Abstract
Due to their high strength and exceptional thermal resistance, boron nitride (BN) nanostructures are a promising new type of reinforcement for medical applications. Among these, cubic BN (c-BN) nanoparticles are crystalline-shaped structural, combining extremely high hardness and thermal stability, which make them highly effective reinforcements in polymers. Their incorporation enhances material performance by increasing density, load-bearing capacity, and wear resistance. This study examines the influences of c-BN nanoparticle addition on the mechanical and wear characteristics of PMMA/c-BN biocomposites. c-BN nanoparticles (NPs) were added to the polymer matrix at different concentrations of 0, 0.5, 1, 2, 3, and 5 wt%. Measurements of bulk density, Vickers microhardness, ultimate compressive stress, yield stress, and wear resistance revealed that the properties of the fabricated composites were significantly improved by the increased concentration of c-BN nanoparticles in the matrix. Notably, the biocomposites containing a 5 wt% c-BN exhibited the highest bulk density, hardness values, and surface roughness of 1.221 ± 0.002 g/cm3, 79.5 ± 1.6 HV, and 125.52 µm, respectively; while the porosity decreased to 0.16% compared to other produced composites. In particular, the biocomposite produced at 3 wt% c-BN displayed the highest ultimate compressive stress, and yield stress: 215 ± 6.17 MPa, and 101.1 ± 2.2 MPa, respectively, with improvements of 99.07%, and 27.85% compared with the PMMA matrix. Additionally, the wear track profiles also showed improved wear resistance; this was confirmed by 3D surface roughness topography and experimental results for SEM images of worn surfaces. Due to their superior mechanical and wear resistance, PMMA biocomposites with c-BN NPs make excellent secondary reinforcement for a variety of medical applications, including dental prostheses.
Introduction
Polymethyl Methacrylate (PMMA) has emerged as a versatile material in various dental applications due to its favorable characteristics, including biocompatibility, esthetics, ease of manipulation, ability to be molded into any shape or size needed for dental restoration, and cost-effectiveness.1–3 PMMA is preferred for denture fabrication due to its excellent biocompatibility and esthetic qualities.4–6 The material can be easily customized to match the patient’s oral anatomy and desired appearance. 7 Moreover, PMMA’s lightweight nature and minimal water absorption contribute to patient comfort and durability. 8 PMMA’s favorable biocompatibility, non-toxic nature, and minimal allergenic potential support its safe use in oral applications which include creating crowns and bridges that look natural and blend in with the rest of the teeth, also used to make dentures and orthodontic appliances such as retainers and aligners, and other dental prosthetics. 9 Also, PMMA can be used in medical implants such as artificial joints and open heart operations (heart valves). PMMA exhibits relatively low tensile strength and fracture toughness, rendering it susceptible to chipping or fracturing under the masticatory forces present in the oral environment.10–12 PMMA’s inherent brittleness and lack of elasticity can lead to inadequate shock absorption, potentially reducing the material’s longevity. 13 Advancements in PMMA-based composites and hybrid materials hold promise for addressing its current limitations. PMMA-based composites have garnered substantial interest in dental applications due to their potential to enhance the mechanical and esthetic characteristics of PMMA while maintaining its favorable biocompatibility. 14 Incorporating ceramic fillers, such as zirconia,15,16 alumina, 17 titanium oxide,18,19 ceramic-metal composites,20,21 titanium (Ti)-based alloys, 22 or graphene, 23 into PMMA matrices imparts improved strength, fracture resistance, and wear characteristics, addressing some of the inherent limitations of pure PMMA. This combination of PMMA and ceramic materials augments dental’s structural integrity, making them a promising avenue for advancing dental materials. Mana Alqahtani 24 investigated the physical and mechanical characteristics of Self-Cured PMMA/hexagonal boron nitride (h-BN) composites at various h-BN concentrations of 0 wt%, 0.5 wt%, 1 wt%, 3 wt%, and 5 wt% h-BN. The authors discovered that as the concentration of h-BN in a polymer matrix increases, the hardness values and flexural strength for manufactured composite specimens increase. The results also provide substantial support for the idea that dental materials with PMMA content and h-BN nanoparticles perform better than those with conventional materials. Also, Alhareb and Ahmad. 25 worked on the mechanical properties of hybrid (Al2O3/ZrO2) nanocomposites into PMMA denture base. The results show the integration of Al2O3/ZrO2 into the PMMA matrix leads to improved flexural strength, fracture toughness, tensile stress, and tensile modulus of this denture material. The results showed that the highest tensile stress, tensile modulus, flexural modulus, and flexural strength are obtained at PMMA/5 wt% of (80/20; Al2O3/ZrO2) of 46.98 MPa, 1.75 GPa, 2.76 GPa, and 100.01 MPa, respectively. Another study found that increasing the concentration of ZrO2 of 0 wt%, 5 wt% and 10 wt% added to the PMMA polymer matrix leads to improved microhardness and wear resistance for PMMA/ZrO2 composites. Also, the highest microhardness and wear resistance is obtained at 10 wt% ZrO2 compared with the 0 wt% ZrO2. 26
Cubic boron nitride (c-BN) demonstrates promising biocompatibility for medical applications.27–31 Renowned for its remarkable hardness resembling that of a diamond, c-BN offers a compelling option for crafting durable dental tools and precision restorative elements.32,33 Additionally, c-BN’s chemical stability and low reactivity with oral tissues suggest it could be used in direct contact with teeth and gums without triggering adverse reactions.34,35 Despite the promising properties of c-BN, no study has been conducted to study the influence of c-BN nano ceramic addition on the characteristics of PMMA/nano c-BN composites. Consequently, the present study focused on producing the heat-cure acrylic resins as a matrix and reinforced with nano c-BN reinforcement using 0.5, 1, 2, 3, and 5 wt% concentrations of c-BN. The produced PMMA and their PMMA/nano c-BN biocomposites were examined by measuring the density, hardness, and compressive properties. Furthermore, the wear properties in terms of wear performance and mechanisms were also investigated.
Materials and methodology
Materials
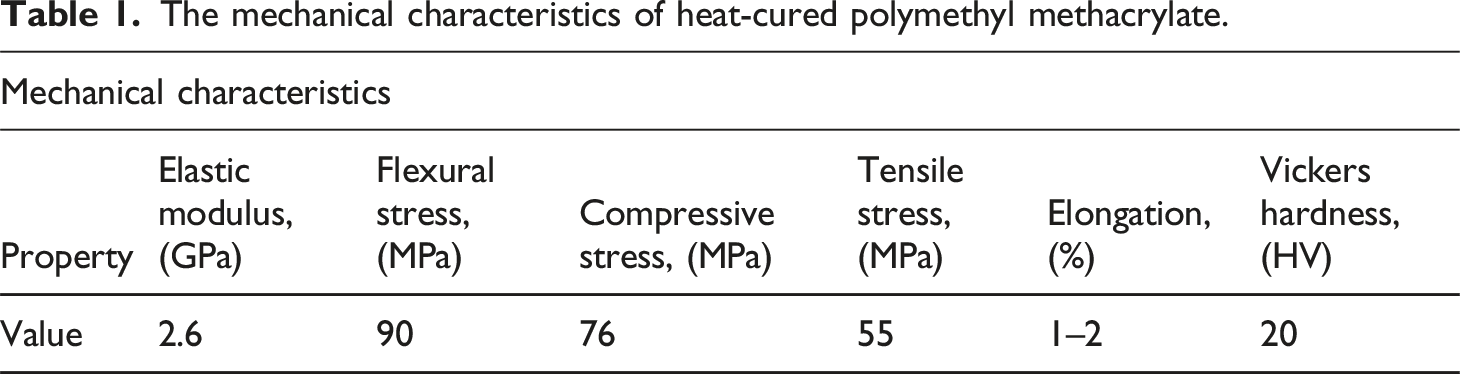
The mechanical characteristics of heat-cured polymethyl methacrylate.
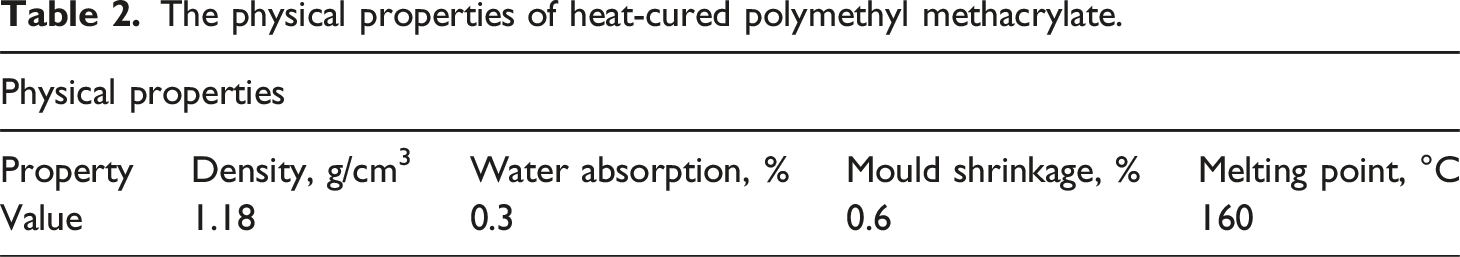
The physical properties of heat-cured polymethyl methacrylate.
