Abstract
This study evaluated the structural and functional properties of poly (vinyl alcohol)/sodium carboxymethyl cellulose (PVA/NaCMC) hydrogels synthesized via the freeze-thawing method using 1% acetic acid (AAc) and distilled water (Dw) as solvents. The results demonstrate that solvent choice and NaCMC concentration significantly influence hydrogel performance, particularly for wound dressings. PVA/NaCMC (AAc)-3.5% emerged as the optimal formulation, exhibiting superior antimicrobial activity, structural stability, and enhanced crosslinking, as evidenced by FT-IR, SEM, and TEM analyses. XRD confirmed higher crystallinity in the AAc-based hydrogel, while swelling tests showed greater water absorption in the Dw-based hydrogel, attributed to its looser network. Antimicrobial tests highlighted the broad-spectrum efficacy of PVA/NaCMC (AAc)-3.5%, inhibiting Gram-positive and Gram-negative bacteria and fungi due to acetic acid’s low pH and hydrogen bond stabilization. These findings emphasize the importance of solvent selection in designing hydrogels for wound care applications requiring infection prevention and structural integrity.
Keywords
Introduction
In recent years, natural polymers such as cellulose, chitosan, starch, and chitin have gained significant attention over synthetic polymers like acrylic acid, acrylamide, and polyethylene glycol in applications such as wound hydrogels, contact lenses, and drug delivery systems, owing to their high hydrophilicity, biocompatibility, non-toxicity, biodegradability, flexibility, and ability to retain moisture.1–5 However, despite these advantages, natural polymers face notable challenges, including poor mechanical strength, limited processability, and insufficient antimicrobial activity, which restrict their broader application in biomedical fields.6–8 Compared to other materials and methods, this study offers a distinct advantage by addressing these limitations through a tailored approach. It focuses on enhancing the antimicrobial properties of hydrogels while maintaining their biocompatibility and physical performance, thereby bridging a significant gap in the current use of natural polymer-based materials. Unlike conventional methods that may rely on synthetic additives or chemical crosslinking agents, this study utilizes acetic acid as a dual-function agent to improve both antimicrobial efficacy and physical properties. The novelty of this research lies in its exploration of acetic acid as a direct antimicrobial-enhancing solvent for PVA/NaCMC hydrogels, which has not been previously reported. Parameters such as water absorption capacity and surface wettability were analyzed through swelling and contact angle measurements, providing critical insights into the structural integrity and potential durability of these hydrogels for biomedical applications. This innovative approach not only improves the functional performance of the hydrogels but also highlights their suitability for use in advanced wound dressing materials.
This study focuses on the synthesis of biomaterials specifically designed for wound dressings, rather than applications such as contact lenses, tissue engineering, or drug delivery. Wound dressings facilitate wound healing by absorbing exudates and maintaining a moist environment, which is critical for tissue repair.6–8 Natural hydrogels used in wound dressings not only exhibit excellent biocompatibility and soft tissue-like characteristics but also support scar tissue regeneration by interacting with the tissue and allowing the transport of oxygen, nutrients, and water-soluble metabolites. 2,9,10 Additionally, the chemical purity and mechanical properties of cellulose-based hydrogels are important for their use in wound dressings and skin replacements.11,12
Carboxymethyl cellulose hydrogels, composed of carboxymethyl cellulose and featuring a significant number of hydroxyl and carboxylic groups, are widely preferred due to their high water/moisture retention capacity, good biodegradability, and low cost.13,14 Carboxymethyl cellulose is a water-soluble derivative of cellulose, which dissolves in water-based solutions, including weak acids like acetic acid. The solubility of carboxymethyl cellulose in acetic acid depends on various factors such as the concentration of acetic acid, temperature, and the degree of substitution of carboxymethyl groups. When carboxymethyl cellulose is added to acetic acid, the carboxymethyl groups interact with acetic acid molecules through hydrogen bonding, which promotes the dissolution of carboxymethyl cellulose in the solution. Additionally, the acidity of acetic acid protonates some hydroxyl groups in cellulose, further facilitating its solubility in acetic acid. 15 Sodium carboxymethyl cellulose (NaCMC), a synthetic polymer derived from carboxymethyl cellulose, exhibits excellent bioadhesive, biodegradable, and biocompatible properties, similar to those of carboxymethyl cellulose. NaCMC, produced through reactions in an alkaline medium, acts as a cellulose derivative and water-soluble cellulose ether. It is widely utilized as a gelling, stabilizing, thickening, and suspending agent across various industries, including food, wound care, medicine, cosmetics, and agriculture.15–19 Carboxymethyl cellulose and NaCMC hydrogels, despite their favorable structural and biocompatibility properties, exhibit poor mechanical strength due to the physical crosslinks formed by the numerous reactive hydroxyl groups in their chains.20,21 To enhance the mechanical strength of these hydrogels, polymer chains must be crosslinked with another biocompatible and water-soluble polymer. Poly (vinyl alcohol) (PVA), frequently used in pharmaceutical and biomedical applications, is well-suited for this purpose. 22 PVA is a semi-crystalline polymer containing hydroxyl groups in its chain, and physical cross-linking between NaCMC and PVA occurs via hydrogen bonding between these hydroxyl groups.23–25 The shared biodegradable properties of NaCMC and PVA make them excellent candidates for blending.23,24 Furthermore, the water solubility of PVA ensures homogeneous mixing with NaCMC, as NaCMC typically uses water as its solvent.21,26–28 However, NaCMC alone does not exhibit significant antimicrobial activity in aqueous media, limiting its use in PVA/NaCMC hydrogels intended for antimicrobial wound dressings.29–32 Acetic acid, a non-toxic, inexpensive, and readily available substance, effectively inhibits various bacteria and fungi responsible for wound infections. Studies have shown that acetic acid demonstrates notable antimicrobial activity and supports wound healing.33–35 For instance, Agrawal et al. 34 demonstrated its effectiveness against a range of fungi and bacteria at a 1% concentration, while McDonnell 35 demonstrated that acetic acid at a 1% concentration effectively inhibited the growth of various fungi and bacteria, thereby accelerating wound healing. Furthermore, their study found that even lower concentrations, such as 0.5%, were sufficient to create a favorable environment for bacterial inhibition. This effect can be attributed to the mechanism by which acetic acid exerts its antimicrobial activity. Vinegar is mainly comprised of acetic acid, a weak organic acid, for which the antimicrobial effect is primarily mediated by its undissociated form. The undissociated acetic acid molecules passively diffuse through the bacterial cell wall and disrupt the intracellular environment by lowering the pH. This acidification interferes with essential metabolic processes and ultimately inhibits bacterial growth. This mechanism highlights the potential of acetic acid as an effective antimicrobial agent, even at relatively low concentrations. The resulting change of the internal pH is believed to have an inhibitory effect on the bacteria by releasing protons. 36 Acetic acid has already been used in the food industry to inhibit food pathogens. Various studies have shown a protective effect of acetic acid on various types of meat, 37 tomatoes, carrots and some salads.38–40 Further studies were also able to proof an inhibitory effect against certain microorganisms such as Enterobacteriaceae. Many scientific research have been carried out on the use of some products containing acetic acid in the fight against microorganisms.40–45 In a study conducted by Ayhan and Bilici 46 the authors were able to show that acetic acid destroys the cell wall structure, causing a loss of ATP in the cell. In another study, it was suggested that polyphenolic compounds may also play a role in the antimicrobial effect of acetic acid. Polyphenols have been shown to bind to the peptidoglycan structure of the cell wall and the phospholipid bilayer in the outer membrane of Gram-negative bacteria, compromising the integrity of the cell. Furthermore, by inhibiting the formation of amino and carboxyl groups of proteins, polyphenols have been shown to interfere with the activity of intracellular bacterial enzymes.47,48 These findings highlight the combined antimicrobial properties of acetic acid and polyphenols, making acetic acid a valuable additive for NaCMC-based hydrogels. Although studies have examined the antimicrobial properties of NaCMC/chitosan composite films prepared using acetic acid as a solvent,30,49 the direct use of acetic acid as a solvent to enhance the antimicrobial properties of PVA/NaCMC hydrogels has not been explored. Previous research has shown that acetic acid, even at low concentrations (1%), can improve the physical and mechanical properties of materials.50–56 However, this study uniquely investigates the role of acetic acid in enhancing the antimicrobial activity of PVA/NaCMC hydrogels, filling a significant gap in the literature.
