Abstract
Developing green fibrous materials with uniformly distributed metal nanoparticles for highly efficient and recyclable catalysis remains a major challenge. Herein, we developed a simple, effective, and green method to immobilize gold nanoparticles (AuNPs) on polydopamine (PDA)-functionalized silk fibers (SFs) for efficient catalytic reduction of 4-nitrophenol (4-NP). The density and size of AuNPs on the PDA-coated SFs can be tuned by adjusting precursor concentration and synthesis duration, respectively. The AuNPs-PDA-SFs catalysts prepared under optimized conditions could catalyze the reduction of 4-NP, 4-nitroaniline (4-NA), and 4-amino-3-nitrophenol (4-A-3-NP) at the apparent rate constants of 0.087, 0.091, and 0.063 min−1, respectively. After six rounds of flow-through reduction of 4-NP, the AuNPs-PDA-SFs could maintain a 4-NP conversion rate greater than 92%, indicating their superior reusability and consistent catalytic activity. Due to the protein properties of SFs, the AuNPs-PDA-SFs can be degraded by enzymes and alkali solutions. This work may provide new insights for designing advanced fiber-supported recyclable catalysts with high catalytic performance and reusability in wastewater treatment.
Introduction
Due to its critical role in almost all chemical processes, catalysis has been widely studied in chemistry and life sciences.1,2 Among these studies, metallic nanoparticles (MNPs) have attracted tremendous attention in the past decade due to their intrinsic catalytic activities that could mimic natural enzymes such as peroxidase, catalase, uricase, and superoxide dismutase. 3 The enzyme-like activities, as well as high stability, bulk synthesis, low cost, and good catalytic efficiency, rendered MNPs promising alternatives to natural enzymes in biochemical and biosensing applications.4,5 In addition to enzyme-like activities, MNPs have been used to catalyze the degradation of some pollutants in wastewater. The reduction of 4-nitrophenol (4-NP), a typical toxic chemical found in wastewater, has been employed as a model system to investigate the catalytic activities of various MNPs.
Although MNPs have demonstrated outstanding potential in wastewater treatment, their practical applications were hindered by their poor recyclability. 6 Great efforts have been dedicated to developing solid-supported catalysts for easy separation and recycle. 7 The Fe3C@nitrogen-doped graphite has been used as a magnetic carrier to support gold nanoparticles (AuNPs), showing a high catalytic activity for 4-NP reduction and excellent magnetic recoverability. 8 A nanocomposite of AuNPs, magnetic γ-Fe2O3, and hydroxyapatite has also been constructed to achieve a highly efficient catalytic reduction of 4-NP and easy magnetic separation. 9 MNPs supported by the magnetic materials can be efficiently separated under the external magnetic field. However, during this process, the catalysts tend to agglomerate, significantly reducing their catalytic performance. The recovery of the catalytic activity requires cumbersome post-processing procedures, including multiple washes and ultrasonic re-dispersion.10,11 In addition, most of these materials are non-degradable, posing a threat to the ecological environment. 12 From a sustainability point of view, there is an urgent need to design degradable MNPs-incorporated catalysts that can withstand repeated separation and recycling.
Since fiber-based network structures possessed the low air-flow or liquid-flow resistance, they have been extensively studied as filter materials for air and water purification. Compared to easily aggregated inorganic or hybrid materials, fiber-based network materials, such as cellulose, 13 chitosan, 14 chitin, 15 carbon fiber, 16 and wool fabrics, 17 could maintain their hierarchically porous structures during the separation and recycling process. Therefore, the employment of fibrous materials as carriers for MNPs may offer high catalytic activities, convenient separation characteristics, and good recyclable properties. As a natural fiber with a long history, silk fiber (SF) has received tremendous attention as a carrier for catalysts due to its excellent biocompatibility, superior mechanical strength, and great biodegradability.17,18 Platinum nanoparticles have been loaded on the surface of SFs via a hydrothermal method, imparting excellent antibacterial properties and catalytic activity. 19 Magnetic ferriferous oxide (Fe3O4) nanoparticles have also been synthesized in situ on regenerated silk fibers (RSFs) using a simple co-precipitation method, demonstrating excellent catalytic activity. 20 Nevertheless, with the conventional adsorption and chemical reduction approaches, the MNPs tend to agglomerate on the fiber surface, reducing their catalytic activities. 21 Additionally, due to the poor surface binding, MNPs may leak out of the SFs during recycling, ultimately reducing their reusability. 22 Therefore, modification of SFs with evenly dispersed MNPs for highly effective recyclable catalysis is still under development, which is a challenging task.