Fabrication of PMMA/c-BN composites
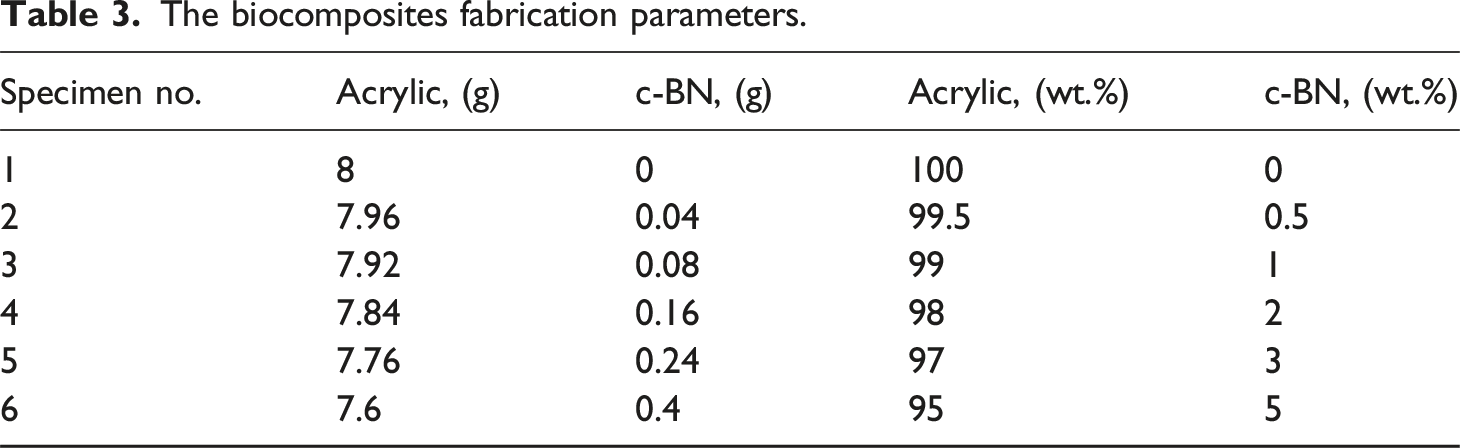
The biocomposites fabrication parameters.
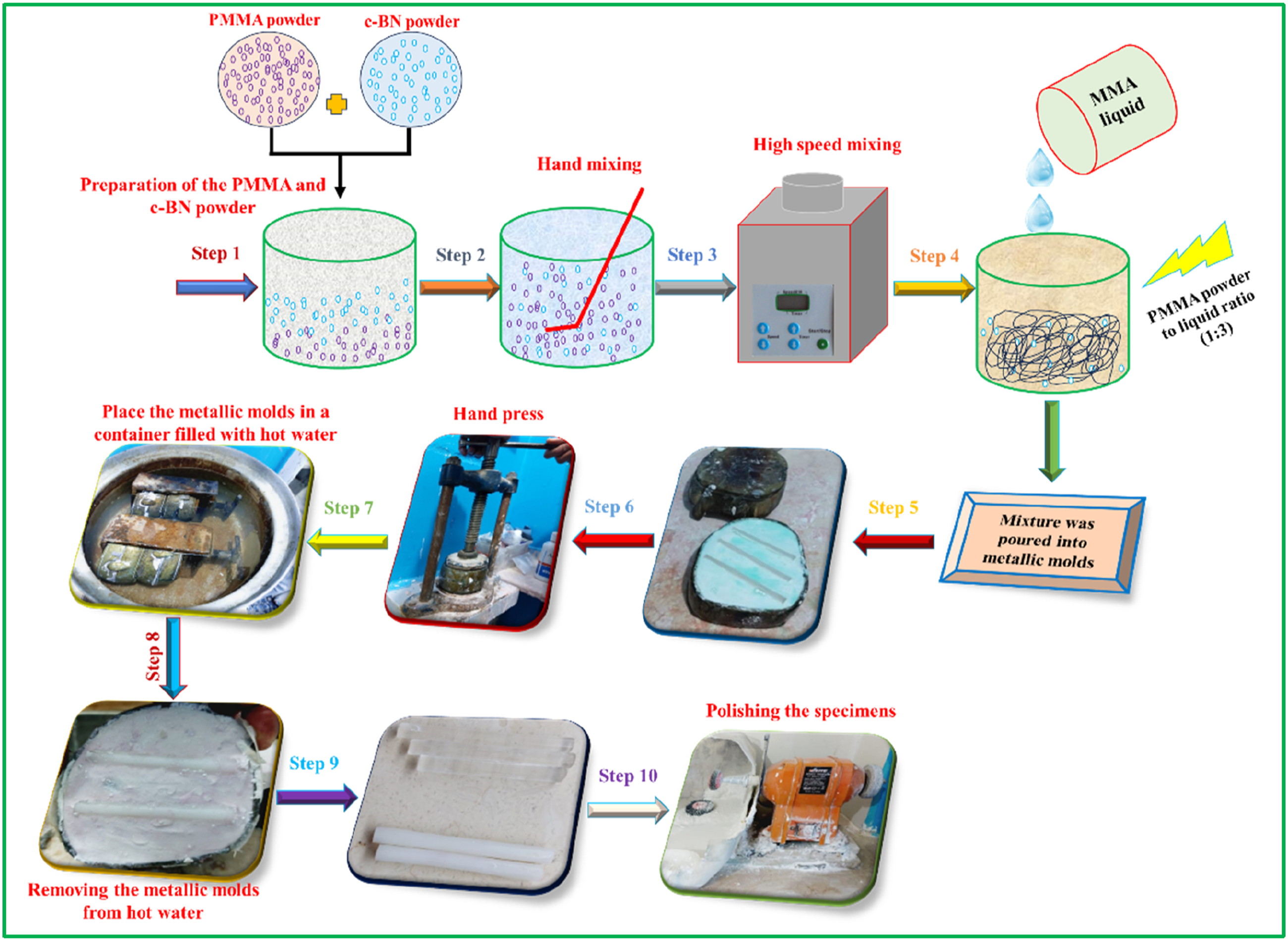
Summarizes the experimental steps to fabricate the PMMA/c-BN composite specimens.
Material characterization
SEM images of the manufactured specimens were captured using a QUANTA Field Emission Gun 250. A sputter coater was utilized to pre-coat the surface of tested specimens with conductive gold (K550X sputter coater, EMITECH, England). A D8 diffractometer was used to obtain X-ray diffraction patterns (XRD) (Bruker, Germany) of samples. At room temperature, measurements were taken with Cu-target Kα radiation (k = 0.154 nm). The Origin software program (version 2022) was used to analyze the XRD pattern, then the Scherrer equation (1) was used to calculate grain size, whereas equation (2) was used to calculate crystallinity. Meanwhile, the Archimedes rule was used to determine the bulk density and porosity of the producing composite specimens.

Then, all manufactured PMMA/ c-BN composites were tested for microhardness on a Vickers Microhardness Tester Machine (BUEHLER 1600-6300 Microhardness Tester, Guiseley, England) with a 100 g load and 10 s of dwell period. Seven points were measured for each sample, where the points were evenly spaced around a circle and at least 0.5 mm away from neighboring indentations to obtain the Vickers microhardness values. 37
Furthermore, the fabricated samples with different c-BN concentrations were subjected to compression tests utilizing an Instron 4208 tensile testing machine with a 0.001 s−1 quasi-static strain rate. All test specimens were cut and prepared according to ASTM D695. Also, three samples were prepared for each concentration, and the average values of the three samples were taken to determine the mechanical properties of PMMA and its composites.
To assess the wear behavior for manufactured composite samples from PMMA/c-BN, the premolar teeth were selected from humans who were free of fractures, cavities, or worn cusps. These teeth had recently been extracted for orthodontic treatment plans and were stored in a 0.1% thymol solution. They were then placed into a mold made of self-cured acrylic resin from Acrostone Co., located in Cairo, Egypt. The mold was inserted into a copper specimen holder with a diameter of 15 mm. The dental surveyor was used to guide the insertion of the natural tooth surfaces, ensuring proper alignment with the long axis of the sample holder.38,39 To prepare for the wear test, the buccal surface of each tooth sample was wet abraded and finished using 1000, 2500, and 4000 grit abrasive paper until reaching a depth of 0.5 mm. This resulted in a flat zone measuring approximately 2–3 mm for loading during the wear test.39,40 Subsequently, the newly developed four stations multimodal Dual-axis ROBOTA chewing simulator integrated with thermo-cyclic protocol operated on a servomotor (Model ACH-09,075DC-T, AD-TECH Technology Co., LTD, Berlin, Germany) was utilized to perform the two-body wear test.
