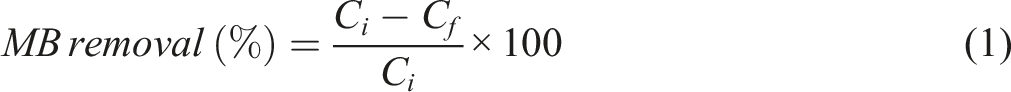Abstract
Water pollution by organic dyes such as methylene blue (MB) poses significant environmental and health risks due to their toxicity and persistence in water bodies. This study addresses the urgent need for effective and sustainable adsorbents by developing a novel adsorbent using recycled poly (ethylene terephthalate) (rPET) and sepiolite, a natural clay mineral, to remove MB dye from aqueous solutions. The rPET/sepiolite adsorbent was synthesized through solution blending, optimizing parameters such as rPET concentration (1, 5, 10, 15 and 20 wt%), blending time (10, 30, 60, 120 and 180 minutes), and sepiolite loading (0, 0.1, 0.25, 0.7, and 0.8 g). The resulting adsorbent was characterized using scanning electron microscopy (SEM), mapping, Brunauer-Emmett-Teller (BET) surface area analysis, thermogravimetric analysis (TGA), Fourier-transform infrared spectroscopy (FTIR), and zeta potential to evaluate its morphological, thermal, and functional properties. The best condition of ad-sorbent, composed of 10 wt% rPET and 0.25 g sepiolite blended for 1 hour, exhibited a hierarchical porous structure, combining micro-, meso-, and macropores, enhancing MB adsorption capacity to 96.7%. The TGA results indicated improved thermal stability, while FTIR confirmed the successful integration of sepiolite into the rPET matrix. These findings demonstrate that the rPET/sepiolite nanocomposite offers a promising, sustainable solution for dye removal in wastewater, contributing to environmental pollution control and plastic waste management. The study highlights the potential of utilizing recycled materials to develop efficient adsorbents, offering a more affordable and sustainable option than traditional techniques.
Keywords
Introduction
The problem of organic dye-induced water pollution has attracted a lot of attention worldwide in recent decades because of the fast expansion of industrialization. The annual discharge of thousands of metric tons of organic dyes into water poses a significant risk to human health due to their carcinogenic and mutagenic properties. Additionally, these dyes cause severe ecological harm by impeding sunlight penetration and inhibiting the photosynthesis of aquatic plants. 1 Consequently, finding an effective solution to decrease the concentration of dye in water has become a pressing issue that needs to be addressed. Numerous methods are currently employed to remove dye pollutants from wastewater, such as oxidation, adsorption, precipitation, and biological or photocatalytic degradation. Among these methods, adsorption remains the most extensively utilized industrially, with an ongoing exploration into diverse dye adsorbents such as activated carbon, 2 lignin-based products3,4 chitosan,5,6 cellulose-based beads, 7 Metal-Organic Frameworks (MOFs)8–11 and other thermos polymerized composites.12,13 However, traditional dye adsorbents produce sludge when they absorb the dye, requiring centrifugation, costly adsorbents, and significant energy usage during polymerization. Hence, creating a filter-type adsorbent presents the benefit of straightforward removal without centrifugation after dye adsorption. In order to effectively remove reactive and acidic dyes with sulfonic acid groups from wastewater dyeing, surface modification is required to improve the dye-molecule bond.
Food packaging, synthetic fibers, electrical insulating materials, films, and bottled containers are just a few of the many products that use poly (ethylene terephthalate) (PET).14,15 Its global yearly output reaches over 70 million tons. 16 Still, only 28.4% of PET is recycled as a renewable resource after brief initial use, with the remainder being disposed of in natural systems. PET exhibits a significant level of chemical stability, resulting in a natural decomposition process that spans roughly 300–450 years. 17 Consequently, the dangers resulting from the accumulation of PET in land and sea ecosystems have become more severe. 18 A viable and sustainable solution to this issue is to recycle post-consumer and post-industrial PET waste, reducing petrochemical raw material usage and carbon emissions. 19
Nanotechnology presents an opportunity to enhance polymer characteristics across various technical fields, including polymeric Nano biomaterials, and among these, layered clay/polymer nanocomposites have been extensively investigated.20,21 Sepiolite is a hydrated magnesium silicate mineral (Mg4Si6O15(OH)2·6H2O) that possesses a microfibrous structure with a high aspect ratio and nanoscale dimensions, qualifying it as a nanofiber by EFSA standards. Its surface properties, characterized by silanol groups (Si-OH) on its external surface, make it particularly appealing for industrial applications, facilitating the preparation of organic-inorganic materials.22,23 Naturally, inexpensive sepiolite has much potential for eliminating dyes from industrial wastewater because it functions as an ion exchanger. Recently, Chinoune et al. (2024) investigated the adsorption behavior of methylene blue (MB) dye on biocomposites of alginate-sepiolite beads, and It was discovered that the amount of sepiolite used affected the improvement of adsorption properties. 24 Researchers found that the nanocomposite can be utilized as an effective and environmentally friendly sorbent for environmental cleanup after closely examining the adsorption behavior of MB onto sepiolite-silver nanoparticles. 25 In the study conducted by other researchers, the impregnation synthesis of NiO/sepiolite and its application for MB dye removal during wastewater treatment is introduced. 26 The adsorptive properties in the NiO/sepiolite composites received interesting results for removing refractory biodegradable organic pollutants.
Despite the extensive exploration of natural and synthetic adsorbents, the potential of materials composed entirely of readily available, non-toxic natural components remains largely underexplored. In particular, the combination of rPET and sepiolite presents an innovative and environmentally sustainable avenue for developing high-performance adsorbents. This study pioneers the fabrication of a novel rPET/sepiolite composite adsorbent through solution blending, optimizing its composition to achieve superior adsorption efficiency. The synthesized adsorbent was systematically evaluated for its effectiveness in removing methylene blue (MB) dye from aqueous solutions. By leveraging the synergistic properties of rPET and sepiolite, this work provides a groundbreaking approach to wastewater treatment, offering a cost-effective, scalable, and eco-friendly alternative to conventional adsorbents. The findings contribute to the advancement of sustainable material applications and address critical challenges in plastic waste valorization and water pollution remediation.
Materials and methods
Materials and reagents
Post-industrial rPET in pellet form (density: 1.38 g/cm3) supplied by Alba Polyester Sdn. Bhd., Malaysia, Sepiolite, which has been commercialized as Pangel S9 (density 2.3 g/cm3) from Tolsa, trifluoroacetic acid (TFA), dichloromethane (DCM) and NaOH from Sigma-Aldrich Chemistry and MB dye was obtained from Hannschweiller Laboratory Chemicals. Throughout this investigation, all of the MB dye solutions were made with distilled water.
Preparation of recycled PET/sepiolite (rPET/sepiolite) adsorbent
Figure 1 shows the preparation of rPET/sepiolite adsorbent via the solution blending method as proposed by (Naksuwan et al., 2015)
27
and (Yu et al., 2024).
22
Naksuwan et al. suggested a 3:7 TFA:DCM ratio, 1g rPET pellet, and 30 mins blending time to achieve a homogenous solution. Optimum sepiolite content of 0.25 g was suggested by Largo et al. (2020).