Mussel adhesive-inspired polydopamine (PDA) has recently attracted broad interest in material science, biology, and medicine. 23 Under alkaline conditions, PDA can be produced by the oxidative polymerization of dopamine, forming a bionic film tightly adhered to various inorganic or organic substrates. 24 Moreover, the rich catechol groups in PDA can chelate metal ions for the in situ synthesis of metallic NPs. 25 The Fe3O4 has been deposited on PDA-coated carboxylated cellulose nanofibers to remove methylene blue and tetracycline hydrochloride from wastewater. The PDA layer could protect Fe3O4 nanoparticles from loss during the reaction, increasing the stability. 26 PDA nanotubes have also been constructed to synthesize AgNPs in situ for the catalytic reduction of methylene blue. 27 Hence, the polymeric layer may be deposited on the SFs to serve as 3-dimensional (3D) matrix containing abundant catechol groups for the production of high-density MNPs. However, the PDA has not been incorporated with SFs to prepare fiber-supported MNPs for highly efficient and recyclable catalysis so far.
Gold nanoparticles (AuNPs) exhibit remarkable activity and selectivity in catalytic processes due to their small-scale effects and large specific surface area.28,29 Herein, we developed a simple, effective, and green method to immobilize AuNPs on the PDA-functionalized SFs for catalyzing the reduction of phenol derivative. The synthesis parameters were explored to optimize the size and surface density of the AuNPs on the PDA-coated SFs. The phenol derivatives were reduced using the as-prepared catalysts to examine their catalytic activity, stability and recoverability. The feasibility of the catalysts in wastewater treatment was also demonstrated with the continuous-flow reduction mode. The PDA-mediated synthesis of AuNPs may facilitate the development of fiber-supported catalysts and adhere to the principles of green chemistry. Additionally, the covalent bonding between the PDA matrix and AuNPs may enhance the activity and reusability of the fiber-supported catalysts. This work could provide a novel method for creating fiber-supported catalytic materials that could be used to degrade organic contaminants in the future.
Experimental
Chemicals
Sodium borohydride (NaBH4), dopamine hydrochloride (DA), chloroauric acid (HAuCl4), 4-nitrophenol (4-NP), sodium hydroxide (NaOH), 4-amino-3-nitrophenol (4-A-3-NP), 4-nitroaniline (4-NA), and papain were bought from Aladdin (Shanghai, China). Hydrochloric acid (HCl) and Tris (hydroxymethyl) aminomethane (Tris) were purchased from Sigma-Aldrich (Shanghai, China).
Preparation of AuNPs-PDA-SFs
The PDA-modified SFs were synthesized according to the previously reported literature with minor modifications (Figure 1).30,31 The raw SFs extracted from silkworm Schematic diagram for the fabrication of AuNPs-PDA-SFs.
Materials characterization
Morphologies of pristine SFs, PDA-coated SFs, and AuNPs-PDA-SFs were characterized with field emission-scanning electron microscope (FE-SEM, JSM-7800F, Joel, Japan) and transmission electron microscope (TEM, JEM-2100, Joel, Japan). The energy diffraction spectra (EDS, INCA X-Max 250) were collected during the FE-SEM tests for elemental analysis. The crystalline patterns of pristine SFs, PDA-coated SFs, and AuNPs-PDA-SFs were obtained using X-ray diffraction spectrometry (XRD-7000, Shimadzu, Japan) in the 2θ range of 10–70°. Fourier transform infrared spectrometry (FT-IR, TENSOR 27, Bruker, Germany) was conducted to characterize the chemical properties of the samples.
AuNPs-PDA-SFs-catalyzed reduction of 4-NP
The AuNPs-PDA-SFs (1 mg) were added into an aqueous solution containing 0.2 mM 4-NP and 20 mM NaBH4. The reduction process was recorded by measuring the UV-Vis spectrum of the solution every 5 min. Continuous shaking and mixing were applied to ensure sufficient contact between the AuNPs-PDA-SFs and 4-NP. Since the concentration of NaBH4 was higher than 4-NP in the mixture, it can be deemed as constant during the reaction.