41
The apparatus enables the simulation of simultaneous horizontal and vertical motions under thermodynamic conditions in temperatures from 5 to 55°C at a 10 s dwell period. The chewing simulator has four chambers, each of which has a lower plastic antagonist holder and an above portion that serves as a sample holder (see Figure 2(a)). Samples were placed within the metal container that can be seen in the chewing simulator in Figure 2(b) upper portion. The buccal surface of premolar teeth was employed in the bottom plastic holder of the device operating as an antagonist Figure 2(c) to examine all the samples under standard circumstances. Also, Figure 2(c) shows how the PMMA specimens were placed on the upper sample holder such that they were in direct touch with the tooth surfaces. A weight of 5 kg was applied, which is equivalent to a chewing force of 49 N. To simulate the clinical activity for 3 months, the samples were put through 37,500 cycles. The specimens were taken out of the holder after 37,500 cycles, washed under running water, and then cleaned in an ultrasonic cleaner for 2 min to remove any debris that had been rubbed off the surface before measurement. Additionally, the wear characteristic of the PMMA and its composites was assessed utilizing weight loss. To demonstrate the varying in weight before and after the wear test, samples were weighed in a Sartorius, Biopharmaceutical, and Laboratories electronic analytical balance at 0.001 gm accuracy. The values of all the installed discs and antagonist specimens were precisely measured thanks to the completely automated calibration system and the micro weighing scale that were both included in this balance. Before being weighed, each mounted specimen was carefully cleaned and dried, also all data was computed and tabulated. (a) Homemade chewing simulator, (b) specimens fixer, and (c) natural tooth fixer.
41

Additionally, the surface roughness of the PMMA matrix and its composites, produced at varying c-BN concentrations, was analyzed using Gwyddion software to determine the surface roughness parameters.
Results and discussion
XRD and SEM results of the c-BN nanoparticles
Figure 3 displays the results for XRD patterns and the SEM image of the c-BN nanoparticles, which encompass the initial materials. The five brightest peaks in the XRD measurements are located at Bragg angles 43°, 50°, 74°, and 89.9°, corresponding to miller indices of (111), (200), (220), and (311), respectively, are pure crystalline c-BN nanoparticles, as shown in Figure 3(a). At the same time, the XRD pattern shows no peaks that correspond to the h-BN phase, indicating that the h-BN content is too small to be detected by XRD measurement. Also, Chen et al.
42
and Yan et al.
43
demonstrated that the XRD results for pristine c-BN powder reveal a cubic phase according to JCPDS No. 35-1365, which this finding is in agreement with our XRD results. Additionally, the calculated crystalline size for c-BN nanopowder is up to about 12 nm, as evidenced by the XRD analysis results displayed in Figure 3. In their investigation of the conversion of BN from cubic to hexagonal under 1-atm helium, Cahill et al.
44
discovered that the XRD result for c-BN shows two peaks at 43.3° (111) and 74.1° (220), which correspond to the cubic phase of BN (PDF 00–035-1365). This is most likely due to preferred orientation, as the large particles settled onto the zero-background holder during preparation. Furthermore, Figure 3(b) illustrates the SEM image of the row c-BN nanoparticles, which indicates the regular shaps with varying particle sizes. (a) XRD patterns and (b) SEM image of raw c-BN NPs.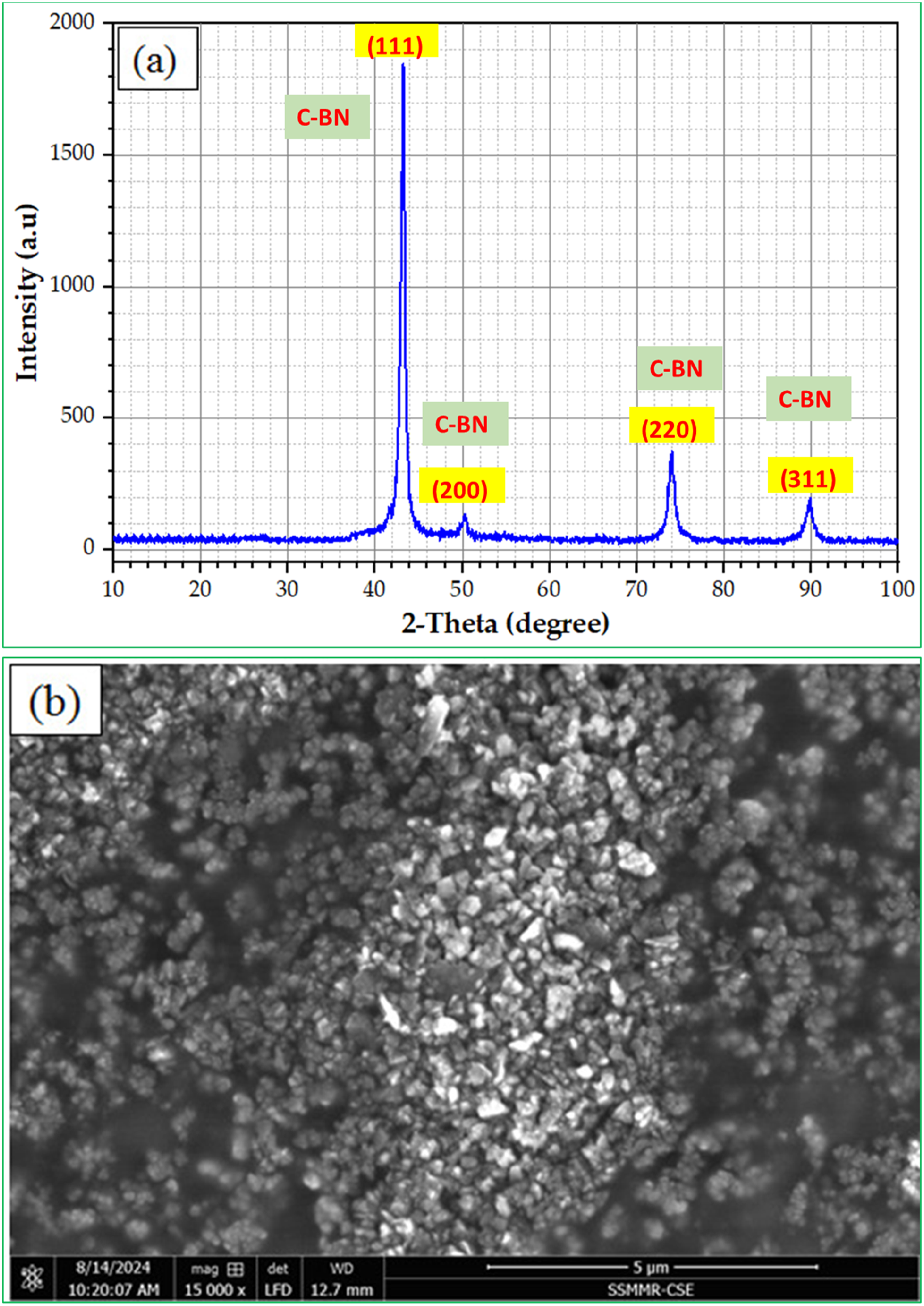
Density measurement for modified composites
The density or weight of the material used to manufacture dentures has a major impact on their performance, durability, and comfort, ensuring a comfortable and secure fit for the patient. As a result, high-density materials are more durable, provide a better fit, comfort, and stability for surrounding teeth and gums, and help prevent further damage or tooth decay, while low-density materials are more susceptible to damage and wear over time. Figure 4 illustrates the influence of varying concentrations of nano-c-BN on bulk and porosity for modified composites. It is clear from Figure 4 that the bulk density increased with increasing c-BN content in modified composites, while porosity decreased. The maximum bulk density value gains are obtained at 5 wt% c-BN (1.221 ± 0.002 g/cm3), whilst the lowest values of bulk density are obtained at 0 wt% c-BN (1.171 ± 0.003 g/cm3). On the other hand, the porosity decreased with increasing c-BN content in modified composites. The highest porosity is obtained at 0 wt% c-BN (1.59664), whilst the lowest values of porosity are obtained at 5 wt% c-BN (0.16353). Hence, the reinforcement effect of c-BN NPs within the PMMA matrix is responsible for the observed increase in bulk density and decrease in porosity with higher c-BN content. The incorporation of c-BN particles in the PMMA matrix during the fabrication process reduces void spaces, resulting in denser biocomposites; the uniform dispersion of nano c-BN enhances packing efficiency, thereby decreasing porosity. At the same time, the high porosity in the 0 wt% c-BN sample is likely due to the absence of filler particles, which results in less efficient packing and more void spaces within the pure PMMA matrix; this effect is diminished as c-BN content increases, improving structural integrity. Lin et al
45
studied the influence of BN (0–30 wt% BN) addition on the apparent porosity of Si3N4-BN composites and reported that the apparent porosity decreased with increasing the BN content in these composites. Similarly, Wang et al.
