Abstract
In this paper, a non-enzymatic hydrogen peroxide (H2O2) sensor, based on Au nanoparticles (AuNPs) electrodeposited on an electrochemically reduced graphene oxide(ERGO)-Nafion modified glass carbon electrode (GCE), was reported. The graphene oxide-(GO-)Nafion nanocomposites were first assembled on the GCE surface to produce a GO-Nafion electrode. GO was then electrochemically reduced to produce an ERGO-Nafion modified GCE (to be subsequently denoted as GCE/ERGO-Nafion). Afterwards, AuNPs were electrodeposited on the modified GCE to form a GCE/ERGO-Nafion/AuNPs electrode. The ERGO-Nafion nanocomposites and the electrodeposited AuNPs were characterized by infrared spectroscopy and scanning electron microscopy (SEM). The results showed that the GO was reduced successfully and the AuNPs were uniformly dispersed on the GCE/ERGO-Nafion electrode with a size of about 48nm. The cyclic voltammograms and the amperometric method were used to evaluate the electrocatalytic properties of the GCE/ERGO-Nafion/AuNPs electrode. The results showed that the modified electrode displayed good electrocatalytic activity in relation to the reduction of H2O2 with a detection limit of 2 μM of (H2O2) based on a signal-to-noise ratio of three. The sensor also indicated great sensitivity (574.8 μA/(mM cm2)), wide linear range (0.02–23 mM), good selectivity and reproducibility, and long-term stability.
1. Introduction
H2O2 is not only a common and very important species in biological systems, on the basis that it is a by-product of several highly selective oxidases, but also an essential mediator in food, pharmaceutical, clinical, industrial and environmental analyses. As such, the rapid and accurate analysis of H2O2 is of great significance in biology, clinical control, the food industry and environmental protection [1]. Among various techniques for H2O2 detection, the electroanalytical technique has aroused attention due to its simple, rapid and cost-effective characteristics compared with other detection methods [2,3]. Enzyme-based electrochemical sensors can result in high sensitivity and good selectivity; however, the most common and serious problem with enzymatic H2O2 sensors are concerned with instability and poor reproducibility. Recently, non-enzymatic H2O2 sensors have been explored in the hope of improving the electrocatalytic activity and selectivity towards the reduction of H2O2. The general principle of non-enzymatic sensors is to use some special materials that can catalyse the decomposition reaction of H2O2 to replace the enzyme. To date, many metal or metal oxide nanoparticles (NPs) have been widely used to develop non-enzymatic electrochemical sensors for H2O2 detection, including gold (Au) NPs [4-6], Ag NPs [7], Pd NPs [8], Pt NPs [2,9,10], SiO2 NPs [3], MnO2, Cu2O and so on. But these electrodes often have drawbacks relating to low sensitivity, narrow linear range and poor stability [11,12]. Therefore, it is necessary to develop highly sensitive and reliable non-enzymatic sensors with wide linear range and excellent electrocatalytic activity for the reduction of H2O2. However, using only a single material makes it hard to achieve superior performance in non-enzymatic sensors. In recent years, a variety of composite materials has become the focus of advanced research in the field of non-enzymatic electrode materials.
Graphene and its derivatives have attracted a tremendous amount of attention from the perspectives of both fundamental science and technology because of a few intriguing attributes they displays, such as great chemical and thermal tolerance, fast electron transportation, high thermal conductivity, excellent mechanical stiffness and good biocompatibility [13]. They promise a wide range of industrial applications, such as in nanocomposites [14], field-effect transistors [15], electromechanical resonators [16], energy storage [17], catalyst support [18] and electrochemical sensors [19-21]. Recently, graphene has been widely used to fabricate electrochemical H2O2 biosensors due to their excellent electrocatalytic activity towards H2O2. In addition, it has been found that the electrocatalytic synergy of graphene and other nanoparticles can extremely improve the performance of the resultant electrochemical sensor. AuNPs are representative metal nanoparticles used in non-enzymatic sensors. Zhang et al. developed a novel non-enzymatic H2O2 sensor by using the graphene/Nafion/Azure I/AuNPs composite-modified GCE (graphene/Nafion/Azure I/AuNPs/GCE) [27]. Graphene was prepared by the oxidation-reduction (using hydrazine, which is very toxic) method. AuNPs were prepared in advance and then absorbed onto the graphene/Nafion/Azure I/GCE. However, the procedure was complicated and the chemically reduced graphene was hard to disperse well, which may have inhibited the performance of graphene.
