG curves. Moreover, the main degradation characteristics are collected in Table 1. Figure 10(a) shows that both the matrix and composites exhibit nearly the same degradation onset temperature around 439°C. Additionally, the DTG of PP and its composites (Figure 10(b)) reveal a peak around 463°C, which is attributed to the breaking of C-C bonds along the main chains,
57
and this peak corresponds to the maximum degradation temperature. This indicates that the addition of PMSQ does not have a significant effect on the maximum degradation temperature of PP. However, an increase in the residual mass is associated with the PMSQ content. For the neat PP, the residual mass is 0.5% (char), while it increases to 4.98%, 8.56%, and 12.28% for the composites with 5, 10, and 15% PMSQ, respectively (Table 1). This linear increase in residual mass is directly related with PMSQ residues which can act as a gas barrier (Figure 10(c)), thereby enhancing the thermal stability of PP.
57
Improved thermal stability with PMSQ addition or other types of fillers was reported in the literature.40,58,59 (a) TGA curves of the neat PP and its composites, (b) DTG curves of the neat PP and its composites, (c) Residual mass curve as a function of PMSQ content.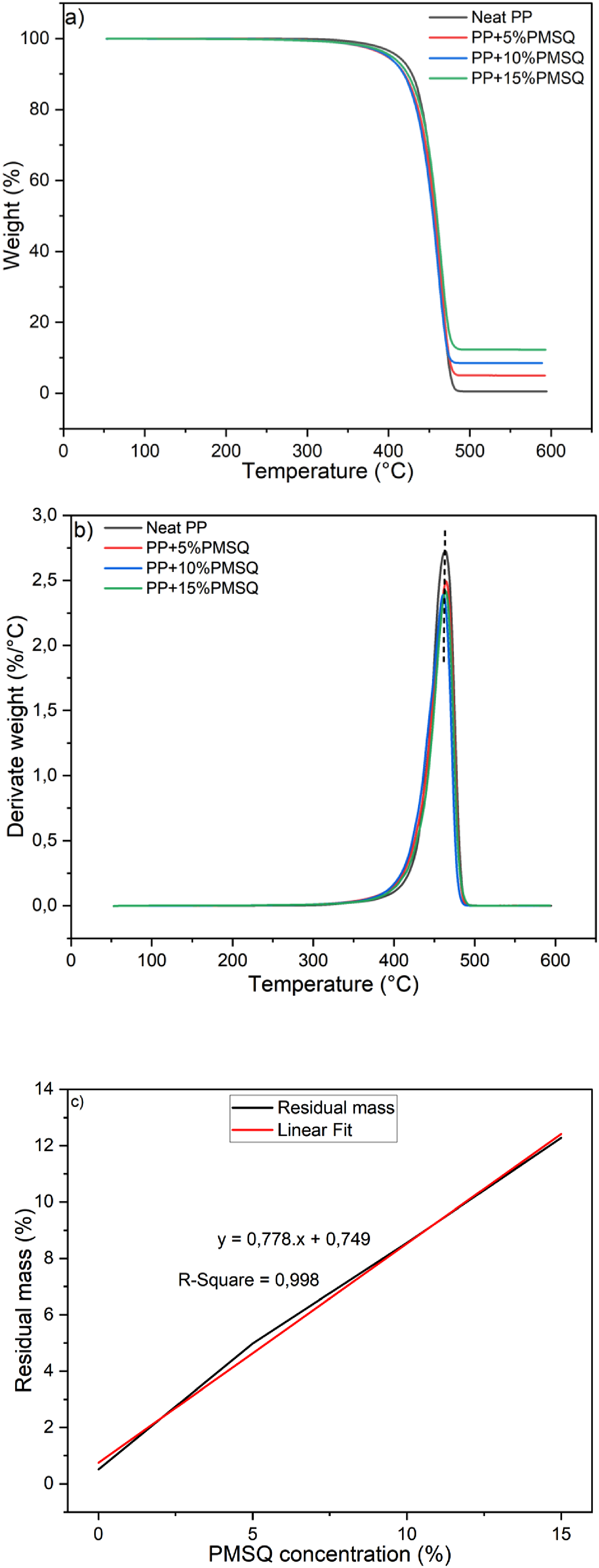
Dynamic mechanical analysis
Figure 11 presents the storage modulus, loss modulus, and tan δ of the matrix (neat PP). Across the α transition, the storage modulus (E′) gradually decreases with increasing temperature significantly dropping (58% reduction) from 2.7 GPa in the glassy region to 1.12 GPa in the rubbery region. This transition corresponds to higher mobility of the polymer chains in the amorphous part of PP, which occurs between −25°C and 40°C resulting in a marked decrease in the storage modulus (E′).