32
The catalytic reduction of 4-NP follows a quasi-first-order kinetics, and the relevant formula is as follows:
The catalytic reduction of AuNPs-PDA-SFs toward 4-A-3-NP and 4-NA was also investigated in the current study using the same methodology as above.
Catalytic performance of AuNPs-PDA-SFs in a continuous-flow column
The bottom of a column was filled with 60 mg AuNPs-PDA-SFs. Then, 5 mL aqueous mixtures containing 2.5 mM 4-NP and 250 mM NaBH4 were added to the column. The liquid flew out from the column at a constant flow rate. The color change of the solution before and after the treatment was recorded to evaluate the reduction of 4-NP.
Degradation of AuNPs-PDA-SFs
The AuNPs-PDA-SFs were immersed in water, papain aqueous solution (20 mg/mL), or sodium hydroxide solution (20 mg/mL), incubating at ambient conditions for 48 h. Digital photographs of the samples were taken at 24-h intervals to illustrate the degradation of the materials.
Results and discussion
Preparation of AuNPs-PDA-SFs
The FE-SEM was employed to observe the surface morphologies of pristine SFs, PDA-coated SFs, and AuNPs-PDA-SFs. The degummed SF was fiber-shaped with a very smooth surface and an average diameter of ∼11 μm (Figure S1A). After a 12-h incubation in the Tris-HCl dopamine solution, the fiber surface became rough, suggesting that the dopamine may polymerize to form a PDA layer on the fiber surface (Figure S1B). The stress-strain curves show that the PDA-coated SFs possess a much higher fracture stress than the pristine SFs (Figure S2), which may be attributed to the formation of the dense polymeric film. Since the particle size and surface density of AuNPs determine the catalytic efficiency,
33
it is crucial to investigate effects of the precursor concentration and the incubation duration on the synthesis process. When the PDA-coated SFs were incubated with a 1 mg/mL HAuCl4 solution, nanoparticles could be produced on the fiber surface (Figure 2(a)). The size of the particles synthesized on the SF surface increased from 27.6 ± 3.5 to 48.5 ± 10.4 nm as the concentration increased from 1 to 5 mg/mL (Figure S3). In addition, several triangular nanoparticles were observed in the 3 and 5 mg/mL groups (Figures 2(b) and (c)). When the concentration further increased to 7 mg/mL, triangle nanoplates appeared on the surface (Figure 2(d)). The results show that the size and the morphology of the AuNPs could be affected by the HAuCl4 concentration. Moreover, to explore the effects of incubation duration on the density and size of the in situ synthesized AuNPs, we incubated the PDA-coated SFs in a 5 mg/mL HAuCl4 solution for 6, 10, 14, and 20 min, respectively. After only 6 min of incubation, the AuNPs, with an average particle size of ∼26.9 nm, were uniformly distributed on the surface (Figures 2(e) and S4A). When the duration increased to 10 min, the average size of the AuNPs rose to ∼48.5 nm (Figures 2(f) and S4B). As the duration elongated to 14 and 20 min, the average particle sizes increased to ∼50.2 and ∼79.1 nm, respectively (Figures 2(g) and (h), S4C and S4D). The catechol groups of PDA chelate the Au ions, forming tiny AuNPs. As the reaction time extends, the Au ions in the solution deposit on the gold seeds to increase the particle size. Based on the above results, it can be concluded that the average size of the AuNPs on PDA-coated SFs could be tuned by controlling the incubation duration. Characterization and catalytic performance of the catalysts. FE-SEM images of AuNPs synthesized on the PDA-coated SFs with different concentrations of HAuCl4 solution (a–d) and different reaction durations (e–h); (i) Effects of the HAuCl4 concentration on the catalytic performance of the AuNPs-PDA-SFs; (j) Effects of the synthesis duration on the catalytic performance of the AuNPs-PDA-SFs.