The freeze-thawing method was used to prepare PVA/NaCMC hydrogel in our study. The Freeze–thawing (FT) method, hydrogels can be obtained by repeated freezing and thawinging processes without crosslinking agents through physical crosslinking.25,57,58 The Freeze–thawinging (FT) method is used to make soft, flexible, and swellable PVA/NaCMC hydrogels.22,24,25 Many hydroxyl groups in the PVA chain and the crystal structure of PVA make duty as a source of hydrogen bonding in the formation of these hydrogels.59,60 Furthermore, the crystal structure of PVA attracted the attention of researchers working on physically cross-linked hydrogels made with repeated freezing and thawinging cycles, and PVA is preferred because of these properties in the freeze-thawinging (FT) process.61–63 In addition to these, the many hydroxy and carboxylic groups of NaCMC allow it to absorb water and moisture.64,65 Prepared for this purpose the PVA/NaCMC hydrogels exhibited distinctive properties such as high water/moisture absorption content, excellent biodegradability, and high tensile strength in many studies.23,66–68
In our study, the antimicrobial properties of PVA/NaCMC hydrogels in both dilute water and 1% acetic acid solvent were investigated. Non-toxic, environmentally friendly, low-cost PVA/NaCMC hydrogels with physical cross-links were obtained by freeze-thawinging. For this purpose, it has been studied both to examine the effect on physical cross-linking by working at different volume percentages of NaCMC and to increase the skin adhesion. To determine both effects on physical crosslinking and antimicrobial, morphological, chemical, physical properties of the obtained PVA/NaCMC wound dressings hydrogel, these hydrogels were characterized by Fourier transfer infrared (FT-IR) spectroscopy, X-ray diffraction (XRD) measurements, scanning electron microscopy (SEM), transmission electron microscopy (TEM), water absorption-swelling tests, contact angle measurements, and antimicrobial tests. Thanks to the antimicrobial activity against different bacterial species of our prepared PVA/NaCMC hydrogels, it will be possible to expand the usage areas of PVA/NaCMC hydrogels.
Materials and Methods
Materials
PVA (typical average MW = 146.000-186.000; 98.0%–98.8% hydrolyzed) and NaCMC (typical average Mw = ∼250.000) were purchased from Sigma-Aldrich Co. Distilled water (DW) was used as the solvent. Acetic acid (glacial 100%, pro analysis) was purchased from Merck (Darmstadt, Germany). These materials were used as received without further purification and functionalization.
Preparation of Hydrogels
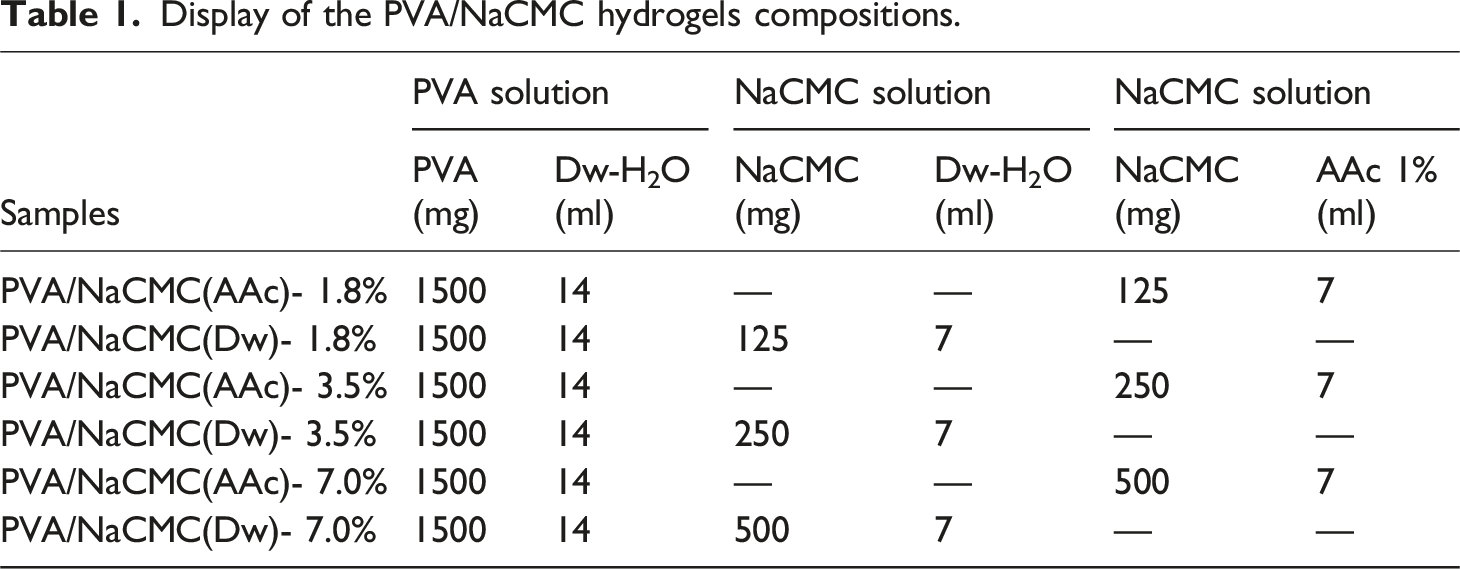
Display of the PVA/NaCMC hydrogels compositions.
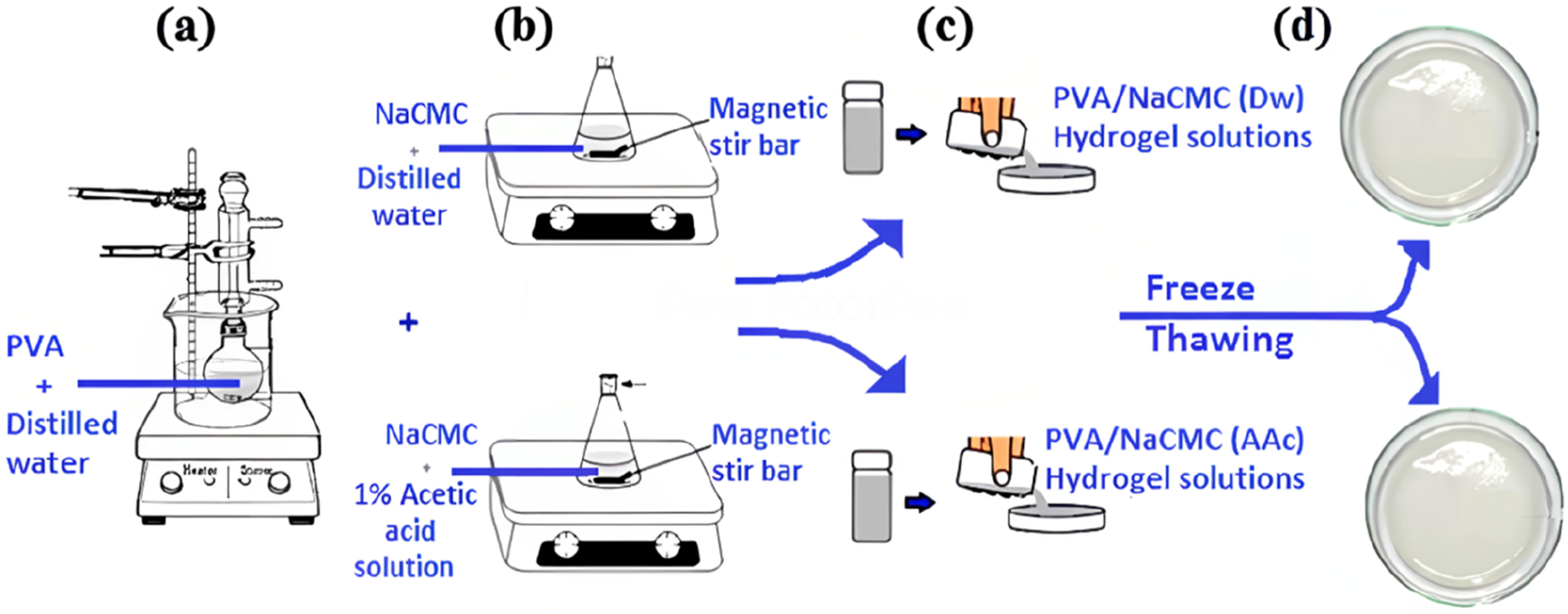
Prepation of PVA solution (a), Prepation of 1.8, 3.5, 7% (w/w) changing amounts of NaCMC with 1% acetic acid and distilled water (b), PVA/NaCMC(Dw) and PVA/NaCMC(Dw) solutions pour into sterile Petri dishes (c), The freezing and thawing cycle temperature range is −20°C to 20°; the freezing and melting times are 18 and 6 h (d).
The two solutions (PVA and NaCMC) were then mixed thoroughly on a magnetic stirrer at 300 rpm until a homogeneous solution was obtained at room temperature (Figure 1(c)). The resulting mixtures were poured into Petri dishes for hydrogel formation. During this process, the PVA ratio was kept constant, while NaCMC concentrations were varied between 1% and 10%. Suitable ratios were determined to be 1.8%, 3.5%, and 7%.
The prepared mixtures were subjected to a freeze-thaw (F-T) cycle, involving freezing at −20°C for 18 hours followed by thawing at 20°C for 6 hours (Figure 1(d)).22,25,66 This freeze-thaw process was repeated three consecutive times to ensure adequate physical crosslinking. After the final thawing step, the hydrogels were fully formed and ready for further analysis. The thawing process was performed at room temperature without any additional drying steps to preserve the hydrogel structure.