60
During this transition, the loss modulus (E″) increases and reaches its maximum at 0.13 GPa, corresponding to a temperature of 10°C, before decreasing again. The loss factor (tan δ) reaches its maximum at 15°C, which is associated to Tα or the glass transition temperature (Tg).
61
In the rubbery region, the storage modulus continues to decrease. In contrast, the loss modulus (E″) decreases while revealing a Tβ peak (between 50 and 100°C), corresponding to chain relaxation in the crystalline portion of PP or slippage between the crystals. DMA results (E′, E″ and tan(δ)) of the neat PP.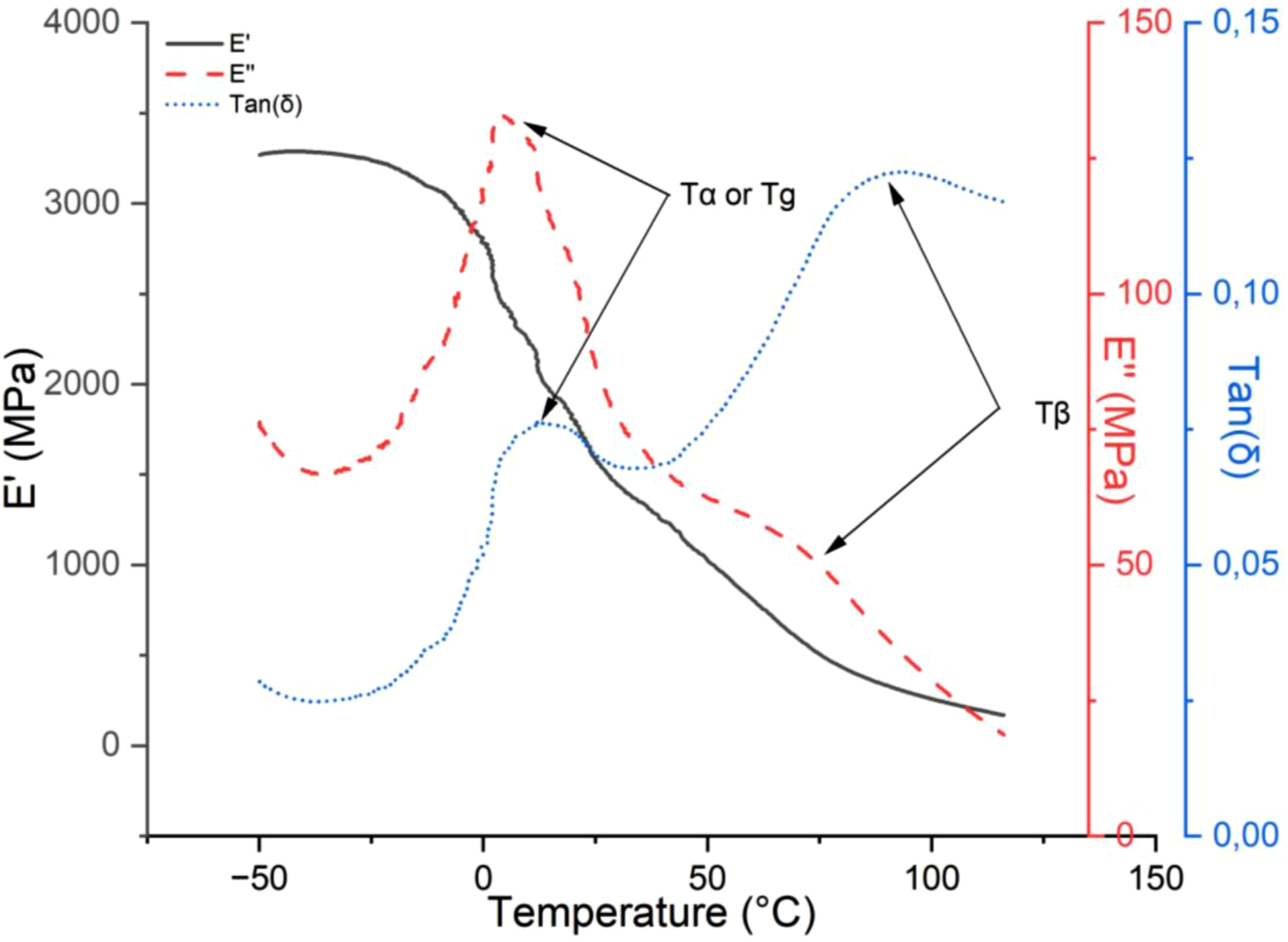
The dynamic mechanical properties of the PP and its composites are presented in Figure 12 and Table 2 for comparison. Since DMA is a very suitable tool to investigate the viscoelastic properties of materials over a wide range of temperature, each parameter is analyzed next. (a) Storage moduli (E′), (b) loss modulus (E″), and (c) loss tangent (tan(δ)) curves for the neat PP and its composites. DMA results of the neat PP and its composites.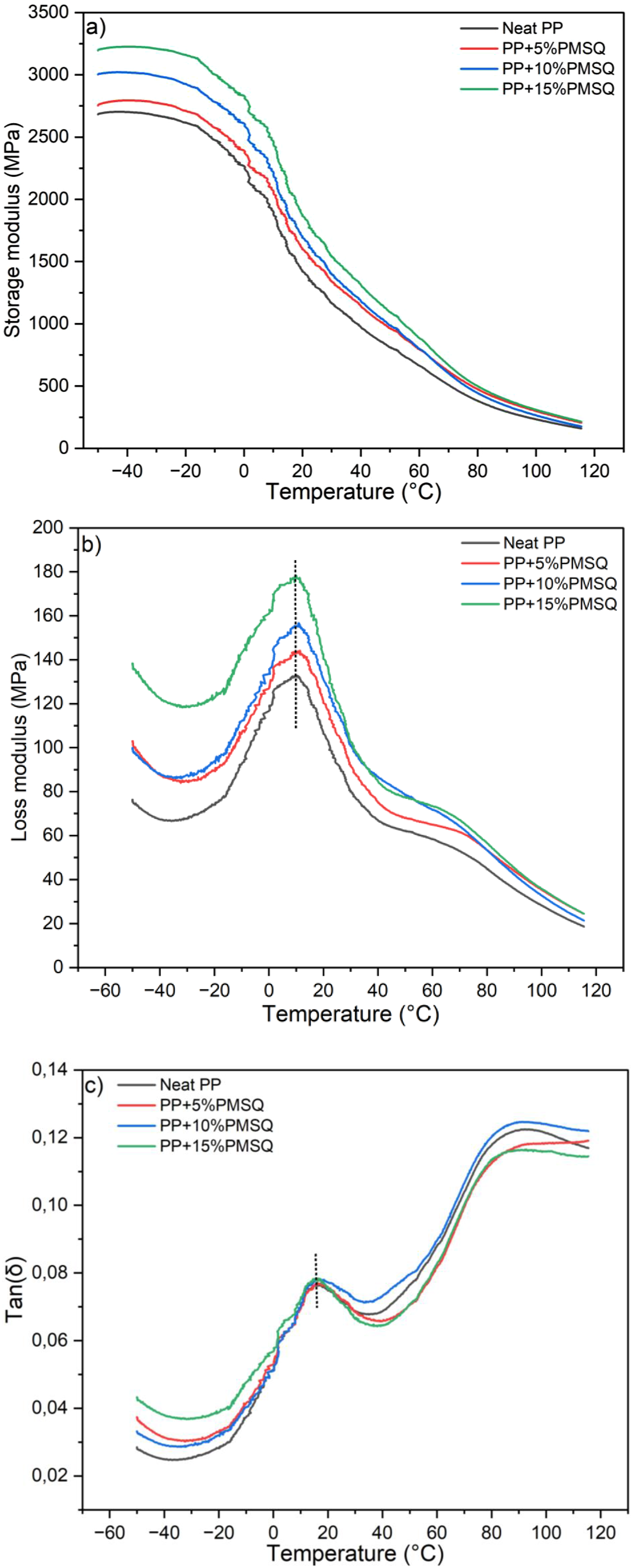
Storage modulus (E′)
Figure 12(a) shows the variation of the storage modulus as a function of temperature for the neat PP and its composites. It is clear that E′ decreases throughout the entire temperature range. A significant drop in E′ is observed between −25°C and 40°C, which can be attributed to the glass transition region of the matrix. This region is also characterized by an increase in chain mobility and free volume, 62 indicating that the matrix softens as temperature increases. 63 However, as the temperature further increases and approaches the melting point, the storage modulus of the composites sharply decreases, tending towards that of the neat matrix. This indicates that the behavior of the storage moduli of the composites is mainly influenced by the matrix.64,65 In the rubbery region, the rate of decrease for the composites is lower than that of the unfilled matrix. Similar results have been reported by Seshweni et al. by adding silica particles to PP. 66
