Abstract
The major purpose of this work is to fabricate electrochemical glucose sensor by employing three electrodes of polydopamine (PDA), PDA-Ag nanoparticles (NPs) and PDA-Ag-CuO nanocomposites. Ag NPs were prepared using PDA as a reducing and stabilizing agent. The fabricated electrodes were evaluated in a phosphate buffer solution working as the electrolyte, spiked with varying concentrations of glucose ranged from 4 to 40 mM using cyclic voltammetry (CV), differential pulse voltammetry (DPV), and electrochemical impedance spectroscopy (EIS). It was noted that Ag NPs and CuO NPs have face centered cubic and orthorhombic structures, respectively. Morphological studies displayed cauliflower shape for PDA, nanoluster spheres for Ag NPs and nanoneedles for CuO NPs ranged from 10 to 35 nm and 30 to 70 nm, respectively. The PDA-Ag NPs nanocomposite electrode exhibited a sensitivity of 1.0 × 10−5 A·mM−1, a correlation coefficient (R2) of 96.5%, a limit of detection (LOD) of 1.73 mM, and a limit of quantification (LOQ) of 5.77 mM. It also demonstrated a relative standard deviation (RSD) of less than 1.5% and a repeatability of 1.6 × 10−5.PDA-Ag-CuO nanocomposite electrode achieved a sensitivity of 9.0 × 10−6 A·mM−1, with a strong linearity (R2 = 99.2%), a calculated detection limit (LOD) of 1.08 mM, and a quantification limit (LOQ) of 3.60 mM. The relative standard deviation (RSD) was calculated to be below 3%, with a repeatability value of 2.8 × 10−5. The interference with glucose by ascorbic acid, urea, cholic acid, and dopamine was assessed, and the results indicated that PDA-Ag NPs-CuO nanocomposite electrode has a high selectivity to glucose.
Introduction
Diabetes is becoming the major health problem in the world. A statistical analysis by the World Health Organization indicates that more than 220 million people must live with diabetes. 1 Diabetes death is estimated to 2.2 by 2030, and patients will be doubled in the next twenty years. Kidney failure, heart disease, and blindness are kinds of diseases that caused by diabetes and the early diagnosis and treatment is necessary to patients. 2 Different techniques have been used to improve an invasive and/or noninvasive device for blood glucose monitoring, like optical spectroscopy, infrared spectroscopy, surface plasmon resonance, Raman spectroscopy and photoacoustic probes. The output results from these techniques should be double checked to direct blood glucose detection; therefore, the direct measure of a blood glucose sensor cannot be replaced by these techniques. Electrochemical sensors offer properties that urgent and important to develop easy, fast, stable, sensitive and highly precise device to detect the glucose. 3
Electrochemical sensors are typically classified into amperometric, potentiometric, electrochemical impedance, and photoelectrochemical types. Emerging developments in nanoscience are paving the way for highly sensitive and selective nanomaterial-based electrochemical sensors. 4 Moreover, functional nanoparticles and polymers play a significant role in enhancing conductivity, catalytic activity, and biocompatibility, thereby accelerating signal transduction and enabling the development of highly sensitive and selective sensors.. 5
Polydopamine (PDA) is a semi-synthetic adhesive polymer formed through the self-polymerization of dopamine (DA), a catecholamine neurotransmitter containing catechol moieties. In alkaline conditions, DA undergoes oxidation followed by spontaneous polymerization, resulting in the formation of PDA. PDA exhibits excellent adhesion properties to a wide range of substrates, along with remarkable biocompatibility, hydrophobicity, and chemical reactivity. 6 Additionally, PDA demonstrates strong reducing capabilities, enabling the in situ reduction of metal salts into metallic nanostructures. These properties make PDA a versatile platform for surface functionalization, biosensing, drug delivery, and catalytic applications.7–9
Ag and CuO NPs have unique properties which includes physical and chemical properties, electrocatalytic activity, antimicrobial, antibacterial, excessive conductivity, thermal stability, non-toxic, biocompatibility, cost effective are broadly used in glucose detection10–16
In the previous research Ag-CuO/rGO (reduced graphene oxide) composite was prepared and fabricated as a nonenzymatic glucose sensor by calcination after the hydrothermal process. Ag and CuO were synthesized
The novelty of this work lies in the development of a glucose sensor using a PDA-Ag-CuO nanocomposite electrode, which combines the unique properties of polydopamine (PDA), silver nanoparticles (Ag NPs), and copper oxide nanoparticles (CuO NPs) to enhance sensor performance. This work is distinguished by its thorough evaluation of the sensor’s performance using cyclic voltammetry (CV), differential pulse voltammetry (DPV), and electrochemical impedance spectroscopy (EIS) in the presence of phosphate buffer as the electrolyte. Furthermore, the study provides a detailed assessment of the sensor’s limit of detection, sensitivity, and selectivity in the presence of various interfering bioanalytes, offering valuable insights into the nanocomposite’s practical application for glucose sensing in complex biological systems.
Materials and methods
Materials
Dopamine hydrochloride was purchased from Sunny Pharmaceutical Company and ammonium hydroxide (25%) was supplied by Sigma. Silver nitrate (99.8%) was supplied from Merk. D-glucose (99.5%) was obtained from Analar, sodium dihydrogen orthophosphate (98%) was purchased from Loba Chemie and disodium hydrogen phosphate dehydrate (98%) was received from Piochem. Copper sulfate pentahydrate (99%) was received from Fisher Scientific and ammonium per sulfate (98%) was supplied from BDH Company. Sodium hydroxide (99.9%) from Carlo Erba, urea (98%) and cholic acid (99%) were purchased from ACROS Company while ascorbic acid tablet was obtained from local pharmacy.
Synthesis and fabrication of PDA, PDA-Ag NPs and PDA-Ag-CuO nanocomposite electrodes
PDA was prepared by adding 25 mL (40 mg/mL) of dopamine hydrochloride to 500 mL water and then ammonium solution was added dropwise to attain pH 8.5 to obtain 2 mg/mL dopamine hydrochloride. This solution was stirred for 24 h at 60οC. The color of aqueous solution was changed to black-brown due to self-polymerization of dopamine. The formed PDA cast on graphite sheet (GE) to produce homogeneous film electrode. PDA-Ag NPs were prepared by dissolving AgNO3 (0.16 g) in 20 mL of PDA as a reducing and stabilizing agent under stirring for 6 h. CuO NPs were prepared by dissolving 10 g of CuSO4.5H2O in 100 mL of distilled water with vigorous stirring until completely dissolution. The product solution was treated with 0.5 g of NaOH and 1.0 g of ammonium persulphate with stirring at 100 °C for 4.0 h. The black precipitate was formed immediately. It was centrifuged and washed 3–4 times with distilled water, then dried the precipitate in oven at 100°C for 24 h.
PDA-Ag-CuO nanocomposite was prepared by physical mixing of PDA-Ag NPs and CuO NPs with 1:1 wt % and then stirred for 30 min. The nanocomposite electrodes were prepared by drop cast on 1 × 1 cm2 graphite sheet then the electrodes were dried in the oven to dry at 60°C for 2 h.
Characterization techniques and measurements
The identification of functional groups, analysis of bonding interactions, and confirmation of chemical group modifications in PDA, PDA-Ag, and PDA-Ag-CuO nanocomposites were carried out using Fourier transform infrared (FTIR) spectroscopy (Spectrum BX 11 Infrared spectrometer FTIR LX 18-5255 Perkin Elmer). The prepared materials were grinded with KBr powder at ratio 1:10 and pressed in a hydraulic press to form translucent pellets. The spectra were detected at the wavenumber range of 4000–400 cm−1. Optical properties of PDA were investigated by Ultraviolet-Visible (UV-VIS) spectroscopy (Evolution 300 double beam scanning UV-visible spectrophotometer, Thermo scientific, USA) in the range of 200–800 nm with Deuterium lamp. The crystal structure of PDA, PDA-Ag NPs and PDA-Ag-CuO nanocomposite were determined by X-ray diffractometer (XRD-7000, Empyrean diffract meter/Analytical) with CuKα radiation powder operating at 40 kV and 30 mA with a scan rate of 10°/min over a range of 2θ between 10° and 90°. The microstructure of PDA, PDA-Ag NPs and PDA-Ag -CuO NPs were investigated using high resolution transmission electron microscope HRTEM (JEOL JEM 2100 F-Japan) equipped with field emission gun (FEG) at an accelerating voltage of 200 kV used for analysis of samples. The morphology of different prepared films was investigated with scanning electron microscopy (SEM “JEOL JSM-6360 LA”, Japan) to study the surface of PDA, PDA/Ag and nanocomposites cast on graphite sheet. Thin films of gold were sputtered onto the samples to get charge free surfaces. Moreover, EDX was used to examine the elemental analysis of the nanocomposite electrodes.
