Abstract
To impart good application performance to chitosan (CS) sizes for high polyester content warp yarns, methyl acrylate (MA) and acrylamide (AM) monomers, with a variation in feed molar ratio from 1:9 to 4:6, were grafted onto the molecular chains of native CS to obtain CS-g-P(MA-co-AM) products, with similar grafting ratios through a K2S2O8-NaHSO3 redox system. Effects of monomer compatibility of MA and AM on sizing performance of the CS-g-P(MA-co-AM) for high polyester content warp were studied. Grafting MA and AM with rational compatibility onto the molecular chains of CS is an effective method to improve the application performance of CS sizes. In view of the overall performance of the CS-g-P(MA-co-AM) sizes, the appropriate feed molar ratio of MA/AM should be 3:7.
Introduction
Starch and its derivatives have been the most commonly-used textile sizes for many decades. It is estimated that more than 630,000 tons of starch is consumed only in sizing operations of the textile industry every year worldwide. 1 However, the SARS-CoV-2 epidemic and locust plague in East Africa and South Asia have greatly impacted the world's grain industry economy. Nearly ten major grain-exporting countries, such as Vietnam, India, Thailand, and Egypt, have claimed to restrict their grain export. 2 The increasingly serious food crisis forced the investigators in the textile field to find ways to achieve the aim of sizing yarns without using human food products. 3 Due to the shortage of petroleum resources and the desiz-ing effluent pollution in the water environment, the uses of synthetic sizes (e.g., PVA) are also limited or even forbidden.4,5 It has become more and more economical to extract inexpensive biopolymers from agriculture and fishery by-products as sizing agents, such as sesbania gum, 6 guar gum, 7 sodium alginate, 8 and chitosan (CS). 9
Chitin is the second largest natural polymer in the world after cellulose. It is estimated that the chitin synthesized in nature reaches nearly 10 billion tons every year. CS is the product of the partial deacetylation of chitin and is the only basic polysaccharide among various natural polysaccharides. CS is a high molecular weight polysaccharide composed mainly of β-(1,4)-linked 2-deoxy-2-amino-D-glucopyranose units and partially of β-(1,4)-linked 2-deoxy-2-acetamido-D-glucopyranose units. At present, CS is mainly extracted from fishery by-products in the ocean. It is reported that 38,000 tons of crab and prawn shells are generated as fishery byproducts annually only in Hainan, 10 an island of China. Chitin accounts for nearly one third of the dry weight of crab and prawn shells. Unfortunately, these fishery by-products have not attracted enough attention and most of them are still disposed of in landfills or dumped back to the ocean. Therefore, if textile researchers, especially those working in coastal countries, make full use of fishery byproducts, considerable economic benefits can be created.
CS offers many good characteristics, such as high film-forming properties, abrasion resistance, antibacterial properties, and biodegradability.11-13 Therefore, CS has been considered by textile researchers as a sizing agent. CS is a polysaccharide with many amino and hydroxyl groups in its molecular chain. It is a strongly polar polymer that shows good sizing performance for natural and regenerated fiber yarns (e.g., cotton and viscose). However, CS has poor affin-ity to synthetic fibers, many of which (e.g., polyester) possess numerous phenyl and ester groups and are thus relatively nonpolar. As a result, the poor affinity greatly narrows the application scope of the CS sizing agent. To modify the CS molecular structure, methyl acrylate (MA) has been grafted onto native CS through the initiation of a K2S2O8-NaHSO3 redox system. This modification decreased the macromo-lecular polarity of the polysaccharide and enhanced the polarity similarity between CS and polyester.
It should be noted that modern sizing operations are carried out in water. This requires the sizing agent to possess water solubility or at least good water dispersibility. Native CS does not possess water solubility in itself and is only able to dissolve in dilute acid (e.g., HOAc or HCl) due to the high regularity and rigidity of the molecular chains and the strong intramolecular and intermolecular hydrogen-bond interactions. 14 On this basis, the introduction of hydrophobic grafted branches (e.g., poly(methyl acrylate), PMA) will unavoidably reduce its water solubility further. Therefore, various amounts of the hydrophilic monomer acrylamide (AM) were also grafted onto the CS simultaneously to maintain the water solubility of the grafted product.