The incorporation of reinforcing agents tends to enhance the stiffness of PP. This improvement is generally attributed to better interfacial adhesion. Strong interfacial bonding facilitates effective stress transfer between the filler and the polymer matrix, thereby improving the storage modulus. 67 In this study, the addition of PMSQ microparticles leads to an increase in the storage modulus with increasing PMSQ content. This increase can be attributed to good interfacial bonding between the filler and the matrix, as well as the strong restrictions on matrix chains induced by the addition of PMSQ. 66 Similar results were observed by Mohanty et al., where fillers played a stress-transfer role, leading to an increase in the storage modulus.40,68,69
In a semi-crystalline matrix, such as PP, chain movements only occur in the amorphous phase across the α transition. However, the crystalline part remains intact until the melting temperature is reached. 70
Loss modulus (E″)
The loss modulus represents the maximum amount of energy dissipated by the composites during deformation. It reflects the viscous properties of the materials, which are influenced by the mobility of polymer molecules inside the composites. 71 The variation of the loss modulus (E″) as a function of temperature is shown in Figure 12(b). Most thermoplastic matrices exhibit two peaks (Tα and Tβ) at different temperatures. 72 It can be observed that the neat PP and its composites exhibit the same viscoelastic behavior and display both transitions. The α transition, around 10°C, corresponds to the mobility of the amorphous phase chains and is associated with the glass transition temperature (Tg) of the matrix. In contrast, the β transition, between 50°C and 90°C, is attributed to the relaxation of the crystalline phase chains of PP in the rubbery plateau. Here, the Tg of the composites is almost identical to that of the neat PP. This indicates that the addition of PMSQ microparticles does not significantly change the relaxation behavior of PP.73,74 However, an increase in the loss modulus peak is observed with PMSQ particles addition. This result can be related to improved thermal stability and more restriction imposed on the polymer chain close to the particles surface.40,63 Consequently, this leads to lower flexibility of the composites. 68 On the other hand, increasing E″ with filler content can be attributed to higher energy dissipation during viscoelastic deformation. 74