28
From those studies as references, the parameters were varied into five ranges to study several effects, which were rPET weight percentage, solvent ratio, sepiolite content, and blending time. Firstly, various weight percentages of rPET (1, 5, 10, 15, 20 wt%) were used in the adsorption test to determine the best weight percentage with the highest MB removal percentage. After the best rPET weight percentage was determined, the effect of TFA and DCM solvent ratios with ratios of 9:1, 7:3, 3:7, 5:5, and 1:9 were varied to dissolve rPET. Since the mixture was allowed to stir at 400 r/min (10, 30, 60, 120, and 180 mins) to achieve a homogeneous polymer solution. The mixture was added with Pangel S9, a natural sepiolite with fibrous clay mineral, (0, 0.1, 0.25, 0.7, and 0.8 g) dispersed in the same solvent under vigorous stirring (400 r/min) to ensure uniform dispersion of the mixture, maintaining continuous stirring to facilitate the interaction between the polymer chains and the nanoclay. The resulting mixture was cast into a petri dish, being air-dried overnight, and subsequently subjected to solvent evaporation, often under controlled temperature and humidity conditions to remove residual solvents completely. The four effects were studied one at a time with the same repeating steps. This process yields rPET/sepiolite adsorbent with enhanced mechanical, thermal, and barrier properties due to the synergistic effects between the PET matrix and the well-dispersed sepiolite clay. This process yields rPET/sepiolite adsorbent with enhanced mechanical, thermal, and barrier properties. The film then crashed using a grinder until the powdery form was produced. Then, it crashed again using a mortar and pestle, which were then collected via a sieve. The recycling of polymers through various techniques and solution blends is discussed by multiple researchers.29–31 Schematic diagram on the preparation of rPET/sepiolite adsorbent.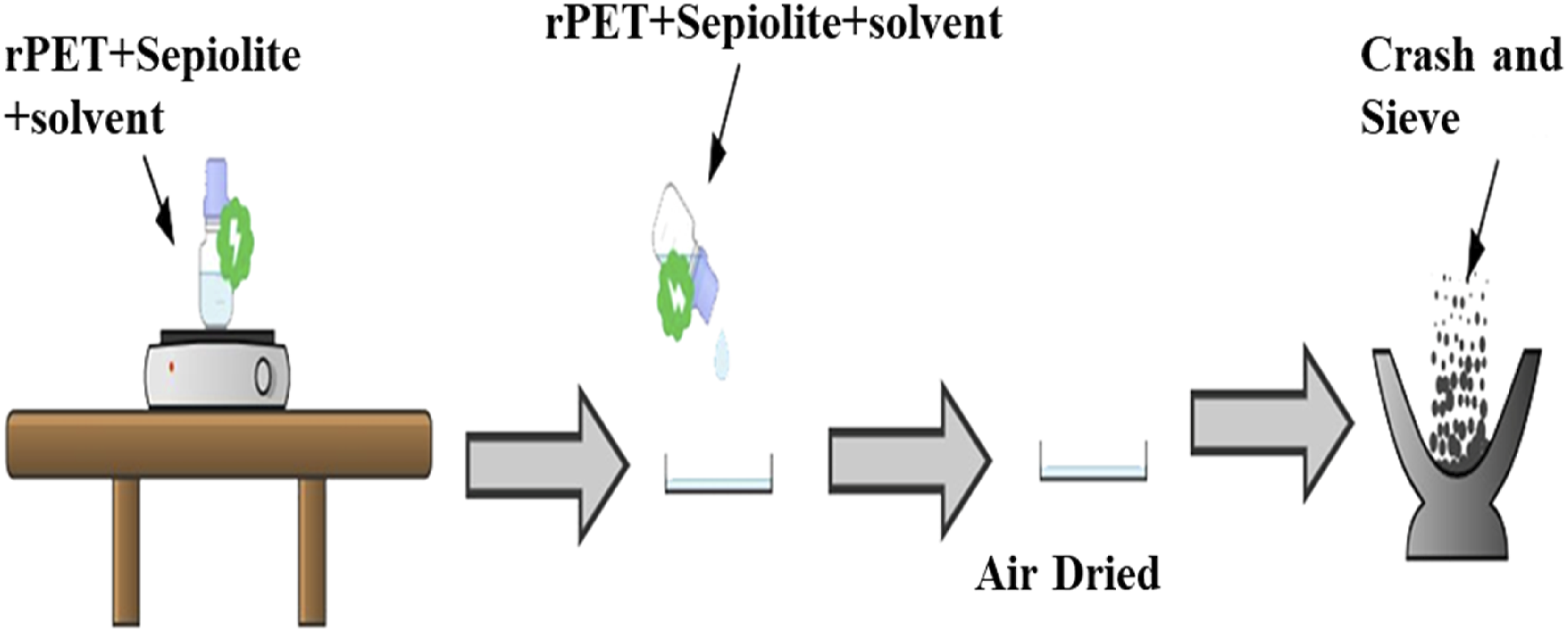
Adsorption of MB dye
The adsorption of MB dye (pH of MB solution is 7) by different formulations of rPET/sepiolite adsorbent was accomplished at room temperature across a 1-h contact time, using a volume of 50 mL of dye solution at a concentration of 10 mg/L and a mass of 1.0 g of adsorbent. By utilizing the calibration curve that the UV/Vis spectrophotometer (Shimadzu UV-1800, Japan) formed at its maximum wavelength (664 nm), the remaining dye concentration was examined, and the percentage removal was determined based on the following equation (1).
Characterization of rPET/sepiolite adsorbent
The Brunauer, Emmett, and Teller (BET) and Barret, Joyner, and Halenda (BJH) methods were used to evaluate textural characteristics like specific surface area, average pore size, and pore size distribution (TriStar two micromeritics, United States). A scanning electron microscope (SEM) (Prox, Phenom-World, Netherlands) was used to examine the samples’ morphological features. The samples were gold-coated under vacuum for 3 minutes at a current of 10 mA before examination. Nitrogen physisorption measurements were conducted at −196°C with a Micromeritics TriStar II 3020 instrument. Fourier transform infrared (FTIR) spectra from Thermo Fisher (Model: Nicolet 6700, United States), within the wavenumber range of 650–4000 cm−1, were used for the sample’s functional groups identification. A thermogravimetric analyzer (TGA) (TG 209 F1, NETZSCH, Germany) was used to assess the thermal stability of the nanocomposite. Measurements were made in a nitrogen atmosphere between 30°C and 800°C at a heating rate of 10°C/min.
Results and discussion
Parametric study on the preparation of rPET/sepiolite adsorbent
Effect of rPET weight percent
According to Figure 2, the highest initial removal rate of MB was achieved with a 10 wt% content of rPET. This adsorbent with 10 wt% rPET, equating to 0.7 g, was shaken with MB solution to determine the best rPET weight percentage. Previous studies indicate that under conditions of a 24-h contact time and a 10 mg/L MB concentration was the optimum condition of the adsorption test32–35 Based on this condition, 10 wt% rPET demonstrated the highest MB removal percentage at 30.85%. The findings demonstrated that a low polymer concentration (<5 wt%) did not yield a consistent internal pore structure, whereas an excessive polymer concentration (>10 wt%) hindered pore formation. Effect of rPET weight percentage on the removal of MB dye (Initial MB concentration = 10 mg/L, the volume of solution = 50 mL, the dosage of adsorbent = 0.7 g, temperature = room temperature).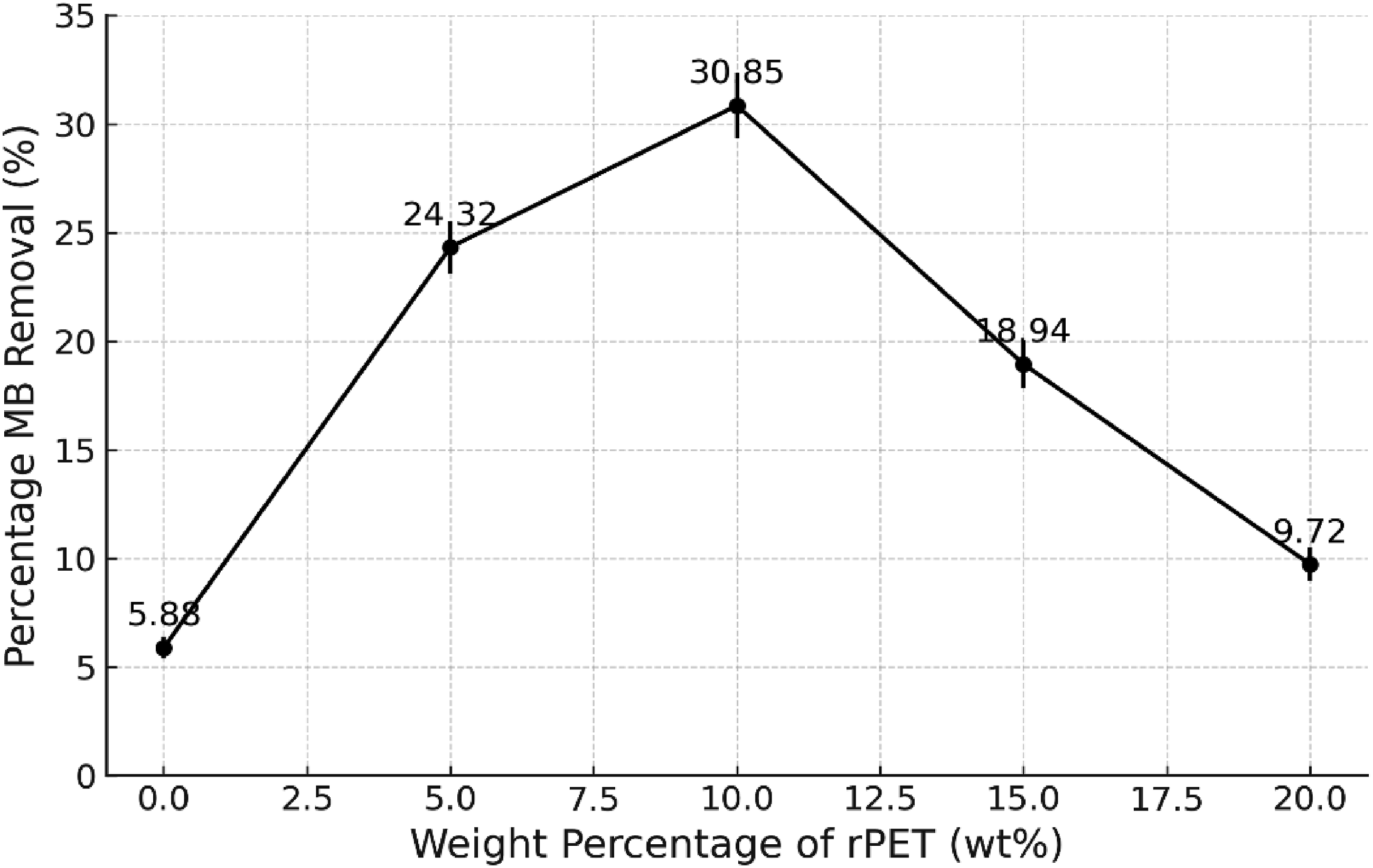
To validate the efficiency of the rPET/sepiolite composite, its methylene blue (MB) adsorption performance was compared to other adsorbents reported in recent literature. Ma et al. (2019) demonstrated that chitosan-Congo red (CS-CR) particles achieved 99.7% removal of MB from a 50 mg/L dye solution using only 1.5 mg of adsorbent, reaching equilibrium within 30 minutes and exhibiting an ultrahigh adsorption capacity of 1457.1 mg/g. 5 Zhao et al. (2022) prepared magnetic acetylated lignin composites (MADL) that achieved over 94% MB removal efficiency in less than 50 minutes and retained high performance even under saline conditions. 4 In contrast, the Zn-MOF adsorbent developed by Gao et al. (2022) focused primarily on Congo red and methyl orange dyes. 10 No removal efficiency for MB was reported in that study. Compared to these, the rPET/sepiolite composite achieved 96.7% MB removal in 60 minutes using 1 g of adsorbent without requiring advanced surface functionalization, magnetic enhancement, or bio-template synthesis. This performance and the composite’s sustainability and low cost underscore its suitability for practical wastewater treatment applications.