46
reported that the porosity increased from 53 to 60.6% with increasing starting PMMA content from 10 to 40 wt% in casting BN/Si3N4 ceramics, while the density decreased due to a large variation in the PMMA microbeads. The physical properties of PMMA and its composites.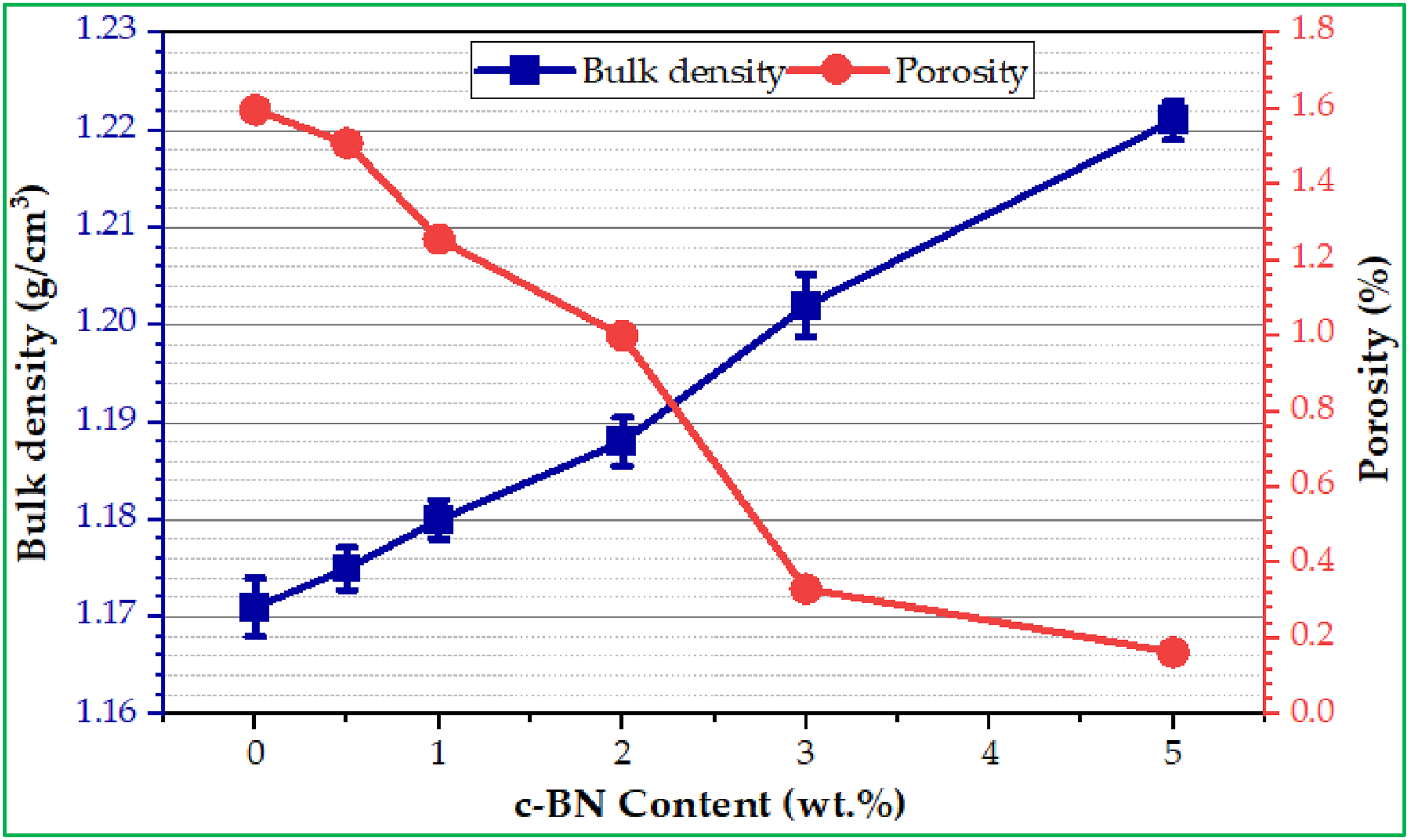
Effect of modified composites on microstructure
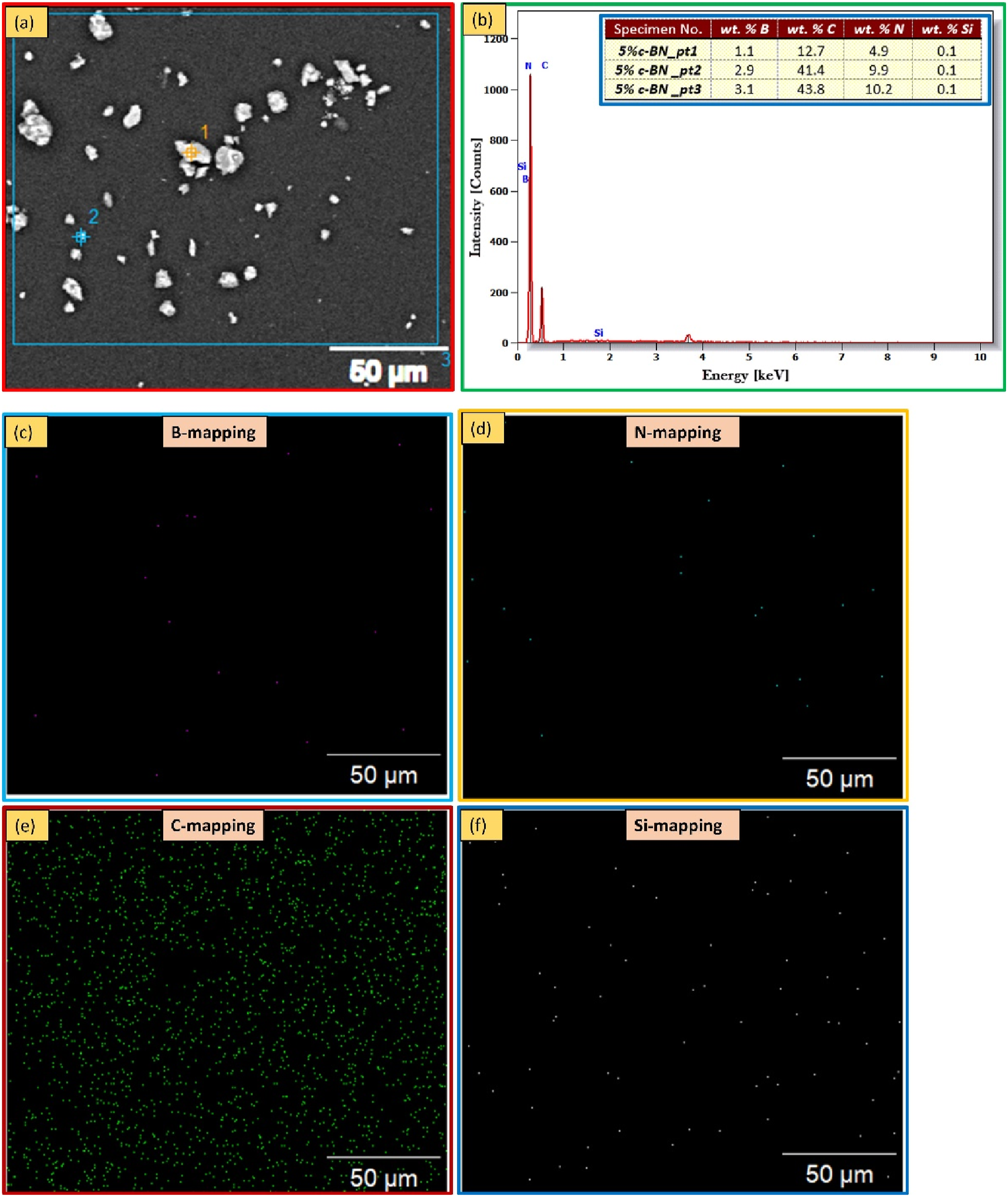
Figure 5(a) displays the SEM micrographs of the initial PMMA, as well as the nanocomposites containing different weight percentages of c-BN: 0.5 wt% (Figure 5(b)), 1 wt% (Figure 5(c)), 2 wt% (Figure 5(d)), 3 wt% (Figure 5(e)), and 5 wt% (Figure 5(f)). The absence of porosity defects (decrease in porosity) in the samples at a macro level suggests that the production procedure effectively produces sound PMMA biocomposites across various c-BN weight percentages, ranging from 0 wt% to 5 wt%. However, an increase in macrosized particles can be observed as the c-BN weight percentage increases which macro-sized originate from the c-BN particles. At a micro level, no defects were observed, except for nanoparticles in the sample containing 5 wt% c-BN. Consequently, The distribution of the c-BN NPs in the PMMA biocomposites reinforced with varying concentrations of c-BN up to 3 wt% is nearly homogeneous, while the sample containing 5 wt% c-BN appeared with few agglomerated particles (Figure 5(f)). EDS analysis for specimen 5 wt%c-BN confirms that these large-size particles are primarily composed of c-BN, while also confirming the presence of c-BN NPs within the acrylic matrix (as shown in Figure 6(a) and (b)). Additionally, both EDS charts display a peak carbon signal, which may be attributed to interference from the instrument or the polymeric material. Also, the EPMA map is used to distribute the elements of the B, N, C, and Si across the interface of PMMA and c-BN for a manufactured composite sample at PMMA/ 5 wt% c-BN is depicted in Figure 6(c)–(f). Figure 6(c) and (d) indicates that B and N diffused into c-BN while C and Si diffused into PMMA (Figure 6(e) and (f)). SEM images of microstructure for fabricated specimens of PMMA/c-BN composites; (a) 0 wt% c-BN,
41
(b) 0.5 wt% c-BN, (c) 1 wt% c-BN, (d) 2 wt% c-BN, (e) 3 wt% c-BN, and (f) 5 wt% c-BN. The EDS spectrum was taken at 5 wt% c-BN is shown in (a and b), and corresponding B, N, C, and Si elemental mapping is shown in (c to f), respectively.
