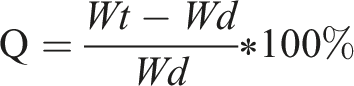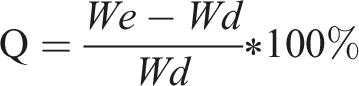Abstract
The traditional freeze-drying method for preparing polyvinyl alcohol (PVA) hydrogels involves a complex process. Conventional one-step drying processes for preparing high-strength PVA hydrogels often yield materials with inadequate mechanical properties and insufficient lubrication. To overcome these limitations, this study introduces polydopamine-modified graphene as a reinforcing nanofiller within PVA hydrogels. The surface modification significantly improves graphene dispersion in the hydrogel matrix, leading to substantial enhancements in mechanical performance. Incorporating just 0.5 wt% polydopamine-modified graphene results in a remarkable 200% increase in tensile strength and a 62% improvement in Young’s modulus compared to unmodified PVA hydrogels. Furthermore, subsequent treatment with a straightforward deep eutectic solvent (DES) and UV-initiated crosslinking synergistically boosts the hydrogel’s overall strength by 500% and reduces its coefficient of friction by 50%, significantly enhancing lubrication properties. This combined strategy of nanomaterial reinforcement and solvent/UV processing presents a facile and effective approach for fabricating robust, high-strength PVA hydrogels with excellent lubricity, holding promise for demanding biomedical and engineering applications.
Introduction
Hydrogels, with their three-dimensional network structures and high water content, are commonly found in biological systems. 1 Recent research has explored the development of biocompatible hydrogels using various materials, including proteins, 2 cellulose, 3 and chitosan.4–6 Due to their unique properties—such as flexibility, biocompatibility, and high water retention—hydrogels have found widespread applications in electronic devices, 7 sensors, 8 and biomedical fields. 9 Among them, vinyl polymer hydrogels like polyvinyl alcohol (PVA), polyacrylamide, and polyacrylic acid stand out for their exceptional mechanical strength and biodegradability.10–12 PVA, a water-soluble polyhydroxy polymer synthesized from vinyl acetate via alcoholysis and hydrolysis, offers excellent biocompatibility, permeability, hydrophilicity, and a low friction coefficient.10,13 These properties make PVA hydrogels highly suitable for applications in artificial organs, wound dressings, shape memory systems, antibacterial materials, flexible sensors, and controlled drug delivery.14–19
However, conventional fabrication methods face significant trade-offs between performance, biocompatibility, and processing efficiency. Physical cross-linking methods for synthesizing PVA-based hydrogels include freeze-thaw cycles, salting-out, and gradient cryogenics.15,20,21 These techniques primarily rely on molecular interactions such as physical entanglement, hydrogen bonding, and hydrophobic and electrostatic forces to establish connectivity.22,23 Hydrogels formed through these weak interactions are typically soft, flexible, self-healing, and free from toxic cross-linker residues, making them highly suitable for biomedical applications, yet suffers from low tensile strength (typically ≤2.3 MPa), protracted processing (>24 h), and structural heterogeneity that compromises optical transparency. Chemical cross-linking enhances mechanical properties and enables rapid in situ gelation, 24 but residual toxic cross-linkers (e.g., glutaraldehyde) pose biocompatibility risks and necessitate complex purification. Radiation cross-linking offers high purity and sterilization benefits but requires expensive equipment and often reduces long-term creep resistance. 25 The hydrogel preparation process, regardless of the method, is often complex. Recently, researchers have introduced a cast-dry method, where PVA hydrogel solutions are directly dried.26,27 Dehydration and drying induce the densification of PVA polymer chains, leading to the formation of crystalline domains. Rehydration endows PVA hydrogels with excellent hydration lubrication properties. 27 This approach offers a simple, non-toxic alternative. However, the hydrogels produced using this method tend to exhibit lower mechanical strength and poorer frictional performance. Therefore, finding ways to enhance the properties of these hydrogels is crucial for simplifying preparation and expanding their range of applications.
Incorporating cross-linking additives into PVA solutions provides a simplified method for preparing hydrogels with enhanced properties.28,29 These additives act as physical cross-linking sites, and their increased specific surface area compensates for the reduced cross-linking density in the polymer. 30 This approach minimizes energy dissipation, allowing the material to endure substantial deformations while improving its mechanical properties. However, achieving uniform dispersion of non-biological additives within the hydrogel remains a significant challenge.
To enhance the performance of hydrogels prepared via the cast-dry method, we propose the utilization of dopamine-modified graphene to synthesize polydopamine-modified graphene-doped PVA hydrogels through a one-step drying process. This method yields hydrogels with significantly improved mechanical properties and lubrication. Surface modification of the graphene results in an increased number of hydrogen bonding sites, while the coupling interactions among polymer units effectively transmit and distribute stress and strain. This leads to elastomers and gels with superior toughness, strength, and robustness. Incorporation of various amounts of polydopamine-modified graphene results in a 76% increase in tensile strength and a 62% enhancement in Young’s modulus. Furthermore, treating the hydrogels prepared by the one-step method with deep eutectic solvents further augments their mechanical strength and lubrication properties.
Experiment details
Chemical reagent
PVA (polymerization degree 1750 ± 50), were purchased from Sinopharm Chemical Reagent (Shanghai) Co., Ltd. Acrylic Acid (AAc), Sodium Chloride (NaCl) Choline Chloride (ChCl), Tris(hydroxymethyl)aminomethane (Tris), and Photoinitiator 2959 was purchased from Shanghai Aladdin Biochemical Technology. Graphene was purchased from Shanghai Wokai Biotechnology Co., Ltd. Hydrochloric Acid (HCl) was purchased from Shanghai Zhenqi Chemical Reagent. Dopamine Hydrochloride (DA) was purchased from Shanghai Macklin Biochemical Technology.
Preparation of polydopamine-modified graphene (PDA-G)
Graphene was ultrasonically cleaned with deionized water for 30 min to remove surface impurities and ensure uniform dispersion in the solution. It was then immersed in dopamine buffer solution for 24 h for deposition. The deposited substrate was centrifuged to remove unreacted chemicals and washed multiple times with deionized water. The modified graphene was subsequently dried in vacuum oven at 60°C.
Preparation of PVA hydrogels
Preparation of PVA hydrogel
PVA dissolves in deionized water at 90°C (with a concentration of 90 wt%). Subsequently, the PVA hydrogel was poured into a petri dish and dried in an oven at 60°C for 48 h. The dried hydrogel was immersed in deionized water to reach swelling equilibrium before further performance testing.
Preparation of PVA-PDA-G hydrogel
Different concentrations (0.1 wt%, 0.3 wt%, 0.5 wt%) of PDA-G were weighed and placed into 10 wt% PVA solution. To form dispersions. The resulting homogeneous solution was poured into a petri dish and films were formed at 60°C until weight equilibrium was reached. These films were subsequently peeled off and soaked in deionized water for further performance evaluation. The PVA -G hydrogel was prepared refer to the above method.