In this paper, a non-enzymatic H2O2 sensor, based on AuNPs electrodeposited on an ERGO-Nafion modified GCE was reported. The GO-Nafion nanocomposites were first assembled on the GCE surface to produce the GO-Nafion electrode. Then, GO was electrochemically reduced. Afterwards, AuNPs were electrodeposited on the modified GCE to form the GCE/ERGO-Nafion/AuNPs electrode (Figure 1). The procedure for the preparation of the GCE/ERGO-Nafion/AuNPs electrode was simple and very environmentally friendly. ERGO was well dispersed in the Nafion matrix, while AuNPs were tightly bound to the ERGO-Nafion. The synergies between the ERGO and AuNPs led to a significant level of performance from the proposed non-enzymatic H2O2 sensor with a low detection limit, great sensitivity, wide linear range, good selectivity and reproducibility, and long-term stability.

Scheme of fabrication of an electrochemically reduced graphene oxide-Nafion/AuNPs modified electrode
2. Experimental
2.1 Material
The GO used in this experiment was synthesized from natural graphene powder by a modified Hummers method [22] and characterized as described in our previous works [23]. Nafion (5% EtOH solution) was purchased from Alfa Aesar Chemical. Other chemicals were purchased from local chemical suppliers. A series of phosphate buffered solution (PBS; 0.4 M) was used as a supporting electrolyte and prepared by mixing a solution of 0.4 M Na2HPO4 and 0.4 M NaH2PO4. The solutions were deoxygenated by nitrogen before the experiments. All solutions were prepared with ultrapure water, purified by the Millipore-Q System (>18 MΩ cm).
2.2 Apparatus
All electrochemical experiments were performed by a CHI660a electrochemical analyser (Chen Hua Instruments, Shanghai, China), which used a conventional three-electrode system with a bare or modified glass carbon electrode (Φ = 3 mm) as the working electrode, an Ag/AgCl (0.3 M KCl solution) electrode as the reference electrode, and a platinum wire as the auxiliary electrode. The cyclic voltammetric experiments were performed in a quiescent solution. Electrochemical impedance spectroscopy (EIS) was performed in a 0.01 M KCl solution containing 10 mM of K3[Fe(CN)6]/K4[Fe(CN)6] (1:1) at frequencies ranging from 0.01 Hz to 100 kHz at open circle potential. The amplitude of the applied sine wave potential in each case was 5 mV. The amperometric experiments were carried out in a continuous stirred solution using a magnetic stirrer (300rpm).
Fourier transform infrared (FT-IR) spectra were obtained using a Spectra Two (PerkinElmer) spectrophotometer within the spectral range of 4,000 cm−1 to 400 cm−1 with an attenuated total reflection (ATR) accessory. SEM observations were carried out with a LEO-1503 field emission scanning electron microscope (German).
2.3 Construction of the GCE/ERGO-Nafion/AuNPs electrode
GO (10mg) was dissolved in 10 mL of Nafion ethanol solution (0.2%) and treated with ultrasound for 30 min to obtain a GO-Nafion mixture suspension. The GCE was carefully polished with 1.0, 0.3 and 0.05 μm γ-Al2O3 powders in sequence and rinsed thoroughly with pure water between each polishing step, after which it was ultrasonicated in pure water, ethanol and then pure water for 5 min and dried with nitrogen gas. Six microlitres of above-mentioned GO-Nafion suspension was cast onto the surface of the GCE and air-dried. The electrochemical reduction of GO was performed by immersing the GCE/GO-Nafion electrode into a 0.5 M KCl solution by potential scanning from −0.8V to 1.7 V for 20 cycles. After washing with pure water, the electrode was immersed into a 0.5 M H2SO4 solution containing 0.4 mg/mL of HAuCl4. Electrodeposition of Au was carried out by potential scanning from −0.55V to −0.95V for 15 cycles. The obtained electrode (labelled GCE/ERGO-Nafion/AuNPs) was then washed with pure water and stored at 4 °C prior to use. For comparison, the GCE/GO-Nafion, GCE/ERGO-Nafion and GCE/AuNPs electrodes were also prepared with the same procedure as described above.