After the preparation of a series of CS-g-P(MA-co-AM) with various feed molar ratios of MA/AM and the evaluation of major application performance of the grafted CS sizes (i.e., apparent viscosity, contact angles of sizing paste on polyester fibers, mechanical properties of sizing film, tensile strength and elongation, abrasion resistance, and the hairiness amount of the sized polyester/cotton blended yarns), influences of the monomer compatibility on sizing performance of the CS-g-P(MA-co-AM) were revealed. Finally, the appropriate grafting ratio is recommended for the preparation of CS-g-P(MA-co-AM), which possesses good sizing performance for high polyester content warp.
Experimental
Materials
Chitin was extracted from crab and prawn shells collected from a local seafood restaurant after demineralization and deproteinization using 1 M of HCl and 10% of NaOH solution, respectively. All chemicals were provided by Sinopharm Chemical Reagent Co. Ltd. Native CS, of which the degree of deacetylation was 90%, was prepared from the chitin through deacetylation using 45% of NaOH solution at 100 °C. The native CS was dried completely, pulverized, and stored in desiccator. MA, AM, K2S2O8, NaHSO3, and paradioxybenzene were used as grafting monomers, oxidant, reductant, and terminator in the graft copolymerization, respectively. All the reagents used were analytical pure grades. The polyester/ cotton 65/35 (T/C65/35) warp (19.5 tex) yarns were obtained from Qingfeng Textile Co. Ltd. and Yuanda Textile Co. Ltd., and were used for the sizing experiment. Poly(methacrylate-co-acrylic acid) (P(MA-co-AA), solid content: 24%) and grafted starch sizes (starch-g-P(MA-co-AA), grafting ratio: 22%) were kindly provided by Jingwei Green Sizes Co. Ltd., Jining, China, to size the T/C65/35 blended yarns.
Graf Copolymerization
Before grafting, 10 g of native CS were dispersed in 1% of dilute HOAc in water. All dilute HOAc used in the study was 1%. Ten, the dispersion was transferred into a 250-mL four-neck flask. The flask was maintained at 60 °C in a water bath and the dispersion was deoxygenated by passing N2 through the solution for at least 30 min. The initiators, including the oxidant (K2S2O8) and the reductant (NaHSO3), were dissolved in the dilute HOAc, respectively. The MA and AM monomers, oxidant, and reductant solutions were added into the flask simultaneously. The feed molar ratio of MA/AM monomers was in a range of 1:9 to 4:6. To eliminate the influence of graft-ing ratio (GR) of the modified CS on the sizing performance, the GR of CS-g-P(MA-co-AM) was controlled at similar values. According to the previous report on feather keratin grafted with MA and acrylic acid (AA) monomers, 15 sizing properties of the grafted keratin on polyester yarns were satisfactory when the GR was around 27.5%. Therefore, the GR of the CS-g-P(MA-co-AM) was controlled within a fixed range (27.5% ± 1.5%) after many tentative graft copolymer-ization experiments. The total feed weights of MA and AM monomers were 5.3, 5.5, 5.6, and 6.0 g when the feed molar ratios of MA/AM monomers were 1:9, 2:8, 3:7, and 4:6, respectively. The addition was completed in 10-20 min and final bath ratio of the CS to the dilute HOAc was 1:10. The concentration of K2S2O8 was 0.037 M and the molar ratio of K2S2O8/NaHSO3 was 1:1.5. Graf polymerization was carried out under vigorous stirring using a mechanical stirrer at 1000 rpm under a nitrogen atmosphere for 2 h. About 2% of paradioxybenzene solution was added to terminate the graft copolymerization. The product was filtered, washed thoroughly with distilled water, dried in a vacuum oven, ground to a fine powder, and stored in a desiccator. Each graft copo-lymerization was repeated three times.
Measurement of Grafting Parameters
Grafting parameters of the CS-g-P(MA-co-AM), such as conversion of monomers to polymer (MC), grafting ratio (GR), and grafting efficiency (GE) were measured. The detailed measurement processes were described in the Supporting Information (SI), available from the author upon request.