Detection of glucose
The glucose detection could be applied by PDA-Ag NPs and PDA-Ag-CuO nanocomposite cast on graphite sheet electrodes which measured at different glucose concentrations from 4 to 40 mM in PBS with 1 mM of K4[Fe(CN)6]·3H2O. Potassium ferricyanide in PBS is used as a supporting electrolyte, as it enhances the electroactivity of the PDA-Ag-CuO nanocomposite electrode, and PBS maintains a stable pH because of its buffering capacity. The prepared electrodes were measured by CV, DPV and EIS techniques using AUTOLAB controlled Potentiostat/Galvanostat (PGSTAE204 Metrohm, Netherlands) driven with NOVA software. Ag/AgCl electrode, modified graphite sheet and a platinum wire had been used as reference electrode, working electrode and counter electrode, respectively.
Results and discussion
The absorption spectrum of PDA is presented in Figure 1(a). The characteristic absorption sharp peak at 280 nm is assign to π−π* electronic transition of catechol.
20
To confirm the chemical structure and functional group of PDA, PDA-Ag NPs and PDA-Ag-CuO nanocomposite, FTIR spectra are carried out and the binding interactions between PDA, Ag NPs and CuO NPs are characterized as illustrated in Figure 1(b). The broad peak at υ 3563 cm−1 is assigned to the stretching vibration of hydroxyl and N-H bonds in the catechol groups of PDA. The observed peaks at υ 1637 cm−1 and υ 1523 cm−1 are attributed to C=O stretching and C=C stretching of indole groups, respectively. The stretching absorption peaks of C-O and C-N have appeared at υ 1289 cm−1 and υ 1111 cm−1, respectively. The observed peak at υ 1403 cm−1 is referred to C-H starching.
21
On the other hand, the absorption peak of PDA at υ 3563 cm−1 is shifted to lower frequency at υ 3478 cm−1 for PDA-Ag NPs. This result is due to the formation of chemical bond between Ag NPs and oxygen of catechol group and hydroxyl group which oxidized to quinone group or carbonyl group, while Ag+ is reduced to Ag0.22,23 Moreover, the absorption peak of PDA at υ 3563 cm−1 is shifted to lower frequency at υ 3407 cm−1 for PDA-Ag-CuO nanocomposite due to the electrostatic interaction of CuO NPs with oxygen of catechol groups. The appearance peak at υ 620 cm−1 confirms the stretching of CuO.24,25 (a) UV–vis absorption spectrum of PDA; (b) FTIR spectra of PDA, PDA–Ag NPs, and PDA–Ag–CuO nanocomposite; (c) XRD patterns of PDA, PDA–Ag NPs, and PDA–Ag–CuO nanocomposite.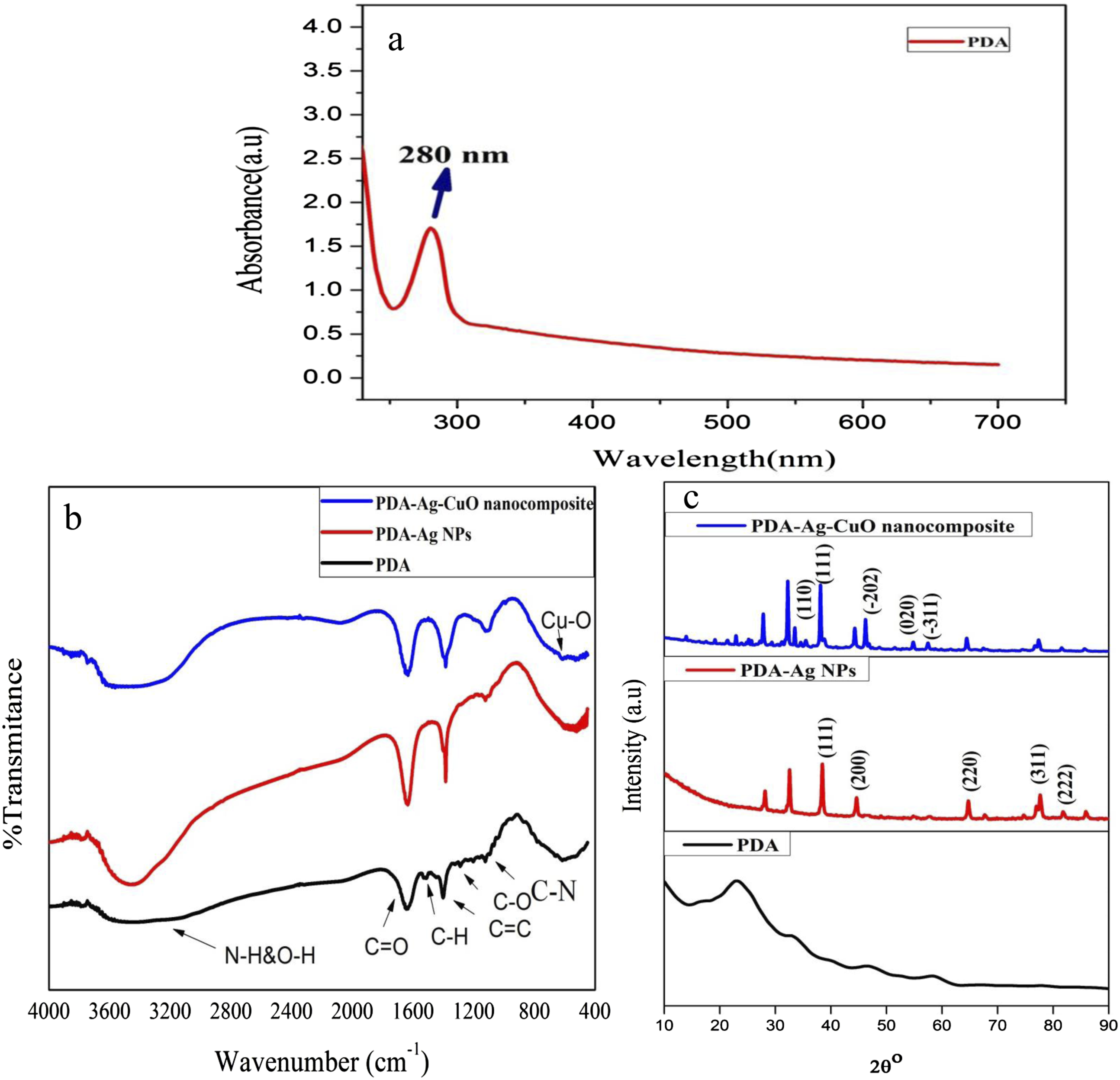
XRD patterns of PDA, PDA-Ag NPs and PDA-Ag-CuO nanocomposite are presented in Figure 1(c). It is noted that the broad diffraction peak observed at 23.3° can be ascribed to the amorphous nature of PDA structure. The peaks at 38.9°, 44.66°, 64.88°, 77.92° and 82.12° for PDA-Ag NPs are corresponded to (111), (200), (220), (311) and (222) crystalline planes, respectively of face centered cubic of Ag NPs. The shift in 2θ o compared with Ag metal is due to the reduction of Ag+ to Ago by PDA and the interaction of Ag NPs with catechol group of PDA. 23 Moreover, the main characteristic diffraction peaks of CuO NPs at 32.6°, 38.1°, 46.2°, 54.7° and 57.5° are assigned to (110), (111), (−202), (020) and (−311) of orthorhombic CuO. Therefore, it is noticed that the sharp peak of PDA existed in the nanocomposites and this is evidence on the presence of physical or chemical interaction between PDA and Ag NPs or PDA and CuO NPs. The chemical bond from the interaction between Ag NPs and CuO NPs with the oxygen of hydroxyl group of catechol group on the surface of PDA group by physical mixing of PDA-Ag-CuO nanocomposite can be occurred. 26
The proposed mechanism of PDA polymerization and reduction of Ag ions is represented in Scheme 1. The proposed mechanism of dopamine polymerization is considered by self-assembly or covalent oxidative polymerization. Firstly, the catechol group is oxidized to form dopamine quinone in alkaline aqueous medium condition. Subsequently, the dopamine quinone is cyclized by intramolecular Michael addition mechanism to form leucodopamine. Schematic diagram of polymerization mechanism of dopamine and the interaction of PDA with Ag NPs and CuO NPs.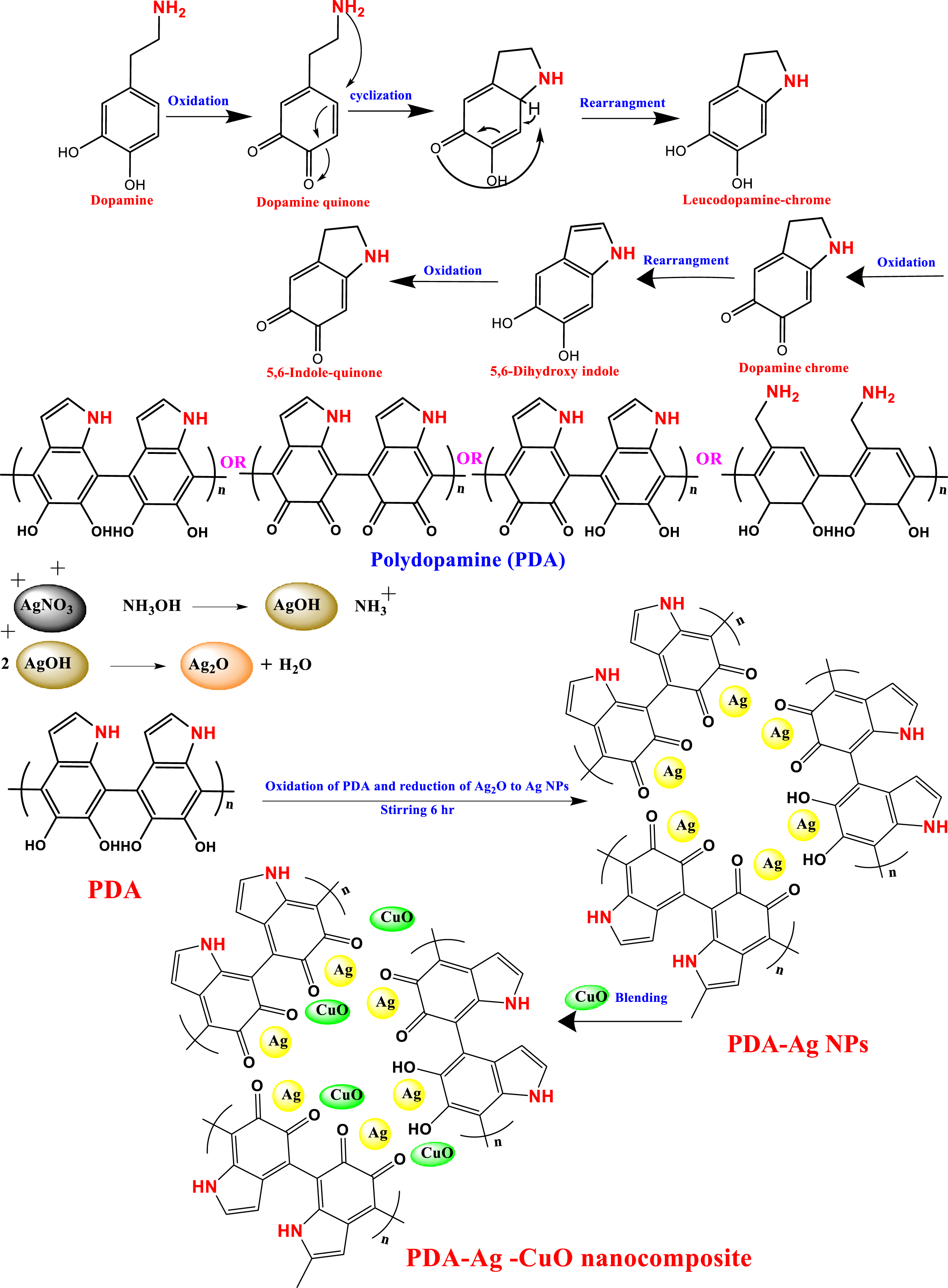