Effect of solvent ratio
TFA, or a mixture of TFA and DCM, can dissolve PET, a thermoplastic polymer. The effect of solvent ratio was determined based on the previous studies by Pulido et al. (2019)
36
and Yu el. Al. (2024).
37
The aim of the TFA:DCM ratio was studied to determine the best solvent ratio that can dissolve rPET and sepiolite in the shortest period, as well as to use the least TFA ratio to lessen the usage of corrosive solvent in the preparation of the adsorbent. Thus, five ranges of solvent ratios were used. Figure 3 shows the best solvent ratio of 3:7 (TFA:DCM) with 95.8% MB removal. Effect of solvent ratio on MB dye removal (Initial MB concentration = 10 mg/L, solution volume = 50 mL, adsorbent dosage = 1 g, temperature = room temperature).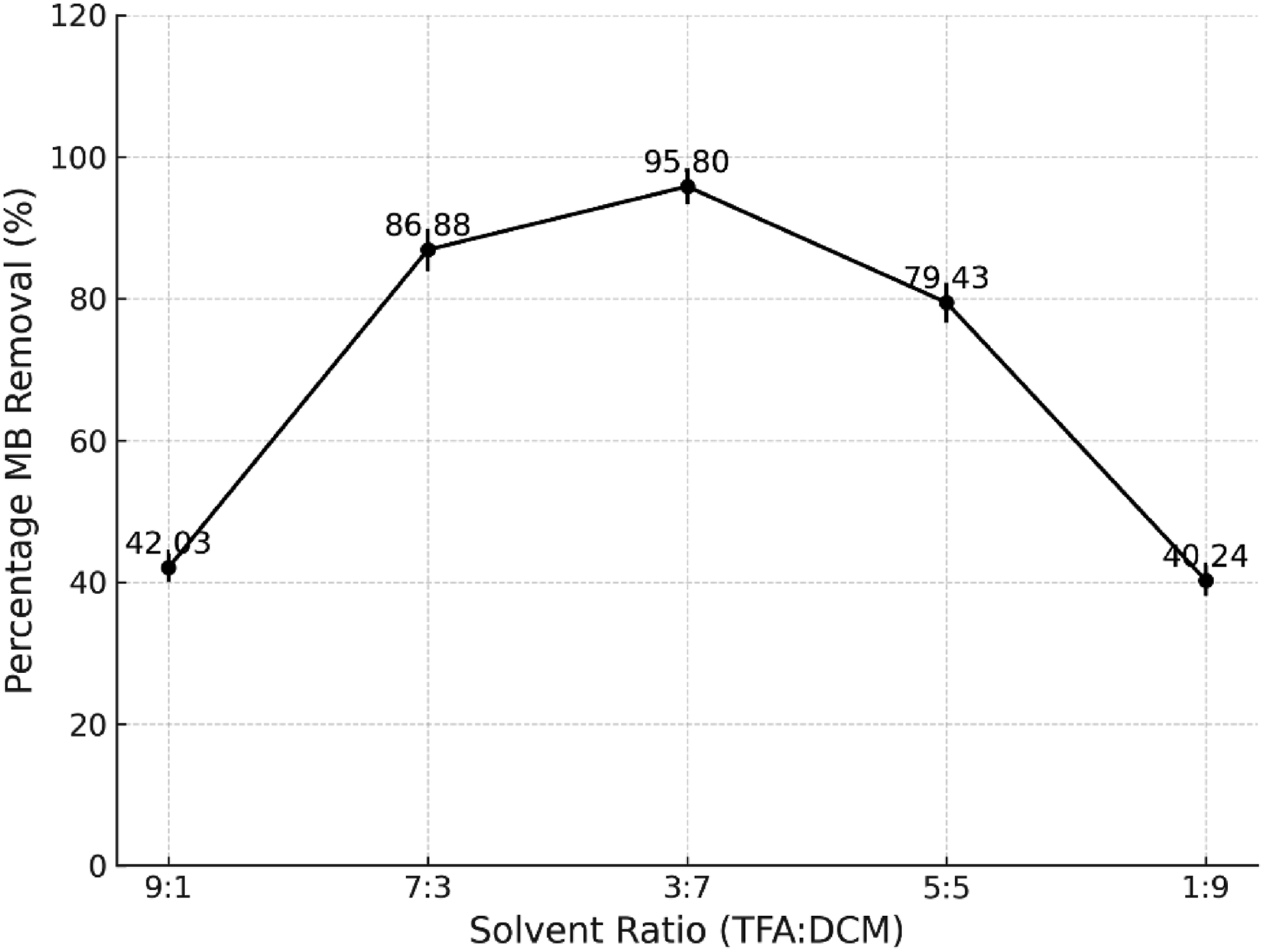
Effect of sepiolite loading
Figure 4 shows the highest MB percentage removal was at 0.25 g loading of sepiolite into rPET with 96.69% removal. The initial maximum sepiolite amount of 0.8 g was determined through trial and error based on its solubility in the rPET solution. Beyond 0.8 g, the solution became excessively thick. Alternatively, Largo et al. investigated the use of sepiolite clay mineral as an adsorbent in the adsorptive removal of cationic and anionic dyes through molecular dynamic simulation and experimental methods.
28
They used 0.25 g/L of sepiolite throughout the study. The findings of the adsorption isotherm experiments and the intermolecular interaction energies show that MB dye has a high adsorption efficiency on the sepiolite surface in a basic solution. Thus, this explained the results where sepiolite content of 0.25 g was the best loading content onto rPET. Effect of Sepiolite loading onto rPET on MB dye removal. (Initial MB concentration = 10 mg/L, the volume of solution = 50 mL, the dosage of adsorbent = 1 g, temperature = room temperature).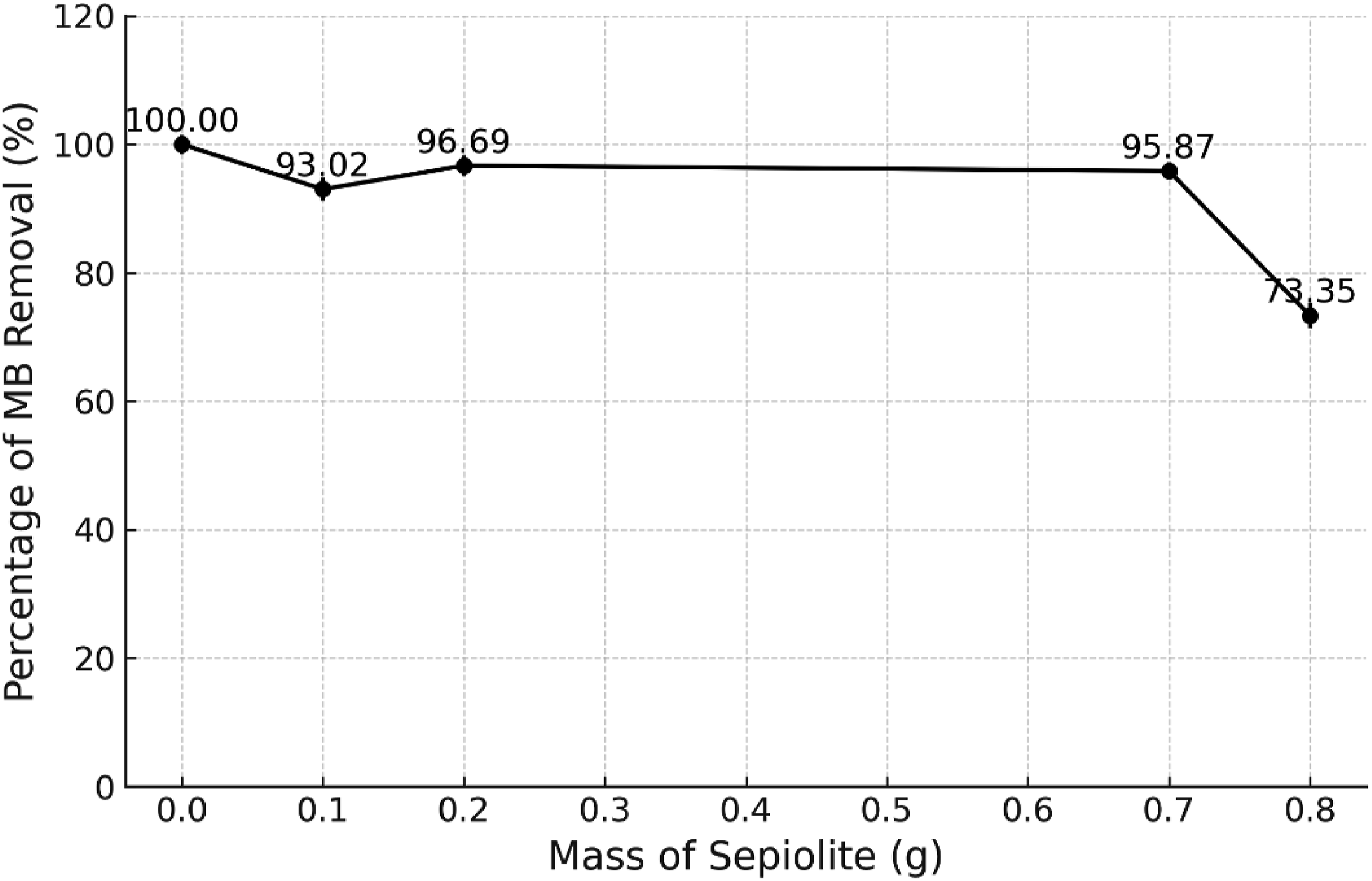
Effect of blending time
Figure 5 illustrates the preparation of rPET with sepiolite over 10, 30, 60, 120 and 180 minutes. The best blending time was found to be 60 minutes. The MB percentage removal was increased as the blending time in adsorbent preparation rose from 10 to 30 minutes, suggesting enhanced mixing and availability of active adsorption sites. Blending time, or the duration of mixing during adsorbent preparation, is crucial in determining the adsorbent’s effectiveness. Proper blending ensures uniform distribution of activating agents and the development of desired porosity and surface area, essential for optimal adsorption capacity.