Preparation of SE-PVA, SE-PVA-G, SE-PVA-PDA-G hydrogels
A deep eutectic solvent (DES) was first prepared by mixing acrylic acid (AAc) and choline chloride (ChCl) at a molar ratio of 3:1, followed by ultrasonication for 30 min until the mixture became uniform and transparent. The PVA hydrogels with optimum mechanical properties from varying content modified graphene were selected, along with pure PVA and graphene-doped hydrogels, for immersion in the DES. The PVA, PVA-G, and PVA-PDA-G hydrogels were soaked in the AAc/ChCl DES for 24 h to replace the water, and were named as SE-PVA, SE-PVA-G, and SE-PVA-PDA-G, respectively, before mechanical testing.
Preparation of SE-C-PVA-PDA-G hydrogel
The PVA-DG hydrogel was immersed in a DES containing 1 wt% photoinitiator 2959 for 24 h and then exposed to UV light for 10 min to achieve crosslinking. The hydrogel was named SE-C-PVA-PDA-G.
Microstructure characterization of PVA hydrogel
Samples of PVA-G, PVA-PDA-G, SE-PVA, SE-PVA-G, and SE-PVA-PDA-G hydrogels were prepared and allowed to swell to equilibrium in deionized water. They were then treated by vacuum freeze-drying for 12 h. Scanning electron microscopy was performed under vacuum to analyze the microstructure of the samples, comparing the different types of hydrogels to select the group with the most optimal morphology.
X-ray diffraction is utilized to determine the crystal structure by analyzing the diffraction patterns between X-rays and crystals. By comparing the X-ray diffraction patterns of PVA hydrogel with those of standard substances, a qualitative analysis of the phase composition of the PVA hydrogel can be performed. Quantitative analysis of the phase composition can be achieved by analyzing the diffraction intensity data of the PVA hydrogel.
Fourier Transform Infrared Spectroscopy (FT-IR) generates an absorption spectrum based on the absorption of specific wavelengths of infrared light by the vibrational transitions of bonded atoms in a compound’s molecules. This technique is primarily used for structural analysis, qualitative identification, and quantitative analysis. FT-IR is employed for structural analysis and qualitative assessment of the hydrogel.
Swelling and water content test of PVA hydrogel
The hydrogel samples were initially dried at 60°C to a constant weight and recorded as Wd. Subsequently, the hydrogels were immersed in deionized water, with samples taken at intervals of 5, 10, 15, 30, and 60 min. After removing surface water, the samples were weighed and recorded as Wt. The process continued until the weight stabilized as We. Swelling ratios were calculated, swelling curves plotted, and the hydrogel with the optimal swelling performance was identified.
The swelling ratio (Q) was calculated using the following formula:
The Water Content (H) was calculated using the following formula:
Tensile performance test of PVA hydrogel
The WDW-2 Microcomputer-controlled electronic universal testing machine was used to conduct uniaxial tensile and cyclic tensile experiments on the hydrogel sample. The size of the samples to be tested was 60 mm × 7 mm × 2 mm. The experimental rates of uniaxial stretching and cyclic stretching were both 10 mm/min. Under cyclic stretching conditions, the strain was set to 100%, and cyclic loading and unloading were performed 10 times. The data was recorded to obtain the stress-strain curve.
Friction performance test of PVA hydrogel
UMT-Ⅱ (American) multifunctional friction and wear testing machine was used to test the sliding tribological properties of hydrogel. 6 mm zirconia ball served as a counterpart.
Results and discussion
The dispersion of graphene materials in heterogeneous matrices is fundamentally challenged by their inherent stacked lamellar configuration and strong interlayer van der Waals forces. Compounding this limitation, the paucity of surface functional groups in pristine graphene induces interfacial incompatibility with polymeric systems, substantially impeding effective matrix adhesion. In this study, surface treatment of graphene using polydopamine (PDA) modification was employed to treat graphene surfaces, significantly reducing van der Waals forces between graphene layers and enhancing compatibility with polymers, resulting in uniformly dispersed graphene-polymer composites. 31 The chemical structure of both graphene and dopamine-modified graphene was characterized. Figure S1 displays the infrared spectra of PDA-G and G. The peak at 1690 cm−1 corresponds to the stretching vibration of the C = O bond in the amide groups of polydopamine, and the peak at 780 cm−1 corresponds to the bending vibration of the C-H bond in the aromatic ring. Figures S2 illustrated the dispersion states, showing that GR began to precipitate and disperse unevenly in water over time, whereas PDA-G maintained good dispersion. This indicates that modified graphene is more uniformly and stably dispersed, demonstrating successful surface modification by polydopamine.
