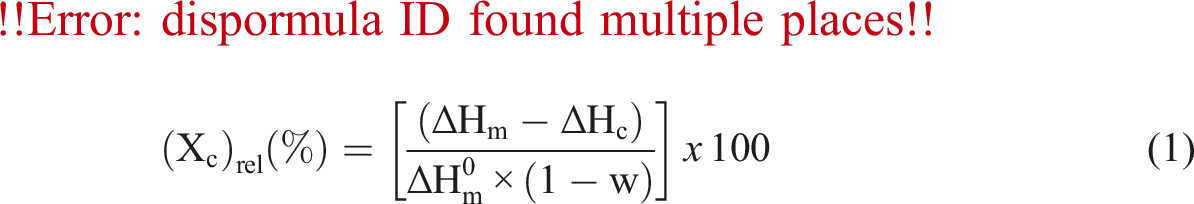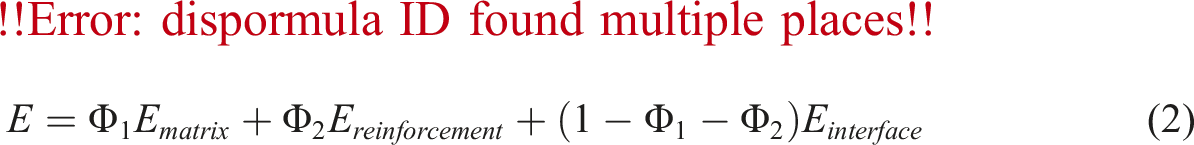Abstract
The objective of this study is to enhance the fundamental structural characteristics of thermoplastic polyester elastomer (TPEE) material. For this purpose, two distinct boron-based particles, namely boric acid (BA) and hexagonal boron nitride (hBN), were utilised as reinforcing materials. The production of boron-based particle-reinforced TPEE matrix composites was achieved through the process of extrusion and injection molding. The investigation of the wear behavior of the composites was conducted through the implementation of adhesive wear tests, while the mechanical properties were analyzed through the execution of tensile and three-point bending tests. Furthermore, the thermal properties were examined through the utilization of differential scanning calorimetry and thermogravimetric analysis. The findings of the study indicate that the coefficient of friction values of the TPEE decreased with the addition of both BA and hBN. The incorporation of both BA and hBN into TPEE, as well as an increase in the weight ratio of these particles, resulted in a notable enhancement in tensile strength, elastic modulus and flexural stress at yield values. The addition of BA to TPEE resulted in an increase in the crystallization of the TPEE matrix, while the addition of hBN to TPEE led to a reduction in the crystallization of the TPEE matrix. The incorporation of both BA and hBN elevated the decomposition temperatures of the TPEE, thereby enhancing its thermal stability. However, hBN demonstrated superior performance compared to BA, potentially due to its lower particle size distribution. Nevertheless, it was concluded that, depending on the intended application of the end product, both types of particles can be effectively utilized to enhance the performance of TPEE.
Keywords
• hBN offers superior performance to TPEE compared to BA. • hBN has superior performance compared to BA. • Boron based particles have the potential to extend the applications of TPEE.Highlights
Introduction
Thermoplastic elastomers (TPEs) are significant engineering materials that integrate the elastic characteristics of elastomers with the advantageous processability and strength attributes of thermoplastics. In contrast to conventional elastomers, TPEs demonstrate noteworthy heat resistance, as evidenced by numerous studies.1–5 While TPEs exhibit elastomeric behavior at room temperature, they can be molded like thermoplastics at elevated temperatures. The impact strength of these materials is particularly impressive, even at relatively low temperatures, which makes them suitable for use in many applications where rubber is currently employed. 6 Thermoplastic polyester elastomers (TPEEs), which belong to the TPE family, are materials that bridge the gap between rubbery elastomers and rigid plastics. They possess a combination of soft and hard segments, which allows them to exhibit properties that are intermediate between those of rubbery elastomers and rigid plastics. The mechanical and thermal properties of these materials are contingent upon the composition of the soft and hard segments that comprise them. As a consequence of the physical interaction between the hard segments within the TPEE matrix, hard crystalline structures are formed by transient cross-linking. In this manner, TPEE displays elastomeric characteristics. However, the hard crystalline structure is subject to degradation at elevated temperatures and the reduction in physical interaction results in the emergence of thermoplastic properties. The soft amorphous endows the material with low-temperature flexibility and thermal stability. TPEE materials demonstrate excellent flexural fatigue strength and are suitable for use over a wide temperature range. Furthermore, these materials are resistant to tearing, impact and creep. Furthermore, they demonstrate exceptional resilience to damage even when subjected to the presence of hydrocarbons and a range of other fluids. One commercially available TPEE, Hytrel®, is the most widely used type of TPEE. It which consists of a hard crystalline segment of poly (butylene terephthalate) (PBT) and a soft amorphous segment based on long poly (ether glycol) chains.2,7,8 TPEE materials possess favorable mechanical and thermal properties, rendering them applicable in numerous industries, particularly the automotive sector.1,2,5,9–13 As a result, TPEE materials are often utilized in automotive components such as gears and sprockets, industrial hoses requiring high strength, sealing products, belts, damping elements, shock and sound absorbing devices, fasteners, fiber optic cables, sports equipment, and electrical and electronic appliance parts.2–4,12,14–16
However, despite the superior properties of TPEE materials, they have are unable to meet the requirements for applications where wear occurs in the unreinforced form due to their low wear resistance. Accordingly, the mechanical, thermal, and tribological performances of these materials can be enhanced by modifying their microstructure with various reinforcements.13,17–21 It is established that the mechanical properties and wear resistance of unreinforced polymers are enhanced by the uniform distribution of reinforcing materials within the structure.2,6 It is therefore feasible to develop an efficacious material that fulfils the aforementioned criteria by incorporating particles into TPEE.
However, in order to develop this efficacious material by ensuring homogeneous distribution of reinforcing materials in the structure, it is necessary to understand the mechanisms that come to the fore during the mixing of polymer and reinforcing material. During the melt mixing procedure, many interactions can occur between the reinforcing material and the polymer and two of these interactions are of particular note. The first is covalent bonding, which rarely occurs spontaneously and only occurs with special surface treatments. Secondly, Van der Waals forces, which are secondary forces. It is imperative to note that these forces are instrumental in ensuring the adsorption of polymer chains to the active sites of the filler surface. The adsorption of polymer molecules on the surface of the reinforcement material through these forces leads to the formation of a layer with different properties than the matrix polymer on the reinforcement surface. This layer, termed the interfacial layer, plays a pivotal role in the properties of the composite. Therefore, it can be concluded that the maximum performance of the composite is only possible when the wetting of the reinforcing material by the polymer is perfect. 22
In recent years, the use of inorganic minerals as reinforcing materials in polymers has emerged as a significant area of research due to their capacity to enhance mechanical, electrical, and thermal properties, while offering a cost-effective alternative. 23 In particular, platelet types of these inorganic materials have been demonstrated to enhance the wear resistance of the polymers to which they are added by acting as solid lubricants during wear due to their layered structure and weak Van der Waals interactions between their adjacent layers.24,25 The objective of this study was to investigate the effect of boric acid (BA) and hexagonal boron nitride (hBN), two boron-based inorganic minerals, on the wear resistance, mechanical performance, and thermal properties of TPEE material. hBN is one of the most common structures of boron nitride and has a graphite-like structure. In the layered structure of hBN, the molecules in each layer are bonded to each other by intermolecular strong covalent bonds, while the layers are bonded to each other by weak Van der Waals forces. Due to the ease with which these layers slide, the addition of hBN facilitates the improvement of wear resistance in the polymer. Furthermore, it exhibits favorable chemical and thermal stability, accompanied by low thermal expansion and high thermal shock resistance.24,26,27 Furthermore, BA is frequently employed as a solid lubricant due to its favorable lubricating characteristics derived from its layered structure. Similarly, the B, O, and H atoms within the layer are densely packed and covalently bonded to one another, while the layers are held together by weak Van der Waals forces. Consequently, the layers are readily displaced during wear, thereby providing lubrication to the system.28,29
A number of studies have been conducted to investigate effect of incorporating particles of varying sizes and types into the TPEE matrix on the characteristics of TPEE.5,12,13,17,19,20,30–34 In a study conducted by Sreekanth et al., 19 the effect of incorporating fillers, including mica and fly ash, at varying concentrations on the mechanical, thermal, electrical, rheological, and morphological characteristics of Hytrel® was examined. The findings indicated that the mechanical properties, including flexural strength and modulus, as well as thermal and electrical properties, exhibited an enhancement with an increase in filler concentration. In another study, Iyer et al. 31 examined the effect of boron nitride incorporation on the characteristics of three distinct elastomer types, including Hytrel®. The findings of the study indicated that the mechanical and thermo-mechanical properties of all three elastomer types exhibited improvement, while the coefficient of thermal expansion demonstrated a reduction with the addition of boron nitride. In a separate study, Pandey et al. 5 incorporated graphene oxide (GO) into Hytrel® at various weight ratios, with the objective of examining the impact of GO addition on the mechanical and thermal properties of the material. The findings of this study indicated that the incorporation of GO into Hytrel® resulted in enhanced thermal stability and improved mechanical and thermomechanical properties.