38
The reduction of MB percentage removal which was observed during 120 and 180 minutes, may be attributed to particle aggregation, decreased porosity, or saturation of adsorption sites over prolonged blending durations. Extended blending times can lead to the agglomeration of adsorbent particles, resulting in larger aggregates that diminish the available surface area for adsorption. This aggregation reduces the number of active sites accessible for MB molecules, thereby decreasing adsorption efficiency.
39
In conclusion, the best preparation condition of the adsorbent was 10 wt% rPET with 0.25 g sepiolite in a 3:7 ratio of TFA:DCM in 60 minutes of blending time. Effect of blending time during preparation of rPET/sepiolite adsorbent on the removal of MB (Initial MB concentration = 10 mg/L, the volume of solution = 50 mL, the dosage of adsorbent = 1g, temperature = room temperature).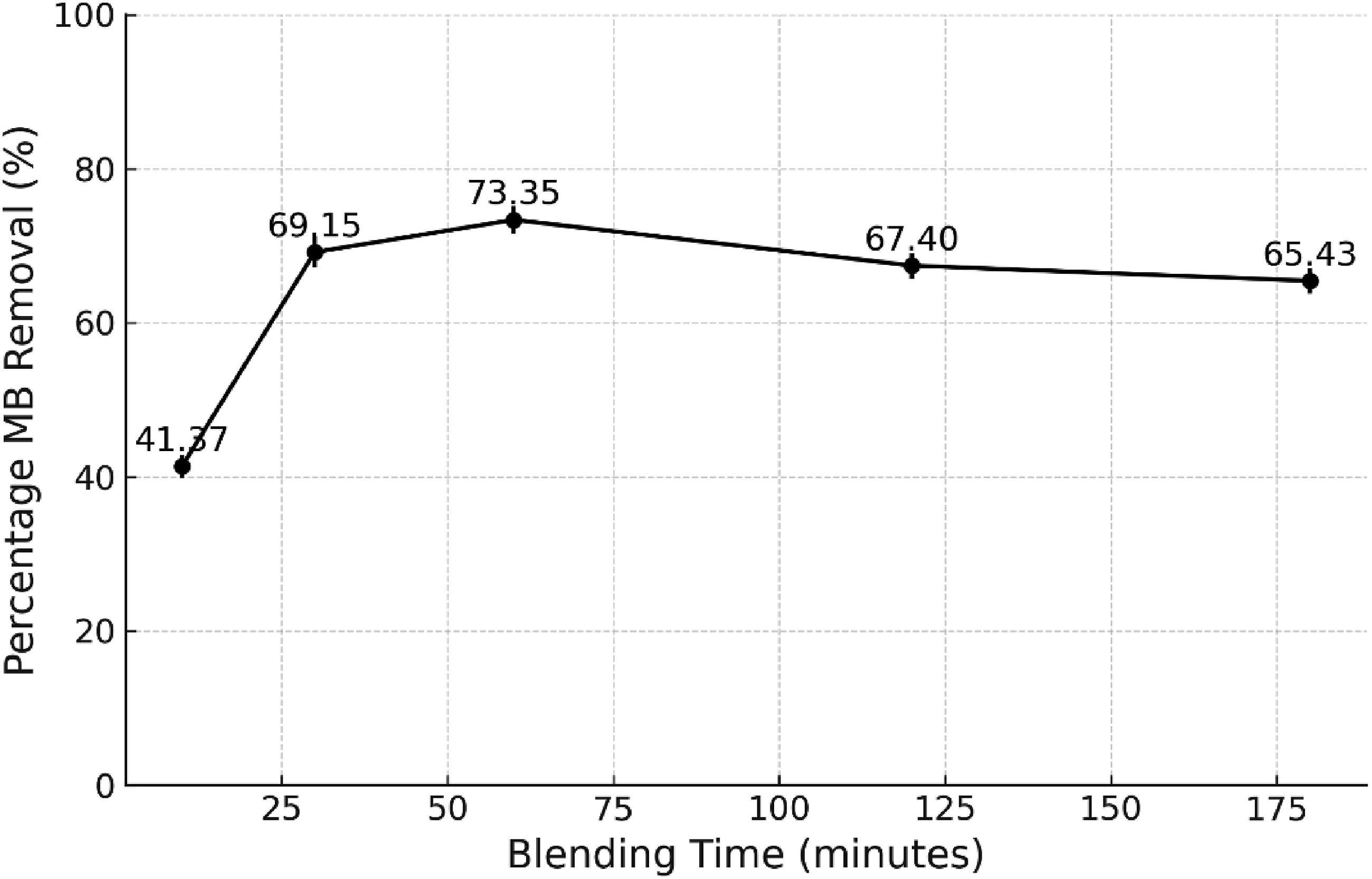
rPET/sepiolite adsorbent characterization discussion
Scanning electron microscopy (SEM) & mapping
Phase separation and thermodynamic instability occur at the polymer interface when a non-solvent is added, resulting in the formation of a polymer-rich matrix and a polymer-poor pore region.
40
SEM pictures (Figure 6) demonstrate that the porosity of the PET matrix decreases as the DCM proportion rises.
41
Tsai et al. (2021) noted that DCM’s volatility leads to skin formation, which resists solvent and non-solvent diffusion, slowing lamination and precipitation, thus reducing microvoids.
42
Surface morphology varies with different solvent compositions, affecting porosity and texture. The concentration of PET in the casting solution affected the porosity of the precipitated PET matrix. SEM images reveal that Figure 6(a) shows a rougher surface with irregular pores (4.7–13.1 μm) and more particulate matter compared to Figure 6(b), which has a smoother, more cohesive surface with elongated pores (2.7–9.2 μm). Figure 6(a) also has a higher concentration of small particles and a granular texture, while Figure 6(b) exhibits a more fibrous texture with smoother transitions. The surface morphology of the (a) rPET and (b) rPET/Sepiolite adsorbent was prepared via solution blending. (×2500 magnification).
As shown in Figure 6(a), increasing the polymer concentration reduced microvoid formation and promoted the development of sponge-like structures. 40 As a result, the ideal parameters for creating the casting solution in the subsequent rPET/sepiolite adsorbent preparation were determined to be 10-weight percent rPET and a TFA:DCM ratio of 3:7 (v/v).
As illustrated in Figures 7 and 8, mapping reveals the elemental composition of the adsorbents. Both samples exhibit carbon signals consistent with their rPET base material. In Figure 7, the incorporation of sepiolite is evidenced by the presence of silica (Si) and magnesium (Mg) in the elemental maps. Trace amounts of fluorine (F) are detected and attributed to residual TFA used as a solvent during the solution blending process. However, the fluorine content appears to be relatively low. Mapping of the (a) rPET adsorbent and (b), (c), (d) are the elements contained in the adsorbent prepared via solution blending. Mapping of the (a) rPET/sepiolite adsorbent and (b), (c), (d), (e), and (f) are the elements contained in the adsorbent prepared via solution blending.

Brunauer-emmett-teller (BET)
rPET adsorbent
Figure 9(a) shows that the nitrogen adsorption-desorption isotherm of rPET used for MB removal has mesoporous characteristics, indicated by the Type IV isotherm with a hysteresis loop. It is effective at adsorbing methylene blue because of its substantial rise near saturation pressure and gradual increase at lower pressures, which indicate a large surface area and pore volume. The presence of mesopores (2–50 nm) indicates the material’s suitability for larger molecules, highlighting its potential for environmental remediation. BET surface area analysis in the 0.05 to 0.35 pressure range confirms the adsorbent’s effectiveness.