To enable facile fabrication of high-performance slippery hydrogels, we developed a synergistic strategy integrating nano-doped reinforcement with drying-induced densification, as schematically presented in Figure 1. Specifically, polydopamine-modified graphene is used as a physical cross-linker, while the densification and crystallization of PVA polymer chains are induced by dehydration during drying, followed by rehydration. In detail, a PVA sol containing polydopamine-modified graphene is dehydrated and dried at room temperature to form a PVA xerogel (Figure 1(c)). During the dehydration and shrinkage process, the rearrangement of the polymer network leads to the formation of dense crystalline domains. The dopamine structure in PDA contains abundant functional groups, such as phenolic hydroxyl (-OH) and amine (-NH2), which form strong hydrogen bonding interactions with the hydroxyl (-OH) groups of PVA chains, thereby enhancing the cohesion of the hydrogel. Schematic illustration of PVA(a), PVA-G (b)and PVA-PDA-G(c) hydrogels preparation.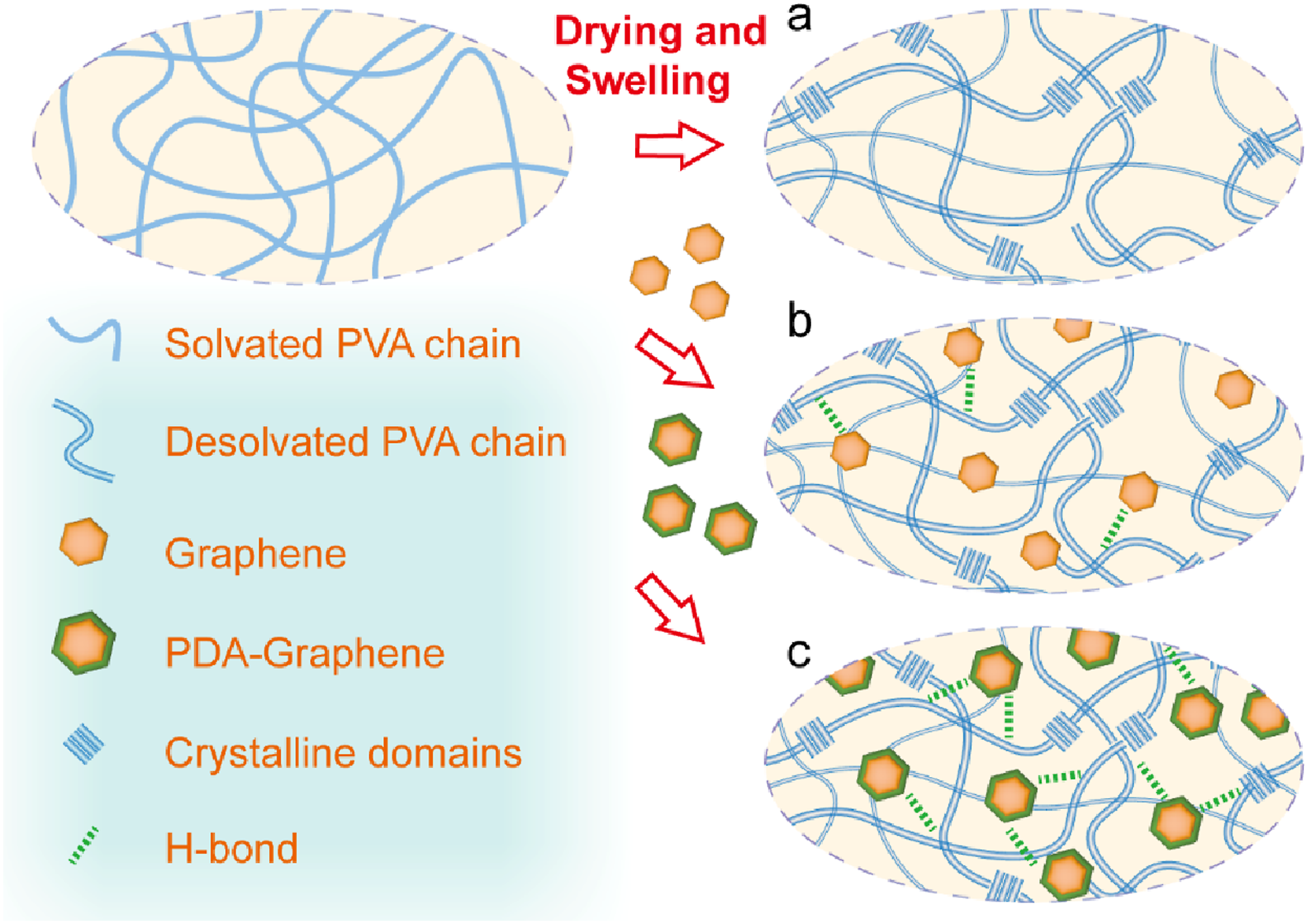
X-ray diffraction (XRD) and Raman patterns were utilized to confirm the presence of crystalline domains and the successful incorporation of graphene in the hydrogels. As shown in Figures S3 and S4, all PVA hydrogels display a distinct diffraction peak at 2θ = 19.7°, corresponding to the (101) plane of semi-crystalline PVA. Furthermore, two minor peaks at 2θ = 22.9° and 40.8° correspond to the (200) and (102) planes of PVA crystalline domains, respectively, highlighting the existence of ordered crystalline structures within the hydrogels. Notably, the characteristic graphene peak at 2θ = 25.77° is retained in the graphene-doped hydrogels, indicating effective dispersion of graphene throughout the hydrogel matrix. The Raman characterization results are shown in Figure S4. Compared to the PVA hydrogel, both PVA-G and PVA-PDA-G hydrogels display characteristic graphene peaks at 1580 cm−1 and 2700 cm−1. The peak at 1580 cm−1 corresponds to the G peak, which is associated with the vibration of sp2 carbon atoms in graphene. The peak at 2700 cm−1 represents the 2D peak, confirming the successful incorporation of graphene into the hydrogel. However, the peak shapes of PVA-G and PVA-PDA-G hydrogels differ noticeably, which can be attributed to the modification of the graphene surface by polydopamine.
To investigate the microstructural changes in PVA hydrogels with the addition of modified graphene, scanning electron microscopy (SEM) was employed to examine the microstructures of PVA hydrogel, PVA-G hydrogel, and PVA-PDA-G hydrogel. As shown in Figure 2, PVA-G hydrogels doped with graphene exhibited smaller pore structures compared to the pristine PVA hydrogel. The modified graphene-doped hydrogel exhibited refined pore architecture due to hydrophilic moieties on functionalized graphene surfaces acting as supplementary crosslinking sites. This surface modification substantially elevates crosslink density within the hydrogel network. This substantially increases the crosslink density of the hydrogel network. From a microstructural perspective, the incorporation of modified graphene enhances the network density while reducing the pore size, leading to a more compact and robust hydrogel structure. SEM pictures of PVA(a), PVA-G (b) and PVA-PDA-G(c) hydrogels.
The drying, reswelling, and rehydration processes of hydrogels play pivotal roles in determining their storage stability and lubrication performance. The degree of crosslinking significantly impacts the swelling behavior of hydrogels. Upon doping with graphene, the hydrogel exhibited a swelling ratio of 210%, which was comparable to that of pure PVA. However, the incorporation of polydopamine-modified graphene enhances the swelling ratio, which increased further with higher concentrations, reaching up to 230%. As illustrated in Figure 3(b), the water content of the hydrogel increased from 48% to 57%. The densification of the polymer network makes it difficult for water molecules to infiltrate and fill the polymer chains during the rehydration process of the hydrogel. The same principle applies to unmodified graphene, which forms only limited crosslinks with PVA molecular chains and struggles to disrupt the crystalline domains of PVA. As a result, the water content and swelling ratio of PVA hydrogels remained relatively low. However, with the incorporation of hydrophilic modified graphene, the functional groups on the graphene surface interact with the hydrogel’s molecular chains through hydrogen bonding. This interaction releases some polymer chains that originally formed crystalline domains, allowing them to be filled with water molecules. Consequently, the swelling ratio and water content increase with the addition of modified graphene. Swelling curve and water content of hydrogels.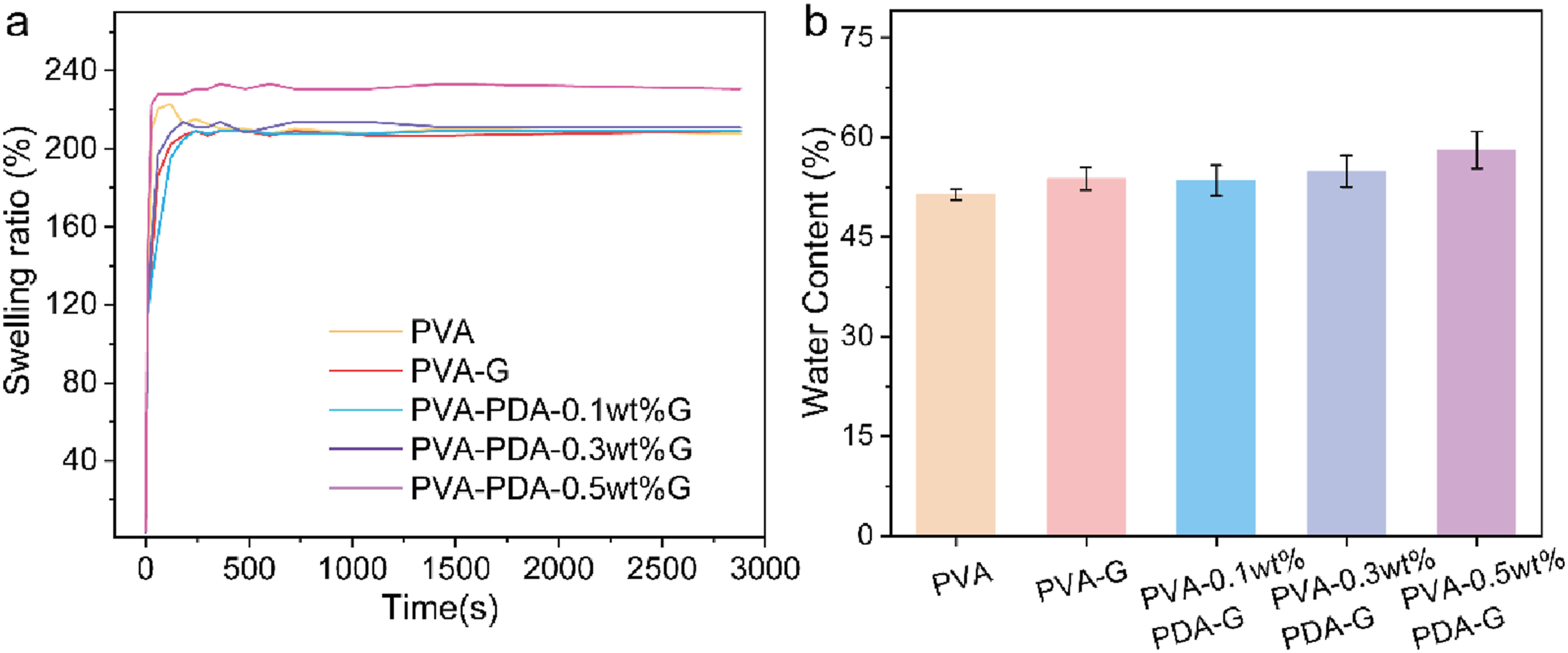
The mechanical properties of hydrogels are critically influenced by the introduction and surface modification of nanofillers, as demonstrated by the stress-strain curves in Figure 4(a). Incorporation of unmodified graphene (PVA-G) enhanced the tensile strength of PVA hydrogels from 2 MPa to 3.2 MPa and fracture strain from 410% to 590%, while polydopamine-modified graphene (PVA-PDA-G) further elevated the tensile strength to 4.2 MPa. This reinforcement is attributed to the layered graphene architecture restricting polymer chain slippage and delaying network fracture, thereby improving ductility and load-bearing capacity.