FTIR Characterization
A Fourier transform infrared (FTIR) spectrometer was used to verify the grafting of MA and AM onto CS. Previous reports indicated that acetone and water were good solvents for PMA and polyacrylamide (PAM), respectively.16,17 The grafted CS was successively extracted by acetone and water for 48 h (acetone for 24 h and water for 24 h) to ensure the thorough removal of the homopolymers (i.e., PMA and PAM). Measurements were taken on a Thermo Nicolet (Avatar 380) FTIR spectrophotometer through the diffuse reflectance technique with a spectral resolution of 2 cm−1 for 64 scans.
Elemental Analysis
An elemental analyzer (Euro EA3000, Italy) was used to measure the carbon, hydrogen, nitrogen, and sulfur contents of native CS and the purified CS-g-P(MA-co-AM). For carbonizing the samples, high temperature combustion was used. Characterization was carried out according to Li's report. 18
Measurement of Application Properties of Sizing Paste
Main application properties of the native and grafted CS sizing pastes, such as apparent viscosity and contact angle on polyester fiber, were measured in the study. The detailed measurement processes are described in the SI, available from the author upon request.
Preparation of Sizing Films and Measurement of Their Application Properties
Sizing film was cast with 400 g of the CS paste (6% w/w) and the surface morphology of the film was observed by scanning electron microscopy (SEM). The main mechanical properties of the sizing films (i.e., tensile strength, elongation, and wear resistance) were evaluated in the study. The preparation method of sizing films, SEM observations, and the detailed measurement processes of mechanical properties of the films are described in the SI, available from the author upon request.
In addition, the dissolution time of the sizing films in hot water was measured to evaluate the desizability of the grafted CS. The sizing films were cut into rectangular strips (100 × 20 mm). In general, desizability of a sizing agent could be measured as the time span required to break its sizing film in hot water. A dead weight of 0.5 g was fixed at one end of each strip for stretching the strip in the water bath and half of the strip with dead weight was saturated in distilled water at 80 °C. The dissolution time required to break the strip was measured in a span beginning from the time when one end of the strip was put into water bath and ending at the time when the strip was broken. Ten samples were measured to obtain the mean value.
TGA Characterization
Termostability of native and grafted CS sizing films was studied using thermogravimetric analysis (TGA). TGA and derivative thermogravimetry (DTG) curves of the native and grafted samples were obtained using a TG/DTA6300 (Japan Seiko Co. Ltd.) integrated thermogravimetric analyzer. About 5 mg of the sample was heated at 10 °C/min in a range of 20 °C to 800 °C under an air atmosphere.
Sizing Operation and Measurement of Application Properties of Sized Yarns
The sizing experiment for T/C65/35 warp was carried out using a GA392 laboratory single-yarn sizing machine (Tongyuan Textile Machinery Co. Ltd.). Size add-on, mechanical properties, and the hairiness amount of the sized yarns were measured in the study. The detailed measurement processes are described in the SI, available from the author upon request.
Statistical Analysis
The data were analyzed using SAS software (SAS Institute Inc., Cary, NC, USA). Tukey's HSD test was used to compare the multiple means in the investigation. The confidence interval was set at 95% and a p value smaller than 0.05 was considered to be a statistically significant difference by Tukey's HSD test. According to the operation results of the analysis software, small letters, such as a, b, c, were inserted above or below the data points in the appropriate figures. The data points with the same small letter were not statistically significantly different from each other.
Results and Discussion
FTIR Analysis of Native and Grafted CS
FTIR spectra of native CS and the CS-g-P(MA-co-AM) are shown in Fig. 1. In addition to all of the characteristic peaks appearing in the spectrum of native CS, such as the ones at ∼3380 and ∼1680 cm−1 due to vibrations of hydroxyl and amide carbonyl groups, 19 respectively, a new characteristic absorption band of methyl ester carbonyl groups at ∼1730 cm−1 could be observed in the spectrum of CS-g-P(MA-co-AM). The peak confirmed the grafting of MA onto molecular chains of CS. 20 In addition, the intensity of the peak at ∼1680 cm−1 in the spectrum of the grafted CS increased markedly. The increase was caused by the stronger vibration of amide carbonyl groups. It is understood that the introduction of PAM branches would increase the amount of amide in the resulting CS. Thus, the increase in the intensity of the peak at ∼1680 cm−1 was regarded as evidence for the grafting of AM onto molecular chains of CS.