Loss factor or tan(δ)
The ratio between E″ and E′ is defined as tan(δ) or the loss/damping factor. This parameter reflects a balance between the elastic and viscous response in a material. 52
Figure 12(c) shows the variation of tan(δ) as a function of temperature for the neat PP and its composites. Again, a similar behavior is observed for all samples. The peak (maximum value of tan δ) in the glassy region generally represents the glass transition temperature (Tg), which is around 15°C. This value does not change between the matrix and the composites indicating that under load, the behavior of the composites is not modified compared to the neat matrix. Additionally, a polymer matrix and its corresponding composite may exhibit similar glass transition temperatures when their interactions are weak or negligible. 75 Seki et al. obtained similar results by adding graphene into PP. 76 However, the magnitude of the damping peaks in the composites increases with the PMSQ concentration compared to the neat matrix. In other words, the energy dissipation increases with the reinforcement content. Similar results were reported by Seshweni et al. 66 Generally, good dispersion of the particles (reinforcement) leads to higher energy dissipation. 40 Finally, the transition around 90°C is attributed to the relaxation of the crystalline phase. 61 At this temperature, an increase in the intensity of the tan(δ) peak is observed, especially for PP+10PMSQ, indicating enhanced damping behavior in the rubbery plateau region. 77 In contrast, the addition of 5% and 15% PMSQ leads to a reduction of the peak intensity, suggesting decreased damping capacity. This result can be attributed to insufficient PMSQ content at 5 wt% to establish effective matrix-filler interactions, and to filler higher level at 15 wt% hindering the mobility of polymer chains.