11
Additionally, the hydrophilic PDA modification significantly increases interfacial contact area and introduces supplementary crosslinking sites, promoting a uniform, dense network topology with enhanced crosslink density that optimizes tensile strength.
32
Corresponding modulus evolution (Figure 4(c)) revealed that unmodified graphene induced only a marginal modulus increase, whereas PDA surface functionalization dramatically elevated the compressive modulus by 400% (from 3.2 MPa to 16 MPa), underscoring the pivotal role of nanofiller-matrix compatibility in maximizing stress transfer efficiency. Compared with currently reported PVA hydrogels fabricated via simple preparation methods, the PVA-PDA-G hydrogel exhibits superior mechanical properties.33,34 Tensile curve (a), elongation at break (b) and elastic modulus (c)of different hydrogels; tensile curve (d), elongation at break (e) and elastic modulus (f) of PVA-PDA-G hydrogels with different concentrations of PDA-G.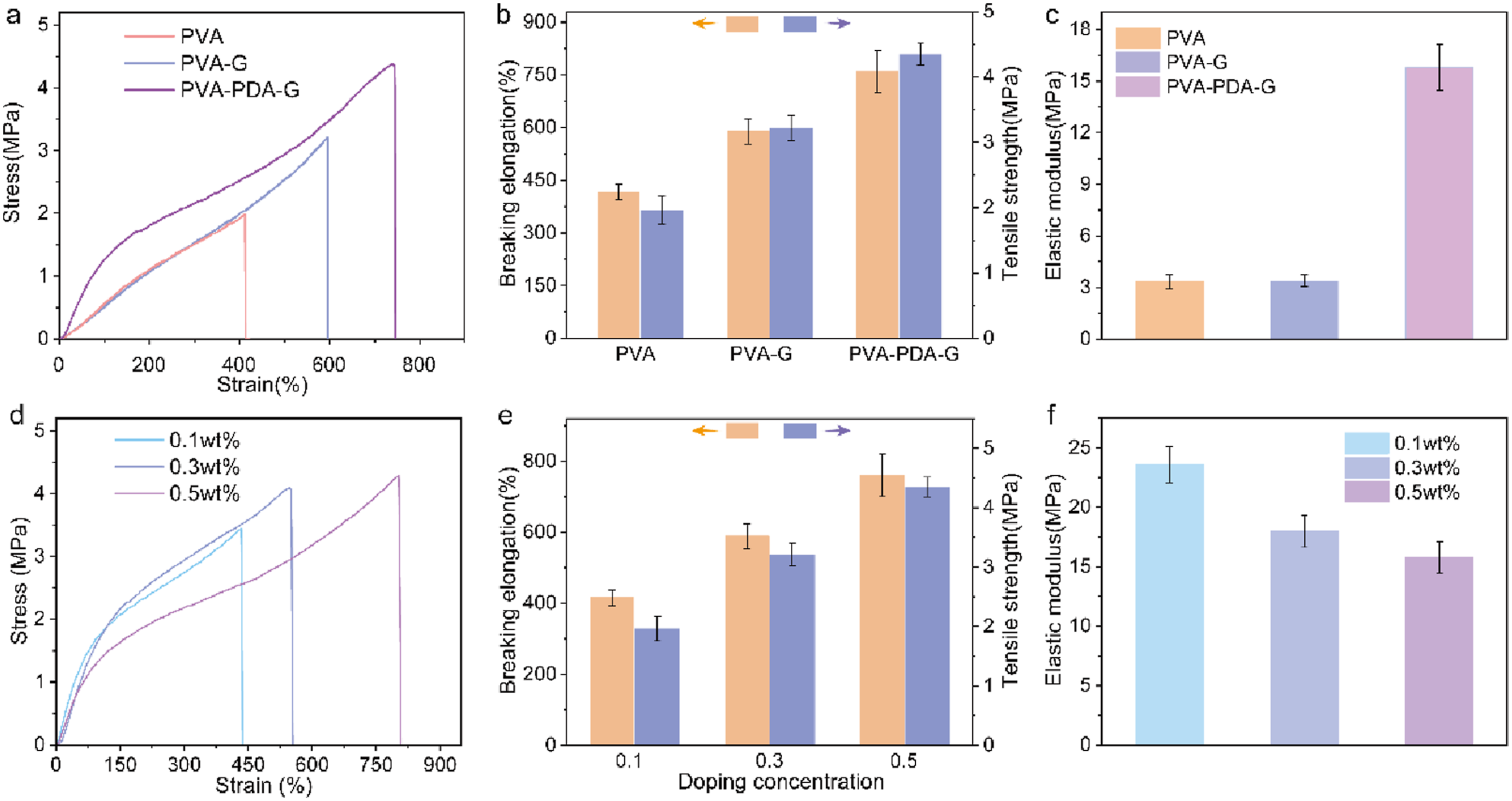
Hydrophilic modified graphene possesses excellent mechanical properties and can be uniformly dispersed within the PVA matrix, forming strong interfacial interactions with polymer chains. Under external stress, graphene effectively transfers and distributes the load, enhancing the tensile strength of the material. The two-dimensional layered structure of graphene serves as a skeleton within the matrix, preventing the propagation of microcracks and improving the material’s resistance to damage. Moreover, modified graphene can act as a template, guiding the alignment of PVA chains and thereby increasing the elongation at break. The addition of flexible graphene sheets also mitigates stress concentration during stretching, further improving the material’s ductility.