Effect of modified composites on microhardness value
Hardness testing of dentures plays a vital role in their effectiveness and durability. In the case of dentures or dental restorations, the hardness value of a biocomposite is used to determine its ability to resist wear, fracture, and cracking, and to maintain its shape over time. Also, if the denture material is too soft (low durometer), it will wear down quickly or deform under pressure. Conversely, denture materials that are too hard can cause discomfort or irritation in the mouth. Figure 7 depicts the effect of introducing (nano-c-BN) particles with varying weight percentages on the hardness values of the modified composites (PMMA/c-BN). Each sample underwent seven tests on various regions, all of which were conducted simultaneously and in the middle of the sample rather than on the periphery, finally, the average measured value was taken. It can be seen that the microhardness values of modified composite samples (PMMA/c-BN) increase with the increase of the percentage of nanoparticles (c-BN), reflecting that the high hardness and brittleness of c-BN particles compared to low-hardness PMMA matrix materials. Moreover, the high wettability between the matrix and these particles (c-BN) causes the surface of the modified composites to become a layered composite harder, resulting in restricted movement of the matrix and low resistance to stress applied to it. Therefore, as the wt.% of c-BN content in modified biocomposites increases, the microhardness values increase.
24
Consequently, the microhardness value increased from (24.3 ± 1.9 HV) for the (without c-BN) sample to a higher hardness value (79.5 ± 1.6 HV) for the composite samples (PMMA - 5 wt% c-BN). The highest hardness values are obtained at 5 wt% c-BN, while the lowest hardness values are obtained at 0 wt% c-BN, as shown in Figure 7. Microhardness values for fabricated specimens of PMMA/c-BN biocomposites.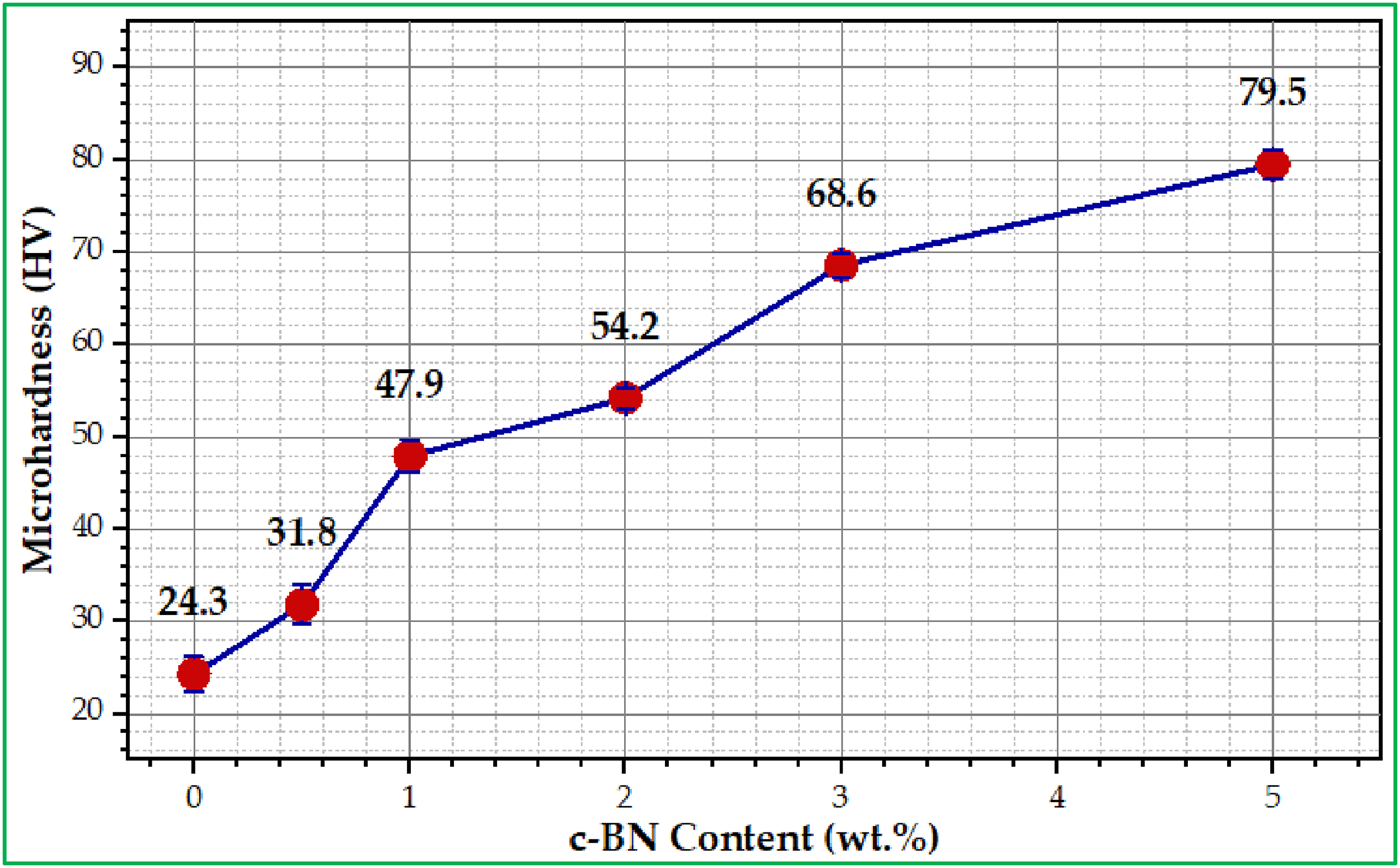
Effect of modified composites on mechanical behavior
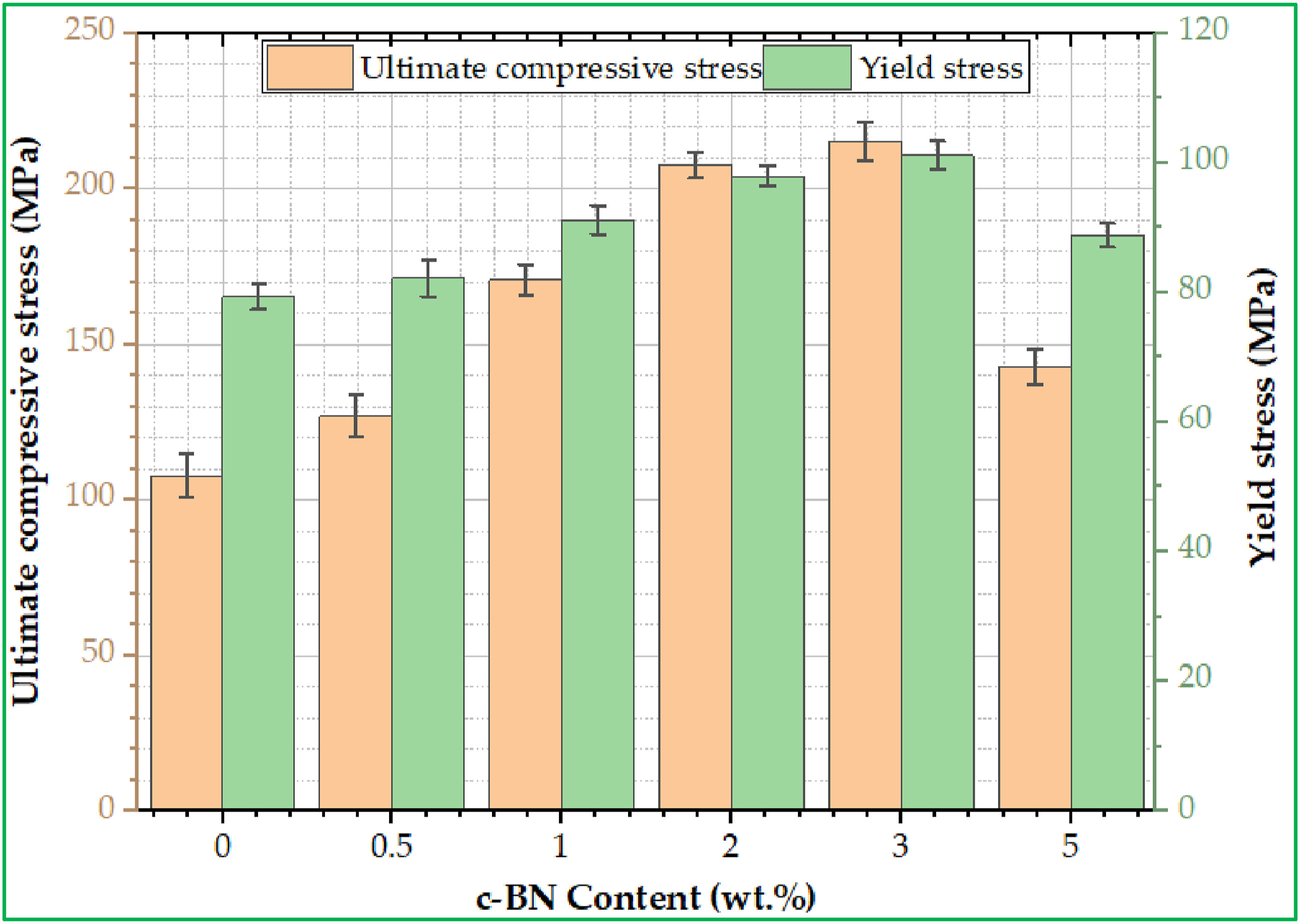
The lifetime and toughness of materials used in manufacturing dental prosthetics such as teeth and restorations can be highly impacted by the mechanical behavior (ultimate compressive stress (UCS), and yield stress (YS)) of the materials used. For example, the materials used in the fabrication of dental prosthetics must be powerful enough (high UCS and YS) to withstand the high forces of chewing and biting inside the mouth during eating. Thus, low values of UCS and YS for the material selected may result in damaged teeth and highly expensive dental to treat the teeth over time, whereas the high UCS and yield stress lead to cause issues such as cracks and fractures in the teeth. Besides, when choosing the materials from which teeth are made the appropriate regard for many factors such as the oral health status of the patient, the patients age, and lifestyle habits is necessary. These factors help when selecting a dental prosthetic material with optimal UCS and yield stress to ensure its high performance, effectiveness, and sustainability for a long time. Figures 8 and 9, demonstrate the impact of adding c-BN particles with varying concentrations of 0, 0.5, 1, 2, 3, and 5 wt% c-BN on both the mechanical characteristics of PMMA/c-BN biocomposites for dentures base materials. The stress-strain curves displayed in Figure 8 depict the results of the ultimate compressive stress test conducted on the samples. It’s clear that as the concentration of c-BN content within PMMA/c-BN biocomposites increases up to 3 wt% c-BN, the UCS and YS values increase while decreasing at 5 wt% c-BN, as shown in Figure 9. This is due to the homogeneity of c-BN particle distribution