3. Results and Discussion
3.1 Characterization of the modified electrode
The electrode was modified by casting the GO-Nafion suspension on the surface of the GCE, after which GO was electrochemically reduced. FT-IR spectroscopy was used to confirm reduction. To exclude the interference of Nafion, the same amount of GO suspension was cast on the surface of GCE, after which it was reduced electrochemically. IR spectra of GO and electrochemically reduced GO (ERGO) are shown in Figure 2. Peaks at 3,300 cm−1, 1,729 cm−1, 1,624 cm−1 and 1,056 cm−1 were respectively ascribed to O-H stretching vibration, C=0 stretching vibration, C=C stretching vibration and C-O symmetry vibration. After chemically reduction, only peaks at 1,624 cm−1 and 1,056 cm−1 were observed. The disappearance of the peak at 3,300 cm−1 and 1,729 cm−1 indicates that the hydroxyl, carboxyl and carbonyl group was completely removed, although a small amount of the epoxy groups was retained.
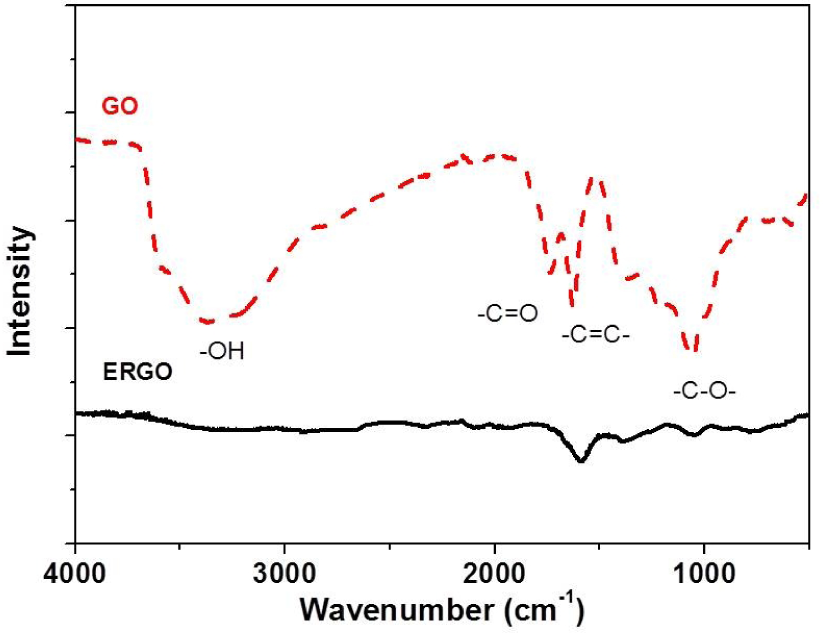
FT-IR spectra of GO and ERGO
The surface morphology of the modified electrodes was examined by SEM, as illustrated in Figure 3. GO (Figure 3A) has a typically folded and wrinkled sheet structure, which looks like soft and thin gauze. GO-Nafion looks very similar to that of GO. The SEM image of ERGO-Nafion on the electrode shows a tiled thick sheet with a wrinkled surface. AuNPs were electrodeposited on the GCE/ERGO-Nafion electrodes. The SEM image in Figure 3D shows numerous spherical nanoparticles on the surface of the electrodes; these nanoparticles are composed of Au. The average diameter of the AuNPs is about 48 nm (Inset of Figure 3D). These small AuNPs provided a large accessible surface area for the subsequent electrocatalytic reaction of analytes.

SEM images of (A) freeze-dried GO, (B) freeze-dried GO-Nafion, (C) ERGO-Nafion on the surface of GCE and (D) AuNPs on ERGO-Nafion. Inset: size distribution of AuNPs.