Characterization
Swelling Test
The swelling degree of the hydrogel samples was determined using the gravimetric method. The water uptake and swelling behavior of the hydrogels were evaluated by comparing the weights (W) of the hydrogels before (WD) and after (WS) immersion in distilled water. Hydrogel samples were cut into 2 × 2 cm2 pieces, dried at room temperature for 24 hours, and their dry weights (WD) were measured using a high-precision balance.
Each sample was immersed in distilled water at room temperature for a total duration of 24 hours. Swelling capacity measurements were recorded at specific time intervals of 1, 3, 5, 7, 12, and 24 hours. At each interval, the hydrogels were removed from the water, and excess surface water was gently blotted off using filter paper. The wet mass (WS) was recorded for each sample.
The swelling degree (SD) was calculated using the following formula69,70:
Contact Angle Measurement
The contact angle of the hydrogels was measured using a DataPhysics OCA contact angle instrument equipped with a high-resolution camera to evaluate the wettability of the samples. A 5 µl drop of distilled water was placed on the hydrogel surface using a micro-syringe. The drop was allowed to stabilize for 5 seconds, after which the contact angle was measured. The angle formed between the water drop and the hydrogel surface was captured by the camera, and the value was recorded.71,72 All measurements were conducted at room temperature to ensure consistency in environmental conditions. To minimize variability and ensure reproducibility, the contact angle was measured on at least five different areas of the hydrogel surface for each sample. The average contact angle value was calculated and used for further analysis. This approach ensures that the recorded contact angle represents the average wettability of the hydrogel rather than localized surface variations.
Antimicrobial Activity Test
In this study totally eight microorganisms (Escherichia coli ATCC 25922, Pseudomonas aeruginosa ATCC 27853, Klebsiella pneumoniae ATCC 70603, Staphylococcus aureus ATCC 43300 (MRSA), Salmonella enteritidis ATTC 13076, Sarcina lutea ATCC 9341, Bacillus cereus ATTC 11778, Candida albicans NRRL Y-417) were used to determine of antimicrobial potential of chemical compounds. Standard microorganisms were obtained from Microbiology Research Laboratory of Vocational School of Health Services, Selcuk University. Broth micro dilution method was conducted for antimicrobial activity of chemical compounds according to Balouiri, Sadiki and Ibnsouda. 73
The chemicals were initially prepared at different concentrations and added into the first wells of microplates containing 100 µl of Mueller Hinton Broth. Then, twofold dilutions of the chemicals were made by dispensing the solutions to the remaining wells. Then 100 µl chemical compounds were transferred to first well of the plate and serial dilution was done by transferring of 100 µl volume mixture via multichannel pipette. When the chemical-medium mixture was ready then fresh microorganism inoculum prepared from 0.5 Mc Farland turbidity and final concentration 5 × 105 were added to each well. Plates were sealed and incubated in an incubator at 37°C for 18-24 hours (C. albicans was incubated for 2 days at 28°C). Gentamicin was used as positive control. After incubation period 20 µl of 2,3,5 Tri phenyl Tetrazolium Chloride solution (0.5%) loaded to each well for detecting of minimum inhibitory concentration of test compounds. The MIC is the lowest concentration of antimicrobial agent that completely inhibits growth of the organism in tubes or micro dilution wells as detected by the unaided eye. 74
Scanning Electron Microscopy (SEM)
The morphological properties of the hydrogels were investigated using scanning electron microscopy (SEM, Jeol JSM 6510 LV). For SEM analysis, the samples were frozen in liquid nitrogen to ensure brittleness and subsequently fractured. He prepared samples were mounted onto SEM sample stubs and sputter-coated with gold under a 10⁻2 bar vacuum for 60 seconds at a voltage range of 10–15 mA to enhance surface conductivity prior to imaging. During SEM imaging, the microstructure of the fracture surfaces of the hydrogels was analyzed and recorded. This method provided detailed insights into the internal structure of the hydrogels, facilitating the characterization of their surface morphology. SEM analysis involved morphological investigations conducted for each hydrogel formulation. Each sample was frozen in liquid nitrogen, fractured, and images were captured from different fracture surfaces. The SEM images obtained were compared across various surfaces to evaluate potential microstructural variations. To enhance data reliability, at least three different regions of each sample were imaged. This approach allowed for a more comprehensive evaluation of the hydrogel surface morphology and minimized experimental errors.
Transmission Electron Microscopy (TEM)
Transmission electron microscopy (TEM) analysis was performed using a JEOL-JEM 2100 microscope operating at an accelerating voltage of 100 kV and a GATAN-ORIUS camera for high-resolution imaging. For sample preparation, thin sections (∼70 nm) of the hydrogels were obtained using an ultramicrotome equipped with a diamond knife. To enhance contrast, the sections were stained with uranyl acetate and lead citrate before being mounted onto copper grids. T This method enabled a detailed examination of the nanoscale architecture and structural uniformity of the hydrogels. For each hydrogel formulation, images were captured from three different regions, and the one with the best contrast and clarity was selected for analysis. This approach minimized variability and ensured an accurate and reliable representation of the internal structure of the hydrogels.
Fourier-Transformed Infrared Spectroscopy (FT-IR)
The crosslinking and functional groups of the hydrogels were analyzed using Fourier Transform Infrared Spectroscopy (FT-IR, Bruker, Waltham, MA, USA) within the wavenumber range of 4000–400 cm⁻1. Hydrogel samples were cut into 5 × 5 mm2 squares and dried at room temperature to remove residual moisture. FT-IR spectra were recorded with 16 scans per sample to ensure high-resolution data. For each hydrogel formulation, measurements were conducted on three different regions of a single sample, and the results were compared to verify the homogeneity of the hydrogels. Once homogeneity was confirmed, one representative FT-IR spectrum from these regions was selected and plotted for each formulation. This approach enhanced the reliability of the analysis and ensured that the FT-IR spectrum accurately represented the overall structure of the hydrogels.
X-Ray Diffraction
The crystalline structure of the hydrogels was analyzed using X-ray Diffraction (XRD) with a Bruker D8 Advance diffractometer (Bruker, Germany). The measurement conditions included a CuKα radiation source (λ = 1.540 Å)), an operating voltage of 30 kV, a current of 10 mA, and a scanning speed of 2°/min. The XRD measurements were performed at the Advanced Technology Research and Application Center of Selcuk University. For each hydrogel formulation, measurements were conducted on three different regions of a single sample to account for potential structural variations. Once homogeneity was confirmed across the regions, one representative XRD pattern was selected and plotted for each formulation. This approach ensured that the XRD pattern accurately reflected the overall crystalline structure of the hydrogels.