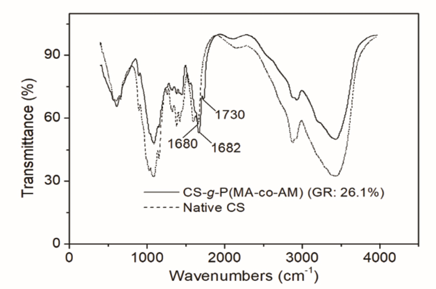
FTIR spectra of purified native CS and CS-g-P(MA-co-AM).
Effects of Monomer Compatibility on Grafting Parameters
Fig. 2 depicts the effects of monomer compatibility of MA and AM on monomer conversion (MC), grafting ratio (GR), and grafting efficiency (GE) of the CS-g-P(MA-co-AM). With the increase in the feed molar ratio of MA/AM, all the MC values were around 99.5% and showed no significant change. To eliminate the influence of the GR of CS-g-P(MA-co-AM) on the sizing performance, the GR of the grafted CS synthesized by various feed molar ratios of MA/AM were controlled at similar values (∼27.5%). As for GE, it gradually decreased in a range of 52.1% to 43.2% with the increase in the feed molar ratio of MA/AM.
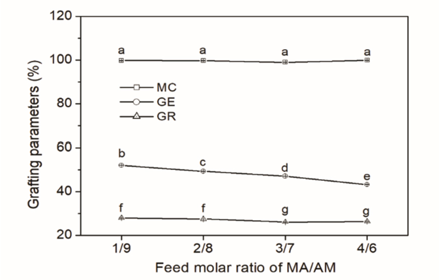
Grafting parameters of CS-g-P(MA-co-AM). 1:9, 2:8, 3:7, and 4:6 are the feed molar ratios of MA/AM used in the graft copolymerizations that comprise the CS-g-P(MA-co-AM) sizes.
In general, an adequately long polymerization time can convert nearly all the monomers into polymer under appropriate polymerization conditions, such as adequate amounts of initiators and proper temperature. Polymerization time (2 h) was long enough in the graft copolymerization due to the high MC (∼99.5%). In the copolymerization, the bonding of CS macromolecule free radicals with MA and AM provided the basis of the formation of grafted branches (i.e., PMA and PAM). The methyl ester group in MA occupies a larger space than the amide group in AM. Thus, it is more difficult for MA to be grafted onto the molecular chains of CS due to its greater steric hindrance when MA and AM are both in the vicinity of the CS free radicals. During the grafting of MA and AM onto the molecular chains of the CS, homopolymerization and graft copolymerization occurred simultaneously, which are a pair of typical competitive reactions. More MA monomers tended to join in the homopolymerization and then formed homopolymers than AM ones. When the MC was nearly the same, the more the homopolymers formed, the fewer branches were grafted onto the backbone of the substrate. The increase in the amount of the homopolymers formed by MA unavoidably led to the decrease in the GE.
Elemental Analysis of Native and Grafted CS
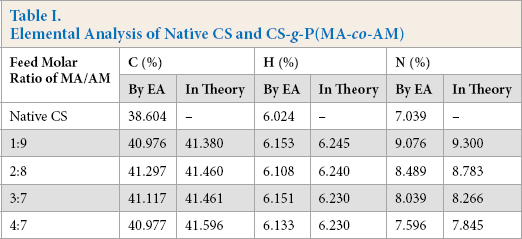
Elemental contents of carbon, hydrogen, and nitrogen of native CS and the grafted CS were measured using elemental analysis (EA). The results are displayed in Table I. Based on the GR evaluated through titration and Soxhlet extraction, and the contents of the three elements of native CS measured by EA, the elemental contents of the grafted CS in theory were calculated and also displayed in Table I. The three elements of CS-g-P(MA-co-AM) were higher than those of the native CS because there were less of these elements in the native CS than in PMA (C (%): 55.81 and H (%): 6.98) or PAM (C (%): 50.70, H (%): 7.04, and N (%): 19.72) branches of the grafted CS. In addition, the elemental contents of the grafted CS measured by EA were in good agreement with those calculated in theory, and thus, the GR of the grafted CS was evaluated accurately in the measurement of grafting parameters.