3.2 Electrochemical properties of the modified electrode
EIS was performed in order to investigate the interface properties of modified electrodes. Figure 4 displays the EIS profiles of different modified electrodes, in which the Nyquist plots are shown with the real part (Z′) on the X-axis and the imaginary part (–Z″) on the Y-axis. It is well known that the semicircle part of the plot, which appears at high frequencies, corresponds to the electron transfer limited process; therefore, the diameter of the semicircle portion corresponding to the electron-transfer-limited process is equal to the electron- transfer-resistance (
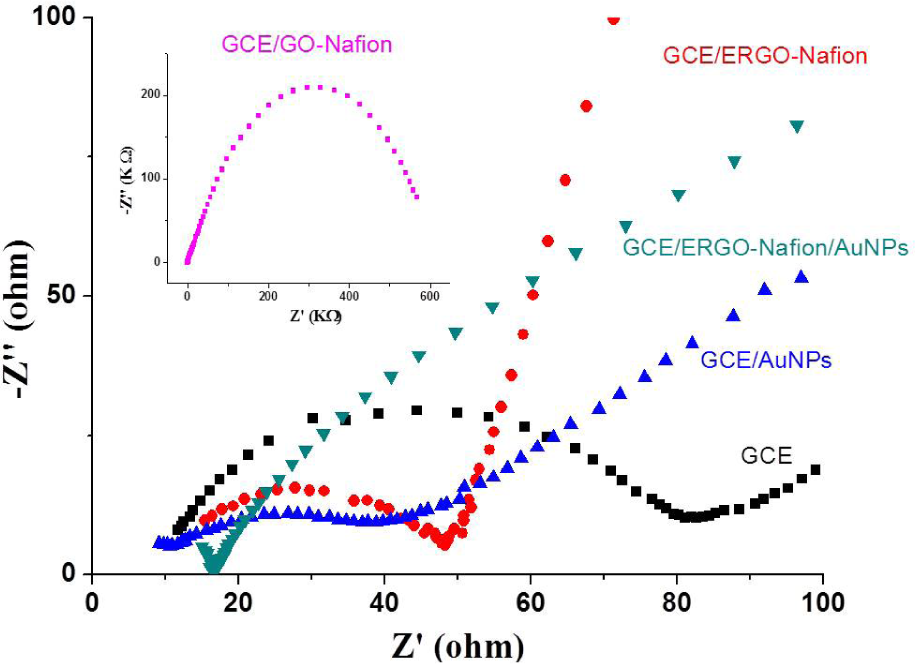
Nyquist plots of the EIS spectra of GCE, GCE/AuNPs, GCE/ERGO-Nafion and GCE/ERGO-Nafion/AuNPs electrodes in a 10mM KCl aqueous solution containing 10 mM Fe(CN)63–4−(1:1). The frequency range is from 10 mHz to 100 kHz. Inset: EIS profile of GCE/GO-Nafion.
3.3 Electrocatalytic properties of the modified electrode
Figure 5 shows the CVs curves of the GCE, GCE/GO-Nafion, GCE/ERGO-Nafion, GCE/AuNPs and the GCE/ERGO-Nafion/AuNPs electrode in 0.4 M of PBS at pH 7.4 containing 10 mM of H2O2. There was an obvious reduction peak at about −0.50 V for the GCE/ERGO-Nafion/AuNPs electrode (curve e), which corresponds to the reduction of H2O2 [24,25]. According to the previous report [26], the cathodic peak can be attributed to the reduction of H2O2 into H2O However, no obvious peak current was observed on the bare GCE electrode (curve a), the GCE/GO-Nafion electrode (curve b) and the GCE/ERGO-Nafion electrode (curve c), which indicates that AuNPs played a crucial role in the catalysed reduction of H2O2. There were also evident currents on the GCE/AuNPs electrode (curve d), but the current was much smaller than that for GCE/ERGO-Nafion/AuNPs (curve e), indicating that ERGO-Nafion can significantly improve the performance of the sensor. In fact, the ERGO-Nafion nanocomposites with a wrinkled surface provided a large surface area to produce a significant quantity of AuNPs, as well as synergistically catalysed the reduction of H2O2 with AuNPs, which accordingly resulted in a large catalytic current. It was noticeable that the peak potential for the GCE/ERGO-Nafion/AuNPs electrode was at −0.50 V, which was more positive than that for the GCE/AuNPs electrode (–0.59 V). The positive reduction peak potential indicated a fast electron transfer rate, along with good electrocatalytic activity in relation to the reduction of H2O2 for the GCE/ERGO-Nafion/AuNPs electrode.