Result and Characterization Data
Swelling Performance of Hydrogels
The swelling behavior of PVA/NaCMC hydrogels was systematically analyzed to investigate the effects of solvent type and NaCMC concentration on water absorption capacity and gel stability. The results, presented in Figure 2 and Table 2, reveal a strong dependence on both the solvent used (acetic acid or distilled water) and the NaCMC content. The swelling degree was determined by monitoring weight changes over time after immersing the hydrogels in distilled water at room temperature for 24 hours. For each hydrogel formulation, measurements were taken from three different regions of the samples, and the average values were calculated to represent the characteristic swelling behavior of each formulation. This methodological approach ensured robust and reliable results by accounting for potential heterogeneities within the hydrogel structures.30,49 Percentage swelling degree as a function of time for the series of PVA/NaCMC hydrogels. Water contact angle and swelling degree results of PVA/NaCMC hydrogels.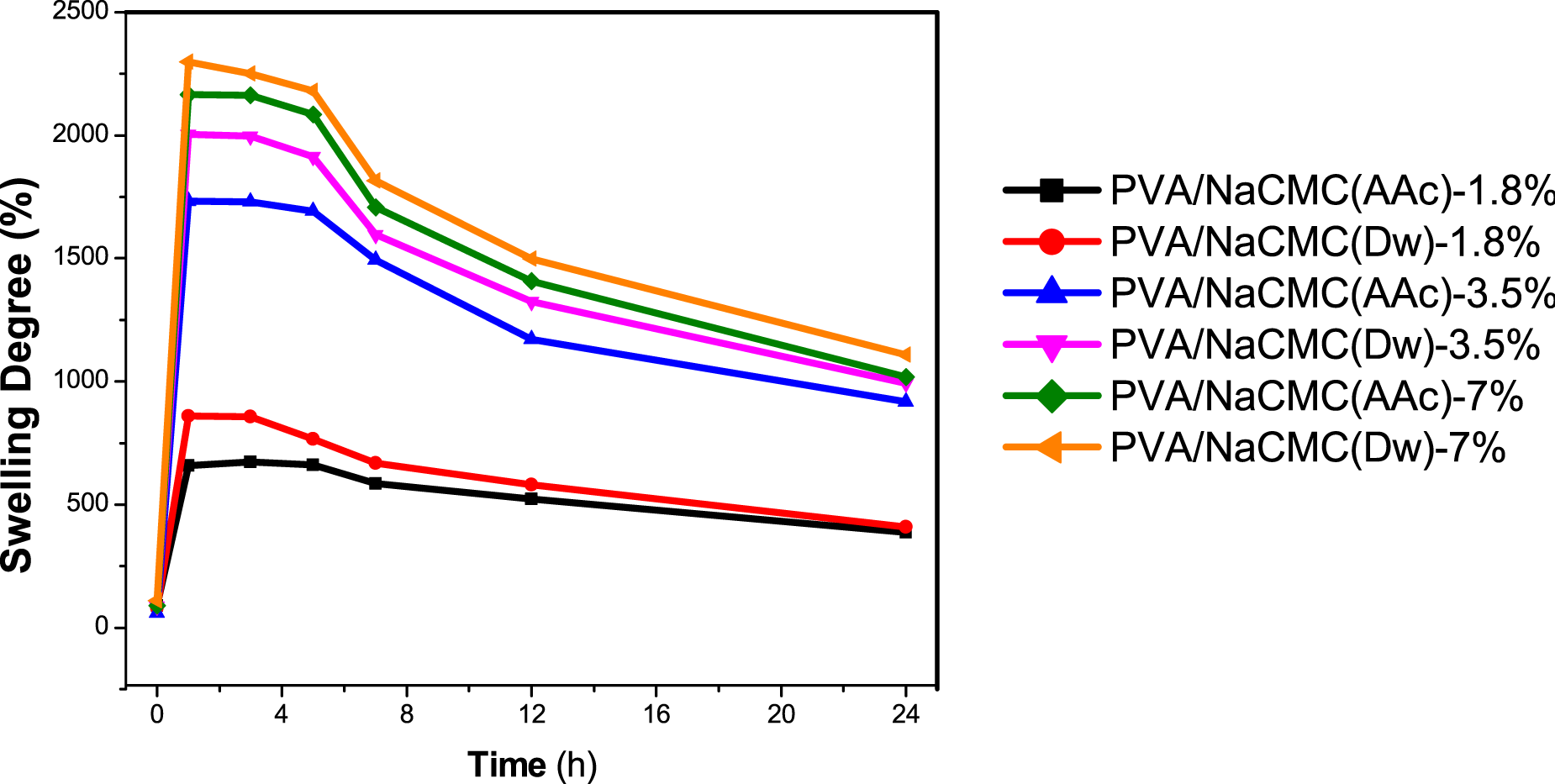

Hydrogels prepared with distilled water (PVA/NaCMC (Dw)) exhibited consistently higher swelling degrees compared to those prepared with acetic acid (PVA/NaCMC (AAc)). For example, at approximately 1 hour, the average swelling degree of PVA/NaCMC (Dw)-7% reached a maximum value of 2300 ± 95.39, while PVA/NaCMC (AAc)-7% demonstrated a slightly lower degree of 2164 ± 84.25. This discrepancy can be attributed to the looser network structure of PVA/NaCMC(Dw) hydrogels, as evidenced by the lower crosslinking density observed in XRD patterns (Figure 6(b)).59,68 Distilled water, as a solvent, allows NaCMC to retain its classical high water absorption properties due to less constrained hydrogen bonding. Conversely, PVA/NaCMC (AAc) hydrogels exhibited a more compact network structure, resulting in slightly lower swelling degrees. This enhanced crosslinking in PVA/NaCMC (AAc) hydrogels was confirmed by the increased intensity of -OH groups in FT-IR spectra (Figure 6(a)), driven by interactions between the carboxymethyl groups of NaCMC and acetic acid.28,59 Despite the slightly lower swelling degree, the antimicrobial properties imparted by acetic acid provide an additional functional advantage, as discussed in later sections. 49
All hydrogels demonstrated a rapid increase in swelling degree within the first hour, followed by stabilization between the 1st and 3rd hours. For instance, the swelling degree of PVA/NaCMC (AAc)-1.8% was 660 ± 6.93 at ∼1 hour and remained consistent at 662 ± 5.5 at ∼5 hours. Beyond the 5th hour, a gradual decline in swelling degree was observed across all formulations, likely due to structural relaxation or degradation. By the 24th hour, gel deterioration was evident, highlighting the need for optimized hydrogel stability for long-term applications.28,75
Hydrogels containing higher NaCMC concentrations (7%) exhibited the highest swelling degrees at ∼1 hour. However, SEM analysis (Figure 4) indicated aggregation in these formulations, negatively affecting their physical properties and reducing their suitability for wound dressing applications.23,76 This issue was particularly evident in PVA/NaCMC (Dw)-7% and PVA/NaCMC (AAc)-7%, where NaCMC aggregates disrupted uniform swelling behavior and compromised structural integrity. In contrast, hydrogels with 3.5% NaCMC demonstrated balanced performance. For instance, PVA/NaCMC (AAc)-3.5% showed an average swelling degree of 1730 ± 113.91, while PVA/NaCMC (Dw)-3.5% exhibited a slightly higher value of 2004 ± 45.54. These formulations avoided aggregation, maintained consistent swelling behavior, and demonstrated superior suitability for biomedical applications.30,49,59,68 Additionally, while the swelling degrees of PVA/NaCMC (AAc)-3.5% and PVA/NaCMC (Dw)-3.5% were comparable, PVA/NaCMC (AAc)-3.5% displayed significantly higher antimicrobial activity due to the effects of acetic acid. This dual functionality—effective water absorption and antimicrobial properties—positions PVA/NaCMC (AAc)-3.5% as a promising candidate for wound dressing materials, capable of hydrating wound sites and preventing microbial infections.28,75
Contact Angle Measurement