After that the leucodopamine is oxidized to form dopamine chrome and then reduced to produce the indole form 5,6-dihydroxyindole and then oxidized again to form 5,6-dihydroxyquinone, so the polymerization could be in different forms of indole or quinone.27,28 Polydopamine has the ability to reduce silver ions to Ag NPs by its phenolic hydroxyl group. Finally, PDA-Ag NPs is physically mixed with CuO NPs to form PDA-Ag-CuO nanocomposite as illustrated in Scheme 1. Electrostatic interactions between Ag NPs and CuO NPs with hydroxyl (O-H) and carbonyl (C=O) functional groups play a crucial role in enhancing the sensor’s performance. The presence of these functional groups facilitates attractive forces, leading to improved adhesion and stability of the nanocomposite. These interactions contribute to the efficient catalytic activity and sensitivity of the sensor in glucose detection
The morphological features of graphite sheet, PDA, PDA-Ag NPs and PDA-Ag-CuO nanocomposite cast onto graphite sheet electrode are displayed in Figure 2. SEM image of graphite sheet appears as irregular plates with smooth surface as illustrated in Figure 2(a). Figure 2(b) represents the formation of uniform spherical structure like cauliflower shape of PDA. Figure 2(c) exhibits small shiny spheres of Ag NPs that decorates the PDA surface with a diameter range from 10 to 35 nm.
29
Figure 2(d) shows the same uniform sphere of PDA which decorated by luster small spheres of Ag NPS and small rectangular shape (rods) of CuO NPs with a diameter from 30 to 70 nm.
30
The SEM images confirm that Ag NPs and CuO NPs are homogeneously dispersed on the surface of PDA without any aggregation. This is due to the steric stabilization of particles on the surface of PDA and the positively charged onto PDA could prevent the aggregation of NPs by improving mutual electrostatic repulsion. SEM images are (a) graphite sheet, (b) PDA, (c) PDA-Ag NPs and (d) PDA-Ag NPs -CuO NPs nanocomposite.
PDA-Ag NPs are investigated using TEM, SEAD and EDX as illustrated in Figure 3. Ag NPs are successfully committed and uniformly dispersed on PDA. The size of Ag NPs in the matrix of PDA is ranged from 10 to 20 nm with a spherical shape as shown in Figure 3(a) and (b). This observation confirms that there is no aggregation of Ag NPs on PDA surface, and the size range of Ag NPs is compatible to that measured from SEM images. Although the ultrasonication process of PDA-Ag NPs for 30 min before the imaging, Ag NPs are attached to the PDA and no free particle are found, which provides evidence that the bonding between Ag NPs and PDA is fixed and stable. This strong bond confirms that this nanostructure could not destroy.
31
SAED pattern of Ag NPs image shows concentric rings with intermittent dots, which indicates that these nanoparticles are crystalline as presented in Figure 3(c). The chemical composition of PDA-Ag NPs is examined by EDX spectrum and different kinds of peaks in the EDX spectrum, which corresponding to carbon, nitrogen, oxygen and silver elements. This approves the existence of Ag NPs on the surface of the PDA. The elemental atoms depicted in the insert table also confirm that the Ag particles are presented with the PDA matric as illustrated in Figure 3(d).
32
TEM (a and b) images of PDA-Ag NPs at different magnifications, (c)- SAED of Ag NPs and (d)- EDX of PDA-Ag NPs.
Figure 4 shows TEM images of PDA-Ag-CuO nanocomposite after physical mixing of PDA-Ag NPs and CuO NPs to obtain the nanocomposite for fabrication of the glucose electrode sensor. PDA-Ag-CuO nanocomposite composite has small uniform spherical shape for Ag NPs with a diameter from 10 to 20 nm and nanorods and nanoneedles of CuO NPs that has a diameter from 30 to 60 nm as presented in Figure 4(a).
33
This nanocomposite consists of a crystalline nature of Ag NPs and CuO NPs spread on the amorphous matrix of PDA that appears by studying the SAED of nanocomposite as shown in Figure 4(b).
34
EDX pattern as presents that the elements in this composite are carbon, nitrogen, oxygen, copper and silver as illustrated in the table that inserted in Figure 4(c).