A review of the literature reveals that the effect of hBN incorporation into a TPEE matrix on wear resistance has yet to be explored. Furthermore, the effect of BA incorporation on TPEE characteristics remains unexplored. For this reason, the objective of this study is to enhance the tribological performance and concurrently improve the mechanical and thermal properties of TPEE-based materials, which have a broad range of applications, particularly in the automotive industry, in the presence of boron-based reinforcing materials, namely hBN and BA. Consequently, the performance a previously utilized polymer was enhanced, and the efficacy of inorganic micro/nano additives was assessed and compared.
Materials and methods
Materials
The matrix material TPEE was procured from DuPont under the brand name Hytrel® 7246 (form: pellet, density: 1.26 g/cm3, melting temperature: 218°C, glass transition temperature: 32°C). One of the boron-based particles, hBN (density: 2.3 g/cm3, purity: 99.97%, particle size: 50-120 nm, form: powder), was procured from BORTEK® Boron Technologies and Mechatronic Inc. (Turkey). The other boron-based particle, BA (H3BO3 orthoboric acid), has the following characteristics: density 1.435 g/cm3, particle size 0.125-1.000 mm, form powder, melting temperature 171°C. The material was supplied by the ETİ Maden Operations General Directorate (Turkey). The silane coupling agent (3-aminopropyl)tri-ethoxysilane (APTES) (molecular weight: 221.37 g/mol, purity: 98%, density: 0.948 g/mL) was procured from Alfa Aesar.
Silanization of hBN and BA particles
The surface of the BA and hBN particles was functionalised with APTES, a silane coupling agent. In order to silanise the hBN nanoparticles, a solution of 100 mL (50/50) ethanol/distilled water was prepared and 1 mL of silane was added. This solution was then stirred at 75°C for 15 minutes using a magnetic stirrer in order to obtain a stable suspension system. Subsequently, 10 g of hBN was introduced to the solution, which was then stirred at 75°C for 1 hour. The solution was then subjected to an ethanol wash in order to remove any residual silane that might still be present on the surface of the hBN particles. Subsequently, the product was subjected to a drying process at room temperature for 24 hours, followed by 12 hours at 80°C in a vacuum oven.
In order to silanise BA particles, a solution of 100 mL (50/50) ethanol/distilled water was prepared and 1 mL of silane was added. This solution was then stirred at 75°C for 15 minutes using a magnetic stirrer. Once the stirring process was complete, 10 g of BA was added to the solution. Subsequently, the solution was permitted to dry at room temperature and then subjected to vacuum drying at 80°C for 12 hours in a vacuum oven.
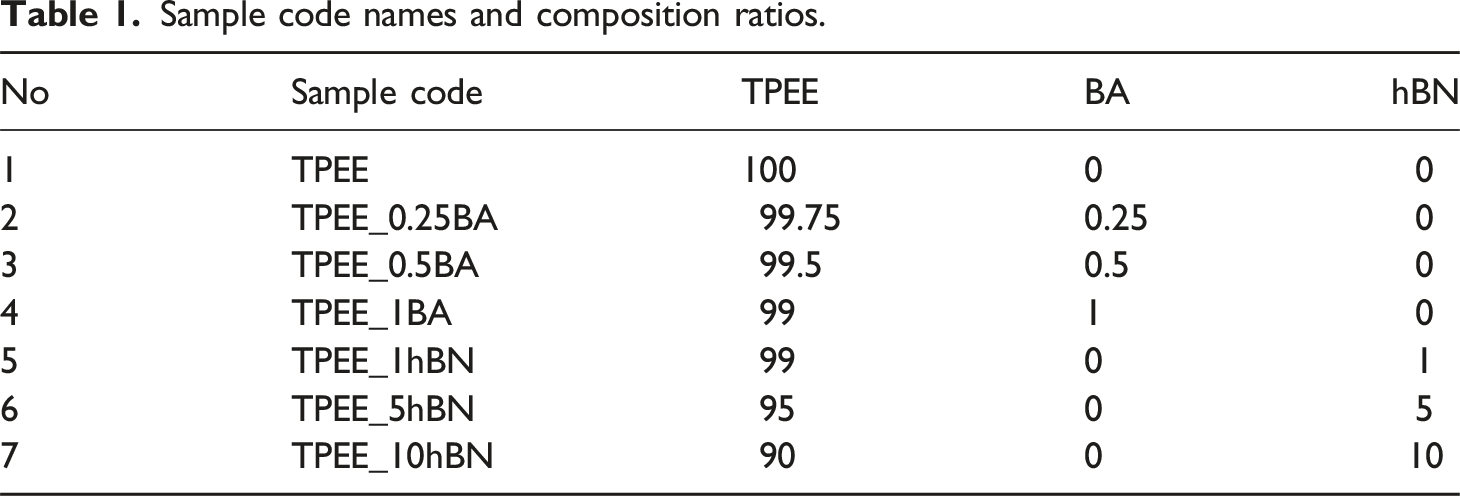
Composite preparation
Sample code names and composition ratios.
Following the compounding process, the test specimens were produced in a laboratory-scale injection moulding machine with fixed moulding conditions of 245°C barrel temperature, 45°C mould temperature, and 10 bar injection pressure.
Characterization of composites
Adhesive wear tests were conducted in accordance with the ASTM G99-23 standard at room temperature using a standard modular pin-on-disk tribometer (Nanovea T50, maximum load capacity 50 N, maximum rotational speed 500 rpm) to determine the coefficient of friction (COF) curves of the test specimens. The specimen was positioned and secured on a rotating disk at a disk speed of 100 rpm, a friction radius of 5 mm, a contact load of 20 N, and a sliding distance of 150m. The COF between the 3 mm radius ceramic ball and each of the differently formulated test specimens was measured and plotted throughout the experiment.
Tensile tests were conducted in accordance with the ISO 527/2-5A standard using a Shimadzu AG-X tensile tester with a capacity of 10 kN at a crosshead speed of 5 mm/min at room temperature. The tensile strength of each sample was recorded, and at least five samples of each formulation were tested in order to calculate the mean value.
Three-point bending tests were conducted on an Instron 4411 tensile tester with a capacity of 5 kN at a rate of 2 mm/min, in accordance with the ISO-178 standard. The flexural stress at yield data were obtained from the test specimens, with a minimum of five specimens of each formulation tested to calculate the average.