CV curves of (a) GCE, (b) GCE/GO-Nafion, (c) GCE/ERGO-Nafion, (d) GCE/AuNPs and (e) GCE/ERGO-Nafion/AuNPs electrode in a 0.4 M PBS solution (pH 7.4) containing 10 mM of H2O2 at a scan rate of 50 mV/s
Moreover, the effect of a varying scan rate for the electrochemical performance of the modified electrode was also examined by cyclic voltammetry (CV). Figure 6A depicts the CVs of the produced GCE/ERGO-Nafion/AuNPs electrode in a 0.4 M PBS solution (pH 7.4) containing 10 mM H2O2 at a scan rate between 10 and 200 mV/s, for which the reduction current peak appeared to correspond to the reduction of H2O2 The dependence between the response current and scan rate are plotted in Figure 6B. As can be seen, the peak current is linear to the square root of the scan rate from 10 to 200 mV/s. This result revealed that the electron transfer of GCE/ERGO-Nafion/AuNPs displayed a diffusion controlled electrochemical process.
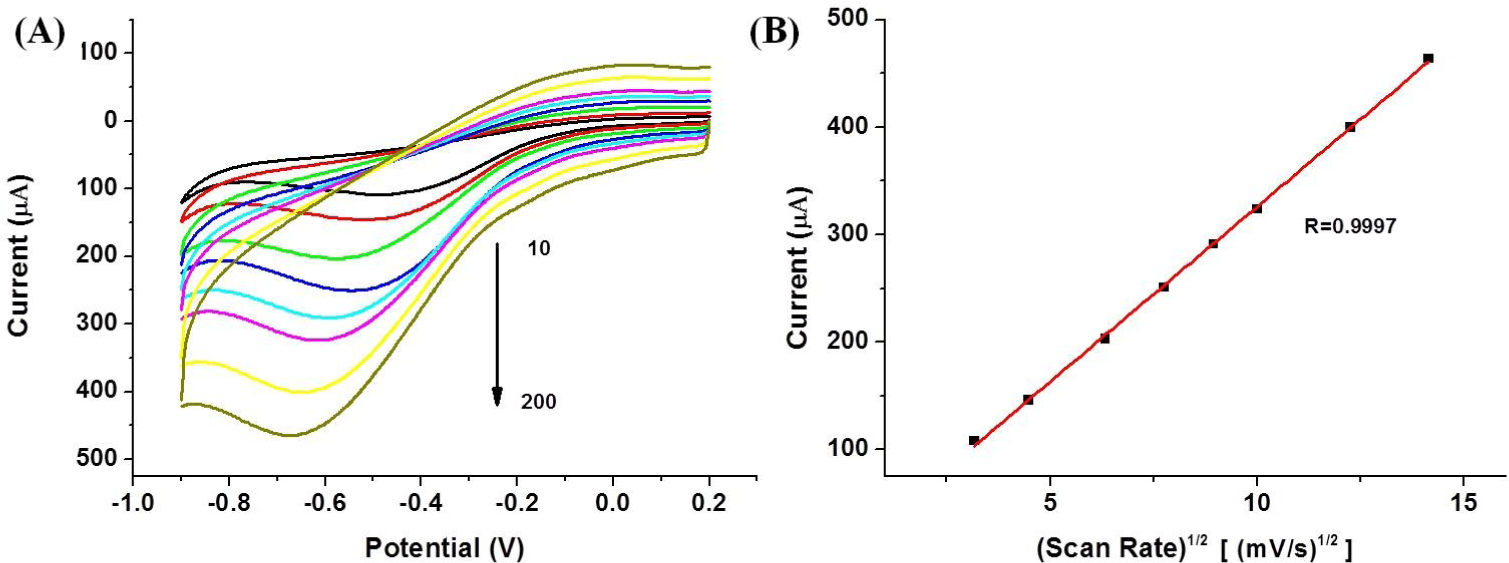
(A) CV curves of the GCE/ERGO-Nafion/AuNPs electrode in a 0.4 M PBS solution (pH 7.4) containing 10 mM H2O2 at a scan rate of 10, 20, 40, 60, 80, 100, 150 and 200 mV/s. (B) Plot of the reduction peak current against the square root of the scan rate.