Since the surface density and size of the SFs-immobilized AuNPs could be tailored, the catalytic performance must be investigated to determine the ideal parameters for synthesizing AuNPs. The linear variation of ln (Ct/C0) against time (Figures 2(i) and (j)) confirms that the catalytic reaction follows quasi-first-order kinetics. When the synthesis duration was fixed at 10 min, the K
Characterization of AuNPs-PDA-SFs
The AuNPs-PDA-SFs prepared with the optimal conditions were characterized using EDS to verify the surface immobilization of PDA and AuNPs. The elements of C, N, and O were well distributed in the EDS mapping images, matching well with the compositions of the SFs and PDA (Figure 3(a)). The immersion in a HAuCl4 solution during the AuNPs thesis may result in the uniform distribution of the Au elements in the mapping image, and the intensity of the yellow color was significantly higher at the locations of AuNPs. The Cl peak could be observed in the EDS curve, which may be due to the adsorption of Cl− on the surface. The TEM and XRD were conducted to further explore the morphology and crystal structure of AuNPs on the PDA-coated SFs, respectively. As shown in Figure 3(b), the SF surface was covered by a thin layer of film embedded with a few nanoparticles, indicating the successful deposition of PDA and incorporation of AuNPs. Two broad bands at 20.45° and 29.26° could be observed in the XRD spectra of the pristine SFs, PDA-coated SFs, and AuNPs-PDA-SFs catalysts (Figure 3(c)), corresponding to crystalline diffractions of silk II and silk Ι.
34
Both of them were the characteristic conformations in the crystal structure of the silk fibroin. Two new diffraction peaks at 38.18° and 44.39° appeared in the spectrum of AuNPs-PDA-SFs, referring to the (111) and (200) crystal planes of the face-centered cubic structured AuNPs, respectively.
35
Since face-selective adsorption of reactants on the catalysts played a crucial role in a catalytic system,
36
the superior crystallinity of the as-prepared AuNPs may contribute to the catalytic performance of the AuNPs-PDA-SFs. The FT-IR spectra of the pristine SFs, PDA-coated SFs, and AuNPs-PDA-SFs catalysts (Figure 3(d)) showed three absorption peaks at 1228, 1515, and 1637 cm−1. The 1228 and 1515 cm−1 peaks could be attributed to the N-H in-plane bending vibration of the secondary amide and C-N stretching, respectively. The peak at 1637 cm−1 corresponds to the C=O stretching vibration of the amide.