The thermal properties of the samples, including the melting temperature (Tm) and enthalpy of melting (ΔHm), were analysed using a TA Instruments-Q200 differential scanning calorimeter (DSC). Thermal scans were conducted between 25°C and 250°C at a rate of 5°C/min in a nitrogen environment. The relative degree of crystallinity (Xc)rel was calculated for each sample using the DSC data and the following equation
29
:
The thermal stability of the samples was investigated utilising a TA Instruments-Q500 thermogravimetric analyser (TGA) at a heating rate of 10°C/min from ambient temperature to 600°C under a nitrogen atmosphere. The weight loss of the samples was recorded as a function of temperature.
The fracture morphology of the specimens was analysed using a JEOL-JCM 6000 scanning electron microscope (SEM). SEM was employed to analyse the fracture surfaces obtained from the tensile test. Prior to the commencement of the morphological analysis, the surfaces to be examined were sputter-coated with gold.
Results and discussion
Adhesive wear test
The effect of BA incorporation into TPEE at concentrations of 0.25, 0.5, and 1 wt% on the COF attribute of the material was examined through an adhesive wear test, and the findings are presented in Figure 1(a)–(b). (a) COF curves of samples containing BA at varying weight ratios; (b) Average COF values of samples containing BA at varying weight ratios.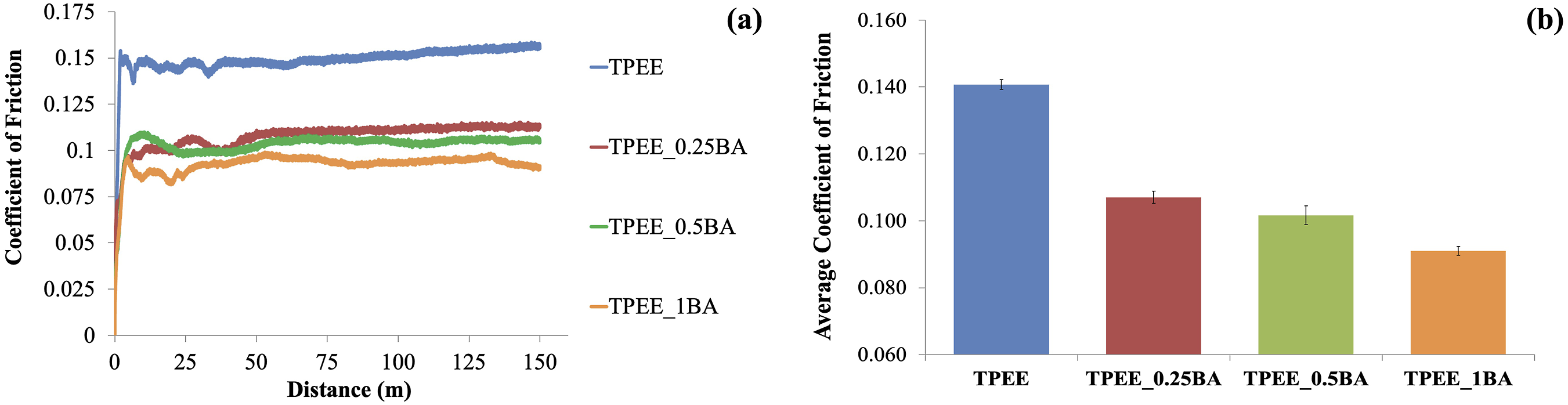
Figure 1(a)–(b) illustrates that the COF values decrease with the addition of BA at varying weight ratios to TPEE. Moreover, the COF exhibits a more pronounced decline with increasing BA content, reaching its lowest value in the sample containing 1 wt% BA. An analysis of Figure 1(a)–(b) reveals that the lubricating properties of BA result in a decrease in the COF of TPEE at all three loading rates of BA.35–37
The effect of hBN incorporation into TPEE at concentrations of 1, 5, and 10 wt% on the COF attribute of the material was examined through an adhesive wear test, and the findings are presented in Figure 2(a)–(b). (a) COF curves of samples containing hBN at varying weight ratios; (b) Average COF values of samples containing hBN at varying weight ratios.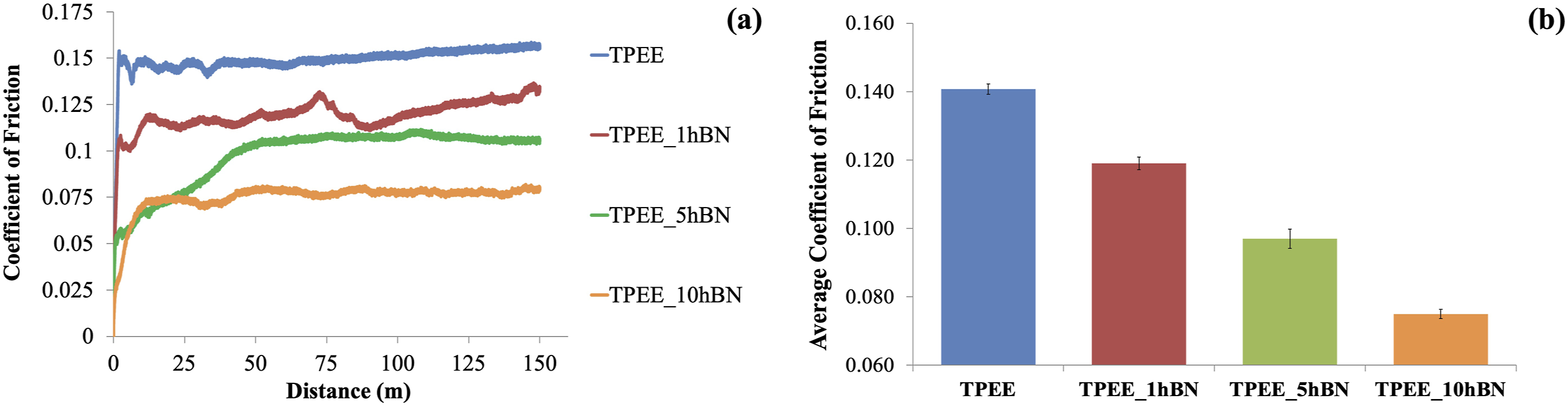
Figure 2(a)–(b) illustrates a notable decline in COF values with the incorporation of hBN at varying weight ratios to TPEE. Moreover, as the quantity of hBN increases, the COF decreases further, reaching a minimum in the sample type containing 10 wt% hBN. An examination of Figure 2(a)–(b) reveals that the addition of hBN, due to its lubricating properties derived from its layered structure, results in a reduction in the COF of the TPEE matrix when reinforced at all three loading rates.24,36,38,39
The findings of the adhesive wear tests conducted on TPEE with the addition of particles indicate that the incorporation of particles enhances the adhesive wear resistance of TPEE. However, when the particle types were compared, the greatest improvement was observed with the addition of 10 wt% hBN. From this perspective, it can be posited that hBN exhibits superior performance in enhancing the adhesive wear resistance compared to BA. This can be attributed to the particle size distribution within the range of 50-120 nm. This is due to the fact that a smaller particle size is associated with a more homogeneous distribution within the polymer, a reduction in agglomeration and an increase in surface area. In contrast, a larger particle size distribution, as observed in BA, is associated with an increase in agglomeration and a reduction in surface area. This is an expected result, given that particles with a more homogeneous dispersion, a reduction in agglomeration and an increase in surface area will exhibit enhanced performance.
As is evident in the extant literature, BA loses water in its structure when exposed to heat energy. At temperatures exceeding 150°C, the material undergoes a transformation into boron oxide (BO), accompanied by the loss of all water molecules. The melting point of crystalline BO has been determined to be 450°C, while amorphous BO begins to soften at 325°C and becomes fluid at 500°C. The resulting boron oxide exhibits superior physical and chemical properties.