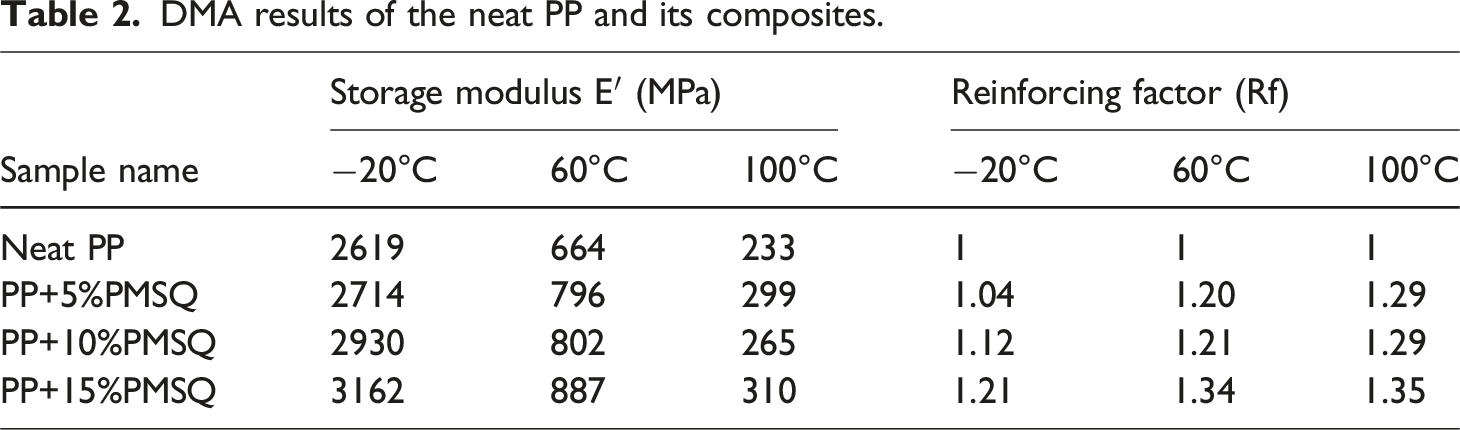
The effect of PMSQ microparticles on the PP matrix can be further quantified using the normalized storage modulus. This parameter, also called the reinforcing factor (Rf), is the ratio between the composite storage modulus to that of the matrix at a same temperature and the results are presented in Table 2. It can be seen that Rf increases with both PMSQ content and temperature due to the reinforcing and stabilizing effect of the fillers. 73 In other words, according to Aurilia et al., this result enables an extension of the processability temperature range of PP. 78
Thermal conductivity
Figure 13 presents the thermal conductivity of the neat PP and its composites over the temperature range of 23°C to 100°C. For all samples, the thermal conductivity increases with temperature. This increase is proportional to the PMSQ content, with the most significant effect observed at 10%. In particular, the thermal conductivity of the neat PP increases from 0.174 to 0.187 W/(m.K) between 50°C and 75°C, which corresponds to a 7% rise. In contrast, the composite with 10% PMSQ shows a substantial (20%) increase, rising from 0.183 to 0.220 W/(m.K) over the same temperature range. This outcome could be related to the improved crystallinity resulting from the addition of 10 wt% PMSQ, as evidenced in DSC data (Table 1).