Elemental Analysis of Native CS and CS-g-P(MA-co-AM)
Solubility of Native and Grafted CS Sizing Films in Hot Water
Fig. 3 shows the effect of monomer compatibility on dissolution time of the CS sizing films in hot water (80 °C). The water-dissolution time of sizing film can indicate the difficulty of desizing, which is carried out in the aqueous phase. The longer the dissolution time in water, the more difficult the desizing. As depicted in Fig. 3, it required less time for the grafted CS film to dissolve in water than that for the native CS film. With the increase in the feed molar ratio of MA/AM, the dissolution time increased gradually.
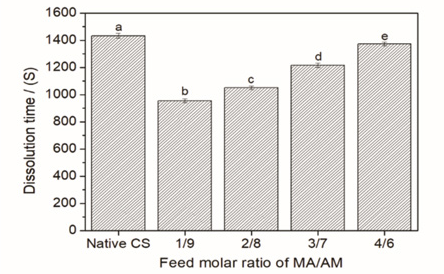
Water dissolution time of sizing films of native CS and CS-g-P(MA-co-AM).
CS is a typical kind of long-chain polymer. The long molecular chains arrange regularly and exhibit high rigidity. Strong intramolecular and intermolecular hydrogen-bond interactions increase the difficulty of the CS dissolving in water. After grafting, PMA and PAM branches could increase the degree of branching of the modified CS. Ten, the intermolecular distance was widened and the intermolecular force of the CS was decreased. Thus, grafting appropriate amounts of acrylic monomers can improve the water-solubility of the CS sizing film. With the increase in the feed molar ratio of MA/AM, the number of the ester groups introduced onto the molecular chains of the CS was increased. The increased number of non-polar ester groups unavoidably reduced the hydrophilicity of the CS and the interaction between the CS and water molecules. As a result, when the GR of the grafted CS were similar, the dissolution time of the grafted CS film in hot water was prolonged, with the increase in the feed molar ratio of MA/AM.
Effects of Monomer Compatibility on Mechanical Properties of Sized Yarns
Effects of monomer compatibility on mechanical properties of the T/C65/35 blended yarns sized by the CS are shown in Fig. 4. All mechanical properties of the yarns sized by the CS-g-P(MA-co-AM), such as tensile strength, elongation, and wear-resistance, were better than those sized by native CS. With the increase in the feed molar ratio of MA/AM in the range of 1:9 to 3:7, the mechanical properties of the sized yarns were improved continuously. However, when the feed molar ratio exceeded 3:7, the mechanical properties began to deteriorate.
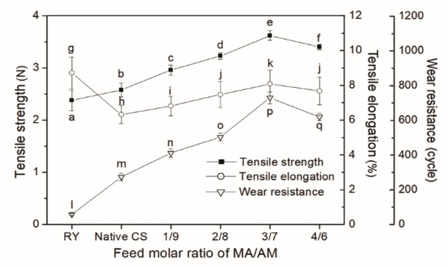
Mechanical properties of raw yarns (RY) and the yarns sized by native CS and CS-g-P(MA-co-AM).
Native CS contains a large number of amino and hydroxyl groups, and thus exhibits strong polarity. In contrast, polyester fiber contains many ester and phenyl groups and belongs to non-polar synthetic fibers. As a result, native CS shows low affinity to polyester fiber due to the difficulty of formation of strong adhesion between a polar-nonpolar pair. 21 In addition, the native CS film formed on the surface of the warp after drying of the sizing paste retains the characteristics of being hard and brittle, which has an adverse effect on the elongation of the warp. Therefore, native CS is not suitable for sizing polyester and high content polyester yarns.
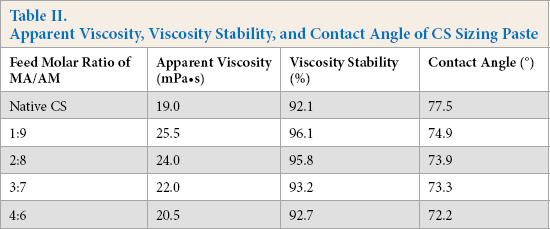
High adhesion of sizing agent to fiber is the basis of forming good cohesion between single fibers in warp yarns and achieving high binding fastness between sizing film and warp yarns. In general, the higher the adhesion of sizing agent to fiber, the better the mechanical properties of the sized yarns. The primary condition to obtain high adhesion is to achieve molecular-level close contact between the adherent and the adhesive. The distance between the two-phase molecules should be less than 0.5 nm. 22 Otherwise, the interface force between the two phases is too small to resist external stress. To reduce the molecular distance between the adhesive and the adherent to such a small degree, it is not enough to rely on external pressure alone. It is also required that the adhesive can wet the adherent well, so that the adhesive can evenly spread out on the surface of the adherent. This explains why improving the wettability of CS sizing paste on polyester fiber is beneficial to increase the adhesion of the CS to polyester and to improve the mechanical properties of the sized polyester warp yarn further. As shown in Table II, the contact angle of the grafted CS sizing paste on polyester fiber decreased with the increase in the feed molar ratio of MA/AM in the range of 1:9 to 4:6. In other words, the wettability of the sizing paste to polyester fiber was improved. 21 Therefore, it could be speculated that the adhesion of the grafted CS to polyester also kept increasing in this feed molar ratio range.