40
To examine the behaviour of both BA and hBN used in this study against heat, TGA analysis was applied to the particles and the results of the analysis are given in Figure 3. TGA thermograms of the BA and the hBN particles.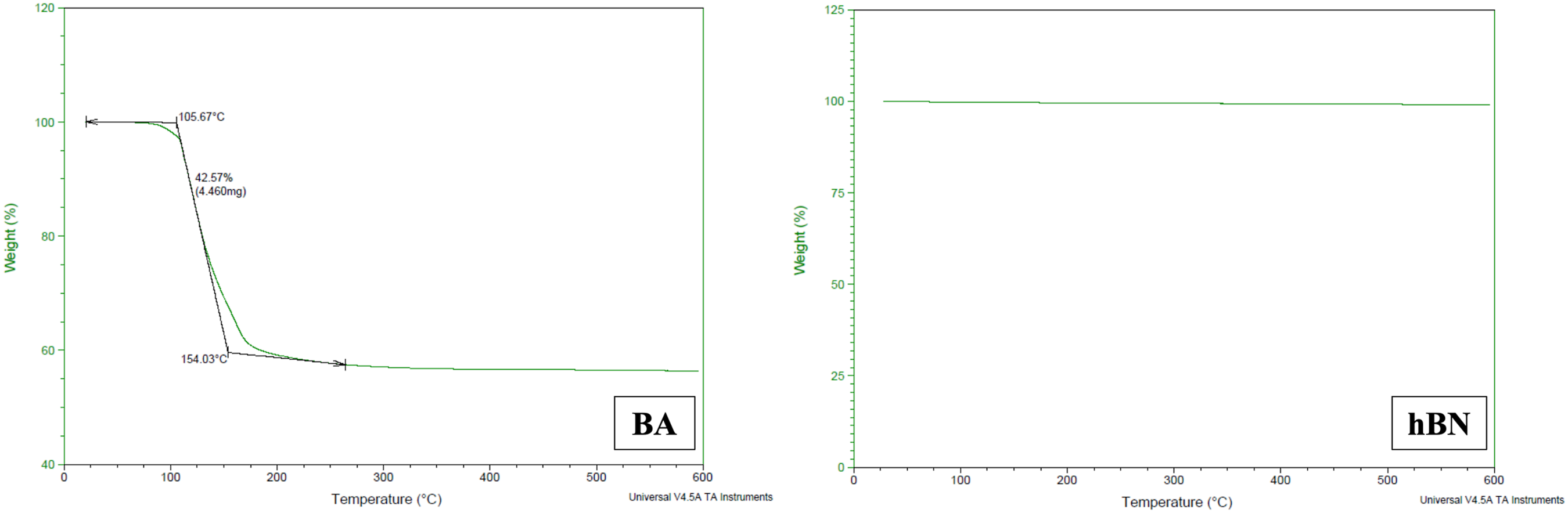
As demonstrated in Figure 3, the dehydration process occurring in the BA structure is clearly evident, while the hBN structure remains unaltered in response to increasing heat energy. Indeed, analysis of the thermogram of BA reveals a precipitous decline in weight commencing at 100°C, with a substantial loss of approximetely 42% in the sample weight at 154°C. This phenomenon can be interpreted as a consequence of the migration of water molecules from the BA structure, leading to its transformation into BO. However, when the temperature is increased further to 600°C, no further decrease in BA weight is observed.
As is evident in the extant literature, certain fillers are known to be thermally sensitive, with a propensity to degrade and/or decompose when exposed to the temperatures experienced during the melting of the polymer during processing. This phenomenon can also result in the agglomeration of these fillers within the polymer. The decrease in particle size during degradation and/or decomposition engenders an increase in the propensity for agglomeration. 22 It can be interpreted that BA is one of these particle types, and that the water in its structure is removed during the TPEE process at a temperature of 245°C. As a result of this, the potential of BA particles to agglomerate increases, and the performance decrease is a consequence of this situation.
Tensile test
The effect of BA incorporation into TPEE at concentrations of 0.25, 0.5, and 1 wt% on the tensile strength value was examined through tensile testing, and the findings are presented in Figure 4(a)–(b). (a) Tensile strength values of samples containing BA at varying weight ratios; (b) Elastic modulus values of samples containing BA at varying weight ratios.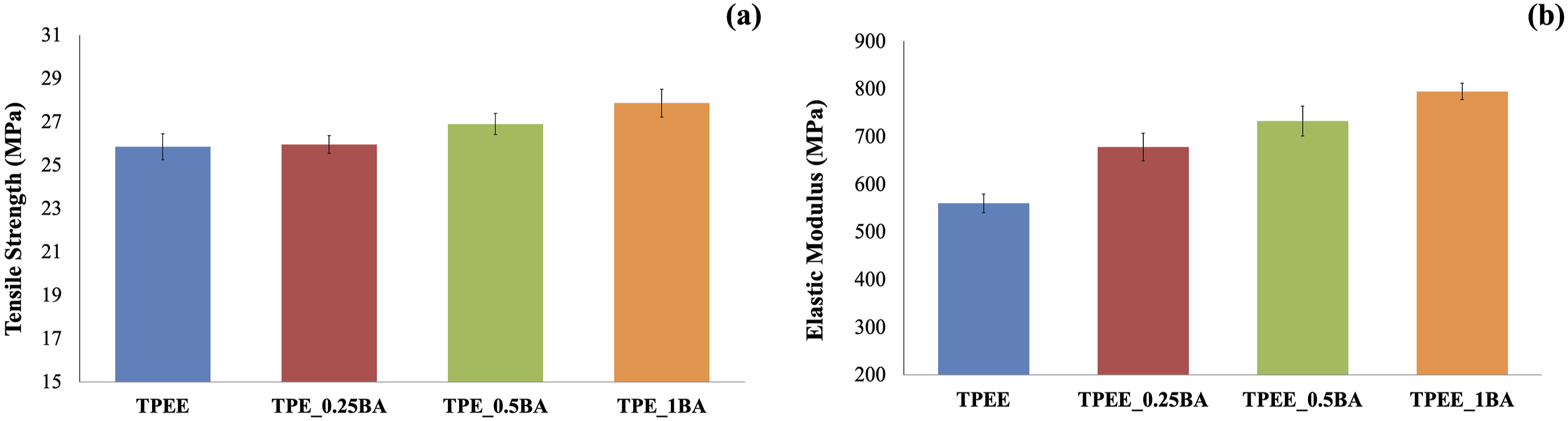
Figure 4(a) demonstrates that the tensile strength value increases with the addition of BA at varying weight ratios to TPEE, and with an increasing weight ratio of BA. The transfer of stress between the matrix and the reinforcing element is the most significant factor influencing the tensile strength of polymer matrix composites. Conversely, the efficiency of stress transfer depends on the uniform dispersion of the reinforcing element within the structure and the interfacial interaction between the matrix and the reinforcing element.41–43 The silanation process enhances the interfacial interaction between the matrix and the reinforcing element, thereby facilitating a more homogeneous distribution of particles within the matrix. It can thus be concluded that the tensile strength of the composite is increased by the addition of BA to TPEE, which is a result of the silanization process applied to BA.44,45
Figure 4(b) illustrates that the modulus value exhibits a consideraly increase with the incorporation of BA into TPEE and with an elevated weight ratio of BA. This enhancement in modulus with rigid particle reinforcement is an anticipated outcome and is elucidated by the rule of mixtures in the existing literature.17,46,47 In accordance with this rule, the modulus of a composite can be calculated using the following equation:
The effect of hBN incorporation into TPEE at concentrations of 1, 5, and 10 wt% on the tensile strength of the material was examined through tensile testing, and the findings are presented in Figure 5(a)–(b). (a) Tensile strength values of samples containing hBN at varying weight ratios; (b) Elastic modulus values of samples containing hBN at varying weight ratios.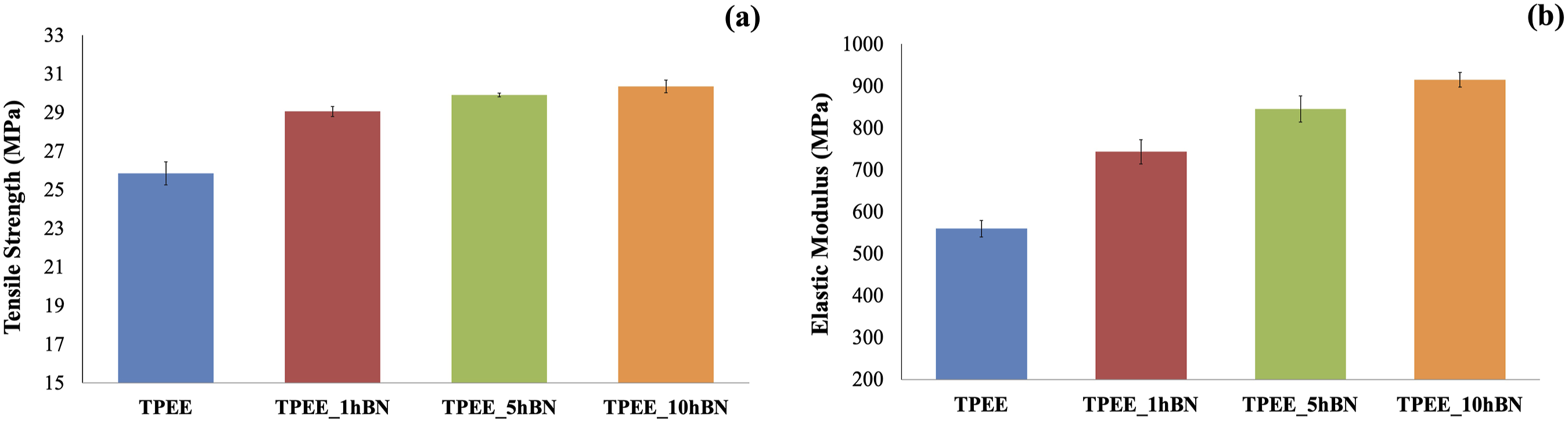