Table 2 and Figure 3 present the contact angle measurements of PVA/NaCMC hydrogels prepared with varying NaCMC concentrations and solvents. In biomedical applications, especially for wound dressings, the optimal contact angle should be less than 90°, ensuring effective absorption of exudates from the wound surface.72,77 In this study, all PVA/NaCMC hydrogel formulations demonstrated low contact angles ranging from 37.11 ± 1.05° to 29.12 ± 0.95°, indicating excellent surface adhesion and wettability. To ensure consistent and reliable results, contact angle measurements were taken from five different regions of each hydrogel formulation, and the recorded values were averaged. This approach minimizes the effect of localized variations and provides a comprehensive evaluation of the hydrogel’s overall wettability. Hydrogels prepared with distilled water (PVA/NaCMC (Dw)) exhibited lower contact angles compared to their acetic acid-based counterparts (PVA/NaCMC (AAc)), indicating a more hydrophilic surface for Dw-based hydrogels. The lower contact angles observed in Dw-based hydrogels suggest the formation of a looser network structure, allowing for easier interaction with water at the surface. For instance, PVA/NaCMC (Dw)-7% had the lowest contact angle of 29.12 ± 0.95°, while PVA/NaCMC (AAc)-7% showed a slightly higher value of 30.50 ± 1.15°. These findings align with the swelling degree results, as Dw-based hydrogels exhibited higher swelling degrees due to the effective utilization of NaCMC’s water absorption capacity. On the other hand, hydrogels prepared with acetic acid (PVA/NaCMC (AAc)) demonstrated higher contact angles, indicating a more hydrophobic surface. This phenomenon is attributed to the denser (physically crosslinked or interaction-driven) network structure formed by strong hydrogen bonding between the carboxymethyl groups of NaCMC and acetic acid molecules. This denser structure restricts water interaction at the surface, resulting in higher contact angles. For example, PVA/NaCMC (AAc)-1.8% showed a contact angle of 37.11 ± 1.05°, whereas PVA/NaCMC (Dw)-1.8% demonstrated a lower contact angle of 32.30 ± 0.98°. The compact network structure in AAc-based hydrogels is further supported by the increased intensity of -OH groups in FT-IR spectra (Figure 6(a)) and the higher crosslinking density observed in XRD patterns (Figure 6(b)). Contact angle measurements for different PVA/NaCMC compositions. (A) PVA/NaCMC (AAc)-1.8%: 37.11°, (B) PVA/NaCMC (Dw)-1.8%: 32.30°, (C) PVA/NaCMC (AAc)-3.5%: 36.97°, (D) PVA/NaCMC (Dw)-3.5%: 31.13°, (E) PVA/NaCMC (AAc)-7%: 30.50°, (F) PVA/NaCMC(Dw)-7%: 29.12°.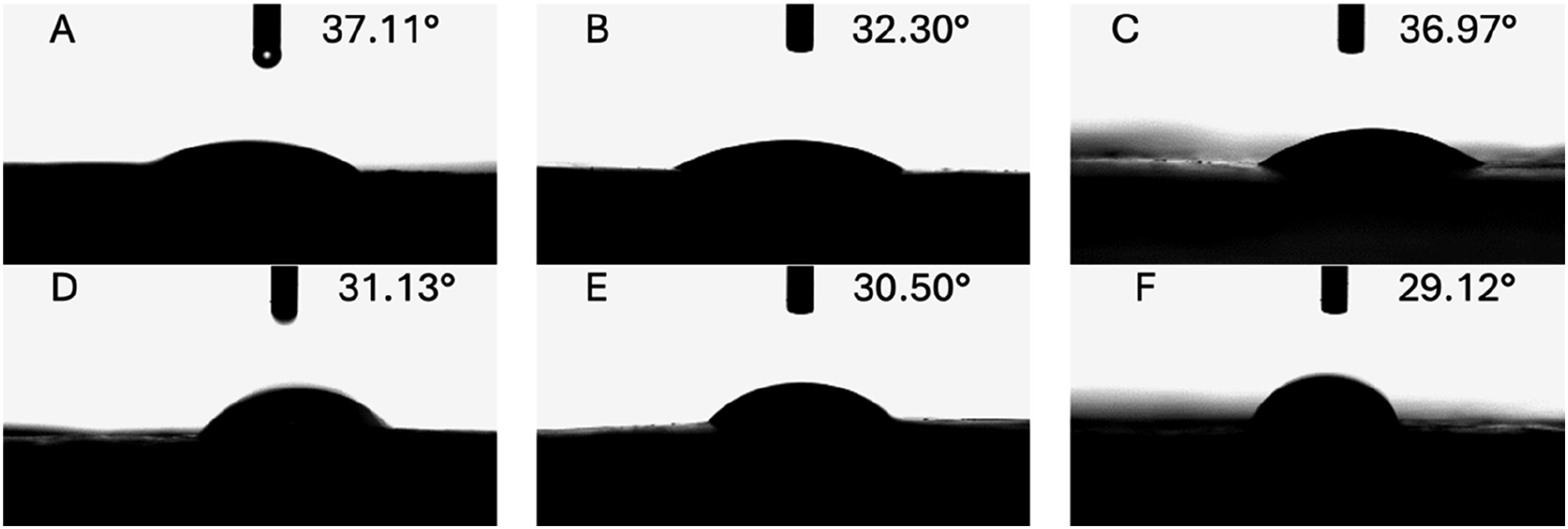
The NaCMC concentration also significantly influences the contact angle of hydrogels. Lower NaCMC concentrations (1.8%) resulted in higher contact angles, suggesting a less hydrophilic surface due to the hydrogel matrix’s limited water absorption capacity. For example, PVA/NaCMC (Dw)-1.8% exhibited a contact angle of 32.30 ± 0.98°, while PVA/NaCMC (AAc)-1.8% showed a higher contact angle of 37.11 ± 1.05° due to the denser network structure. When the NaCMC concentration increased to 3.5%, the contact angles decreased, indicating improved surface hydrophilicity and water interaction capacity. For instance, PVA/NaCMC (Dw)-3.5% had a lower contact angle of 31.13 ± 1.08°, while PVA/NaCMC (AAc)-3.5% demonstrated a slightly higher contact angle of 36.97 ± 1.12°. At higher NaCMC concentrations (7%), the contact angle further decreased, with PVA/NaCMC (Dw)-7% exhibiting the lowest contact angle of 29.12 ± 0.95°. However, SEM analysis (Figure 4) revealed aggregation in these hydrogels, negatively affecting physical stability and surface adhesion properties. Aggregation at high NaCMC concentrations disrupted the structural integrity of the hydrogel matrix, leading to non-uniform swelling behavior. For instance, PVA/NaCMC (Dw)-7% exhibited a swelling degree of 2181.0 ± 18.4% and a contact angle of 29.12 ± 0.95°, but structural disruptions were observed due to aggregation. SEM micrograph of the morphologies of the hydrogels: (A) PVA/NaCMC(AAc)-1.8%, (B)PVA/NaCMC (Dw)- 1.8%, (C) PVA/NaCMC (AAc)- 3.5%, (D) PVA/NaCMC (Dw)- 3.5%, (E) PVA/NaCMC (AAc)- 7%, (F)PVA/NaCMC (Dw)- 7%.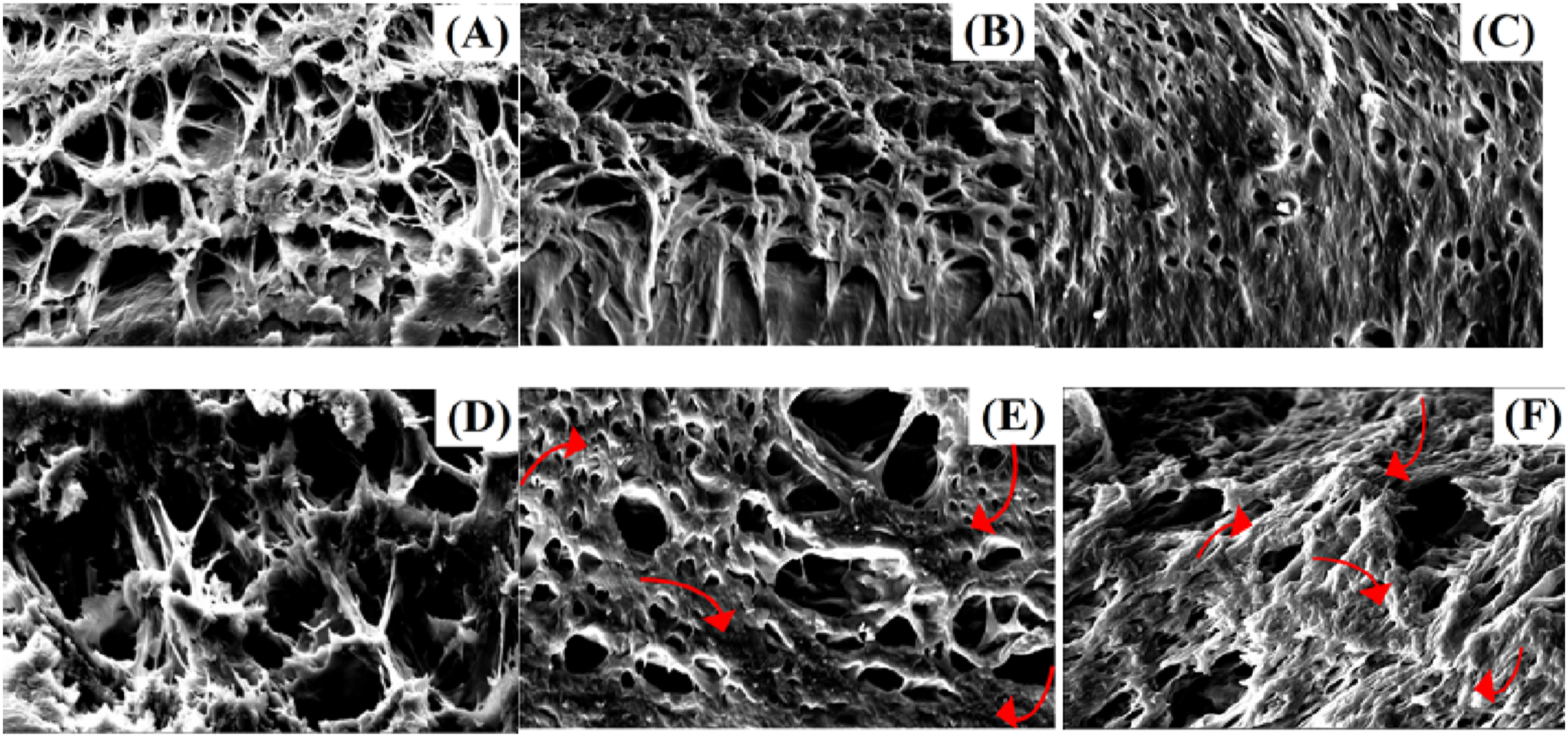
When these findings are combined with the swelling degree results, hydrogels containing 3.5% NaCMC achieved an optimal balance between swelling behavior and surface wettability. For example, PVA/NaCMC (AAc)-3.5% exhibited a swelling degree of 1730.0 ± 15.5% and a contact angle of 36.97 ± 1.12°, while PVA/NaCMC (Dw)-3.5% demonstrated a higher swelling degree of 2004.0 ± 18.9% and a lower contact angle of 31.13 ± 1.08°. These results confirm that PVA/NaCMC (Dw) hydrogels possess a more hydrophilic surface and effectively utilize NaCMC’s water absorption capacity. However, PVA/NaCMC (AAc) hydrogels offer controlled swelling and additional functionality through a denser network structure, making them suitable for specific applications such as antimicrobial wound dressings.
Antimicrobial Activity Test
Antimicrobial activity test results of chemicals obtained from broth micro dilution test against pathogen microorganisms (na: not active).