Apparent Viscosity, Viscosity Stability, and Contact Angle of CS Sizing Paste
Warp yarns are subjected to stretching and bending, are abraded by reed, heddle, and shuttle, and keep rubbing against each other during the weaving process. Therefore, the formation of warp protective layer-sizing film with good mechanical properties is a prerequisite for obtaining sized yarns with good mechanical properties. Besides the adhesion of sizing agent to fiber, the mechanical properties of the sizing film should be taken into serious consideration. SEM images of sizing films of the native CS and CS-g-P(MA-co-AM) are shown in Fig. 5. Fig. 6 depicts mechanical properties of sizing films of the native CS and the grafted CS with various feed molar ratios of MA/ AM. Fig. 6 showed that tensile strength, elongation, and wear-resistance of the CS-g-P(MA-co-AM) sizing film were improved when the feed molar ratio of MA/AM was in the range of 1:9 to 3:7. However, when the feed molar ratio exceeded 3:7, the mechanical properties of the sizing film began to decrease instead due to the remarkable decrease in the water solubility of the grafted CS. The decrease directly led to the deterioration of the mechanical properties of the sized yarns. The results of the mechanical tests for the yarns sized by CS-g-P(MA-co-AM) were in agreement with the changes of the mechanical properties of the sizing film. However, the decreasing degree of mechanical properties of the sized yarn was not as large as that of the sizing film. The reason was that the beneficial influence of increasing the feed molar ratio of MA/AM on the adhesion between the CS and polyester still affected the mechanical properties of the sized yarns positively.

SEM images of sizing films of (a) native CS and CS-g-P(MA-co-AM), of which feed molar ratios of MA/AM are (b) 1:9, (c) 2:8, (d) 3:7, and (e) 4:6 in the graft copolymerization.
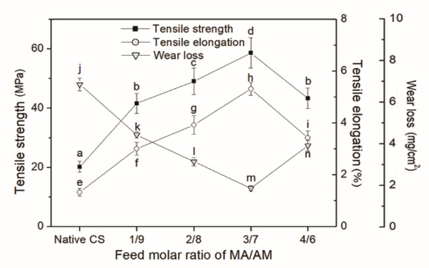
Mechanical properties of native CS and CS-g-P(MA-co-AM) sizing films.