To explore optimal graphene doping, hydrogels with 0.1 wt%, 0.3 wt%, and 0.5 wt% modified graphene were prepared and mechanically tested. Figure 4(d) and (e) compared the tensile strength and elongation at break. The strength of PVA-PDA-0.1 wt%G was 3.41 MPa with 435 % elongation; PVA-PDA-0.3 wt%G showed 4.08 MPa and 545%; PVA-PDA-0.5 wt%G reached 4.31 MPa and 809%. As dopant concentration increased, both strength and elongation enhanced significantly. With the increase in the concentration of modified graphene, more physical crosslinking points are formed within the hydrogel. The hydroxyl and other functional groups on the surface of hydrophilic modified graphene interact with PVA molecular chains through hydrogen bonding, creating additional crosslinking points that enhance the mechanical properties and extensibility of the matrix. Moreover, the incorporation of graphene improves the uniformity and density of the hydrogel’s internal structure, significantly boosting its tensile strength.
As the concentration of modified graphene increased, the modulus of the hydrogel showed a decreasing trend (Figure 4(f)). This may be due to the hydrophilic modified graphene at high concentrations acting similarly to a plasticizer within the hydrogel, enhancing the mobility of molecular chains and potentially interfering with the crystallization process of PVA chains. The hydrophilic nature of the modified graphene may also increase the flexibility of hydrogel, making the matrix more prone to deformation. Additionally, the presence of graphene could promote localized hydration effects, reducing the compactness of the polymer network.
A key application of lubricating hydrogels lies in biological lubrication. This study aims to develop hydrogels with superior lubricating properties using a simple and efficient approach. Tribological characterization was performed to quantitatively evaluate the aqueous lubrication performance and underlying mechanisms of hydrated PVA hydrogels under controlled conditions (sliding frequency: 1 Hz, applied load: 10 N, water lubricant). As delineated in Figure 5(a), the polydopamine-modified graphene composite hydrogel (PVA-PDA-G) exhibited a substantially reduced friction coefficient of 0.125, representing an approximately 34.2% reduction compared to pristine PVA hydrogel (μ = 0.19). In contrast, both unmodified PVA and unmodified graphene-reinforced hydrogels (PVA-G) demonstrated significantly higher friction coefficients. This pronounced tribological enhancement is attributed to the synergistic effects of PDA modification, which improves interfacial compatibility and load-bearing capacity. Critically, the inherent low mechanical strength of pure PVA hydrogels induces excessive deformation under applied load, thereby increasing the real contact area and frictional resistance. The PDA-functionalized graphene reinforcement mitigates this deformation through effective stress distribution, establishing a stable boundary lubrication regime that optimizes friction reduction.
35
This reinforcement mechanism renders the PVA-PDA-G hydrogel tribologically superior to equivalent reinforced PVA hydrogels within the nanofiller-enhanced series.36,37 For PVA-PDA-G hydrogels with superior mechanical strength, a dense internal network structure reduces contact area during friction, thereby lowering resistance caused by elastic deformation.11,38 Pure PVA hydrogels lack surface-active functional groups and lubrication-enhancing materials, so their lubricating performance mainly relies on water lubrication. However, the water film is easily disrupted during sliding, leading to a higher friction coefficient. Unmodified graphene has poor dispersion in the hydrogel, tending to aggregate, which prevents the formation of a uniform lubrication network in the matrix and weakens the friction-reducing effect. The surface of unmodified graphene lacks active functional groups, such as those in polydopamine-modified graphene, which would enhance water retention and improve interfacial lubrication. Although these hydrogels have not yet reached ideal lubrication, they effectively reduce friction in one-step drying method hydrogels. Lubrication properties of slippery PVA hydrogels. Friction test curves (a) and corresponding average coefficient of friction of different PVA hydrogel (b); Friction test curves (c) and corresponding average coefficient of friction (d) of PVA-PDA-G hydrogel with different concentrations of PDA-G.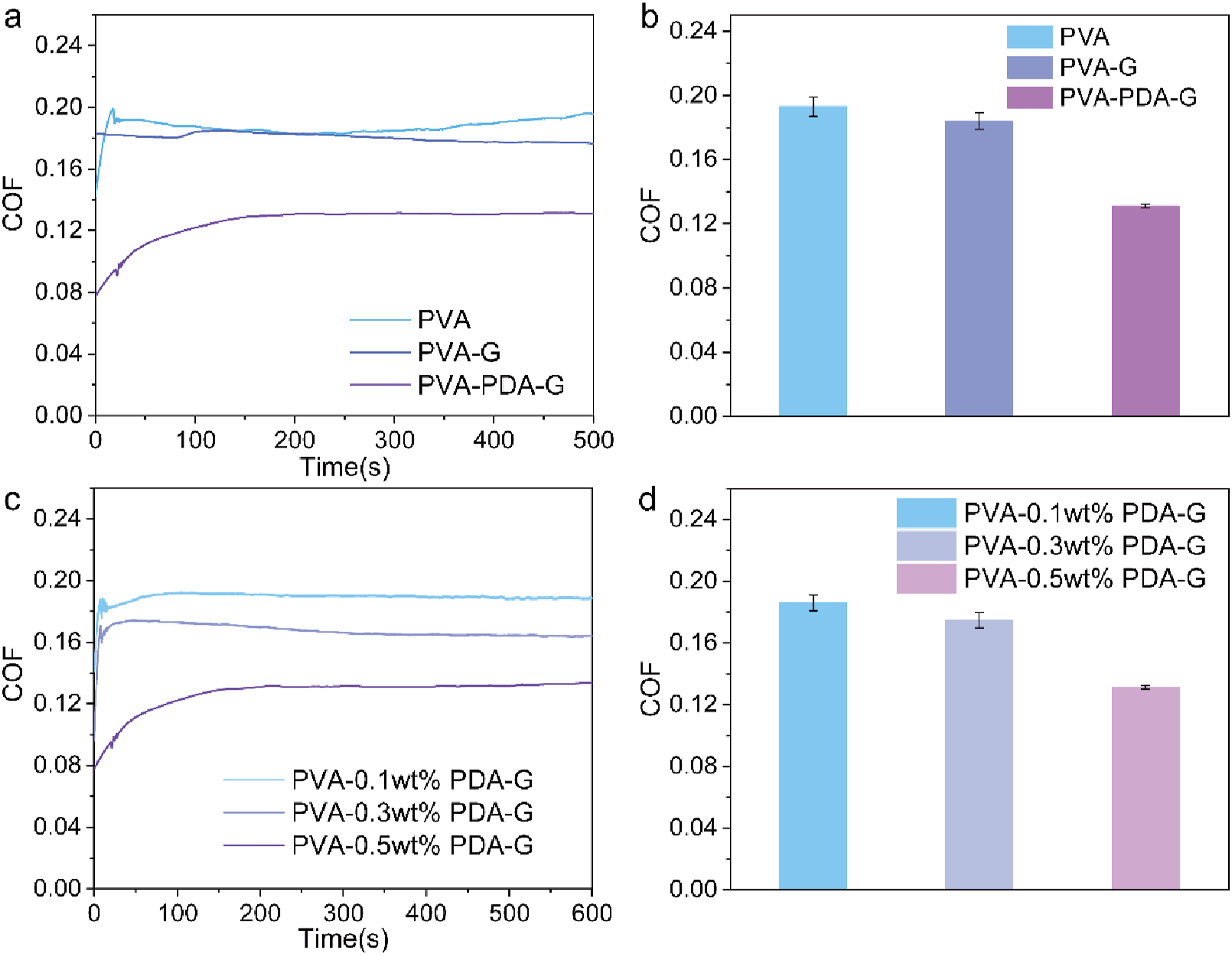
Further tribological testing on 0.1 wt%, 0.3 wt%, and 0.5 wt% modified graphene-doped hydrogels indicated that friction coefficients decrease as graphene concentration increases. At 0.3 wt% and 0.1 wt%, coefficients are 0.19 and 0.17, respectively. At 0.5 wt%, PVA-PDA-G hydrogels reached the lowest friction coefficient of 0.125. Although high-concentration hydrogels have a lower modulus, their increased strength allows them to better distribute and alleviate localized stress concentration during friction. This helps prevent premature surface damage, further reducing friction. As the content of modified components in the hydrogel increases, the interfacial lubrication and water retention properties are enhanced, leading to a more pronounced water-lubricating effect and a significant reduction in the friction coefficient.