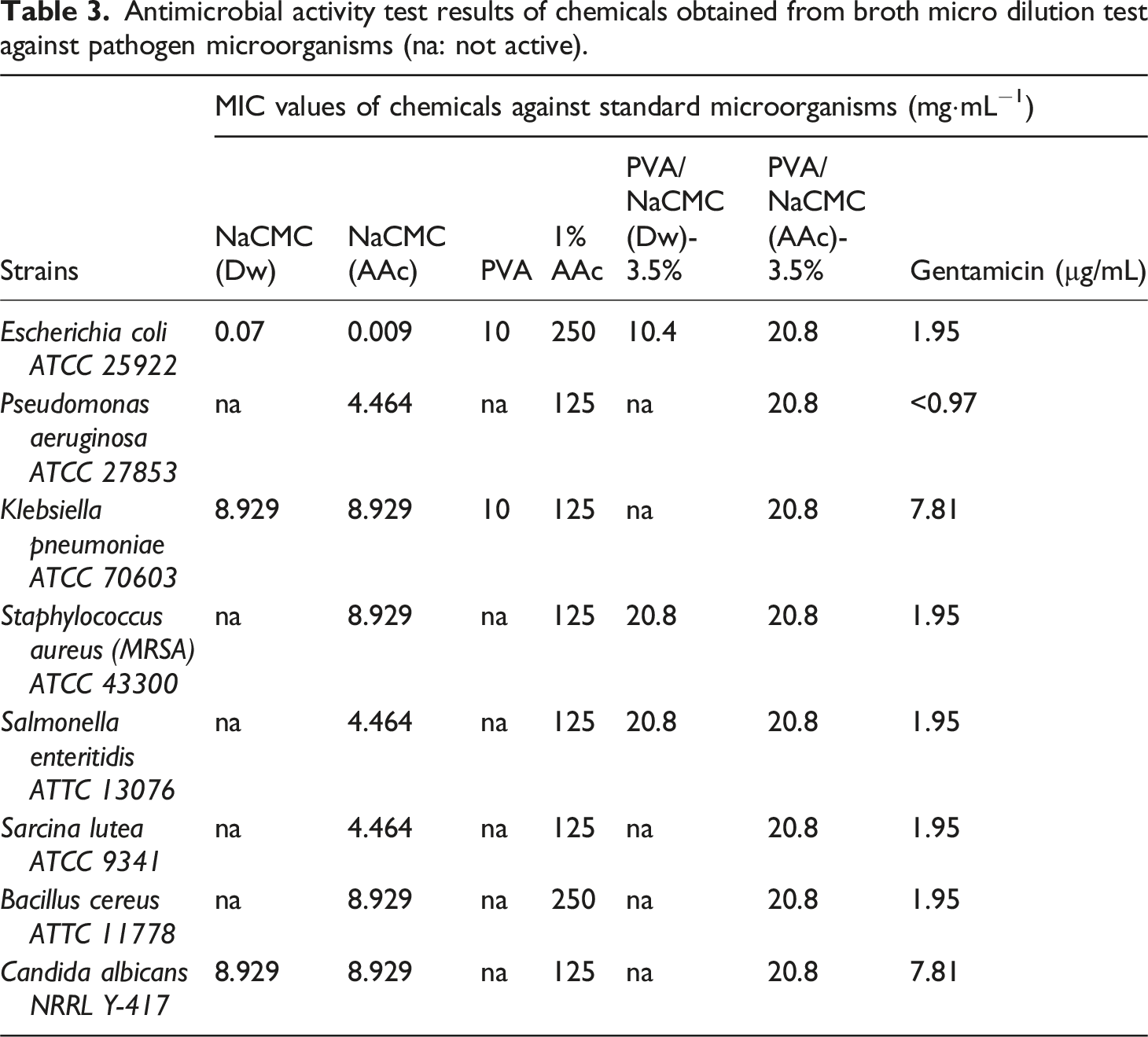
The last formations of hydrogels (PVA/NaCMC(Dw)-3.5% and PVA/NaCMC(AAc)-3.5%) were also subjected to antimicrobial activity tests. The water based compound PVA/NaCMC(Dw)-3.5% was effective only against to E. coli, S. aureus and S. enteritidis. The MIC values were determined as 10.4 and 20.8 mg·mL−1, respectively. When the acetic acid based hydrogel was evaluated, it was seen that this compound was effective for all microorganisms tested in this assay. The MIC value was 20.8 mg·mL−1. These results showed that acetic acid based materials were more effective than water based compound against pathogen microorganisms. Wound site infection is one of the most common infections caused by hospital. It is an important cause of morbidity and mortality. Some studies have shown that Gram-positive microorganisms such as S. aureus (MRSA) and gram-positive microorganisms such as E. coli and P. aeruginosa are most frequently isolated from wound infections.78,79
SEM and TEM Analysis
Figures 4 and 5 present the SEM and TEM analyses of PVA/NaCMC (AAc) and PVA/NaCMC (Dw) hydrogels, highlighting the effect of solvent choice (acetic acid (AAc) or distilled water (Dw)) on the structural properties of NaCMC-based hydrogels. SEM images reveal that both hydrogels demonstrate a successful interaction between PVA and NaCMC, resulting in the formation of a homogeneous network structure. However, the PVA/NaCMC (AAc) hydrogels exhibited a denser and more organized porous network structure,80–82 while the PVA/NaCMC (Dw) hydrogels displayed a less regular structure. The well-defined porous structure in PVA/NaCMC (AAc) hydrogels is attributed to the role of acetic acid as a structuring agent, which enhances crosslinking density and contributes to the formation of more distinct pores. TEM micrograph of the morphologies of the hydrogels (A) PVA/NaCMC (AAc)- 3.5%, (B) PVA/NaCMC (Dw)- 3.5% .
The TEM analysis shown in Figure 5 provides a more detailed view of these structural differences. PVA/NaCMC (AAc) hydrogels exhibited more uniform and evenly distributed pores, while PVA/NaCMC (Dw) hydrogels showed a less homogeneous pore structure, reflecting a lower crosslinking density. These structural differences align with the swelling and contact angle results, emphasizing the significant impact of solvent choice on the water absorption capacity and surface properties of the hydrogels. Consistent with the SEM and TEM observations, the looser structure of PVA/NaCMC (Dw) hydrogels facilitated higher water uptake. Conversely, the denser and more crosslinked structure of PVA/NaCMC (AAc) hydrogels limited water absorption but improved structural stability.
PVA/NaCMC (AAc) hydrogels, with their tightly crosslinked network, provide Structural stability, making them suitable for applications requiring durability. On the other hand, PVA/NaCMC (Dw) hydrogels offer superior swelling performance and surface hydrophilicity due to their higher water absorption capacity. These contrasting properties highlight the critical role of solvent choice in tailoring hydrogel characteristics for biomedical applications.
FTIR Analysis
Figure 6(a) illustrates the FT-IR spectra of PVA, NaCMC, PVA/NaCMC (AAc)-3.5%, and PVA/NaCMC (Dw)-3.5% hydrogels. In the FT-IR spectrum of PVA, the broad band observed around 3288 cm⁻1 corresponds to the stretching vibrations of hydroxyl (-OH) groups,76,83 while a prominent peak at 2908 cm⁻1 indicates the stretching vibrations of aliphatic C-H groups. 83 Additionally, the peaks at 1744 cm⁻1 and 1088 cm⁻1 are attributed to the vibrations of carbonyl (C = O) and C-O bonds, respectively.60,76,83 In the FT-IR spectrum of NaCMC, the broad band in the range of 3200–3400 cm⁻1 represents the stretching vibrations of -OH groups. Asymmetric stretching vibrations of COO⁻ groups are observed at 1600 cm⁻1, while symmetric stretching vibrations are noted at 1400 cm⁻1.61,76 Furthermore, the bands detected between 1020 and 1080 cm⁻1 correspond to the stretching vibrations of C-O bonds.60,83 These spectral features distinctly highlight the characteristic structures of PVA and NaCMC.
The FT-IR spectra of PVA/NaCMC(Dw)-3.5% and PVA/NaCMC(AAc)-3.5% hydrogels, inter-molecular hydrogen bonds between PVA and NaCMC are evidenced by a broad band observed around 3000–3600 cm⁻1, representing the -OH stretching vibrations. 60 The band around 2900 cm⁻1 corresponds to the C-H stretching vibrations. 60 Additionally, the presence of a strong absorption band at 1600 cm⁻1 reflects the asymmetric stretching vibrations of the carboxylate (COO⁻) group, while the absorption band at 1400 cm⁻1 corresponds to the symmetric stretching vibrations of the COO⁻ group. 83 The bands observed around 1020–1080 cm⁻1 indicate the presence of O-H groups, likely associated with increased hydrogen bonding resulting from both crosslinking via cyclic freezing/thawing and the solvent’s crosslinking effect.24,60 The FT-IR spectrum of PVA/NaCMC (AAc)-3.5% hydrogels demonstrates stronger and more intense -OH bands compared to PVA/NaCMC (Dw)-3.5%, signifying a denser crosslinked network facilitated by acetic acid. This denser network enhances the formation of hydrogen bonds. Conversely, the PVA/NaCMC (Dw)-3.5% hydrogels exhibit less intense -OH bands, indicating a looser network structure. These spectral differences clearly illustrate the significant impact of solvent choice on hydrogen bonding and crosslinking density.