43
BET plot of rPET on (a) N2 adsorption-desorption isotherms of freeze-dried BC based on the first adsorption-desorption run immediately after degassing, (b) distributions in constant pressure and adsorptive fouling experiments.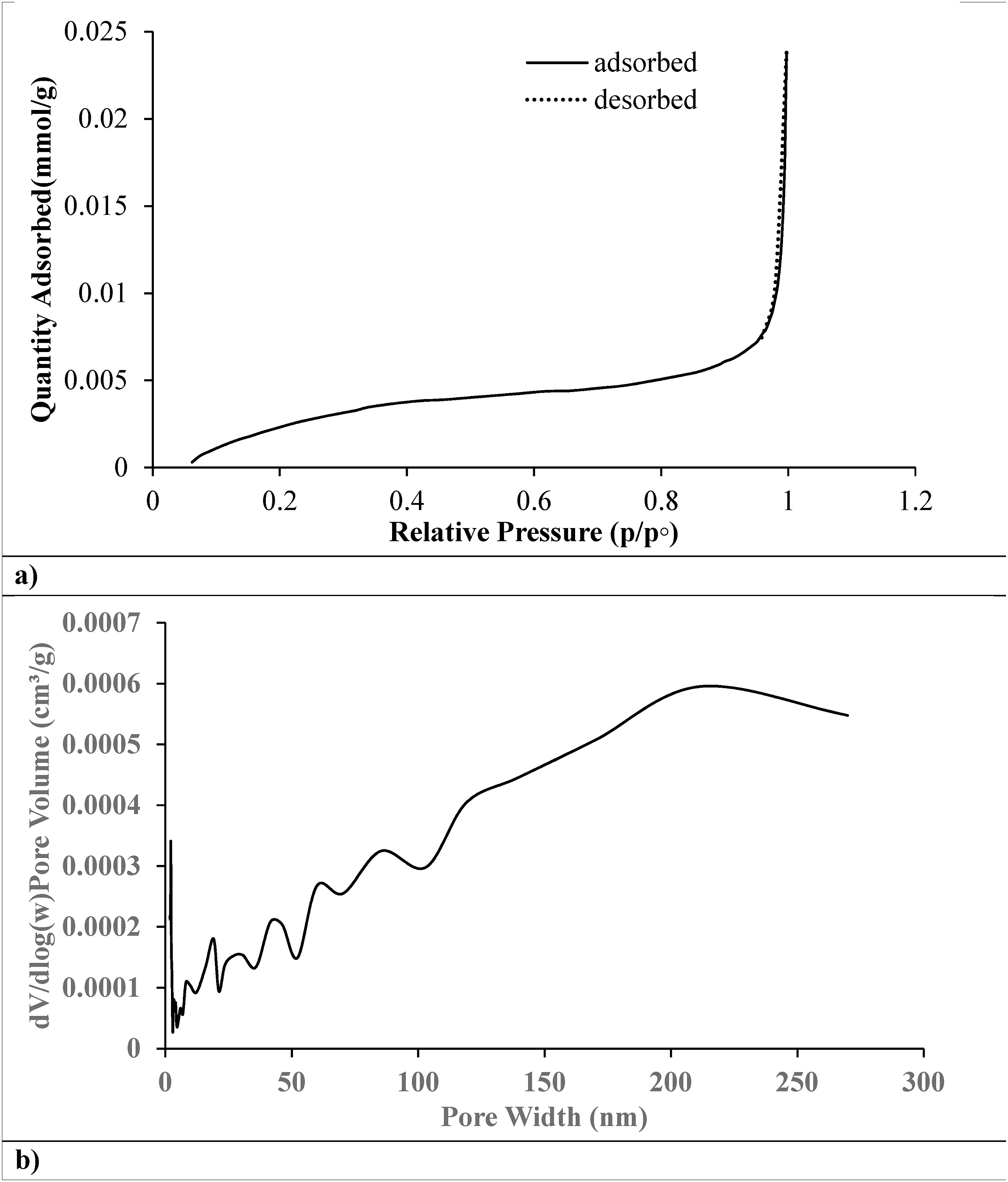
Figure 9(b) reveals that the rPET adsorbent has a heterogeneous pore structure with mesopores and macropores (>50 nm) crucial for MB adsorption. The broad pore size distribution, particularly in the 150–200 nm range, suggests enhanced capacity for larger dye molecules. This distribution allows capillary condensation and easier diffusion of larger molecules, optimizing the material’s performance in water treatment. 44 These features highlight the recycled PET’s potential as a sustainable, high-performance adsorbent for environmental remediation.
Sepiolite
Figure 10(a) shows that the nitrogen adsorption-desorption isotherm for the sepiolite adsorbent used to remove methylene blue displays a Type IV isotherm with a clear hysteresis loop typical of mesoporous materials. The gradual increase in adsorption at lower pressures suggests the presence of micropores, while the sharp rise near saturation pressure indicates capillary condensation within the mesopores. The slight separation between the adsorption and desorption branches in the hysteresis loop points to slit-shaped pores or networks, enhancing the material’s adsorption capability. Sepiolite is a viable choice for wastewater treatment since it effectively removes methylene blue, with a maximum adsorption capacity of about 25 mmol/g.
45
BET plot of sepiolite on (a) N2 adsorption-desorption isotherms of freeze-dried BC based on the first adsorption-desorption run immediately after degassing, (b) distributions in constant pressure and adsorptive fouling experiments.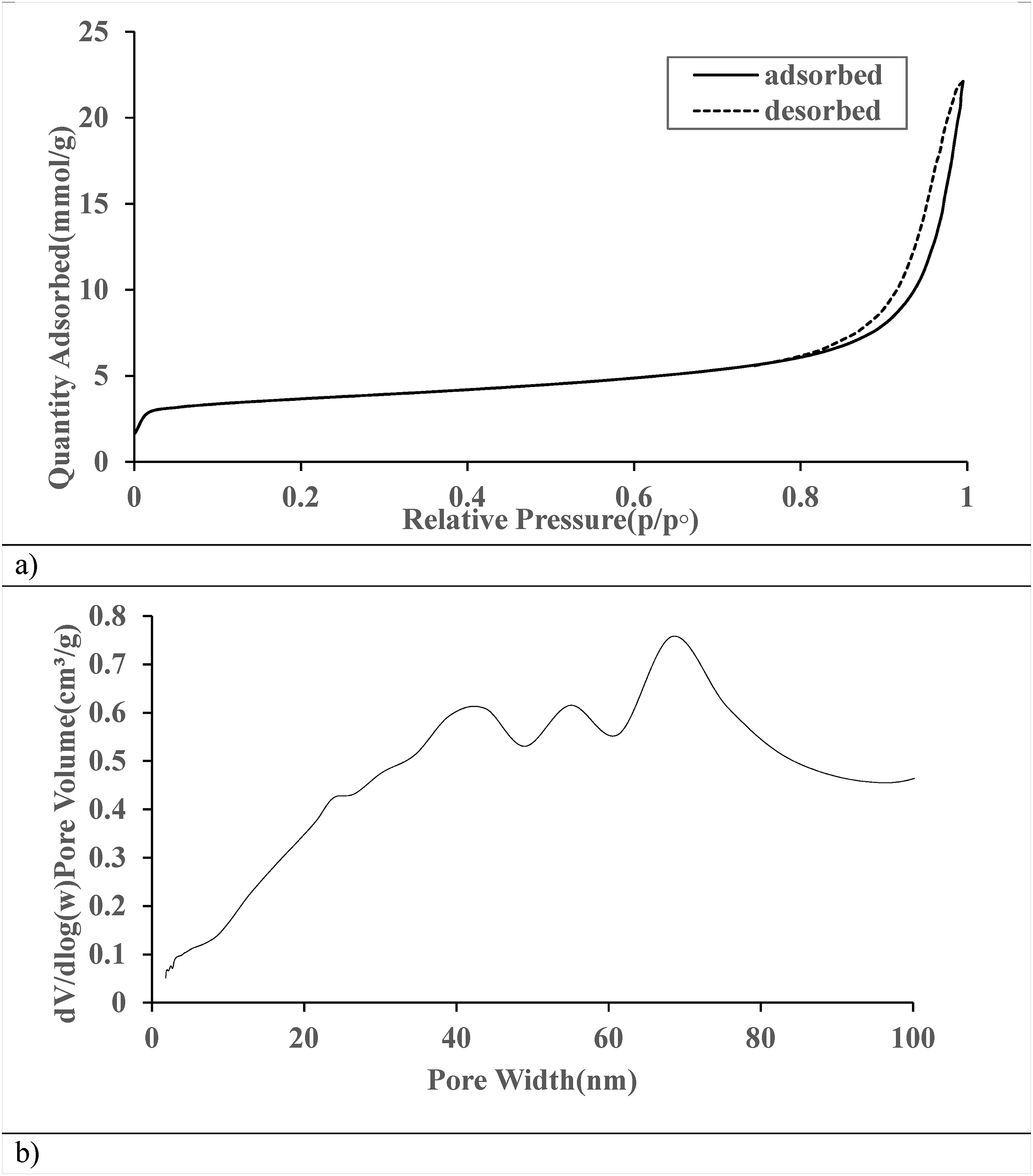
Figure 10(b) shows that the pore size distribution of sepiolite is complex and multimodal, indicating its effectiveness in methylene blue adsorption. The graph reveals a wide range of pore sizes, with notable peaks at about 40 nm, 55 nm, and a prominent one at 70 nm, suggesting a hierarchical structure that improves adsorption kinetics. 46 The gradual increase in pore volume from 0 to 30 nm indicates the presence of micropores and small mesopores, essential for initial adsorption stages and molecular sieving. The dominant mesoporous character, with significant pore volume in the 20–80 nm range, is advantageous for methylene blue adsorption, allowing efficient diffusion and access to internal sites. 47 Larger mesopores and potential macropores beyond 80 nm may facilitate rapid mass transfer and prevent pore blocking during adsorption. 48 This diverse pore structure, combining micro-, meso-, and macropores, likely gives sepiolite a high adsorption capacity, fast kinetics, and good regeneration properties for methylene blue removal from water. 49
rPET/sepiolite adsorbent
Figure 11(a) demonstrates that the nitrogen adsorption-desorption isotherm of the rPET/sepiolite adsorbent has a Type II isotherm with an H3 hysteresis loop, indicating a complex pore structure.