79
According to Barkhad et al., the incorporation of fillers in thermoplastic matrices can increase its crystallinity, which in turn improves thermal conductivity.
80
However, as reported by Dawson et al., the intrinsic thermal conductivity of PP shows minimal sensitivity to temperature below 100°C. Therefore, the observed increase in conductivity is mainly due to the effect of PMSQ.
81
Thermal conductivity of the neat PP and its composites.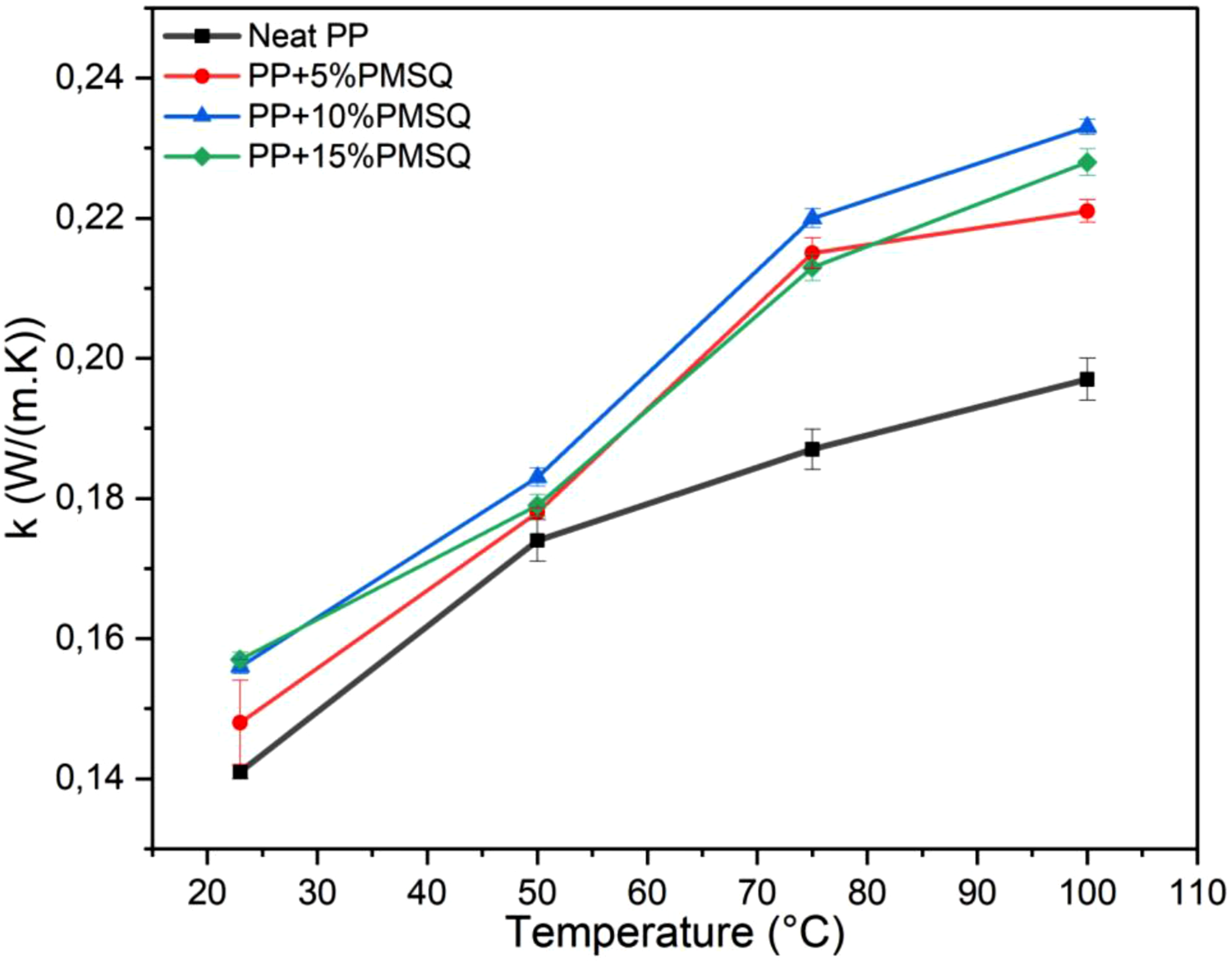
The addition of 15 wt% PMSQ leads to a decrease in thermal conductivity compared to the composite containing 10 wt% PMSQ. This reduction may be attributed to a decrease in crystallinity, as showed in DSC measurements (Table 1), likely caused by a more disordered structure resulting from the higher PMSQ content. A higher concentration of filler can hinder the orientation of polymer chains, thereby reducing heat transfer efficiency. It is well documented in the literature that thermal conductivity is strongly influenced by both the degree of crystallinity,82,83 and the orientation of molecular chains.84,85 Nevertheless, the conductivity increase becomes more pronounced above 50°C due to higher polymer chain mobility (Figure 13).