37
The PDA-modified SFs exhibited two new peaks at 1620 and 1502 cm−1, which may be assigned to the N-H stretching vibration of the secondary amine and the benzene ring skeleton vibration in the PDA structure, respectively.
38
Characterization of the catalysts prepared under optimal conditions. (a) FE-SEM and EDS mapping images of AuNPs-PDA-SFs; (b) TEM image of AuNPs-PDA-SFs; (c) XRD patterns of pristine SFs (a), PDA-coated SFs (b), and AuNPs-PDA-SFs (c); (d) FT-IR spectra of pristine SFs (a), PDA-coated SFs (b), and AuNPs-PDA-SFs (c).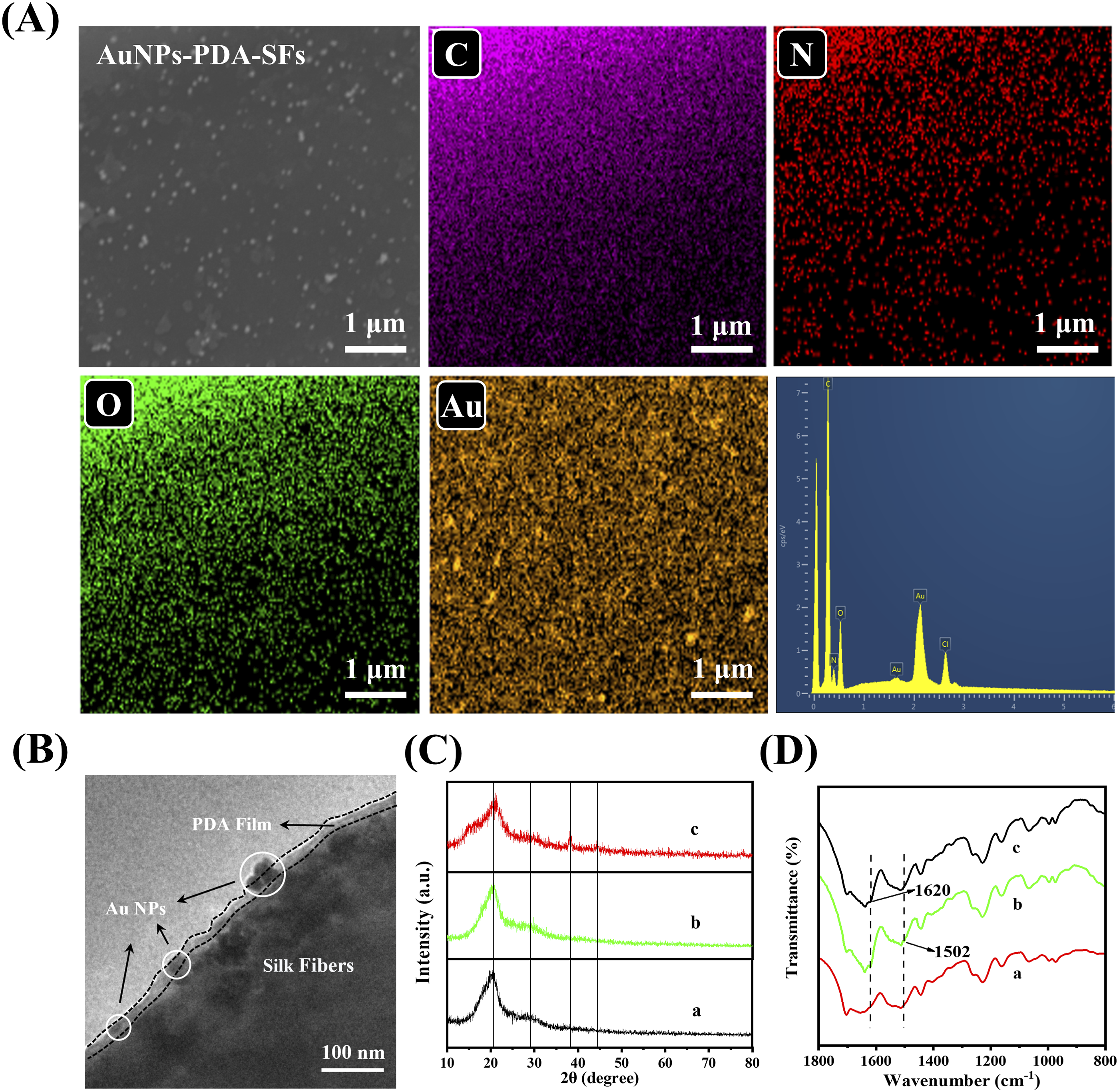
Catalytic performance of AuNPs-PDA-SFs
The 4-NP reduction was conducted in the presence of AuNPs-PDA-SFs and NaBH4 to evaluate the catalytic performance. Under catalysis, the electrons can be transferred from the BH4– group (donor) to the 4-NP (acceptor), reducing 4-NP into 4-AP. If the system lacked AuNPs-PDA-SFs, the catalytic process of 4-NP will be affected and have difficulty to be reduced (Figure 4(a)). When the AuNPs-PDA-SFs were added, the absorbance of 4-NP at 400 nm gradually decreased with time. During this process, two new peaks appear at 226 and 300 nm, which can be identified as characteristic peaks of 4-aminophenol (4-AP). After 25 min, the absorbance at 400 nm was relatively weak, indicating the efficient degradation of 4-NP (Figure 4(b)). The K
Catalytic performance of the AuNPs-PDA-SFs on different phenol derivatives. UV–Vis spectra of 4-NP solutions without catalysts (a) and with catalysts (b); (c) plots of ln (Ct/C0) against time for 4-NP; UV–Vis absorption spectra of 4-NA solutions without catalysts (d) and with catalysts (e); (f) Plots of ln (Ct/C0) against time for 4-NA; UV–Vis spectra of 4-A-3-NP solution without catalysts (g) and with catalysts (h); (i) Plots of ln (Ct/C0) against time for 4-A-3-NP.
Catalytic performance of AuNPs-PDA-SFs in a continuous-flow mode
To demonstrate the potential of the AuNPs-PDA-SFs in practical water treatment, they were used as fillers to cover the bottom of a glass burette for continuous-flow catalysis. Since the contact time between the pollutant and the catalyst was much shorter in the continuous flow mode than in the suspension mode, based on the literature and the findings in our preliminary tests, higher doses of AuNPs-PDA-SFs (60 mg), 4-NP (2.5 mM) and NaBH4 (250 mM) were employed to accelerate the reduction process.