TGA of Native and Grafted CS Sizing Films
Fig. 7 reveals the thermal degradation behaviors of native CS and the grafted CS sizing films. As shown in the thermogram of the native CS (Fig. 7a), the degradation process could be divided into three main stages: (1) moisture removal from CS (< 120 °C), (2) thermal degradation of CS (220-400 °C), and (3) carbonization and carbon oxidation (> 480 °C). In Stage 1, both bound water and absorbed water of CS were removed. The weight loss percentage was 11.1% at the end of Stage 1. In Stage 2, the molecular chains of CS were decomposed by thermal oxidation, the glycosidic bonds were broken, and adjacent hydroxyl groups were dehydrated to ethers. The weight loss rate reached the maximum at 292 °C and the weight loss percentage was 67.3% at the end of Stage 2. In Stage 3, CS was carbonized and some carbon was oxidized.23-25
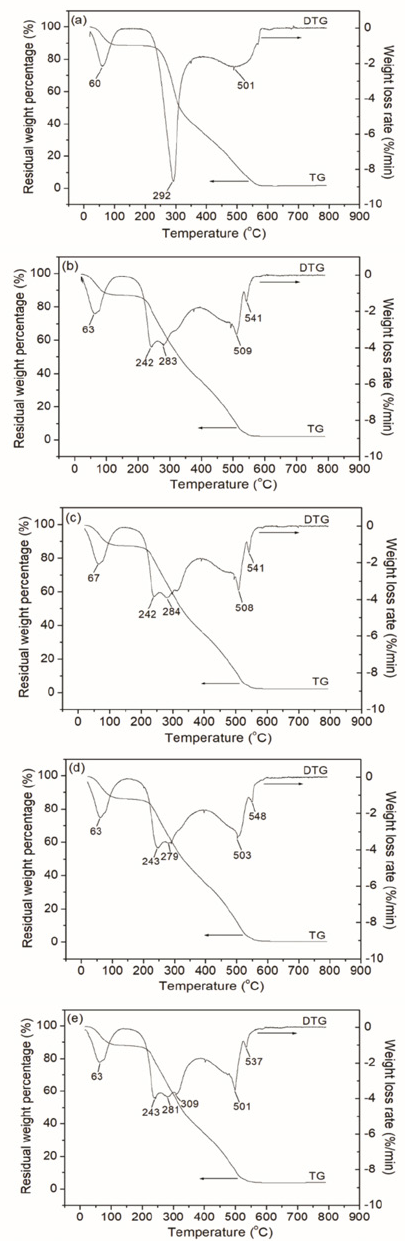
TGA thermograms of sizing films of (a) native CS and CS-g-P(MA-co-AM), of which feed molar ratios of MA/AM are (b) 1:9, (c) 2:8, (d) 3:7, and (e) 4:6 in the graft copolymerization.
Compared with the thermogram of native CS film, thermal degradation trends of the grafted CS films were quite similar in Stage 1. The weight loss percentage was 12.6% at the end of Stage 1. However, the thermal degradation trends of the native and grafted CS in Stage 2 and Stage 3 were different. In Stage 2, the grafted CS peaks in the DTG curve were more numerous than that for native CS. The appearance of more peaks could be attributed to the breakage of the C-C bond of the backbone and the terminal C=C bond of PMA and PAM branches, and the imine reaction between amide groups of PAM branches. The weight loss rate reached the maximum at 242 °C and the weight loss percentage was 64.8% at the end of Stage 2. In Stage 3, CS backbone, and PMA and PAM branches, were all carbonized.26-28
More peaks could be observed in the DTG curve of the grafted CS in the stage due to the difference of carbonization temperature of the backbone and graft branches. As for the four kinds of grafted CS sizing films, they showed similar thermal degradation behaviors because of the nearly same grafting ratio. It should be noted that the highest temperature the CS was exposed to during the sizing process occurred in the drying chamber for sized yarns (∼110 °C). In the temperature range of 20 °C to 120 °C, the native and grafted CS sizing films possess similar thermal stability based on thermogram analysis.
Comparison of Mechanical Properties of the Yarns Processed by Various Sizes
In addition to the grafted CS, the T/C65/35 blended yarns were sized by P(MA-co-AA) and starch-g-P(MA-co-AA). Mechanical properties of the yarns sized by the three copolymers are displayed in Table III. The mechanical properties of the yarns sized by the grafted CS were all higher than those of the yarns sized by P(MA-co-AA) and starch-g-P(MA-co-AA), except that the tensile elongation was slightly less than that of the yarns sized by P(MA-co-AA).
Mechanical Properties of the Yarns Sized by Various Sizing Agents
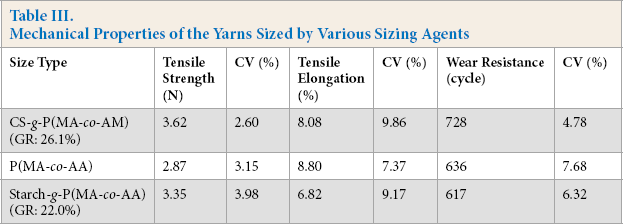
Taking only the mechanical properties into consideration, the grafted CS was more suitable for sizing high content polyester yarns than the other two sizes. Moreover, CS is less expensive and more biodegradable than the polyacrylic copolymer, which is a non-renewable petroleum-based size. Starch and its derivatives have been the most commonly-used textile sizes for a long time. However, as mentioned above, many Asian and African countries are seriously troubled by grain problems due to the SARS-CoV-2 epidemic and locust plague. As a result, chitosan has great potential for becoming a good substitute for starch, especially in coastal countries.