Electrical resistivity
In this study, only the electrical resistivity of the neat polypropylene and the composite with 10% PMSQ (optimum content) are presented. A more comprehensive analysis will be addressed in a forthcoming article.
Figure 14 presents the electrical resistivity of PP and PP+10% PMSQ. This parameter is inversely proportional to the electrical conductivity, meaning that when resistivity increases, conductivity decreases and vice versa.
86
An order of magnitude increase in resistivity (10x) is observed for PP after the addition of 10% PMSQ. Additionally, it is noteworthy that the electrical resistivity of neat polypropylene remains almost constant over the measured time interval. In contrast, the incorporation of PMSQ results in a significant increase in electrical resistivity over time. This increase can be attributed to the insulating properties of the PMSQ particles. Indeed, PMSQ exhibits characteristics similar to those of silica (SiO2), which has a very low electrical conductivity of 10−17 S/cm.
87
The incorporation of this type of reinforcement into a thermoplastic matrix can contribute to increase the thermal conductivity of the composite and to improve/maintaining its insulating properties. Similar results were reported for the incorporation of aluminium nitride (AlN) and boron nitride (BN) particles at 10% into PA6 resulting in a composite with a 36% higher thermal conductivity. However, the electrical conductivity remained low (72.1 × 10−9 S/m), which is only 3% higher than the neat matrix. This slight variation indicates that the electrical insulation properties were maintained and the authors attributed this result to the insulating properties of the filler used.
88
Electrical resistivity of the neat PP and PP+10% PMSQ.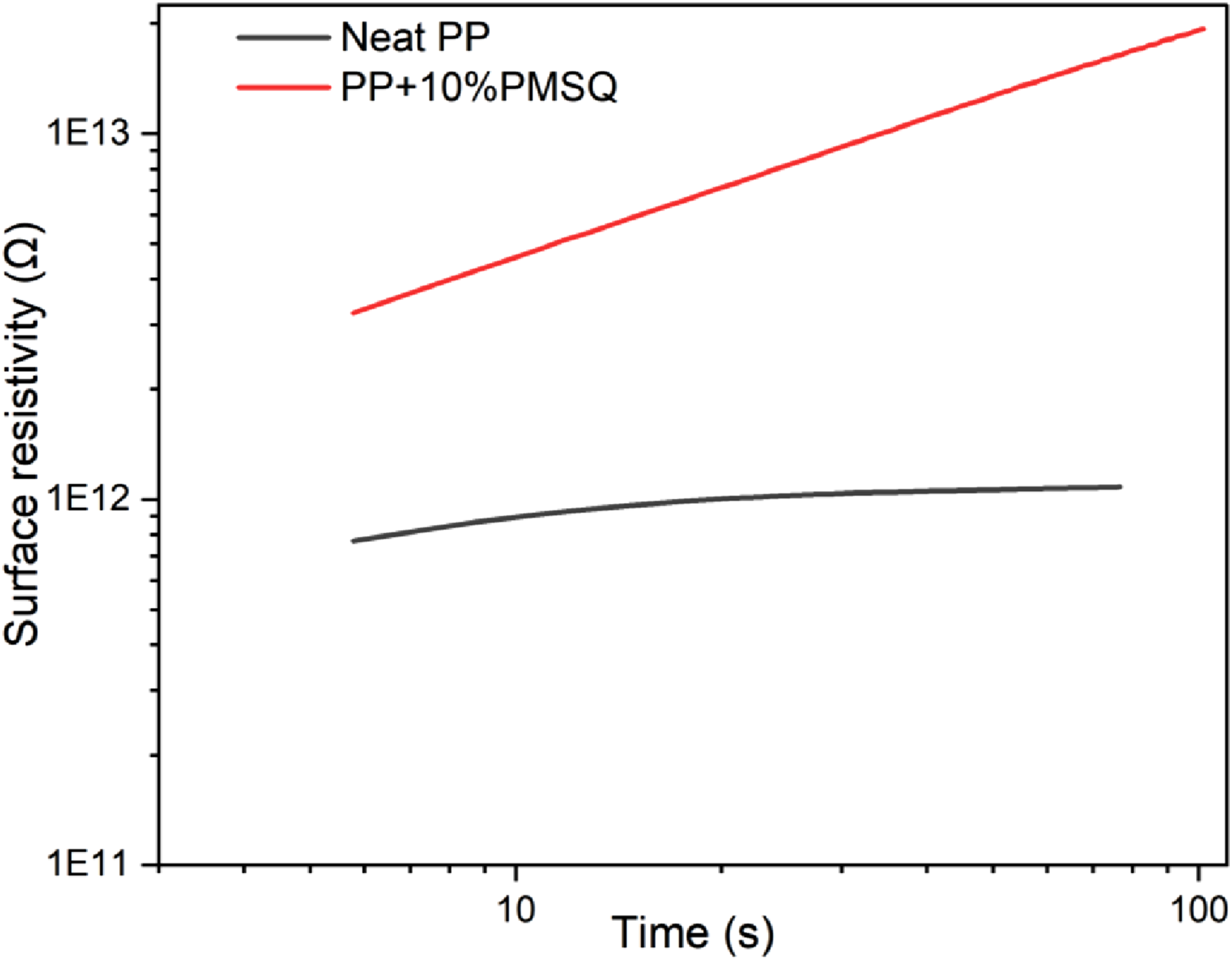
Conclusion