50
The gradual rise in adsorption at low pressures suggests the presence of micropores and small mesopores, which are essential for initially adsorbing methylene blue. The sharp uptake at high pressures points to large mesopores and macropores, which aid in rapid mass transfer during adsorption.
20
The hysteresis loop indicates capillary condensation in mesopores, enhancing the adsorbent’s capacity. The slight divergence in the adsorption and desorption branches at lower pressures may suggest pore network effects or ink-bottle-shaped pores.
51
This isotherm reveals a hierarchical porous structure with micro-, meso-, and macropores. It is beneficial for efficient methylene blue adsorption due to its high capacity, fast kinetics, and good access to adsorption sites.
11
BET plot of rPET/Sepiolite on (a) N2 adsorption-desorption isotherms of freeze-dried BC based on the first adsorption-desorption run immediately after degassing, (b) distributions in constant pressure and adsorptive fouling experiments.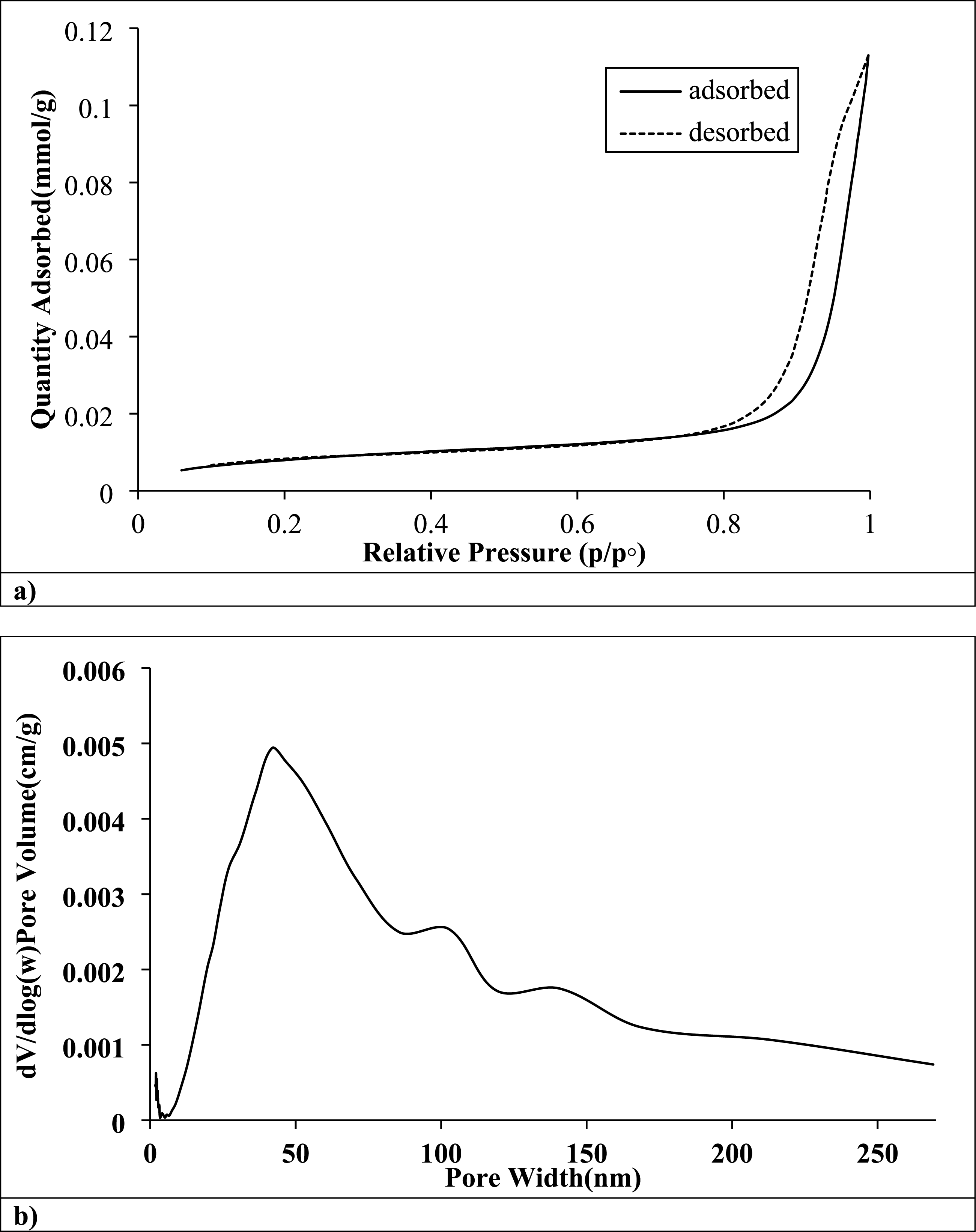
Figure 11(b) demonstrates that the pore size division of the rPET/sepiolite adsorbent has a complex and hierarchical porous structure ideal for methylene blue adsorption. The peak at around 40 nm indicates mesopores, crucial for efficient dye diffusion and adsorption. 46 Smaller pores below 20 nm suggest micropores contributing to a high surface area and rapid initial adsorption. 52 The broader distribution beyond 100 nm points to larger mesopores and macropores, facilitating mass transfer and preventing pore blockage during adsorption. 48 This multimodal pore structure, combining micro-, meso-, and macropores, is especially advantageous for MB removal, providing high capacity, fast kinetics, and good access to adsorption sites. 53 The mechanical characteristics of the material are probably improved by adding rPET to sepiolite, and functional groups may be added to enhance adsorption efficiency. 20 The gradual decrease in pore volume for pores larger than 50 nm suggests a well-controlled synthesis process, balancing surface area and pore accessibility, essential for maintaining high adsorption efficiency across different contaminant concentrations. 54
Thermogravimetric analysis (TGA)
The kinetics of degradation are shown in Figure 12 by the curves at various heating rates. Thermal analysis of rPET, Sepiolite, and rPET/sepiolite adsorbent was conducted through TGA by heating from the ambient temperature to 700°C. As shown in Figure 12(a), the significant mass loss of rPET was 70% at the temperature ranging from 380 to 460°C. This suggests that rPET has greater thermal stability because it begins to decompose at a higher temperature. This might be because rPET has a chemical structure known to have a higher heat resistance.