Figure 5(a) demonstrates that the tensile strength value increases with the addition of hBN at varying weight ratios to TPEE. The highest tensile strength value is observed in the composite containing 10 wt% hBN. As previously stated, the enhancement in tensile strength upon the incorporation of hBN can be attributed to two key factors. Firstly, the transfer of stress from the matrix to the reinforcing material facilitates the strengthening of the composite. Secondly, the favourable interfacial interaction between the matrix and the reinforcing material, facilitated by silanization, 48 also contributes to the composite’s tensile strength. 48
Figure 5(b) illustrates that the modulus value increases considerably with the incorporation of hBN to TPEE and with an increasing weight ratio of hBN. This is attributed to the fact that the modulus value of the hBN particle is higher than that of the TPEE polymer, and the modulus of the interfacial phase between hBN and TPEE is enhanced as a consequence of silanization, in accordance with the rule of mixtures previously outlined. 17
A comparison of the particle types revealed that the greatest increase was achieved with the addition of 10 wt% hBN. In this regard, it can be stated that hBN exhibits superior performance in enhancing the tensile strength and modulus values in comparison to BA. This can be attributed to the lower range of particle size distribution observed in hBN.
As illustrated in Figure 6, the SEM micrographs reveal that BA particles are distributed within the TPEE matrix as larger agglomerates, exhibiting a more regionalised localisation. In contrast, hBN particles are positioned in the TPEE matrix as smaller particles, displaying a more homogeneous distribution. SEM micrographs of tensile fractured surfaces of samples.
Three point bending test
The effect of BA and hBN incorporation into TPEE on flexural stress at yield was examined through a three-point bending test, with the findings presented in Figure 7(a)–(b). Flexure stress at yield values of samples containing (a) BA (b) hBN at varying weight ratios.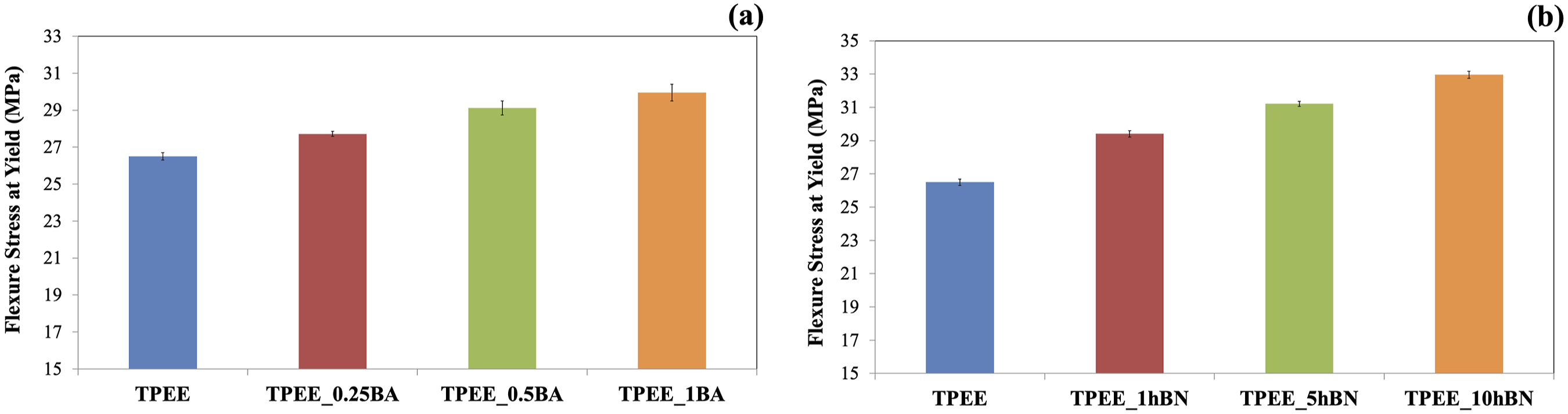
Figure 7(a) and (b) demonstrate that the flexural stress at yield increases with the incorporation of both BA and hBN to TPEE, as well as with increasing weight ratios of these reinforcements. The highest flexural stress at yield is observed in composites containing 1 wt% BA and 10 wt% hBN.
The incorporation of reinforcement represents a crucial element in the flexural strength of polymers, as this property is contingent upon the capacity of the material to withstand bending loads. The incorporation of reinforcement into the polymer and the subsequent increase in reinforcement amount result in enhanced resistance to bending loads. 27 It can thus be seen from Figure 7(a) and (b) that flexural stress at yield of TPEE increase with the addition of particles.
Furthermore, the interfacial interaction between the matrix and the reinforcement, as well as the homogeneous distribution and orientation of the reinforcement within the matrix, are the primary determinants of the flexural properties of the composite. The unmodified and hydrophilic particles may not be evenly distributed within the matrix phase, potentially forming agglomerates. This results in the application of flexural stress being concentrated at specific points within the matrix phase, with the potential for cracks to propagate into the unreinforced regions of the matrix. This is a significant factor contrubuting to the propagation of cracks, which ultimately results in a decline in flexural strength and potential failure of the matrix. The surface modification and the homogeneous distribution of particles serves to prevent crack propagation in the composite structure, thereby leading to a significant improvement in flexural strength properties.27,49–51 An evaluation of Figure 7(a) and (b) in consideration of this point indicates that the addition of both BA and hBN to the TPEE matrix increases the flexural stress at the yield value of TPEE. Nevertheless, the greater increase in flexural stress at yield observed in the composites containing hBN can be attributed to the lower particle size distribution of hBN.