and their high wettability with PMMA. Additionally, the increase in UCS and YS can be attributed to the fact that the c-BN particles exhibit high UCS and YS compared to PMMA, as shown in Figure 9. According to Figure, the highest UCS and YS were 215 ± 6.17 MPa, and 101.1 ± 2.2 MPa (for 3 wt% c-BN), respectively. These values were approximately 99.07% higher than PMMA without the nanoparticles for UCS and 27.85% higher for YS. It is obvious that adding more c-BN NPs to modified composites increased the UCS and YS. Thus, the interphase that exists between inorganic nanoparticles and the organic polymer matrix has a significant effect on the strength of polymeric nanocomposites. Conversely, increasing the c-BN concentration in the PMMA matrix from 3 to 5 wt% promotes c-BN particle aggregation (Figure 5(f)) and degrades the mechanical characteristics of UCS and YS. Agglomeration in the 5 wt% c-BN sample causes stress concentration points, weak interfaces, and microstructural flaws that serve as starting points for crack propagation and failure under applied loads, which is why this degradation has occurred. Measuring the mechanical behavior of a specific ceramic material not only provides information about its ability to resist crack growth but also affects its thermal shock resistance,
47
as well as important for the usefulness of materials in the oral cavity.48,49 Furthermore, studying the fracture behavior mechanisms of brittle materials using stress-strain data can often provide valuable information on the role of crystalline phases in the matrix. Stress-strain curve for fabricated specimens of PMMA/c-BN biocomposites. Mechanical behavior for fabricated specimens of PMMA/c-BN biocomposites.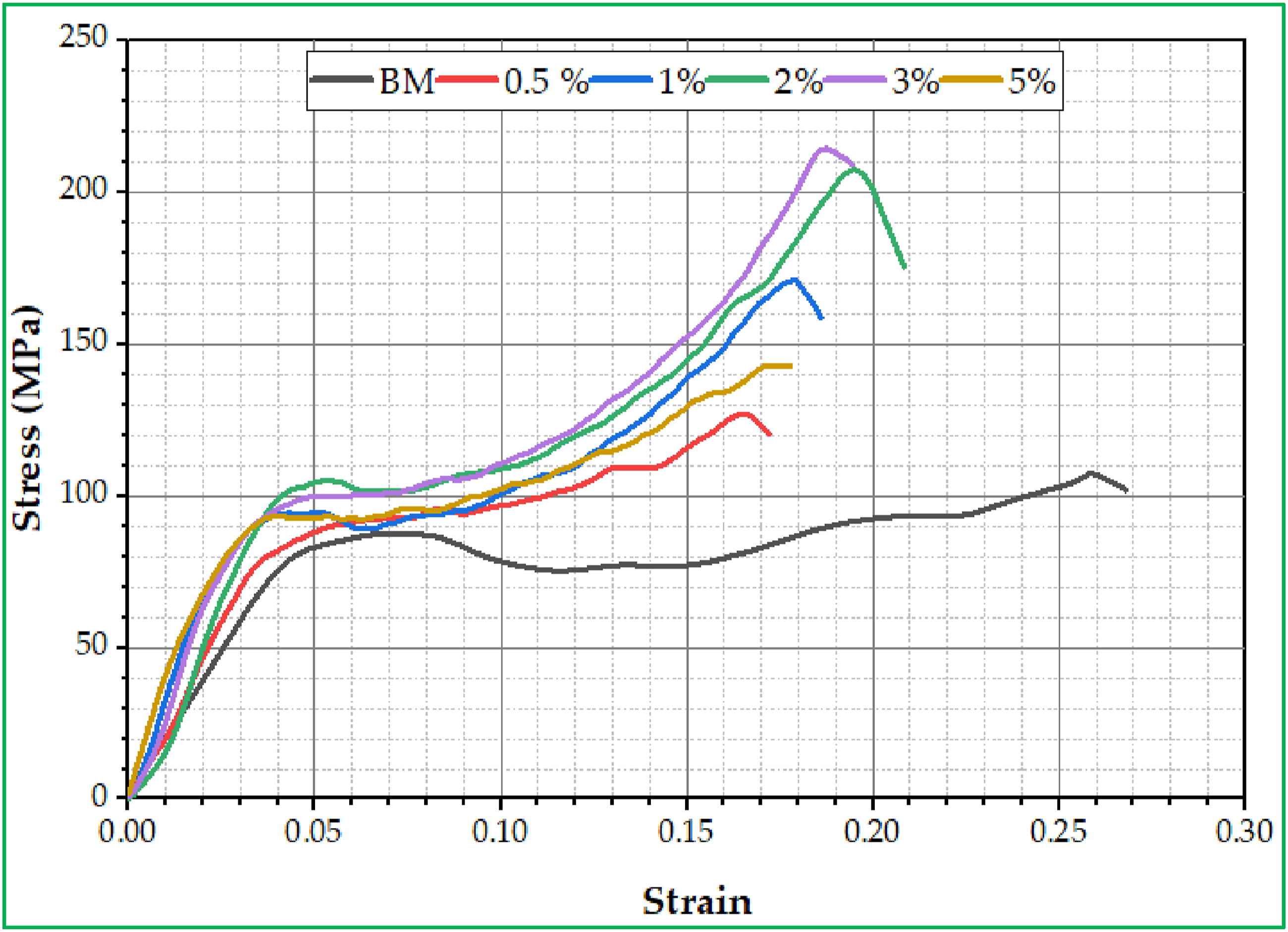
Effect of c-BN loading on wear loss of PMMA composites
In general, the study of the wear characteristic of biocomposites used in medical sectors is of very importance, particularly materials used in the sectors of dentistry because it helps in evaluating the force and lifetime of dental materials and restorations. Consequentially, the wear test offers more information regarding the ability of materials to withstand abrasion, and deterioration as time passes,
50
through the simulation of chewing forces inside the mouth. Also, various factors during the wear test are assessed; surface roughness (surface condition), quantity of material loss, and changes in the physical properties of materials used. For instance, during the wear test, if the materials used showed low wear resistance or deterioration, this refers to the materials not being suitable for use in high-stress areas region inside the mouth, such as molars.51,52 Figure 10 shows the weight loss of PMMA/c-BN biocomposites. The findings of the wear test and weight loss both show that the inclusion of c-BN nanoparticles significantly improved the wear resistance and that the resistance to wear increased with increasing the concentration of c-BN nanoparticles in modified biocomposites, except for the PMMA-5 wt% c-BN the resistance to wear decreased. The matrix modification was strengthened as a result of the metallurgical changes brought about by the addition of ceramic particles. The two main factors that contribute most to the wear resistance of the PMMA/c-BN made by powder metallurgy are (1) the modified matrix’s tiny grain size and (2) the composites’ fabrication’s strengthening because of the fine dispersion of c-BN particles. In addition, the primary wear mechanisms of polymeric materials are adhesion and abrasion wear. Although ceramic materials and polymers tend not to adhere to stick together, a transferred material film can often form on the ceramic surface, which is the hardest material, thereby adhesion stronger.