Hydrogels are often applied in various liquid environments, prompting an evaluation of their friction behavior in different media. Figure 6 presents the friction test results for the PVA-PDA-0.5 wt%G hydrogel under constant conditions of 10 N and 1 Hz. The findings show that when deionized water is used as the friction medium, the friction coefficient of the hydrogel remains relatively high at 0.136. However, a significant reduction in the friction coefficient is observed when simulated body fluid (SBF) and calf serum are used, with calf serum providing the lowest value. Friction test curves (a) and corresponding average coefficient of friction (d) of PVA-PDA-G hydrogel at different water lubricants. Friction test curves (b) and (e) corresponding average coefficient of friction of PVA-PDA-G hydrogel at different applied frequency. Friction test curves (c) and (f) corresponding average coefficient of friction of PVA-PDA-G hydrogel at different applied load.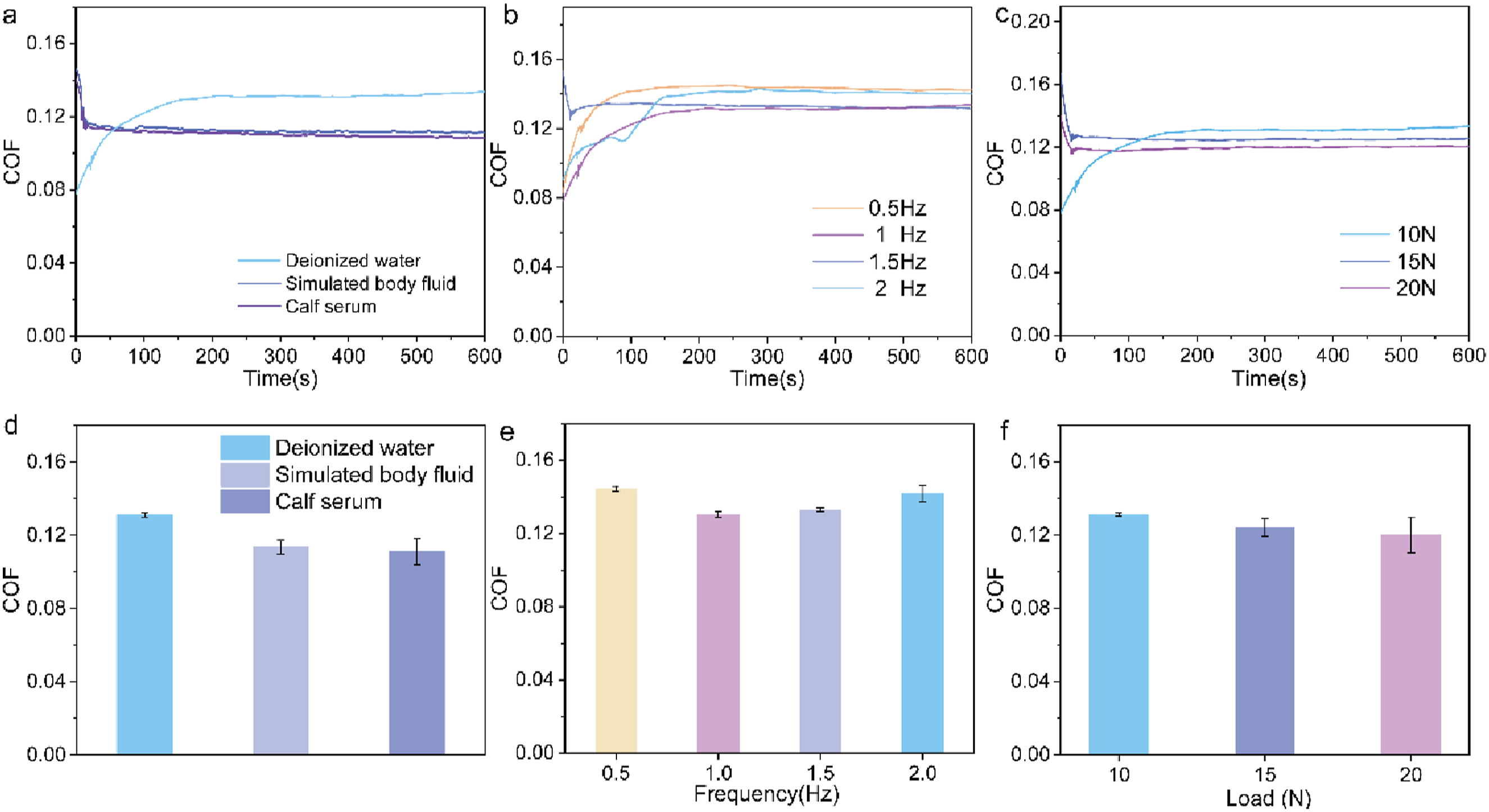
With a constant 10 N sliding load and deionized water, the impact of varying sliding frequencies was explored, as shown in Figure 6(b) and (e). As frequency rose from 0.5 Hz to 2 Hz, the average friction coefficient decreased first and then increased. Under low-frequency conditions, the molecular chains and water within the hydrogel have sufficient time to rearrange and flow, thereby reducing the shear resistance. This reduction in energy dissipation leads to a decreasing trend in the friction coefficient. Low-frequency vibrations allow the hydrogel to release more water to the interface, enhancing lubrication and reducing friction. As the frequency increases, the molecular chains in the hydrogel cannot complete their rearrangement in time, exhibiting a phenomenon similar to “hardening.” This increases shear resistance, resulting in a higher friction coefficient. Under high-frequency conditions, the water within the hydrogel cannot respond quickly to vibrations, weakening the lubrication effect and increasing interfacial friction.