39
The mixture with the bright yellow color could be converted into a colorless solution during the 6-min flow-through process (Figure 5(a)). The AuNPs-PDA-SFs-catalyzed flow-through reduction of 4-NP could be ideally repeated in three independent experiments (Figure S8). As the flow rate increases, the adsorbent may become denser, ultimately affecting the catalytic performance. Moreover, the AuNPs-PDA-SF catalysts were recyclable. The conversion rate could be maintained at a level greater than 92% even after six rounds of flow-through 4-NP reduction (Figure 5(b)). After each cycle of catalytic reduction, the absorbance of the flow-through solution at 400 nm was below 0.1, which was much lower than the initial one, proving the efficient degradation of 4-NP (Figure 5(c)). The catalysts show high catalytic activity in the treatment of the flow phase, making it feasible for the industrial application. It should be noticed that the robustness of the setup should be considered in the industrial scale applications. Stronger materials, such as iron or aluminum, should be utilized to prepare the catalytic columns for better robustness. Catalytic performance of the AuNPs-PDA-SFs in a continuous-flow mode. (a) Real-time measurement of AuNPs-PDA-SFs-catalyzed reduction of 4-NP in the continuous-flow mode; (b) conversion efficiency of the recycled AuNPs-PDA-SFs catalysts; (c) absorbance of the solution after each round of flow-through treatment.
Degradation of AuNPs-PDA-SFs
To investigate the degradability, the as-prepared catalysts were soaked in water, papain solution, and NaOH solution (Figure 6). During incubation in water under ambient conditions for 48 h, there is no noticeable change in AuNPs-PDA-SFs, showing excellent stability in water. However, when the catalysts were soaked in papain or NaOH solution for 24 h, the AuNPs-PDA-SFs became loose, indicating the partial degradation of the fibers. The majority of AuNPs-PDA-SFs in the enzyme solution and virtually all of the AuNPs-PDA-SFs in the NaOH solution were degraded as the incubation duration increased to 48 h, proving the great degradability of the AuNPs-PDA-SF catalysts in both protease and alkaline solutions. As shown in Figure 5, the AuNPs-PDA-SF catalysts could be repeatedly used several times for degrading polluted water without any washing or drying process. During the catalyst fabrication process, PDA (a biocompatible coating) is immobilized on the SF surface, which enables the reductant-free synthesis of AuNPs. In addition, the SF substrate can be completely degraded during post-processing without any side effects on the environment. Therefore, the synthesis and post-processing of the catalyst does not cause serious environmental problems compared to conventional catalysts. The prepared AuNPs-PDA-SFs are more promising in terms of catalytic performance, recyclability and biodegradability than previously reported catalysts (Table S1). In the future, AuNPs-PDA-SFs could also be a multi-functional platform for combining tumor photothermal therapy and antibacterial infection. Degradation of AuNPs-PDA-SFs in different solution.
Conclusion
Herein, AuNPs were successfully immobilized on PDA-coated SFs to reduce 4-NP, 4-NA, and 4-A-3-NP. The high density and homogeneous synthesis of AuNPs can be achieved on the PDA-functionalized SFs. By controlling reaction parameters, such as the precursor concentration and reaction duration, the density and size of the AuNPs could be tuned. The best catalytic performance could be achieved at a HAuCl4 concentration of 5 mg/mL and a reaction duration of 10 min. The catalysts could be used as fillers to prepare a glass burette-based column for continuous-flow catalysis. After six recycling rounds, the AuNPs-PDA-SFs catalyst could maintain 4-NP conversion rate greater than ∼92%, verifying its stability and reusability. Moreover, the AuNPs-PDA-SFs could be degraded in alkaline and enzymatic solutions within 48 h. To the best of our knowledge, this is the first report on the PDA-mediated synthesis of AuNPs catalysts supported by SFs that exhibit outstanding reusability, high catalytic performance, and remarkable degradability. This work has produced a novel SF-supported catalyst for wastewater treatment that is simple to separate and recover and has an effective catalytic effect.
Supplemental Material
Supplemental Material - Polydopamine-mediated in situ synthesis of gold nanoparticles uniformly distributed on silk fibers as reusable catalysts for efficient 4-nitrophenol reduction
Supplemental Material for Polydopamine-mediated in situ synthesis of gold nanoparticles uniformly distributed on silk fibers as reusable catalysts for efficient 4-nitrophenol reduction by Zhendong Zhang, Jing Xiao, Chunyou Wang, Fangmiao Song, Wei Sun, Chenhui Wang, Zhisong Lu and Yan Zhang in Journal of Industrial Textiles.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the specific research fund of Innovation Platform for Academicians of Hainan Province (YSPTZX202126), the National Natural Science Foundation of China (No. 52273123), and the Natural Science Foundation of Chongqing, China (CSTB2022NSCQ-MSX0474).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