35
Also, the bond between Ag NPs and CuO NPs with the catechol group on the surface of PDA is very strong after the sonication of the nanocomposite for 30 min as appeared in the figure and this proves the reacted components are highly attached together. HRTEM images of (a) PDA-Ag-CuO nanocomposite at different magnifications, (b)- SAED of nanocomposite and (c)- EDX of nanocomposite and (d)- EDX of PDA-Ag NPs.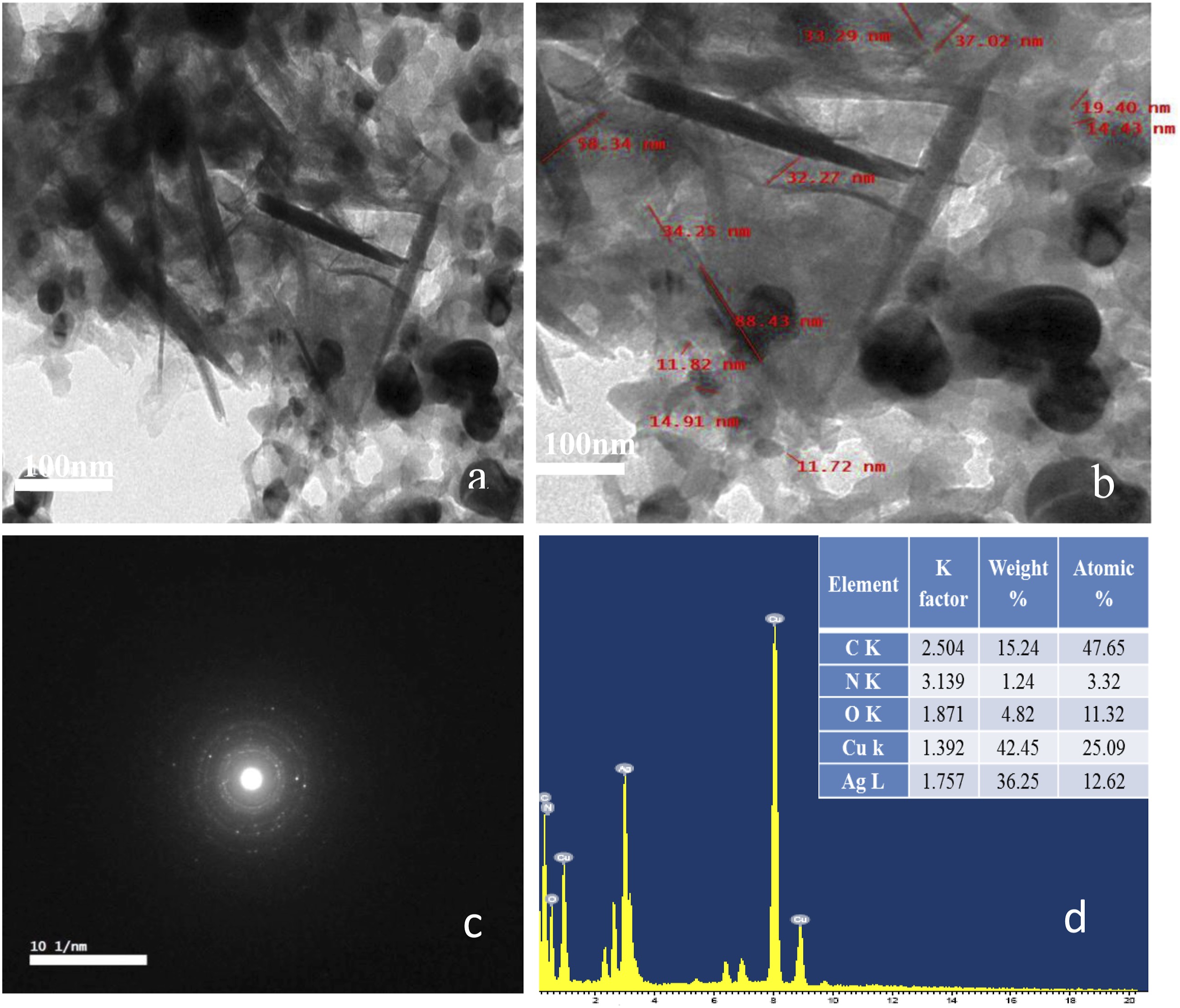
Electrochemical sensing of glucose
The oxidation and reduction peaks of different prepared electrodes of the graphite sheet, PDA-Ag NPs and PDA-Ag-CuO nanocomposite cast on graphite sheet in presence of PBS at pH 7 with 1 mM potassium ferricyanide and 10 mM glucose are studied by CV curves as illustrated in Figure 5. The potential window of CV is adjusted from −0.4 to 1 V. The oxidation peak current of pristine graphite sheet is 0.99 mA at 0.4 V, for PDA-Ag NPs electrode is 1 mA at 0.36 V and 4 mA at 0.78 V and for PDA-Ag-CuO nanocomposite is 4.8 mA at 0.6 V. While the reduction current peak is −1.1 mA at 0.23 V for pristine graphite sheet, 12 mA at 0.68 V and −5 mA at −0.02 V for PDA-Ag NPs electrode and 4.8 mA at 0.1 V for PDA-Ag-CuO nanocomposite . It is noticed that the oxidation and reduction current of the nanocomposite of PDA-Ag-CuO nanocomposite electrode has the highest oxidation current of 4.8 mA at 0.6 V and reduction current of 4.8 mA at 0.1 V compared to the other electrodes. The electrochemical reaction is irreversible due to glucose and its oxidation intermediates can adsorb onto the electrode surface, causing slower electron transfer due to blocking or fouling effects on the active sites of the Ag NPs and CuO NPs, further increasing ΔEp.
36
Therefore, this comparative study between the three electrodes by applying CV measurements has proved that the PDA/Ag/CuO NPs nanocomposite is the optimum electrode for `glucose sensor. CVs of different electrodes the pristine graphite sheet, nanocomposites of PDA-Ag NPs and PDA-Ag-CuO nanocomposite cast on graphite sheet in PBS of pH 7 with 10 mM glucose.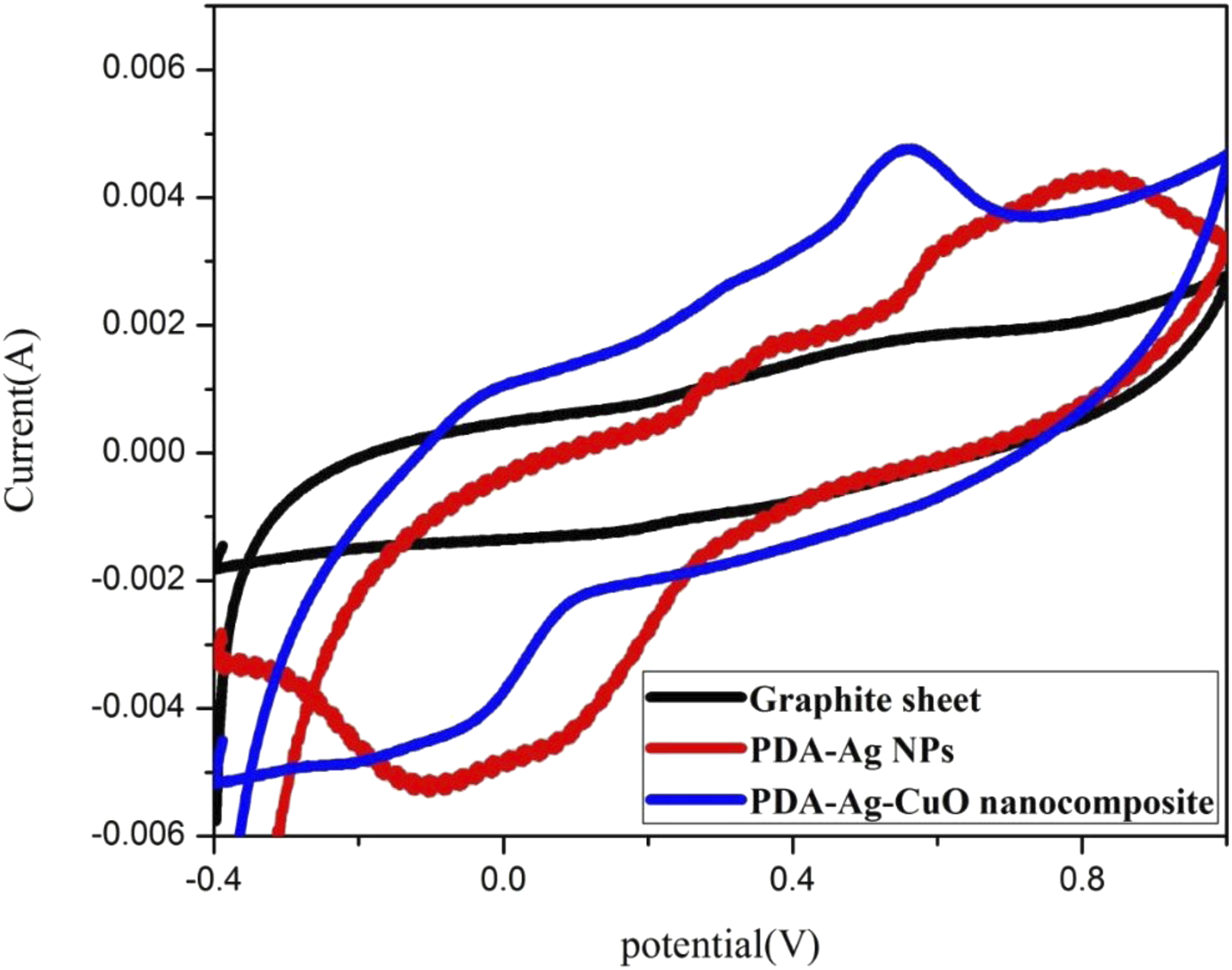
These results can be explained for PDA-Ag NPs electrode, there is an enhancement of current due to the oxidation reduction reaction of Ag NPs and glucose. Ag NPs is oxidized to Ag2O by losing electrons then Ag2O is oxidized to AgO by losing electrons. Finally, AgO chemically reacts with glucose to produce Ag2O and glucolactone according the following reactions as illustrated in Scheme 2.