55
Finally, after a gradual weight reduction process, only about 10% of the rPET quantity remained resilient at 700 degrees Celsius. (a) TGA curves weight losses versus temperature and (b) derivatives weight of rPET, Sepiolite, and rPET/Sepiolite.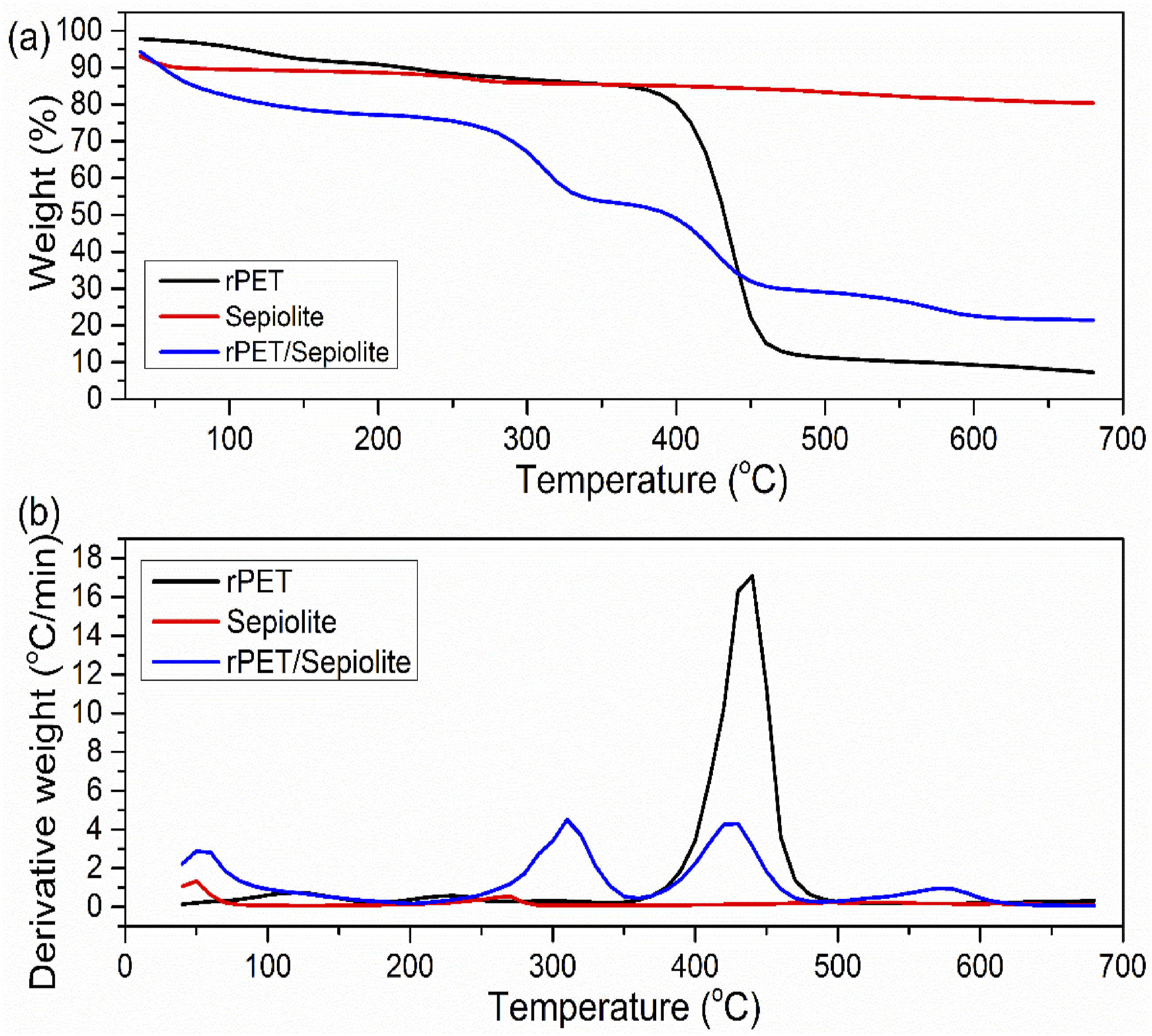
The TGA profile of sepiolite exhibited an initial weight loss of approximately 1.8% in the temperature range of 30 °C–105 °C, with the most significant decomposition occurring at 47°C. This weight reduction is primarily attributed to the evaporation of physically adsorbed water, a phenomenon widely reported in previous studies. 56 The second weight-loss stage occurred between 270 and 660°C, where 7.6% of the total mass was lost, with the peak degradation rate observed at ∼284°C. This phase is associated with intermolecular and intramolecular transformations, involving the oxidation of complex structures and forming metal oxides and spinel phases. 57 Notably, beyond 650°C, sepiolite retained approximately 81.5% of its initial mass, underscoring its exceptional thermal stability. Such robustness makes sepiolite an excellent candidate for high-temperature applications, particularly as a stable support material for catalytic and adsorption processes.
The TGA profile of the rPET/sepiolite adsorbent, depicted in Figure 12(a), revealed two primary stages of thermal decomposition. The first weight loss, attributed to moisture evaporation, occurred between 30 and 115°C, with a 4.4% mass reduction, a trend similar to that of sepiolite. However, a notable shift in peak decomposition temperature was observed, with the maximum weight loss rate at 63.4°C, significantly higher than the corresponding peak for sepiolite. This shift is likely associated with eliminating chlorine residues from dichloromethane (DCM) used during solution blending, which influenced the moisture retention behavior of the composite. 58 The second thermal decomposition stage, characteristic of rPET degradation, extended beyond 350°C, indicating a gradual thermal breakdown of polymer chains. Beyond 700°C, approximately 80% of the adsorbent’s weight remained, confirming that the incorporation of sepiolite enhanced the thermal resistance of the rPET matrix. This synergistic thermal stability suggests that the rPET/sepiolite composite inherits the robust heat tolerance of sepiolite, making it a promising material for high-temperature applications in wastewater treatment and adsorption technologies.
Furthermore, the TGA findings validate the successful incorporation of sepiolite into the rPET matrix, reinforcing the composite structure and minimizing premature thermal degradation. The relatively high residual mass also indicates strong interfacial interactions between rPET and sepiolite, which could improve the adsorption efficiency and long-term durability of the adsorbent under various environmental conditions.
Fourier transform infrared spectroscopy (FTIR)
The FTIR spectra of rPET, sepiolite, and rPET/sepiolite adsorbent were recorded with a 4000 – 400 cm-1 wavelength range, as shown in Figure 13. In rPET, the ester linkage is the most significant feature, leading to strong carbonyl (C = O) and C-O stretching vibrations. These groups are essential for the polyester’s mechanical properties, crystallinity, and thermal stability.
59
In contrast, sepiolite’s FTIR spectrum is dominated by silicate-related vibrations (Si-O-Si) stemming from its clay mineral structure. The O-H stretching vibrations indicate the presence of hydroxyl groups, essential for the adsorption properties of sepiolite.
60
The interaction between Mg and OH in the sepiolite structure gives rise to specific peaks that differentiate it from other silicates. rPET/sepiolite’s adsorbent spectra show additional bands due to organic functional groups (e.g., CH2 or CH3 groups from organic modifiers), further altering the FTIR spectrum. These modifications often aim to improve the dispersion of sepiolite in polymer matrices or enhance its compatibility with organic phases. The FTIR spectra of rPET/sepiolite adsorbent reveal their respective functional groups, which are closely tied to their chemical compositions and structural characteristics. IR spectra of rPET, sepiolite, and rPET/sepiolite adsorbent.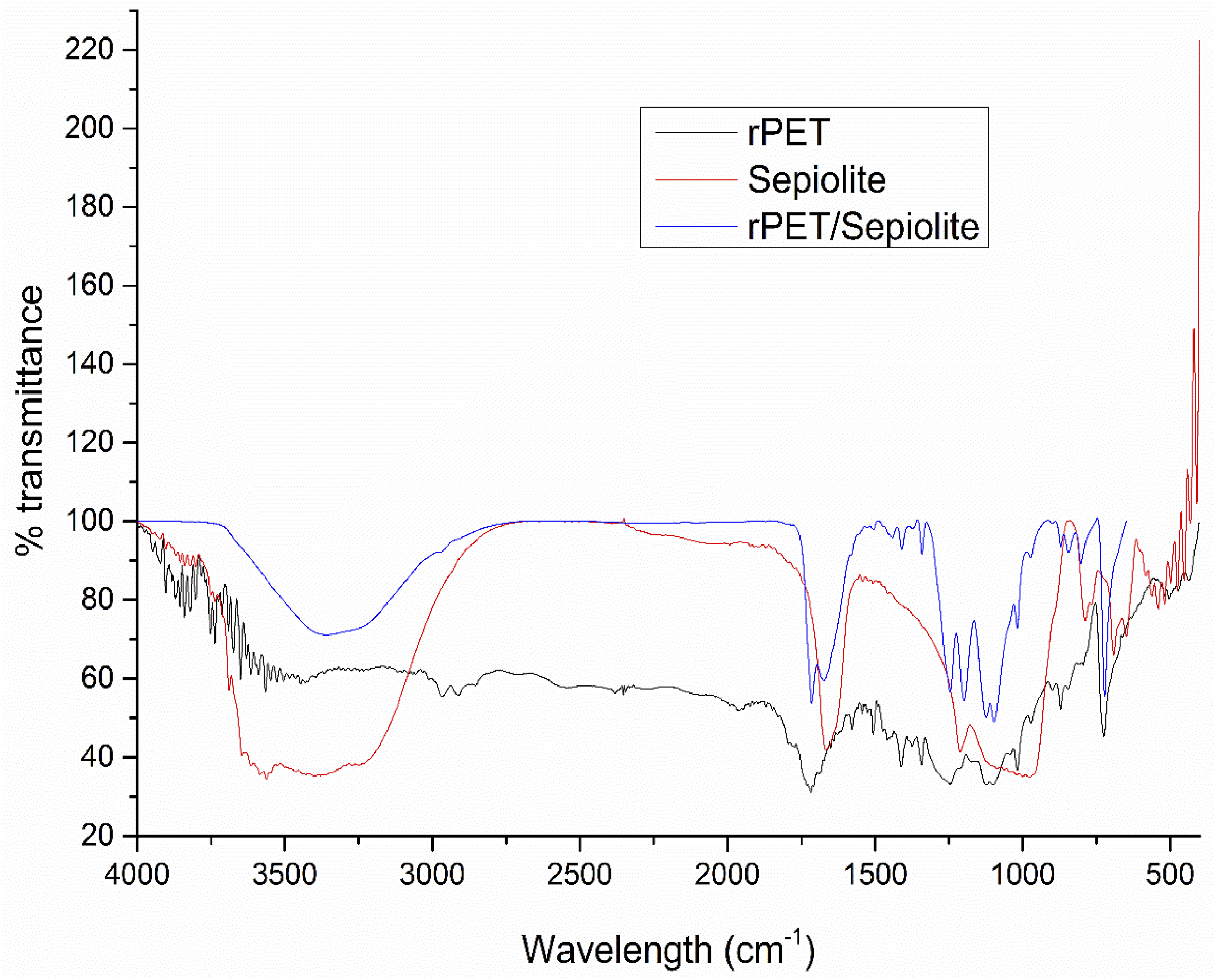
In the spectrum of sepiolite, a broad absorption band centered around 3430 cm−1 corresponds to the O–H stretching vibrations of surface silanol (Si-OH) groups and adsorbed water. This band is also visible in the composite but appears slightly shifted and less intense, suggesting hydrogen bonding interactions between the hydroxyl groups of sepiolite and the ester functionalities in rPET. The rPET spectrum shows a distinct peak near 1715 cm−1, assigned to the C = O stretching vibration of ester groups in the PET backbone. 59 This peak is retained in the composite but exhibits minor shifts and intensity changes, indicating possible dipole-dipole or hydrogen bonding interactions between rPET and sepiolite. In the 1010–1040 cm−1 region, strong Si–O–Si stretching bands appear in both the sepiolite and composite spectra, confirming the structural integrity of sepiolite within the blend. 60 The fingerprint region below 800 cm−1 shows overlapping bands originating from Si–O bending in sepiolite and out-of-plane C–H bending in rPET, further supporting successful incorporation. These functional groups collectively facilitate MB dye adsorption through electrostatic attraction between negatively charged Si–O- groups and cationic MB molecules, hydrogen bonding via surface hydroxyls, and potential π–π interactions between the aromatic rings of MB and PET. Therefore, the FTIR results support the role of surface functionalities and interfacial interactions in enhancing the adsorption performance of the rPET/sepiolite composite.