Differential scanning calorimetry analysis
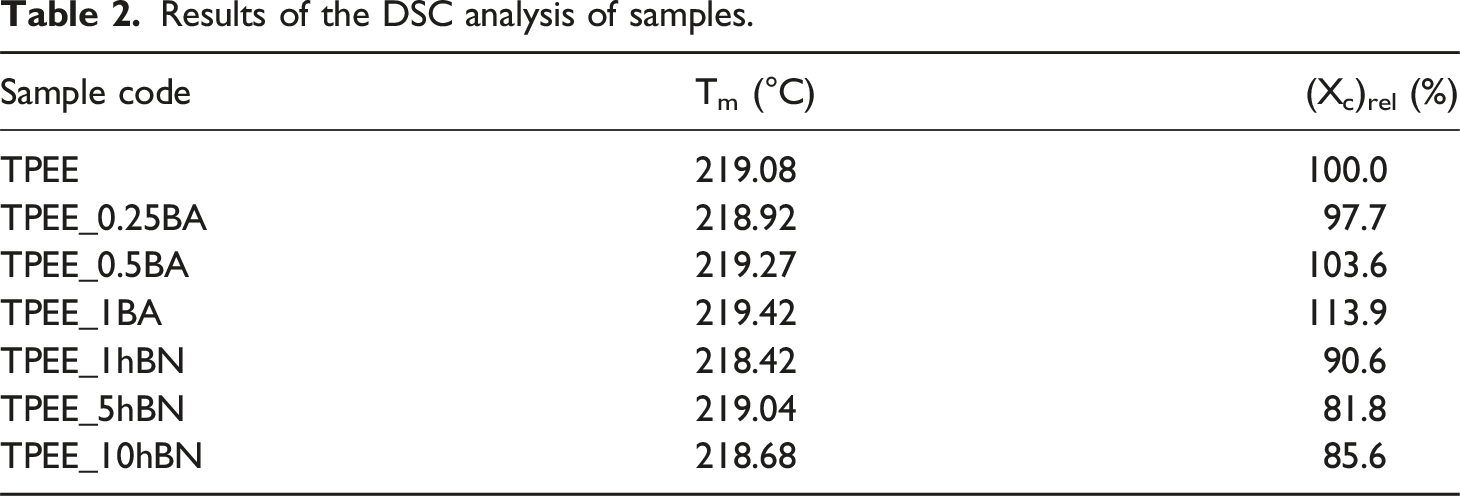
Results of the DSC analysis of samples.
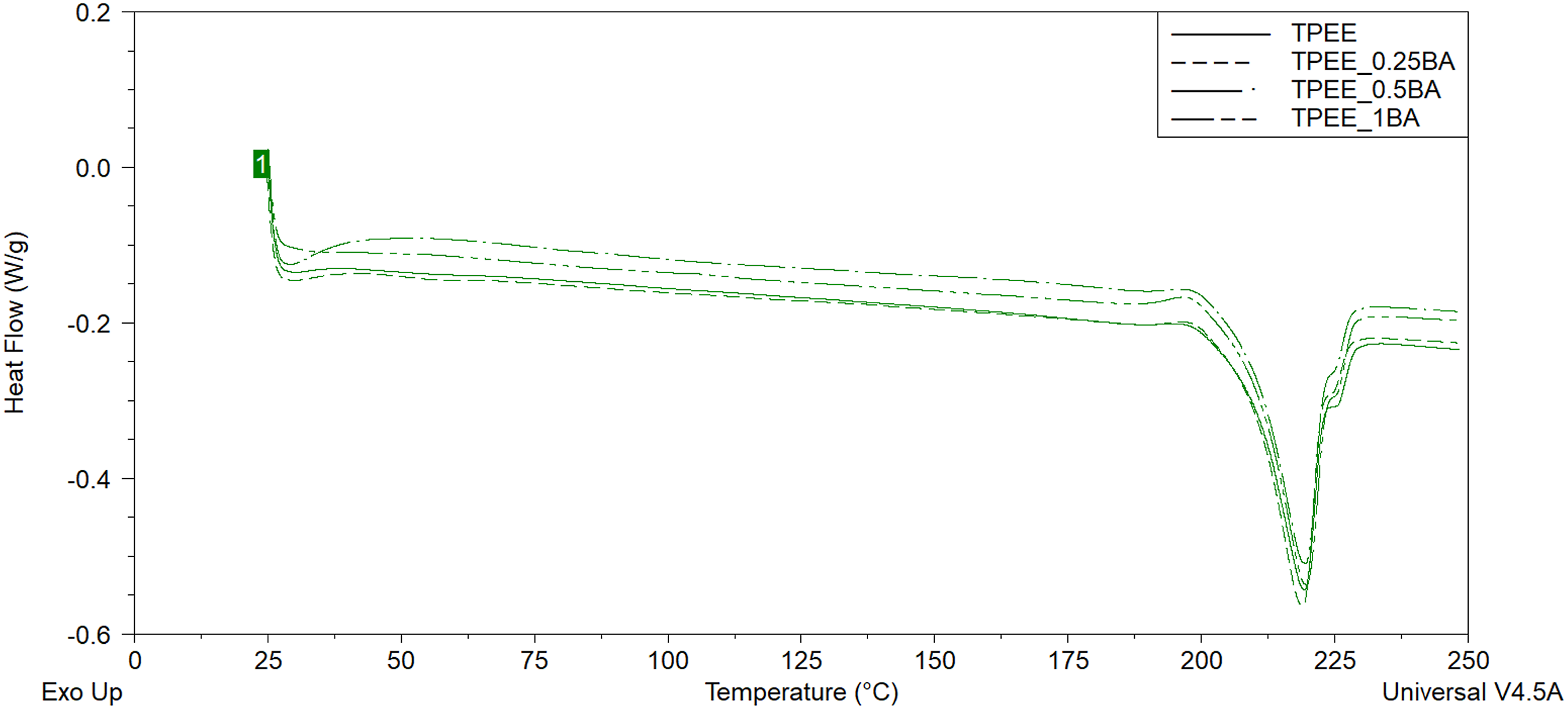
DSC thermograms of unreinforced TPEE and BA-reinforced samples.