53
Figure 10 also demonstrates that PMMA-3 wt% c-BN composites exhibited the highest wear resistance (lowest weight loss) in comparison to other combinations under the tested conditions. The enhancement noticed in this investigation can be ascribed to the elevated degree of hardness displayed by the c-BN nanoparticles relative to the surrounding resin matrix. This attribute confers upon them a heightened resistance to wear and makes them less susceptible to abrasion by an antagonist. Conversely, pure PMMA recorded the highest weight loss compared to other combinations. Weight loss for fabricated specimens of PMMA/c-BN composites.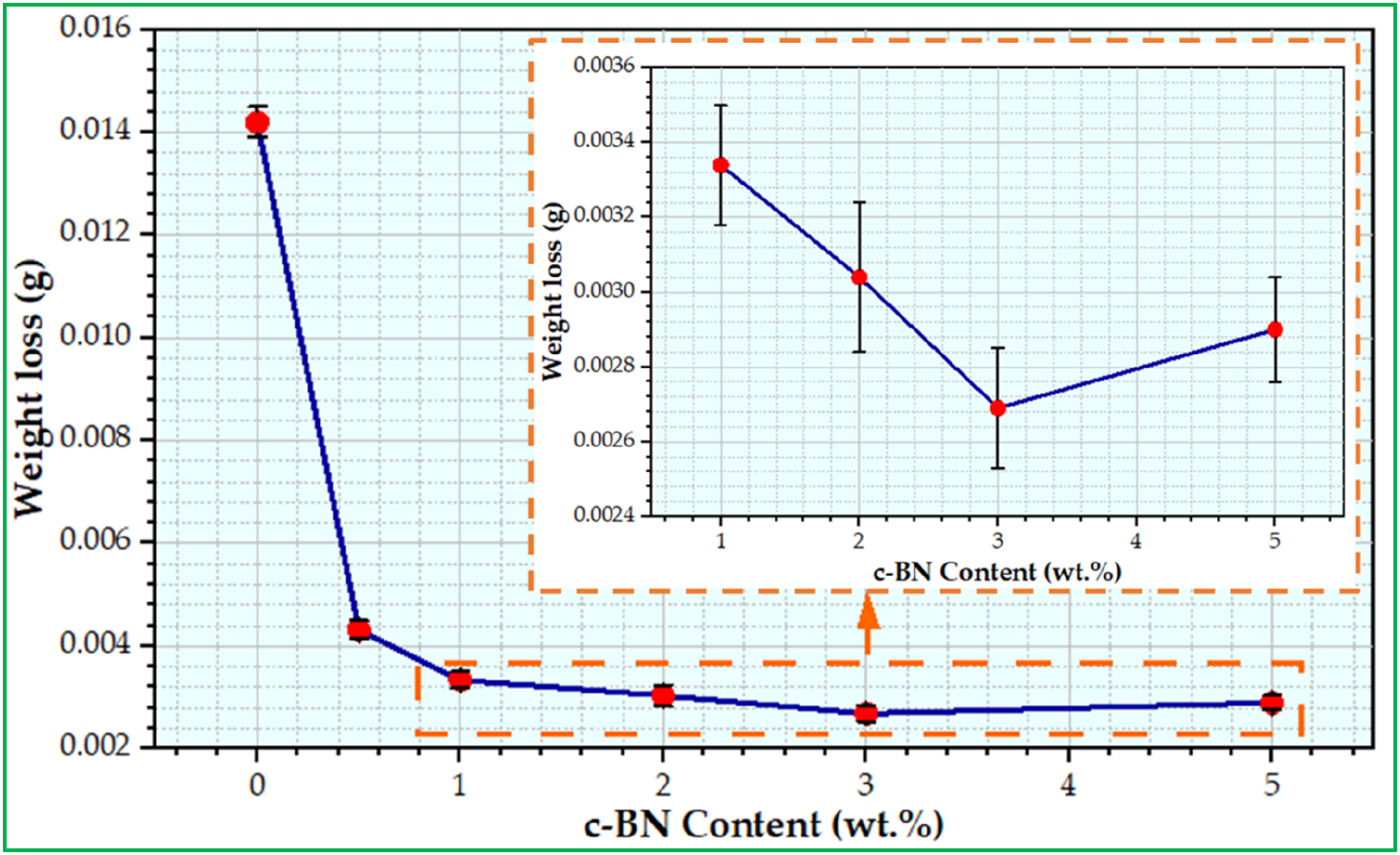
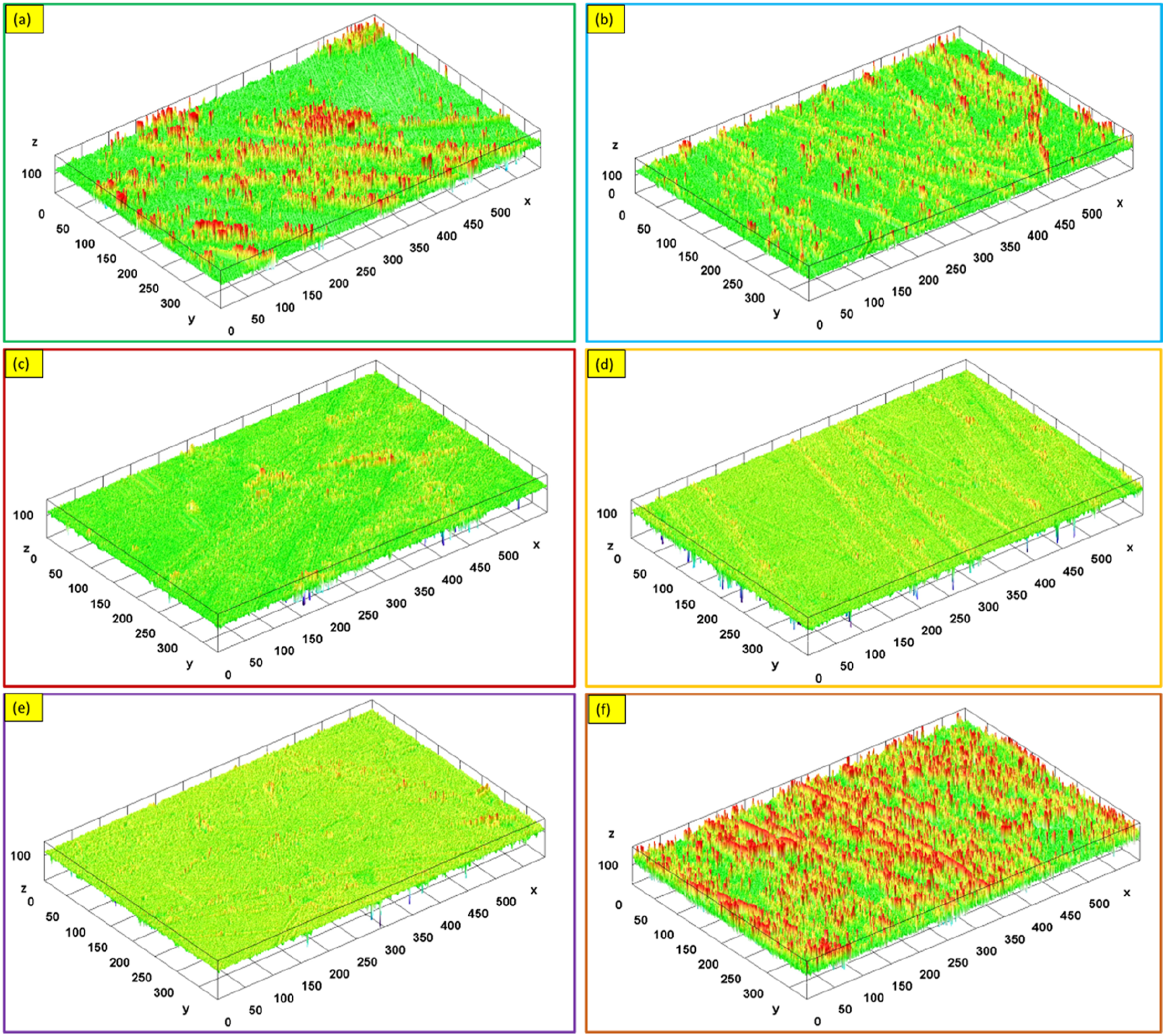
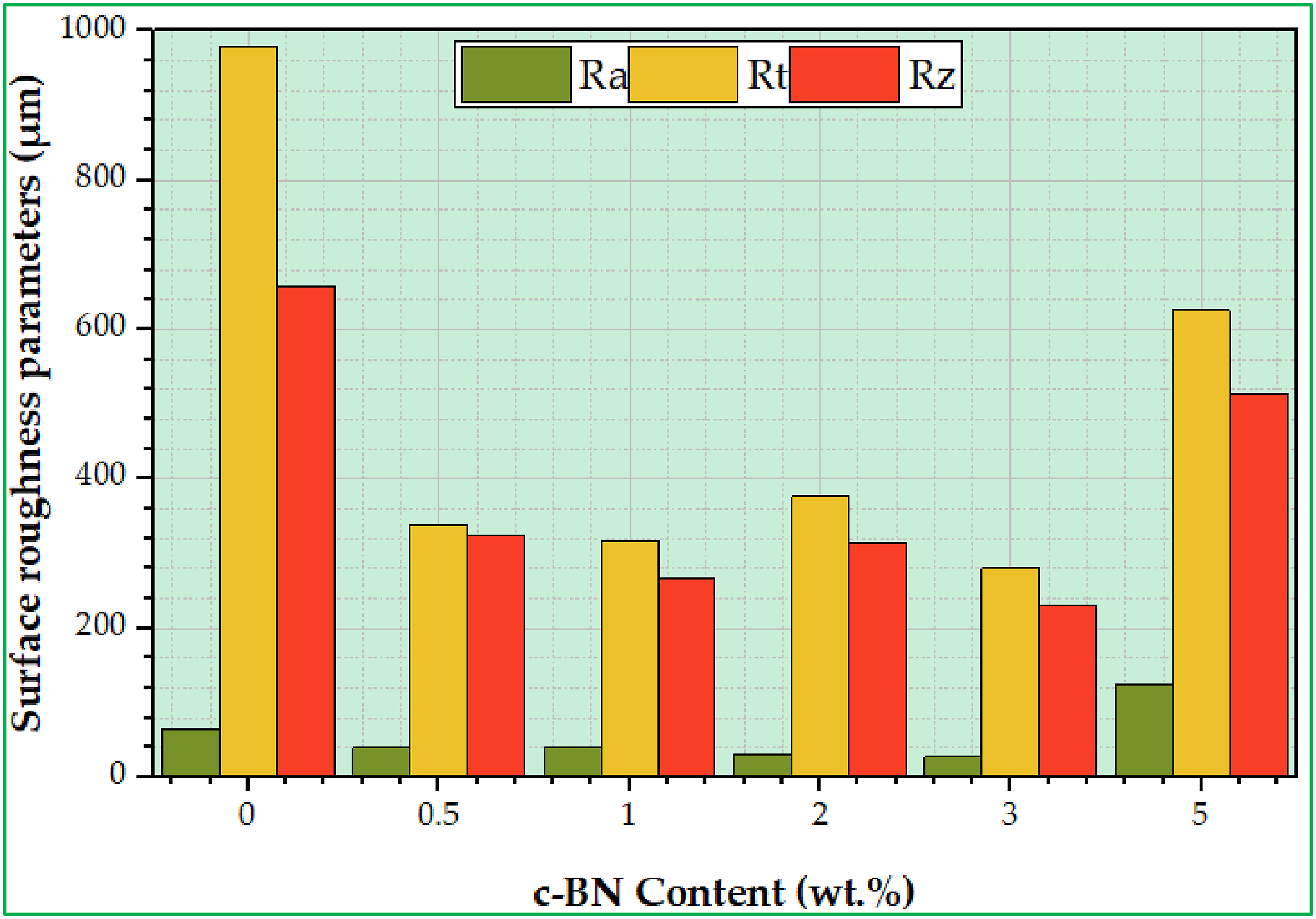
The examination of the SEM micrographs of the worn-out surfaces provides a clearer understanding of the impacts of the different c-BN concentrations, as shown in Figure 11. As the c-BN concentration decreases in PMMA/c-BN biocomposites, noticeable deep tear marks appear on the surface of the samples, as depicted in Figure 11(a)–(d). Additionally, there are numerous delaminations and significant weight loss observed in the wear trace. Consequently, biocomposite samples containing 0 wt% c-BN to 2 wt% c-BN experience more severe damage, while the biocomposite samples containing 5 wt% c-BN is more deep wear tracks. Moreover, craters can be observed after matrix delamination and transfer of removed material. Furthermore, the analysis of wear traces indicates that all types of composite wear are related to abrasive and adhesive wear modes. In the vicinity of PMMA/c-BN composites, scratches, and microcracks in the polymer matrix are visible in 3 wt% c-BN biocomposite samples compared to other c-BN concentrations. An increase in c-BN concentration results in an improvement in both the polymer matrix and wear resistance, except PMMA-5 wt% c-BN sample. The wear trace is found to be shallower, and the wear mechanism occurs in the form of surface sweeping, as shown in Figure 11(d) and (e), while a deeper wear trace is obtained in Figure 11(f). Also, Figure 12(a)–(f) depicts the 3D surface roughness topography for the PMMA and its fabricated biocomposites from PMMA/c-BN with varying c-BN concentrations. The 3D surface roughness images were drawn by analysis and turn of the 2D SEM-worn surface by Gwyddion software to determine the surface roughness parameters like average roughness (Ra), the total surface roughness (Rt), and maximum peak height (Rz) as well as to evaluate the surface characteristics for PMMA/c-BN biocomposites. It’s clear that the surface roughness appeared to be very smooth with an increase in the c-BN into PMMA matrix up to 3 wt% as depicted in Figure 12(a)–(d), while the sample fabricated with 5 wt% c-BN appeared more rough surface (Figure 12(f)). Based on the surface roughness results shown in Figure 13, the PMMA composite without c-BN was found to have higher roughness parameters (Ra, Rt, and Rz). As the concentrations of c-BN NPs increased up to 3 wt%, the surface quality improved and the roughness values decreased, leading to better results in terms of roughness parameters. This can be due to the homogeneous dispersion of c-BN, the addition of