37
And in case of PDA-Ag-CuO nanocomposite electrode, there are two redox reactions, where the first one has occurred resulted from the reaction between PDA-Ag NPs and glucose as above mentioned before. The other redox reaction is attributed to reaction between CuO NPs and glucose through three steps. CuO NPs is oxidized to Cu(OH)2 with losing electrons by hydroxyl group, then Cu(OH)2 is also oxidized to CuOOH by hydroxyl group in a reversible reaction and finally, CuOOH gains electron and is reduced to Cu(OH)2. Glucose is oxidized by losing electrons and converts to glucolactone as the following mechanism as shown in Figure 2.
37
Mechanism of detection of glucose by PDA-Ag NPs electrode and PDA-Ag-CuO nanocomposite.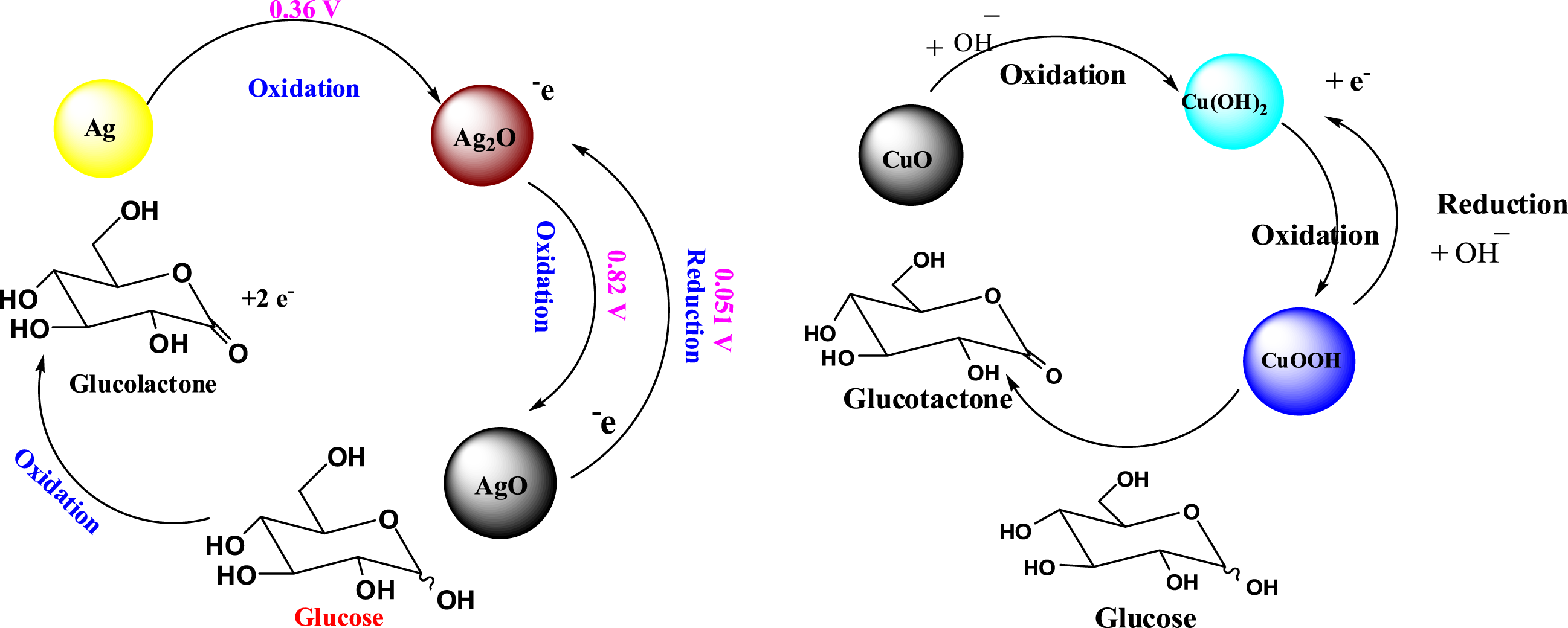
Nyquist curves of different electrodes of the pristine graphite sheet, nanocomposites of PDA-Ag NPs and PDA-Ag-CuO nanocomposite in PBS of pH 7 as electrolyte with 10 mM glucose are shown in Figure 6. In electrochemical systems, the pattern of Nyquist plots for faradaic impedance spectra over a wide range of frequencies usually involves both the semicircle (at this frequency region the electrochemical process is controlled by charge transfer phenomena) and the straight line (at this frequency region the electrochemical process is controlled by mass transfer phenomena). However, for graphite sheet, PDA-Ag NPs and PDA-Ag-CuO nanocomposite electrodes, the straight lines are only detected due to the mass transfer phenomenon. Graphite sheet, PDA-Ag NPs and PDA-Ag-CuO nanocomposite electrodes have series resistance of 8.5, 13.5 and 10.2 Ohm, respectively. It is noticed that PDA-Ag-CuO nanocomposite electrode has the lower resistance value than PDA-Ag NPs electrode. This is maybe Ag NPs and