With a fixed sliding frequency of 1 Hz and deionized water as the medium, different loads’ effects on PVA-PDA-G hydrogels’ lubrication were studied. As shown in Figure 6(c), increasing the load from 10 N to 20 N keeps the friction coefficient around 0.125. Load increases slightly reduce the coefficient due to enhanced lubricant action from expelled hydrogel fluids at greater contact areas. However, due to the network structure of the hydrogel being able to retain a certain amount of water, the change in lubrication effect is relatively limited, and the friction coefficient does not decrease significantly.
Currently, researchers have proposed various post-treatment methods for PVA to enhance its performance, among which solvent exchange is a highly effective and simple approach. We treated the PVA-PDA-G hydrogel with a deep eutectic solvent (DES), replacing the original good solvent in the gel with a poor solvent (Figure 7(a)). This process disrupts polymer-solvent interactions while enhancing polymer-polymer interactions, thereby facilitating the formation and growth of crystalline domains. Using acrylic acid as a hydrogen bond donor in DES allows for secondary engineering of the polymer network into a semi-interpenetrating network structure, endowing it with multiple unparalleled functionalities. Schematic illustration of SE -PVA-PDA-G and SE-C-PVA-PDA-G hydrogels preparation (a). SEM pictures of PVA-PDA-G (b), SE -PVA-PDA-G (c), and SE-C-PVA-PDA-G (d) hydrogels.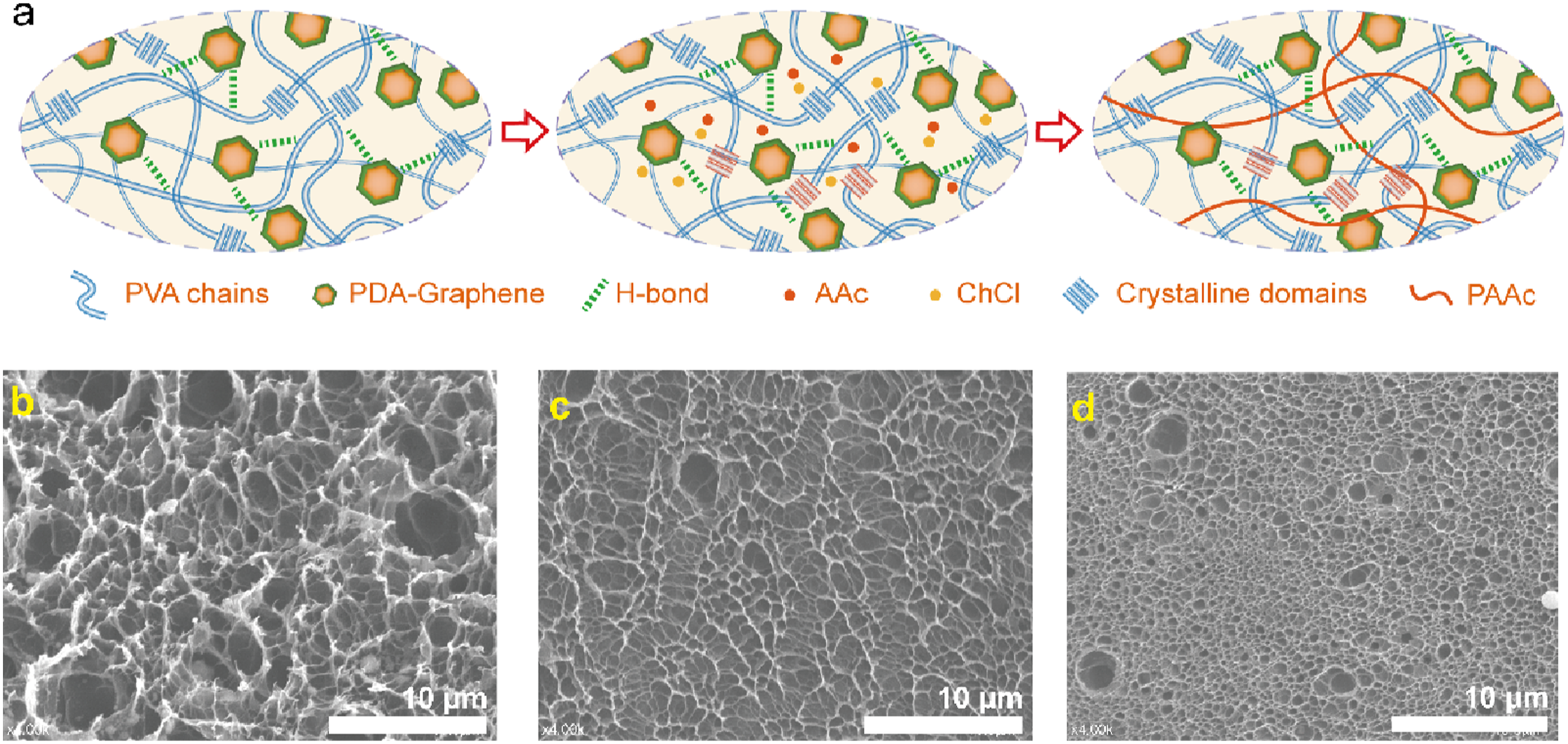
Further enhancements and optimizations were achieved for PVA-PDA-G hydrogels using deep eutectic solvents (DES) and UV-initiated polymerization. The resulting hydrogels are referred to as SE-PVA-PDA-G after DES treatment and SE-C-PVA-PDA-G following UV polymerization. 39 These treatments impart distinct structural and mechanical improvements to the hydrogels.
Post-DES treatment, SE-PVA-PDA-G exhibits a loose porous structure due to the solvent exchange process (Figure 7(c)). UV-induced crosslinking further enhances the internal architecture by forming a dense, three-dimensional network with increased porosity. Additionally, UV polymerization facilitates the in-situ formation of polyacrylic acid (PAA) within the hydrogel matrix, resulting in a denser dual-network structure that significantly reinforces the hydrogel’s mechanical properties.