This study examined the effect of incorporating polymethylsilsesquioxane (PMSQ) microparticles into polypropylene (PP) on its thermal, thermo-mechanical, and electrical properties. The composites, prepared by melt blending with PMSQ contents ranging from 0 to 15 wt%, showed several notable trends. DSC analysis revealed a slight reduction in melting temperature without significant changes in crystallinity. TGA results indicated that thermal stability (Tonset and Tmax) remained largely unchanged. DMA showed a clear improvement in stiffness, with a 32% increase in storage modulus at 15 wt% PMSQ, while the glass transition temperature remained stable. Thermal conductivity increased by 20% with 10 wt% PMSQ, although higher loading (15%) led to a decrease likely due to lower crystallinity and particles agglomeration. Electrical resistivity measurements confirmed that PP’s insulating properties were preserved across all formulations.
These results demonstrated that PMSQ microparticles can significantly enhance the thermal performance of PP without compromising its electrical insulation, making the composites well-suited for real-world applications in electronics, energy storage systems, and electric vehicles, where both heat dissipation and electrical insulation are essential.
Heat dissipation and thermal management in devices such as electronic components, energy storage systems, automotive and aerospace industries require both thermally conductive and electrically insulating materials. This study has successfully developed a composite material that is both thermally conductive and electrically insulating. However, efforts are needed to further increase the thermal conductivity of this material to reach the required value (1 W/(m.K)) for effective thermal management in these applications. In addition, it is important to note that the increase in thermal conductivity obtained in this study is significantly lower than that reported using thermally conductive fillers.
Footnotes
Acknowledgements
The authors would like to thank the Natural Sciences and Engineering Research Council of Canada (RGPIN-2016-05689) for its financial support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This was supported by Natural Sciences and Engineering Research Council of Canada (RGPIN-2016-05689).