CuO NPs are found in the nanoscale as proved by SEM and TEM to enhance the active surface area and electroactivity, so the presence of CuO with Ag NPs enhances the current.38–40 Nyquist plot of graphite sheet, PDA-Ag NPs and PDA-Ag-CuO nanocomposite electrodes in PBS of pH 7 as electrolyte with 10 mM glucose.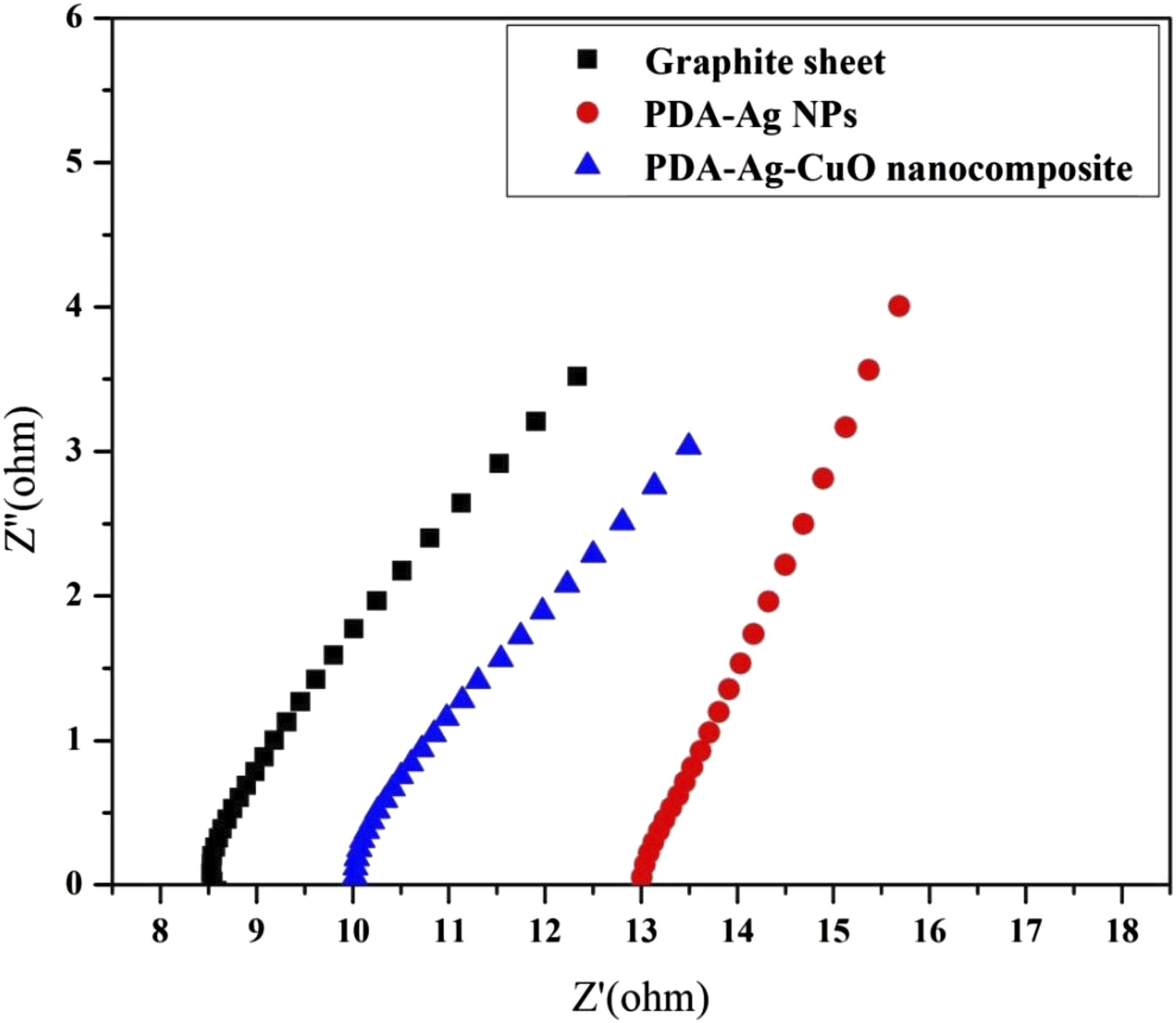
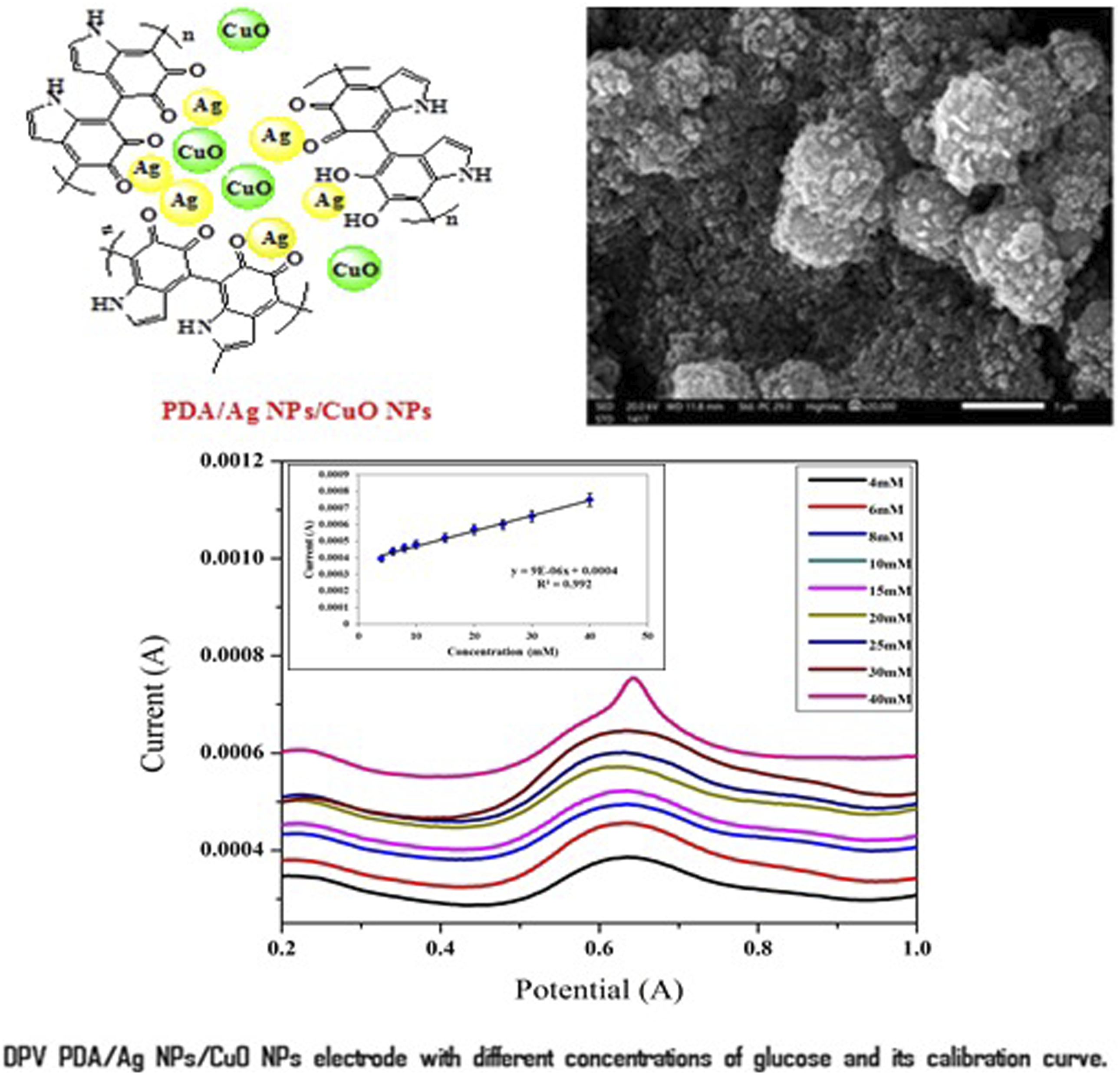
PDA-Ag NPs electrode is applied as a glucose sensor by measuring DPV at different glucose concentrations from 6 to 30 mM and the calibration curve is plotted from current peaks as illustrated in Figure 7. It is observed that high glucose concentration enhances the current peak due to redox reaction between glucose and Ag NPs according to the mechanism which previously mentioned in CV measurement. This redox reaction enhances the charge transfer between PDA-Ag NPs electrode and glucose and the current value could be increased.
41
It’s observed that there is peak shift with the change of glucose concentration due to the adsorption of the glucose onto the electrode surface, causing blocking the active sites of the Ag NPs and CuO NPs. From the calibration curve, it is found that PDA-Ag NPs electrode has R2 of 96 % with a sensitivity of 1 × 10−5A mM−1 cm−2; LOD and LOQ of the PDA-Ag NPs electrode for glucose detection were determined to be 1.73 mM and 5.77 mM, respectively, by applying the data to equations (1) and (2) presented below: DPV curves of PDA-Ag NPs electrode with different concentrations of glucose and its calibration curve.

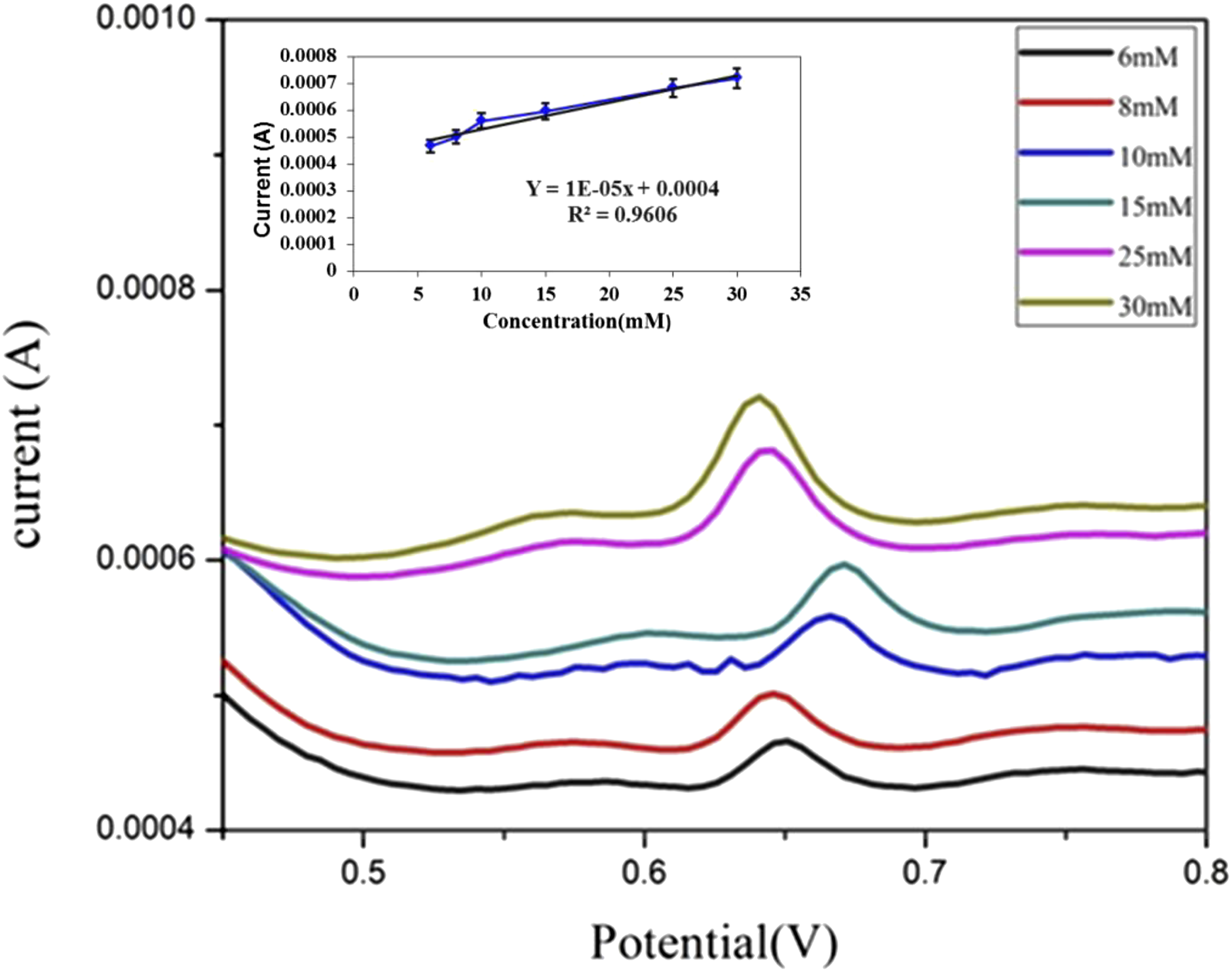
The precision limit is estimated by calculating the repeatability (r) according to the following equation (3)
43
:
It is noted that r of this electrode for 6 mM glucose is 1.6 × 10−5.