3.4 Calibration, linearity and detection limit
The typical steady state current response of the GCE/ERGO-Nafion/AuNPs electrode to the successive injection of H2O2 is given in Figure 7A. The reduction current rose rapidly when H2O2 was added and reached a maximum steady state value within 1 s. The fast response indicates a quick diffusion of H2O2 into ERGO-Nafion/AuNPs nanocomposites [27] and a quick electron transfer of the AuNPs enhanced by ERGO-Nafion. Figure 7B shows the calibration curve of the amperometric response. The response of the electrode has a good linear relationship with H2O2 in the range from 20 μM to 23.0 mM (R=0.998), which is much wider than that of other H2O2 biosensors, such as an inner filter effect-based fluorescent assay [28], an Au/AuNPs/PEI/CNTs-COOH/DNA electrode[29] and a carboxyl functionalized graphene oxide (GO-COOH)- 3,3′, 5,5′-tetramethylbenzidine-based electrochemical sensor [30]. The detection limit was estimated to be 2.0 μM, based on a signal-to-noise ratio of three. The linear response range, the sensitivity and detection limit for H2O2 detection of the modified electrode prepared in the course of this work meet the demands of the practical application for the determination of H2O2 concentrations.
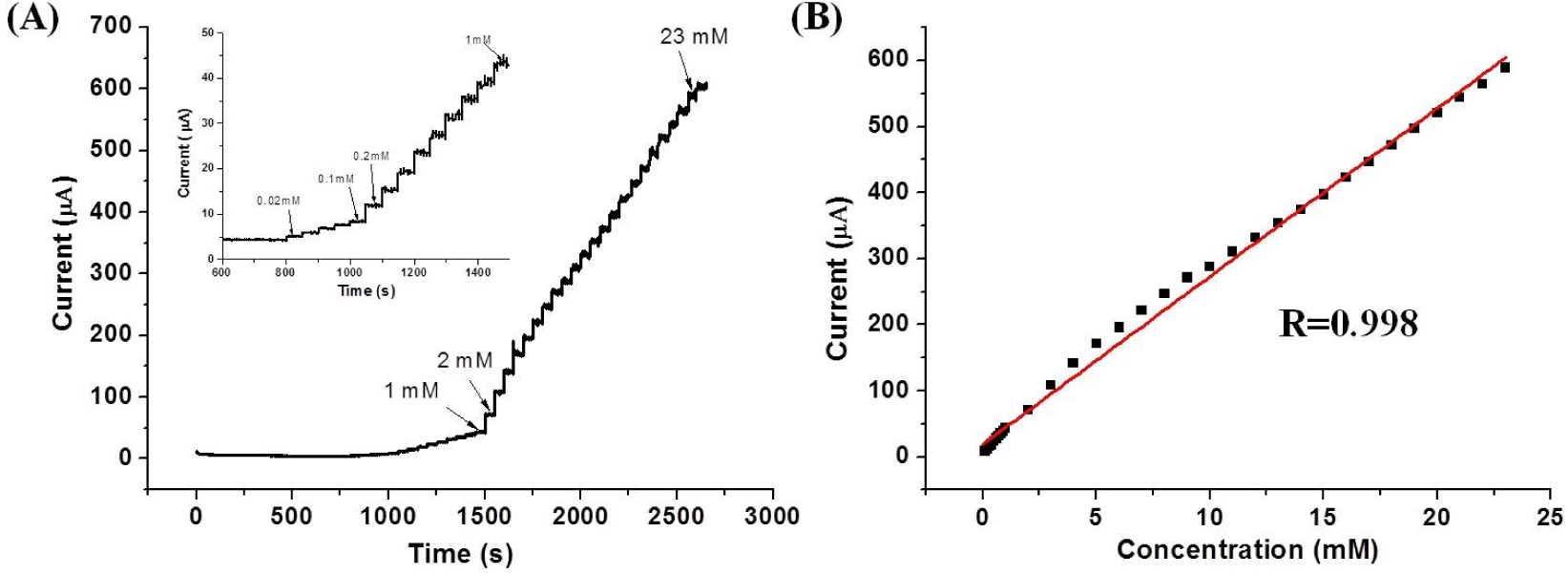
(A) Typical steady state response of the GCE/ERGO-Nafion/AuNPs electrode to the successive injection of H2O2 into the stirred PBS. (B) The plot of the calibration curve. Applied potential is −0.500 V and the supporting electrolyte is 0.4 M PBS at pH 7.4.