Zeta potential
The isoelectric point (IEP) of rPET is essential in understanding its surface charge behavior in aqueous environments, especially for applications involving adsorption, catalysis, and other interfacial interactions. The IEP of PET can vary depending on its processing history and degree of recycling, as these factors influence surface functional groups and, consequently, the surface charge.
Studies indicate that the IEP of PET generally falls in the acidic pH range, around pH 3-4, though this can vary based on surface modifications and degradation during recycling. For instance, when PET undergoes recycling, ester linkages at the surface can hydrolyze, exposing carboxyl and hydroxyl groups that shift the IEP towards lower pH values. 61 The PET surface is positively charged at pH values below the IEP and negatively charged above, which affects how it interacts with charged species in solution. 62
These surface properties are crucial when recycled PET is used in applications like wastewater treatment, where the interaction with contaminants can depend heavily on the pH relative to the IEP. Consequently, modifying the IEP through surface treatments or blending recycled PET with other polymers can help tailor its surface charge properties for specific applications. 63
The IEP of rPET/sepiolite adsorbent represents a critical parameter in understanding their surface properties and adsorption behavior. When the zeta potential equals zero, the surface charge neutrality occurs, and the material exhibits minimal electrostatic stability. Based on Figure 14, the IEP of rPET/sepiolite adsorbents typically falls in the acidic pH range, usually between pH 2-4. However, this can differ based on the specific surface modifications and processing conditions used in recycling PET and incorporating sepiolite. The surface charge characteristics of these adsorbents are fascinating because they combine the properties of both rPET, which tends to have a negative surface charge at neutral pH due to exposed carboxyl groups, and sepiolite, a natural magnesium silicate with a unique fibrous morphology that contributes to its surface charge distribution. Incorporating sepiolite into rPET matrices can significantly alter the IEP compared to either material alone, as the clay mineral’s surface hydroxyl groups and structural magnesium ions influence the overall surface charge behavior. Zeta potential of rPET/sepiolite adsorbent.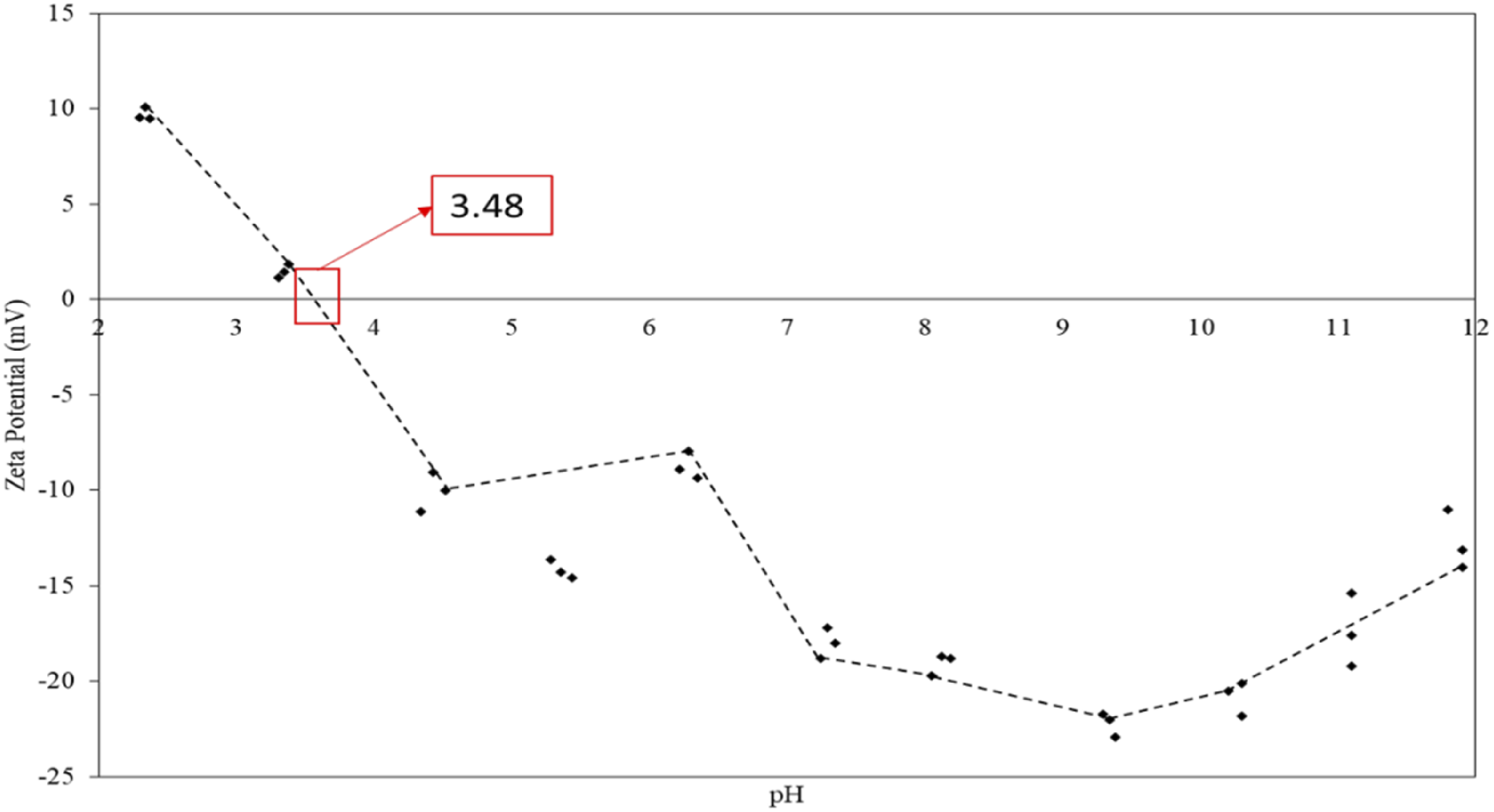
Conclusions
This research effectively developed a novel adsorbent using rPET and sepiolite via solution blending to eliminate MB dye from aqueous solutions effectively. The optimal preparation conditions were determined to be 10 wt% rPET with 0.25 g of sepiolite blended for 1 hour. This resulted in an adsorbent with a hierarchical porous structure that significantly enhanced adsorption capacity, achieving a maximum removal efficiency of 96.7%. Characterization analyses, including SEM, mapping, BET, TGA, FTIR, and zeta potential, confirmed the adsorbent’s improved thermal stability, increased surface area, and successful integration of sepiolite into the rPET matrix. These properties contributed to its high adsorption performance, making it a viable option for sustainable wastewater treatment.
The findings demonstrate the potential of using recycled materials, such as PET, to create effective adsorbents, providing an environmentally friendly and cost-efficient solution to address plastic waste and water pollution. This approach supports circular economy principles by repurposing waste materials for environmental remediation. Future research should explore the application of rPET/sepiolite adsorbent in treating various dyes and pollutants, evaluate their long-term stability and reusability, and assess their performance in real industrial wastewater conditions. Overall, this study contributes to advancing sustainable materials for environmental management, promoting a greener approach to water pollution control.
From an environmental standpoint, using recycled PET (rPET) and Sepiolite to fabricate adsorbents presents a sustainable and eco-conscious approach to wastewater treatment. rPET, derived from post-consumer plastic waste, reduces the environmental burden associated with plastic pollution and contributes to resource conservation and circular economy practices by revalorizing waste materials into functional adsorbents. On the other hand, sepiolite is a naturally occurring clay mineral known for its non-toxicity, high surface area, and chemical stability. It is abundantly available and has minimal environmental impact during extraction and use. Moreover, both rPET and sepiolite can be regenerated and reused, reducing the need for frequent replacement and disposal. Combining these materials into a hybrid adsorbent offers an environmentally benign alternative to synthetic or heavily processed adsorbents, aligning well with sustainable development goals and current efforts to mitigate water pollution using green materials.
Footnotes
Acknowledgements
This work was supported by the Deanship of Scientific Research, Vice Presidency for Graduate Studies and Scientific Research at King Faisal University, Saudi Arabia (Grant No.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Deanship of Scientific Research, Vice Presidency for Graduate Studies and Scientific Research at King Faisal University, Saudi Arabia Grant No. (KFU242442). The authors extend their appreciation to Abdulmonem Alrashed Humanitarian Foundation (FSP-2-0017).
Data Availability Statement
The raw data for reproducing these findings are supplied with the article. However, it should only be shared on request until the project is completed.