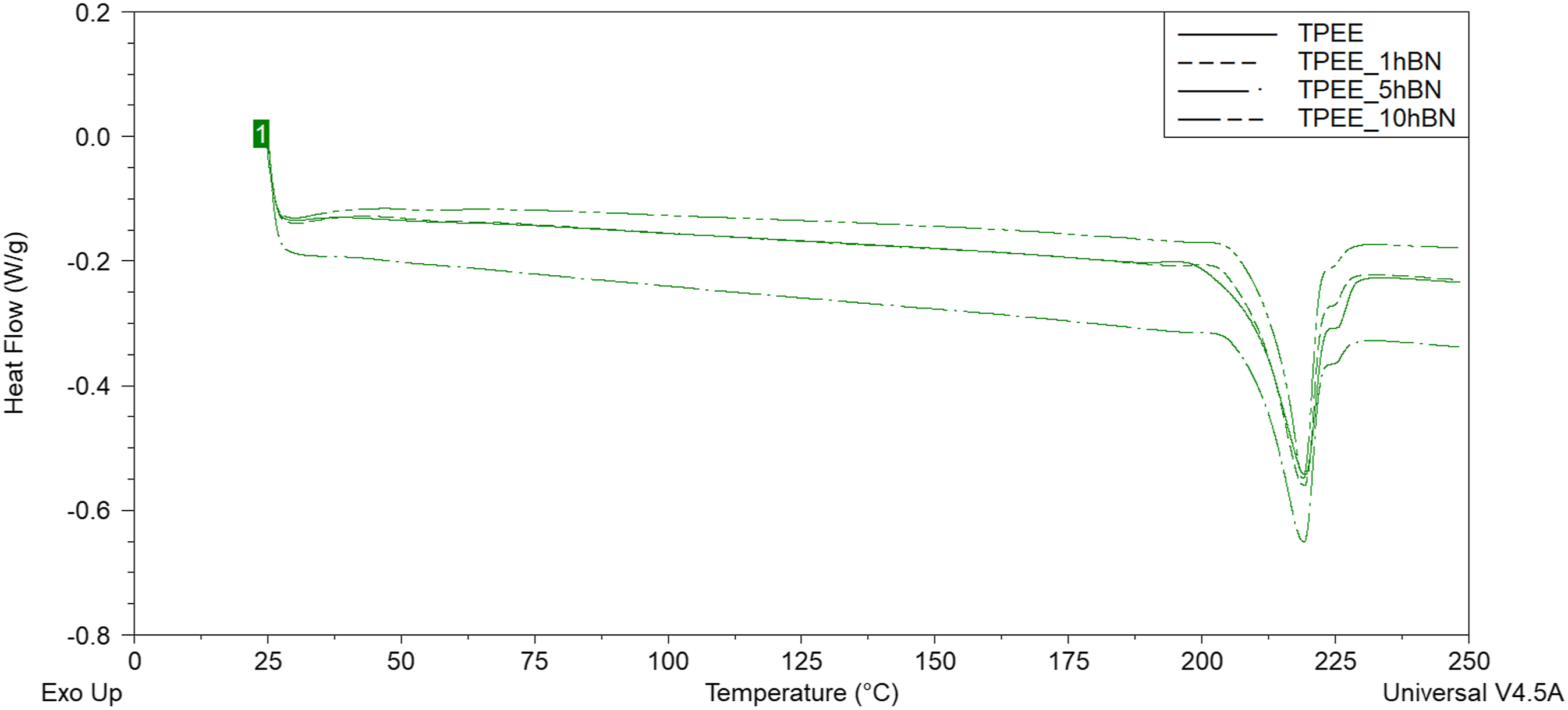
DSC thermograms of unreinforced TPEE and hBN-reinforced samples.
Table 2 illustrates that the addition of BA and hBN at varying weight ratios to TPEE does not result in a notable alteration in the melting temperature values. However, the (Xc)rel (%) value initially exhibits a decrease with the addition of 0.25 wt% BA to TPEE. Conversely, the (Xc)rel (%) value increases as the weight ratio of BA increases to 0.5 and 1 wt%. The highest (Xc)rel (%) value is reached at 1 wt% BA addition. Two distinct potential effects of particle incorporation into a polymer on the (Xc)rel (%) value have been identified in the literature. The first of these effects is that the particles act as nucleating agents in the polymer, forming heterogeneous nucleation sites and thus increasing the percent crystallinity of the structure by facilitating the crystallisation of polymer chains.27,28,52,53 Another potential mechanism proposed in the literature is that particles, when distributed homogeneously within the polymeric matrix, can act as a physical barrier, impeding or delaying the crystallisation and crystal growth of polymer chains. This, in turn, may result in a reduction in the degree of crystallinity.27,28,54–56 The results presented in Table 2 demonstrate that the addition of BA to TPEE at low weight ratio resulted in homogeneous dispersed within the matrix due to the low amount of BA. This prevented the crystallisation of the chains by penetrating between the polymer chains. However, as the quantity of BA present in the material increased, these particles began to function as nucleating agents, thereby enhancing the percent crystallinity of TPEE by forming heterogeneous nucleation sites. Furthermore, Table 2 reveals that the (Xc)rel (%) value declines with the incorporation of hBN into TPEE, irrespective of the weight ratio of hBN. This result shows that when hBN is added to TPEE, the hBN particles are homogeneously dispersed in the matrix and penetrate between the polymer chains, preventing the the chains from crystallizing.
In consideration of the thermal property alterations indicated by DSC analysis, it can be proposed that hBN, exhibiting a lower particle size distribution than BA, is more homogeneously dispersed within the TPEE matrix, intercalating between polymer chains and impeding crystallization by constraining chain mobility across all three weight ratios. Conversely, it can be posited that BA, whose particle size distribution is greater than that of hBN, may have exhibited an enhanced propensity to agglomerate with the increase in weight ratio. These agglomerates may have facilitated the crystallization of the TPEE matrix by acting as heterogeneous nucleation agents.
Thermogravimetric analysis
TGA results of samples.
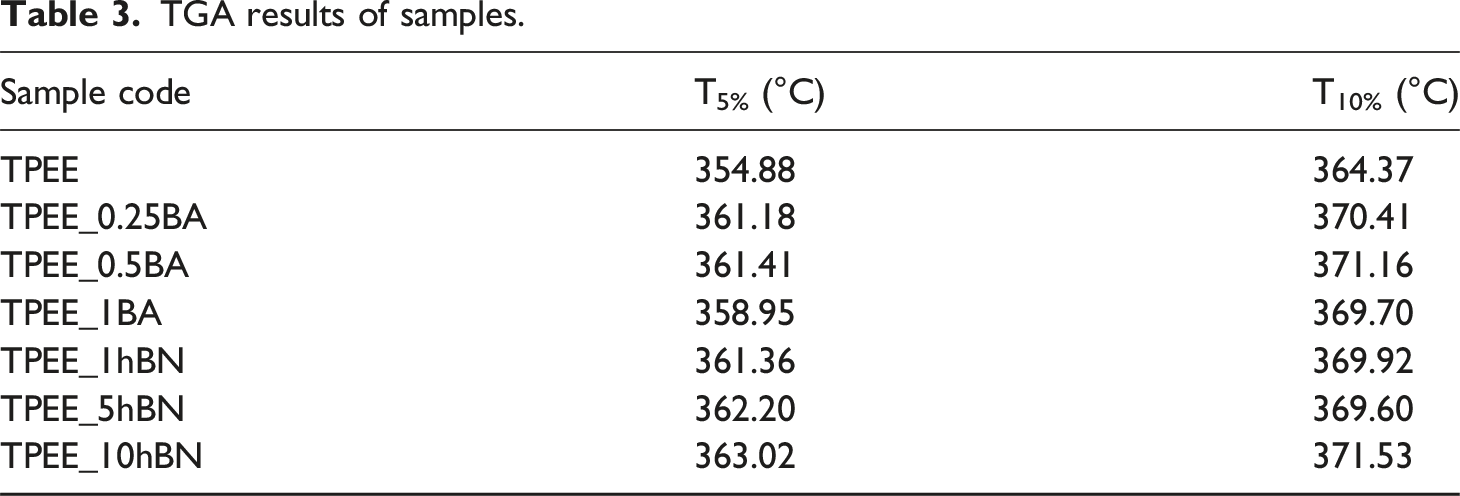
TGA thermograms of unreinforced TPEE and BA-reinforced samples.