As illustrated in Figure 8(a), the tensile properties of SE-PVA-PDA-G and SE-C-PVA-PDA-G are markedly improved compared to the original PVA-PDA-G hydrogels. SE-C-PVA-PDA-G demonstrates exceptional tensile strength of 10.28 MPa and elongation at break of 1052.89%, reflecting a significant increase in strength and a moderate rise in elongation. In comparison, SE-PVA-PDA-G achieves a tensile strength of 6.89 MPa and an elongation of 947.95%, both of which surpass the corresponding values for untreated PVA-PDA-G. These results highlight the synergistic effects of DES treatment and UV polymerization in enhancing the mechanical performance and structural integrity of the hydrogels. Tensile curve (a) and elastic modulus (b) of different hydrogels. Friction test curves (c) and corresponding average coefficient of friction of different PVA hydrogel (d).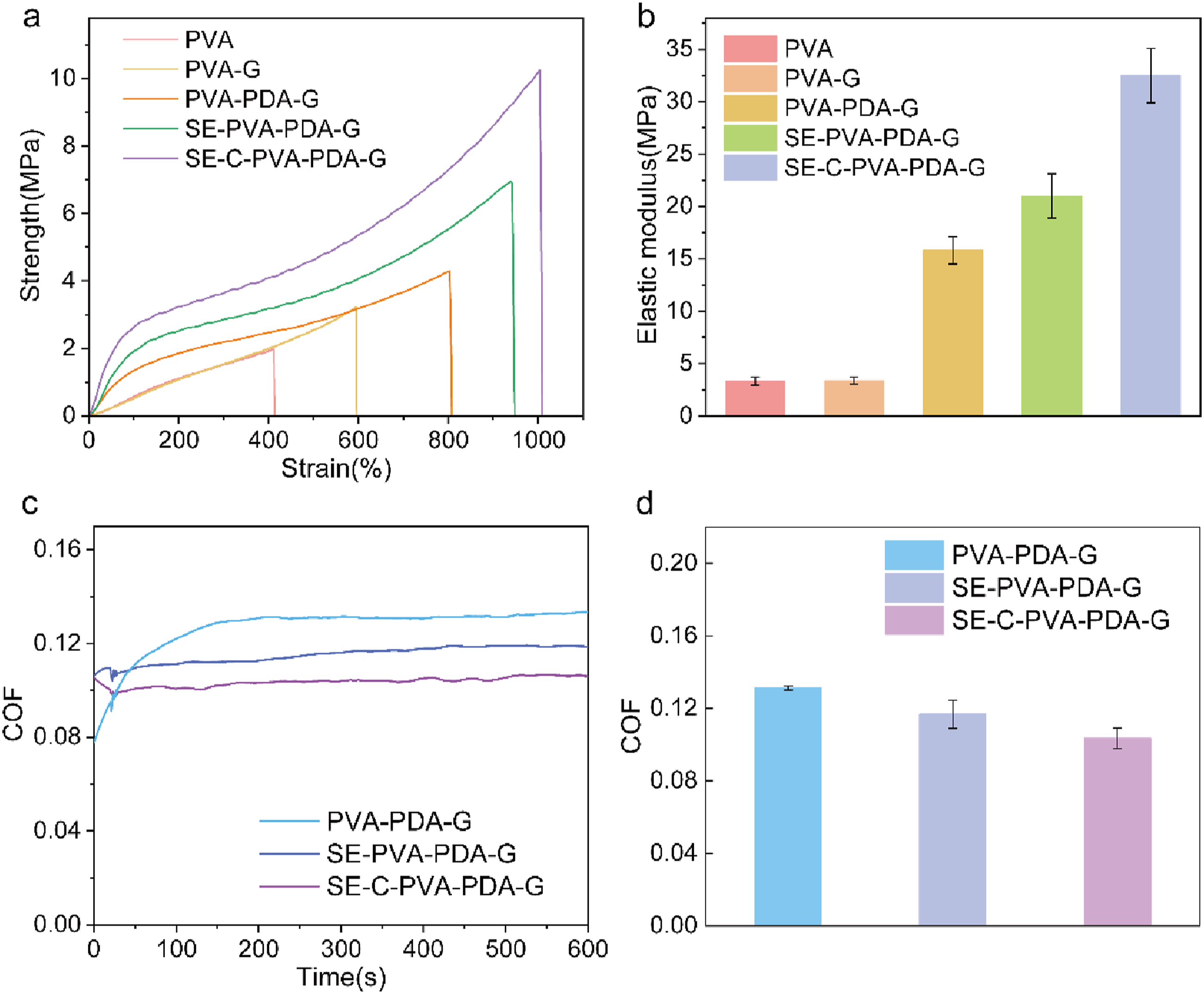
The strength of SE-C-PVA-PDA-G is twice that of PVA-PDA-G, and its elongation is 1.25 times better than the base group. The elastic modulus significantly increased after treatment with deep eutectic solvents (DES) and UV-crosslinking, highlighting that these processes considerably enhance the performance of hydrogels produced. The deep eutectic solvent molecules locally modify the hydrogel’s microenvironment, facilitating a rearrangement of its internal structure, which leads to an overall improvement in mechanical properties. UV light triggers the polymerization of monomers within the hydrogel, resulting in the formation of a secondary crosslinked network. This additional crosslinking increases the hydrogel’s crosslinking density, enhancing its mechanical strength and rigidity under applied external forces.
Figure 8(c) and (d) depict the friction performance curves for SE-PVA-PDA-G and SE-C-PVA-PDA-G hydrogels. Both materials exhibit reduced friction coefficients, with SE-C-PVA-PDA-G showing a coefficient of 0.095 and SE-PVA-PDA-G at 0.118. Both values are lower than that of PVA-PDA-G, demonstrating that the DES and UV-crosslinking treatments effectively reduce friction and improve lubrication properties. DES treatment enhances the hydrogel’s mechanical strength through molecular reorganization, concurrently improving dimensional stability and deformation resistance. This modification critically preserves structural integrity during tribological stress by mitigating fatigue-induced fracture. Upon formation of the secondary poly(acrylic acid) (PAA) network, interfacial restructuring occurs: PAA chains facilitate hydrogen-bonded hydration layers at the hydrogel surface, substantially elevating equilibrium water content (EWC) and surface hydrophilicity. The augmented hydrophilicity promotes enhanced water absorption/retention kinetics, which synergistically reduces boundary friction coefficients through improved hydrodynamic lubrication. Furthermore, the PAA network confers dual-phase structural enhancements: (1) topological stabilization via interpenetrating network architecture, and (2) tunable elasticity enabling sustained formation of low-shear aqueous boundary films during prolonged sliding contact.
Conclusion
Current fabrication of high-strength lubricating hydrogels is fundamentally constrained by intricate preparation protocols that severely compromise their practicality. PVA hydrogels produced via one-step drying exhibit notably deficient mechanical robustness and suboptimal lubricity. To transcend these limitations, we engineered polydopamine-modified graphene nanofillers into the PVA matrix using this streamlined drying methodology. The modified graphene demonstrates exceptional aqueous dispersibility, orchestrating uniform reinforcement throughout the hydrogel network. This strategic integration yields a remarkable 39% reduction in the friction coefficient for one-step dried hydrogels. Furthermore, deep eutectic solvent treatment coupled with in situ acrylic monomer polymerization within the system synergistically achieves a twofold enhancement in strength and an additional 30% friction reduction. Although super-lubrication remains unrealized, these breakthroughs establish critical design principles for hydrogel optimization. This nanofiller-mediated crosslinking paradigm pioneers a microstructure-directed strategy for engineering load-adaptive hydrogels, opening transformative pathways for advanced artificial joints and high-demand biomedical interfaces.
Supplemental Material
Supplemental Material - Simple construction of PVA hydrogels based on dehydration drying method via polydopamine-modified graphene nanocomposites
Supplemental Material for Simple construction of PVA hydrogels based on dehydration drying method via polydopamine-modified graphene nanocomposites by Haiyan Feng, Jiawei Li, Xubing Wei, Kai Chen, Xinyue Zhang, Cunao Feng, Xiaowei Li, Dekun Zhang in Journal of Thermoplastic Composite Materials.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by National Natural Science Foundation of China (Grants 52305238, 52475233, 52405239), the Open Fund of State Key Laboratory of Solid Lubrication, Lanzhou Institute of Chemical Physics (Grant LSL 2201), Jiangsu Funding Program for Excellen Postdoctoral Talent.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.