3.5 Selectivity, reliability, reproducibility and stability
The interferences of some electroactive compounds, which are commonly present in physiological or food samples, may prevent the accurate determination of H2O2 concentration. The common interfering species in physiological samples are ascorbic acid and uric acid, while they are ethanol and methanol in food samples. As such, these four particular interfering species were used to evaluate the selectivity of the GCE/ERGO-Nafion/AuNPs electrode. Figure 8 shows the CV curves of the GCE/ERGO-Nafion/AuNPs electrode in a 10 mM H2O2 solution, as well as 10-fold ascorbic acid (AA), 10-fold uric acid (UA), 10-fold ethanol (EtOH) and 10-fold methanol (MeOH). An obvious reduction peak of the electrode appears in the 10 mM H2O2 solution, while there was no obvious peak observed in the AA, UA, EtOH and MeOH solutions. This indicates that the proposed electrochemical sensor has a high selectivity. The main reason for such significant selectivity can be related to the use of low detection potential in this study, which greatly reduced the responses of common interferences. The reliability of the GCE/ERGO-Nafion/AuNPs electrode was performed by measuring the current response upon the addition of different amounts of H2O2 into a 0.13 mM solution. The results are summarized in Table 1. The relative standard deviation of the measured value to the truth value was less than 5%. The reproducibility and stability of the biosensor were performed by measuring the current response of the sensor upon 10mM of H2O2. The average relative standard deviation was not more than 0.47% for 20 successive determinations using the same electrode (Figure S2), while it was 2.3% for five different biosensors (Figure S3), which were fabricated independently by the same procedure described in Section 2.3, validating the good reproducibility of the method. The response current maintained 93% of its initial electrochemical response after storage at 4 °C for five days (Figure S4).
The current response upon the addition of different amounts of H2O2 into a 0.13 mM H2O2 solution


CV curves of the GCE/ERGO-Nafion/AuNPs electrode in a 0.4 M PBS solution (pH 7.4) containing 10 mM H2O2, 100 mM AA, 100 mM MeOH, 100 mM EtOH and 100 mM Glu, respectively
4. Conclusion
A non-enzymatic H2O2 sensor, based on AuNPs electrodeposited on an ERGO-Nafion modified GCE was reported. GO can be successfully electroreduced. AuNPs were uniformly dispersed on the GCE/ERGO-Nafion electrode with a size of about 48 nm. The proposed GCE/ERGO-Nafion/AuNPs electrode displayed good electrocatalytic activity in relation to the reduction of H2O2 with a detection limit of 2 μM of H2O2 (S/N=3). Moreover, the sensor also showed great sensitivity (574.8 μA/(mM cm2)), wide linear range (0.02–23 mM), good selectivity and reproducibility, and long-term stability.
Footnotes
5. Acknowledgements
The authors would like to express their gratitude for the financial support received from the National Natural Science Foundation of China (grant numbers 61171020, 61376125 and 21203247).