PVA/NaCMC (AAc)-3.5% and PVA/NaCMC (Dw)-3.5% hydrogels were not only chosen for FT-IR analysis but also due to their balanced and reliable results in other characterization methods. This NaCMC concentration demonstrated superior structural integrity, surface properties, and water absorption performance in SEM, contact angle, and swelling tests. Specifically, SEM analysis revealed that hydrogels containing 3.5% NaCMC exhibited a more uniform pore structure, minimizing aggregation issues, while achieving optimal performance in contact angle and swelling measurements. For instance, the PVA/NaCMC (Dw)-3.5% hydrogel exhibited higher water absorption (2004 ± 18.9%) and lower contact angle (31.13 ± 1.08°), while PVA/NaCMC (AAc)-3.5% exhibited denser crosslinking with a lower water absorption capacity (1730 ± 15.5%) and a higher contact angle (36.97 ± 1.12°).
X-Ray Diffraction
PVA/NaCMC (AAc)-3.5% and PVA/NaCMC (Dw)-3.5% hydrogels were selected for XRD analysis as they demonstrated the most optimal performance among the hydrogels based on SEM micrographs, contact angle measurements, swelling degree, and antimicrobial activity results. This analysis aimed to validate the structural characteristics and investigate the influence of solvent selection on the crystalline structures of the hydrogels via X-ray diffraction (XRD) patterns. Figure 6(b) presents the XRD diffractograms of these hydrogels. Both hydrogels exhibited a distinct peak at 20°, confirming their semicrystalline structure. In contrast, pure NaCMC is known to exhibit a lower intensity peak at 20.5°.76,83 The higher intensity of the 20° peak in PVA/NaCMC hydrogels is attributed to PVA’s relatively higher crystallinity compared to NaCMC, despite PVA not being fully crystalline.76,83 When acetic acid (AAc) was used as the solvent, the XRD pattern of PVA/NaCMC (AAc)-3.5% displayed a sharper and more pronounced peak at 20°, which is associated with increased crystallinity. This increase in crystallinity can be explained by the formation of more organized and regular regions in the polymer network due to tighter crosslinking. The alignment of NaCMC and PVA chains, facilitated by acetic acid as a structuring agent, contributed to this enhanced crystallinity. 84 In contrast, the XRD pattern of PVA/NaCMC (Dw)-3.5% exhibited a broader peak at 20°, indicating a less crystalline structure. This observation aligns with SEM results, which revealed a more porous and irregular network structure. The use of distilled water as the solvent resulted in higher swelling capacity and less compact crosslinking, leading to reduced crystallinity in these hydrogels.
When XRD results are considered alongside other analyses, the solvent’s influence on the crystallinity and structural properties of PVA/NaCMC hydrogels becomes evident. The higher crystallinity observed in PVA/NaCMC (AAc)-3.5% highlights its superiority in terms of structural integrity, whereas the lower crystallinity of PVA/NaCMC (Dw)-3.5% aligns with its higher water absorption capacity and surface hydrophilicity. These findings demonstrate the role of XRD analysis in complementing other characterization methods, providing a comprehensive understanding of the structural and functional performance of hydrogels.
Discussion
This study systematically evaluated the structural and functional properties of PVA/NaCMC hydrogels prepared using 1% acetic acid (AAc) and distilled water (Dw). The findings demonstrate that solvent choice and NaCMC concentration significantly influence hydrogel performance, particularly in biomedical applications such as wound dressings. Among the tested formulations, the PVA/NaCMC (AAc)-3.5% hydrogel emerged as the optimal candidate due to its superior balance of antimicrobial activity and structural properties (Figures 6(a) and 6(b); Table 2). (a) FT-IR spectrum of PVA, NaCMC, PVA/NaCMC(Dw)-3.5%, PVA/NaCMC(AAc)-3.5%, (b). XRD patterns of PVA/NaCMC (Dw) and PVA/NaCMC (AAc) the hydrogels samples.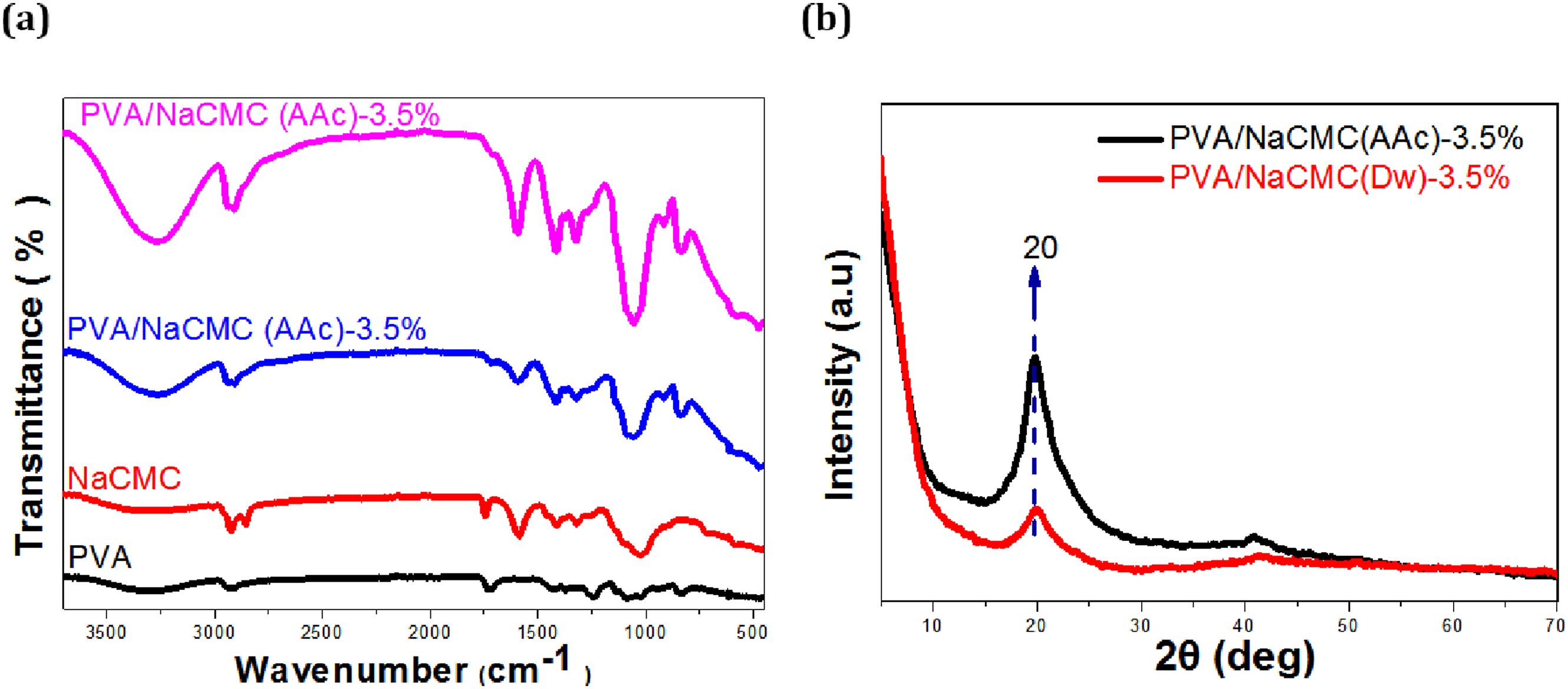
The FT-IR spectrum (Figure 6(a)) provided critical insights into the chemical interactions of the hydrogels. Both PVA/NaCMC (AAc)-3.5% and PVA/NaCMC (Dw)-3.5% hydrogels exhibited broad -OH stretching bands in the 3000–3600 cm⁻1 region, indicative of intermolecular hydrogen bonding between PVA and NaCMC.23,59,60,68,76,83 However, the -OH band intensity was higher in the AAc-based hydrogel, reflecting a denser hydrogen-bonded network. This observation aligns with SEM and XRD results, confirming the role of acetic acid in enhancing crosslinking and structural organization.66,68,76 Furthermore, the COO⁻ stretching vibrations at 1600 cm⁻1 (asymmetric) and 1400 cm⁻1 (symmetric) emphasized the strong interactions between NaCMC’s carboxyl groups and acetic acid.23,60,66 In contrast, the weaker COO⁻ bands in the Dw-based hydrogel suggested a looser network structure.28,83
SEM micrographs (Figure 4) revealed that the PVA/NaCMC (AAc)-3.5% hydrogel exhibited a clean and organized pore structure, indicative of a densely crosslinked network, whereas the PVA/NaCMC (Dw)-3.5% hydrogel displayed a more porous and irregular structure.80,83 TEM analysis (Figure 5) supported these findings, showing a more uniform network in the AAc-based hydrogel compared to the less compact structure of its Dw-based counterpart.