The relative standard deviation (RSD) values of PDA-Ag NPs electrode for different glucose concentrations are less than 1.5%. It can be concluded that the PDA-Ag NPs electrode has high precision limit. 42 Also as observed from the calibration curve, PDA-Ag NPs electrode detect glucose from 6 to 30 mM and this is not achieved the real exact range of glucose in all diabetics body and this electrode is not applicable to detect glucose.
DPV curves and the calibration curve of PDA-Ag-CuO nanocomposite electrode is investigated at different glucose concentrations in PBS of pH 7 and 1 mM potassium ferricyanide as exhibited in Figure 8. The current peaks values of different glucose concentrations are detected for PDA-Ag-CuO nanocomposite electrode ranged from 4 to 40 mM. The current peaks values are increased with the enhanced of glucose concentration owing to proposed mechanism of oxidation and reduction reaction of Ag NPs and CuO NPs. The oxidation reaction of glucose provides electrons that responsible of reaction and enhances the produced current.43–45 It is observed that PDA-Ag NPs electrode has R2 of 99.2% with a sensitivity of 9 × 10−6 μA mM−1, LOD of 1.08 mM and LOQ of 3.6 mM. The precision of PDA-Ag-CuO nanocomposite nanocomposite electrode are provided by measuring RSD % < 3% for different glucose concentrations. The repeatability is found to be less than 2.8 × 10−5 and this result validates the highly precision of this electrode.46,47 These results proves that nanocomposite of PDA-Ag-CuO nanocomposite cast on graphite sheet electrode in presence of PBS of pH 7 and 1 mM potassium ferricyanide has high sensitivity and precision for glucose detection, in addition to the ability of this electrode to cover a wide range of glucose concentration from 4 to 40 mM which is compatible with healthy and diabetics. DPV curves of PDA-Ag-CuO nanocomposite electrode with different concentrations of glucose and its calibration curve.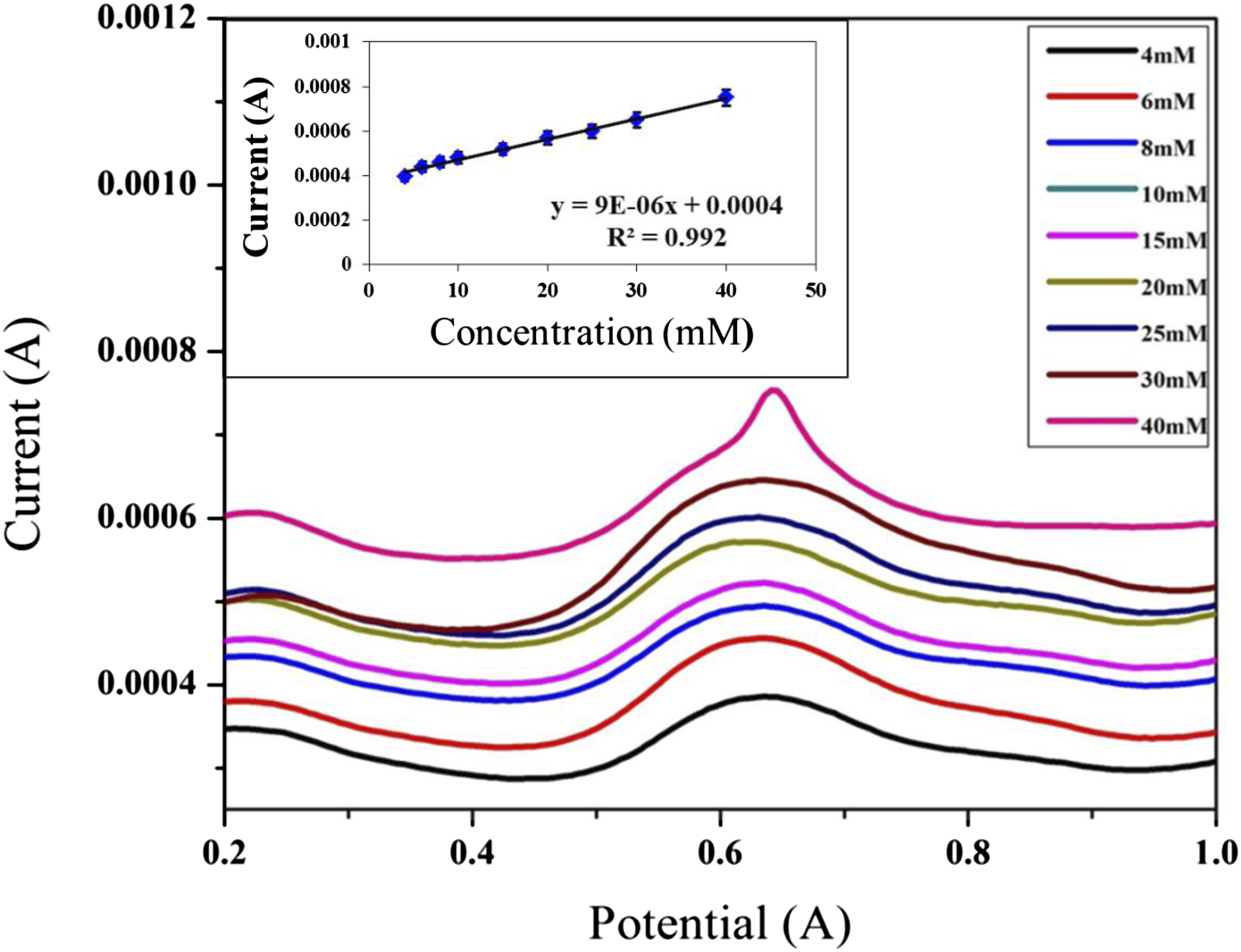
The selectivity of nanocomposite electrode for glucose sensing
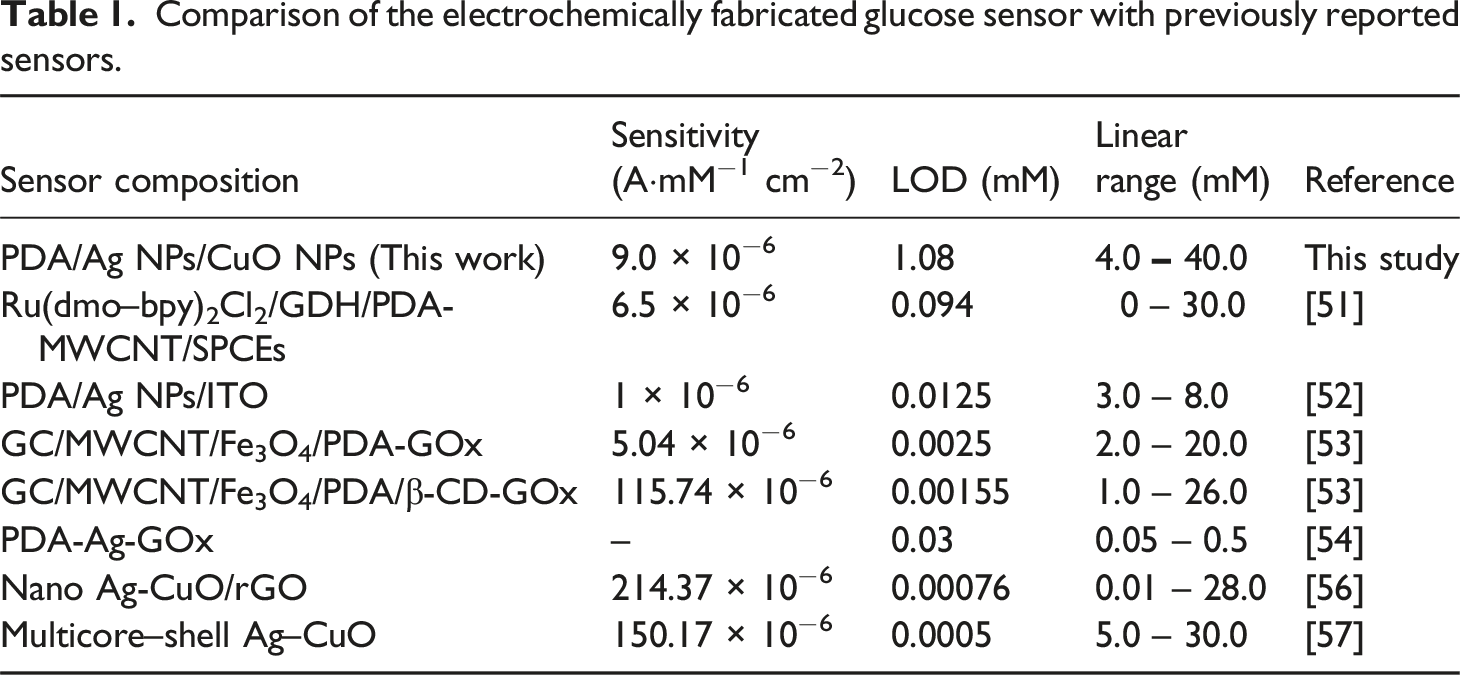
To assess the selectivity of the PDA-Ag-CuO nanocomposite electrode for glucose sensing, its electrochemical responses were recorded in the presence of 10 mM of common blood biomolecules such as ascorbic acid (AA), urea (U), cholic acid (CA), and dopamine (DA). The current responses were normalized to evaluate the electrode’s specificity. As shown in Figure 9, the PDA-Ag-CuO nanocomposite electrode generated a significantly stronger current response for glucose compared to the other analytes, reflecting its high selectivity. The normalized current values for urea, cholic acid, dopamine, and ascorbic acid were approximately 63%, 46%, 36%, and 41%, respectively. This pronounced selectivity toward glucose can be ascribed to the specific interactions and favorable surface affinity between glucose molecules and the functional groups present in the nanocomposite, which facilitate more efficient electron transfer and glucose oxidation compared to other interfering species.48–50 Table 1 gives the comparison of the performance of