c-BN nanoparticles, and the superior characteristics of c-BN, whereas nonhomogenous dispersion and high concentrations of c-BN result in agglomeration (Figure 12(f)) of the c-BN and increased roughness, as illustrated in Figure 13. Thus, with the decreased surface roughness (Ra, Rt, and Rz) for PMMA/c-BN biocomposites used to make dentures, the patient feels more comfortable due to low microorganisms adhering to the denture surface as well as the low possibility chance of a bad odor, it will be easier to remove dentures without impacting patients' oral health, and the frictional force between teeth will be reduced.
54
Likewise, a denture’s cleanliness and appearance are negatively impacted by increased surface roughness (Ra, Rt, and Rz), which permits the growth of bacteria and stains. Worn-out surface for fabricated specimens of PMMA/c-BN composites; (a) 0 wt% c-BN, (b) 0.5 wt% c-BN, (c) 1 wt% c-BN, (d) 2 wt% c-BN, (e) 3 wt% c-BN, and (f) 5 wt% c-BN. 3D surface roughness for fabricated specimens of PMMA/c-BN composites; (a) 0 wt% c-BN, (b) 0.5 wt% c-BN, (c) 1 wt% c-BN, (d) 2 wt% c-BN, (e) 3 wt% c-BN, and (f) 5 wt% c-BN. Surface roughness parameters for fabricated PMMA/c-BN biocomposites.
Conclusions
The incorporation of c-BN NPs into the PMMA matrix significantly affects the physical, mechanical, and tribological characteristics of the produced biocomposites. The bulk density of the biocomposites produced increases progressively with increasing the concentration of c-BN in the polymer matrix from 0 to 5 wt% c-BN, reaching the highest value of 1.221 ± 0.002 g/cm3 at 5 wt% c-BN, compared to 1.171 ± 0.003 g/cm3 for 0 wt% c-BN (PMMA matrix). Conversely, porosity decreases with increasing C-BN NPs in the PMMA matrix, with the highest values of porosity (1.6%) obtained at 0 wt% c-BN and the lowest porosity (0.16%) at 5 wt% c-BN. The SEM analysis for biocomposites reveals a homogenous dispersion of c-BN NPs within the fabricated PMMA matrix for c-BN concentrations up to 3 wt%. But, at PMMA-5 wt% c-BN, slight agglomeration is observed, which is evidenced by EDS analysis and mapping, confirming the interaction between PMMA and c-BN.
The mechanical characteristics, including hardness values, ultimate compressive stress, and yield stress of the PMMA/c-BN composites are dramatically improved with increasing the c-BN concentration in the polymer matrix compared with PMMA-0 wt% c-BN. The highest hardness value, reaching 79.5 ± 1.6 HV, is achieved at PMMA-5 wt% c-BN, whereas the UCS and YS reach their values of 215 ± 6.17 MPa and 101.1 ± 2.2 MPa, respectively, at PMMA-3 wt% c-BN. Similarly, the wear resistance increases with increasing the concentrations of c-BN in the polymer matrix, reaching its highest at PMMA-3 wt% c-BN, which exhibits an improvement of up to 81% compared with the PMMA matrix. The surface roughness parameters (Ra, Rt, and Rz) decrease with increasing concentrations of c-BN in the polymer matrix, improving the frictional force between teeth and low microorganisms adhering to the denture surface and the low possibility chance of a bad odor. The lowest roughness parameters are obtained at PMMA-3 wt% c-BN, whereas the highest is at pure PMMA.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