Effects of Monomer Compatibility on the Hairiness Amount of Sized Yarns
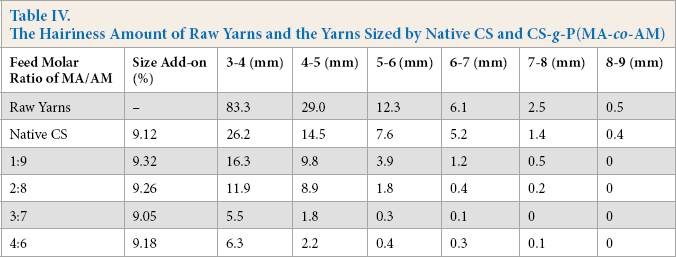
Effects of monomer compatibility on the hairiness amount of the yarns sized by CS are shown in Table IV. The hairiness amount of the yarns sized by CS-g-P(MA-co-AM) was all less than that of the yarns sized by native CS. With the increase in the feed molar ratio of MA/AM in the range of 1:9 to 3:7, the hairiness amount of the yarns sized by the grafted CS continued to decrease. However, when the feed molar ratio exceeded 3:7, the hairiness amount started to increase instead.
The amount of the hairiness of the sized yarns is determined by the adhesion of sizing agent to fiber. It is an effective way to reduce the hairiness of warp yarns by improving adhesion. Due to the grafting of PMA branches containing many ester groups onto the backbone of native CS, the grafted CS had better adhesion to polyester fiber than native CS. In addition, the hairiness amount of the sized yarns is also related to apparent viscosity of sizing paste. Excessively low viscosity will decrease the resistance that the hairiness encounters when warp yarns are going through sizing paste and negatively impact the clinging of the hairiness to the yarn body. 16 As shown in Table II, the grafting of PAM branches efficiently increased the apparent viscosity of the CS sizing paste due to the enhancement in the intermolecular force between the CS and water. Higher adhesion to polyester and higher apparent viscosity of the sizing paste helped the grafted CS possess better hairiness reduction ability than native CS.
As for CS-g-P(MA-co-AM), in the case of having a similar GR of the modified CS, the higher the feed molar ratio of MA/AM, the greater the number of ester groups were grafted onto the molecular chain of CS. Thus, grafted CS synthesized with a higher feed molar ratio of MA/AM had better adhesion to polyester fiber. As shown in Table IV, the amount of hairiness kept decreasing when the feed molar ratio ranged from 1:9 to 3:7. However, the introduction of excess ester groups onto the CS would decrease the apparent viscosity of CS paste to a low value. For instance, when the feed molar ratio of MA/AM reached 4:6, the viscosity of the grafted CS paste decreased to only 20.5 mPa·s due to the poor water solubility of the CS. Therefore, the hairiness amount started to increase instead.
The Hairiness Amount of Raw Yarns and the Yarns Sized by Native CS and CS-g-P(MA-co-AM)
Conclusion
The monomer compatibility of MA and AM had significant impacts on the application performance of CS-g-P(MA-co-AM) for warp yarn sizing. Grafting parameters, apparent viscosity and viscosity stability, contact angle, mechanical properties of the sizing film, tensile strength and elongation, wear resistance, and the hairiness amount of the sized polyester/cotton (65/35) blended yarns had a close relationship to the feed molar ratio of MA/AM. The grafted CS with similar GR exhibited better sizing properties for high content polyester warp yarns when the feed molar ratio of MA/AM was 3:7.
Grafting appropriate amount of MA onto native CS is a simple and efficient way to improve the major sizing performance of CS, especially yarn elongation and the hairiness reduction ability. Applicable objects of CS sizes were extended from natural fiber yarns to synthetic fiber yarns through the introduction of PMA and PAM grafted branches onto the backbone of native CS. The graft copolymerization of chitosan with MA and AM monomers provides a new way to meet the demand of sizing warp without human food from fishery byproducts.
Footnotes
Acknowledgments
The work was supported financially by the Technological Research Project for Public Welfare of Zhejiang Province (LGG21E030005) and the Postdoctoral Research Program of Zhejiang Province. Financial sponsors do not endorse the views expressed in this publication.