the fabricated sensor and sensors from the literature. The PDA/Ag NPs/CuO NPs-based sensor offers a compelling combination of sensitivity and a wide linear detection range (4.0–40.0 mM), making it highly suitable for real biological applications, especially for monitoring elevated glucose levels in diabetic patients. However some reported sensors achieve lower LODs, the broad detection range and the simplicity of fabrication using low-cost, readily available materials highlight the sensor’s practicality and scalability for future clinical use.51–57 DPV curves of PDA-Ag-CuO nanocomposite electrode in 10 mM for different biomolecules and the normalization bar chart. Comparison of the electrochemically fabricated glucose sensor with previously reported sensors.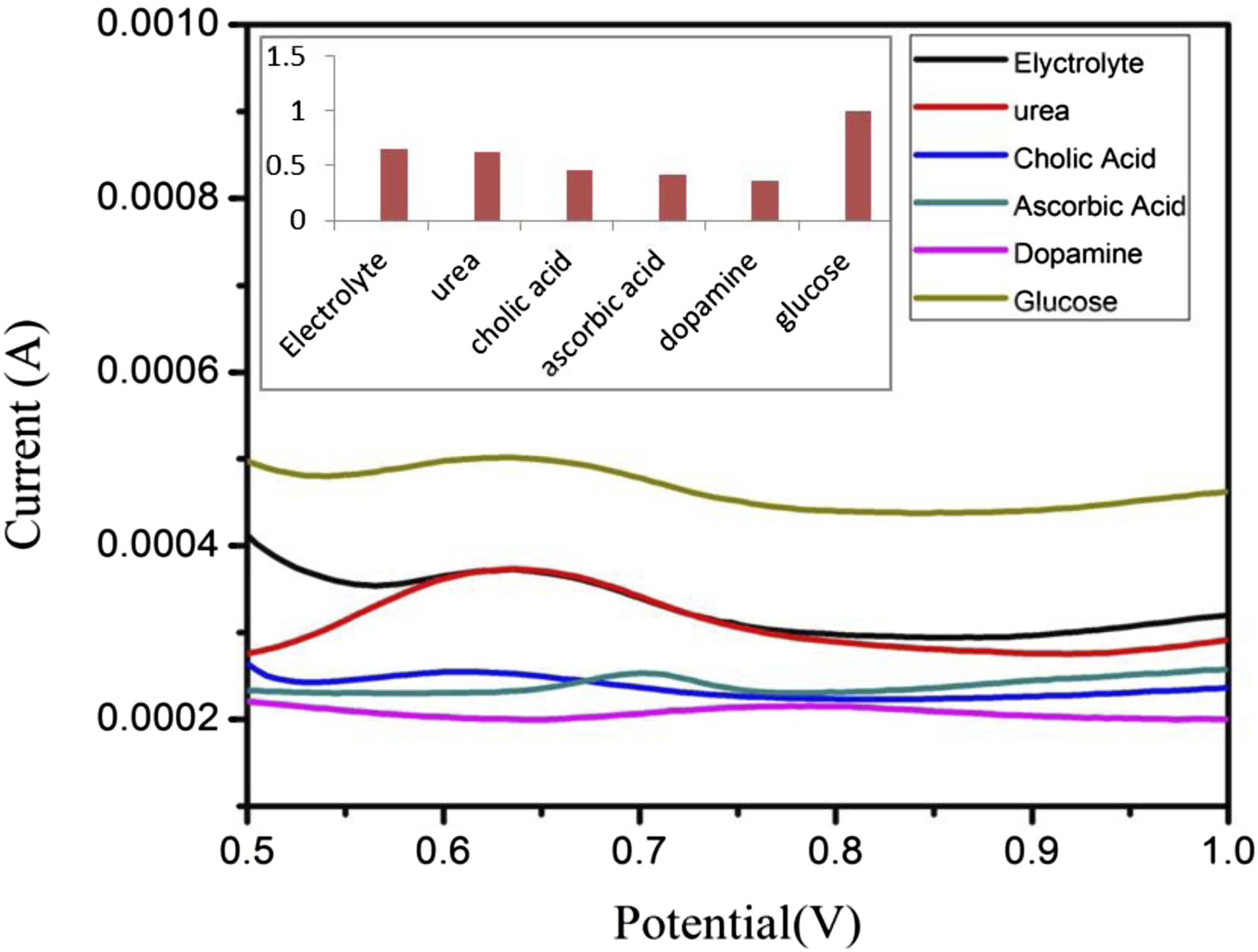
Conclusion
In this work, an innovative electrochemical biosensor for glucose detection was developed through the fabrication of two electrode types composed of PDA-Ag NPs and PDA-Ag-CuO nanocomposites, deposited onto graphite sheet substrates. XRD analysis confirmed that the amorphous nature of PDA and Ag nanoparticles exhibited a face-centered cubic (FCC) structure, while CuO nanoparticles possessed an orthorhombic crystalline form. SEM imaging revealed a cauliflower-like morphology for PDA, spherical Ag nanoparticles (ranging from 10 to 35 nm) for PDA-Ag NPs, and nanoneedle-shaped CuO particles (30–70 nm) for PDA-Ag-CuO nanocomposites, along with dispersed Ag NPs and PDA features.
The PDA-Ag NPs electrode exhibited a sensitivity of 1 × 10−5 AmM−1cm−2, a correlation coefficient (R2) of 96.5%, a limit of detection (LOD) of 1.73 mM, and a limit of quantification (LOQ) of 5.77 mM. Additionally, it demonstrated good reproducibility with a relative standard deviation (RSD) below 1.5% and a repeatability of 1.6 × 10−5 A. Meanwhile, the PDA-Ag-CuO nanocomposite electrode achieved a sensitivity of 9 × 10−6 A•mM−1•cm−2, a higher R2 of 99.2%, an improved LOD of 1.08 mM, and an LOQ of 3.60 mM, with acceptable reproducibility (RSD < 3%) and repeatability (2.8 × 10−5 A).
Selectivity tests involving typical interfering biomolecules such as ascorbic acid, urea, cholic acid, and dopamine demonstrated a significantly higher current response toward glucose, confirming the electrode’s strong selectivity and its capacity to discriminate glucose from other analytes in complex biological matrices.
Furthermore, a comparative analysis with previously reported sensors highlighted that the developed PDA/Ag NPs/CuO NPs-based sensor offers a highly competitive profile in terms of sensitivity and detection range. Notably, it provides a broader linear range (4.0–40.0 mM) compared to many other sensors, reinforcing its potential for practical glucose monitoring applications.
Compared to existing non-enzymatic glucose sensors, the fabricated electrodes offer competitive or improved sensitivity and excellent selectivity, suggesting their suitability for advanced biosensing platforms, including clinical diagnostics and portable or wearable glucose monitoring systems.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