XRD analysis (Figure 6(b)) demonstrated that both hydrogels possessed a semi-crystalline structure with a characteristic peak at 20°, attributed to PVA’s partial crystallinity. However, the PVA/NaCMC (AAc)-3.5% hydrogel exhibited a sharper and more intense peak, reflecting increased crystallinity and crosslinking. Acetic acid acted as a structuring agent, aligning NaCMC and PVA chains into a more ordered network. Conversely, the broader peak in the PVA/NaCMC (Dw)-3.5% hydrogel indicated an amorphous structure and looser crosslinking 29,85.
When the swelling degrees of PVA/NaCMC (Dw)-3.5% and PVA/NaCMC (AAc)-3.5% hydrogels were compared, it was observed that PVA/NaCMC (Dw)-3.5% hydrogels reached a higher swelling degree of 2004.0 ± 18.9%. In contrast, PVA/NaCMC(AAc)-3.5% hydrogel exhibited a lower swelling degree of 1730.0 ± 15.5% compared to PVA/NaCMC(Dw)-3.5%, but its dense cross-linked network contributed to the increased structural stability.28,76,80,83 Contact angle measurements (Figure 3, Table 2) supported these findings. The PVA/NaCMC (Dw)-3.5% hydrogel exhibited a lower contact angle of 31.13 ± 1.08°, indicating higher hydrophilicity, while the PVA/NaCMC (AAc)-3.5% hydrogel had a slightly higher contact angle of 36.97 ± 1.12°.
PVA/NaCMC (AAc)-3.5% hydrogels demonstrated superior antimicrobial activity compared to water-based hydrogels, making them promising candidates for infection-preventive applications. The low pH of acetic acid and its ability to stabilize hydrogen bonds disrupted microbial cell membranes and inhibited metabolic processes.33,34 As shown in Table 3, the PVA/NaCMC (Dw)-3.5% hydrogel was effective against Escherichia coli (E. coli), Staphylococcus aureus (S. aureus), and Salmonella enteritidis (S. enteritidis), with Minimum Inhibitory Concentration (MIC) values of 10.4 mg·mL⁻1 and 20.8 mg·mL⁻1, respectively. In contrast, the PVA/NaCMC (AAc)-3.5% hydrogel exhibited antimicrobial activity against all tested microorganisms, including Gram-positive bacteria (S. aureus), Gram-negative pathogens (E. coli, Pseudomonas aeruginosa), and Candida albicans. The MIC value for the AAc-based hydrogel was consistently 20.8 mg·mL⁻1 for all pathogens, highlighting its broad-spectrum efficacy. 86 Wound infections caused by these pathogens are significant sources of morbidity and mortality. Previous studies,33,45 have shown that Gram-positive organisms (e.g., MRSA) and Gram-negative bacteria (e.g., P. aeruginosa) are common causes of wound infections. Compared to similar studies, the use of acetic acid in this work aligns with findings that organic acids disrupt microbial cell walls and enhance wound healing outcomes.33,35 Additionally, the densely crosslinked network of the PVA/NaCMC (AAc)-3.5% hydrogel contributed to its stability, a feature less pronounced in the PVA/NaCMC (Dw)-3.5% hydrogel, which had a looser network but higher swelling capacity.
The PVA/NaCMC hydrogels developed in our study demonstrated a swelling capacity of 2004 ± 18.9% in a distilled water-based environment, significantly surpassing the performance of CMCNa-based hydrogels (1273%) reported by Nasution et al. 86 In terms of antimicrobial activity, our PVA/NaCMC hydrogels were effective against microorganisms such as E. coli, S. aureus, and S. enteritidis, with minimum inhibitory concentration (MIC) values determined to be 10.4–20.8 mg·mL⁻1 in the distilled water-based system and 20.8 mg·mL⁻1 in the acetic acid-based system. These results represent a notable improvement compared to the limited antimicrobial activity of the CMCNa hydrogels reported by Nasution et al. Additionally, in terms of surface hydrophilicity, the distilled water-based PVA/NaCMC hydrogels exhibited a contact angle of 31.13 ± 1.08°, demonstrating superior hydrophilic properties compared to the contact angles (≥40°) of CMCNa hydrogels in the literature. These findings underscore the significant potential of PVA/NaCMC hydrogels for biomedical applications due to their high swelling capacity, broad-spectrum antimicrobial activity, and enhanced hydrophilicity. The study conducted by Oliveira et al. on propolis-containing hydrogels reported phenol and flavonoid release, resulting in 80%–90% bacterial inhibition against S. aureus and a maximum swelling capacity of 580%. 28 However, in our study, 2004 ± 18.9% swelling capacity and effective antimicrobial activity were achieved without the addition of any biologically active agents, relying solely on solvent differences. Furthermore, the absence of biologically active compounds allowed for cost-effective production and the development of a simpler manufacturing process. These results highlight that high-performance hydrogels can be achieved without the need for biologically active additives, demonstrating the feasibility of developing efficient, cost-effective, and easily producible alternatives. In the study by Bao et al., the enhancement of hydrogel antimicrobial and mechanical properties was achieved through the incorporation of metal nanoparticles (e.g., AgNPs, ZnONPs), crosslinking agents (e.g., citric acid, glutaraldehyde), and plant extracts. 87 While these methods expanded the application areas of the hydrogels, they also introduced challenges related to environmental impact, biocompatibility, and cost. In contrast, our study achieved a swelling capacity of 2004 ± 45.54%, significantly exceeding the maximum 900% swelling capacity reported by Bao et al., by merely optimizing the pH and ionic strength of the solvent environment. Despite the absence of crosslinking agents, our PVA/NaCMC hydrogels demonstrated satisfactory mechanical strength and flexibility. Furthermore, antimicrobial activity was achieved without the addition of metal nanoparticles or plant extracts, minimizing toxicity risks and offering a biocompatible and environmentally friendly alternative.
The results of this study align with and extend findings in the literature regarding the impact of crosslinking and solvent choice on hydrogel properties. Previous studies16,22,24,25,28,59,60,66,76,80,83,86,87 the antimicrobial potential of PVA-based hydrogels but noted challenges in achieving sufficient crosslinking while maintaining water absorption. In contrast, this study overcame these limitations by formulating the PVA/NaCMC (AAc)-3.5% hydrogel, which enhanced structural stability and antimicrobial efficacy while maintaining adequate swelling behavior. The novelty of this work lies in systematically evaluating solvent choice and NaCMC concentration. The hydrophilicity and structural stability balance observed in the PVA/NaCMC (AAc)-3.5% hydrogel underscores the critical role of acetic acid in enhancing crosslinking and structural organization.
This study demonstrates the significant impact of solvent choice on the structural and functional properties of PVA/NaCMC hydrogels. PVA/NaCMC (AAc)-3.5% hydrogels exhibited superior antimicrobial activity, Structural stability, and structural integrity, making them ideal for applications requiring durability and infection prevention. Conversely, PVA/NaCMC (Dw)-3.5% hydrogels, with higher hydrophilicity and water absorption capacity, are better suited for hydration-focused applications. By integrating structural, chemical, and functional analyses, this work emphasizes the importance of solvent choice in designing biomaterials for biomedical applications.
Building on these findings, future research should explore the effects of reducing acetic acid concentrations and investigate alternative biocompatible solvents. Specifically, studying the impacts of low concentrations, such as 1% acetic acid, on hydrogel structural stability and antimicrobial efficacy could lead to optimized formulations with reduced acid content. Additionally, incorporating nanocomposite fillers offers a promising approach to enhancing the Structural durability and functionality of hydrogels. These nanofillers could not only improve the structural integrity of nanocomposite hydrogels but also significantly enhance their antimicrobial properties, providing an effective solution against infections caused by resistant microorganisms. Diversifying functional features is another critical step for advanced wound care applications. Endowing hydrogel wound dressings with analgesic properties could improve patient comfort and support the healing process. Understanding the behavior of these hydrogels under physiological conditions and evaluating their long-term stability and biodegradability are necessary. In conclusion, functionalizing PVA/NaCMC hydrogels with nanocomposites and pharmacological agents offers exciting opportunities to develop multifunctional wound dressing materials with enhanced antimicrobial and therapeutic properties. The use of such hydrogels in advanced wound care applications could provide new solutions that optimize infection management, pain control, and healing rates, ultimately improving patients’ quality of life.
Footnotes
Author contributions
Project Administration: [Ahmet OKUDAN]; Experimental Work: [Buse ÇOPUR, Şeyda BAYRAKTAR]; Antimicrobial Studies and Analysis: [Ahmet UYSAL, Erdoğan GÜNEŞ]; Writing-Original Draft: [Buse ÇOPUR, Şeyda BAYRAKTAR, Ahmet OKUDAN, Ahmet UYSAL, Erdoğan GÜNEŞ]; Writing-Review & Editing: [Buse ÇOPUR, Ahmet OKUDAN].
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Selcuk University Research Foundation (project number: BAP-20401100).