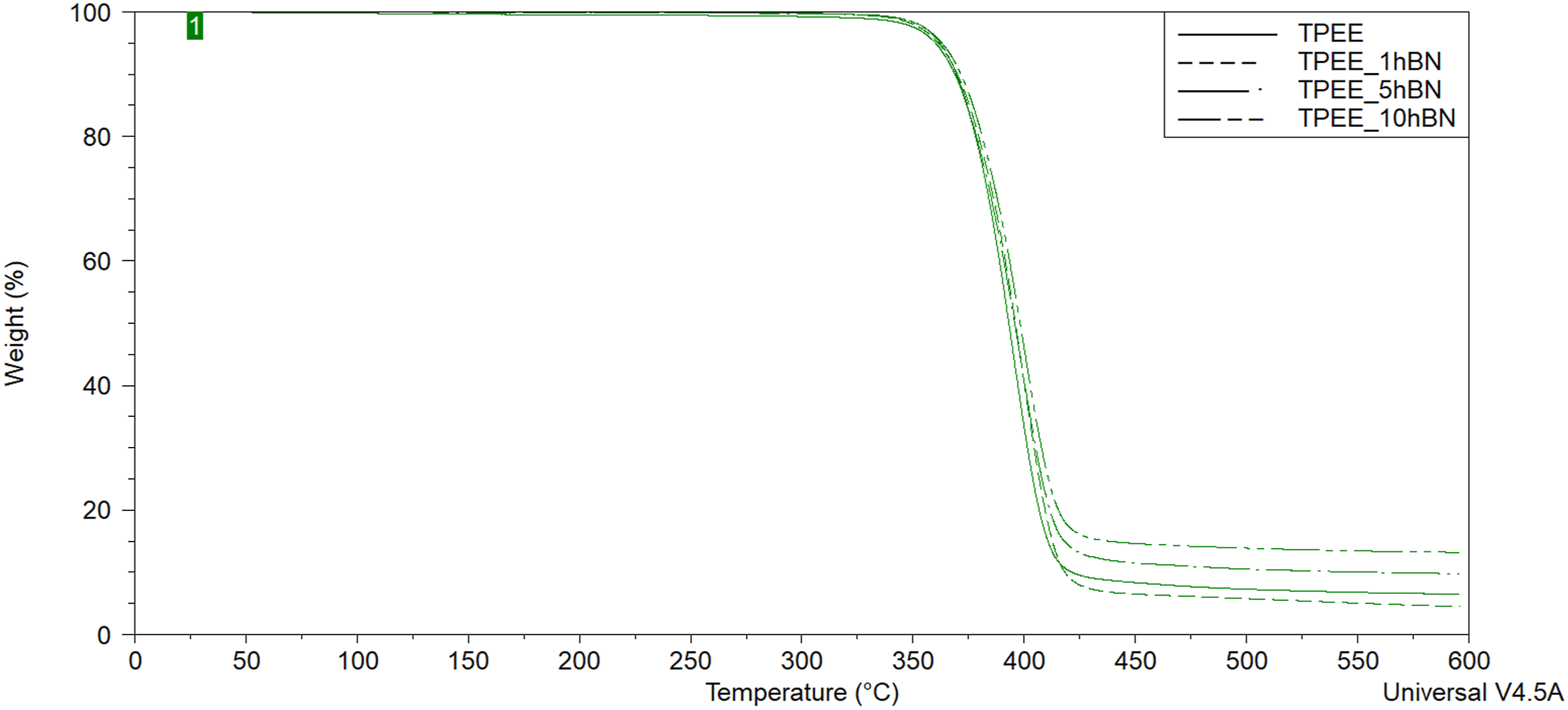
TGA thermograms of unreinforced TPEE and hBN-reinforced samples.
T5% denotes the temperature at which a 5% weight loss is observed in the sample, whereas T10% represents the temperature at which a 10% weight loss occurs in the sample.
Upon examination of Table 3, it becomes evident that the incorporation of BA into TPEE, leads to an increase in both T5% and T10% degradation temperatures, thereby enhancing the thermal stability of TPEE. This improvement reaches approximately 7°C at both T5% and T10% temperatures in composites containing 0.5 wt% BA. Additionally, another result observed from Table 3 is that with the addition of hBN to TPEE and with increasing hBN weight ratio, both degradation temperatures increase, thus improving the thermal stability of TPEE. This improvement reached approximately 8°C at T5%.
The observed increase in heat decomposition temperatures with the addition of BA and hBN is consistent with the expected result. As has been documented in the literature, the thermal conductivity and heat capacity of boron-based inorganic particles, such as BA and hBN, are superior to those of unreinforced TPEE. Consequently, in the context of thermal energy transfer to materials comprising these particles, the reinforcing materials in question are capable of absorbing a greater quantity of heat due to their elevated heat capacity in comparison to the matrix material. Furthermore, they are able to dissipate the absorbed heat with greater ease within the structure due to their superior thermal conductivity in comparison to the matrix material. Consequently, they enhance the thermal stability of the material in which they are embedded. Furthermore, due to their layered structure, they can act as a physical barrier during the degradation process of the material in which they are embedded, trapping degradation products between the layers and delaying the degradation process of the material. 27
It can thus be concluded that the BA and hBN particles incorporated into the TPEE matrix in this study serve to protect the TPEE matrix to a certain extent against thermal degradation. This is due to the elevated heat capacity and thermal conductivity of the particles, as well as their layered structure. Consequently, they facilitate the maintenance of the thermal stability of TPEE.
Conclusions
The objective of this study was to investigate the effect of the addition of varying weight ratios of boron-based particles (BA and hBN) on the tribological, mechanical, and thermal properties of TPEE the composites.
The results of the adhesive wear test demonstrated that the COF values of the material decreased with the addition of both BA and hBN. However, a comparison of the performance of the two boron-based particles in improving wear resistance by reducing COF revealed that hBN exhibited superior performance to BA.
The data obtained from the tensile test demonstrated that the incorporation of both BA and hBN into TPEE, as well as an increase in the weight ratio of these particles, resulted in an notable enhancement in the tensile strength and elastic modulus values. Furthermore, the three-point bending test demonstrated that the incorporation of BA and hBN into TPEE, along with an increase in their weight ratio, led to an enhancement in flexural stress at yield. However, when the improvement in the particle types’ performances on the mechanical properties was compared, it was observed that hBN exhibited a higher performance.
The results of DSC analysis indicated that the addition of BA to TPEE resulted in an increase in the crystallization of the TPEE matrix. Conversely, the addition of hBN to TPEE led to a reduction in the crystallization of the TPEE matrix. The enhancing effect of BA on crystallization can be attributed to its role as a nucleation agent, a consequence of its structural properties. The inhibiting effect of hBN on crystallization can be attributed to the homogeneity of its dispersion in the TPEE matrix, which is a result of its low particle size distribution and homogeneous dispersion between the polymer chains.
The TGA results, which were used to determine the change in the thermal stability of TPEE with the addition of BA and hBN to TPEE, demonstrated that the incorporation of both BA and hBN elevated the decomposition temperatures of the material, thereby enhancing its thermal stability.
In conclusion, the present study has demonstrated that the tribological, mechanical, and thermal properties of TPEE can be enhanced through the incorporation of boron-based particles. Furthermore, it was concluded that the size distributions and agglomeration tendencies of the particle types utilised in the study were effective on the composite properties. Moreover, from the test results obtained, it was concluded that the tendency of BA to agglomerate increased due to dehydration during composite production and that this situation affected its performance. While the specific particle type to be utilized may be contingent upon the intended application of the end product, it has been evidenced that both particles can expand the scope of TPEE utilization and enhance its performance in the designated area by improving the intrinsic properties of TPEE.
In view of the findings of this study, it is therefore recommended that the use of hBN-reinforced TPEE matrix composites be given due consideration, particularly within the automotive and aerospace sectors, where the requirement for elevated mechanical and tribological performance, coupled with optimal thermal conductivity, is paramount. Conversely, the utilisation of BA-reinforced TPEE matrix composites is advised within the domains of biomedical engineering, electrical and electronics, and household goods, where the demands on mechanical performance may be less stringent, yet flammability and tribological performance assume significant importance.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by Türkiye Bilimsel ve Teknolojik Araştırma Kurumu (The Scientific and Technological Research Council of Turkey) under the 1505-University-Industry Collaboration Support Program (Project No: 5160067).